Abstract
Cardiomyocytes proliferate profusely during early development and for a brief period after birth in mammals. Within a month after birth, this proliferative capability is dramatically reduced in mammals unlike lower vertebrates where it persists into adult life. The zebrafish, for example, retains the ability to regenerate the apex of the heart following resection by a mechanism predominantly driven by cardiomyocyte proliferation. Differences in proliferative capacity of cardiomyocytes in adulthood between mammals and lower vertebrates are closely liked to ontogenetic or phylogenetic factors. Elucidation of these factors has the potential to provide enormous benefits if they lead to the development of therapeutic strategies that facilitate cardiomyocyte proliferation. In this review, we highlight the differences between Mammalian and Zebrafish cardiomyocytes, which could explain at least in part the different proliferative capacities in these two species. We discuss the advantages of the zebrafish as a model of cardiomyocyte proliferation, particularly at the embryonic stage. We also identify a number of key molecular pathways with potential to reveal key steps in switching cardiomyocytes from a quiescent to a proliferative phenotype.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ischaemic heart disease is the commonest cause of heart failure in developed countries either as a consequence of acute myocardial infarction or chronic ischaemic damage [1]. In addition the impact of an ageing population with degenerative cardiac disease associated with hypertension or valvular heart disease also contributes to cardiomyocyte dysfunction, loss and ultimately heart failure [2–4]. In the face of injury and progressive loss of cardiomyocytes, the human heart typically responds with hypertrophy combined with hyperplasia of non-cardiomyocyte cell populations in the heart including fibroblasts [5].
Embryonic and fetal hearts of most vertebrates, including mammals, typically show some degree of regenerative capacity, although studies in neonatal mice suggest it can last for up to 7 days after birth [6]. The limited capacity of the adult mammalian heart to restore lost cardiomyocytes following injury contrasts starkly with the highly effective regenerative process in adult hearts from lower vertebrates including amphibians and fish. Recently, the underlying mechanisms of heart regeneration in the adult zebrafish have become clearer with evidence that mature cardiomyocytes dedifferentiate, re-enter the cell cycle and then proliferate to support repair and replacement of injured myocardium [7, 8]. The zebrafish embryo has also contributed significantly to elucidate mechanisms of cardiac growth and development [9]. Despite being a two-chambered organ, the zebrafish heart exhibits many similarities to the mammalian heart and cellular and molecular studies clearly illustrate the common evolutionary origin of these structures [10]. Since the regenerative process appears to reactivate developmental pathways [11], understanding zebrafish cardiac development also helps us to understand the powerful cardiac regenerative ability in the adult zebrafish heart. A key question is whether a better understanding of the zebrafish cardiac regenerative process provides important insights into the mammalian response to injury and repair, and it remains an exciting challenge to determine if the regenerative capacity of the mammalian heart could be harnessed as a therapeutic strategy with significant clinical value.
There is no doubt, however, that the zebrafish represents an interesting model in which to study cardiomyocyte proliferation and cardiomyocyte hyperplasia following injury. One major advantage of utilizing zebrafish embryos for studies of cardiogenesis is that the embryo is able to survive for several days without a functioning cardiovascular system. This permits the study of cardiac phenotypes that would otherwise be lethal in mammalian model systems [12]. This review examines the differences and similarities in the response of mammals and zebrafish to cardiac injury. We also highlight several unique properties of the zebrafish embryo as a model of cardiomyocyte proliferation.
Cardiomyocyte proliferation: from early embryos to adulthood
Embryonic development
The heart is the first organ to visibly form and function during embryogenesis [13]. In Vertebrates, cardiogenesis is a morphologically complex process that involves sequential heart primordia migration, folding, looping, septation and maturation to form the chambered heart. The following description of heart development applies mainly to the mammalian heart although many aspects also apply to the zebrafish, and chicken heart. During embryogenesis, proliferation of new cardiomyocytes is the main source for the heart growth. Cardiomyocytes proliferate along the heart tube walls and within the atrioventricular septum. In particular, the outer surface of the heart, also called the compact region [14], achieves the highest proliferative rate. The epicardium, the thin layer of cells enveloping the heart, provides a source of mitogenic signaling that stimulate proliferation of cardiomyocytes within the compact zone [15]. A key epicardial-derived regulator of cardiac growth includes retinoic acid and its related receptors [16]. Newly formed cardiomyocytes thicken the ventricular wall and organize fingerlike projections along the inner ventricle surface giving rise to trabeculae, structures that increase force of contraction and improve oxygen and nutrient exchange for the heart itself. The endocardium, the specialized single-cell inner layer of the heart, also provides fundamental growth signals for embryonic cardiomyocytes, including peptides of the neuregulin family and their related tyrosine kinase receptors [17]. These growth factors also play an important role in promoting the normal “ballooning” of the outer curvature of the ventricle [18]. The intense cardiomyocyte proliferative activity observed during embryonic heart growth is accompanied by increasing intra-cavity shear forces that contribute to the shaping of the early heart [19].
Fetal to adulthood transition
While cardiomyocytes divide extensively and rapidly during fetal life, in mammals they lose their proliferative capacity shortly after birth. The proliferative activity of murine cardiomyocytes starts to decrease around E10–12 [20]. One key molecular player at this stage is Jumonji (jarid2) that acts to inhibit cardiomyocyte proliferation through repression of cyclin D1 expression. Jumonji appears to repress cyclin D1 transcription by recruiting histone H3–K9 methyltransferases, G9a and GLP, to the cyclin D1 promoter [21]. Indeed, jarid2 mutant mice demonstrate increased proliferation and overexpression of cyclin D1 in cardiomyocytes at E10 [22] [20].
In humans, cardiomyocyte proliferative capacity is lost by a few months after birth when cardiomyocytes withdraw from the cell cycle and remain in G0 stage, apparently indefinitely [23–26], a process called terminal differentiation. Downregulation of several fetal genes and upregulation of genes responsible for the adult phenotype play important roles in this process. Cyclin-dependent kinases (CDKs) play a core functional role in the cell cycle machinery. Sequential activation of different CDKs, forming complexes with their specific cyclins, allows progression of the cell cycle. In mammals, CDK4/6–cyclin D is activated in phase G1, CDK2–cyclin E in phase G1/S, CDK2/1–cyclin A in phase S/G2 and CDK1–cyclin B in phase M. Diminished CDK activity leads to attenuation or cessation of the cell cycle. The expression and activity of many cyclins and CDKs change synchronously during embryonic and postnatal developmental stages [27], suggesting a highly orchestrated series of cellular mechanisms controlling their role in proliferation. CDKs are regulated by CDK inhibitors (CKIs) including the INK4 family (p15, p16, p18 and p19) and Cip/Kip family (p21, p27 and p57) [27]. CKIs participate in termination of postnatal mammalian cardiomyocyte cell cycle as demonstrated in p21 and p27 knockout mice, where cardiomyocytes exit the cell cycle at G1-phase [28]. Meis1 is a transcriptional factor that is known to activate p21 and regulate cardiac cell cycle exit [29]. In fact, cardiomyocyte proliferative activity is prolonged after birth in Meis1 KO mice.
The cardiomyocyte cell cycle appears closely coupled to the accumulation of cell mass during development which acts to maintain consistent cell size [30]. In most species, this transition from hyperplastic-to-hypertrophic activity is characterized by changes in degree of ploidy and number of nuclei as cardiomyocytes undergo additional DNA replication followed by cytokinesis and/or karyokinesis [31, 32] (Fig. 1). The higher number of mononucleated and diploid cardiomyocytes in species capable of cardiac regeneration, such as newt [33], zebrafish [34] and rodent fetal and neonates [6, 35–37], suggest a higher proliferation capacity in such cells.
Cardiomyocyte cellular structure across species. Zebrafish and newt are mostly mononucleated and diploid [59, 154]; an organization that seems to favour a higher proliferative response to injury. Rodents show either mono- or bi-nucleated diploid cardiomyocytes [38]. Following stress or injury, mostly, these cardiomyocytes respond with hypertrophy; however, only those mononucleated cells appear to initiate proliferation. Human cardiomyocytes are mostly mononucleated and tetraploids. Limited data in young humans up to 20 years old, suggest that cardiomyocytes have some proliferative capacity [51, 52]. However, in later life, hypertrophy is the predominant response to injury in human
Indeed, Bersell et al. [38] demonstrated that only mononucleated cardiomyocytes that respond to the activation of the neuregulin 1/ErbB4 pathway after cardiac injury in mice initiate cardiomyocyte proliferation. Adult human cardiomyocytes are mostly mononucleated and tetraploid (4n), and adult mice cardiomyocytes are mainly binucleated and diploid (2n) [39] (Fig. 1). Indeed, several reports have shown a gradual decrease in the incorporation of radiolabeled thymidine soon after birth, coinciding with the formation of binucleated cardiomyocytes in mice [35, 40]. This process is associated with an increase in myofibril density and the formation of mature intercalated discs [41].
Fetal to adulthood temporal transition represents the divide between cardiac regenerating and non-regenerating species and it is, therefore, a key stage in which to study differences in cell cycle exit between species.
Adulthood
During the twentieth century, it was believed that the heart is a post-mitotic organ and cardiac growth in the adult was attributed exclusively to cardiomyocyte hypertrophy [42–45]. However, since the 1990s, evidence of cardiomyocyte proliferation in adult human hearts has been gradually accepted [46–48]. Quaini and co-workers demonstrated the presence of proliferating cell nuclear antigen, a marker of the G1–S cell cycle phases, in adult human hearts with ischemic and dilated cardiomyopathy. Evidence of metaphasic chromosomes together with cytokinesis was demonstrated in normal myocardium and in ischemic and dilated cardiomyopathy and in myocardial infarction [46, 47]. Considerable disagreement remains on the frequency of these cellular events in adult normal and diseased adult myocardium [49]; however, there appears to be a clear cardiac cellular response to disease and injury [39, 50]. The measured rate of cell division is influenced by the methods used to detect DNA synthesis and count cardiomyocytes [35, 50]. Radiocarbon birth dating has shown that cardiomyocyte turnover is approximately 1% over 20–40-year-old hearts [51]. A more recent study using new confocal imaging and optical dissector method suggested that cardiomyocyte division contributes to heart growth in young humans up to 20 years of age, with a cardiomyocyte turnover of approximately 1.9% [52]. Some reports have shown that adult rat cardiomyocytes can proliferate in culture after treatment with FGF1 concomitant with p38MAP kinase inhibition [25]. Indeed, normal adult cardiomyocytes rarely appear to re-enter the cell cycle and proliferate [41], and terminally differentiated mammalian cardiomyocytes show a predominantly hypertrophic response to mitogenic stimuli [53, 54]. One significant difference between embryonic and adult heart is the expression of cell cycle promoters, such as CDKs, cyclins, and proto-oncogenes, markedly expressed in embryonic hearts, corresponding to high cardiomyocyte cell cycle activity, while their expression is lower in the adult heart. In general, negative cell cycle regulatory genes, such as CDK inhibitors, are upregulated in adult hearts, where cardiomyocyte cell cycle activity is extremely low [41].
There is now a well-recognized link between mammalian cardiomyocyte hypertrophy and proliferation which involves a complex series of interconnected signaling pathways, including JAK, PLC, JNK, ERK, calcineurin, STAT, RAS, and MEF2 [5]. In addition to an increase in cellular RNA and protein, hypertrophy results in transcriptional reprogramming that closely resembles the fetal gene program that is known to drive hyperplasia in the developing fetus [55, 56]. It is hypothesized that cardiomyocyte hypertrophy, without hyperplasia, in mammals might be the result of a fundamental block in karyokinesis and cytokinesis by which the adult cardiomyocyte is unable to disassemble sarcomeres, uncouple from neighbouring cells and divide [57].
Mammals and zebrafish
Compared to mammals, cardiomyocytes from lower vertebrates, including teleost fish and salamander, show high proliferative capacity in adulthood [58]. The molecular mechanisms underlying this trait are not well understood. Better understanding could potentially provide important therapeutic targets for a range of cardiac disorders where cardiomyocyte loss plays a major role. A relevant difference between mammalians and zebrafish is that, in the latter, cardiomyocytes do not undergo cytokinetic mitosis. In fact, the majority of cardiomyocytes in adult zebrafish continue to have a single nucleus and a diploid genome (2n), similar to that observed in fetal mammalian hearts (vide supra) and associated with significant proliferative ability [59]. There is a common set of genes that drive growth and development of the embryonic heart in mammals [60] and fish [61]. A similar gene program is activated in the adult mammalian heart following injury or haemodynamic stress [5, 62, 63]. Many of these genes also appear to be activated in response to resection of the ventricle apex in the zebrafish [8, 64]. While this process has been studied in a variety of mammalian animal models, less is known about the related processes in the adult human heart.
Becker and colleagues [65] created and analysed a transgenic zebrafish embryo carrying a mutation in the troponin (tnnt2) gene associated with hypertrophic cardiomyopathy (HCM) in humans. An important finding in this model was that although the mutant gene resulted in abnormal sarcomeric organization, similar to that observed in humans and mouse models with HCM, the zebrafish exhibited a hyperplastic rather than a hypertrophic phenotype. Thus, the response of the zebrafish to stress or injury appears to be, primarily, a hyperplastic response, in contrast to mammals where the response is almost exclusively one of hypertrophy.
The heart’s response to injury: hypertrophy versus hyperplasia
In mammals
A variety of mechanisms are present in all organisms for dealing with tissue damage resulting from injury or disease. Mammals can regenerate liver, pancreas and skin, and can partially repair injury of peripheral nerves or skeletal muscle, but retain poor regenerative ability of other organs [66].
While the adult mammalian heart is considered a terminally differentiated organ, the fetal heart retains a remarkable proliferative capacity, capable of regenerating cardiomyocytes following injury [67, 68]. Porrello and colleagues [6] have shown that neonatal mouse cardiomyocytes can regenerate following surgical resection of the apex of the left ventricle. However, by day 7, the regenerative capacity is lost and replaced by a more classical fibrotic response resulting in scar tissue and impaired cardiac function. This finding has re-energized the search for mechanisms that underpin cell cycle arrest in the mammalian heart, with the intention of pursuing key molecular targets that might induce mature mammalian cardiomyocytes to re-enter the cell cycle.
Following transmural myocardial infarction (MI) in adult rats and human, the typical response to injury is inflammation and ventricular remodeling with a fibrotic scar forming at the site of MI [69, 70] (see Table 1). Reparative fibrosis appearing within and around the MI region is essential to safeguard the structural integrity of infarcted tissue, although interstitial fibrosis in non-infarcted myocardium alters tissue stiffness and can lead to ventricular dysfunction [71]. Following such injury, mammalian myocardium typically responds with cardiomyocyte hypertrophy combined with hyperplasia of non-cardiomyocyte cell populations in the heart including fibroblasts [5]. Cardiac hypertrophy, characterized by an increased cardiomyocyte size, enhanced protein synthesis and re-organization of the sarcomere [72], is acknowledged as an adaptive response intended to preserve cardiac function in the face of increasing haemodynamic stress. This physiological response normalizes wall tension and is necessary in the short term to allow the animal to adapt to the initial myocardial loss or haemodynamic load [5]. However, more prolonged stress converts this to a pathological process where a prolonged stimulus to cardiac hypertrophy leads to abnormal cellular responses within the myocardium typified by interstitial fibrosis, a switch to less-efficient myosin types and abnormal calcium handling [73]. These features result in so-called adverse remodeling of the ventricle and, in addition to contractile dysfunction, can lead to congestive cardiac failure and its fatal sequelae of progressive pump failure or sudden arrhythmic death. Apoptosis is also a feature of cardiac hypertrophy particularly when associated with congestive heart failure [74]. However, evidence has emerged that the adult mammalian heart does contain a small population of progenitor cells capable of differentiating into cardiomyocytes [75]. These cells might play a role in the replacement of lost cardiomyocytes [76–78], although they do not appear to be activated as part of a replacement process [59].
Nevertheless, the presence of active DNA synthesis [35], even if low, and the presence of putative progenitor cells within the hearts of adult mammals, gives rise to the possibility that proliferation of adult cardiomyocytes could be stimulated therapeutically as part of a process that could support repair and recovery. Why existing cardiac progenitor cell populations are unable to repair cardiac muscle in response to injury and, most importantly, how to trigger this untapped resource, are not yet clear.
Currently, many laboratories and institutions around the world are focused on the engraftment of various types of progenitor cells into infarcted hearts to achieve myocardial tissue renewal [79–83]. To date, most of these studies suggest that, while there may be small improvements in cardiac function, there is little evidence, in both human and mouse models, that such progenitors truly engraft and mature into active cardiomyocytes [84, 85]. There is evidence, however, that they may increase angiogenesis, leading many to believe that progenitor cells release a variety of growth factors that contribute to the response to injury by stimulating growth and repair of non-cardiomyocyte-derived cells [86–88].
The zebrafish
While mammals lose the ability to regenerate the heart within a few weeks after birth, lower vertebrates, such as amphibians and fish, retain this ability to regenerate the heart, and, indeed, most of their organs, following significant injury or loss of tissue well into adulthood (see table). In the zebrafish, resection of up to 20% of the ventricle apex results in complete regeneration and repair of the ventricle within 60 days [89]. Zebrafish heart regeneration proceeds through injury-induced proliferation of cardiomyocytes which retain their capacity to divide and proliferate postnatally [33, 89–91]. BrdU labelling studies have shown BrdU-positive cardiomyocytes along the leading edge of the regenerating heart. Lepilina and co-workers [92] suggested that regenerating myocardium arises and matures from undifferentiated cardiomyocyte progenitor cells of epicardial origin. In contrast, two more recent genetic fate-mapping studies [7, 11] unambiguously demonstrated that pre-existing committed cardiomyocytes are, in fact, the main source of the cells contributing to cardiac regeneration in the zebrafish.
Thus, although cellular mechanisms involved in cardiac regeneration have recently been unraveled, the molecular pathways that might be involved in initiating and maintaining the cardiomyocyte response to injury remain uncertain. Further studies are required to clarify these mechanisms that induce a hyperplastic, and not a hypertrophic, response in this setting. Genetic similarity between humans and zebrafish [93, 94] support the notion that cardiac regenerative pathways can be dissected in the fish model.
Zebrafish embryo as a model of cardiomyocyte proliferation
The zebrafish heart is emerging as an increasingly flexible model to study many developmental, genetic and acquired cardiac disorders [95]. Zebrafish heart development is well characterized [9, 96–98] adding to numerous early publications on this topic [99, 100]. Despite having only two chambers, the fish heart retains many of the structural and developmental components of the mammalian heart [101], including a three-layer ventricular wall (epicardium, myocardium and endocardium) from three days post fertilization [102, 103]. The early mammalian heart is derived from first and second heart fields [102, 103], although there is evidence that a population of lateral mesoderm-derived haemangioblasts represent an evolutionary antecedent of the second heart field in zebrafish [104].
The zebrafish embryonic heart is particularly suited to studying aspects of cardiomyocyte proliferation for a number of important reasons. First, the embryonic heart in the early stages is composed of only a few hundred cardiomyocytes [105, 106], and this allows accurate and reproducible approaches to count total cardiomyocyte number in either chamber or both. In addition, transgenic technology using fluorescent proteins [107, 108] can be targeted to developing cardiomyocytes, which allows the ready visualization of in vivo development and function of these cells [109].
The small size of the zebrafish embryo permits exchange of gases by passive diffusion, allowing their survival and relatively normal development for several days even in the absence of a functioning heart and circulation [110]. This permits the phenotypic analysis of embryos with severe or lethal cardiovascular mutations and defects [111, 112] which would otherwise be extremely difficult to assess in higher vertebrates where they would be highly likely to die in utero [13].
There are no well-described models of myocardial infarction or cardiac hypertrophy in lower vertebrates akin to those developed and exploited over many years in small mammals [113]. To circumvent this problem, researchers have developed more radical approaches of resecting a piece of the ventricle. This was first reported in salamanders [114] and then in the zebrafish, where approximately 20% of the ventricle apex can be resected, and regrowth will occur within 60 days [89]. This model has now been reproduced in many laboratories around the world and has become a standard approach to studying the molecular and cellular mechanisms associated with cardiomyocyte proliferation leading to what has come to be known as cardiac regeneration. However, this resection model is only feasible in a low-pressure heart such as that found in lower vertebrate species and in the early postnatal time period in mice. In a high-pressure haemodynamic system, typical of adult mammalian hearts, acute bleeding from the resection margin results in rapid death. Not only does this indicate that the injury response to heart resection is different in zebrafish due to the underlying physiology, but it also poses the question of whether repair of a resected piece of heart by cardiomyocyte proliferation is likely to convey an evolutionary advantage for mammals since it would most likely result in exsanguination from the heart itself.
Arguably, therefore, the best option for a mammalian heart, working at higher physiological pressures, and faced with a large zone of injury is to generate an area of scar tissue as quickly as possible in order to heal the infarcted territory and avoid the risk of myocardial rupture and bleeding that would ensue [115]. In addition, since this ventricle apex resection is not as physiologically or clinically relevant as a model [116], others have used cryoinjury in which liquid nitrogen is used to injure a localised region of ventricle in a regional manner similar to myocardial infarction [117, 118]. Following this cryoinjury, the adult zebrafish heart demonstrates features consistent with regeneration including cardiomyocyte proliferation, but in this case, there is also a more readily observed area of scar which requires up to 3 months for complete repair.
Mechanisms controlling organ and tissue regeneration are conserved between larvae and adults at the cellular and molecular levels, suggesting that the regenerative machinery directing cell proliferation in response to injury may exist from early developmental stages [119]. Indeed, amputation of either the adult or embryo zebrafish fin, a widely used model of tissue regeneration, induces proliferation of progenitor cells from the amputation margin [120]. In the same model, at molecular level, transcription factors and components of various signaling pathways normally upregulated during adult fin regeneration [121] are also increased during larval regeneration [119–121].
We have recently developed and validated a model of heart injury and recovery in the zebrafish embryo using highly targeted laser injury [122] (supplementary movie). The use of embryos significantly shortens the injury response and subsequent repair time period. We have demonstrated a striking ability of the zebrafish embryonic heart to regenerate and recover cardiac function by 24 h post-laser injury. Using this approach, the zebrafish embryo can be used in a high-throughput model system to study human disease. For example, figure S1 clearly shows a protocol to study heart response to injury that can be completed in an experiment over a period of only 4–5 days. Embryos can be injected at 1–2 cell stage with one or more compounds, such as labelled mRNA, for overexpressing genes, or CRISPR/Cas 9 for gene knockout or morpholino oligonucleotides for gene knockdown (figure S1A). Otherwise, embryos can be exposed to small molecules and pharmacological compounds of interest by simply adding these to the medium at the desired developmental stage (figure S1B). Embryos can then be injured, for example using a targeted laser at 72 hpf as described, followed by collection of data of cardiac function, including ejection fraction and tail blood flow (figure S1C). Using transgenic lines expressing fluorochrome driven by cardiac markers, such as tg(myl:gfp)y1 in figure S1D, hundreds of embryonic hearts can be isolated in a few minutes, for immunohistochemistry, gene and protein analysis (Figure S1E). The laser injury technique also lends itself to a high-throughput approach; it is possible to create a standardised level of heart injury to the ventricle in approximately 50 embryos per hour. We have also applied the laser-induced injury to different cardiac regions and structures in the zebrafish larva, including bulbus arteriosus and atrioventricular valve and have demonstrated the potential of the zebrafish to be used for various models of cardiac disease [123]. Although the overlying tissue, the pericardium in this case, is also partially injured, this is proportionately less compared to existing surgical approaches (apical resection, cryoinjury) in adults requiring damaging tissue layers covering the heart, making our model more myocardial-specific.
Using the laser-induced injury model, we have explored a number of molecular pathways that could enhance recovery from myocardial injury. In particular, we explored the relationships between cyclin-dependent kinase (CDK)9, La-related protein (LARP)7 and the positive transcription elongation factor (P-TEF)b complex of which they are molecular partners. CDK9 has been implicated in mammal cardiac hypertrophy [124]. In our experiments, we first downregulated CDK9 and or LARP7 by pharmacological or morpholino knockdown and found that they had mainly opposite effects, to CDK9 strongly reducing its action and LARP7 slightly increasing cardiomyocyte proliferation [125]. In separate experiments, we found that prior CDK9 downregulation impaired the cardiac recovery from laser injury, whereas LARP7 knockdown did not. Interestingly, co-injection of LARP7 and CDK9-targeted morpholinos rescued the CDK9 phenotype, both in terms of cardiomyocyte proliferation and cardiac function. We concluded that LARP7 acts to maintain CDK9 in an inactive state in the P-TEFb complex and that suppressing LARP7 activity results in a derepression of CDK9 that ultimately leads to a more active P-TEFb complex. Studying the repair process following injury during cardiogenesis and development clearly raises some questions of relevance of these experiments to the mammalian heart. However, compared to the adult, the zebrafish larva not only offers the advantage to study hundreds of animals over a short time period, but it also allows the possibility to study severe developmental cardiac defects which would otherwise be extremely challenging to study in mammalian models. High-throughput drug screening programs, for example, have been successfully used in zebrafish to explore novel therapeutic candidates before moving forward to more expensive mammalian model systems [126–128].
Molecular pathways linked to cardiomyocyte proliferation as targets for drug development
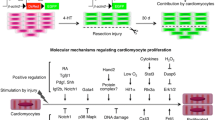
Several molecular pathways are under investigation for their potential ability to inhibit or activate cardiomyocyte growth and or proliferation and could be harnessed to develop heart disease therapies (Fig. 2). In some cases, the role of these pathways in cardiomyocyte proliferation has been unraveled in zebrafish. For example, a balance between CDK9 and its repressors, including LA-Related Protein 7, can switch on or off cardiomyocyte proliferation in zebrafish [125]. However, CDK9 knockdown affects somatic growth and development of a number of key embryonic structures including the brain, heart, eye and blood vessels [129]. For therapeutic strategies, this raised the question to develop tissue- or cell-specific anti-CDK9 drugs. The activity of the P-TEFb complex, a key driver for transcription formed by CDK9 and cyclin T, is increased in cardiac hypertrophy in mammals [124]. In addition, the CDK9 activity is derepressed by the dissociation of 7SK small nuclear RNA [130, 131] and HEXIM1 [132, 133], two CDK9 inhibitors. HEXIM1 knockout mice die during fetal development and exhibit all the genetic and physical hallmarks of cardiac hypertrophy [134]. CDK9 has also been shown to regulate cell cycle and is involved in cardiomyocytes differentiation from mice embryonic stem cells [135]. Mahmoud and colleagues [29] showed that in neonatal and adult cardiomyocytes, Meis1 deficiency increases cardiomyocyte numbers, whereas Meis1 overexpression activates CDK inhibitors INK4b-ARFINK4a and Cdkn1a genes, which leads to cell cycle arrest because of inhibition of CDKs. miRNAs are also potential targets for cardiomyocyte proliferation therapies (Fig. 2) [136]. As miRNAs generally repress gene expression by promoting mRNA degradation and/or by inhibiting translation, the final effects on cardiomyocte proliferation are mediated by inhibiting or activating the cell cycle [137, 138]. For example, miR-590 and miR-17/92 clusters promote cardiomyocyte proliferation by inhibiting the proliferation repressors Homer protein homolog 1 (Homer1) and Homeodomain-only protein x (Hopx), whereas miR-15 family represses cardiomyocyte proliferation by inhibiting the proliferative activator Checkpoint kinase 1 (Check1). Furthermore, the muscle-specific microRNA-1 (miR-1), which normally keeps CDK9 derepressed at the transcriptional level [139], was downregulated at a very early stage, following cardiac hypertrophy induced in a mouse model of aortic constriction-induced hypertrophy.
Also, the Hippo/Yap pathway plays an essential role in the regulation of heart development and postnatal cardiomyocyte proliferation [140], highlighting the potential for enhancing cardiac regeneration (Fig. 2) [141]. Indeed, modulation of this pathway in the neonatal heart has been shown to prolong the regenerative window. However, Hippo/Yap also appears to influence cardiomyocyte autophagy and apoptosis [142]. Among the mitogens, IGF2 has been shown to activate cardiomyocyte proliferation and is required for zebrafish heart regeneration [143], whereas TGFβ/activin signaling plays important roles in cardiomyocyte proliferation and scar formation [144].
Modulation of inflammation may provide a key therapeutic strategy to drive heart regeneration. Indeed, the types of macrophage present in the regenerating neonatal mouse heart may provide essential stimuli for angiogenesis and regeneration [145].
Hypoxia, redox signaling and metabolic phenotypes are also key regulators of cardiomyocyte proliferation and cardiac renewal [146]. Puente et al. [147] have recently shown that cell cycle arrest in postnatally terminally differentiated cardiomyocytes is triggered by mitochondrial reactive oxygen species-mediated oxidative DNA damage. This suggests that ROS may play key roles in cell cycle regulation and differentiation during cardiac development. In turn, this suggests that cells responsible for cellular turnover in the heart, such as immature and/or mature myocytes or progenitor population may need an environment with a lower oxygen concentration to proliferate efficiently. The heart may be unique in the nature of this oxygen-sensitive response. On the one hand, it has high O2 consumption [148], and on the other hand, its cardiac progenitor cells appear to benefit from hypoxic preconditioning which improves survival and homing of engrafted cells into an infarcted territory [149]. Indeed, the epicardium and subepicardium regions contain multipotent progenitor cells [150] which could represent a novel hypoxic niche of the heart. These cells express Hif1α and respond to hypoxia by increasing cell proliferation and, thus, provide a source of new cardiac cells following injury including fibroblasts, perivascular smooth muscle cells [151] and cardiomyocytes after thymosin β4 activation [144, 145, 152]. A better understanding of these and other molecular mechanisms would allow us to develop exciting new strategies to improve cardiomyocyte proliferation and cardiac regeneration.
Future perspectives
The question remains as to how drugs or therapeutic stategies could be used to support cardiac repair in disease conditions where the heart was failing or at risk of failing following injury, stress or cell loss. Clearly, a key time for treatment would be around the time of an acute myocardial infarction where many previous studies of bone-marrow-derived stem cells have been extensively tested over the last 10 years [153] with overall, relatively disappointing results.
The possibility of directly harnessing cellular mechanisms to achieve therapeutic benefit in patients with both heart disease remains elusive. To date, the possibility of providing therapies targeted at enhancing cardiomyocyte proliferation that could either be administered acutely, at the time of acute injury such as infarction, or in the long term to patients with chronic left ventricular systolic dysfunction remains an important but elusive target. The key issue would be whether such drugs could be administered orally or intravenously and whether their benefits outweighed the risk of harm. At this stage, we are a long way from knowing the answers to these questions since no viable candidate drug has yet been developed despite a number of interesting pathways currently undergoing further studies. Any such drug with the capacity to reactivate a highly differentiated cardiomyocyte into a more active proliferative state is likely to carry a significant risk of inducing unregulated cell growth either in the target tissue or in other exposed tissues. The risk of neoplasia is, therefore, clear unless the drug can be highly targeted to a specific cardiac pathway or delivered in a highly localised manner directly to the heart.
References
Go AS, Mozaffarian D, Roger VL, Benjamin EJ et al (2013) Heart disease and stroke statistics–2013 update: a report from the American Heart Association. Circulation 127(1):e6–e245
Berenji K, Drazner MH, Rothermel BA, Hill JA (2005) Does load-induced ventricular hypertrophy progress to systolic heart failure? Am J Physiol Heart Circ Physiol 289(1):H8–H16
Haider AW, Larson MG, Benjamin EJ, Levy D (1998) Increased left ventricular mass and hypertrophy are associated with increased risk for sudden death. J Am Coll Cardiol 32(5):1454–1459
Lips DJ, deWindt LJ, van Kraaij DJ, Doevendans PA (2003) Molecular determinants of myocardial hypertrophy and failure: alternative pathways for beneficial and maladaptive hypertrophy. Eur Heart J 24(10):883–896
Heineke J, Molkentin JD (2006) Regulation of cardiac hypertrophy by intracellular signalling pathways. Nat Rev Mol Cell Biol 7(8):589–600
Porrello ER, Mahmoud AI, Simpson E, Hill JA et al (2011) Transient regenerative potential of the neonatal mouse heart. Science 331(6020):1078–1080
Jopling C, Sleep E, Raya M, Marti M et al (2010) Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 464(7288):606–609
Mercola M, Ruiz-Lozano P, Schneider MD (2011) Cardiac muscle regeneration: lessons from development. Genes Dev 25(4):299–309
Staudt D, Stainier D (2012) Uncovering the molecular and cellular mechanisms of heart development using the zebrafish. Annu Rev Genet 46:397–418
Jensen B, Wang T, Christoffels VM, Moorman AF (2013) Evolution and development of the building plan of the vertebrate heart. Biochim Biophys Acta 1833(4):783–794
Kikuchi K, Holdway JE, Werdich AA, Anderson RM et al (2010) Primary contribution to zebrafish heart regeneration by gata4(+) cardiomyocytes. Nature 464(7288):601–605
Burggren WW, Pinder AW (1991) Ontogeny of cardiovascular and respiratory physiology in lower vertebrates. Annu Rev Physiol 53:107–135
Stainier DY (2001) Zebrafish genetics and vertebrate heart formation. Nat Rev Genet 2(1):39–48
Chen H, Zhang W, Li D, Cordes TM et al (2009) Analysis of ventricular hypertrabeculation and noncompaction using genetically engineered mouse models. Pediatr Cardiol 30(5):626–634
Pennisi DJ, Ballard VL, Mikawa T (2003) Epicardium is required for the full rate of myocyte proliferation and levels of expression of myocyte mitogenic factors FGF2 and its receptor, FGFR-1, but not for transmural myocardial patterning in the embryonic chick heart. Dev Dyn 228(2):161–172
Guadix JA, Ruiz-Villalba A, Lettice L, Velecela V et al (2011) Wt1 controls retinoic acid signalling in embryonic epicardium through transcriptional activation of Raldh2. Development 138(6):1093–1097
Zhao YY, Sawyer DR, Baliga RR, Opel DJ et al (1998) Neuregulins promote survival and growth of cardiac myocytes. Persistence of ErbB2 and ErbB4 expression in neonatal and adult ventricular myocytes. J Biol Chem 273(17):10261–10269
Christoffels VM, Habets PE, Franco D, Campione M et al (2000) Chamber formation and morphogenesis in the developing mammalian heart. Dev Biol 223(2):266–278
Hove JR, Koster RW, Forouhar AS, Acevedo-Bolton G et al (2003) Intracardiac fluid forces are an essential epigenetic factor for embryonic cardiogenesis. Nature 421(6919):172–177
Toyoda M, Shirato H, Nakajima K, Kojima M et al (2003) jumonji downregulates cardiac cell proliferation by repressing cyclin D1 expression. Dev Cell 5(1):85–97
Shirato H, Ogawa S, Nakajima K, Inagawa M et al (2009) A jumonji (Jarid2) protein complex represses cyclin D1 expression by methylation of histone H3-K9. J Biol Chem 284(2):733–739
Takeuchi T, Kojima M, Nakajima K, Kondo S (1999) jumonji gene is essential for the neurulation and cardiac development of mouse embryos with a C3H/He background. Mech Dev 86(1–2):29–38
Bicknell KA, Coxon CH, Brooks G (2007) Can the cardiomyocyte cell cycle be reprogrammed? J Mol Cell Cardiol 42(4):706–721
Busk PK, Hinrichsen R (2003) Cyclin D in left ventricle hypertrophy. Cell Cycle 2(2):91–95
Engel FB (2005) Cardiomyocyte proliferation: a platform for mammalian cardiac repair. Cell Cycle 4(10):1360–1363
Poolman RA, Brooks G (1998) Expressions and activities of cell cycle regulatory molecules during the transition from myocyte hyperplasia to hypertrophy. J Mol Cell Cardiol 30(10):2121–2135
Ikenishi A, Okayama H, Iwamoto N, Yoshitome S et al (2012) Cell cycle regulation in mouse heart during embryonic and postnatal stages. Dev Growth Differ 54(8):731–738
Tane S, Ikenishi A, Okayama H, Iwamoto N et al (2014) CDK inhibitors, p21(Cip1) and p27(Kip1), participate in cell cycle exit of mammalian cardiomyocytes. Biochem Biophys Res Commun 443(3):1105–1109
Mahmoud AI, Kocabas F, Muralidhar SA, Kimura W et al (2013) Meis1 regulates postnatal cardiomyocyte cell cycle arrest. Nature 497(7448):249–253
Neufeld TP, Edgar BA (1998) Connections between growth and the cell cycle. Curr Opin Cell Biol 10(6):784–790
Li F, Wang X, Capasso JM, Gerdes AM (1996) Rapid transition of cardiac myocytes from hyperplasia to hypertrophy during postnatal development. J Mol Cell Cardiol 28(8):1737–1746
Li JM, Poolman RA, Brooks G (1998) Role of G1 phase cyclins and cyclin-dependent kinases during cardiomyocyte hypertrophic growth in rats. Am J Physiol 275(3 Pt 2):H814–H822
Matz DG, Oberpriller JO, Oberpriller JC (1998) Comparison of mitosis in binucleated and mononucleated newt cardiac myocytes. Anat Rec 251(2):245–255
Kikuchi K, Poss KD (2012) Cardiac regenerative capacity and mechanisms. Annu Rev Cell Dev Biol 28:719–741
Soonpaa MH, Kim KK, Pajak L, Franklin M et al (1996) Cardiomyocyte DNA synthesis and binucleation during murine development. Am J Physiol 271(5 Pt 2):H2183–H2189
Li F, Wang X, Bunger PC, Gerdes AM (1997) Formation of binucleated cardiac myocytes in rat heart: I. Role of actin-myosin contractile ring. J Mol Cell Cardiol 29(6):1541–1551
Poss KD (2007) Getting to the heart of regeneration in zebrafish. Semin Cell Dev Biol 18(1):36–45
Bersell K, Arab S, Haring B, Kuhn B (2009) Neuregulin1/ErbB4 signaling induces cardiomyocyte proliferation and repair of heart injury. Cell 138(2):257–270
Laflamme MA, Murry CE (2011) Heart regeneration. Nature 473(7347):326–335
Clubb FJ Jr, Bishop SP (1984) Formation of binucleated myocardial cells in the neonatal rat. An index for growth hypertrophy. Lab Invest 50(5):571–577
Pasumarthi KB, Field LJ (2002) Cardiomyocyte cell cycle regulation. Circ Res 90(10):1044–1054
Karsner HT, Saphir O, Todd TW (1925) The State of the cardiac muscle in hypertrophy and atrophy. Am J Pathol 1(4):351–372
Linzbach AJ (1960) Heart failure from the point of view of quantitative anatomy. Am J Cardiol 5:370–382
Macmahon HE (1937) Hyperplasia and regeneration of the myocardium in infants and in children. Am J Pathol 13(5):845–854
Zak R (1974) Development and proliferative capacity of cardiac muscle cells. Circ Res 35(2):17–26
Beltrami AP, Urbanek K, Kajstura J, Yan SM et al (2001) Evidence that human cardiac myocytes divide after myocardial infarction. N Engl J Med 344(23):1750–1757
Kajstura J, Zhang X, Reiss K, Szoke E et al (1994) Myocyte cellular hyperplasia and myocyte cellular hypertrophy contribute to chronic ventricular remodeling in coronary artery narrowing-induced cardiomyopathy in rats. Circ Res 74(3):383–400
Quaini F, Cigola E, Lagrasta C, Saccani G et al (1994) End-stage cardiac failure in humans is coupled with the induction of proliferating cell nuclear antigen and nuclear mitotic division in ventricular myocytes. Circ Res 75(6):1050–1063
Soonpaa MH, Field LJ (1998) Survey of studies examining mammalian cardiomyocyte DNA synthesis. Circ Res 83(1):15–26
Soonpaa MH, Field LJ (1997) Assessment of cardiomyocyte DNA synthesis in normal and injured adult mouse hearts. Am J Physiol 272(1 Pt 2):H220–H226
Bergmann O, Bhardwaj RD, Bernard S, Zdunek S et al (2009) Evidence for cardiomyocyte renewal in humans. Science 324(5923):98–102
Mollova M, Bersell K, Walsh S, Savla J et al (2013) Cardiomyocyte proliferation contributes to heart growth in young humans. Proc Natl Acad Sci USA 110(4):1446–1451
Bicknell KA, Surry EL, Brooks G (2003) Targeting the cell cycle machinery for the treatment of cardiovascular disease. J Pharm Pharmacol 55(5):571–591
Capasso JM, Bruno S, Cheng W, Li P et al (1992) Ventricular loading is coupled with DNA synthesis in adult cardiac myocytes after acute and chronic myocardial infarction in rats. Circ Res 71(6):1379–1389
Olson EN (2004) A decade of discoveries in cardiac biology. Nat Med 10(5):467–474
Olson EN, Schneider MD (2003) Sizing up the heart: development redux in disease. Genes Dev 17(16):1937–1956
Ahuja P, Sdek P, MacLellan WR (2007) Cardiac myocyte cell cycle control in development, disease, and regeneration. Physiol Rev 87(2):521–544
Martin-Puig S, Fuster V, Torres M (2012) Heart repair: from natural mechanisms of cardiomyocyte production to the design of new cardiac therapies. Ann N Y Acad Sci 1254:71–81
Wills AA, Holdway JE, Major RJ, Poss KD (2008) Regulated addition of new myocardial and epicardial cells fosters homeostatic cardiac growth and maintenance in adult zebrafish. Development 135(1):183–192
Xu H, Baldini A (2007) Genetic pathways to mammalian heart development: recent progress from manipulation of the mouse genome. Semin Cell Dev Biol 18(1):77–83
Brand T (2003) Heart development: molecular insights into cardiac specification and early morphogenesis. Dev Biol 258(1):1–19
Taegtmeyer H, Sen S, Vela D (2010) Return to the fetal gene program: a suggested metabolic link to gene expression in the heart. Ann N Y Acad Sci 1188:191–198
Alexander JM, Bruneau BG (2010) Lessons for cardiac regeneration and repair through development. Trends Mol Med 16(9):426–434
Lien CL, Harrison MR, Tuan TL, Starnes VA (2012) Heart repair and regeneration: recent insights from zebrafish studies. Wound Repair Regen 20(5):638–646
Becker JR, Deo RC, Werdich AA, Panakova D et al (2011) Human cardiomyopathy mutations induce myocyte hyperplasia and activate hypertrophic pathways during cardiogenesis in zebrafish. Dis Model Mech 4(3):400–410
Alcon A, Cagavi Bozkulak E, Qyang Y (2012) Regenerating functional heart tissue for myocardial repair. Cell Mol Life Sci 69(16):2635–2656
Allukian M 3rd, Xu J, Morris M, Caskey R et al (2013) Mammalian cardiac regeneration after fetal myocardial infarction requires cardiac progenitor cell recruitment. Ann Thorac Surg 96(1):163–170
Drenckhahn JD, Schwarz QP, Gray S, Laskowski A et al (2008) Compensatory growth of healthy cardiac cells in the presence of diseased cells restores tissue homeostasis during heart development. Dev Cell 15(4):521–533
Anversa P, Li P, Zhang X, Olivetti G et al (1993) Ischaemic myocardial injury and ventricular remodelling. Cardiovasc Res 27(2):145–157
Weber KT, Pick R, Jalil JE, Janicki JS et al (1989) Patterns of myocardial fibrosis. J Mol Cell Cardiol 21(Suppl 5):121–131
Sun Y, Weber KT (2000) Infarct scar: a dynamic tissue. Cardiovasc Res 46(2):250–256
Frey N, Olson EN (2003) Cardiac hypertrophy: the good, the bad, and the ugly. Annu Rev Physiol 65:45–79
Aoki H, Sadoshima J, Izumo S (2000) Myosin light chain kinase mediates sarcomere organization during cardiac hypertrophy in vitro. Nat Med 6(2):183–188
Sabbah HN (2000) Apoptotic cell death in heart failure. Cardiovasc Res 45(3):704–712
Rubart M, Field LJ (2006) Cardiac regeneration: repopulating the heart. Annu Rev Physiol 68:29–49
Beltrami AP, Barlucchi L, Torella D, Baker M et al (2003) Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 114(6):763–776
Laugwitz KL, Moretti A, Lam J, Gruber P et al (2005) Postnatal isl1 + cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 433(7026):647–653
Moretti A, Caron L, Nakano A, Lam JT et al (2006) Multipotent embryonic isl1 + progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127(6):1151–1165
Buccini S, Haider KH, Ahmed RP, Jiang S et al (2012) Cardiac progenitors derived from reprogrammed mesenchymal stem cells contribute to angiomyogenic repair of the infarcted heart. Basic Res Cardiol 107(6):301
Chen CH, Wang SS, Wei EI, Chu TY et al (2013) Hyaluronan enhances bone marrow cell therapy for myocardial repair after infarction. Mol Ther 21(3):670–679
Dragneva G, Korpisalo P, Yla-Herttuala S (2013) Promoting blood vessel growth in ischemic diseases: challenges in translating preclinical potential into clinical success. Dis Model Mech 6(2):312–322
Kearns-Jonker M, Dai W, Gunthart M, Fuentes T et al (2012) Genetically engineered mesenchymal stem cells influence gene expression in donor cardiomyocytes and the recipient heart. J Stem Cell Res Ther Suppl 1(00):5
Menasche P (2011) Stem cell therapy for chronic heart failure: lessons from a 15-year experience. C R Biol 334(7):489–496
Povsic TJ, O’Connor CM (2010) Cell therapy for heart failure: the need for a new therapeutic strategy. Expert Rev Cardiovasc Ther 8(8):1107–1126
Nussbaum J, Minami E, Laflamme MA, Virag JA et al (2007) Transplantation of undifferentiated murine embryonic stem cells in the heart: teratoma formation and immune response. FASEB J 21(7):1345–1357
Foubert P, Matrone G, Souttou B, Lere-Dean C et al (2008) Coadministration of endothelial and smooth muscle progenitor cells enhances the efficiency of proangiogenic cell-based therapy. Circ Res 103(7):751–760
Iwasaki H, Kawamoto A, Tjwa M, Horii M et al (2011) PlGF repairs myocardial ischemia through mechanisms of angiogenesis, cardioprotection and recruitment of myo-angiogenic competent marrow progenitors. PLoS One 6(9):e24872
Wang H, Zhang X, Li Y, Ma Y et al (2010) Improved myocardial performance in infarcted rat heart by co-injection of basic fibroblast growth factor with temperature-responsive chitosan hydrogel. J Heart Lung Transplant 29(8):881–887
Poss KD, Wilson LG, Keating MT (2002) Heart regeneration in zebrafish. Science 298(5601):2188–2190
Nag AC, Healy CJ, Cheng M (1979) DNA synthesis and mitosis in adult amphibian cardiac muscle cells in vitro. Science 205(4412):1281–1282
Raya A, Koth CM, Buscher D, Kawakami Y et al (2003) Activation of Notch signaling pathway precedes heart regeneration in zebrafish. Proc Natl Acad Sci USA 100(Suppl 1):11889–11895
Lepilina A, Coon AN, Kikuchi K, Holdway JE et al (2006) A dynamic epicardial injury response supports progenitor cell activity during zebrafish heart regeneration. Cell 127(3):607–619
Howe K, Clark MD, Torroja CF, Torrance J et al (2013) The zebrafish reference genome sequence and its relationship to the human genome. Nature 496(7446):498–503
Kettleborough RN, Busch-Nentwich EM, Harvey SA, Dooley CM et al (2013) A systematic genome-wide analysis of zebrafish protein-coding gene function. Nature 496(7446):494–497
Bakkers J (2011) Zebrafish as a model to study cardiac development and human cardiac disease. Cardiovasc Res 91(2):279–288
Liu J, Stainier DY (2012) Zebrafish in the study of early cardiac development. Circ Res 110(6):870–874
Tu S, Chi NC (2012) Zebrafish models in cardiac development and congenital heart birth defects. Differentiation 84(1):4–16
Yelon D, Weinstein BM, Fishman MC (2002) Cardiovascular system. Results Probl Cell Differ 40:298–321
Stainier DY, Fishman MC (1992) Patterning the zebrafish heart tube: acquisition of anteroposterior polarity. Dev Biol 153(1):91–101
Stainier DY, Lee RK, Fishman MC (1993) Cardiovascular development in the zebrafish. I. Myocardial fate map and heart tube formation. Development 119(1):31–40
Asnani A, Peterson RT (2014) The zebrafish as a tool to identify novel therapies for human cardiovascular disease. Dis Model Mech 7(7):763–767
Hami D, Grimes AC, Tsai HJ, Kirby ML (2011) Zebrafish cardiac development requires a conserved secondary heart field. Development 138(11):2389–2398
Guner-Ataman B, Paffett-Lugassy N, Adams MS, Nevis KR et al (2013) Zebrafish second heart field development relies on progenitor specification in anterior lateral plate mesoderm and nkx2.5 function. Development 140(6):1353–1363
Peterkin T, Gibson A, Patient R (2009) Common genetic control of haemangioblast and cardiac development in zebrafish. Development 136(9):1465–1474
Bostrom P, Mann N, Wu J, Quintero PA et al (2010) C/EBPbeta controls exercise-induced cardiac growth and protects against pathological cardiac remodeling. Cell 143(7):1072–1083
Targoff KL, Schell T, Yelon D (2008) Nkx genes regulate heart tube extension and exert differential effects on ventricular and atrial cell number. Dev Biol 322(2):314–321
Burns CG, Milan DJ, Grande EJ, Rottbauer W et al (2005) High-throughput assay for small molecules that modulate zebrafish embryonic heart rate. Nat Chem Biol 1(5):263–264
Detrich HW 3rd (2008) Fluorescent proteins in zebrafish cell and developmental biology. Methods Cell Biol 85:219–241
Choi WY, Gemberling M, Wang J, Holdway JE et al (2013) In vivo monitoring of cardiomyocyte proliferation to identify chemical modifiers of heart regeneration. Development 140(3):660–666
Chen JN, van Eeden FJ, Warren KS, Chin A et al (1997) Left-right pattern of cardiac BMP4 may drive asymmetry of the heart in zebrafish. Development 124(21):4373–4382
Isogai S, Horiguchi M, Weinstein BM (2001) The vascular anatomy of the developing zebrafish: an atlas of embryonic and early larval development. Dev Biol 230(2):278–301
Weaver J (2011) Genetic origins of birth defects revealed by new animal model. PLoS Biol 9(10):e1001180
Verdouw PD, van den Doel MA, de Zeeuw S, Duncker DJ (1998) Animal models in the study of myocardial ischaemia and ischaemic syndromes. Cardiovasc Res 39(1):121–135
Becker RO, Chapin S, Sherry R (1974) Regeneration of the ventricular myocardium in amphibians. Nature 248(5444):145–147
Nasir A, Gouda M, Khan A, Bose A (2014) Is it ever possible to treat left ventricular free wall rupture conservatively? Interact CardioVasc Thorac Surg 19(3):488–493
Abarbanell AM, Herrmann JL, Weil BR, Wang Y et al (2010) Animal models of myocardial and vascular injury. J Surg Res 162(2):239–249
Gonzalez-Rosa JM, Mercader N (2012) Cryoinjury as a myocardial infarction model for the study of cardiac regeneration in the zebrafish. Nat Protoc 7(4):782–788
Schnabel K, Wu CC, Kurth T, Weidinger G (2011) Regeneration of cryoinjury induced necrotic heart lesions in zebrafish is associated with epicardial activation and cardiomyocyte proliferation. PLoS ONE 6(4):e18503
Kawakami A, Fukazawa T, Takeda H (2004) Early fin primordia of zebrafish larvae regenerate by a similar growth control mechanism with adult regeneration. Dev Dyn 231(4):693–699
Rojas-Munoz A, Rajadhyksha S, Gilmour D, van Bebber F et al (2009) ErbB2 and ErbB3 regulate amputation-induced proliferation and migration during vertebrate regeneration. Dev Biol 327(1):177–190
Sanchez Alvarado A, Tsonis PA (2006) Bridging the regeneration gap: genetic insights from diverse animal models. Nat Rev Genet 7(11):873–884
Matrone G, Taylor JM, Wilson KS, Baily J et al (2013) Laser-targeted ablation of the zebrafish embryonic ventricle: a novel model of cardiac injury and repair. Int J Cardiol 168(4):3913–3919
Matrone G, Maqsood S, Taylor J, Mullins JJ et al (2014) Targeted laser ablation of the zebrafish larval heart induces models of heart block, valvular regurgitation, and outflow tract obstruction. Zebrafish 11(6):536–541
Sano M, Abdellatif M, Oh H, Xie M et al (2002) Activation and function of cyclin T-Cdk9 (positive transcription elongation factor-b) in cardiac muscle-cell hypertrophy. Nat Med 8(11):1310–1317
Matrone G, Wilson KS, Maqsood S, Mullins JJ et al (2015) CDK9 and its repressor LARP7 modulate cardiomyocyte proliferation and response to injury in the zebrafish heart. J Cell Sci 128(24):4560–4571
Letamendia A, Quevedo C, Ibarbia I, Virto JM et al (2012) Development and validation of an automated high-throughput system for zebrafish in vivo screenings. PLoS One 7(5):e36690
Evensen L, Link W, Lorens JB (2010) Imaged-based high-throughput screening for anti-angiogenic drug discovery. Curr Pharm Des 16(35):3958–3963
Gabor KA, Goody MF, Mowel WK, Breitbach ME et al (2014) Influenza A virus infection in zebrafish recapitulates mammalian infection and sensitivity to anti-influenza drug treatment. Dis Model Mech 7(11):1227–1237
Matrone G, Mullins JJ, Tucker CS, Denvir MA (2016) Effects of cyclin dependent kinase 9 inhibition on zebrafish larvae. Cell Cycle 0
Nguyen VT, Kiss T, Michels AA, Bensaude O (2001) 7SK small nuclear RNA binds to and inhibits the activity of CDK9/cyclin T complexes. Nature 414(6861):322–325
Yang Z, Zhu Q, Luo K, Zhou Q (2001) The 7SK small nuclear RNA inhibits the CDK9/cyclin T1 kinase to control transcription. Nature 414(6861):317–322
Michels AA, Fraldi A, Li Q, Adamson TE et al (2004) Binding of the 7SK snRNA turns the HEXIM1 protein into a P-TEFb (CDK9/cyclin T) inhibitor. EMBO J 23(13):2608–2619
Yik JH, Chen R, Nishimura R, Jennings JL et al (2003) Inhibition of P-TEFb (CDK9/Cyclin T) kinase and RNA polymerase II transcription by the coordinated actions of HEXIM1 and 7SK snRNA. Mol Cell 12(4):971–982
Huang F, Wagner M, Siddiqui MA (2004) Ablation of the CLP-1 gene leads to down-regulation of the HAND1 gene and abnormality of the left ventricle of the heart and fetal death. Mech Dev 121(6):559–572
Kaichi S, Takaya T, Morimoto T, Sunagawa Y et al (2011) Cyclin-dependent kinase 9 forms a complex with GATA4 and is involved in the differentiation of mouse ES cells into cardiomyocytes. J Cell Physiol 226(1):248–254
Small EM, Olson EN (2011) Pervasive roles of microRNAs in cardiovascular biology. Nature 469(7330):336–342
Wang J, Martin JF (2014) Macro advances in microRNAs and myocardial regeneration. Curr Opin Cardiol 29(3):207–213
Eulalio A, Mano M, Dal Ferro M, Zentilin L et al (2012) Functional screening identifies miRNAs inducing cardiac regeneration. Nature 492(7429):376–381
Sayed D, Hong C, Chen IY, Lypowy J et al (2007) MicroRNAs play an essential role in the development of cardiac hypertrophy. Circ Res 100(3):416–424
Lin Z, Pu WT (2014) Harnessing Hippo in the heart: Hippo/Yap signaling and applications to heart regeneration and rejuvenation. Stem Cell Res 13(3 Pt B):571–581
Xin M, Kim Y, Sutherland LB, Murakami M et al (2013) Hippo pathway effector Yap promotes cardiac regeneration. Proc Natl Acad Sci USA 110(34):13839–13844
Yamamoto S, Yang G, Zablocki D, Liu J et al (2003) Activation of Mst1 causes dilated cardiomyopathy by stimulating apoptosis without compensatory ventricular myocyte hypertrophy. J Clin Invest 111(10):1463–1474
Huang Y, Harrison MR, Osorio A, Kim J et al (2013) Igf signaling is required for cardiomyocyte proliferation during zebrafish heart development and regeneration. PLoS One 8(6):e67266
Chablais F, Jazwinska A (2012) The regenerative capacity of the zebrafish heart is dependent on TGFbeta signaling. Development 139(11):1921–1930
Aurora AB, Porrello ER, Tan W, Mahmoud AI et al (2014) Macrophages are required for neonatal heart regeneration. J Clin Invest 124(3):1382–1392
Kimura W, Muralidhar S, Canseco DC, Puente B et al (2014) Redox signaling in cardiac renewal. Antioxid Redox Signal 21(11):1660–1673
Puente BN, Kimura W, Muralidhar SA, Moon J et al (2014) The oxygen-rich postnatal environment induces cardiomyocyte cell-cycle arrest through DNA damage response. Cell 157(3):565–579
Santos CX, Anilkumar N, Zhang M, Brewer AC et al (2011) Redox signaling in cardiac myocytes. Free Radic Biol Med 50(7):777–793
Yan F, Yao Y, Chen L, Li Y et al (2012) Hypoxic preconditioning improves survival of cardiac progenitor cells: role of stromal cell derived factor-1alpha-CXCR4 axis. PLoS One 7(7):e37948
Chong JJ, Chandrakanthan V, Xaymardan M, Asli NS et al (2011) Adult cardiac-resident MSC-like stem cells with a proepicardial origin. Cell Stem Cell 9(6):527–540
Zhou B, Honor LB, He H, Ma Q et al (2011) Adult mouse epicardium modulates myocardial injury by secreting paracrine factors. J Clin Invest 121(5):1894–1904
Smart N, Bollini S, Dube KN, Vieira JM et al (2011) De novo cardiomyocytes from within the activated adult heart after injury. Nature 474(7353):640–644
Fisher SA, Brunskill SJ, Doree C, Mathur A et al (2014) Stem cell therapy for chronic ischaemic heart disease and congestive heart failure. Cochrane Database Syst Rev 29(4):CD007888
Bettencourt-Dias M, Mittnacht S, Brockes JP (2003) Heterogeneous proliferative potential in regenerative adult newt cardiomyocytes. J Cell Sci 116(Pt 19):4001–4009
Acknowledgement
This work was supported by the British Heart Foundation Centre Core Award and Research Excellence award and the Medical Research Council (UK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare to have no conflicts of interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
18_2016_2404_MOESM1_ESM.mov
Supplemental movie - Laser pulse injury of the zebrafish embryonic heart – A single laser pulse, in this example at the atrioventricular cushion of a zebrafish embryo at 72hpf, produces an instantaneous cessation of ventricular contraction and gradual recovery of cardiac rhythm over the next few minutes. Laser ablation also results in regurgitation of blood from ventricle to the atrium resulting in flow reversal in the cardinal veins (MOV 1712 kb)
18_2016_2404_MOESM2_ESM.pdf
Figure S1- Scheme outlining the efficacy of zebrafish embryo as an ideal model for a high throughput approach. A. Zebrafish genome can be manipulated by injection at 1-2 cell stage eggs of molecular compounds such as CRISPR/Cas9 or morpholino. In this images, a morpholino tagged with lissamine as tracker was injected and visible under fluorescence light. B. Zebrafish larvae, here at 48hpf, can be exposed to drug and/or the hearts can be injured by laser. C. Cardiac function can be easily assessed later during the development by ejection fraction or tail blood flow analysis. D. Many zebrafish larvae hearts can be easily isolated in a few minutes from a tg(myl7:gfp)y1 line and be used for several applications, including immunostaining, gene and protein expression. (The content in this figure is responsibility of the authors) (PDF 1394 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Matrone, G., Tucker, C.S. & Denvir, M.A. Cardiomyocyte proliferation in zebrafish and mammals: lessons for human disease. Cell. Mol. Life Sci. 74, 1367–1378 (2017). https://doi.org/10.1007/s00018-016-2404-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-016-2404-x