Abstract
Myopia is often described as resulting from a complex set of interactions between genetic and environmental risk factors. Rare forms of strongly familial myopia account for myopia in around 1% of any population. “School myopia”, the most common form in most modern societies, is also influenced by genetic factors, but changes in environmental risk factors appear to be responsible for the major increases in the prevalence of myopia in some parts of East and Southeast Asia. Two major environmental risk factors have been identified—intensive schooling and limited time outdoors, with educational pressures high and time outdoors particularly limited in the parts of East and Southeast Asia afflicted by an epidemic of school myopia. Several other “independent” risk factors for myopia have been reported. The effects of many of these may be mediated by modulation of the two major risk factors, and in future studies on risk factors, mediation analysis needs to be used systematically. In the case of school myopia, we argue that there are two major environmental risk factors, with a limited role for genetic variation and minimal interactions. We propose a method for comparing the impact of identified genetic risk factors with the cumulative effects of environmental exposures. This suggests that genetic risk factors associated with more myopic refractions generally lead to myopia only when combined with exposures to environmental risk factors. The predominant role played by increased educational pressures and limited time outdoors suggests strategies for controlling the current epidemic of myopia by directly reducing educational pressures, perhaps by limiting homework and rote learning in the early school years, combined with increases in time spent outdoors in schools. Identification of distal environmental factors, such as early onset of competition for selective academic streams and schools, and the use of after-school tutorial classes also suggest strategies for reducing the current epidemic through changes at the school system level. When combined with clinical techniques for controlling the progression of myopia, these measures have the potential to markedly reduce the current high prevalence rates of both myopia and sight-threatening pathological high myopia.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
-
Myopia commencing school myopia is found mainly is found mainly in developed societies, with only around 1% of any population affected by predominantly genetic forms of myopia.
-
Exposure to environmental risk factors plays a significant role in the development of “school myopia” myopia, both within and between populations.
-
Within populations, changes in exposure to environmental risk factors are primarily responsible for the emergence of an increasing prevalence of myopia.
-
The major environmental risk factors identified are educational pressures, perhaps best characterized in terms of near work, and limited time outdoors during daylight hours.
-
These risk factors suggest that strategies based on decreases in the amount of near work and increases in the amount of time spent outdoors during school hours are likely to be useful in the controlling the epidemic of myopia.
It is now generally accepted that there is an environmental epidemic of myopia in several parts of East and Southeast Asia, namely Singapore, Japan, South Korea, China, including Hong Kong and Macau, as well as in Taiwan [1,2,3,4,5,6,7,8]. In these parts of the world, the prevalence of myopia in young adults who have completed 12 years of schooling, as most do, is now 70–90%, up from 20 to 30% two or three generations ago. In addition, the prevalence of high and potentially pathological myopia (more than 5 D or 6 D of myopia) is of the order of 10–20% [9,10,11,12]. The simple argument that the speed at which the prevalence of myopia has increased in these locations is not compatible with a predominant role for genetic determination of myopia [1, 2] has generally been accepted, even though genetic variation associated with myopia has been conclusively demonstrated [13]. This emphasizes the need to define the environmental exposures responsible for the rapid increases in prevalence, in order to design preventive interventions.
The term “an epidemic of myopia” covers two distinct, but related, issues. The first is an increase in the prevalence of myopia overall, with a characteristic increase in onset of myopia in children in the early school years, leading to a need to provide optical correction for a significant proportion of the population from an early age. The high prevalence of myopia observed in young adults will gradually become the norm for the entire adult population, as younger more myopic generations replace those who are older and less myopic.
The second issue is an increase in the prevalence of high myopia, which is associated with a higher prevalence of pathological outcomes. The cutoff values used to define high myopia are somewhat arbitrary (generally −5 D or −6 D), since the prevalence of pathological complications rises steeply with severity of myopia. The best cutoff to define high and potentially pathological myopia is one which distinguishes, as effectively as possible, between lower and higher risks of pathology, but no cutoff will do this perfectly [14]. The increase in more severe levels of myopia seems to arise from the fact that myopia has appeared at increasingly early ages as the epidemic has developed [11]. With subsequent progression, the myopic refractive error will increase toward the cutoff for high myopia, a tendency exacerbated by rapid progression in younger children [15]. In these circumstances, slowing the onset of myopia should lead to a disproportionate reduction in high myopia. An important question in this area is whether the risk factors for increased prevalence of myopia are similar to the risk factors for progression, or in practical terms whether increased time outdoors would help to limit progression directly, as well as by delaying the onset of myopia. It is certainly possible to slow the development of high myopia by clinically slowing progression rates, using optical or pharmaceutical interventions, and many of these interventions are now available [16].
This chapter reviews the scientific evidence on risk factors in all these areas, and relates this evidence to strategies for slowing the onset and progression of myopia. We will also mention a number of commonly believed risk factors for myopia that may not have a substantial scientific basis. Myopia is such a common condition in societies with well-developed education systems that there are many popular beliefs about the causes of myopia. These often remain influential, sometimes even when the scientific evidence is against them.
6.1 Key Issues When Studying Risk Factors for Myopia
In assessing the literature on risk factors for myopia, it is essential to be clear about a number of fundamental issues.
6.1.1 Myopia Is Etiologically Heterogeneous
It is now generally accepted that environmental factors have a major role to play in the development of myopia. Nevertheless, there are 100–200 clearly genetic forms of myopia documented in the Online Mendelian Inheritance in Man (OMIM) database (https://www.omim.org/, accessed Jan 30, 2019) that clearly run in families [17]. These are individually quite rare, but account collectively for myopia in around 1% of any population. In a recent detailed analysis, 119 genes were associated specifically with myopia, generally in association with other clinical features [17]. Most of these forms are therefore called syndromic, although in some, myopia seems to be the major clinical feature. The latter cases are often called nonsyndromic [18]. Environmental factors appear to play a rather limited role in these diseases, but their existence does not mean that all forms of myopia are similarly genetic—This issue is further discussed in Chapter 5 etc.
In addition to these genetic forms, in modern societies, myopia generally appears in association with schooling, as “school myopia.” It is this form that has increased in the epidemics of myopia. Genetic variation has some role to play in this form of myopia, with the most recent genetic data identifying variants in over 160 genes associated with spherical equivalent refraction (SER). Many of these variants are associated with the genes involved in the more clearly genetic forms, suggesting that mutations in coding regions of the same genes can lead to highly genetic forms of myopia, or to more modest effects if the mutations affect regulatory regions [17].
Identified single nucleotide polymorphisms (SNPs) in samples of European ancestry account for less than 10% of the variance in refraction [13]. Measured as SNP-heritability, they may account for 20–35% of the phenotypic variance [19]. These values fall well short of the twin study heritability of 80–90% [20,21,22]—an example of the missing heritability seen with many complex traits [23]. The estimated SNP-heritability in East Asian samples is much lower at around 5% [13], as would be expected if the amount of phenotypic variation due to genetic variation had remained constant, but that induced by environmental factors had increased substantially in populations severely affected by the environmentally induced epidemic of myopia. We have recently reviewed this area, and readers can consult these reviews for more details [2, 24].
Given this picture, genetic variation may have little, if any, role to play in the rapid changes that have produced the current epidemic of myopia, but this does not mean that it has no role to play in the determination of variations in refractive error within a population. In fact, some myopia geneticists have argued that environmental factors are important between populations, but within populations, genetic factors are the primary determinants [5, 6, 8]. However, this argument does not make sense, because between-population differences have largely emerged due to selective changes within populations of the kind seen in East and Southeast Asia (Fig. 6.1). The key to understanding lies in the recognition that the balance between genetic and environmental factors is population-specific. In the case of myopia, the evidence suggests that there is little difference between ethnic groups in the levels of SNPs associated with myopia [13], although studies on a wider range of ethnic groups are required. However, environmental exposures can differ substantially between ethnic groups, and can change rapidly. Environmental factors are therefore far more likely to be involved in rapid intergenerational change, or in the rapid emergence of differences between societies—both features of the current epidemic.
Prevalence of myopia in older and more recent cohorts in older and more recent younger cohorts. Data taken from cohorts closest in age to 20 from Breslin et al. [66], French et al. [96], Czepita et al. [71] Wang et al. [58], Attebo et al. [59], Wu et al. [12], Koh et al. [10], Jung et al. [9], Wong et al. [60]. Xu et al. [198], and He et al. [140]
This has an important implication; key insights into the epidemics are most likely to come from comparisons of societies with different prevalence of myopia, rather than from detailed studies within populations. Many epidemiologists are wary of this ecological approach, because of what is known as the “ecological fallacy”—the error that can be made if conclusions are based only upon ecological comparisons. But in the case of myopia, this is not an issue, because education and time outdoors clearly play a role both within and between populations.
6.1.2 Myopia Is a Developmental Condition
Developmental considerations also support the importance of etiological heterogeneity. Some forms of myopia appear in children before they start school. These are often severe forms, with a strong genetic etiology, and are called early onset myopia. In contrast, “school myopia” results from excessive elongation of the eye during the school years. Other ocular components, such as corneal and lens power, are within the normal age-specific range. Myopic refractions are also seen in two other conditions—keratoconus and cataract-related myopic shifts in older people. These conditions are etiologically quite distinct from “school myopia”—the first involving abnormal increases in corneal power, generally during the teen-age years, and the second involving abnormal increases in lens power in older adults. These forms are likely to have a different relationship to pathological myopia than “school myopia,” because they do not involve excessive enlargement of the posterior pole of the eye.
Most children are born hyperopic, although premature children have a tendency toward more myopic refractions [25]. At birth, the distribution of refractions is close to Gaussian, and subsequent development involves clustering of refractions to produce a tighter distribution, combined with reduction in hyperopic refractions [26,27,28]. These changes are often called “emmetropisation” because, as a general rule, refractions move from hyperopic toward emmetropic. However, emmetropia is not the end point for human refractive development under normal conditions. Rather, the preferred end point appears to be a moderate level of hyperopia (1.0–1.3 D), provided that cycloplegic refraction is measured [29]. This mildly hyperopic state can be maintained even in adults in some environments [30]. However, in populations where the prevalence of myopia becomes high, the hyperopic clustering of refractions disappears.
This part of refractive development is consistent with animal studies on experimental myopia that suggest that hyperopic defocus induces faster axial elongation, thus reducing hyperopic refractive error, while myopic defocus can slow axial elongation, and thus might be expected to affect the progression of myopia [31]. Simple expectations from the animal models, namely that myopia would be a self-limiting condition, or that it would be dangerous to correct myopia since it would reduce the error signal, are not consistent with current evidence [32,33,34]. However, there is some evidence that early myopic errors can be cleared [35], and contact lenses designed to impose myopic appear to slow the progression of myopia [36]. Thus, these mechanisms can come into play in particular circumstances, but what these circumstances are is currently unclear.
6.1.3 Cycloplegia and Definitions of Myopia: Important Methodological Issues
It is now generally accepted that cycloplegia is the gold standard for studies with children, with the use of up to three drops of 1% cyclopentolate as a common practice, with monitoring of pupil diameter and the light reflex to assess adequacy of cycloplegia [37]. This issue is particularly important in young children, where the prevalence of myopia is low, and a small amount of pseudomyopia can significantly increase prevalence estimates. It is also important when comparing ethnic groups, because it is harder to achieve cycloplegia in children with darkly pigmented irises. Without cycloplegia, hyperopia is substantially underestimated, emmetropia may be overestimated, with a smaller but significant overestimation of the prevalence of myopia. Suboptimal cycloplegia is likely to lead to errors of lesser magnitude, but in the same direction.
In contrast to studies on children, cycloplegia has generally not been used in studies on adults. Adults over the age of 50 show minimal differences between cycloplegic and noncycloplegic refractions [37, 38], consistent with the evidence that by this age, accommodative capacity has essentially disappeared, resulting in the need for some form of correction for reading. However, in younger adults, cycloplegia is required for accurate refraction, yet it generally has not been used in the larger studies on adults, even if they included young adults.
This emphasis on cycloplegia poses problems, because gaining informed consent for cycloplegia appears to be becoming more difficult. It is worth considering alternatives, and biometric variables like axial length and the ratio of the axial length to corneal radius of curvature (AL/CR ratio) could be used [39,40,41]. Both correlate highly with myopia [42, 43]. Changes in lens power also play a major role in refractive development, but they do not correlate highly with refraction and cannot be measured directly [44]. The advantage of the first two parameters is that they can be measured noninvasively. Their disadvantage is that measurement requires relatively expensive modern equipment, and that there is only a very limited set of data for comparison. But where repeated measures of cycloplegic refraction are envisaged, the highly correlated biometric variables may provide a useful alternative.
It is also important to note that a range of definitions is used in the literature for both myopia and high myopia. Cutoff values for myopia range from −0.25 D to −1.0 D, with some variation in the use of less than, or less than or equal to. Results are sometimes reported in relation to the better eye, worse eye, either eye, or both eyes, depending, to some extent, on the aims of the study. The most common standard for epidemiology is at least −0.5 D in the right eye. Similarly, the prevalence of high myopia is sometimes reported in relation to a cutoff of −5 D or −6 D. Sometimes more stringent cutoffs (−8 D, −10 D or −12 D) are used, which is likely to concentrate more genetic forms of myopia in the sample. Cumberland et al. [45] have therefore called for the development of an approach that “serves research, policy and practice,” but made no specific recommendations. However, standard definitions will be of little use without standardization of cycloplegia. While awaiting agreement in both these areas, if it ever comes, we suggest a pragmatic approach is to publish a distribution of refraction in all studies in sufficient detail that others can apply the cutoff they think is most relevant. Cumberland et al. [45] also emphasized the sensitivity of risk factor analysis to different definitions of myopia, and where appropriate, sensitivity analysis should be used to establish that conclusions are robust [46].
These issues are very relevant to several major studies on risk factors for myopia. Studies on childhood and adolescence, the period during which myopia develops, and on young adults, when myopia stabilizes, are particularly important, since exposures over this period can be most directly related to the development of myopia. In addition, studies on young adults give estimates of prevalence that can be most directly compared to assess whether and to what extent there is an epidemic of myopia. Table 6.1 lists some of the most important studies that used cycloplegia. In general, there is a dearth of systematic evidence on the prevalence of myopia in most populations, particularly in the critical period up to the emergence of young adults. Large studies have rarely been designed to look at refractive errors, and studies reporting on refractive errors from the 1958 British Birth Cohort Study [47], the Avon Longitudinal Study of Parents and Children (ALSPAC) [48,49,50], the British Twins Early Development Study (TEDS) [51], the US-based National Health and Nutrition Evaluation Survey (NHANES) [52], the Korean NHANES [53], the Gutenberg Health Survey [54], and the UK Biobank Study [55] measured noncycloplegic refractions. Data from the European Eye Epidemiology (E3) consortium suffers from the same bias [56, 57]. Major studies of eye diseases, including refractive errors, such as the Beaver Dam Eye Study [58], the Blue Mountains Eye Study [59], and the Tanjong Pagar Eye Study [60] also measured noncycloplegic refractions. However, the Tehran Eye Study measured both cycloplegic and noncycloplegic refractions over a wide age range [61].
In general, studies designed to look specifically at refractive errors in school age children, such as the pioneering studies from Taiwan [11], the RESC (Refractive Error Study in Children) Studies [62], the Singapore Cohort Study of the Risk Factors for Myopia (SCORM) Study [63], the Orinda [64] and Collaborative Longitudinal Evaluation of Ethnicity and Refractive Error (CLEERE) [34] Studies in the United States, the Sydney Myopia Study and the Sydney Adolescent Vascular and Eye Study (SMS and SAVES) [65], the Northern Ireland Childhood Errors of Refraction Study [66], the Ireland Eye Study [67], several studies from China [12, 58, 68, 69] and Poland [70, 71], have measured cycloplegic refractions. The Generation R birth cohort study [72] introduced cycloplegic refractions at an early stage, but after the initial data collection. Studies on pediatric and preschool samples are more limited, but cycloplegia has generally been used [27, 28, 35, 73,74,75,76,77]. Cycloplegia needs to be carefully controlled in these studies, because when the prevalence of myopia is low, even a small amount of pseudomyopia may have a significant effect on the apparent prevalence.
6.1.4 Myopia: The Importance of Causal Pathways
The aim of identifying risk factors is to generate preventive interventions. This requires identification of modifiable causal risk factors, not just associations. Observational epidemiological studies can only establish associations, but far too often, papers on cross-sectional associations conclude with a limitation that conclusions about causality cannot be drawn. This seems to imply that longitudinal data can establish causality, but this is not correct. Parallel longitudinal changes are equally ambiguous, and this makes understanding the epidemic of myopia difficult since many parameters, such as industrialization, urbanization, pollution, incomes, nutrition, and education, have changed over the same period.
Strictly, randomized intervention studies are required to establish causality, but often studies of this kind would be regarded as unethical. In many cases, we therefore have to rely on the nonrandomized trials that occur as a part of social development. For example, it would be unethical to offer differential educational opportunities to children in a randomly selected fashion, but social differences often lead to differential educational opportunities, and these can be highly informative, although they lack randomization. The high prevalence of myopia seen in Jewish males who received an intensive religious education, compared to their sisters who received a less intensive religious education, as well as boys and girls who received a less intensive secular education [78], provides rather compelling evidence of causality. In some cases, Mendelian randomization analysis can be used. Although there are strict limitations on its use [79, 80], it has already been usefully applied to the problem of myopia [81,82,83].
While the limitations of cross-sectional or longitudinal data are clear, avoiding discussing causality misses the point, because whether the association is potentially causal is crucial. Causality is therefore an important issue to discuss. Unfortunately, journals often discourage speculation, both explicitly and through tight word and reference limits. We recognize that wild speculation is not useful—Curtin’s description of the ophthalmologist waking after a sleepless night with “a new and usually more bizarre” hypothesis often rings true [84]. But evidence-based speculation should be encouraged, and subjected to critical review.
A related issue is the distinction between proximal and distal risk factors. Proximal risk factors are those closest to having a direct impact on eye growth, whereas distal factors will modulate the magnitude of more proximal factors. A plausible causal chain leads from education (excessive near work and limited time outdoors) to control of dopamine release and changes in axial elongation. Parental socioeconomic status, increases in the accessibility to education, and social attitudes to education may all increase involvement in education and/or result in reduced time outdoors, changing the prevalence of myopia through a common pathway that probably involves regulation of dopamine release and certainly involves regulation of axial elongation.
6.1.5 Statistical Analysis
There are also some associated considerations in relation to statistical analysis. A very common approach in analysis of risk factors is to identify significant associations in univariate analysis, then put the significant associations into multivariate regression analysis, labeling those factors that remain significant as “independent” risk factors. Often this appears to be done without serious consideration of whether the risk factors make sense in terms of causal pathways, whether they are modifiable, whether they are accurately measureable, whether they are likely to be collinear, whether they could result from reverse causality, and how complete the list of variables is. All these considerations can affect the validity of regression analysis.
Statistical analysis can be used more scientifically to address specific hypotheses about mediation by associated factors [85]. For example, in considering the relationship between myopia, education, and socioeconomic status (SES), it is possible to include SES and then the SES and education to see if there is a reduction in the strength of the association. This would be a sign that education mediates the influence of SES. In general, stepwise procedures need to be increasingly used in analysis. The quality of the variables is also important—whether they are continuous or categorical variables, and whether they can be accurately measured. Even if one factor is entirely mediated by another, if either variable is poorly measured, adjustment may only reduce rather than largely eliminate the effect of the other variable. Thus, the concept of independent risk factors is somewhat problematic [86].
Some of the variables are intrinsically complex. Education, for example, can be assessed in adults as a set of categories (primary school, high school, and university completed) or as a continuous variable (years of education). But intensity of education is also important, since children who obtain higher academic grades at a given level of schooling are more likely to be myopic. No variable captures both duration and intensity effectively. Myopia is associated with both intelligence quotient (IQ) [87] and school results [88]. Both are sometimes included in analyses to measure “innate” potential. But this approach is problematic since IQ shows complex changes over time [89, 90], and like myopia, combines significant twin heritability, considerable missing heritability, and social malleability [91]. There are both phenotypic and genetic correlations between myopia and IQ [92], as well as strong environmental effects. Inclusion of past performance or IQ in analyses as indicators of “innate potential,” may, if these measures are influenced by the same environmental factors that influence myopia, obscure their role.
Regression analyses that combine variables that are more or less proximal also pose a problem. It is well known that axial elongation is the biological basis of myopic shifts in refraction, and thus it correlates highly with SER. If axial length is included in a regression analysis with more distal factors, it will tend to obscure the associations of these more distal factors with refraction.
In summary, there are a number of conceptual and methodological issues that need careful consideration in seeking to tease out the risk factors for myopia. Further progress in the analysis of risk factors for myopia requires more critical approaches in several areas.
6.2 The Main Risk Factors for “School Myopia”: Education and Time Outdoors
We have recently reviewed this topic in detail, and these reviews can be consulted for more detailed references [1, 2, 4, 24]. In one of them, we distinguish between societies where the provision of education is limited and the prevalence of myopia is low (operationally, less than 10% myopia in young adults), at an age when 12 years of schooling would be the norm in the developed world, societies with western style education where the prevalence is between 10 and 60%, and developed countries in East and Southeast Asia, where education is intense, time outdoors is low, and the prevalence of myopia is high at 70–90%, particularly in urban areas.
It seems likely that the gradual transition from a very low prevalence in societies with little formal education to that typical of modern western societies with widespread education is predominantly due to the expansion of educational provision to cover all children, combined with increases in educational standards. Many of the countries in the first group are currently well short of the UN Sustainable Development Goals in Education for 2030 (https://sustainabledevelopment.un.org/sdg4, accessed January 30, 2019). The prevalence of myopia is unlikely to increase in these countries until progress is made in achieving these goals. In contrast, the very high prevalence rates now typical of developed countries in East and Southeast Asia compared to the much more moderate prevalence rates observed in western countries cannot be attributed to more years of education, but are probably related to more intense education, early onset of high homework loads, and much less time outdoors.
Because of the common observation in societies with significant educational provision that the prevalence of myopia increases with age, there is now a widespread assumption that myopia is affected by chronological age. However, the low prevalence of myopia in societies where educational opportunities are limited, suggests that it is not chronological age, but cumulative experience of schooling that is important, because without schooling most children do not become myopic. For this reason, it is often a useful sampling strategy to obtain a large sample of a given age, rather than smaller age-specific samples over a range of ages.
In adults, years of schooling is often used as a variable to look at associations with myopia. Consistent with the preceding arguments, those who have completed university education are more likely to be myopic than those who have completed secondary education, or primary education [55, 93]. Data from the UK Biobank dataset was recently used to address the issue of causality in relation to years of schooling and myopia [83]. The authors used two sets of genetic data—one on SNPs that associated with years of education, the other on SNPs associated with myopia. It was found that people with SNPs associated with more years of education were more likely to be myopic, whereas those with SNPs associated with more myopia did not have more years of education. This provides a clear direction of causation from years of education to myopia. It is important to note that this does not mean that variables such as years of education or myopia are predominantly genetically determined. The percentage of variation in the two parameters explained by identified SNPs was low in both cases.
Rapid historical increases in years of schooling cannot be explained by genetic change but must depend on more distal social changes that made mass education more available, and linked employment and salary to increased skill levels, creating a strong incentive for parents to extend the education of their children. In the west, the expansion of mass education paralleled industrialization and the rise of modern capitalism, and later the introduction of new technologies. Data suggesting that the prevalence of myopia has increased can in fact be traced back to the early years of last century in the United States [94]. In contrast, the rapid economic development of countries in East and Southeast Asia has been based on industrialization supported by rapid development of a skilled work force. This, combined with recent experiences of rural poverty and Confucian concepts of familial obligation and work ethic, appears to have taken educational intensity to a much higher level, producing the epidemic of myopia.
Intensity of schooling shows up in measures of achievements during schooling. There is a general association of higher school performance with more myopia, for a given number of years of schooling [88], with associations of more myopia with involvement in accelerated learning streams within or between schools [95,96,97], and use of after-school tutoring [98]. These factors seem to be associated with the strong international performance of school systems in the countries with high prevalence of myopia [99], and may point to possible control measures. For example, if involvement in accelerated learning streams starts early, this provides an incentive for parents to impose intensive study regimes on children early in life so that they get into the higher level streams, leading to early onset myopia and rapid progression. A possible reform would therefore be to delay the onset of myopia with policies designed to control the early introduction of accelerated or elite streams within or between schools, while controlling intensive academic streams in private schools, private tutorial colleges, or cram schools.
Analysis of the results of the E3 consortium suggested that education was not the whole story [56]. This is not surprising, since the role of time outdoors, discussed below, has been demonstrated. But the analysis rests on an implicit assumption that completion of primary education (defined in this paper as 16 years) posed the same educational load across the many decades of development of education systems. Putting to one side the rather unusual definition of primary school education, it is very unlikely that this is the case. Educational standards at the end of primary school when most children completed schooling to work as unskilled laborers or homemakers were almost certainly lower than they are today, when most children complete many further years of education, and a significant percentage engage in tertiary studies.
Two potentially more proximal factors that may be related to educational intensity have also been considered—reading distance and reading duration without breaks [100]. The first has a long history, dating back to Cohn, who supported the use of headrests to maintain head position while reading. These were abandoned, but it is not clear if that was because they were not effective, or because children refused to use with them. Some studies report a strong independent association of reading distance with myopia, but it is not clear whether the relationship is causal, or whether children decrease their reading distance as they become myopic. The use of headrests has been reintroduced in some schools in China, and many products are available online to control head position. Longitudinal studies are required to establish if close reading positions are adopted prior to, or in parallel with the development of myopia, and randomized trials are required to assess whether controlling reading position does reduce myopia. Reading without breaks has also often been regarded as a risk factor, and the rule of a 10 min break every 30 min forms part of myopia prevention in both Taiwan and mainland China. An intervention study is also important in this area.
6.2.1 The Search for Causal Mechanisms for Education
While there can be no doubt about the causal relationship between education and myopia, the causal pathways are less clear. Near work was generally considered to be a likely mediating factor; however, the evidence on near work has been rather inconsistent, although meta-analyses indicate that there is a relatively small effect [101]. Part of the problem may be that near work is generally estimated from questionnaire responses which are intrinsically not particularly reliable. It is also difficult because the slow development of myopia means that near work estimates need to cover periods of at least some months if not years to be related to significant changes in axial length or refraction. Recent development of instruments for objectively measuring near work may provide better estimates, although considerable thought will need to be given to the sampling frames, given the marked daily, weekly, and seasonal variations in near work demands.
Initial thinking about the causal connection invoked accommodation as the probable driver, but this idea has been increasingly called into question. Some of the important negative evidence came from animal experimentation, which showed that refractive development was not drastically affected by cutting the ciliary nerve [102], and that animals that had no accommodative capacity showed normal patterns of refractive development [103, 104]. The ability of atropine to block axial elongation was initially interpreted as supporting the hypothesis, but this judgment had to be reevaluated, since atropine was effective, even in animals such as chickens where control of accommodation involves nicotinic rather than muscarinic pathways.
Attention therefore shifted to the possibility of control by defocus on the retina, given the clear role for defocus signals in controlling axial elongation demonstrated in animal models [31]. The link to education was provided by the hypothesis that lags in accommodation would result in residual hyperopia, which could stimulate axial elongation. Testing this hypothesis has led to inconsistent results, and Mutti et al. have shown that accommodative lag may be a consequence rather than a cause [105]. There is thus no clear pattern of support for the hypothesis.
It has also been suggested that peripheral rather than central defocus may play the dominant role in controlling eye growth [106]. This hypothesis developed from evidence that trainee pilots with eye shapes that created peripheral hyperopia were more likely to become myopic [107], leading to the idea that peripheral hyperopia leads to the development of myopia. Elegant animal experiments then demonstrated that eyes could normally regulate eye growth, even if the central retina was ablated [108]. However, this hypothesis has not been supported by several lines of evidence. The interpretation of the original evidence on development of myopia has been questioned [109], and while there is a general trend for eyes to develop from more oblate to more prolate, the relationship between eye shape and refraction has been quite variable tend to appear [110]. It remains possible that peripheral hyperopic defocus might drive myopic progression but attempts to control myopic correction by reducing peripheral hyperopic have not been particularly successful. However, selective imposition of myopic defocus in the periphery may provide a method for controlling myopia [111].
A clear causal mechanism linking education to myopia has therefore not been identified, but the causal relationship nevertheless appears to be solid. A recent hypothesis is that the problem may be associated with the predominant use of black text on white background [112]. This hypothesis proposes that the problem lies in the balance of stimulation of ON and OFF visual pathways, with natural scenes leading to balanced stimulation, with black text on white heavily overstimulating OFF pathways. Given that ON-bipolar pathways stimulate release of dopamine from amacrine cells [113], and dopamine agonists can inhibit axial elongation in animal models [114, 115], this is a promising link.
6.2.2 Protection by Time Outdoors
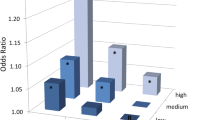
In contrast to the long history of speculation and research about near work, solid evidence that time outdoors was an important factor only became available about 10 years ago. However, since the first presentation of the evidence at the 2006 ARVO meeting, a large amount of evidence supporting the idea has been accumulated [2, 116], and a recent meta-analysis has confirmed the association [117]. Two key pieces of evidence are shown in Figs. 6.2 and 6.3 [118, 119]. The first shows that increased time outdoors can override the impact of increased near work, the second that increased time outdoors can override the impact of parental myopia. There are some reports that have failed to find this association, but these have generally been in situations where the amount of time spent outdoors is low and may be subthreshold for protection. The evidence now includes successful school-based intervention trials that have shown that increases in time outdoors of 40–80 min per day produced significant reductions in incident myopia [46, 120, 121], consistent with expectations from the epidemiological data.
Impacts of tertiles of near work and time outdoors on the odds ratio for myopia. Figure reproduced from Rose et al. [118]
Impact of hours playing sport (and outdoor activity) on the risk of myopia against number of myopic parents. Figure reproduced with permission from Jones et al. [119]
Rose et al. [118] postulated that the mechanism was probably that the brighter light outdoors during daylight hours led to more dopamine release, which in turn inhibited axial elongation. This hypothesis was confirmed by laboratory experiments that demonstrated the inhibitory effects of bright light on myopia, and the protective effect was shown to involve D2-dopamine receptors [122,123,124,125,126]. The plausible possibility that lower vitamin D levels, observed in children who spend less time outdoors, played a causal role has not been supported by further analysis [81, 82, 127]. The hypothesis that the protective effects might be due to a different balance of hyperopic and myopia defocus outdoors as compared to indoors [14], while plausible in terms of the results of animal experimentation, also seems unlikely, given that increasing light intensity alone blocks experimental myopia.
The question of causality has been settled with the randomized intervention trials in children. However, some issues are still unclear. The initial studies were based on distinctions between time spent outdoors and indoors, and were later linked to an operational definition of outdoors as involving light intensities over 1000 lux. Animal studies suggested that light intensities considerably higher, at least 10,000 lux, might be required to produce significant effects, but there is suggestive evidence that lower light intensities might be effective in humans [120, 128]. One intervention trial has even suggested that modest increases in classroom lighting strongly inhibit the development of myopia [129], and this study requires urgent replication. A school-based intervention trial in Taiwan has reported that more modest outdoor light exposures might also reduce incident myopia and, in fact, some research using objective measurements of light suggests that effective exposures of over a few thousand lux are rare outdoors, even though light appears to be regulating axial elongation. It would not be surprising if animal experimentation overestimated the light exposures required for protection, given that the stimulus for eye growth is strong and constant, whereas signals in humans may be more intermittent. It has also been suggested that the timing of the exposures, or their frequency, and parameters such as spectral composition may be important. There is limited and so far inconsistent support for these ideas, but the uncertainty around them does not matter when the simple intervention, increased time outdoors, is considered. These factors may become important if increased time outdoors needs to be supplemented or even replaced by artificial light exposures.
There is also controversy over whether increased time outdoors reduces progression as well as onset of myopia. The initial work did not support this possibility [118, 119, 130], but even at that time, there was considerable evidence that there were seasonal differences in progression that suggested that progression rates were slower in summer than in winter, suggesting regulation of progression consistent with the documented effects of near work and time outdoors [131,132,133,134,135]. More recent reports have provided evidence that more time outdoors may slow progression [120, 136], but more definitive work in this area is required.
6.3 Other Possible Risk Factors for Myopia
A range of other “independent” risk factors that are less obviously related to education have been documented. As well as summarizing the evidence for them, we will discuss whether they are likely to be mediated by exposures associated with education and time outdoors (Table 6.2).
6.3.1 Use of Computers and Smart Phones
In the last few years, use of computers and smart phones has become a routine part of daily life, with digital devices integrated into schooling in advanced countries. Increased digital screen time is often invoked to explain claimed continuing increases in the prevalence and severity of myopia, or at least as explanations for the increasingly early onset of myopia. Somewhat in advance of the available evidence, Taiwan has introduced laws controlling the amount of digital screen time that children of preschool age are allowed, although how regulations of this kind could be enforced is not clear. Dirani et al. [137] have recently asked whether increased digital screen time might be “the single modifiable risk factor for myopia,” accounting for “increased near-work activity and decreased outdoor activity.”
We suggest that this is unlikely, since the epidemic of myopia appeared well before the common use of these electronic devices. The prevalence of myopia was already high in Taiwan and Singapore for children born in the early 1960s, whereas the internet did not become available to the general public until 1993. Early analysis in the Sydney Myopia Study found that use of devices such as Nintendos and Game Boys was associated with less myopia, perhaps because they were predominantly used by academically underperforming boys. It is certainly possible that digital devices have now come to constitute a significant form of near work, and will correlate more closely with education and myopia, and some recent studies have documented significant associations between myopia and digital screen time [51, 138].
However, the historical perspective is important in considering preventive interventions. If limits are placed on the use of digital devices, children may simply revert to older forms of near work, such as reading books. And if digital devices encourage even more time indoors, active steps may need to be taken to get children to break with established behavior patterns and spend more time outdoors. We believe overemphasis on digital screen time may in fact have negative consequences if it leads to neglect of the other important factors. There is currently no evidence that time using digital devices is more dangerous than a similar amount of time reading. However, digital devices may make near work more pervasive and favor indoor lifestyles.
6.3.2 Sex
Many studies have compared the prevalence of myopia in males and females. Older studies have some tendency to report higher prevalence in males, whereas more recent studies more commonly report higher prevalence in females. For example, the Blue Mountains Eye Study reported that older male adults were more myopic than females [59], but the situation was reversed in the Sydney Myopia Study on children [42, 43, 96, 139]. Similarly, the Liwan Eye Study reported that sex differences in older adults were marginal [140], but in younger cohorts, girls were more likely to be myopic than boys [141]. The massive difference between the prevalence of myopia in girls and boys in Orthodox Jewish communities in Israel, where the boys undergo very intensive education from an early age, provides an extreme inverted example of this trend [78]. This variability does not suggest a direct biological link between sex and myopia, but rather suggests that the links may be mediated by social factors such as access to education for girls, which have changed considerably in many places in recent decades.
6.3.3 Ethnicity
Ethnicity or race has often been proposed as a risk factor for myopia, and indeed as evidence for genetic determination of myopia. It is however important to note that ethnicity and race covers both genetic differences, which are small in magnitude compared to the commonalities that make us human, and cultural differences that are often hard to quantify. Epidemiological evidence clearly shows major differences between ethnic groups in the prevalence of myopia, but more detailed analysis shows that these are predominantly due to environmental exposures. For example, the prevalence of myopia is high in the three major ethnic groups in Singapore, Chinese, Indian, and Malay, but in India and Malaysia, the prevalence is much lower [1, 4, 24]. This suggests that it is the environment of Singapore, perhaps most of all the education system and the limited time spent outdoors, that is crucial.
6.3.4 Parental Myopia
One of the best documented risk factors for myopia that is not directly linked to education is having myopic parents. Although shared attitudes to education are just as plausible, it is often assumed that this must be a genetic phenomenon, which is clearly true for the very genetic, highly familial forms of myopia. But the impact of parental myopia is also seen for “school myopia.” Consistently, studies covering a range of different ethnic groups have shown that having one or two myopic parents increases the risk of myopia [142,143,144,145,146,147], although the risk is inevitably lower in populations with a high prevalence of myopia. Wu and Edwards have shown the impact of parental myopia over three generations in China [148].
The effect of parental myopia is often cited as evidence for a genetic contribution to myopia, on the grounds that parents and children share genes. But they also share environments, and the possibility that myopic parents provide their children with environments more conducive for developing myopia, perhaps by placing more emphasis on education, has not been excluded, although early analyses did not support this hypothesis. However, recent studies have shown that children with myopic parents do not have a significantly greater level of myopia-associated SNPs [149], undermining the genetic argument. A more detailed analysis of environmental risk factors with objective measures of near work and time outdoors may be required to explain this association.
6.3.5 Intelligence
Higher intelligence or IQ, and some other cognitive measures, are generally associated with myopia. However, whether they are factors independent of education and time outdoors is not clear. While there are some genetic influences on these factors, as noted previously, like myopia, they are socially malleable, rather than genetically determined [89, 90]. In the SCORM Study, both academic grades and IQ scores appear to be independently associated with myopia [87, 88], and the same result has been obtained in a very large study of Israeli conscripts [150]. But the causal chains are very obscure, with a number of questions to be answered. For example, Are the links explained by the genetic correlations of the phenotypes? Do people with high IQs do more near work? Or is part of their high IQ achieved by more time reading and indoors? Do people with higher IQs need less effort to achieve high grades? Whatever the answer to these questions, reducing intelligence does not provide a route to prevention.
6.3.6 Urban/Rural Differences
Another consistent association of myopia is with urban residence, with children growing up in more rural areas generally being less myopic. In general, educational outcomes for children from rural areas are lower, and they often report less near work and more time outdoors. It is hard to see a direct link between axial elongation and postcode, and we suggest that urban/rural differences are likely to be based on differences in near work and time outdoors.
Population density has been invoked as one factor [151], but a major study in China has found that while there was a positive association of myopia with increased population density [152], even in the area with the lowest population density studied, the prevalence of myopia was still very high. This suggests other factors were more important. Access to green space has also been linked to lower use of spectacles [153].
6.3.7 Pollution
Pollution is one of the factors that has increased markedly since the Second World War in parts of East and Southeast Asia. There are many forms of pollution. Speculation about this factor has been fueled by the high levels of air pollution seem in some parts of China, but in fact, in international terms, Chinese cities rank well behind many cities in South Asia and the Middle East, where the prevalence of myopia is much lower than in Chinese cities (https://www.who.int/airpollution/data/cities/en/, accessed Jan 30, 2019). Increased use of spectacles, presumably for myopia, has been associated with traffic-related pollution [154], but this may in fact be related to the association between urban residence and more myopia, as well as links to socioeconomic status, area of residence, and education [153].
6.3.8 Housing
Nature of housing has also been invoked as a factor. However, the results in this area are currently quite contradictory. In Singapore, more spacious housing was associated with more myopia, possibly because of a causal chain involving socioeconomic status and its associations with education [60, 95]. In contrast, in both Sydney [151] and Hong Kong [155], small apartment dwelling has been associated with more myopia. A causal chain is not as obvious in this case and more complex pathways may need to be considered.
6.3.9 Height
Height has often been considered as a potential risk factor for myopia, and indeed, there have been substantial increases in height in populations over the last 150 years. These increases in height have been generally attributed to more adequate nutrition. It has also been argued that associations between height and myopia might be expected, given that taller people have longer axial lengths, but this argument does not take into account the “emmetropisation” mechanisms that produce substantial convergence of refractive status, despite differences in body stature. While several papers have reported that height is a risk factor for myopia [9, 156], the evidence on this is very inconsistent. Rosner et al. [157] reported that Israeli male military conscripts, who were not myopic, were taller and weighed more than those who were not myopic—the reverse of expectations. Another inconsistency lies in the difference in prevalence of myopia between males and females, with a higher prevalence of myopia being commonly reported in girls in recent studies. If there were a tight biological link between height and refraction, a more consistent relationship would be expected. Social factors affecting nutrition and education may therefore be important. Given the differences in height between the sexes, studies need to be carried out with population stratification wherever possible.
Rahi and colleagues reported that maternal height (and age) was associated with more myopia [47]. In the United Kingdom, height differs by socioeconomic status, with greater heights (and also maternal age) in higher SES groups [158]. Since children from higher SES groups are generally more myopic, these associations need to be tested for mediation by social factors.
6.3.10 Diet
Diets in East and Southeast Asia have changed significantly since the Second World War, and Cordain et al. argued that dietary change could have contributed to the increased prevalence of myopia, backed up by a plausible hypothesis linking insulin resistance, chronic hyperinsulinemia, increased circulating insulin-like growth factor (IGF-1), decreased circulating growth hormone, and decreased retinoid receptor signaling to increases in scleral growth [159]. But in other ways, this hypothesis has gained little support, and expected associations of height, weight, body mass index (BMI) and obesity with myopia have not been consistently observed. Improved diet is associated with greater height and axial length, but it does not appear to have produced increased refraction, consistent with the powerful eye growth control mechanisms that have been reported. It is also worth noting that international variations in overweight and obesity do not parallel the international distribution of myopia, with none of the countries with a high prevalence of myopia making the list of the top 20 countries ranked by percentage of obesity (https://www.who.int/gho/ncd/risk_factors/overweight/en/, accessed Jan 30, 2019) and thus there is little support for a tight biological link between diet and myopia.
6.3.11 Sleep
Associations between sleep and myopia have also been reported, but the evidence is quite inconsistent [160,161,162]. It is clear that children who have heavy study loads after school are likely to get less sleep, both because there is less time available, but also because mental activity close to bed time can disrupt sleep. Lack of sleep is more likely to be a problem in the senior years of school, when homework loads in many parts of East and Southeast Asia are very high. However, sleep deprivation is likely to be less common in the early primary years, when myopia first appears, which reduces the likelihood that it is a major risk factor. Kearney et al. [163] have recently reported that myopes in the Northern Ireland Childhood Errors of Refraction (NICER) Study have lower levels of serum melatonin, which could be linked to disruption of circadian rhythms, which have sometimes been linked to myopia [164], or could be linked to reduced light exposures and dopaminergic function.
6.3.12 Smoking
Maternal smoking was associated with a lower risk of myopia in the SCORM Study from Singapore, but there was no association with paternal smoking, and the number of mothers who smoked was small [165]. In the subsequent Strabismus, Amblyopia and Refractive Error in young Singaporean Children Study (STARS) Study, a stronger negative association with maternal and paternal smoking was reported [166]. A similar protective relationship was reported in a sample from a pediatric ophthalmology clinic, which largely persisted after adjustment for a range of factors including child’s near work activity and parental myopia and education [167]. A detailed study from South Korea reported consistent results for exposure to passive smoke estimated from urinary cotinine level [168], supporting the suggestion that nicotinic pathways are involved in the regulation of eye growth. In contrast, Rahi et al. reported a positive association between maternal smoking in early pregnancy and more myopia [47]. Although some of the associations reported are substantial, given the associations of smoking with SES and education, and lower gestational weight, these studies need to be carefully controlled for confounding.
6.3.13 Birth Order
Associations between myopia and birth order were reported with first-born children tending to be more myopic [49]. However, in educational studies, it is well documented that first-born children generally get more education [169]. A subsequent study on the UK Biobank dataset, adjusted for education, showed that the association between myopia and birth order was reduced but not eliminated [170]. In addition, in China, children from one child families are also more myopic than other children with siblings, and this was attributed to greater parental support for their child’s education [171]. However, the sociology of these differences is very complex, and more work needs to be done to establish that birth order is an independent risk factor.
6.3.14 Season of Birth
Season of birth has also been associated with myopia in several studies, with children born in summer tending to be more myopic [51, 172, 173]. No plausible pathway has been suggested for a direct link. Instead, it has been suggested that children born in summer often start school up to a year younger than their peers born in spring. This could mean first exposure to education and less time outdoors at an age when myopic shifts in refraction, including myopic progression, are larger, resulting in more myopia.
6.3.15 Allergic Conjunctivitis: Hay Fever and Kawasaki Disease
In 2011, Herbort et al. proposed a general association of myopia with inflammatory conditions affecting the choriocapillaris [174]. An association between ocular inflammatory conditions such as uveitis was subsequently demonstrated [175]. Later, a higher risk of myopia was associated with allergic conjunctivitis, and less so allergic rhinitis, atopic dermatitis, and asthma [176]. A large population-based study using the NHANES dataset showed that hay fever was also associated with a higher prevalence of high myopia [177]. A recent report has also associated increased myopia with Kawasaki disease [178], which has conjunctivitis as one of its core diagnostic criteria. This association needs further study.
These studies provide a persuasive case for some link between ocular allergic responses and the development of myopia, and using an animal model. Wei et al. have proposed a mechanism involving increased tumor necrosis factor (TNF)-alpha and interleukins. It does not seem likely that a link between ocular inflammation and myopia can explain the epidemic of myopia in East and Southeast Asia, since there is no parallel between the international distribution myopia and that of allergic rhinoconjunctivitis in children [179]. One possibility is that eye rubbing may lead to myopic refractions through corneal changes, as may be the case with keratoconus [180], but the US study on hay fever ruled out a role for them. The possibility that children with these conditions tend to spend less time outside should be examined. It also seems possible that allergic conditions might add to the incidence and progression of myopia, without being the primary determinant.
6.3.16 Febrile Diseases
Using data from the UK Biobank, Guggenheim et al. reported associations between several childhood diseases and myopia. From a list including pneumonia, encephalitis, meningitis, rheumatic fever, measles, rubella, mumps, diphtheria, and pertussis, rubella, mumps and pertussis were associated with myopia, while measles, rubella, and pertussis were associated with high myopia [181]. The authors argued against a link to educational disruption or limited time outdoors, since not all serious childhood diseases were linked to myopia, but this link, whatever its causes, cannot explain the emergence of the epidemic of myopia. But these findings may have clinical implications that need to be explored.
6.3.17 Fertility Treatment
The British TEDS Study has documented a standard range of social variables, with level of maternal education, summer birth, hours spent playing computer games surviving full multivariate regression analysis, with associations with socioeconomic status, educational attainment, reading enjoyment, and cognitive variables showing associations at multiple stages in the life-course analysis. A unique feature of the analysis was the protective associations of fertility treatment detected in the final analysis [51]. The authors ruled out associations with parental education, and the causes are currently obscure.
6.4 Popular Beliefs About the Causes of Myopia
There are many popular beliefs in the causes of myopia around the world, which have presumably arisen because the development of myopia and its progression is often observed by parents, who naturally seek explanations. In the western world, a common belief is that reading in dim light, or under the bed clothes causes vision to deteriorate, but this outcome, and these behaviors might indeed be common in those who like reading books, and there is some evidence that reading for pleasure is a risk factor for myopia. We have not attempted a systematic survey in this area, but in China, there seem to be many beliefs of this kind, perhaps because the prevalence of myopia has increased so conspicuously. One commonly encountered belief is that myopia is associated with reading and writing postures that violate the “foot, fist, inch” rule, that is the eyes should be one foot from the book, the chest should be one fist from the desk, and the fingers should be 1 inch from the nib of the pen. This is a variant on the idea that bad posture while reading leads to the development of myopia, which has widespread currency, but has never been proven. A similar common belief is that reading while riding on public transport is dangerous, but again this has never been tested. Other, intuitively unlikely ideas include the development of myopia in children who read on their back, or their front, or who read extracurricular books with font sizes greater than standard textbooks. These have occasionally been looked at in scientific papers, and associations have sometimes been reported. These proposed factors need to be rigorously tested epidemiologically, and if they survive that sort of testing, they need to be subjected to standard Randomised Control Trial (RCT) testing. Unfortunately, without any positive evidence, two of these have been written into China’s National Myopia Prevention Plan.
6.5 Comparing Genetic and Environmental Effects
Comparing the impact of genetic to environmental effects is difficult, because they are often quantified in different ways, with genetic effects generally determined in relation to variation in refraction, while evidence on the impact of environmental effects is generally related to prevalence of myopia. Genetic studies have identified myopia-associated SNPs that account for slightly less than 10% of the variance in refraction, although these factors are very precisely measured [13]. Compared to the possibly misplaced expectations from twin studies with heritability estimates in the range of 80–90%, there is obviously a large amount of what has come to be called missing heritability, although it is actually the identified genetic variation which is missing. Use of the SNP-heritability approach suggests that Genome-Wide Association Study (GWAS) has the potential to explain more like 20–35% of the variance [19]. It is often suggested that improvements in methodology and increases in sample size may help to close the gap, although there is a law of diminishing returns with increased sample sizes. There is little evidence that other forms of genetic variation such as copy number variations or rare genetic effects of large size have a role to play.
In contrast, the measurement of risk factor exposures is much less precise, largely involving estimates derived from questionnaires. These questionnaires not only suffer from the general problem of recall bias, but in relation to near work and time outdoors, they also ask for a very difficult task to be performed, namely estimating average exposures. Not surprisingly, such questionnaires are more accurate when estimates of recent exposures over a limited time frame are compared to measurements obtained using objective instruments, but even then, discrepancies are significant. We are therefore not yet in a position to calculate individual environmental risk factor effect sizes. This may explain why attempts to quantify the associations between estimates of near work and time outdoors on the one hand and myopia on the other explain only low percentages of the variance [19], despite the evidence that changes in them appear to be responsible for the major increases in prevalence of myopia seen recently.
We suggest that while awaiting more quantitative data on environmental risk exposures, the change in mean SER from the age of 5–6 to the end of schooling may be taken to represent the cumulative effect of relevant environmental exposures over this time. This assumption seems reasonable, given that children exposed to only limited schooling generally develop little myopia. The only exception that we have found to this statement is the increase in prevalence of myopia in Eskimo and Inuit children after exposure to rudimentary schooling in extreme northern latitudes [182,183,184]. This may be an exception due to the limited light exposures available during winter, combined with reductions in the amount of time spent outdoors.
The data from the RESC Study in Nepal, where there is little development of myopia and limited schooling, appears to be an average shift of −0.02 D/year with minimal schooling [185]. This figure is about −0.16 D/year of schooling in the Sydney Myopia Study. East Asian studies suggest a total change that is much greater. In the Guangzhou RESC Study, refraction changed by at least −3.00 D over 10 years, or a change of −0.3 D/year [141]. In the Shandong Eye Study [12], the change was closer to −4.00 D over 10 years, or −0.4 D/year. One limitation of this simple analysis is that it assumes linear changes across the years of schooling, which may not be the case. These values compare with estimates from the UK Biobank Study on refractive shifts associated with years of schooling, which suggest shifts from epidemiological analysis of −0.16 D/year, while the Mendelian randomization analysis gives a change of −0.28 D/year [83]. It is encouraging that these figures are in the same ballpark when children have been exposed to schooling, but it needs to be recognized that these figures are only first approximations.
Accepting these estimates as the best currently available, we can then attempt to use them to understand the onset of myopia. Age of onset may be a very significant parameter, since it has decreased markedly during the emergence of the myopia epidemics [11], appears to have similar genetic associations to the final level of myopia in adults [13, 186], and appears to be a good predictor of final myopia [187].
In several studies that have used rigorous cycloplegia, refractions at the age of 5–7 are concentrated in the range of 1.0–1.3 D. This range has been documented for children in East Asia [12, 46, 75, 188] and in western societies [43, 76, 96]. The similarity of the values reported for children of East Asian and European backgrounds is striking, given the massively different prevalence rates that emerge later. In other reports, mean SER has been lower, and we suggest that, in many of these cases, inadequate cycloplegia may be involved. This means that the challenge is to explain how genetic factors and environmental factors can produce myopic shifts of 1.5 D or more to reach the threshold for myopia of −0.5 D. The steps involved in moving from this baseline value to the threshold for myopia and onto final SER at the age of 20 are summarized in Fig. 6.4.
Environmentally driven axial elongation and myopic shifts in refraction lead to final refraction. Rate of axial elongation and myopia shifts in refraction increase prior to the onset of myopia, and remain elevated but decline with age [199, 200]. This increase may begin when SER reaches the low hyperopic range [201]. The earlier the onset, the greater the progression
In the ALSPAC Study [149] on average, participants carried around 130 risk alleles, with most of the distribution fitting within ±15 alleles [149]. If the average number of risk alleles corresponds to a mean SER of +1.0 to 1.3 D, with an average effect size of −0.1 D/risk allele, few participants would reach the myopia cutoff of −0.5 D on purely genetic grounds, and several years of schooling would be required to reach the threshold. Modeling by Ghorbani-Mojarrad et al. [149] also suggests that additive genetic effects are unlikely to produce “school myopia,” without additional contributions from environmental factors. In contrast, in East Asian environments, 2–4 years of schooling would bring many children past the myopia cutoff. This is likely to happen more rapidly for those who are already at risk genetically.
This picture is based on a number of assumptions and needs extensive further development in relation to the size and linearity of effects, but it provides a framework for understanding one of the distinctive characteristics of the current epidemic of “school myopia,” the importance of environmental exposures and the modest impact of genetic risk. It also suggests that more work needs to be done on the determinants of spherical equivalent at the age of 5–7 years, and determinants of the age of onset of myopia, because the latter is a significant predictor of subsequent progression and final myopia [187].
Gene–environment interactions also need to be taken into account. It must be remembered that simple environmental regulation of gene expression is not an example of gene–environment interactions. Gene–environment interactions are characterized by situations in which genetic risk and environmental risk combine to produce impacts which are greater or less than expected from their individual effects—or in other words that expression of different alleles of a given gene are differentially affected by different environments. Verhoeven et al. [189] have examined the specific case of the interaction of genetic risk and educational risk, reporting major interactions, with the combination of high genetic risk and high educational risk producing very high risk greater than the arithmetic sum of the individual risks, using the synergy analyses of Rothman [190,191,192]. Cortina-Borjas et al. [193] have argued that this method produces many false cases of gene–environment interactions, in part because when using odds ratios, risks are more likely to be multiplicative. From our analysis of the data in the paper by Verhoeven et al., the interactions in this paper are close to multiplicative, and we suggest that a more detailed analysis is required. A search for more specific cases has produced some more specific examples of gene–environment interactions [194,195,196,197], but the cases reported appear to be rare. More work is clearly needed in this complex area.
6.6 Conclusions
Our overview of risk factors for myopia has identified education and limited time outdoors as the major risk factors for myopia. We suggest that the evidence on these two risk factors is now very strong. These two factors both suggest evidence-based approaches to control of myopia, such as increased time outdoors and decreased near work time. These two factors then appear to converge to regulate eye growth, through cellular and biological pathways that require further definition, but appear to involve regulation of the rate of dopamine release as one component, and axial elongation as another.
Many other risk factors for myopia have been proposed. Many of them may be more distal social factors such as parental and social attitudes to education, provision of educational opportunities, and organization of school systems and schools, and may be mediated by the educational and time outdoor exposures that children receive.
We suggest that future studies in this area need to become more rigorous in several ways. Cycloplegia needs to the required standard in new studies. Mediation analysis needs to become a standard part of risk factor analysis, used to define causal pathways. As a minimum, new studies also need to collect data on educational exposures and time outdoors, increasingly using the new methods for collection of objective data that are becoming available. These higher standards need to be enforced by more rigorous review and publication processes.
While there are many aspects of the risk factors for myopia that require further analysis, the picture that we currently have provides important insight into the characteristics of the current epidemics. In particular, we can now see why genetic effects on “school myopia” are so slight, despite the evidence for a large number of SNPs of small effect size associated with myopia.
The evidence for a major role of education and time outdoors has given us insight into effective means of control, some of which are currently being implemented in myopia control rather than school myopia programs, such as programs aimed to increase time outdoors. Many other interventions at more distal levels also can be envisaged that might reduce early competition for privileged places in schools. When these are combined with clinical interventions to control myopia progression, and there are now many available, in principle, we now understand how to turn back the epidemic of myopia.
But understanding does not automatically lead to success in prevention; after all we have known how to correct refractive errors for some time, but uncorrected refractive error is still a major cause of visual impairment. Research into more effective means of preventing the onset and progression of myopia needs to, and will undoubtedly continue. But the next big challenge may already be to identify and overcome the barriers to implementation, so that we can ensure that we achieve what increasingly appears to be a realizable goal—the prevention of “school myopia.”
References
Morgan I, Rose K. How genetic is ‘school myopia’? Prog Retin Eye Res. 2005;24:1–38.
Morgan IG, French AN, Ashby RS, Guo X, Ding X, He M, et al. The epidemics of myopia: aetiology and prevention. Prog Retin Eye Res. 2018;62:134–49.
Morgan IG, He M, Rose KA. Epidemic of pathological myopia: what can laboratory studies and epidemiology tell us? Retina. 2017;37:989–97.
Morgan IG, Ohno-Matsui K, Saw SM. Myopia. Lancet. 2012;379:1739–48.
Hysi PG, Wojciechowski R, Rahi JS, Hammond CJ. Genome-wide association studies of refractive error and myopia, lessons learned, and implications for the future. Invest Ophthalmol Vis Sci. 2014;55:3344–51.
Wojciechowski R. Nature and nurture: the complex genetics of myopia and refractive error. Clin Genet. 2011;79:301–20.
Wojciechowski R, Cheng CY. Involvement of multiple molecular pathways in the genetics of ocular refraction and myopia. Retina. 2017;38:91–101.
Wojciechowski R, Hysi PG. Focusing in on the complex genetics of myopia. PLoS Genet. 2013;9:e1003442.
Jung SK, Lee JH, Kakizaki H, Jee D. Prevalence of myopia and its association with body stature and educational level in 19-year-old male conscripts in Seoul, South Korea. Invest Ophthalmol Vis Sci. 2012;53:5579–83.
Koh V, Yang A, Saw SM, Chan YH, Lin ST, Tan MM, et al. Differences in prevalence of refractive errors in young Asian males in Singapore between 1996-1997 and 2009-2010. Ophthalmic Epidemiol. 2014;21:247–55.
Lin LL, Shih YF, Hsiao CK, Chen CJ. Prevalence of myopia in Taiwanese schoolchildren: 1983 to 2000. Ann Acad Med Singap. 2004;33:27–33.
Wu JF, Bi HS, Wang SM, Hu YY, Wu H, Sun W, et al. Refractive error, visual acuity and causes of vision loss in children in Shandong, China. The Shandong Children Eye Study. PLoS One. 2013;8:e82763.
Tedja MS, Wojciechowski R, Hysi PG, Eriksson N, Furlotte NA, Verhoeven VJM, et al. Genome-wide association meta-analysis highlights light-induced signaling as a driver for refractive error. Nat Genet. 2018;50:834–48.
Flitcroft DI. The complex interactions of retinal, optical and environmental factors in myopia aetiology. Prog Retin Eye Res. 2012;31:622–60.
Sankaridurg PR, Holden BA. Practical applications to modify and control the development of ametropia. Eye. 2014;28:134–41.
Huang J, Wen D, Wang Q, McAlinden C, Flitcroft I, Chen H, et al. Efficacy comparison of 16 interventions for myopia control in children: a network meta-analysis. Ophthalmology. 2016;123:697–708.
Flitcroft DI, Loughman J, Wildsoet CF, Williams C, Guggenheim JA, Consortium C. Novel myopia genes and pathways identified from syndromic forms of myopia. Invest Ophthalmol Vis Sci. 2018;59:338–48.
Young TL. Molecular genetics of human myopia: an update. Optom Vis Sci. 2009;86:E8–E22.
Guggenheim JA, St Pourcain B, McMahon G, Timpson NJ, Evans DM, Williams C. Assumption-free estimation of the genetic contribution to refractive error across childhood. Mol Vis. 2015;21:621–32.
Baird PN, Schache M, Dirani M. The GEnes in Myopia (GEM) study in understanding the aetiology of refractive errors. Prog Retin Eye Res. 2010;29:520–42.
Hammond CJ, Snieder H, Gilbert CE, Spector TD. Genes and environment in refractive error: the twin eye study. Invest Ophthalmol Vis Sci. 2001;42:1232–6.
Lyhne N, Sjolie AK, Kyvik KO, Green A. The importance of genes and environment for ocular refraction and its determiners: a population based study among 20-45 year old twins. Br J Ophthalmol. 2001;85:1470–6.
Manolio TA, Collins FS, Cox NJ, Goldstein DB, Hindorff LA, Hunter DJ, et al. Finding the missing heritability of complex diseases. Nature. 2009;461:747–53.
Morgan IG, Rose KA. Myopia: is the nature-nurture debate finally over? Clin Exp Optom. 2019;102:3–17.
Varughese S, Varghese RM, Gupta N, Ojha R, Sreenivas V, Puliyel JM. Refractive error at birth and its relation to gestational age. Curr Eye Res. 2005;30:423–8.
Cook RC, Glasscock RE. Refractive and ocular findings in the newborn. Am J Ophthalmol. 1951;34:1407–13.
Mayer DL, Hansen RM, Moore BD, Kim S, Fulton AB. Cycloplegic refractions in healthy children aged 1 through 48 months. Arch Ophthalmol. 2001;119:1625–8.
Mutti DO, Mitchell GL, Jones LA, Friedman NE, Frane SL, Lin WK, et al. Axial growth and changes in lenticular and corneal power during emmetropization in infants. Invest Ophthalmol Vis Sci. 2005;46:3074–80.
Morgan IG, Rose KA, Ellwein LB, Refractive Error Study in Children Survey G. Is emmetropia the natural endpoint for human refractive development? An analysis of population-based data from the refractive error study in children (RESC). Acta Ophthalmol. 2010;88:877–84.
Sorsby A, Sheridan M, Leary GA, Benjamin B. Vision, visual acuity, and ocular refraction of young men: findings in a sample of 1,033 subjects. Br Med J. 1960;1:1394–8.
Wallman J, Winawer J. Homeostasis of eye growth and the question of myopia. Neuron. 2004;43:447–68.
French AN, Morgan IG, Mitchell P, Rose KA. Risk factors for incident myopia in Australian schoolchildren: the Sydney adolescent vascular and eye study. Ophthalmology. 2013;120:2100–8.
Ma X, Congdon N, Yi H, Zhou Z, Pang X, Meltzer ME, et al. Safety of spectacles for children’s vision: a cluster-randomized controlled trial. Am J Ophthalmol. 2015;160:897–904.
Zadnik K, Sinnott LT, Cotter SA, Jones-Jordan LA, Kleinstein RN, Manny RE, et al. Prediction of juvenile-onset myopia. JAMA Ophthalmol. 2015;133:683–9.
Ehrlich DL, Atkinson J, Braddick O, Bobier W, Durden K. Reduction of infant myopia: a longitudinal cycloplegic study. Vis Res. 1995;35:1313–24.
Lam CS, Tang WC, Tse DY, Tang YY, To CH. Defocus incorporated soft contact (DISC) lens slows myopia progression in Hong Kong Chinese schoolchildren: a 2-year randomised clinical trial. Br J Ophthalmol. 2014;98:40–5.
Morgan IG, Iribarren R, Fotouhi A, Grzybowski A. Cycloplegic refraction is the gold standard for epidemiological studies. Acta Ophthalmol. 2015;93:581–5.
Fotouhi A, Morgan IG, Iribarren R, Khabazkhoob M, Hashemi H. Validity of noncycloplegic refraction in the assessment of refractive errors: the Tehran Eye Study. Acta Ophthalmol. 2012;90:380–6.
He X, Zou H, Lu L, Zhao R, Zhao H, Li Q, et al. Axial length/corneal radius ratio: association with refractive state and role on myopia detection combined with visual acuity in Chinese school children. PLoS One. 2015;10:e0111766.
Tideman JWL, Polling JR, Vingerling JR, Jaddoe VWV, Williams C, Guggenheim JA, et al. Axial length growth and the risk of developing myopia in European children. Acta Ophthalmol. 2018;96:301–9.
Guo Y, Liu LJ, Tang P, Lv YY, Feng Y, Xu L, et al. Outdoor activity and myopia progression in 4-year follow-up of Chinese primary school children: The Beijing Children Eye Study. PLoS One. 2017;12:e0175921.
Ip JM, Huynh SC, Robaei D, Kifley A, Rose KA, Morgan IG, et al. Ethnic differences in refraction and ocular biometry in a population-based sample of 11-15-year-old Australian children. Eye. 2008;22:649–56.
Ojaimi E, Rose KA, Morgan IG, Smith W, Martin FJ, Kifley A, et al. Distribution of ocular biometric parameters and refraction in a population-based study of Australian children. Invest Ophthalmol Vis Sci. 2005;46:2748–54.
Iribarren R. Crystalline lens and refractive development. Prog Retin Eye Res. 2015;47:86–106.
Cumberland PM, Bountziouka V, Rahi JS. Impact of varying the definition of myopia on estimates of prevalence and associations with risk factors: time for an approach that serves research, practice and policy. Br J Ophthalmol. 2018;102:1407–12.
He M, Xiang F, Zeng Y, Mai J, Chen Q, Zhang J, et al. Effect of time spent outdoors at school on the development of myopia among children in China: a randomized clinical trial. JAMA. 2015;314:1142–8.
Rahi JS, Cumberland PM, Peckham CS. Myopia over the lifecourse: prevalence and early life influences in the 1958 British birth cohort. Ophthalmology. 2011;118:797–804.
Deere K, Williams C, Leary S, Mattocks C, Ness A, Blair SN, et al. Myopia and later physical activity in adolescence: a prospective study. Br J Sports Med. 2009;43:542–4.
Guggenheim JA, McMahon G, Northstone K, Mandel Y, Kaiserman I, Stone RA, et al. Birth order and myopia. Ophthalmic Epidemiol. 2013;20:375–84.
Guggenheim JA, Northstone K, McMahon G, Ness AR, Deere K, Mattocks C, et al. Time outdoors and physical activity as predictors of incident myopia in childhood: a prospective cohort study. Invest Ophthalmol Vis Sci. 2012;53:2856–65.
Williams KM, Kraphol E, Yonova-Doing E, Hysi PG, Plomin R, Hammond CJ. Early life factors for myopia in the British Twins Early Development Study. Br J Ophthalmol. 2019. https://doi.org/10.1136/bjophthalmol-2018-312439.
Vitale S, Ellwein L, Cotch MF, Ferris FL, Sperduto R. Prevalence of refractive error in the United States, 1999-2004. Arch Ophthalmol. 2008;126:1111–9.
Kim EC, Morgan IG, Kakizaki H, Kang S, Jee D. Prevalence and risk factors for refractive errors: Korean National Health and Nutrition Examination Survey 2008-2011. PLoS One. 2013;8:e80361.
Wolfram C, Hohn R, Kottler U, Wild P, Blettner M, Buhren J, et al. Prevalence of refractive errors in the European adult population: the Gutenberg Health Study (GHS). Br J Ophthalmol. 2014;98:857–61.
Cumberland PM, Bao Y, Hysi PG, Foster PJ, Hammond CJ, Rahi JS, et al. Frequency and distribution of refractive error in adult life: methodology and findings of the UK Biobank Study. PLoS One. 2015;10:e0139780.
Williams KM, Bertelsen G, Cumberland P, Wolfram C, Verhoeven VJ, Anastasopoulos E, et al. Increasing prevalence of myopia in Europe and the impact of education. Ophthalmology. 2015;122:1489–97.
Williams KM, Verhoeven VJ, Cumberland P, Bertelsen G, Wolfram C, Buitendijk GH, et al. Prevalence of refractive error in Europe: the European Eye Epidemiology (E(3)) Consortium. Eur J Epidemiol. 2015;30:305–15.
Wang Q, Klein BE, Klein R, Moss SE. Refractive status in the Beaver Dam Eye Study. Invest Ophthalmol Vis Sci. 1994;35:4344–7.
Attebo K, Ivers RQ, Mitchell P. Refractive errors in an older population: the Blue Mountains Eye Study. Ophthalmology. 1999;106:1066–72.
Wong TY, Foster PJ, Hee J, Ng TP, Tielsch JM, Chew SJ, et al. Prevalence and risk factors for refractive errors in adult Chinese in Singapore. Invest Ophthalmol Vis Sci. 2000;41:2486–94.
Hashemi H, Fotouhi A, Mohammad K. The age- and gender-specific prevalences of refractive errors in Tehran: the Tehran Eye Study. Ophthalmic Epidemiol. 2004;11:213–25.
Negrel AD, Maul E, Pokharel GP, Zhao J, Ellwein LB. Refractive error study in children: sampling and measurement methods for a multi-country survey. Am J Ophthalmol. 2000;129:421–6.
Saw SM, Carkeet A, Chia KS, Stone RA, Tan DT. Component dependent risk factors for ocular parameters in Singapore Chinese children. Ophthalmology. 2002;109:2065–71.
Zadnik K. The Glenn A. Fry Award Lecture (1995). Myopia development in childhood. Optom Vis Sci. 1997;74:603–8.
Ojaimi E, Rose KA, Smith W, Morgan IG, Martin FJ, Mitchell P. Methods for a population-based study of myopia and other eye conditions in school children: the Sydney Myopia Study. Ophthalmic Epidemiol. 2005;12:59–69.
Breslin KM, O’Donoghue L, Saunders KJ. A prospective study of spherical refractive error and ocular components among Northern Irish schoolchildren (the NICER study). Invest Ophthalmol Vis Sci. 2013;54:4843–50.
Harrington SC, Stack J, Saunders K, O’Dwyer V. Refractive error and visual impairment in Ireland school children. Br J Ophthalmol. 2019. https://doi.org/10.1136/bjophthalmol-2018-312573.
Li SM, Liu LR, Li SY, Ji YZ, Fu J, Wang Y, et al. Design, methodology and baseline data of a school-based cohort study in Central China: the Anyang Childhood Eye Study. Ophthalmic Epidemiol. 2013;20:348–59.
Zheng Y, Ding X, Chen Y, He M. The Guangzhou twin project: an update. Twin Res Hum Genet. 2013;16:73–8.
Czepita D, Mojsa A, Zejmo M. Prevalence of myopia and hyperopia among urban and rural schoolchildren in Poland. Ann Acad Med Stetin. 2008;54:17–21.
Czepita D, Zejmo M, Mojsa A. Prevalence of myopia and hyperopia in a population of Polish school children. Ophthalmic Physiol Opt. 2007;27:60–5.
Tideman JWL, Polling JR, Jaddoe VWV, Vingerling JR, Klaver CCW. Environmental risk factors can reduce axial length elongation and myopia incidence in 6- to 9-year-old children. Ophthalmology. 2019;126:127–36.
Ehrlich DL, Braddick OJ, Atkinson J, Anker S, Weeks F, Hartley T, et al. Infant emmetropization: longitudinal changes in refraction components from nine to twenty months of age. Optom Vis Sci. 1997;74:822–43.
Giordano L, Friedman DS, Repka MX, Katz J, Ibironke J, Hawes P, et al. Prevalence of refractive error among preschool children in an urban population: the Baltimore Pediatric Eye Disease Study. Ophthalmology. 2009;116:739–46.
Guo X, Fu M, Ding X, Morgan IG, Zeng Y, He M. Significant axial elongation with minimal change in refraction in 3- to 6-year-old chinese preschoolers: the Shenzhen Kindergarten Eye Study. Ophthalmology. 2017;124:1826–38.
Mutti DO, Sinnott LT, Lynn Mitchell G, Jordan LA, Friedman NE, Frane SL, et al. Ocular component development during infancy and early childhood. Optom Vis Sci. 2018;95:976–85.
Wen G, Tarczy-Hornoch K, McKean-Cowdin R, Cotter SA, Borchert M, Lin J, et al. Prevalence of myopia, hyperopia, and astigmatism in non-Hispanic white and Asian children: multi-ethnic pediatric eye disease study. Ophthalmology. 2013;120:2109–16.
Zylbermann R, Landau D, Berson D. The influence of study habits on myopia in Jewish teenagers. J Pediatr Ophthalmol Strabismus. 1993;30:319–22.
Paternoster L, Tilling K, Smith GD. Genetic epidemiology and Mendelian randomization for informing disease therapeutics: conceptual and methodological challenges. PLoS Genet. 2017;13:e1006944.
Smith GD, Ebrahim S. Mendelian randomization: prospects, potentials, and limitations. Int J Epidemiol. 2004;33:30–42.
Cuellar-Partida G, Lu Y, Kho PF, Hewitt AW, Wichmann HE, Yazar S, et al. Assessing the genetic predisposition of education on myopia: a Mendelian randomization study. Genet Epidemiol. 2016;40:66–72.
Cuellar-Partida G, Williams KM, Yazar S, Guggenheim JA, Hewitt AW, Williams C, et al. Genetically low vitamin D concentrations and myopic refractive error: a Mendelian randomization study. Int J Epidemiol. 2017;46:1882–90.
Mountjoy E, Davies N, Plotnikov D, Davey Smith G, Rodriguez S, Williams C, Guggenheim J, Atan D. Education and myopia: a Mendelian randomisation study. BMJ. 2018;361:k2022.
Curtin BJ. The myopias: basic science and clinical management. Philadelphia: Harper and Row; 1985.
Lee H, Herbert RD, McAuley JH. Mediation analysis. JAMA. 2019;321(7):697–8.
Brotman DJ, Walker E, Lauer MS, O’Brien RG. In search of fewer independent risk factors. Arch Intern Med. 2005;165:138–45.
Saw SM, Tan SB, Fung D, Chia KS, Koh D, Tan DT, et al. IQ and the association with myopia in children. Invest Ophthalmol Vis Sci. 2004;45:2943–8.
Saw SM, Cheng A, Fong A, Gazzard G, Tan DT, Morgan I. School grades and myopia. Ophthalmic Physiol Opt. 2007;27:126–9.
Shayer M, Ginsburg D. Thirty years on--a large anti-Flynn effect? (II): 13- and 14-year-olds. Piagetian tests of formal operations norms 1976-2006/7. Br J Educ Psychol. 2009;79:409–18.
Shayer M, Ginsburg D, Coe R. Thirty years on - a large anti-Flynn effect? The Piagetian test volume & heaviness norms 1975-2003. Br J Educ Psychol. 2007;77:25–41.
Sauce B, Matzel LD. The paradox of intelligence: heritability and malleability coexist in hidden gene-environment interplay. Psychol Bull. 2018;144:26–47.
Williams KM, Hysi PG, Yonova-Doing E, Mahroo OA, Snieder H, Hammond CJ. Phenotypic and genotypic correlation between myopia and intelligence. Sci Rep. 2017;7:45977.
Au Eong KG, Tay TH, Lim MK. Education and myopia in 110,236 young Singaporean males. Singap Med J. 1993;34:489–92.
Lee KE, Klein BE, Klein R, Wong TY. Changes in refraction over 10 years in an adult population: the Beaver Dam Eye study. Invest Ophthalmol Vis Sci. 2002;43:2566–71.
Quek TP, Chua CG, Chong CS, Chong JH, Hey HW, Lee J, et al. Prevalence of refractive errors in teenage high school students in Singapore. Ophthalmic Physiol Opt. 2004;24:47–55.
French AN, Morgan IG, Burlutsky G, Mitchell P, Rose KA. Prevalence and 5- to 6-year incidence and progression of myopia and hyperopia in Australian schoolchildren. Ophthalmology. 2013;120:1482–91.
Ma Y, Qu X, Zhu X, Xu X, Zhu J, Sankaridurg P, et al. Age-specific prevalence of visual impairment and refractive error in children aged 3-10 years in Shanghai, China. Invest Ophthalmol Vis Sci. 2016;57:6188–96.
Tsai DC, Fang SY, Huang N, Hsu CC, Chen SY, Chiu AW, et al. Myopia development among young schoolchildren: the myopia investigation study in Taipei. Invest Ophthalmol Vis Sci. 2016;57:6852–60.
Morgan IG, Rose KA. Myopia and international educational performance. Ophthalmic Physiol Opt. 2013;33:329–38.
Ip JM, Saw SM, Rose KA, Morgan IG, Kifley A, Wang JJ, et al. Role of near work in myopia: findings in a sample of Australian school children. Invest Ophthalmol Vis Sci. 2008;49:2903–10.
Huang HM, Chang DS, Wu PC. The association between near work activities and myopia in children-a systematic review and meta-analysis. PLoS One. 2015;10:e0140419.
Wildsoet C. Neural pathways subserving negative lens-induced emmetropization in chicks--insights from selective lesions of the optic nerve and ciliary nerve. Curr Eye Res. 2003;27:371–85.
McBrien NA, Moghaddam HO, New R, Williams LR. Experimental myopia in a diurnal mammal (Sciurus carolinensis) with no accommodative ability. J Physiol. 1993;469:427–41.
McBrien NA, Moghaddam HO, Reeder AP. Atropine reduces experimental myopia and eye enlargement via a nonaccommodative mechanism. Invest Ophthalmol Vis Sci. 1993;34:205–15.
Mutti DO, Mitchell GL, Hayes JR, Jones LA, Moeschberger ML, Cotter SA, et al. Accommodative lag before and after the onset of myopia. Invest Ophthalmol Vis Sci. 2006;47:837–46.
Smith EL. Prentice award lecture 2010: a case for peripheral optical treatment strategies for myopia. Optom Vis Sci. 2011;88:1029–44.
Hoogerheide J, Rempt F, Hoogenboom WP. Acquired myopia in young pilots. Ophthalmologica. 1971;163:209–15.
Smith EL, Hung LF, Huang J. Relative peripheral hyperopic defocus alters central refractive development in infant monkeys. Vis Res. 2009;49:2386–92.
Atchison DA, Rosen R. The possible role of peripheral refraction in development of myopia. Optom Vis Sci. 2016;93:1042–4.
Sng CC, Lin XY, Gazzard G, Chang B, Dirani M, Lim L, et al. Change in peripheral refraction over time in Singapore Chinese children. Invest Ophthalmol Vis Sci. 2011;52:7880–7.
Smith EL, Hung LF, Huang J, Arumugam B. Effects of local myopic defocus on refractive development in monkeys. Optom Vis Sci. 2013;90:1176–86.
Aleman AC, Wang M, Schaeffel F. Reading and myopia: contrast polarity matters. Sci Rep. 2018;8:10840.
Boelen MK, Boelen MG, Marshak DW. Light-stimulated release of dopamine from the primate retina is blocked by 1-2-amino-4-phosphonobutyric acid (APB). Vis Neurosci. 1998;15:97–103.
Iuvone PM, Tigges M, Stone RA, Lambert S, Laties AM. Effects of apomorphine, a dopamine receptor agonist, on ocular refraction and axial elongation in a primate model of myopia. Invest Ophthalmol Vis Sci. 1991;32:1674–7.
McCarthy CS, Megaw P, Devadas M, Morgan IG. Dopaminergic agents affect the ability of brief periods of normal vision to prevent form-deprivation myopia. Exp Eye Res. 2007;84:100–7.
French AN, Ashby RS, Morgan IG, Rose KA. Time outdoors and the prevention of myopia. Exp Eye Res. 2013;114:58–68.
Xiong S, Sankaridurg P, Naduvilath T, Zang J, Zou H, Zhu J, et al. Time spent in outdoor activities in relation to myopia prevention and control: a meta-analysis and systematic review. Acta Ophthalmol. 2017;95:551–66.
Rose KA, Morgan IG, Ip J, Kifley A, Huynh S, Smith W, et al. Outdoor activity reduces the prevalence of myopia in children. Ophthalmology. 2008;115:1279–85.
Jones LA, Sinnott LT, Mutti DO, Mitchell GL, Moeschberger ML, Zadnik K. Parental history of myopia, sports and outdoor activities, and future myopia. Invest Ophthalmol Vis Sci. 2007;48:3524–32.
Wu PC, Chen CT, Lin KK, Sun CC, Kuo CN, Huang HM, et al. Myopia prevention and outdoor light intensity in a school-based cluster randomized trial. Ophthalmology. 2018;125:1239–50.
Wu PC, Tsai CL, Hu CH, Yang YH. Effects of outdoor activities on myopia among rural school children in Taiwan. Ophthalmic Epidemiol. 2010;17:338–42.
Ashby R, Ohlendorf A, Schaeffel F. The effect of ambient illuminance on the development of deprivation myopia in chicks. Invest Ophthalmol Vis Sci. 2009;50:5348–54.
Ashby RS, Schaeffel F. The effect of bright light on lens compensation in chicks. Invest Ophthalmol Vis Sci. 2010;51:5247–53.
Karouta C, Ashby RS. Correlation between light levels and the development of deprivation myopia. Invest Ophthalmol Vis Sci. 2015;56:299–309.
Smith EL, Hung LF, Arumugam B, Huang J. Negative lens-induced myopia in infant monkeys: effects of high ambient lighting. Invest Ophthalmol Vis Sci. 2013;54:2959–69.
Smith EL, Hung LF, Huang J. Protective effects of high ambient lighting on the development of form-deprivation myopia in rhesus monkeys. Invest Ophthalmol Vis Sci. 2012;53:421–8.
Guggenheim JA, Williams C, Northstone K, Howe LD, Tilling K, St Pourcain B, et al. Does vitamin D mediate the protective effects of time outdoors on myopia? Findings from a prospective birth cohort. Invest Ophthalmol Vis Sci. 2014;55:8550–8.
Read SA, Collins MJ, Vincent SJ. Light exposure and eye growth in childhood. Invest Ophthalmol Vis Sci. 2015;56:6779–87.
Hua WJ, Jin JX, Wu XY, Yang JW, Jiang X, Gao GP, et al. Elevated light levels in schools have a protective effect on myopia. Ophthalmic Physiol Opt. 2015;35:252–62.
Jones-Jordan LA, Sinnott LT, Cotter SA, Kleinstein RN, Manny RE, Mutti DO, et al. Time outdoors, visual activity, and myopia progression in juvenile-onset myopes. Invest Ophthalmol Vis Sci. 2012;53:7169–75.
Cui D, Trier K, Munk R-MS. Effect of day length on eye growth, myopia progression, and change of corneal power in myopic children. Ophthalmology. 2013;120:1074–9.
Deng L, Gwiazda J, Thorn F. Children’s refractions and visual activities in the school year and summer. Optom Vis Sci. 2010;87:406–13.
Deng L, Pang Y. The role of outdoor activity in myopia prevention. Eye Sci. 2015;30:137–9.
Donovan L, Sankaridurg P, Ho A, Chen X, Lin Z, Thomas V, et al. Myopia progression in Chinese children is slower in summer than in winter. Optom Vis Sci. 2012;89:1196–202.
Gwiazda J, Deng L, Manny R, Norton TT, Group CS. Seasonal variations in the progression of myopia in children enrolled in the correction of myopia evaluation trial. Invest Ophthalmol Vis Sci. 2014;55:752–8.
Sanchez-Tocino H, Villanueva Gomez A, Gordon Bolanos C, Alonso Alonso I, Vallelado Alvarez A, Garcia Zamora M, et al. The effect of light and outdoor activity in natural lighting on the progression of myopia in children. J Fr Ophtalmol. 2019;42:2–10.
Dirani M, Crowston JG, Wong TY. From reading books to increased smart device screen time. Br J Ophthalmol. 2019;103:1–2.
Saxena R, Vashist P, Tandon R, Pandey RM, Bhardawaj A, Gupta V, et al. Incidence and progression of myopia and associated factors in urban school children in Delhi: The North India Myopia Study (NIM Study). PLoS One. 2017;12:e0189774.
Ojaimi E, Robaei D, Rochtchina E, Rose KA, Morgan IG, Mitchell P. Impact of birth parameters on eye size in a population-based study of 6-year-old Australian children. Am J Ophthalmol. 2005;140:535–7.
He M, Huang W, Li Y, Zheng Y, Yin Q, Foster PJ. Refractive error and biometry in older Chinese adults: the Liwan eye study. Invest Ophthalmol Vis Sci. 2009;50:5130–6.
He M, Zeng J, Liu Y, Xu J, Pokharel GP, Ellwein LB. Refractive error and visual impairment in urban children in Southern China. Invest Ophthalmol Vis Sci. 2004;45:793–9.
Edwards MH. Effect of parental myopia on the development of myopia in Hong Kong Chinese. Ophthalmic Physiol Opt. 1998;18:477–83.
Ip JM, Huynh SC, Robaei D, Rose KA, Morgan IG, Smith W, et al. Ethnic differences in the impact of parental myopia: findings from a population-based study of 12-year-old Australian children. Invest Ophthalmol Vis Sci. 2007;48:2520–8.
Liang CL, Yen E, Su JY, Liu C, Chang TY, Park N, et al. Impact of family history of high myopia on level and onset of myopia. Invest Ophthalmol Vis Sci. 2004;45:3446–52.
Mutti DO, Mitchell GL, Moeschberger ML, Jones LA, Zadnik K. Parental myopia, near work, school achievement, and children’s refractive error. Invest Ophthalmol Vis Sci. 2002;43:3633–40.
Xiang F, He M, Morgan IG. The impact of parental myopia on myopia in Chinese children: population-based evidence. Optom Vis Sci. 2012;89:1487–96.
Xiang F, He M, Morgan IG. The impact of severity of parental myopia on myopia in Chinese children. Optom Vis Sci. 2012;89:884–91.
Wu MM, Edwards MH. The effect of having myopic parents: an analysis of myopia in three generations. Optom Vis Sci. 1999;76:387–92.
Ghorbani Mojarrad N, Williams C, Guggenheim JA. A genetic risk score and number of myopic parents independently predict myopia. Ophthalmic Physiol Opt. 2018;38:492–502.
Rosner M, Belkin M. Intelligence, education, and myopia in males. Arch Ophthalmol. 1987;105:1508–11.
Ip JM, Rose KA, Morgan IG, Burlutsky G, Mitchell P. Myopia and the urban environment: findings in a sample of 12-year-old Australian school children. Invest Ophthalmol Vis Sci. 2008;49:3858–63.
Zhang M, Li L, Chen L, Lee J, Wu J, Yang A, et al. Population density and refractive error among Chinese children. Invest Ophthalmol Vis Sci. 2010;51:4969–76.
Dadvand P, Sunyer J, Alvarez-Pedrerol M, Dalmau-Bueno A, Esnaola M, Gascon M, et al. Green spaces and spectacles use in schoolchildren in Barcelona. Environ Res. 2017;152:256–62.
Dadvand P, Nieuwenhuijsen MJ, Basagana X, Alvarez-Pedrerol M, Dalmau-Bueno A, Cirach M, et al. Traffic-related air pollution and spectacles use in schoolchildren. PLoS One. 2017;12:e0167046.
Choi KY, Yu WY, Lam CHI, Li ZC, Chin MP, Lakshmanan Y, et al. Childhood exposure to constricted living space: a possible environmental threat for myopia development. Ophthalmic Physiol Opt. 2017;37:568–75.
Saw SM, Chua WH, Hong CY, Wu HM, Chia KS, Stone RA, et al. Height and its relationship to refraction and biometry parameters in Singapore Chinese children. Invest Ophthalmol Vis Sci. 2002;43:1408–13.
Rosner M, Laor A, Belkin M. Myopia and stature: findings in a population of 106,926 males. Eur J Ophthalmol. 1995;5:1–6.
Bann D, Johnson W, Li L, Kuh D, Hardy R. Socioeconomic inequalities in childhood and adolescent body-mass index, weight, and height from 1953 to 2015: an analysis of four longitudinal, observational, British birth cohort studies. Lancet Public Health. 2018;3:e194–203.
Cordain L, Eaton SB, Brand Miller J, Lindeberg S, Jensen C. An evolutionary analysis of the aetiology and pathogenesis of juvenile-onset myopia. Acta Ophthalmol Scand. 2002;80:125–35.
Jee D, Morgan IG, Kim EC. Inverse relationship between sleep duration and myopia. Acta Ophthalmol. 2016;94:e204–10.
Zhou Z, Morgan IG, Chen Q, Jin L, He M, Congdon N. Disordered sleep and myopia risk among Chinese children. PLoS One. 2015;10:e0121796.
Ayaki M, Torii H, Tsubota K, Negishi K. Decreased sleep quality in high myopia children. Sci Rep. 2016;6:33902.
Kearney S, O’Donoghue L, Pourshahidi LK, Cobice D, Saunders KJ. Myopes have significantly higher serum melatonin concentrations than non-myopes. Ophthalmic Physiol Opt. 2017;37:557–67.
Chakraborty R, Ostrin LA, Nickla DL, Iuvone PM, Pardue MT, Stone RA. Circadian rhythms, refractive development, and myopia. Ophthalmic Physiol Opt. 2018;38:217–45.
Saw SM, Chia KS, Lindstrom JM, Tan DT, Stone RA. Childhood myopia and parental smoking. Br J Ophthalmol. 2004;88:934–7.
Iyer JV, Low WC, Dirani M, Saw SM. Parental smoking and childhood refractive error: the STARS study. Eye. 2012;26:1324–8.
Stone RA, Wilson LB, Ying GS, Liu C, Criss JS, Orlow J, et al. Associations between childhood refraction and parental smoking. Invest Ophthalmol Vis Sci. 2006;47:4277–87.
Nam GE, Hwang BE, Lee YC, Paik JS, Yang SW, Chun YH, et al. Lower urinary cotinine level is associated with a trend toward more myopic refractive errors in Korean adolescents. Eye. 2017;31:1060–7.
Booth AJ, Kee HJ. Birth order matters. The effect of family size and birth order on educational attainment. J Popul Econ. 2009;22:367–97.
Guggenheim JA, Williams C, UK Biobank Eye and Vision Consortium. Role of educational exposure in the association between myopia and birth order. JAMA Ophthalmol. 2015;133:1408–14.
Zhao L, Zhou M. Do only children have poor vision? Evidence from China’s one-child policy. Health Econ. 2018;27:1131–46.
Mandel Y, Grotto I, El-Yaniv R, Belkin M, Israeli E, Polat U, et al. Season of birth, natural light, and myopia. Ophthalmology. 2008;115:686–92.
McMahon G, Zayats T, Chen YP, Prashar A, Williams C, Guggenheim JA. Season of birth, daylight hours at birth, and high myopia. Ophthalmology. 2009;116:468–73.
Herbort CP, Papadia M, Neri P. Myopia and inflammation. J Ophthalmic Vis Res. 2011;6:270–83.
Lin HJ, Wei CC, Chang CY, Chen TH, Hsu YA, Hsieh YC, et al. Role of chronic inflammation in myopia progression: clinical evidence and experimental validation. EBioMedicine. 2016;10:269–81.
Wei CC, Kung YJ, Chen CS, Chang CY, Lin CJ, Tien PT, et al. Allergic conjunctivitis-induced retinal inflammation promotes myopia progression. EBioMedicine. 2018;28:274–86.
Shafer BM, Qiu M, Rapuano CJ, Shields CL. Association between hay fever and high myopia in United States adolescents and adults. Eye Contact Lens. 2017;43:186–91.
Kung YJ, Wei CC, Chen LA, Chen JY, Chang CY, Lin CJ, et al. Kawasaki disease increases the incidence of myopia. Biomed Res Int. 2017;2017:2657913.
Ait-Khaled N, Pearce N, Anderson HR, Ellwood P, Montefort S, Shah J, et al. Global map of the prevalence of symptoms of rhinoconjunctivitis in children: The International Study of Asthma and Allergies in Childhood (ISAAC) phase three. Allergy. 2009;64:123–48.
Gordon-Shaag A, Millodot M, Kaiserman I, Sela T, Barnett Itzhaki G, Zerbib Y, et al. Risk factors for keratoconus in Israel: a case-control study. Ophthalmic Physiol Opt. 2015;35:673–81.
Guggenheim JA, Williams C, Eye UKB, Vision C. Childhood febrile illness and the risk of myopia in UK Biobank participants. Eye. 2016;30:608–14.
Morgan RW, Munro M. Refractive problems in Northern natives. Can J Ophthalmol. 1973;8:226–8.
Morgan RW, Speakman JS, Grimshaw SE. Inuit myopia: an environmentally induced “epidemic”? Can Med Assoc J. 1975;112:575–7.
Young FA, Leary GA, Baldwin WR, West DC, Box RA, Harris E, et al. The transmission of refractive errors within eskimo families. Am J Optom Arch Am Acad Optom. 1969;46:676–85.
Pokharel GP, Negrel AD, Munoz SR, Ellwein LB. Refractive error study in children: results from Mechi zone, Nepal. Am J Ophthalmol. 2000;129:436–44.
Verhoeven VJ, Hysi PG, Wojciechowski R, Fan Q, Guggenheim JA, Hohn R, et al. Genome-wide meta-analyses of multiancestry cohorts identify multiple new susceptibility loci for refractive error and myopia. Nat Genet. 2013;45:314–8.
Chua SY, Sabanayagam C, Cheung YB, Chia A, Valenzuela RK, Tan D, et al. Age of onset of myopia predicts risk of high myopia in later childhood in myopic Singapore children. Ophthalmic Physiol Opt. 2016;36:388–94.
Lan W, Zhao F, Lin L, Li Z, Zeng J, Yang Z, et al. Refractive errors in 3-6 year-old Chinese children: a very low prevalence of myopia? PLoS One. 2013;8:e78003.
Verhoeven VJ, Buitendijk GH, Consortium for Refractive E, Myopia, Rivadeneira F, Uitterlinden AG, et al. Education influences the role of genetics in myopia. Eur J Epidemiol. 2013;28:973–80.
Rothman KJ. Synergy and antagonism in cause-effect relationships. Am J Epidemiol. 1974;99:385–8.
Rothman KJ. The estimation of synergy or antagonism. Am J Epidemiol. 1976;103:506–11.
Rothman KJ. Estimation versus detection in the assessment of synergy. Am J Epidemiol. 1978;108:9–11.
Cortina-Borja M, Smith AD, Combarros O, Lehmann DJ. The synergy factor: a statistic to measure interactions in complex diseases. BMC Res Notes. 2009;2:105.
Fan Q, Guo X, Tideman JW, Williams KM, Yazar S, Hosseini SM, et al. Childhood gene-environment interactions and age-dependent effects of genetic variants associated with refractive error and myopia: The CREAM Consortium. Sci Rep. 2016;6:25853.
Fan Q, Verhoeven VJ, Wojciechowski R, Barathi VA, Hysi PG, Guggenheim JA, et al. Meta-analysis of gene-environment-wide association scans accounting for education level identifies additional loci for refractive error. Nat Commun. 2016;7:11008.
Fan Q, Wojciechowski R, Kamran Ikram M, Cheng CY, Chen P, Zhou X, et al. Education influences the association between genetic variants and refractive error: a meta-analysis of five Singapore studies. Hum Mol Genet. 2014;23:546–54.
Tkatchenko AV, Tkatchenko TV, Guggenheim JA, Verhoeven VJ, Hysi PG, Wojciechowski R, et al. APLP2 regulates refractive error and myopia development in mice and humans. PLoS Genet. 2015;11:e1005432.
Xu L, Li J, Cui T, Hu A, Fan G, Zhang R, et al. Refractive error in urban and rural adult Chinese in Beijing. Ophthalmology. 2005;112(10):1676–83.
Mutti DO, Sinnott LT, Mitchell GL, Jones-Jordan LA, Moeschberger ML, Cotter SA, et al. Relative peripheral refractive error and the risk of onset and progression of myopia in children. Invest Ophthalmol Vis Sci. 2011;52(1):199–205.
Xiang F, He M, Morgan IG. Annual changes in refractive errors and ocular components before and after the onset of myopia in Chinese children. Ophthalmology. 2012;119(7):1478–84.
Thorn F, Gwiazda J, Held R. Myopia progression is specified by a double exponential growth function. Optom Vis Sci. 2005;82(4):286–97.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2020 The Author(s)
About this chapter
Cite this chapter
Morgan, I.G., French, A.N., Rose, K.A. (2020). Risk Factors for Myopia: Putting Causal Pathways into a Social Context. In: Ang, M., Wong, T. (eds) Updates on Myopia. Springer, Singapore. https://doi.org/10.1007/978-981-13-8491-2_6
Download citation
DOI: https://doi.org/10.1007/978-981-13-8491-2_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-8490-5
Online ISBN: 978-981-13-8491-2
eBook Packages: MedicineMedicine (R0)