Abstract
The cellular prion protein (PrPC) is well-known for its involvement, under its pathogenic protease-resistant form (PrPSc), in a group of neurodegenerative diseases, known as prion diseases. PrPC is expressed in nervous system, as well as in other peripheral organs, and has been found overexpressed in several types of solid tumors. Notwithstanding, studies in recent years have disclosed an emerging role for PrPC in various cancer associated processes. PrPC has high binding affinity for 37/67 kDa laminin receptor (RPSA), a molecule that acts as a key player in tumorigenesis, affecting cell growth, adhesion, migration, invasion and cell death processes. Recently, we have characterized at cellular level, small molecules able to antagonize the direct PrPC binding to RPSA and their intracellular trafficking. These findings are very crucial considering that the main function of RPSA is to modulate key events in the metastasis cascade. Elucidation of the role played by PrPC/RPSA interaction in regulating tumor development, progression and response to treatment, represents a very promising challenge to gain pathogenetic information and discover novel specific biomarkers and/or therapeutic targets to be exploited in clinical settings. This review attempts to convey a detailed description of the complexity surrounding these multifaceted proteins from the perspective of cancer hallmarks, but with a specific focus on the role of their interaction in the control of proliferation, migration and invasion, genome instability and mutation, as well as resistance to cell death controlled by autophagic pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The cellular prion protein (PrPC) is a cell surface glycoprotein mainly expressed in central nervous system. Nevertheless, it is ubiquitously expressed in various organs and systems of all known mammals [1]. PrPC has been intensively studied for its involvement in a group of neurodegenerative disorders, known as prion diseases and characterized by misfolding of PrPC into the pathological isoform PrP scrapie (PrPSc) [2]. PrPC is 253 amino acid residues long and it is composed of a “disordered” unstructured N-terminal region and a “structured” C-terminal domain (Fig. 1A). Features of N-terminal domain are an octapeptide repeat involved in the binding of divalent ions, a central hydrophobic tract and two cleavage sites (α and β); the structured domain consists of three α-helices and two short antiparallel β-sheet strands [3, 4].
3D models of the full length human PrPC and RPSA proteins. A PrPC structure consists of N-terminal unstructured domain and C-terminal structured domain. The dashed red lines indicate two regions on PrPC involved in RPSA binding: the 53–93aa region as an indirect HSPG-dependent interacting domain and the 144–179aa region as direct interaction domain. B RPSA structure consists of a globular N-terminal domain and an intrinsically disordered, unstructured C-terminal domain. The two main binding sites for PrPC are indicated in the figure with dashed lines (in red), namely, the 161–180aa and 209–229aa region. To generate 3D models AlphaFold software was used producing a per-residue confidence score (pLDDT) between 0 and 100. Protein regions below 50 pLDDT may be unstructured in isolation
PrPC undergoes various types of post-translational modifications, among which attachment of a glycosyl–phosphatidylinositol (GPI) moiety, that allows PrPC anchoring to the outer leaflet of the plasma membrane, and several proteolytic processes that generate various truncated or soluble forms of the protein [3, 5]. Altogether, glycosylation and proteolytic processes generate a variety of isoforms that may underlie the wide PrPC functions, ranging from neuronal development to peripheral nerve myelin maintenance, differentiation, stress protection, mitochondrial homeostasis, and cell adhesion. As a GPI-anchored protein, PrPC is largely dependent on its receptors to mediate the aforementioned functions. Among these receptors, a high binding affinity for PrPC is shown by the 37/67 kDa laminin receptor (LR), originally identified as a non-integrin cell surface receptor for laminin-1 (LM), the major component of basement membranes. It was also known as ribosomal protein SA (RPSA), since it was discovered as a component of the 40S ribosome [6]. Afterwards, this protein has been identified as an interactor for both the cellular PrPC isoform and the pathological misfolded PrPSc [7, 8].
The human RPSA gene encodes for the 37 kDa LR, consisting of a globular N-terminal domain and of an intrinsically disordered C-terminal domain (Fig. 1B). The 37 kDa LR is considered the cytosolic precursor of the higher molecular weight species included the 67 kDa LR; nevertheless, it is still unclear what post-translational modifications are responsible for the conversion of 37 kDa LR precursor into the higher molecular weight isoforms. Interestingly, the 67 kDa LR is a type II transmembrane protein that consists of three domains: the intracellular N-terminal domain, a transmembrane domain, and an extracellular C-terminal domain. The N-terminal domain interacts with the TGF-inhibited membrane-associated protein (TIMAP), thus representing a possible mechanism modulating several downstream signal transduction pathways. The 161–180aa region of RPSA, also known as peptide G, serves as binding site for LM, heparan sulphate proteoglycans (HSPG) and PrPC, as well as the 209–229aa region that similarly binds LM and PrPC (Fig. 1B). Mapping analysis in yeast two-hybrid system and cell-binding assay identified two sites on PrPC involved in the interaction with RPSA. Interestingly, the 144–179aa region, also termed PrPLRPbd1, mediates a direct interaction between PrPC and RPSA, whereas the 53–93aa region, PrPLRPbd2, mediates an indirect HSPG-dependent interaction [9].
The structural versatility of RPSA correlates with its multifunctionality, indeed its functions are surprisingly diverse including the well-known cell anchoring via laminins, as well as ribosomal biogenesis and participation to translation, pre-rRNA processing, roles in cellular migration, invasion, viability and growth, cytoskeleton organization, chromatin and histone binding (reviewed in DiGiacomo and Meruelo) [10].
Beyond their physiological roles, recent findings revealed the involvement of both PrPC and RPSA in pathological condition, such as cancer [8, 10,11,12].
While the relationship between PrPC and the pathogenesis of prion disorders has been largely established, the link between PrPC and cancer progression was first reported when PrPC gene (PRNP) was identified as one of the most expressed in pancreatic cancer (PC) cells [13]. Afterwards, PrPC over-expression has been associated with diverse types of solid cancers, such as colorectal cancer (CRC) and glioma [12]. PrPC acts as a key factor to enhance the malignant properties of tumors, being involved in several aspects of cancer biology and sustaining many cancer hallmarks. For instance, PrPC enhances tumorigenesis promoting proliferative signaling, evasion from growth suppressors, resistance to cell death, angiogenesis, migration and metastasis, metabolic reprogramming, evasion from immune surveillance and genome instability. Data relating to the biology of PrPC in tumor, according to each hallmark of cancer, are well-summarized in three recent reviews [14,15,16]. Increasing evidence demonstrated that PrPC contributes to tumorigenesis via multiple pathways, such as phosphatidyl-inositol-3 kinase (PI3K)-Akt, extracellular signal regulated kinase (ERK) and Notch, by regulating tumor growth, differentiation, and resistance to conventional therapies. In addition, PrPC has been shown to protect glioma cells from autophagic cell death by targeting mammalian target of rapamycin (mTOR) activity [17]. This finding has prompted us to report about the involvement of PrPC in the autophagic pathway related to cancer.
Recently, 48 somatic mutations in PRNP were identified in cancer patients [18], among which 8 were reported to be pathogenic for prion diseases, and that cancer patients carrying mutations of PRNP may produce PrPSc and may not be diagnosed with prion disease. From these findings it is likely that both the physiological and the pathological form of PrPC can play a role in the biology of cancer.
Interestingly, many human PrPC variants carrying a pathogenic mutation are trapped in intracellular compartments and have an impaired transport to the cell surface [19,20,21]. In addition, PrPC mutants associated with Gerstmann–Straussler–Scheinker human prion disease, seem to affect the cytoskeleton tubulin expression, inducing its down-regulation and disruption of microtubules structures [22]. These latter are crucial not only for intracellular protein transport but also for chromosome segregation during cell division. Thus, the variant amyloidogenic forms of PrPC, through impairment of cytoskeletal proteins, might influence chromosome disjunction with consequences on genome ploidy and stability, which represent one of the cancer hallmarks.
However, it is still unknown whether PrPC mutants can have an impaired trafficking in cancer cells and whether they are able, as compared to wild-type PrPC, to establish interaction with RPSA.
Similar to PrPC, RPSA expression has been found increased in neoplastic cells, as compared to their normal counterparts, and directly correlated with several hallmarks of cancer, such as maintenance of cell viability, increase of cellular proliferation, as well as enhanced invasive and metastatic potential [10].
One of the first papers showing that both PrPC and its interactor RPSA were upregulated in cancer, derives from data by Zhou et al. in gastric cancer (GC) [23]. These authors suggested that PrPC and RPSA synergically act to influence GC biology in a manner that patients with high level of both proteins had the poorest prognosis (see the section “Proliferative signaling, invasion and metastasis mediated by PrPC/RPSA interaction”).
Moreover, at a cellular level, an interaction between PrPC and RPSA was first established in a two-hybrid screening of a HeLa cDNA expression library for PrPC-interacting proteins [9]. The pathogenic significance of PrPC/RPSA interaction has been revealed in tissues and cells from scrapie-infected versus non-infected mice [24]. In particular, in the brain tissue, high levels of PrPSc accumulation corresponded to high levels of RPSA, which have been then described to play a crucial role in prion propagation. Although the important role of RPSA in mediating binding and internalization of PrPC and its involvement in pathological mechanisms of prion diseases has been clearly demonstrated [25, 26], the specific role of the cross-talk between PrPC and RPSA in cancer has not been addressed yet, and requires further investigation. Since growing evidence is demonstrating that both these proteins have per se a role in carcinogenesis, our aim in this review is to shed light on the hypothesis that they could synergically modulate the biological processes responsible for the neoplastic transformation, thus representing valid therapeutic targets. Interestingly, Pesapane et al. [27] identified small molecules, targeting the peptide G domain of RPSA, able to inhibit RPSA interaction with LM in normal human kidney cells. Subsequently, we reported that these small inhibitors impaired RPSA interaction with PrPC in neuronal cells, as well as in non-neuronal cells, antagonizing the direct PrPC binding to RPSA via the peptide G domain [28]. These findings are very crucial considering that the main function of RPSA is to enhance tumor cell adhesion to the LM of basement membranes and cell migration, two key events in the metastasis cascade.
The study of the mechanisms underlying the PrPC/RPSA interaction might help not only in understanding the pathogenesis of different cancers but also for developing prophylactic and therapeutic measures, as well as for diagnosis.
The primary aim of this review is to focus on the role of PrPC/RPSA interaction in the control of cancer hallmarks, such as proliferation, migration and invasion, genome instability and mutation, as well as resistance to cell death modulated by autophagic pathway. This need derives from highlighting the cancer hallmarks mainly shared by the two proteins.
Moreover, to provide a broad overall view on the topic, we also report more recent findings concerning the individual involvement of the two molecules in the aforementioned hallmarks. We believe that this work can represent the basis for future studies on this topic.
PrPC and RPSA involvement in proliferative signaling, invasion and metastasis
The role of PrP C in proliferation
Several works supported a role of PrPC in contexts outside the brain by regulating myelin homeostasis, immune system processes and cancer progression [4]. An over-expression of PRNP, unrelated to age, gender and stage, was initially reported in diverse types of solid cancers notably glioblastoma (GBM), pancreatic ductal adenocarcinoma (PDAC) and lung adenocarcinoma (LAC), CRC, GC and breast cancer (BC) [29, 30]. Later, a series of evidence from in vitro and in vivo experimental models suggested the PrPC involvement in tumor biology, affecting fundamental properties of cancer cells first and foremost a sustained cell growth [31, 32]. The inappropriate cancer cell proliferation results from a deregulation of intracellular networks that in physiological situations orchestrate transient cell growth and division during embryonic development and homeostatic maintenance of tissues. In this section, we only focused on the sustaining proliferative signaling pathways involved in cell viability maintenance without including the apoptotic process, a distinct barrier to aberrant cancer cell proliferation, that falls beyond the focus of this review.
Due to PrPC physiological cellular localization (cell surface, Golgi, nuclear and cytosolic forms), it is not surprising that it modulates a wide range of molecules including cell growth mediators and signal intracellular transducers [4, 33, 34], all potential drivers of altered cell proliferation. The role of PrPC over-expression in cancer cell proliferation and the related molecular mechanisms involved, have been established in several experimental models and addressed in recent reviews [12, 16]. One of the most frequently activated signaling pathways in cancer is the PI3K/Akt/mTOR pathway [35]. In human GC cell line, the ectopic expression of PrPC promoted cellular proliferation, at least partially, through activation of PI3K/Akt pathway. As revealed by western blotting analysis, phosphorylated Akt was markedly up-regulated, an effect inhibited by pharmacological blockade of Akt through LY294002 inhibitor [36]. Moreover, the over-expression of PrPC activated cell cycle machinery up-regulating Cyclin D1 at both mRNA and protein levels, thus enhancing G0/G1 to the S phase transition [36] or G2/M vs G1/S phase, as reported for glioma cells [37]. Interestingly, octapeptide repeat region in the N-terminal of PrPC plays a pivotal role in promoting the PrPC-dependent proliferative effect in GC cells, indeed its deletion almost completely abolished the cellular proliferation [36]. PrPC-dependent proliferation has been well-documented also in human BxPC3 and Panc 02.03 PDAC cell lines [38]. Upon PrPC silencing, the two cell lines proliferated slower than the control cells and when inoculated in nude mice a significant decrease of tumor growth was observed.
Wang and collaborators [39] reported that, at molecular level, PrPC silencing downregulated the levels of Notch signaling pathway (commonly activated in tumor) in pancreatic cancer cells, enhancing Notch1 proteasomal degradation and consistently suppressing its downstream transcriptional factor target, Hes1. It was also observed a reduction of Ki-67, a key marker of cell proliferation.
A modulation of Ki-67 has been previously proven both in GBM patients and U87 GBM cells xenografted tumors by Lopes and collaborators [40]. They also found a link between PrPC and heat-shock protein 70 (Hsp70)–Hsp90-organizing protein (HOP, aka stress inducible phosphoprotein 1, STI1 or STIP1), resulting in a modulation of GBM cell proliferation through ERK1/2 and PI3K pathways.
More recently, it has been investigated the subcellular localization of PrPC in PDAC tissues by electron microscopy: cells with higher expression of PrPC (compared with control), revealed a peculiar stoichiometric increase of the PrPC in the nuclear compartment, suggesting a direct role as a transcriptional modulator of several pathways involved in tumorigenesis [41]. Accordingly to pathology results, PrPC expression in PDAC tissues correlated with cancer stage, confirming its role as marker of aggressiveness [42].
In human PC3 prostate cancer cell line, a correlation between PrPC and the amyloid precursor protein (APP) and its proteolytic fragments (Aβ peptides) has been reported [43]. The use of an antibody that recognizes the 92–110 epitope within PrPC, that is known to bind Aβ, reduced the tumor growth in PC3 xenografts mice, suggesting a PrPC-dependent tumor-promoting effect of Aβ (see also the section “The role of PrPC in invasion and metastasis”). Moreover, we found that a reduction of Aβ in fibroblasts from patients affected by a genetic form of Alzheimer’s disease, was achieved through modulation of RPSA using a specific small molecule inhibitor [44]. In the context of cancer, our finding is promising if we consider the possibility to modulate RPSA and Aβ levels.
In ovarian cancer (OC) tissues and cell lines, it has been reported a decreased PrPC expression both at mRNA and protein level. Next, in vitro experiments showed that PRNP overexpression decreased the proliferation capacity of two OC cell lines suggesting, unlike other types of solid tumor, a potential tumor-suppressive role of PNRP [45]. This is the only study currently published in total disagreement with the oncogenic role of the PrPC in other cancer types, suggesting how complicated the function and the regulation of PrPC in cancer can be. However, most of the data of this study were based on three public databases, so further in vitro and in vivo experiments will be needed to consolidate these findings.
In tumor microenvironment (TME) of most solid cancers are included cancer stem-like cells (CSCs) which enable tumor growth and progression [31]. A number of evidence indicate that PrPC sustains CSCs in vitro and in vivo proliferation rate of several cancer cells, such as CRC and GBM as described later [46,47,48].
The role of RPSA in proliferation
In common with PrPC, several investigations have clearly demonstrated an increase of RPSA expression in epithelium-derived cancers, as compared to the corresponding normal tissue [8, 49, 50].
SiRNA mediated RPSA knock-down resulted in a significant decrease of viability in different cancer cell lines, such as BC and oesophageal cancer cells [51], pancreatic cancer and neuroblastoma cells [52], melanoma [53] and CRC cells [54]. Regarding proliferation-promoting processes, the main mechanism first explored in cancer cells expressing high levels of RPSA was apoptosis. Since this mechanism is beyond the focus of our review, we only describe the involvement of RPSA in proliferation by maintaining cell viability and controlling survival pathways.
An alternative proposed mechanism of resistance to programmed cell death by which RPSA maintains cell viability is through the high binding with the Midkine, a heparin binding multifunctional protein, overexpressed in a variety of human carcinoma correlating with a poor prognosis. The complex RPSA-Midkine translocated into the nucleus [55], where it could be involved in the maintenance of nuclear stability and in turn in cellular survival [50, 51]. Interestingly, RPSA was also associated with cell adhesion-mediated drug resistance (CAM-DR) in an in vitro and in vivo model of GC drug-resistance cell line. RPSA stimulated Focal Adhesion Kinase (FAK) phosphorylation and the PI3K/Akt and MEK/ERK1/2 survival pathways [56]. Similar results were observed in multidrug resistance CRC cell line SW480 [57].
The role of PrP C in invasion and metastasis
In the progression of tumor with aggressive phenotype, the cells acquire the ability to disseminate into the body by different strategies. The cells become invasive and migratory and via bloodstream and/or lymph nodes colonize ectopic tissues generating metastases. All the process is very complex involving both the action of specific cancer cell intracellular pathways and the support from cells in the TME [32]. In this context, cancer cells activate an intrinsic mechanism, namely, epithelial mesenchymal transition (EMT), through which cells able to migrate from the primary tumor, invade surrounding tissues, and reach distant sites [58]. All these processes have been associated with PrPC overexpression in several types of cancer cells [12, 16, 29].
PrPC localization in lipid rafts of the plasma membrane [59] is consistent with its engagement in cell–matrix interactions, in adhesion and migration, cellular processes in which is also involved filamin A (FLNA), an actin binding protein which links cell mechanics and signaling [60].
PrPC is a secretory protein which possesses the N-terminal signal peptide that is cleaved to generate pro-PrP, whereas at the C-terminus contains the signal peptide for GPI–anchor attachment (GPI–PSS). This signal is then cleaved to generate the GPI-anchored form of PrPC. In contrast to GPI-anchored PrP isoform, PrPC exists as pro-PrP in most of the human PDAC cell lines (BxPC-3 and Panc 02.03) [38] and in melanoma M2 cell line [61]. Here, the presence of the uncleaved GPI–PSS allows the protein itself to become an integral transmembrane bound protein, able to establish interaction with β1-integrin and FLNA, with a consequent arrangement of actin cytoskeleton. This event provides a migratory phenotype to these cancer cell lines. More recently, to further investigate how PrPC modulates melanoma biology, the same group reported that pro-PrP promotes cancer cell migration and invasion in in vitro and in vivo models, binding two partners known for their involvement in tumor metastasis, namely, Insulin-like growth factor-1 receptor (IGF-1R) and E3 ligase c-Cbl, and promoting autophagy, which is discussed later [62]. Interestingly, pro-PrP contains its GPI–PSS, a crucial sequence of ten aminoacids, that allows the building of trimeric complex pro-PrP/c-Cbl/IGF-1R, and without which the migratory phenotype is mitigated [62]. In several PDAC cell lines (BxPC-3, Capan-1, Panc10.5, Panc-1) pro-PrP isoform interacts with the transmembrane protein Notch, as well as FLNA. Overexpression of pro-PrP increases Notch expression, enhancing cancer cell migration and invasion ability, an effect that can be blocked by a Notch inhibitor [39]. More recently, a molecular and functional link between PrPC and caveolin 1 (CAV1), the protein coat of caveolar plasma membrane identified as marker of EMT [63], has been reported in human PC3 prostate cancer cells, LoVo and MDST8 colon cancer cell lines [43]. Using cell-based assays, the authors found that PrPC up-regulated the expression of CAV1 and, by proximity ligation assays, that the two proteins colocalized in the two aforementioned colon cancer cells, reminiscent of that reported in 1C11 neuronal cells [4]. Further studies will be needed to strengthen these findings and to understand the role of PrPC–CAV1 interaction in the context of cancer. In the same study, the authors disclosed that PrPC controlled the expression of APP and Aβ peptides. Using an antibody that blocked PrPC–Aβ interaction, it has been established that Aβ–PrPC signaling regulated pathways involved in the mesenchymal subtype of colon cancer cell line, in agreement with a recent finding that identified melanoma-secreted Aβ as a promoter of brain metastasis [64].
PrPC expression increases lamellipodia formation, protrusions of cell membrane organized by actin filaments and considered motor which pulls the cell forward during the process of cell migration and invasion capability of cultured LAC cell lines. These effects were inhibited when cells were treated with the Janus kinase (JNK) inhibitor SP600125, suggesting that PrPC promotes lung cancer cell migration through upregulation of JNK signaling [30].
During invasion process, it is required the activity of matrix metalloproteinase's (MMPs), enzymes that break down extracellular matrix to permit the passage of cancer cells. PrPC overexpression enhances migration and invasion of GC cell lines via activation both of MMP-11 and mitogen activated protein kinase (MAPK) signaling, one of the principal pathways involved in cancer cell migration [65, 66]. Similar results have been reported in BC cells [67].
Among the components of TME, a key role for the progression of the tumor is played by CSCs. When GBM cells were cultured as neurospheres, they expressed both stemness markers, such as CD133 and high levels of PrPC; in addition, the two proteins co-localized in plasma membrane of GBM stem-like cells (GSCs) [48]. In PrPC-silenced GSCs the authors observed a decrease of CD133 expression and of cell surface adhesion proteins, such as E-cadherin and integrin α6, resulting in a reduced migration on laminin. Collectively, these results provided that PrPC may stabilize cell adhesion molecules on CSCs cell surface. In the field of CRC, Du and collaborators [46] revealed that PrPC expression defines a subpopulation of CD44-positive CSCs (PrPC+CD44+) with high liver metastatic capability in orthotopic xenograft models, an effect significantly inhibited by administration of PrPC monoclonal antibodies. PrPC+/CD44+ cells showed an elongated mesenchymal-like morphology characteristic of the EMT phenotype accompanied by an upregulated expression of mesenchymal genes (N-cadherin and Twist), as well as a downregulated gene expression of epithelial markers (E-cadherin). In a recent work, PrPC silencing attenuated the characteristic molecular landscape of a CRC–CSCs subpopulation with mesenchymal phenotype, through the recruitment of the Hippo pathway effectors YAP and TAZ and the TGFβ pathway, suggesting that PrPC deregulation could contribute to a CSCs phenotype in CRC [68]. Moreover, the same group has identified Integrin Linked Kinase (ILK) as close effector of PrPC, whose expression controls, mesenchymal phenotype of CRC–CSCs subtype, sustaining adhesion, migration, as well as invasion [69].
Finally, in the context of intercellular communication, the release of exosomes by cancer cells has been associated with several hallmark features of cancer, influencing tumor growth and metastasis [70]. Under hypoxic conditions, CRC cells secreted PrPC-expressing exosomes which were able to significantly increase the expression of CSCs markers, such as ALDH1A, Nanog and Oct4, measured by flow cytometric analysis and to sustain tumor progression in an in vitro model [71].
The role of RPSA in invasion and metastasis
RPSA plasma membrane localization allows its binding to one of the principal components of extracellular matrix, LM, a glycoprotein involved in several biological processes, such as adhesion, migration and growth. Several proteolytic enzymes are activated as a consequence of this binding, resulting in the degradation of the basal lamina and in the following migration and invasion of tumor cells [72, 73]. To investigate metastatic potential conferred by RPSA in malignant melanoma cells, RPSA nontoxic specific antibody, IgG1–iS18, has been used [74]. Flow cytometric analysis revealed that melanoma cells displayed high levels of RPSA on their cell surface and the treatment with IgG1–iS18 antibody determined a significant reduction of the adhesive potential to LM and the invasive capacity of malignant melanoma cells, confirming previous results reported in several types of cancer cells [75,76,77,78]. For instance, in A549 lung cancer cell line, downregulation of RPSA with specific siRNA, inhibited cellular migration activity, as evaluated by scratch motility assay [79]. From a mechanistic point of view, proteomic analysis performed in pancreatic cancer cells, revealed that RPSA interacted with Integrin alpha 6 (ITGA6) and that they colocalized on the plasma membrane as showed by immunofluorescence analysis. Interestingly, although RPSA interacted with ITGA6, they promoted cellular migration by activating two different signaling pathways, namely, PI3K and MAPK, respectively [80]. More recently, Gresseu and collaborators [81] reported that neurospheres derived by U87 glioblastoma cell culture, expressed increased levels of RPSA, of EMT genes, such as SNAIL and fibronectin, as well as stemness markers SOX2 and CD133, compared to corresponding 2D culture cells. These data suggested a correlation between RPSA expression both with EMT process and the acquisition of CSC phenotype.
Proliferative signaling, invasion and metastasis mediated by PrP C /RPSA interaction
The observation that PrPC and RPSA are overexpressed in GC and that the two proteins interact with high affinity [82], led Zhou's group to analyze their expression levels in a cohort of 238 patients affected by GC by combining the tissue microarray technique and immunohistochemical method. It has been also evaluated their correlation with the clinicopathological characteristics of patients to understand if their co-expression could be used as a marker of poor prognosis [23]. They found that tissues with high expression of PrPC also had a high rate of upregulated expression of RPSA and for these patients the prognosis was the poorest. Later, the interaction between PrPC and RPSA has been assessed both by co-localization, examined by immunofluorescence staining, and co-immunoprecipitation experiments in GC cell lines and tissues [83].
The biological effects of PrPC/RPSA interaction were for the first time evaluated in the development of Merlin-deficient tumors by Provenzano and collaborators [84]. To demonstrate that the observed PrPC overexpression was due to Merlin deficiency, they reintroduced wild-type Merlin gene in cancer cells with consequent strongly reduction of PrPC levels. The increased PrPC expression was caused by its upregulated transcription and not by decreased proteasomal degradation. PrPC knockdown induced a reduction of Ki67-positive proliferating cells, as well as of cyclin D1, accompanied by a decreased expression and activation of ERK1/2, FAK and Akt. In addition, cancer cells were less capable of adhering to the cell matrix. All these cellular events were mediated by PrPC acting via RPSA [84]. In addition, the same authors demonstrated that PrP synthetic peptide (aminoacid residues 105–120 of the human PrP) was able to protect schwannoma cells from H2O2-mediated cell stress. This study represents one of the first evidence for signaling of PrPC via RPSA with the engagement of downstream Erk1/2, PI3K/Akt and FAK signaling pathways to control survival, proliferation and cell–matrix–adhesion in schwannoma cells, suggesting PrPC and its interactor as potential therapeutic targets for schwannomas. Moreover, the same group [84] described a strong release of PrPC through exosomes from schwannoma cells and, together with an independent paper from Guitart et al. [85], reporting the presence of both PrPC and RPSA in exosomes deriving from neuronal cells, reinforced the idea to possibly modulate the two proteins for fight cancer.
Genome instability and mutation: the role of PrPC and RPSA
The aberrant proliferation of cancer cells correlates with both an increased rate of genomic changes and accumulation of mutations in multiple genes regulating cell division and tumor suppression, further conferring advantage to the neoplastic transformation of cells. This cancer hallmark is known as genomic instability [31].
PrP C in DNA damage response (DDR)
PrPC has been largely demonstrated to be involved in the maintenance of genomic stability after exposure to oxidative stress and genotoxic stimuli as a key factor in the DDR. The requirement of PrPC to preserve genome stability in response to genotoxic insult, has been shown in mouse brain and in human neuroblastoma cells by Bravard et al. [86]. The results obtained in both experimental models, indicated PrPC as a strong activator of DNA damage repair through stimulation of the base excision repair enzyme apurinic/apyrimidinic endonuclease (APE1) activity in the nucleus. Moreover, genotoxic stress, as well as oxidative stress, leads to transcriptional activation of PRNP, further reinforcing the scenario of PrPC involvement in DDR [86,87,88]. PrPC upregulation has been also reported in response to copper-induced oxidative stress by ataxia telangiectasia-mutated (ATM)-mediated pathway [88]. PrPC was reported as marker of cellular stress to aneuploidy in colon cancer screening by Domingues et al. [89]. In this study, conducted in HCT116 CRC cells upon serum deprivation, the levels of PrPC increased in aneuploid cells and were protective against serum-deprivation induction of death. The main message deriving from this research was that the upregulation of PrPC in aneuploid cells was a consequence of the oxidative stress associated with this genomic alteration. These findings fit well with the data from Bernardino et al. [90], where PrPC expression conferred protective role against irradiation. Collectively, these studies sustain that upon DNA injury, PrPC is induced and its main job is to protect cells from DNA damage, hence it is clearly emerging a role for PrPC in balancing DNA damage and repair [89, 90]. Thus, PrPC has been proposed to represent one of the protein needle of the balance between cell survival and growth, two critical events in cancer cells. In this context, it should be noted that expression of genes for prion protein changes in certain cancers, such as glioblastoma, melanoma, pancreatic and GC [29, 91]. The protective function of PrPC could be crucial to prevent the long-term accumulation of DNA damage in neurons; however, since the ubiquitous expression of PrPC in mammalian tissue, it is likely that DNA protective pathway can be active in other cell types.
RPSA in DDR
Similar to PrPC, RPSA receptor is involved in DDR by controlling the nucleolar localization of the double-strand break repair factors RNF8 and BRCA1 [92]. RPSA interacts with the ubiquitin ligase RNF8 in the nuclei of HEK293T cells, whereas upon γ-irradiation, RNF8 dissociates from RPSA and translocates into the nucleoplasm to participates to DNA repair, thus suggesting a putative role for RPSA in RNF8 anchoring to nucleolus. Likewise, BRCA1 nucleolar localization requires both RPSA and RNF8, although their interaction has not been demonstrated yet.
Altogether, these studies highlighted in DDR a physiological role of PrPC and RPSA, which can exert a protective function against the accumulation of DNA damage. In a pathological context, the over-expression of PrPC and RPSA frequently reported in several cancer types, can be the consequence of the genomic instability and it likely represents a mechanism through which the cells enhance the DDR to counteract the accumulation of DNA damage. On the contrary, one could also speculate that the unbalanced expression level of PrPC and RPSA can contribute to genomic instability causing a dysfunctional response to DNA damage and replication stress. Hence, to date, it still remains under debate whether the upregulation of PrPC and RPSA in pathological context could be a mechanism that sustains or counteracts DDR and genomic instability.
PrP C in chromatin remodeling
Beyond the recruitment of the repair factors, another important aspect of DDR includes the chromatin remodeling. Although PrPC is a cell surface protein, it is well-established that it can translocate to the nuclei [93], where it shows high affinity for polyanionic molecules, such as nucleic acids (reviewed in Silva et al.) [94, 95]. The ability of PrPC to interact with nucleic acids has been extensively demonstrated both in vitro [96] and in vivo [97], and this interaction can lead to conformational changes of both the protein and the nucleic acid molecule itself. Indeed Bera et al. [96] demonstrated that PrPC is able to bend and unwind DNA, resembling the same structural changes of DNA induced by proteins involved in transcriptional regulation.
Beside the interaction with nucleic acids, it was also demonstrated the interaction of PrPC with chromatin histones, such as H1 and H3 [93], strengthening previous data on PrPC involvement in transcriptional regulation in the nucleus.
RPSA in chromatin remodeling
RPSA as well, potentially interacts with chromatin, both directly interacting with DNA, as demonstrated in in vitro study [98] and more tightly through histones [99]. Hence, among the RPSA binding proteins identified in NIH3T3 cell extracts, there is a large group of proteins involved in DNA/chromatin maintenance. Histones H2A, H3 and H4 were all identified to be interactors of RPSA. In addition, SWI/SNF complex subunit SMARCC1/BAF155, which is thought to regulate transcription through alteration of chromatin structure, was isolated as RPSA interactor [98]. Collectively, these data suggest that the association of RPSA with both histones and chromatin modifying proteins, results in chromatin maintenance or modification.
PrP C regulating genomic stability
The aforementioned studies are focused on the involvement of the PrPC isoform in several aspects of carcinogenesis, i.e., DDR and chromatin remodeling. However, to date, we cannot conclude whether amyloid directly or indirectly affect carcinogenesis, since only the expression levels of genes for amyloidogenic proteins and not the presence of amyloids in the cells were considered. PrPC, as well as other amyloidogenic proteins, such as amyloid beta peptide (Aβ), tau and α-synuclein, all associated with neurodegenerative diseases, may undergo conformational transitions, during which the content in β-sheet structures increases, and form amyloid oligomers, which are able to grow via interactions between the same regions of protein molecules generating protofibrils [100]. These structures are then combined into fibrils and amyloid aggregates [101]. PrPC in its PrPSc form, has a β-sheet proportion of about 43%, approximately 14 times greater than in its monomeric form PrPC (3%) [102]. A prion-like prionogenic protein, such as tau and α-synuclein, is able to form different inherited conformational variants of aggregates which possess diverse biological properties known as prion strains [103].
Moreover, as reported in a recent review from Andreychuk et al. [104], studies in yeast and bacteria, suggest that prion and prion-like proteins [100, 105] may play a role in regulating DNA replication, thus being directly involved in maintaining the genome stability [106].
At this point a question arises: how can an amyloid protein-like PrPC, affect the stability of the genome in human cells? One possible consideration might be based on the fact that the transition of a protein into the amyloid state seems to affect the microtubules-driven chromosome segregation during mitosis and meiosis [107]. The dynamic instability is a critical point for the function of this class of cytoskeleton filaments. Alteration of microtubules polymerization, as well as spindle-assembly and chromosome segregation, has been associated with the yeast prion protein Sup35p mutations [108]. In mammals, a direct interaction between PrPC and tubulin has been identified in extracts from porcine brains [109], and in vitro experiments using standard light scattering measurements and electron microscopy, showed that full length recombinant PrPC induces a rapid increase in tubulin oligomers associated with sheets of protofilaments, accompanied by reduction of the length of microtubules [110]. These data contributed to postulate that inducing formation of stable tubulin oligomers, PrPC may act as an inhibitor of microtubule assembly. Moreover, PrPC mutants associated with Gerstmann–Straussler–Scheinker human prion disease, seems to yield even a stronger effect on the down-regulation of tubulin expression, with respect to the normal PrPC counterpart, inducing disruption of microtubules structures [22]. It is tempting to speculate that the amyloid forms of the protein, through impairment of cytoskeletal proteins, can cause chromosome nondisjunction during cell division with consequences on genome ploidy.
RPSA regulating genomic stability
A similar interaction with cytoskeletal proteins has been reported for RPSA, as well [111]. RPSA directly interacts with tubulin in NIH3T3 cells acting as a tethering protein, holding the ribosome to tubulin, which is crucial for protein biosynthesis. Moreover, RPSA directly binds to actin, which instead is critical for cell migration. These data indicate that interactions between RPSA and the cytoskeleton, are vital in mediating two cellular processes linked to cancer: protein synthesis and migration. However, whether RPSA interaction with cytoskeleton can influence genome ploidy still remains an open issue.
In this context, another important source of aneuploidy is the cancer-relevant change termed tetraploidization, that has been strictly related to telomere crisis [112]. Telomere-driven tetraploidization occurs in human cells undergoing crisis and promotes transformation of cells [112, 113]. At the ends of the chromosomes, the repetitive DNA sequences known as telomeres, are subjected to the “end replication” problem of DNA polymerases and are maintained by the ribonucleoprotein DNA polymerase complex, telomerase [114]. Telomere crisis occurs during tumorigenesis when depletion of telomere reserve leads to telomere fusion, whose consequence is the formation of dicentric chromosomes, which have been proposed to drive genome instability [115].
In addition to their role in telomeres extension, telomerase and its enzymatic subunit, known as human telomerase reverse transcriptase (hTERT), possess other extra-telomeric functions, such as DNA repair and cell proliferation, which all together converge towards preservation of cell viability [116]. Interestingly, hTERT overexpression and telomerase activity have been detected in most cancer types [117,118,119,120], and among the various proteins found to interact and control hTERT/telomerase functioning in human cancer cell lines, the group of Naidoo et al. [121] identified RPSA. The authors postulated that RPSA upregulation may enhance telomerase activity [122, 123]. Reversely, siRNA-mediated RPSA knock-down impeded telomerase activity in breast and lung cancer cells [79, 121]. Likewise, a significant decrease in telomerase activity and hTERT expression levels was observed in late-stage CRC cells via RPSA siRNA technology, resulting in genetic instability and activation of DDR pathway [54, 124].
Resisting cell death through modulation of autophagic pathway: what about the role of PrPC and RPSA?
Resistance to apoptotic cell death was the first cancer hallmark to be connected to PrPC, nearly 20 years ago [125]. The authors described the upregulation of PRNP transcripts in adriamycin-resistant GC cells, as compared to the normal counterpart. In various cell types, both apoptotic and autophagic cell death pathways can be induced by serum deprivation. Thus, here we will exclusively report and discuss data on the involvement of PrPC and RPSA in autophagic mechanism, mainly in cancer.
It is important to highlight that in cancer cells, three distinct pathways of active autophagy can be induced by amino acid starvation: chaperone-mediated autophagy (CMA) dependent by mTOR inactivation, selective endosomal microautophagy independent of mTOR inactivation and the canonical, ATG protein-assisted macroautophagy, that can be activated by mTOR-dependent signaling. All the three main autophagy pathways likely work in concert and may partially compensate for each other.
Autophagy, also termed type II programmed cell death, is a lysosome-mediated catabolic pathway that allows the elimination of damaged organelles, misfolded protein, thus contributing to cellular homeostasis in non-transformed cell. In cancer, autophagy has a dual role acting both as tumor suppressor and tumor promoter. In fact, autophagy, by eliminating damaged cells and organelles, counteracts malignant transformation during early tumorigenesis. Conversely, in tumoral cells, the autophagic pathway can sustain the survival of cells in the presence of several stress stimuli (for example, hypoxia) representing a tumor-promoting mechanism. The bipolar role of autophagy in cancer is described in more detail in a recent review [126].
PrP C in autophagy
One of the first finding concerning the involvement of PrPC in the autophagic process, has been reported in neuronal cells by Oh et al. [127], which have shown higher autophagic activity in Zürich I mouse PRNP0/0 hippocampal neuronal cells than in wild-type mouse cells. This mechanism was retarded by the introduction of PrPC into PRNP0/0 cells, but not by reintroduction of PrPC lacking the N-terminal octapeptide repeat region, suggesting that this protein region plays a critical role in modulating autophagy exhibited by PrPC. Moreover, Oh et al. [128] provided the first evidence that the deficiency of PrPC may impair autophagic flux via H2O2-induced oxidative stress in hippocampal cells.
The evidence about the PrPC-mediated modulation of autophagy in neurodegeneration has been well-established; however, many of the mechanisms described in neurodegenerative diseases might be shared with tumorigenesis. Indeed, in recent years, several studies have highlighted the correlation between PrPC and autophagic flux modulation in different tumor types.
In tumor cells, the crucial challenge to depict the crosstalk between PrPC and carcinogenesis, is to identify the proteins interacting with PrPC. Among the multiple molecular interactions that PrPC physiologically performs, the ones shared with the autophagic activity are those involved in tumor carcinogenesis of glial cells. Such activity has been identified on mTOR through which PrPC operates for neuroprotection via PI3K/Akt pathways [129].
As reported in the above sections, Akt–PI3K–mTOR signaling axis is among the most frequently deregulated pathway in cancer cells, thus sustaining abnormal proliferation. At the same time, mTOR is considered a master regulator of the autophagic flux through phosphorylation of several substrates.
As described by Akhavan et al. [130], GBM can be considered as a pathology characterized by defects in mTOR activity, at the early stage of its development. mTOR acts both as upstream regulator and as a downstream effector of PI3K. A constitutive PI3K pathway activation is a hallmark of GBM [131].
The role of PrPC in autophagic processes can represent a crucial event in glioma tumorigenesis as reviewed in Armocida et al. [132]. Another report on the ability of PrPC to modulate autophagic cell death in glial tumor cells, comes from studies by Barbieri et al. [17], where PrPC silencing resulted in inhibition of the kinase activity of mTOR, promoting autophagic cell death.
More recently, Lenzi and collaborators [133] provided evidence that rapamycin induced a persistent clearance of PrPC in GBM cells, and removal of PrPC was consistent with enhanced autophagy flux, which efficiently clears such an aggregate prone protein.
As recently reported by Li and collaborators [62], in human PC cell lines and in melanoma M2 cell line, the presence of the uncleaved GPI–PSS in pro-PrP allows the protein to bind the E3 ligase c-Cbl facilitating the IGF-1R ubiquitination for its degradation and consequent autophagic activation through Akt–ULK1 signaling axis, driving melanoma lung metastasis. Importantly, disruption of pro-PrP/c-Cbl/IGF-1R complex using a synthetic peptide, induced a reduction of cancer cell autophagy and mitigated tumor aggressiveness. These findings provide a therapeutic approach for treating human cancers expressing pro-PrP.
RPSA in autophagy
Concerning the role of RPSA in autophagic process, we have recently proposed a pivotal role for RPSA in modulation of autophagic pathway in neuronal cells [134, 135]. The so-called “canonical” autophagic process is orchestrated by the hierarchical and coordinated activity of autophagy-related genes ATG, and the autophagic machinery is modulated by the upstream ULK1/2 complex and the PI3K-class 3/beclin 1 (PI3KC3/BECN1) complex, all together representing the pre-initiation complexes.
Beside this, it is now well-established that ATG proteins may play a role distinct from canonical autophagy and defined as non-canonical pathway driving unconventional microtubule-associated protein 1A/1B-light chain 3 (LC3)-lipidation on endosomes [136,137,138]. LC3, in its lipidated form, is a protein associated with the membrane of forming autophagosomes, whose source of formation mainly derives from plasma membrane and endocytic vesicles [139].
From previous observations, it is now evident that LC3 lipidation can occur independently of canonical autophagy regulators, involving endosomal compartments [139]. In agreement with the above described role of endosomes in autophagic events, we found that inhibition of RPSA by NSC48478, reflected on the upregulation of a large cluster of endosomal-related genes and was accompanied by the induction of LC3 lipidation on Rab5-positive-endosomes. Our findings indicate that RPSA inhibition stimulates a “non-canonical” autophagic pathway, thus sustaining the hypothesis of RPSA-mediated modulation of autophagic process [134, 135].
Interestingly, RPSA is expressed on the cell surface and is internalized through early endosomes. These compartments resulting from the endocytic pathways, have been shown to generate signaling platforms. However, it is not known whether the early endosomes resulting from the internalization of RPSA can exhibit such cell signaling property.
The YIGSR pentapeptide sequence in the β1 chain of laminin-1 has been revealed as the binding site for RPSA [140], and the administration of LM or YIGSR induced internalization of RPSA into early endosomes of adrenal pheochromocytoma PC12 cells. This caused a sustained generation of cell signaling that increased the survival of cells from death induced by serum starvation [141], which is known to induce autophagic cell death.
Beside the evidence of autophagic modulation through non-canonical route, we and other groups demonstrated the role of RPSA as mediator of cellular pathway including autophagy. Several studies demonstrated the correlation between RPSA and the modulation of Akt–PI3K, as well as MAPK/ERK signaling pathway, which, as described above, are crucial regulators of canonical autophagic pathway. For instance, we demonstrated that the inhibition of RPSA inactivates Akt in human skin fibroblasts from familial Alzheimer’s disease [44].
Moreover, our previous finding concerning the inactivation of MAPK/ERK signaling induced by RPSA inhibition in neuronal cells [142] could be of great interest, if one considers the possibility that non-canonical autophagy could generate endo-lysosomal signaling hubs [143, 144]. As such, this hypothesis might provide insight into the efficacy and function of RPSA inhibitor as an autophagy-modulating drug that possesses the property of endosomal lipidation of LC3.
Similar to the above described effects, A375SM melanoma cells expressing reduced level of LR, showed a significantly increased phosphorylation of ERK, JNK and p38, regardless of the exposure to exogenous laminin [145]. To note, Akt/PI3K and MAPK signaling converges on the regulation of mTOR. Hence, the regulation of growth signals, such as Akt–PI3K and MAPK, links RPSA not only to pro-survival signaling but also to autophagic flux.
To date, the interplay between RPSA and signaling pathways controlling autophagy in the context of tumorigenesis still remains underestimated; however, altogether, the aforementioned studies could help to identify molecular mechanisms that can be in common with a complex pathology, such as cancer.
The effect on autophagy modulation of PrPC linked to its receptor RPSA in cancer is even less investigated. Recently Luo et al. [83] demonstrated that MGr1–Ag/37LRP (that is the precursor of RPSA) may interact with PrPC and promote the PrPC-induced multi-drug-resistance in GC through PI3K/Akt pathway. Thus, knockdown of MGr1-Ag/37LRP significantly attenuated PrPC induced multi-drug-resistance by sensitizing drug-induced apoptosis through inhibition of Akt. In this study, the authors explored the contribution of MGr1-Ag/37LRP to PrPC mediated multi-drug-resistance in GC without investigating a possible downstream effect on autophagic pathway. However, in our opinion, this study lays the foundation to further investigation about the hypothesis that PrPC and RPSA could jointly coordinate the autophagic pathway.
Several studies have advanced the hypothesis that diverse signaling pathways controlled by PI3-kinase/Akt, PKC, Fyn and Erk1/2, all implicated in the control of autophagic pathways, are modulated by PrPC expression and interaction with other partners, such as RPSA (reviewed in Mehrpour and Codogno) [33]. In this scenario, such a complex could be destroyed by PrPC toxic mutants (lacking N-terminal region, or the central domain of PrPC [146, 147]) possibly by competing for the binding of some complex components yet failing to interact with the signal transducing factors.
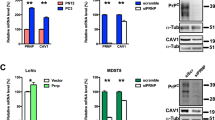
A schematic summary of PrPC and RPSA influences on cellular events related to tumorigenesis up to this point discussed, is provided in Fig. 2.
Summary of the data supporting the involvement of PrPC and RPSA in the regulation of several cancer hallmarks. Proteins and signaling pathways modulated by PrPC and RPSA in cancer cells are reported. The features shared by the two proteins related to cancer hallmarks have been highlighted in bold. Scheme created using BioRender.com
Concluding remarks
Beyond the undoubted involvement of PrPC in neurological disorders [2], many proofs in recent years showed that it is overexpressed in diverse solid tumors and plays a role in the onset and development of cancer influencing cellular events, such as genome instability with consequent gene mutation, proliferation, migration and invasion together with resistance to autophagic death [16, 148]. Among its wide number of ligands, PrPC interacts with RPSA protein, a versatile non integrin LM receptor whose overexpression in solid and haematological malignancies has been associated with the potential invasive and metastatic cell phenotype [10, 50]. Compelling data suggest that PrPC and RPSA can be considered on their own as potential therapeutic targets [12, 149] and Fig. 3 provides a strong visual support to this idea.
Silencing of PrPC expression through antisense oligonucleotide-based strategies could be helpful in the field of cancer therapy, as reported for certain type of tumors [48, 148]. Recently, pro-PrP has been pinpointed as potential therapeutic target to reduce melanoma metastasis in vitro and in vivo, using a peptide that inhibited GPI–PSS of pro-PrP and some of its interacting partners [62]. Application of monoclonal antibodies against PrPC in preclinical models of cancer cells, such as mesenchymal colon cancer [43] or CSCs–CRC [46], has proven to be a well-founded strategy to mitigate cellular aggressive phenotype with minimal toxicity.
RPSA interaction with LM, the main extracellular matrix glycoprotein, makes the receptor a promoter of invasive and metastatic characteristics of cancer cells. Specific antibodies for RPSA and siRNAs targeting RPSA are two therapeutic strategies employed in many tumorigenic in vitro cell lines and both effective in inhibiting the characteristics of multiple cancer hallmarks [8, 50]. Recently, small molecules targeting RPSA and able to interfere with cell binding to LM, have been identified by in silico techniques. The authors focused on the peptide G of RPSA [27], a sequence of twenty aminoacids that binds both LM and PrPC with high affinity and mediates several cellular events linked to carcinogenesis. The best compound, named NSC47924, resulted cell-permeable and selectively inhibited cell adhesion, migration and invasion of cancer cell lines [27]. Next, the same small inhibitor was used by Sarnataro et al. [28] to control trafficking and interaction between PrPC and RPSA, as well as the activation of “non-canonical” autophagic pathway in neuronal cells [134].
Both PrPC and RPSA show an intrinsically disordered protein region (IDPR) (Fig. 1). It is known that IDPRs are commonly involved in a wide range of biological functions, which complement the ones attributed to ordered and structured protein domains (reviewed in Uversky 2013) [150]. Disordered domains are protein regions that do not assume unique three-dimensional structures and have been found to play different roles in the modulation of binding partners function and in promotion of supra-molecular complexes. Thus, multifunctionality of PrPC and RPSA might be attributed to this characteristic feature which make them involved in a wide range of physiological and pathological roles.
In conclusion, despite evidence related to PrPC and RPSA interaction in cancer are beginning to be unveiled, future investigations may allow the development of therapeutic strategies to target this complex specifically.
Availability of data and materials
Not applicable.
Abbreviations
- APE1:
-
Apurinic/apyrimidinic Endonuclease 1
- APP:
-
Amyloid precursor protein
- ATG:
-
Autophagy-related gene
- ATM:
-
Ataxia–telangiectasia mutated
- Aβ:
-
Amyloid beta
- BC:
-
Breast cancer
- CAM-DR:
-
Cell adhesion-mediated drug resistance
- CAV1:
-
Caveolin 1
- CMA:
-
Chaperone-mediated autophagy
- CRC:
-
Colorectal cancer
- CSCs:
-
Cancer stem-like cells
- DDR:
-
DNA damage response
- EMT:
-
Epithelial mesenchymal transition
- ERK:
-
Extracellular signal regulated kinase
- FAK:
-
Focal adhesion kinase
- FLNA:
-
Filamin A
- GBM:
-
Glioblastoma
- GC:
-
Gastric cancer
- GPI:
-
Glycosyl-phosphatidylinositol
- GPI–PSS:
-
GPI peptide signaling sequence
- GSCs:
-
Glioblastoma stem-like cells
- HOP:
-
Hsp70–Hsp90-organizing protein
- HSPG:
-
Heparan sulphate proteoglycans
- hTERT:
-
Human telomerase reverse transcriptase
- IDPR:
-
Intrinsically disordered protein region
- IGF-1R:
-
Insulin-like growth factor-1 receptor
- ILK:
-
Integrin linked kinase
- ITGA6:
-
Integrin alpha 6
- JNK:
-
Janus kinase
- LAC:
-
Lung adenocarcinoma
- LC3:
-
Microtubule-associated protein 1A/1B-light chain 3
- LM:
-
Laminin-1
- LR:
-
Laminin receptor
- MAPK:
-
Mitogen activated protein kinase
- MMP:
-
Matrix metalloproteinase
- mTOR:
-
Mammalian target of Rapamycin
- OC:
-
Ovarian cancer
- PC:
-
Pancreatic cancer
- PDAC:
-
Pancreatic ductal adenocarcinoma
- PI3K:
-
Phosphatidyl-inositol-3 kinase
- PI3KC3/BECN1:
-
PI3K-class 3/beclin 1
- PrPC :
-
Cellular prion protein
- PrPSc :
-
Scrapie prion protein
- RPSA:
-
Ribosomal protein SA
- TIMAP:
-
Transforming growth factor-β inhibited membrane associated protein
- TME:
-
Tumor microenvironment
References
Zanusso G, Liu D, Ferrari S, Hegyi I, Yin X, Aguzzi A, Hornemann S, Liemann S, Glockshuber R, Manson JC et al (1998) Prion protein expression in different species: analysis with a panel of new mAbs. Proc Natl Acad Sci USA 95:8812–8816. https://doi.org/10.1073/pnas.95.15.8812
Aguzzi A, Heppner FL (2000) Pathogenesis of prion diseases: a progress report. Cell Death Differ 7:889–902. https://doi.org/10.1038/sj.cdd.4400737
Sarnataro D, Pepe A, Zurzolo C (2017) Cell biology of prion protein. Progr Mol Biol Transl Sci 150:57–82. https://doi.org/10.1016/bs.pmbts.2017.06.018
Hirsch TZ, Martin-Lannerée S, Mouillet-Richard S (2017) Functions of the prion protein. Prog Mol Biol Transl Sci 150:1–34. https://doi.org/10.1016/bs.pmbts.2017.06.001
Linsenmeier L, Altmeppen HC, Wetzel S, Mohammadi B, Sftig P, Glatzel M (2017) Diverse functions of the prion protein—does proteolytic processing hold the key? Biochim Acta Mol Cell Res 1864:2128–2137. https://doi.org/10.1016/j.bbamcr.2017.06.022
Tohgo A, Takasawa S, Munakata H, Yonekura H, Hayashi N, Okamoto H (1994) Structural determination and characterization of a 40 kDa protein isolated from rat 40 S ribosomal subunit. FEBS Lett 340:133–138. https://doi.org/10.1016/0014-5793(94)80188-6
Gauczynski S, Peyrin JM, Haïk S, Leucht C, Hundt C, Rieger R, Krasemann S, Deslys JP, Dormont D, Lasmézas CI et al (2001) The 37-kDa/67-kDa laminin receptor acts as the cell-surface receptor for the cellular prion protein. EMBO J 20:5863–5875. https://doi.org/10.1093/emboj/20.21.5863
Jovanovic K, Chetty CJ, Khumalo T, Da Costa DB, Ferreira E, Malindisa ST, Caveney R, Letsolo BT, Weiss SF (2015) Novel patented therapeutic approaches targeting the 37/67 kDa laminin receptor for treatment of cancer and Alzheimer’s disease. Expert Opin Ther Pat 25:567–582. https://doi.org/10.1517/13543776.2015.1014802
Hundt C, Peyrin JM, Haïk S, Gauczynski S, Leucht C, Rieger R, Riley ML, Deslys JP, Dormont D, Lasmézas CI et al (2001) Identification of interaction domains of the prion protein with its 37-kDa/67-kDa laminin receptor. EMBO J 20:5876–5886. https://doi.org/10.1093/emboj/20.21.5876
DiGiacomo V, Meruelo D (2016) Looking into laminin receptor: critical discussion regarding the non-integrin 37/67-kDa laminin receptor/RPSA protein. Biol Rev Camb Philos Soc 91:288–310. https://doi.org/10.1111/brv.12170
Christen B, Damberger FF, Perez DR, Hornemann S, Wuthich K (2013) Structural plasticity of the cellular prion protein and implications in health and disease. Proc Natl Acad Sci USA 110:8549–8554. https://doi.org/10.1073/pnas.1306178110
Go G, Lee SH (2020) The cellular prion protein: a promising therapeutic target for cancer. J Mol Sci 21:9208. https://doi.org/10.3390/ijms21239208
Han H, Bearss DJ, Browne LW, Calaluce R, Nagle RB, Von Hoff DD (2002) Identification of differentially expressed genes in pancreatic cancer cells using cDNA microarray. Cancer Res 62:2890–2896
Ding M, Chen Y, Lang Y, Cui L (2021) The role of cellular prion protein in cancer biology: a potential therapeutic target. Front Oncol 11:742949. https://doi.org/10.3389/fonc.2021.742949
Mouillet-Richard S, Ghazi A, Laurent-Puig P (2021) The cellular prion protein and the hallmarks of cancer. Cancers (Basel) 13:5032. https://doi.org/10.3390/cancers13195032
Ryskalin L, Biagioni F, Busceti CL, Giambelluca MA, Morelli L, Frati A, Fornai F (2021) The role of cellular prion protein in promoting stemness and differentiation in cancer. Cancers (Basel) 13:170. https://doi.org/10.3390/cancers13020170
Barbieri G, Palumbo S, Gabrusiewicz K, Azzalin A, Marchesi N, Spedito A, Biggiogera M, Sbalchiero E, Mazzini G, Miracco C et al (2011) Silencing of cellular prion protein (PrPC) expression by DNA-antisense oligonucleotides induces autophagy-dependent cell death in glioma cells. Autophagy 7:840–853. https://doi.org/10.4161/auto.7.8.15615
Kim YC, Won SY, Jeong BH (2020) Identification of prion disease-related somatic mutations in the prion protein gene (PRNP) in cancer patients. Cells 9:1480. https://doi.org/10.3390/cells9061480
Ivanova L, Barmada S, Kummer T, Harris DA (2001) Mutant prion proteins are partially retained in the endoplasmic reticulum. J Biol Chem 276:42409–42421. https://doi.org/10.1074/jbc.M106928200
Campana V, Sarnataro D, Fasano C, Casanova P, Paladino S, Zurzolo C, (2006) Detergent-resistant membrane domains but not the proteasome are involved in the misfolding of a PrP mutant retained in the endoplasmic reticulum. J Cell Sci 119:433–442. https://doi.org/10.1242/jcs.02768
Campana V, Caputo A, Sarnataro D, Paladino S, Tivodar S, Zurzolo, (2007) Characterization of the properties and trafficking of an anchorless form of the prion protein. J Biol Chem 282:22747–22756. https://doi.org/10.1074/jbc.M701468200
Wang ZY, Shi Q, Wang SB, Tian C, Xu Y, Guo Y, Chen C, Zhang J, Dong XP (2013) Co-expressions of casein kinase 2 (CK2) subunits restore the down-regulation of tubulin levels and disruption of microtubule structures caused by PrP mutants. J Mol Neurosci 50:14–22. https://doi.org/10.1007/s12031-012-9845-y
Zhou L, Shang Y, Liu C, Li J, Hu H, Liang C, Han Y, Zhang W, Liang J, Wu K (2014) Overexpression of PrPc, combined with MGr1-Ag/37LRP, is predictive of poor prognosis in gastric cancer. Int J Cancer 135:2329–2337. https://doi.org/10.1002/ijc.28883
Rieger R, Edenhofer F, Lasmezas CI, Weiss S (1997) The human 37-kDa laminin receptor precursor interacts with the prion protein in eukaryotic cells. Nat Med 3:1383–1388. https://doi.org/10.1038/nm1297-1383
Morel E, Andrieu T, Casagrande F, Gauczynski S, Weiss S, Grassi J, Rousset M, Dormont D, Chambaz J (2005) Bovine prion is endocytosed by human enterocytes via the 37 kDa/67 kDa laminin receptor. Am J Pathol 167:1033–1042. https://doi.org/10.1016/S0002-9440(10)61192-3
Kolodziejczak D, Da Costa DB, Zuber C, Jovanovic K, Omar A, Beck J, Vana K, Mbazima V, Richt J, Brenig B et al (2010) Prion interaction with the 37-kDa/67-kDa laminin receptor on enterocytes as a cellular model for intestinal uptake of prions. J Mol Biol 402:293–300. https://doi.org/10.1016/j.jmb.2010.06.055
Pesapane A, Di Giovanni C, Rossi FW, Alfano D, Formisano L, Ragno P, Selleri C, Montuori N, Lavecchia A (2015) Discovery of new small molecules inhibiting 67 kDa laminin receptor interaction with laminin and cancer cell invasion. Oncotarget 6:18116–18133. https://doi.org/10.18632/oncotarget.4016
Sarnataro D, Pepe A, Altamura G, De Simone I, Pesapane A, Nitsch L, Montuori N, Lavecchia A, Zurzolo C (2016) The 37/67 kDa laminin receptor (LR) inhibitor, NSC47924, affects 37/67 kDa LR cell surface localization and interaction with the cellular prion protein. Sci Rep 6:24457. https://doi.org/10.1038/srep24457
Hinton C, Antony H, Hashimi SM, Munn A, Wei MQ (2013) Significance of prion and prion-like proteins in cancer development, progression and multi-drug resistance. Curr Cancer Drug Targets 13:895–904. https://doi.org/10.2174/156800961131300092
Lin SC, Lin CH, Shih NC, Liu HL, Wang WC, Lin KY, Liu ZY, Tseng YJ, Chang HK, Lin YC et al (2020) Cellular prion protein transcriptionally regulated by NFIL3 enhances lung cancer cell lamellipodium formation and migration through JNK signaling. Oncogene 39:385–398. https://doi.org/10.1038/s41388-019-0994-0
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
Hanahan D (2022) Hallmarks of cancer: new dimensions. Cancer Discov 12:31–46. https://doi.org/10.1158/2159-8290.CD-21-1059
Mehrpour M, Codogno P (2010) Prion protein: from physiology to cancer biology. Cancer Lett 290:1–23. https://doi.org/10.1016/j.canlet.2009.07.009
Shafiq M, Da Vela S, Amin L, Younas N, Harris DA, Zerr I, Altmeppen HC, Svergun D, Glatzel M (2022) The prion protein and its ligands: Insights into structure-function relationships. Biochim Biophys Acta Mol Cell Res 1869:119240. https://doi.org/10.1016/j.bbamcr.2022.119240
Liu P, Cheng H, Roberts TM, Zhao JJ (2009) Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov 8:627–644. https://doi.org/10.1038/nrd2926
Liang J, Pan Y, Zhang D, Guo C, Shi Y, Wang J, Chen Y, Wang X, Liu J, Guo X et al (2007) Cellular prion protein promotes proliferation and G1/S transition of human gastric cancer cells SGC7901 and AGS. FASEB J 21:2247–2256. https://doi.org/10.1096/fj.06-7799com
Zhuang D, Liu Y, Mao Y, Gao L, Zhang H, Luan S, Huang F, Li Q (2012) TMZ-induced PrPc/par-4 interaction promotes the survival of human glioma cells. Int J Cancer 130:309–318. https://doi.org/10.1002/ijc.25985
Li C, Yu S, Nakamura F, Yin S, Xu J, Petrolla AA, Singh N, Tartakoff A, Abbott DW, Xin W et al (2009) Binding of pro-prion to filamin A disrupts cytoskeleton and correlates with poor prognosis in pancreatic cancer. J Clin Invest 119(9):2725–2736. https://doi.org/10.1172/JCI39542
Wang Y, Yu S, Huang D, Cui M, Hu H, Zhang L, Wang W, Parameswaran N, Jackson M, Osborne B et al (2016) Cellular prion protein mediates pancreatic cancer cell survival and invasion through association with and enhanced signaling of notch1. Am J Pathol 186:2945–2956. https://doi.org/10.1016/j.ajpath.2016.07.010
Lopes MH, Santos TG, Rodrigues BR, Queiroz-Hazarbassanov N, Cunha IW, Wasilewska-Sampaio AP, Costa-Silva B, Marchi FA, Bleggi-Torres LF, Sanematsu PI et al (2015) Disruption of prion protein-HOP engagement impairs glioblastoma growth and cognitive decline and improves overall survival. Oncogene 34:3305–3314. https://doi.org/10.1038/onc.2014.261
Bianchini M, Giambelluca MA, Scavuzzo MC, Di Franco G, Guadagni S, Palmeri M, Furbetta N, Gianardi D, Funel N, Ricci C et al (2021) Detailing the ultrastructure’s increase of prion protein in pancreatic adenocarcinoma. World J Gastroenterol 27:7324–7339. https://doi.org/10.3748/wjg.v27.i42.7324
Bianchini M, Giambelluca MA, Scavuzzo MC, Di Franco G, Guadagni S, Palmeri M, Furbetta N, Gianardi D, Funel N, Pollina LE et al (2020) The occurrence of prion protein in surgically resected pancreatic adenocarcinoma. Pancreatology 20:1218–1225. https://doi.org/10.1016/j.pan.2020.08.004
Mouillet-Richard S, Martin-Lannerée S, Le Corre D, Hirsch TZ, Ghazi A, Sroussi M, Pilati C, de Reyniès A, Djouadi F, Vodovar N et al (2022) A proof of concept for targeting the PrPC - Amyloid β peptide interaction in basal prostate cancer and mesenchymal colon cancer. Oncogene 41:4397–4404. https://doi.org/10.1038/s41388-022-02430-7
Bhattacharya A, Izzo A, Mollo N, Napolitano F, Limone A, Margheri F, Mocali A, Minopoli G, Lo Bianco A, Di Maggio F et al (2020) Inhibition of 37/67kDa laminin-1 receptor restores APP maturation and reduces amyloid-β in human skin fibroblasts from familial Alzheimer’s disease. J Pers Med 10:232. https://doi.org/10.3390/jpm10040232
Hu K, Zhang X, Zhou L, Li J (2022) Downregulated prnp facilitates cell proliferation and invasion and has effect on the immune regulation in ovarian cancer. J Immunol Res 2022:3205040. https://doi.org/10.1155/2022/3205040.eCollection
Du L, Rao G, Wang H, Li B, Tian W, Cui J, He L, Laffin B, Tian X, Hao C et al (2013) CD44-positive cancer stem cells expressing cellular prion protein contribute to metastatic capacity in colorectal cancer. Cancer Res 73:2682–2694. https://doi.org/10.1158/0008-5472.CAN-12-3759
Corsaro A, Bajetto A, Thellung S, Begani G, Villa V, Nizzari M, Pattarozzi A, Solari A, Gatti M, Pagano A et al (2016) Cellular prion protein controls stem cell-like properties of human glioblastoma tumor-initiating cells. Oncotarget 7:38638–38657. https://doi.org/10.18632/oncotarget.9575
Iglesia RP, Prado MB, Cruz L, Martins VR, Santos TG, Lopes MH (2017) Engagement of cellular prion protein with the co-chaperone Hsp70/90 organizing protein regulates the proliferation of glioblastoma stem-like cells. Stem Cell Res Ther 8:76. https://doi.org/10.1186/s13287-017-0518-1
Cloutier G, Sallenbach-Morrissette A, Beaulieu JF (2019) Non-integrin laminin receptors in epithelia. Tissue Cell 56:71–78. https://doi.org/10.1016/j.tice.2018.12.005
Vania L, Morris G, Otgaar TC, Bignoux MJ, Bernert M, Burns J, Gabathuse A, Singh E, Ferreira E, Weiss SFT (2019) Patented therapeutic approaches targeting LRP/LR for cancer treatment. Expert Opin Ther Pat 29:987–1009. https://doi.org/10.1080/13543776.2019.1693543
Khumalo T, Ferreira E, Jovanovic K, Veale RB, Weiss SF (2015) Knockdown of LRP/LR induces apoptosis in breast and oesophageal cancer cells. PLoS ONE 10:e0139584. https://doi.org/10.1371/journal.pone.0139584
Chetty CJ, Ferreira E, Jovanovic K, Weiss SFT (2017) Knockdown of LRP/LR induces apoptosis in pancreatic cancer and neuroblastoma cells through activation of caspases. Exp Cell Res 360:264–272. https://doi.org/10.1016/j.yexcr.2017.09.016
Rebelo TM, Vania L, Ferreira E, Weiss SFT (2018) siRNA - Mediated LRP/LR knock-down reduces cellular viability of malignant melanoma cells through the activation of apoptotic caspases. Exp Cell Res 368:1–12. https://doi.org/10.1016/j.yexcr.2018.04.003
Vania L, Morris G, Ferreira E, Weiss SFT (2021) Knock-down of LRP/LR influences signalling pathways in late-stage colorectal carcinoma cells. BMC Cancer 21:392. https://doi.org/10.1186/s12885-021-08081-3
Salama RH, Muramatsu H, Zou K, Inui T, Kimura T, Muramatsu T (2001) Midkine binds to 37-kDa laminin binding protein precursor, leading to nuclear transport of the complex. Exp Cell Res 270:13–20. https://doi.org/10.1006/excr.2001.5341
Sun L, Liu L, Liu X, Wang Y, Li M, Yao L, Yang J, Ji G, Guo C, Pan Y et al (2014) MGr1-Ag/37LRP induces cell adhesion-mediated drug resistance through FAK/PI3K and MAPK pathway in gastric cancer. Cancer Sci 105:651–659. https://doi.org/10.1111/cas.12414
Lu CL, Xu J, Yao HJ, Luo KL, Li JM, Wu T, Wu GZ (2016) Inhibition of human 67-kDa laminin receptor sensitizes multidrug resistance colon cancer cell line SW480 for apoptosis induction. Tumor Biol 37:1319–1325. https://doi.org/10.1007/s13277-015-3873-5
Brabletz T, Kalluri R, Nieto MA, Weinberg RA (2018) EMT in cancer. Nat Rev Cancer 18:128–134. https://doi.org/10.1038/nrc.2017.118
Sarnataro D, Campana V, Paladino S, Stornaiuolo M, Nitsch L, Zurzolo C (2004) PrP(C) association with lipid rafts in the early secretory pathway stabilizes its cellular conformation. Mol Biol Cell 15:4031–4042. https://doi.org/10.1091/mbc.e03-05-0271
Zhou J, Kang X, An H, Lv Y, Liu X (2021) The function and pathogenic mechanism of filamin A. Gene 784:145575. https://doi.org/10.1016/j.gene.2021.145575
Li C, Yu S, Nakamura F, Pentikäinen OT, Singh N, Yin S, Xin W, Sy MS (2010) Pro-prion binds filamin A, facilitating its interaction with integrin beta1, and contributes to melanomagenesis. J Biol Chem 285:30328–30339. https://doi.org/10.1074/jbc.M110.147413
Li H, Zhang J, Ke JR, Yu Z, Shi R, Gao SS, Li JF, Gao ZX, Ke CS, Han HX et al (2022) Pro-prion, as a membrane adaptor protein for E3 ligase c-Cbl, facilitates the ubiquitination of IGF-1R, promoting melanoma metastasis. Cell Rep 41:111834. https://doi.org/10.1016/j.celrep.2022.111834
Li J, Zhao W, Akbani R, Liu W, Ju Z, Ling S, Vellano CP, Roebuck P, Yu Q, Eterovic AK et al (2017) Characterization of human cancer cell lines by reverse-phase protein arrays. Cancer Cell 31:225–239. https://doi.org/10.1016/j.ccell.2017.01.005
Kleffman K, Levinson G, Rose IVL, Blumenberg LM, Shadaloey SAA, Dhabaria A, Wong E, Galán-Echevarría F, Karz A, Argibay D et al (2022) Melanoma-secreted amyloid beta suppresses neuroinflammation and promotes brain metastasis. Cancer Disco 12:1314–1335. https://doi.org/10.1158/2159-8290.CD-21-1006
Pan Y, Zhao L, Liang J, Liu J, Shi Y, Liu N, Zhang G, Jin H, Gao J, Xie H et al (2006) Cellular prion protein promotes invasion and metastasis of gastric cancer. FASEB J 20:1886–1888. https://doi.org/10.1096/fj.06-6138fje
Huang C, Jacobson K, Schaller MD (2004) MAP kinases and cell migration. J Cell Sci 117:4619–4628. https://doi.org/10.1242/jcs.01481
Gil M, Kim YK, Kim KE, Kim W, Park CS, Lee KJ (2016) Cellular prion protein regulates invasion and migration of breast cancer cells through MMP-9 activity. Biochem Biophys Res Commun 470:213–219. https://doi.org/10.1016/j.bbrc.2016.01.038
Le Corre D, Ghazi A, Balogoun R, Pilati C, Aparicio T, Martin-Lannerée S, Marisa L, Djouadi F, Poindessous V, Crozet C et al (2019) The cellular prion protein controls the mesenchymal-like molecular subtype and predicts disease outcome in colorectal cancer. EBioMedicine 46:94–104. https://doi.org/10.1016/j.ebiom.2019.07.036
Ghazi A, Le Corre D, Pilati C, Taieb J, Aparicio T, Didelot A, Dedhar S, Mulot C, Le Malicot K, Djouadi F et al (2021) Prognostic value of the PrPC-ILK-IDO1 axis in the mesenchymal colorectal cancer subtype. Oncoimmunology 10:1940674. https://doi.org/10.1080/2162402X.2021.1940674
Kalluri R, LeBleu VS (2020) The biology, function, and biomedical applications of exosomes. Science 367(6478):eaau6977. https://doi.org/10.1126/science.aau6977
Yun CW, Lee JH, Go G, Jeon J, Yoon S, Lee SH (2021) Prion protein of extracellular vesicle regulates the progression of colorectal cancer. Cancers (Basel) 13:2144. https://doi.org/10.3390/cancers13092144
Ardini E, Sporchia B, Pollegioni L, Modugno M, Ghirelli C, Castiglioni F, Tagliabue E, Ménard S (2002) Identification of a novel function for 67-kDa laminin receptor: increase in laminin degradation rate and release of motility fragments. Cancer Res 62:1321–1325
Pupa SM, Ménard S, Forti S, Tagliabue E (2002) New insights into the role of extracellular matrix during tumor onset and progression. J Cell Physiol 192:259–267. https://doi.org/10.1002/jcp.10142
Munien C, Rebelo TM, Ferreira E, Weiss SF (2017) IgG1-iS18 impedes the adhesive and invasive potential of early and late stage malignant melanoma cells. Exp Cell Res 351:135–141. https://doi.org/10.1016/j.yexcr.2017.01.009
Zuber C, Knackmuss S, Zemora G, Reusch U, Vlasova E, Diehl D, Mick V, Hoffmann K, Nikles D, Fröhlich T et al (2008) Invasion of tumorigenic HT1080 cells is impeded by blocking or downregulating the 37-kDa/67-kDa laminin receptor. J Mol Biol 378:530–539. https://doi.org/10.1016/j.jmb.2008.02.004
Omar A, Reusch U, Knackmuss S, Little M, Weiss SF (2012) Anti-LRP/LR-specific antibody IgG1-iS18 significantly reduces adhesion and invasion of metastatic lung, cervix, colon and prostate cancer cells. J Mol Biol 419:102–109. https://doi.org/10.1016/j.jmb.2012.02.035
Khumalo T, Reusch U, Knackmuss S, Little M, Veale RB, Weiss SFT (2013) Adhesion and invasion of breast and oesophageal cancer cells are impeded by anti-LRP/LR-specific antibody IgG1-iS18. PLoS ONE 8:e66297. https://doi.org/10.1371/journal.pone.0066297
Chetty C, Khumalo T, Da Costa Dias B, Reusch U, Knackmuss S, Little M, Weiss SF (2014) Anti-LRP/LR specific antibody IgG1-iS18 impedes adhesion and invasion of liver cancer cells. PLoS ONE 9:e96268. https://doi.org/10.1371/journal.pone.0096268
Bignoux MJ, Otgaar TC, Bernert M, Weiss SFT, Ferreira E (2023) Downregulation of LRP/LR with siRNA inhibits several cancer hallmarks in lung cancer cells. FEBS Open Bio 13:323–340. https://doi.org/10.1002/2211-5463.13544
Wu Y, Tan X, Liu P, Yang Y, Huang Y, Liu X, Meng X, Yu B, Wu M, Jin H (2019) ITGA6 and RPSA synergistically promote pancreatic cancer invasion and metastasis via PI3K and MAPK signaling pathways. Exp Cell Res 379:30–47. https://doi.org/10.1016/j.yexcr.2019.03.022
Gresseau L, Roy ME, Duhamel S, Annabi B (2022) A Signaling crosstalk links SNAIL to the 37/67 kDa Laminin-1 receptor ribosomal protein SA and regulates the acquisition of a cancer stem cell molecular signature in U87 glioblastoma neurospheres. Cancers (Basel) 14:5944. https://doi.org/10.3390/cancers14235944
Zuber C, Knackmuss S, Rey C, Reusch U, Röttgen P, Fröhlich T, Arnold GJ, Pace C, Mitteregger G, Kretzschmar HA et al (2008) Single chain Fv antibodies directed against the 37 kDa/67 kDa laminin receptor as therapeutic tools in prion diseases. Mol Immunol 45:144–151. https://doi.org/10.1016/j.molimm.2007.04.030
Luo G, Wang W, Wu Q, Lu Y, Su T, Gu N, Li K, Wang J, Du R, Zhao X et al (2017) MGr1-Antigen/37 kDa laminin receptor precursor promotes cellular prion protein induced multi-drug-resistance of gastric cancer. Oncotarget 8:71630–71641. https://doi.org/10.18632/oncotarget.17795
Provenzano L, Ryan Y, Hilton DA, Lyons-Rimmer J, Dave F, Maze EA, Adams CL, Rigby-Jones R, Ammoun S, Hanemann CO (2017) Cellular prion protein (PrPC) in the development of Merlin-deficient tumors. Oncogene 36:6132–6142. https://doi.org/10.1038/onc.2017.200
Guitart K, Loers G, Buck F, Bork U, Schachner M, Kleene R (2016) Improvement of neuronal cell survival by astrocyte-derived exosomes under hypoxic and ischemic conditions depends on prion protein. Glia 64(6):896–910. https://doi.org/10.1002/glia.22963
Bravard A, Auvré F, Fantini D, Bernardino-Sgherri J, Sissoëff L, Daynac M, Xu Z, Etienne O, Dehen C, Comoy E et al (2015) The prion protein is critical for DNA repair and cell survival after genotoxic stress. Nucleic Acids Res 43:904–916. https://doi.org/10.1093/nar/gku1342
Shyu WC, Lin SZ, Saeki K, Kubosaki A, Matsumoto Y, Onodera T, Chiang MF, Thajeb P, Li H (2004) Hyperbaric oxygen enhances the expression of prion protein and heat shock protein 70 in a mouse neuroblastoma cell line. Cell Mol Neurobiol 24:257–268. https://doi.org/10.1023/b:cemn.0000018620.41913.d2
Qin K, Zhao L, Ash RD, McDonough WF, Zhao RY (2009) ATM-mediated transcriptional elevation of prion in response to copper-induced oxidative stress. J Biol Chem 284:4582–4593. https://doi.org/10.1074/jbc.M808410200
Domingues PH, Nanduri LSY, Seget K, Venkateswaran SV, Agorku D, Viganó C, von Schubert C, Nigg EA, Swanton C, Sotillo R et al (2017) Cellular prion protein PrPC and Ecto-5’-Nucleotidase are markers of the cellular stress response to aneuploidy. Cancer Res 77:2914–2926. https://doi.org/10.1158/0008-5472.CAN-16-3052
Bernardino-Sgherri J, Siberchicot C, Auvré F, Busso D, Brocas C, El Masri G, Lioutsko A, Ferri F, Radicella JP, Romeo PH et al (2021) Tumor resistance to radiotherapy is triggered by an ATM/TAK1-dependent-increased expression of the cellular prion protein. Oncogene 40:3460–3469. https://doi.org/10.1038/s41388-021-01746-0
Manni G, Lewis V, Senesi M, Spagnolli G, Fallarino F, Collins SJ, Mouillet-Richard S, Biasini E (2020) The cellular prion protein beyond prion diseases. Swiss Med Wkly 150:w20222. https://doi.org/10.4414/smw.2020.20222
Guerra-Rebollo M, Mateo F, Franke K, Huen MS, Lopitz-Otsoa F, Rodríguez MS, Plans V, Thomson TM (2012) Nucleolar exit of RNF8 and BRCA1 in response to DNA damage. Exp Cell Res 318:2365–2376. https://doi.org/10.1016/j.yexcr.2012.07.003
Strom A, Wang GS, Picketts DJ, Reimer R, Stuke AW, Scott FW (2011) Cellular prion protein localizes to the nucleus of endocrine and neuronal cells and interacts with structural chromatin components. Eur J Cell Biol 90:414–419. https://doi.org/10.1016/j.ejcb.2010.11.015
Silva JL, Lima LM, Foguel D, Cordeiro Y (2008) Intriguing nucleic-acid-binding features of mammalian prion protein. Trends Biochem Sci 33:132–140. https://doi.org/10.1016/j.tibs.2007.11.003
Silva JL, Gomes MP, Vieira TC, Cordeiro Y (2010) PrP interactions with nucleic acids and glycosaminoglycans in function and disease. Front Biosci (Landmark edition) 15:132–150. https://doi.org/10.2741/3611
Bera A, Roche AC, Nandi PK (2007) Bending and unwinding of nucleic acid by prion protein. Biochemistry 46:1320–1328. https://doi.org/10.1021/bi0620050
Mangé A, Crozet C, Lehmann S, Béranger F (2004) Scrapie-like prion protein is translocated to the nuclei of infected cells independently of proteasome inhibition and interacts with chromatin. J Cell Sci 117:2411–2416. https://doi.org/10.1242/jcs.01094
Kinoshita K, Kaneda Y, Sato M, Saeki Y, Wataya-Kaneda M, Hoffmann A (1998) LBP-p40 binds DNA tightly through associations with histones H2A, H2B, and H4. Biohem Biophys Res Comm 253:277–282. https://doi.org/10.1006/bbrc.1998.9699
Venticinque L, Meruelo D (2012) Comprehensive proteomic analysis of nonintegrin laminin receptor interacting proteins. J Proteome Res 11:4863–4872. https://doi.org/10.1021/pr300307h
Sarnataro D (2018) Attempt to untangle the prion-like misfolding mechanism for neurodegenerative diseases. Int J Mol Sci 19:3081. https://doi.org/10.3390/ijms19103081
Wickner RB, Shewmaker F, Edskes H, Kryndushkin D, Nemecek J, McGlinchey R, Bateman D, Winchester CL (2010) Prion amyloid structure explains templating: how proteins can be genes. FEMS Yeast Res 10:980–991. https://doi.org/10.1111/j.1567-1364.2010.00666.x
Pan KM, Baldwin M, Nguyen J, Gasset M, Serban A, Groth D, Mehlhorn I, Huang Z, Fletterick RJ, Cohen FE et al (1993) Conver- sion of alpha-helices into beta-sheets features in the formation of the scrapie prion proteins. Proc Natl Acad Sci U SA 90:10962–10966. https://doi.org/10.1073/pnas.90.23.10962
King CY (2001) Supporting the structural basis of prion strains: induction and identification of [PSI] variants. J Mol Biol 307:1247–1260. https://doi.org/10.1006/jmbi.2001.4542
Andreychuk YV, Zadorsky SP, Zhuk AS, Stepchenkova EI, Inge-Vechtomov SG (2020) Relationship between type I and type II template processes: amyloids and genome stability. Mol Biol (Mosk) 54:750–775. https://doi.org/10.31857/S002689842005002X
Pepe A, Avolio R, Matassa DS, Esposito F, Nitsch L, Zurzolo C, Paladino S, Sarnataro D (2017) Regulation of sub-compartmental targeting and folding properties of the prion-like protein Shadoo. Sci Rep 7:3731. https://doi.org/10.1038/s41598-017-03969-2
Molina-García L, Gasset-Rosa F, Moreno-Del Álamo M, Fernández-Tresguerres ME, Moreno-Díaz de la Espina S, Lurz R, Giraldo R (2016) Functional amyloids as inhibitors of plasmid DNA replication. Sci Rep 6:25425. https://doi.org/10.1038/srep25425
Basu J, Williams BC, Li Z, Williams EV, Gold-Berg ML (1998) Depletion of a Drosophila homolog of yeast Sup35p disrupts spindle assembly, chromosome segregation, and cytokinesis during male meiosis. Cell Motil Cytoskeleton 39:286–302. https://doi.org/10.1002/(SICI)1097-0169(1998)39:4%3c286::AID-CM4%3e3.0.CO;2-1
Borchsenius AS, Tchourikova AA, Inge-Vechtomov SG (2000) Recessive mutations in SUP35 and SUP45 genes coding for translation release factors affect chromosome stability in Saccharomyces cerevisiae. Curr Genet 37:285–291. https://doi.org/10.1007/s002940050529
Nieznanski K, Nieznanska H, Skowronek KJ, Osiecka KM, Stepkowski D (2005) Direct interaction between prion protein and tubulin. Biochem Biophys Res Commun 334:403–411. https://doi.org/10.1016/j.bbrc.2005.06.092
Nieznanski K, Podlubnaya ZA, Nieznanska H (2006) Prion protein inhibits microtubule assembly by inducing tubulin oligomerization. Biochem Biophys Res Commun 349:391–399. https://doi.org/10.1016/j.bbrc.2006.08.051
Venticinque L, Jamieson KV, Meruelo D (2011) Interactions between laminin receptor and the cytoskeleton during translation and cell motility. PLoS ONE 6:e15895. https://doi.org/10.1371/journal.pone.0015895
Davoli T, Denchi EL, de Lange T (2010) Persistent telomere damage induces bypass of mitosis and tetraploidy. Cell 141:81–93. https://doi.org/10.1016/j.cell.2010.01.031
Davoli T, de Lange T (2012) Telomere-driven tetraploidization occurs in human cells undergoing crisis and promotes transformation of mouse cells. Cancer Cell 21:765–776. https://doi.org/10.1016/j.ccr.2012.03.044
Harley CB, Villeponteau B (1995) Telomeres and telomerase in aging and cancer. Curr Opin Genet Dev 5:249–255. https://doi.org/10.1016/0959-437x(95)80016-6
Maciejowski J, Li Y, Bosco N, Campbell PJ, de Lange T (2015) Chromothripsis and kataegis induced by telomere crisis. Cell 163:1641–1654. https://doi.org/10.1016/j.cell.2015.11.054
Saretzki G (2014) Extra-telomeric functions of human telomerase: cancer, mitochondria and oxidative stress. Curr Pharm Des 20:6386–6403. https://doi.org/10.2174/1381612820666140630095606
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL, Coviello GM, Wright WE, Weinrich SL, Shay JW (1994) Specific association of human telomerase activity with immortal cells and cancer. Science 266:2011–2015. https://doi.org/10.1126/science.7605428
Zhang PH, Zou L, Tu ZG (2006) RNAi-hTERT inhibition hepatocellular carcinoma cell proliferation via decreasing telomerase activity. J Surg Res 131:143–149. https://doi.org/10.1016/j.jss.2005.09.017
Maggisano V, Celano M, Lombardo GE, Lepore SM, Sponziello M, Rosignolo F, Verrienti A, Baldan F, Puxeddu E, Durante C et al (2017) Silencing of hTERT blocks growth and migration of anaplastic thyroid cancer cells. Mol Cell Endocrinol 448:34–40. https://doi.org/10.1016/j.mce.2017.03.007
Lombardo GE, Maggisano V, Celano M, Cosco D, Mignogna C, Baldan F, Lepore SM, Allegri L, Moretti S, Durante C et al (2018) Anti-hTERT siRNA-loaded nanoparticles block the growth of anaplastic thyroid cancer xenograft. Mol Cancer Ther 17:1187–1195. https://doi.org/10.1158/1535-7163.MCT-17-0559
Naidoo K, Malindisa ST, Otgaar TC, Bernert M, Da Costa Dias B, Ferreira E, Reusch U, Knackmuss S, Little M et al (2015) Knock-down of the 37kDa/67kDa laminin receptor LRP/LR impedes telomerase activity. PLoS ONE 10:e0141618. https://doi.org/10.1371/journal.pone.0141618
Otgaar TC, Ferreira E, Malindisa S, Bernert M, Letsolo BT, Weiss SFT (2017) 37 kDa LRP::FLAG enhances telomerase activity and reduces senescent markers in vitro. Oncotarget 8:86646–86656. https://doi.org/10.18632/oncotarget.21278
Bignoux MJ, Cuttler K, Otgaar TC, Ferreira E, Letsolo BT, Weiss SFT (2019) LRP::FLAG rescues cells from Amyloid-β-Mediated cytotoxicity through increased TERT levels and telomerase activity. J Alzheimers Dis 69:729–741. https://doi.org/10.3233/JAD-190075
Vania L, Rebelo TM, Ferreira E, Weiss SFT (2018) Knock-down of LRP/LR promotes apoptosis in early and late stage colorectal carcinoma cells via caspase activation. BMC Cancer 18:602. https://doi.org/10.1186/s12885-018-4531-2
Zhao Y, You H, Liu F, An H, Shi Y, Yu Q, Fan D (2002) Differentially expressed gene profiles between multidrug resistant gastric adenocarcinoma cells and their parental cells. Cancer Lett 185:211–218. https://doi.org/10.1016/s0304-3835(02)00264-1
Yun CW, Jeon J, Go G, Lee JH, Lee SH (2020) The dual role of autophagy in cancer development and a therapeutic strategy for cancer by targeting autophagy. Int J Mol Sci 22(1):179. https://doi.org/10.3390/ijms22010179
Oh JM, Shin HY, Park SJ, Kim BH, Choi JK, Choi EK, Carp RI, Kim YS (2008) The involvement of cellular prion protein in the autophagy pathway in neuronal cells. Mol Cell Neurosci 39:238–247
Oh JM, Choi EK, Carp RI, Kim YS (2012) Oxidative stress impairs autophagic flux in prion protein-deficient hippocampal cells. Autophagy 8:1448–1461. https://doi.org/10.4161/auto.21164
Santos TG, Lopes MH, Martins VR (2015) Targeting prion protein interactions in cancer. Prion 9:165–173. https://doi.org/10.1080/19336896.2015.1027855
Akhavan D, Cloughesy TF, Mischel PS (2010) mTOR signaling in glioblastoma: lessons learned from bench to bedside. Neuro Oncol 12:882–889. https://doi.org/10.1093/neuonc/noq052
Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Siu IM, Gallia GL et al (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321:1807–1812. https://doi.org/10.1126/science.1164382
Armocida D, Busceti CL, Biagioni F, Fornai F, Frati A (2023) The role of cellular prion protein in glioma tumorigenesis could be through the autophagic mechanisms: a narrative review. Int J Mol Sci 2:1405. https://doi.org/10.3390/ijms24021405
Lenzi P, Busceti CL, Lazzeri G, Ferese R, Biagioni F, Salvetti A, Pompili E, De Franchis V, Puglisi-Allegra S, Frati A et al (2023) Autophagy activation associates with suppression of prion protein and improved mitochondrial status in glioblastoma cells. Cells 12:221. https://doi.org/10.3390/cells12020221
Limone A, Veneruso I, Izzo A, Renna M, Bonavita R, Piscitelli S, Calì G, De Nicola S, Riccio P, D’Argenio V et al (2022) Activation of non-canonical autophagic pathway through inhibition of non-integrin laminin receptor in neuronal cells. Cells 11:466. https://doi.org/10.3390/cells11030466
Limone A, Veneruso I, D’Argenio V, Sarnataro D (2022) Endosomal trafficking and related genetic underpinnings as a hub in Alzheimer’s disease. J Cell Physiol 237:3803–3815. https://doi.org/10.1002/jcp.30864
Florey O, Eun KS, Sandoval CP, Haynes CM, Overholtzer M (2011) Autophagy machinery mediates macroendocytic processing and entotic cell death by targeting single membranes. Nat Cell Biol 13:1335–1343. https://doi.org/10.1038/ncb2363
Florey O, Overholtzer M (2012) Autophagy proteins in macroendocytic engulfment. Trends Cell Biol 22:374–380. https://doi.org/10.1016/j.tcb.2012.04.005
Jacquin E, Leclerc-Mercier S, Judon C, Blanchard E, Fraitag S, Florey O (2017) Pharmacological modulators of autophagy activate a parallel noncanonical pathway driving unconventional LC3 lipidation. Autophagy 13:854–867. https://doi.org/10.1080/15548627.2017.1287653
Puri C, Renna M, Bento CF, Moreau K, Rubinsztein DC (2013) Diverse autophagosome membrane sources coalesce in recycling endosomes. Cell 154:1285–1299. https://doi.org/10.1016/j.cell.2013.08.044
Domogatskaya A, Rodin S, Tryggvason K (2012) Functional diversity of laminins. Annu Rev Cell Dev Biol 28:523–553. https://doi.org/10.1146/annurev-cellbio-101011-155750
Gopalakrishna R, Gundimeda U, Zhou S, Bui H, Davis A, McNeill T, Mack W (2018) Laminin-1 induces endocytosis of 67KDa laminin receptor and protects Neuroscreen-1 cells against death induced by serum withdrawal. Biochem Biophys Res Commun 495:230–237. https://doi.org/10.1016/j.bbrc.2017.11.015
Bhattacharya A, Limone A, Napolitano F, Cerchia C, Parisi S, Minopoli G, Montuori N, Lavecchia A, Sarnataro D (2020) APP maturation and intracellular localization are controlled by a specific inhibitor of 37/67 kDa Laminin-1 receptor in neuronal cells. Int J Mol Sci 21:1738. https://doi.org/10.3390/ijms21051738
Martinez-lopez N, Athonvarangkul D, Mishall P, Sahu S, Singh R (2013) Autophagy proteins regulate ERK phosphorylation. Nat Commun 4:2799. https://doi.org/10.1038/ncomms3799
Barrow-McGee R, Kishi N, Joffre C, Ménard L, Hervieu A, Bakhouche BA, Noval AJ, Mai A, Guzmán C, Robbez-Masson L et al (2016) Beta 1-integrin-c-Met cooperation reveals an inside-in survival signalling on autophagy-related endomembranes. Nat Commun 7:11942. https://doi.org/10.1038/ncomms11942
Givant-Horwitz V, Davidson B, Reich R (2004) Laminin-induced signaling in tumor cells. Cancer Res 64:3572–3579. https://doi.org/10.1158/0008-5472.CAN-03-3424
Shmerling D, Hegyi I, Fischer M, Blättler T, Brandner S, Götz J, Rülicke T, Flechsig E, Cozzio A, von Mering C, Hangartner C et al (1998) Expression of amino-terminally truncated PrP in the mouse leading to ataxia and specific cerebellar lesions. Cell 93:203–214. https://doi.org/10.1016/s0092-8674(00)81572-x
Baumann F, Tolnay M, Brabeck C, Pahnke J, Kloz U, Niemann HH, Heikenwalder M, Rülicke T, Bürkle A, Aguzzi A (2007) Lethal recessive myelin toxicity of prion protein lacking its central domain. EMBO J 26:538–547. https://doi.org/10.1038/sj.emboj.7601510
Grimaldi I, Leser FS, Janeiro JM, da Rosa BG, Campanelli AC, Romão L, Lima FRS (2022) The multiple functions of PrPC in physiological, cancer, and neurodegenerative contexts. J Mol Med (Berl) 100(10):1405–1425. https://doi.org/10.1007/s00109-022-02245-9
Pesapane A, Ragno P, Selleri C, Montuori N (2017) Recent advances in the function of the 67 kDa laminin receptor and its targeting for personalized therapy in cancer. Curr Pharm Des 23:4745–4757. https://doi.org/10.2174/1381612823666170710125332
Uversky VN (2013) The most important thing is the tail: multitudinous functionalities of intrinsically disordered protein termini. FEBS Lett 587:1891–1901. https://doi.org/10.1016/j.febslet.2013.04.042
Acknowledgements
The authors would like to thank all of those whose fruitful research has contributed in any way to the elucidation of the role of PrPC and RPSA in cancer pathogenesis and therapy.
Funding
Open access funding provided by Università degli Studi di Napoli Federico II within the CRUI-CARE Agreement. The author(s) received financial support for the authorship and publication of this article by the contract (so-called "transformative") negotiated by CARE–CRUI and signed by the “Federico II” University Center for Libraries, Italy.
Author information
Authors and Affiliations
Contributions
Conceptualization and design: DS and SB. Writing of the first draft: DS, SB, VM, AL. Figures: AL and VM. Final editing of the text: DS and SB.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors declare a conflict of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Limone, A., Maggisano, V., Sarnataro, D. et al. Emerging roles of the cellular prion protein (PrPC) and 37/67 kDa laminin receptor (RPSA) interaction in cancer biology. Cell. Mol. Life Sci. 80, 207 (2023). https://doi.org/10.1007/s00018-023-04844-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04844-2