Abstract
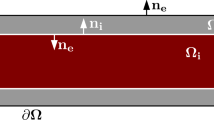
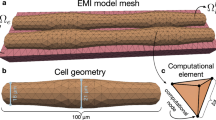
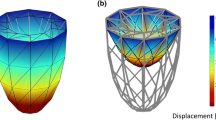
We aim to extend existing models of single-cell mechanics to the EMI framework, to define spatially resolved mechanical models of cardiac myocytes embedded in a passive extracellular space. The models introduced here will be pure mechanics models employing fairly simple constitutive laws for active and passive mechanics. Future extensions of the models may include a coupling to the electrophysiology and electro-diffusion models described in the other chapters, to study the impact of spatially heterogeneous ion concentrations on the cell and tissue mechanics.
Chapter PDF
Similar content being viewed by others
Reference
Ambrosi D, Pezzuto S (2012) Active stress vs. active strain in mechanobiology: constitutive issues. Journal of Elasticity 107(2):199–212
2. Campbell KS (2009) Interactions between connected half-sarcomeres produce emergent mechanical behavior in a mathematical model of muscle. PLoS computational biology 5(11):e1000560
3. Campbell SG, Lionetti FV, Campbell KS, McCulloch AD (2010) Coupling of adjacent tropomyosins enhances cross-bridge-mediated cooperative activation in a markov model of the cardiac thin filament. Biophysical journal 98(10):2254–2264
4. Chase PB, Macpherson JM, Daniel TL (2004) A spatially explicit nanomechanical model of the half-sarcomere: myofilament compliance affects ca 2+-activation. Annals of biomedical engineering 32(11):1559–1568
5. Guccione J, McCulloch A,Waldman L (1991) Passive material properties of intact ventricular myocardium determined from a cylindrical model. Journal of Biomechanical Engineering 113(1):42–55, https://doi.org/10.1115/1.2894084
Holzapfel GA (2000) Nonlinear solid mechanics: a continuum approach for engineering.Wiley
7. Jæger KH, Tveito A (2020) Derivation of a cell-based mathematical model of excitable cells. In: Tveito A, Mardal KA, Rognes ME (eds) Modeling excitable tissue - The EMI framework, Simula Springer Notes in Computing, SpringerNature
8. Laadhari A, Ruiz-Baier R, Quarteroni A (2013) Fully eulerian finite element approximation of a fluid-structure interaction problem in cardiac cells. Int J Numer Meth Engng 96:712–738
9. LeGrice IJ, Smaill B, Chai L, Edgar S, Gavin J, Hunter PJ (1995) Laminar structure of the heart: ventricularmyocyte arrangement and connective tissue architecture in the dog. American Journal of Physiology-Heart and Circulatory Physiology 269(2):H571–H582
Mijailovich SM, Nedic D, Svicevic M, Stojanovic B,Walklate J, Ujfalusi Z, GeevesMA(2017) Modeling the actin. myosin atpase cross-bridge cycle for skeletal and cardiac muscle myosin isoforms. Biophysical journal 112(5):984–996
11. Nakagome K, Sato K, Shintani SA, Ishiwata S (2016) Model simulation of the spoc wave in a bundle of striated myofibrils. Biophysics and physicobiology 13:217–226
12. Nash MP, Panfilov AV (2004) Electromechanical model of excitable tissue to study re-entrant cardiac arrhythmias. Progress in Biophysics and Molecular Biology 85(2-3):501–522
13. Rice JJ,Wang F, Bers DM, de Tombe PP (2008) Approximate Model of Cooperative Activation and Crossbridge Cycling in Cardiac Muscle Using Ordinary Differential Equations. Biophysical Journal 95(5):2368–2390
14. Ruiz-Baier R, Gizzi A, Rossi S, Cherubinie C, Laadhari A, Filippi S, Quarterone A (2014) Mathematical modelling of active contraction in isolated cardiomyocytes. Mathematical medicine and biology 31:259–283
Sundnes J, Lines GT, Cai X, Nielsen BF, Mardal KA, Tveito A (2007) Computing the electrical activity in the heart, vol 1. Springer Science & Business Media
Telle Å (2020) Software for EMI – Modeling cardiac mechanics on a sub-cellular scale. https://doi.org/10.5281/zenodo.3702168, URL https://doi.org/10.5281/zenodo.3702168
17. Tveito A, Jæger KH, Lines GT, Paszkowski Ł, Sundnes J, Edwards AG, M¯aki-Marttunen T, Halnes G, Einevoll GT (2017) An evaluation of the accuracy of classical models for computing the membrane potential and extracellular potential for neurons. Frontiers in computational neuroscience 11:27
Tveito A, Jager KH, Kuchta M, Mardal KA, Rognes ME (2017) A cell-based framework for numerical modeling of electrical conduction in cardiac tissue. Frontiers in Physics 5:48, https://doi.org/10.3389/fphy.2017.00048, URL https://www.frontiersin.org/article/10.3389/fphy.2017.00048
19. Usyk TP, LeGrice IJ, McCulloch AD (2002) Computational model of three-dimensional cardiac electromechanics. Computing and Visualization in Science 4(4):249–257
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2021 The Author(s)
About this chapter
Cite this chapter
Telle, Å., Wall, S.T., Sundnes, J. (2021). Modeling Cardiac Mechanics on a Sub-Cellular Scale. In: Tveito, A., Mardal, KA., Rognes, M.E. (eds) Modeling Excitable Tissue. Simula SpringerBriefs on Computing(), vol 7. Springer, Cham. https://doi.org/10.1007/978-3-030-61157-6_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-61157-6_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-61156-9
Online ISBN: 978-3-030-61157-6
eBook Packages: Mathematics and StatisticsMathematics and Statistics (R0)