Abstract
The discoveries that cerebrospinal fluid participates in metabolic perivascular exchange with the brain and further drains solutes to meningeal lymphatic vessels have sparked a tremendous interest in translating these seminal findings from animals to humans. A potential two-way coupling between the brain extra-vascular compartment and the peripheral immune system has implications that exceed those concerning neurodegenerative diseases, but also imply that the central nervous system has pushed its immunological borders toward the periphery, where cross-talk mediated by cerebrospinal fluid may play a role in a range of neoplastic and immunological diseases. Due to its non-invasive approach, magnetic resonance imaging has typically been the preferred methodology in attempts to image the glymphatic system and meningeal lymphatics in humans. Even if flourishing, the research field is still in its cradle, and interpretations of imaging findings that topographically associate with reports from animals have yet seemed to downplay the presence of previously described anatomical constituents, particularly in the dura. In this brief review, we illuminate these challenges and assess the evidence for a glymphatic-lymphatic coupling. Finally, we provide a new perspective on how human brain and meningeal clearance function may possibly be measured in future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
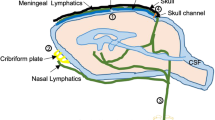
The study of lymphatic vessels in dura mater has a long history, but in recent time, this was facilitated by the introduction of specific markers for lymphatic endothelial cells [1]. In 2015, animal studies unveiled for the first time that tracer in cerebrospinal fluid (CSF) drains directly into lymphatic vessels within the dura, primarily alongside the dural venous sinuses [2, 3]. It was furthermore demonstrated that meningeal lymphatic drainage assumes a pivotal role not only in eliminating solutes from the subarachnoid space (SAS), but also from the brain itself, implying that brain solute clearance along perivascular pathways is dependent on meningeal lymphatic vasculature. As such, later studies showed that experimental ablation of meningeal lymphatic drainage was followed by delayed clearance of both tau [4] and amyloid-beta [5] from the animal brain.
In this review, we provide as background a short summary of current, co-existing concepts for brain perivascular clearance, including the glymphatic system, which currently seems to have the largest momentum. Furthermore, we describe shortly main elements of the breakthrough research findings of dural lymphatic anatomy and function in rodents. In humans, we identify previous anatomical research on dura, particularly the parasagittal dura and its constituents. From this, we critically illuminate translational magnetic resonance imaging (MRI) investigations in humans that have endeavored to image the intricate interplay between brain perivascular drainage and lymphatic drainage from the craniospinal compartment. We particularly scrutinize potential pitfalls in the interpretation of applied MRI studies within this emerging research field. Finally, we recommend a new method to assess meningeal CSF clearance capacity in research and the clinic.
The glymphatic system and competing models
The initial elucidation of the glymphatic system in rodents [6] has significantly influenced imaging of human brain clearance along perivascular pathways over the past decade, fostering a burgeoning realm of translational research [7]. One of the main breakthrough observations in humans was that exchange between cerebrospinal fluid (CSF) and brain tissue is extensive and profound, and clearance from both CSF and brain differs between individuals as well as between patient groups, including those with idiopathic normal pressure hydrocephalus (iNPH)-associated dementia [8, 9]. Nevertheless, some unresolved inquiries remain. Presently, the extent to which perivascular solute clearance contributes to overall brain clearance remains ambiguous when juxtaposed with other well-established pathways, including solute excretion across the blood–brain-barrier (BBB), local proteolytic cleavage, and microglial resorption [10]. The role of impaired glymphatic function has been postulated as a causal factor and consequence within a spectrum of neurological disorders [11, 12]. However, the glymphatic paradigm faces direct contradictions from researchers attempting to reproduce the experimental framework [13] and is challenged by alternative hypotheses for perivascular clearance, such as intramural periarterial drainage (IPAD) and periarterial mixing [10, 14] (Fig. 1). Despite these variances, all competing frameworks now seem to converge on the consensus that the influx of cerebrospinal fluid (CSF) from the brain surface serves as the principal propellant within these pseudo-lymphatic clearance systems. In previous reports of the IPAD system, CSF influx along arteries was not supported, based on the belief that cortex lacked perivascular spaces [15], but this view seem now to have been abandoned. A shared characteristic is further the scarcity of evidence for a direct anatomical connection between the brain and lymphatic vessels at the meninges and, ultimately, the bloodstream.
Competing model frameworks for understanding perivascular clearance. All models currently accept CSF influx along penetrating arteries and mixing with the interstitial fluid, but drainage routes from brain differ substantially. In the glymphatic system (A), interstitial solutes are cleared along veins into CSF or directly to meningeal lymphatic vessels, in the IPAD model (B) drainage is within the artery wall, and in the mixing model (C), solutes diffuse along a concentration gradient along periarterial spaces back into CSF, facilitated by mixing from physiological motion. Illustration from Zhao et al. (Physiology) [14] (reuse by permission from CCC RightsLink® service)
In the primary conceptualization of the glymphatic system, the question of additional drainage routes from brain, whether directly into the subarachnoid space (SAS) or traversing cortical veins to eventual lymphatic vessels remained open. In the IPAD model [16], meningeal lymphatic vessels are by-passed, as solute drainage within the arterial wall muscle layer is supposed to persist uninterrupted and travel opposite of the blood stream within the carotid artery wall directly to cervical lymph nodes, albeit without direct evidence of such connections. Conversely, periarterial mixing entails solutes diffusing from the brain interstitium along their concentration gradient directly into periarterial spaces, where pulsations facilitate the retrograde pumping of solutes back into the SAS.
Curiously, if the influx of CSF into the brain is of paramount importance, the notion that brain waste is cleared back into CSF at the surface as to a common sink, and potentially recirculated into the brain, might appear counterintuitive. Therefore, the anticipation that brain perivascular drainage connects directly with meningeal lymphatic vessels has supposedly influenced interpretation of imaging findings from the human setting.
Imaging of meningeal lymphatic structures and their functional characteristics in rodents
In their seminal study from 2015, Louveau and coworkers detected that fluorescent CSF tracer drained into vessels adjacent to the sagittal and transverse dural sinuses (dorsal regions) that were positive for several lymphatic endothelial cell markers [2]. These vessels were considered to represent initial lymphatic vessels, covering less of the tissue and forming a less complex network composed of narrow vessels compared to other body regions. Diameter of the vessels was shown to be at micrometer scale, and it was hypothesized that the vessels may add to CSF filtration via arachnoid granulations, which are prominent in the perisinous region. Finally, it was proposed that meningeal lymphatic vessels may represent the second step in the drainage of interstitial fluid from the brain parenchyma after primary drainage into CSF. In the same year, Aspelund et al. [3] in another seminal study observed that the dye they injected into brain parenchyma drained into CSF along paravenous routes and entered the CSF space before drainage to deep cervical lymph nodes, primarily via lymphatic vessels that were exiting through foramina at the skull base. In later studies, lymphatic vessels were reported to be the major outflow pathway for CSF [17] and that ventral meningeal lymphatic vessels at the skull base were the hotspots for lymphatic drainage of CSF, not those in the dorsal perisinus region [18]. It was more recently demonstrated that the parasagittal dural stroma is rather an important hub for brain-immune cross-talk, where circulating immune cells can enter its stroma directly from the venous sinuses and here be presented to brain specific antigens by local dendritic cells [19]. Moreover, arachnoid granulations have been shown to communicate with this stroma and also to harbor immune cells, suggesting these trans-arachnoid passage ways may also represent lymphatic conduits with a neuroimmune role [20].
The human parasagittal dura
Rodent and human brain differ in many aspects, not only in the most obvious ways concerning size and gyration patterns; but also clearance of tracer from CSF occurs of a time scale of days in humans [8], compared to a few hours in rats [21], with the pig brain comparing closer to the human [22]. Functionally, it has also been provided evidence that CSF lymphatic efflux routes may differ; while nasal lymphatic efflux is consistently shown to be of major importance in rodents [23], an in vivo CSF tracer study in humans detected no efflux through the cribriform plate to nasal mucosa [24], which contradicts previous non-MRI studies [25, 26]. There may also be species differences regarding impact of sleep and circadian rhythms on meningeal clearance function. Hence, lymphatic efflux of CSF tracer examined by near-infrared fluorescence imaging, was faster in awake than anesthetized mice [27]. In humans, on the other hand, there was in one patient cohort no effect of one night`s sleep deprivation on efflux of CSF tracer to parasagittal dura [28]. In the brain, however, CSF tracer dynamics was affected by both acute sleep deprivation [29] and chronic sleep impairment [30]. Further translational research in humans therefore seem mandated.
Fox et al. performed in 1996 a detailed anatomical study of the parasagittal dura based on formalin-fixed human autopsy specimens. In addition to intradural arachnoid granulations, they reproduced presence of an extensive network of intradural, endothelial-lined channels stemming from the superior sagittal sinus along nearly its entire length. These channels were first described by Weed in 1917 [31], and has later been demonstrated by others [32,33,34]. Fox et al. assessed the dimensions of intradural channels, which ranged from 0.02 to 2.0 mm and either connected to the superior sagittal sinus or lateral lacunae, which extended up to 3 cm from midline. The network of intradural channels was unrelated to cortical veins and was proposed to represent a pathway for the flow of CSF from arachnoid granulations to the superior sagittal sinus. Meningeal veins also appeared distinct from the intradural channels, but not branches of meningeal veins, which arborized medially and to some extent connected there with intradural channels. Intradural channels have a superior location to arachnoid granulations [32], which may imply their role in trans-dural CSF efflux to skull bone marrow [35]. A review by Mack et al. [36] also emphasized the extensive venous network within dura, as well as the meningeal arteries and accompanying meningeal veins, which are superficially located in the outer aspect of dura, and finally capillaries, that are present throughout the dura and are extremely rich in the parasagittal region. Dural lymphatic vessels were identified to be at micrometer scale in the parasagittal dura from histological analysis of whole-mount human specimens [37]. Recently, dural lymphatic vessels were also demonstrated utilizing immunohistochemistry in specimens retrieved from patients [1, 38]; the lymphatic vessels were seen in specimens from patients and obtained about 1.5 cm lateral to the parasagittal dura, and also in more remote locations. Here, lymphatic vessels were both localized nearby blood vessels as well as in distance to blood vessels. Given the methodological challenges with identifying lymphatic vessels using formalin-fixed tissues [1], it remains to be determined the relationship between intradural channels in parasagittal dura and lymphatic vessels.
Attempts to visualize meningeal lymphatics in parasagittal dura with MRI
Regarding imaging of human meningeal lymphatics, MRI has typically been the preferred methodology due to its non-invasive nature, and has mainly focused on the parasagittal dural region. Various MRI based approaches have been performed, by both assessing enhancement patterns of MRI contrast agents administered intravenously and intrathecally, but also without any use of contrast agent. In any case, since blood vessels, intradural arachnoid granulations and intradural channels are abundant in the parasagittal dura, these need to be accounted for in attempts to visualize lymphatic vessels in this region. Accordingly, a commonly acknowledged challenge with MRI of lymphatic vessels in other body regions (MR lymphangiography) has been to separate contrast enhancing blood vessels from lymphatic vessels. For superficial body lymphatic vessels, this has been accomplished by obtaining MRI after injecting contrast agent into the cutis to visualize lymphatic channels through a 30 min period, and subsequently, an MRI 2–3 min after intravenous contrast to define veins [39]. Lymphatic vessels in limbs will typically show progressive contrast enhancement with the greatest degree after 30 min and appear with a characteristic irregular, beaded pattern. Veins have typically a smooth, uniform caliber and are visible 2–3 min after intravenous injection. MRI contrast agents have typically a half-life in blood of approximately two hours, are therefore present within the blood circulation and thus also in peripheral tissue for several hours [40].
Several reports of MRI studies claim to have directly visualized dural lymphatic vessels after administration of intravenous contrast [41,42,43,44,45,46,47], some with addition of the precautious prefix “putative” [47]. Typically, single, mm-thick tubular structures are demonstrated in 2D or 3D and conclusions about their lymphatic origin typically rely on a topography fitting with descriptions of dural lymphatic networks in rodents or neuropathological data. Strikingly, there are to our knowledge no studies discussing the possibility that tubular shaped structures at MRI might represent veins or derive from other known, abundant constituents in the region. There is also broadly a lack of considerations that vessels visualized at MRI are orders of magnitude larger in diameter than what has been described in animal [2, 3, 19] as well as in human [1, 38, 48] studies. In one human study, lymphatic vessel diameter at various location remote from sinus veins varied from approximately 20–40 µm [38] (Fig. 2), and in another, meningeal lymphatic vessels adjacent to the superior sagittal sinus ranged from 8 to 119 µm [48]. Thus, lymphatic vessels at MRI should not be expected to have diameter larger than ~ 0.1 mm, and often even less, which is far below the typical MR image resolution (~ 1 mm).
Human dural lymphatic vessel from the upper frontal convexity. Co-localization of the lymphatic markers lymphatic vessel endothelial hyaluronan receptor 1 (LYVE-1) and podoplanin (PDPN) confirms the presence of meningeal lymphatic vessels in dura mater of patients (panels J–L are magnification of panels G–I). The vessels have a diameter at micrometer scale and would not be expected to be delineated at mm-scale MRI. Courtesy of Fig. 7 G–L from Quesada et al. FBCNS) [1], reprinted under the terms of the Creative Commons Attribution License (CC BY)
On the contrary, a human CSF tracer study has demonstrated direct efflux from CSF into the parasagittal dura through 48 h, but could not depict signs of the tracer collecting into any tubular structures whatsoever, only diffuse enhancement [28, 49]. Peak enhancement in parasagittal dura occurred at 24 h after intrathecal tracer injection and was highly correlated with enhancement in adjacent CSF (Fig. 3). Another study using same methodology, rather interpreted a similar, diffuse enhancement pattern as “putative” meningeal lymphatic vessels [50], but without providing evidence for enhancement in tubular structures per se.
The parasagittal dura (PSD) is visible as hyperintense tissue at FLAIR (A and C). PSD (B) volume varies widely between subjects for unknown reasons, but has in addition to stroma been shown to contain arachnoid granulations, blood vessels, intradural channels, lateral lacunae and lymphatic vessels, the latter at micrometer scale. After injection of a CSF tracer (MRI contrast agent), PSD enhances visibly with tracer after 24 and 48 h in a diffuse fashion, but not in the shape of tubular structures. Tracer enhancement in PSD is dependent on tracer enhancement in the adjacent cerebrospinal fluid (CSF) space and is not visible before tracer has reached the CSF compartment (D–F). At 24 h (G) and 48 h (H), From Eide PK and Ringstad (Brain Research) [28], reused with permission under the terms of the Creative Commons CC-BY license
To exclude venous signals at MRI, “black blood” techniques have been applied to darken signal from slow flow within the venous vasculature [41]. However, shine-through of signal from veins with very slow flow, or artifacts from slow flowing blood in smaller vessels is one major confounding factor in the interpretation of high signal in tubular structures [51]. Whereas accompanying contrast-enhanced MR venography has further been added to subtract signals from veins that were visible at MRI [41], venous structures of microscopic size with very low flow velocity, or having caliber well below the image resolution, may not be depicted. Still, diffuse contrast enhancement in parasagittal dural regions was widely interpreted as lymph. However, blood vessels in the dura lack tight junctions and allow extravasation of solutes as large as 43 kDa [52], while for instance gadobutrol has molecular weight ~ 0.6 kDa [40]. Therefore, intravenous contrast is expected to enhance in the extra-vascular stroma of the parasagittal dura through these abundant, leaky vessels. Further sources of contrast enhancement in the parasagittal dura, whether diffuse or in shape of tubular vessels, may be derived from enrichment within networks of dural veins and capillaries, and also as proposed by Park et al. [32], enrichment within intradural channels. Absinta et al. [37] observed that a blood pool agent (gadofosveset) did not leak into the parasagittal dura, contrary to the enhancement which was observed in dura after intravenous administration of gadobutrol. Even though the enhancement they observed may be considered diffuse and at millimeter scale as revealed by 2D (Fig. 4) and not least by 3D representations, and qualitatively comparable to the enhancement in the choroid plexuses, they interpreted this as lymphatic vessels.
Examples of interpretation of lymphatic tissue in parasagittal dura. In a intravenous contrast enhancement in parasagittal dura is interpreted as lymphatic vessels (red arrows), while blood flow in superior sagittal sinus is suppressed (From Absinta et al. eLife 2017) [37]. In b all hyperintense FLAIR-signal in parasagittal dura (arrowheads) was accepted as lymph (From Albayram et al. Nature Communications, 2022) [54]. Figure elements are reused with permission under the Creative Commons International License
To this end, it can be argued that imaging of lymph by use of intravenous contrast-enhanced MRI therefore awaits further validation. Future imaging studies must be interpreted with larger care and by acknowledging existence of other sorts of tubular structures than lymphatic vessels being present in the region. While it should be expected that dural lymphatic vessels indeed exist also in the human parasagittal dura, it remains unknown whether the recently reported lymphatic vessels in human dura were within or lateral to the parasagittal dura [1]. In this regard, it should be kept in mind that the volume of parasagittal dura varies extensively between individuals [53], and the basis for this is currently not well understood. As discussed previously, meningeal lymphatic vessels within the parasagittal dura region may rather be important for immunological surveillance of the brain more than in CSF drainage [17, 19]. This is supported by human CSF tracer studies, showing that CSF flux around the upper brain convexities differs substantially between individuals [8], and with sometimes very minute amounts of CSF tracer entering from the upper SAS adjacent into the parasagittal dura [49], suggesting this is not a major efflux route. Notwithstanding this, exchange between SAS and parasagittal dura as part of neuroimmunlogical surveillance may still take place.
A method for a non-contrast approach to visualize meningeal lymph has also been suggested, where literally any hyperintense FLAIR signal outside the brain at MRI was interpreted as lymph [54]. The basis for this understanding was that increasing protein content in an external container was positively associated with FLAIR signal. Since lymph is protein rich, a hyperintense FLAIR signal was therefore accepted to derive from lymph only. As outlined above, the parasagittal dura contains many elements that can contribute to the hyperintense FLAIR signal, including small blood vessels, the lateral lacuna, intradural arachnoid granulations, and the fibrous stroma of the parasagittal dura. Interpretation of a hyperintense FLAIR signal as lymph is therefore highly presumptuous. Furthermore, even hyperintense FLAIR signals from the CSF spaces near the skull base were interpreted as ventral meningeal vessels; however, this has later been dismissed as CSF flow artifacts [55]. Finally, the authors interpreted a continuous, high FLAIR signal in the walls of neck blood vessels as lymph, too, and the continuity with similar signals at the skull base was considered to represent connections between cervical lymph nodes and the intracranial meningeal lymphatic system. A similar interpretation was reproduced in another study from the same group [56]. Again, lymph is not an exclusive source of a high FLAIR signal, and for instance connective tissue and vasa vasorum in neck vessel walls are least as likely sources. Ultimately, the reasoning behind accepting any structure with high FLAIR signal outside brain as lymph was heavily contested by two independent commentaries [55, 57]. Examples of contrast and non-contrast based interpretations of lymph at MRI are given in Fig. 4.
It remains to be determined how abundant lymphatic vessels are in human dura. Given the recent observations that fixation method extensively affects the ability to visualize lymphatic vessels in human dura [1], care should be taken when interpreting findings from formalin-fixed dura of cadavers. Accordingly, a recent study failed to identify lymphatic structures in the perisinus region, but rather tubular intradural vascular channels which stained positively with markers of lymphatic vessels that connected lateral interdural arachnoid granulations with the venous circulation [58]. In another work, it was concluded that arachnoid granulations that traverse the dura-arachnoid stroma not only serve as CSF reservoirs, but are also enriched with immune cells, supporting that perisinus tissues can represent important immune hubs at the brain surface [20]. Kutomi and Takeda also found that arachnoid granulations in pigs were lined by lymphatic endothelial cells, while dura mater in mouse was devoid of arachnoid granulations and “totally underdeveloped” compared to pig or human [59].
Imaging evidence of glymphatic–lymphatic coupling
In humans, MRI was used for the first time to demonstrate in vivo drainage of a CSF tracer (the MRI contrast agent gadobutrol) to the lymphatic system, where the tracer enhanced subtly in cervical lymph nodes with peak after 24 h. Peak enhancement coincided with peak tracer enhancement in brain, and the finding was inferred to indicate that the tracer was cleared directly from perivascular spaces in brain to lymph after its enhancement in brain [60]. This tracer is inert, confined outside the intact BBB, and should not be expected to be cleared via other routes than the perivascular. However, later studies using the same methodology demonstrated clearly that also levels of tracer in CSF are highly associated with levels in brain at all time points through 24–48 h, well into clearance phase, suggesting that CSF clearance rate is a major determinate for brain tracer clearance. Moreover, the same CSF tracer appeared in blood with a peak merely a few hours after its intrathecal distribution, preceding peak enhancement in cervical lymph nodes by far, and also preceding its enhancement at the upper brain convexities. This sequential pattern clearly suggests that a substantial proportion of tracer was already resorbed form the spinal canal and at the skull base via meningeal lymphatic vessels. Tracer levels in CSF under the cranial vertex was a prerequisite for, and correlated with, enhancement in the parasagittal dura, clearly indicating direct efflux from CSF directly to dura, not from the brain. These human observations of efflux from CSF to meninges also resonate well with previous animal findings [2, 3], but do not exclude the possibility of an anatomically uninterrupted clearance pathway from brain to meningeal lymph.
Indeed, observations from intravenous contrast-enhanced MRI have been inferred to support a direct anatomical link between brain paravascular pathways and meningeal lymphatic vessels. Naganawa et al. demonstrated age-dependent leakage of contrast agent around the vein of Labbè [61], as well as from cortical veins at the upper convexity, into CSF at inversion recovery MRI [62]. Later, it was suggested that the contrast leaked into a space connecting the glymphatic system and meningeal lymphatics [63]. Sennfält and coworkers had a similar interpretation of this phenomenon in a recent paper [45], where they conclude that their findings suggested that interstitial fluid drainage was visible on conventional MRI and drained from brain parenchyma via cortical perivenous spaces to dural meningeal lymphatics in the parasagittal dura. As such, this drainage pathway remained separate from CSF. Observations were typically made within 30 min after intravenous contrast administration. However, this stands in sharp contradistinction to in vivo observations of CSF tracer clearance from brain, which occurs for hours and days [8]. It seems very likely that this discrepancy can be derived from an alternative cause behind the perivenous enhancement; a direct leakage of contrast agent through the venous wall. Leptomeningeal blood vessels have endothelial cells connected by tight junctions, but are not ensheathed by astrocytic foot processes, and paracellular junctions between these endothelial cells vary in tightness and can be up to 2.8 nm large [64]. Even within brain tissue, brain vessels are to some degree leaky to contrast agents, and previously established leakage constants indicate that gadolinium concentration in brain tissue after intravenous administration can be close to what is reported after intrathecal administration [65]. At arterial spin labeling MRI with ultra-long echo times, substantial and rapid exchanges of water between blood vessels and CSF at the brain surface was demonstrated [66]. Furthermore, MRI contrast agent has been shown to leak directly into CSF and the subarachnoid space at multiple locations [67,68,69]. Perivenous enhancement after intravenous administration was also observed in a case series after temporary opening of the blood–brain-barrier using focused ultrasound [70]. However, the phenomenon was shown to occur in merely a fraction of subjects, and it was not discussed whether the outskirts of the applied ultrasound energy field might have induced increased venous wall permeability. To this end, human MRI studies have so far only provided circumstantial evidence of an uninterrupted, compartmentalized clearance pathway from brain to lymph, and observations appear biased by expectations from theoretical frameworks.
Meningeal immunity and CSF-mediated interactions with the CNS is a flourishing research field with intense ongoing debates. Recently, it was described a previously unrecognized, fourth meningeal layer denoted subarachnoid lymphatic-like membrane (SLYM) [71]. The SLYM layer was shown to contain Prox1 + cells and to encase subarachnoid blood vessels. By its impermeability to molecules larger than 3 kD, and thereby functionally dividing the subarachnoid space into compartments, the SLYM layer would also enable for a directionalized CSF movement at the brain surface toward brain perivascular spaces, possibly linking meningeal immunity with the brain more directly. However, two later studies possibly contradicted the SLYM by claiming that Prox1 + cells were part of the arachnoid mater [72, 73]. While the possible existence of SLYM yet seems unresolved, a previous human MRI study demonstrated sign of periarterial propagation of CSF tracer within the subarachnoid space, as implicated by a SLYM layer [9]. Future human MRI studies utilizing CSF tracer and high temporal resolution could possibly resolve some of these outstanding questions.
When considering coupling between the glymphatic and lymphatic system, MRI investigators further need to be aware of a potential CSF-mediated cross-talk between the CNS and spinal dural lymphatic vessels. Spinal absorption of CSF has for long been well established to occur [74], but clearance pathways were more obscure. Recently, a continuum of meningeal and epidural lymphatic vessels in the vertebral (spinal) canal was described, with lateral exits along blood vessels and spinal nerves [75]. The spinal lymphatic vasculature was shown to interact immunologically with the spinal cord and furthermore to be particularly dense compared to the one that covers the cranial dura mater, leading to the possible explanation that spinal lymphatic vessels strongly contribute to CSF absorption [76]. A concurring study revealed that following intraventricular tracer injection in mice, tracer propagated down the spinal canal and was cleared from the subarachnoid space predominantly from the sacral spinal canal to lymphatic vessels and further to sacral and iliac lymph nodes [77]. Substantial spinal clearance was corroborated by a human CSF tracer study, where intrathecally injected MRI contrast agent at the lumbar level appeared in blood with peak concentration far earlier than peak contrast enrichment around the upper brain convexities. This constellation of findings suggests that substantial CSF clearance occurs from the spinal canal as well as the skull base. A further analysis of MRI data suggested that up to 2/3 of injected tracer in humans was resorbed via the thecal sac [78].
Imaging evidence of coupling between CNS and bone marrow
It is remarkable that direct dural-bone marrow connections have been demonstrated physically as well as functionally, enabling for mobilization of myeloid cells into meninges and CNS parenchyma following injury and neuroinflammation [79]. Toward skull bone marrow, CSF efflux was shown for the first time in any species at human MRI [35] and was later confirmed in mice with meningitis, here also revealing that CSF and bacteria exited along perivascular spaces of dural blood vessels and through sub-millimeter skull channels [80]. The same channels directly provided for backwards migration of leukocytes to meninges. Thus, brain-derived danger signals in CSF can be sampled by regional marrow. It was recently demonstrated that the mouse skull has the most distinct transcriptomic profile compared with other bones, corroborated by the finding that skull bone marrow reflected inflammatory brain responses in patients with various neurological disorders, including inflammatory, ischemic, and neurodegenerative diseases [81]. These compelling observations in humans were accomplished by use of positron emission tomography, and it remains to be seen whether dedicated MRI could reveal bone marrow changes in response to disease.
Two-way interactions between the brain and peripheral (meningeal) and central (bone marrow) immune components have wide implications for potential mechanisms in, and modifying of, neuroinflammation, neoplastic disease and neurodegeneration [82]. Furthermore, when central and peripheral immune constituents can interact directly with the meninges and also be distributed in CSF, new hypotheses may emerge about mechanisms behind symptoms associated with common viral illnesses, including headache and sickness behavior. No human study has yet investigated CSF efflux to vertebral bone marrow, neither in the normal physiological situation nor in disease, which may eventually be accomplished with MRI contrast agents or a radioactive compound utilized as CSF tracer.
CSF clearance as measure of meningeal lymphatic function
The acknowledgment that lymphatic vessels are the major outflow pathway of small and large CSF tracer substances, renders CSF clearance a potentially impactful marker of meningeal lymphatic clearance capacity [17]. For endogenous solutes with a role in neurodegenerative disease, one study demonstrated that impaired functional lymphatics was associated with reduced extracellular tau clearance from CNS [4]. It was proposed that the glymphatic and lymphatic systems work in tandem to clear tau to the periphery, where tau first was cleared from ISF to CSF before uptake by the dural lymphatic system. Interestingly, despite complete lack of lymphatic vessels, tau was still cleared from brain to the periphery, suggesting that other, but minor clearance routes for tau may exist. Indeed, a tau transporter over the BBB has been characterized [83]. In a Parkinson’s disease animal model, ligation of cervical lymphatic vessels aggravated accumulation of alfa-synuclein [84]. In rodents, amyloid-β is to large extent cleared from brain via other pathways than the perivascular route, including degradation by microglia and via dedicated transporters at the BBB, and merely about ¼ of amyloid-β is cleared via CSF [85, 86].
It is therefore likely that the ability to measure CSF clearance rate would provide a suitable marker of lymphatic clearance capacity, but perhaps particularly for tau, which is deposited in brain in Alzheimer`s disease and other tauopathies. Feasibility of measuring CSF clearance rate in humans has been shown for CSF-to-blood clearance of intrathecally injected MRI contrast agent gadobutrol [87], a gadolinium–based contrast agent. Intrathecal injection of gadobutrol is “off-label” use, but the methodology can likely be converted to radiopaque contrast agents, that are generally approved for intrathecal injection, and have many molecular properties similar to gadobutrol. These features include molecular size, hydrophilic properties, the lack of binding to or reacting with proteins, and being contained outside the intact blood–brain-barrier after distribution in CSF. Thus, clearance over the blood–brain-barrier is negligible and largely anticipated to occur via meningeal lymphatic channels, both from the intracranial space and the spinal canal. Prospective safety studies of intrathecal gadobutrol in amount 0.25–0.50 mmol have shown side effects that are comparable to those reported after use of intrathecal radiopaque agents, and also after spinal punctures alone [88,89,90]. These observations are corroborated by the experiences from clinical use of MRI contrast agents in dose 1.0 mmol and less [91]. Remarkably, the methodology has revealed that CSF clearance not only differs between patient groups, but also among individuals within the same patient category [92] (Fig. 5). A methodological strength is that it measures overall CSF clearance and is thus independent on knowledge about particular clearance pathways, a question that is still debated [10, 93]. Being a surrogate marker of endogenous solutes of much larger size, it has yet not been validated against established biomarkers in CSF, but was associated with plasma biomarkers of neurodegeneration [94], and also with their fluctuations in plasma after sleep deprivation [95]. The methodology still lacks validation against clinical end points, such as prognostication of disease and disease progression, but can already be a marker of CSF clearance capacity for tailoring of dosage in intrathecal treatment regimens [92], since drugs in CSF must also be expected to be mainly removed along lymphatic pathways, equally to CSF itself.
Pharmakokinetic variability in CSF-to-blood clearance of gadobutrol for reference patients (REF) (A), patients with pineal cysts (PC) (B), arachnoid cysts (AC) (C), spontaneous intracranial hypotension (SIH) (D), idiopathic intracranial hypertension (IIH) (E), idiopathic normal pressure hydrocephalus (iNPH) (F), communicating hydrocephalus (Comm HC) (G) and non-communicating hydrocephalus (Non-comm HC) (H). Black lines provide the mean predicted concentration of intrathecally injected gadobutrol in blood over time. From: Hovd MH et al. (Fluids and Barriers CNS) [92], reused with permission under the terms of the Creative Commons CC-BY license
Concluding remarks
Pioneering research in animal models has positioned glymphatic brain clearance and its interplay with meningeal lymphatics at the forefront of CNS research. MRI stands out as an attractive tool to image those two systems and their coupling. MRI particularly profits from the ability to acquire a bigger perspective both anatomically and functionally within the craniospinal compartment, throughout an extended time span, and in vivo. However, both obvious and more subtle species differences mandate translation of previous animal research findings to humans. In future research, human MRI studies should liberate themselves from proposed frameworks now seemingly restraining perspectives on how imaging findings are interpreted, and better appreciate the extreme complexity of these systems. So far, interpretations of imaging findings within this young research field appear heavily biased by a desire to reproduce findings from animal studies, and very few negative studies are reported. As discussed in this review, MRI findings that have been accepted to represent lymph can least as likely be derived from other sources. Likewise, no convincing evidence has yet been provided for a direct coupling between the glymphatic system and meningeal lymphatic function, and currently, CSF is more likely to represent the intermediate step between those two systems. Meningeal lymphatic clearance function is likely the main determinate of CSF clearance rate and thereby glymphatic clearance rate along perivascular pathways. Measurements of CSF tracer clearance to blood can therefore potentially serve as surrogate marker of meningeal lymphatic clearance function. Currently, it is yet unclear to what degree glymphatic-lymphatic brain clearance contributes to total brain clearance, and how this fraction may differ between CNS regions. In this context, MR imaging of CSF flow patterns at the CNS surface emerges with new relevance, as large inter-individual variations in humans have already been revealed. Whether for instance CSF flow obstruction at the brain surface is also associated with compromise of solute clearance or immunological function, should be assessed in future research.
Data availability
Not applicable (review manuscript with no use of new original data).
References
Vera Quesada CL, Rao SB, Torp R, Eide PK (2023) Immunohistochemical visualization of lymphatic vessels in human dura mater: methodological perspectives. Fluids Barriers CNS 20(1):23. https://doi.org/10.1186/s12987-023-00426-3
Louveau A, Smirnov I, Keyes TJ, Eccles JD, Rouhani SJ, Peske JD et al (2015) Structural and functional features of central nervous system lymphatic vessels. Nature 523(7560):337–341. https://doi.org/10.1038/nature14432
Aspelund A, Antila S, Proulx ST, Karlsen TV, Karaman S, Detmar M et al (2015) A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J Exp Med 212(7):991–999. https://doi.org/10.1084/jem.20142290
Patel TK, Habimana-Griffin L, Gao X, Xu B, Achilefu S, Alitalo K et al (2019) Dural lymphatics regulate clearance of extracellular tau from the CNS. Mol Neurodegener 14(1):11. https://doi.org/10.1186/s13024-019-0312-x
Da Mesquita S, Louveau A, Vaccari A, Smirnov I, Cornelison RC, Kingsmore KM et al (2018) Functional aspects of meningeal lymphatics in ageing and Alzheimer’s disease. Nature 560(7717):185–191. https://doi.org/10.1038/s41586-018-0368-8
Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA et al (2012) A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Sci Transl Med 4(147):147ra11. https://doi.org/10.1126/scitranslmed.3003748
Agarwal N, Lewis LD, Hirschler L, Rivera LR, Naganawa S, Levendovszky SR et al (2023) Current Understanding of the anatomy, physiology, and magnetic resonance imaging of neurofluids: update from the 2022 “ISMRM imaging neurofluids study group” Workshop in Rome. J Magn Reson Imaging: JMRI. https://doi.org/10.1002/jmri.28759
Ringstad G, Valnes LM, Dale AM, Pripp AH, Vatnehol SS, Emblem KE et al (2018) Brain-wide glymphatic enhancement and clearance in humans assessed with MRI. JCI Insight. https://doi.org/10.1172/jci.insight.121537
Ringstad G, Vatnehol SAS, Eide PK (2017) Glymphatic MRI in idiopathic normal pressure hydrocephalus. Brain: J Neurol 140(10):2691–2705. https://doi.org/10.1093/brain/awx191
Hladky SB, Barrand MA (2022) The glymphatic hypothesis: the theory and the evidence. Fluids Barriers CNS 19(1):9. https://doi.org/10.1186/s12987-021-00282-z
Rasmussen MK, Mestre H, Nedergaard M (2022) Fluid transport in the brain. Physiol Rev 102(2):1025–1151. https://doi.org/10.1152/physrev.00031.2020
Nedergaard M, Goldman SA (2020) Glymphatic failure as a final common pathway to dementia. Science 370(6512):50–56. https://doi.org/10.1126/science.abb8739
Smith AJ, Yao X, Dix JA, Jin BJ, Verkman AS (2017) Test of the ‘glymphatic’ hypothesis demonstrates diffusive and aquaporin-4-independent solute transport in rodent brain parenchyma. Elife. https://doi.org/10.7554/eLife.27679
Zhao L, Tannenbaum A, Bakker E, Benveniste H (2022) Physiology of glymphatic solute transport and waste clearance from the brain. Physiology (Bethesda). https://doi.org/10.1152/physiol.00015.2022
Engelhardt B, Vajkoczy P, Weller RO (2017) The movers and shapers in immune privilege of the CNS. Nat Immunol 18(2):123–131. https://doi.org/10.1038/ni.3666
Albargothy NJ, Johnston DA, MacGregor-Sharp M, Weller RO, Verma A, Hawkes CA, Carare RO (2018) Convective influx/glymphatic system: tracers injected into the CSF enter and leave the brain along separate periarterial basement membrane pathways. Acta Neuropathol 136(1):139–152. https://doi.org/10.1007/s00401-018-1862-7
Ma Q, Ineichen BV, Detmar M, Proulx ST (2017) Outflow of cerebrospinal fluid is predominantly through lymphatic vessels and is reduced in aged mice. Nat Commun 8(1):1434. https://doi.org/10.1038/s41467-017-01484-6
Ahn JH, Cho H, Kim JH, Kim SH, Ham JS, Park I et al (2019) Meningeal lymphatic vessels at the skull base drain cerebrospinal fluid. Nature 572(7767):62–66. https://doi.org/10.1038/s41586-019-1419-5
Rustenhoven J, Drieu A, Mamuladze T, de Lima KA, Dykstra T, Wall M et al (2021) Functional characterization of the dural sinuses as a neuroimmune interface. Cell 184(4):1000–16 e27. https://doi.org/10.1016/j.cell.2020.12.040
Shah T, Leurgans SE, Mehta RI, Yang J, Galloway CA, de Mesy Bentley KL et al (2023) Arachnoid granulations are lymphatic conduits that communicate with bone marrow and dura-arachnoid stroma. J Exp Med. https://doi.org/10.1084/jem.20220618
Iliff JJ, Lee H, Yu M, Feng T, Logan J, Nedergaard M, Benveniste H (2013) Brain-wide pathway for waste clearance captured by contrast-enhanced MRI. J Clin Investig 123(3):1299–1309. https://doi.org/10.1172/jci67677
Bechet NB, Shanbhag NC, Lundgaard I (2021) Glymphatic pathways in the gyrencephalic brain. J Cereb Blood Flow Metab 41(9):2264–2279. https://doi.org/10.1177/0271678X21996175
Proulx ST (2021) Cerebrospinal fluid outflow: a review of the historical and contemporary evidence for arachnoid villi, perineural routes, and dural lymphatics. Cell Mol Life Sci 78(6):2429–2457. https://doi.org/10.1007/s00018-020-03706-5
Melin E, Eide PK, Ringstad G (2020) In vivo assessment of cerebrospinal fluid efflux to nasal mucosa in humans. Sci Rep 10(1):14974. https://doi.org/10.1038/s41598-020-72031-5
Johnston M, Zakharov A, Papaiconomou C, Salmasi G, Armstrong D (2004) Evidence of connections between cerebrospinal fluid and nasal lymphatic vessels in humans, non-human primates and other mammalian species. Cerebrospinal Fluid Res 1(1):2. https://doi.org/10.1186/1743-8454-1-2
de Leon MJ, Li Y, Okamura N, Tsui WH, Saint-Louis LA, Glodzik L et al (2017) Cerebrospinal fluid clearance in Alzheimer disease measured with dynamic PET. J Nucl Med 58(9):1471–1476. https://doi.org/10.2967/jnumed.116.187211
Ma Q, Ries M, Decker Y, Muller A, Riner C, Bucker A et al (2019) Rapid lymphatic efflux limits cerebrospinal fluid flow to the brain. Acta Neuropathol 137(1):151–165. https://doi.org/10.1007/s00401-018-1916-x
Eide PK, Ringstad G (2021) Cerebrospinal fluid egress to human parasagittal dura and the impact of sleep deprivation. Brain Res 1772:147669. https://doi.org/10.1016/j.brainres.2021.147669
Eide PK, Vinje V, Pripp AH, Mardal KA, Ringstad G (2021) Sleep deprivation impairs molecular clearance from the human brain. Brain: J Neurol 144(3):863–874. https://doi.org/10.1093/brain/awaa443
Eide PK, Pripp AH, Berge B, Hrubos-Strom H, Ringstad G, Valnes LM (2022) Altered glymphatic enhancement of cerebrospinal fluid tracer in individuals with chronic poor sleep quality. J Cereb Blood Flow Metab 42(9):1676–1692. https://doi.org/10.1177/0271678X221090747
Weed LH (1917) An anatomical consideration of the cerebro-spinal fluid. Anat Rec 12(4):461–496. https://doi.org/10.1002/ar.1090120405
Park M, Park JP, Kim SH, Cha YJ (2022) Evaluation of dural channels in the human parasagittal dural space and dura mater. Ann Anat 244:151974. https://doi.org/10.1016/j.aanat.2022.151974
Han H, Tao W, Zhang M (2007) The dural entrance of cerebral bridging veins into the superior sagittal sinus: an anatomical comparison between cadavers and digital subtraction angiography. Neuroradiology 49(2):169–175. https://doi.org/10.1007/s00234-006-0175-z
Johnson MD (2021) Evaluation of fluid channels in the human dura and superior sagittal sinus in older patients. Neurosci Lett 746:135649. https://doi.org/10.1016/j.neulet.2021.135649
Ringstad G, Eide PK (2021) Molecular trans-dural efflux to skull bone marrow in humans with cerebrospinal fluid disorders. Brain: J Neurol. https://doi.org/10.1093/brain/awab388
Mack J, Squier W, Eastman JT (2009) Anatomy and development of the meninges: implications for subdural collections and CSF circulation. Pediatr Radiol 39(3):200–210. https://doi.org/10.1007/s00247-008-1084-6
Absinta M, Ha SK, Nair G, Sati P, Luciano NJ, Palisoc M et al (2017) Human and nonhuman primate meninges harbor lymphatic vessels that can be visualized noninvasively by MRI. Elife. https://doi.org/10.7554/eLife.29738
Vera Quesada CL, Rao SB, Torp R, Eide PK (2023) Widespread distribution of lymphatic vessels in human dura mater remote from sinus veins. Front Cell Dev Biol 11:1228344. https://doi.org/10.3389/fcell.2023.1228344
Mitsumori LM, McDonald ES, Wilson GJ, Neligan PC, Minoshima S, Maki JH (2015) MR lymphangiography: how i do it. J Magn Reson Imaging: JMRI 42(6):1465–1477. https://doi.org/10.1002/jmri.24887
Cheng KT (2004) Gadobutrol. Molecular imaging and contrast agent database (MICAD). Bethesda (MD): National Center for Biotechnology Information (US)
Jacob L, de Brito NJ, Lenck S, Corcy C, Benbelkacem F, Geraldo LH et al (2022) Conserved meningeal lymphatic drainage circuits in mice and humans. J Exp Med. https://doi.org/10.1084/jem.20220035
Filippopulos FM, Fischer TD, Seelos K, Dunker K, Belanovic B, Crispin A et al (2022) Semiquantitative 3T brain magnetic resonance imaging for dynamic visualization of the glymphatic-lymphatic fluid transport system in humans: a pilot study. Invest Radiol 57(8):544–551. https://doi.org/10.1097/RLI.0000000000000870
Gabr RE, Lincoln JA, Hasan KM, Kramer LA (2023) Functional assessment of the dural lymphatic vessels using dynamic contrast MRI in multiple sclerosis. Brain Behav 13(7):e3042. https://doi.org/10.1002/brb3.3042
Kim JH, Yoo RE, Choi SH, Park SH (2023) Non-invasive flow mapping of parasagittal meningeal lymphatics using 2D interslice flow saturation MRI. Fluids Barriers CNS 20(1):37. https://doi.org/10.1186/s12987-023-00446-z
Sennfalt S, Thrippleton MJ, Stringer M, Reyes CA, Chappell F, Doubal F et al (2023) Visualising and semi-quantitatively measuring brain fluid pathways, including meningeal lymphatics, in humans using widely available MRI techniques. J Cereb Blood Flow Metab. https://doi.org/10.1177/0271678X231179555
Zhang M, Tang J, Xia D, Xue Y, Ren X, Huang Q et al (2023) Evaluation of glymphatic-meningeal lymphatic system with intravenous gadolinium-based contrast-enhancement in cerebral small-vessel disease. Eur Radiol 33(9):6096–6106. https://doi.org/10.1007/s00330-023-09796-6
Naganawa S, Ito R, Kawamura M, Taoka T, Yoshida T, Sone M (2023) Association between the putative meningeal lymphatics at the posterior wall of the sigmoid sinus and delayed contrast-agent elimination from the cerebrospinal fluid. Magn Reson Med Sci: MRMS. https://doi.org/10.2463/mrms.mp.2022-0110
Safiye Çavdar BK, Altınöz D, Söyler G, Cingöz A, Ali Gürses İ, Özkan M, Aslıyüksek H, Çakır H (2023) Lymphatic vessels accompanying dorsal and basal dural sinuses in the human brain. J Chem Neuroanat. https://doi.org/10.1016/j.jchemneu.2023.102357
Ringstad G, Eide PK (2020) Cerebrospinal fluid tracer efflux to parasagittal dura in humans. Nat Commun 11(1):354. https://doi.org/10.1038/s41467-019-14195-x
Zhou Y, Cai J, Zhang W, Gong X, Yan S, Zhang K et al (2020) Impairment of the glymphatic pathway and putative meningeal lymphatic vessels in the aging human. Ann Neurol 87(3):357–369. https://doi.org/10.1002/ana.25670
Lindenholz A, van der Kolk AG, Zwanenburg JJM, Hendrikse J (2018) The use and pitfalls of intracranial vessel wall imaging: how we do it. Radiology 286(1):12–28. https://doi.org/10.1148/radiol.2017162096
Balin BJ, Broadwell RD, Salcman M, El-Kalliny M (1986) Avenues for entry of peripherally administered protein to the central nervous system in mouse, rat, and squirrel monkey. J Comp Neurol 251(2):260–280. https://doi.org/10.1002/cne.902510209
Melin E, Ringstad G, Valnes LM, Eide PK (2023) Human parasagittal dura is a potential neuroimmune interface. Commun Biol 6(1):260. https://doi.org/10.1038/s42003-023-04634-3
Albayram MS, Smith G, Tufan F, Tuna IS, Bostanciklioglu M, Zile M, Albayram O (2022) Non-invasive MR imaging of human brain lymphatic networks with connections to cervical lymph nodes. Nat Commun 13(1):203. https://doi.org/10.1038/s41467-021-27887-0
Ringstad G, Eide PK (2023) The pitfalls of interpreting hyperintense FLAIR signal as lymph outside the human brain. Nat Commun 14(1):4913. https://doi.org/10.1038/s41467-023-40508-2
Keser Z, Smith G, Cagil E, Tufan F, Albayram O, Albayram MS (2023) High-resolution MRI to noninvasively characterize drainage around the carotid artery into the cervical lymph nodes. J Neuroimaging 33(1):102–108. https://doi.org/10.1111/jon.13056
Naganawa S, Kato Y, Yoshida T, Sone M (2023) Fluid signal suppression characteristics of 3D-FLAIR with a T2 selective inversion pulse in the skull base. Nat Commun 14(1):4915. https://doi.org/10.1038/s41467-023-40507-3
Yagmurlu K, Sokolowski J, Soldozy S, Norat P, Cirak M, Tvrdik P et al (2022) A subset of arachnoid granulations in humans drain to the venous circulation via intradural lymphatic vascular channels. J Neurosurg 136(3):917–926. https://doi.org/10.3171/2021.2.JNS204455
Kutomi O, Takeda S (2020) Identification of lymphatic endothelium in cranial arachnoid granulation-like dural gap. Microscopy (Oxf) 69(6):391–400. https://doi.org/10.1093/jmicro/dfaa038
Eide PK, Vatnehol SAS, Emblem KE, Ringstad G (2018) Magnetic resonance imaging provides evidence of glymphatic drainage from human brain to cervical lymph nodes. Sci Rep 8(1):7194. https://doi.org/10.1038/s41598-018-25666-4
Ohashi T, Naganawa S, Ogawa E, Katagiri T, Kuno K (2019) Signal intensity of the cerebrospinal fluid after intravenous administration of gadolinium-based contrast agents: strong contrast enhancement around the vein of labbe. Magnetic Reson Med Sci: MRMS 18(3):194–199. https://doi.org/10.2463/mrms.mp.2018-0043
Naganawa S, Nakane T, Kawai H, Taoka T (2019) Age dependence of gadolinium leakage from the cortical veins into the cerebrospinal fluid assessed with whole Brain 3D-real inversion recovery MR imaging. Magn Reson Med Sci: MRMS 18(2):163–169. https://doi.org/10.2463/mrms.mp.2018-0053
Naganawa S, Ito R, Taoka T, Yoshida T, Sone M (2020) The space between the pial sheath and the cortical venous wall may connect to the meningeal lymphatics. Magn Reson Med Sci: MRMS 19(1):1–4. https://doi.org/10.2463/mrms.bc.2019-0099
Cassella JP, Lawrenson JG, Firth JA (1997) Development of endothelial paracellular clefts and their tight junctions in the pial microvessels of the rat. J Neurocytol 26(8):567–575. https://doi.org/10.1023/A:1015438624949
Ringstad G, Valnes LM, Vatnehol SAS, Pripp AH, Eide PK (2023) Prospective T1 mapping to assess gadolinium retention in brain after intrathecal gadobutrol. Neuroradiology 65(9):1321–1331. https://doi.org/10.1007/s00234-023-03198-7
Petitclerc L, Hirschler L, Wells JA, Thomas DL, van Walderveen MAA, van Buchem MA, van Osch MJP (2021) Ultra-long-TE arterial spin labeling reveals rapid and brain-wide blood-to-CSF water transport in humans. Neuroimage 245:118755. https://doi.org/10.1016/j.neuroimage.2021.118755
Deike-Hofmann K, Reuter J, Haase R, Paech D, Gnirs R, Bickelhaupt S et al (2019) Glymphatic pathway of gadolinium-based contrast agents through the brain: overlooked and misinterpreted. Invest Radiol 54(4):229–237. https://doi.org/10.1097/RLI.0000000000000533
Ohashi T, Naganawa S, Iwata S, Kuno K (2021) Distribution of gadolinium-based contrast agent after leaking into the cerebrospinal fluid: comparison between the cerebral cisterns and the lateral ventricles. Magn Reson Med Sci: MRMS 20(2):175–181. https://doi.org/10.2463/mrms.mp.2020-0016
Naganawa S, Nakane T, Kawai H, Taoka T, Kawaguchi H, Maruyama K et al (2020) Detection of IV-gadolinium leakage from the cortical veins into the CSF using MR fingerprinting. Magn Reson Med Sci: MRMS 19(2):141–146. https://doi.org/10.2463/mrms.mp.2019-0048
Meng Y, Abrahao A, Heyn CC, Bethune AJ, Huang Y, Pople CB et al (2019) Glymphatics visualization after focused ultrasound-induced blood-brain barrier opening in humans. Ann Neurol 86(6):975–980. https://doi.org/10.1002/ana.25604
Mollgard K, Beinlich FRM, Kusk P, Miyakoshi LM, Delle C, Pla V et al (2023) A mesothelium divides the subarachnoid space into functional compartments. Science 379(6627):84–88. https://doi.org/10.1126/science.adc8810
Pietila R, Del Gaudio F, He L, Vazquez-Liebanas E, Vanlandewijck M, Muhl L et al (2023) Molecular anatomy of adult mouse leptomeninges. Neuron. https://doi.org/10.1016/j.neuron.2023.09.002
Mapunda JA, Pareja J, Vladymyrov M, Bouillet E, Helie P, Pleskac P et al (2023) VE-cadherin in arachnoid and pia mater cells serves as a suitable landmark for in vivo imaging of CNS immune surveillance and inflammation. Nat Commun 14(1):5837. https://doi.org/10.1038/s41467-023-41580-4
Edsbagge M, Tisell M, Jacobsson L, Wikkelso C (2004) Spinal CSF absorption in healthy individuals. Am J Physiol Regul Integr Comp Physiol 287(6):R1450–R1455. https://doi.org/10.1152/ajpregu.00215.2004
Antila S, Karaman S, Nurmi H, Airavaara M, Voutilainen MH, Mathivet T et al (2017) Development and plasticity of meningeal lymphatic vessels. J Exp Med 214(12):3645–3667. https://doi.org/10.1084/jem.20170391
Jacob L, Boisserand LSB, Geraldo LHM, de Brito NJ, Mathivet T, Antila S et al (2019) Anatomy and function of the vertebral column lymphatic network in mice. Nat Commun 10(1):4594. https://doi.org/10.1038/s41467-019-12568-w
Ma Q, Decker Y, Muller A, Ineichen BV, Proulx ST (2019) Clearance of cerebrospinal fluid from the sacral spine through lymphatic vessels. J Exp Med 216(11):2492–2502. https://doi.org/10.1084/jem.20190351
Vinje V, Zapf B, Ringstad G, Eide PK, Rognes ME, Mardal KA (2023) Human brain solute transport quantified by glymphatic MRI-informed biophysics during sleep and sleep deprivation. Fluids Barriers CNS 20(1):62. https://doi.org/10.1186/s12987-023-00459-8
Cugurra A, Mamuladze T, Rustenhoven J, Dykstra T, Beroshvili G, Greenberg ZJ et al (2021) Skull and vertebral bone marrow are myeloid cell reservoirs for the meninges and CNS parenchyma. Science 373:6553. https://doi.org/10.1126/science.abf7844
Pulous FE, Cruz-Hernandez JC, Yang C, Kaya Z, Paccalet A, Wojtkiewicz G et al (2022) Cerebrospinal fluid can exit into the skull bone marrow and instruct cranial hematopoiesis in mice with bacterial meningitis. Nat Neurosci 25(5):567–576. https://doi.org/10.1038/s41593-022-01060-2
Kolabas ZI, Kuemmerle LB, Perneczky R, Forstera B, Ulukaya S, Ali M et al (2023) Distinct molecular profiles of skull bone marrow in health and neurological disorders. Cell 186(17):3706–25 e29. https://doi.org/10.1016/j.cell.2023.07.009
Rustenhoven J (2021) A privileged brain. Science 374(6567):548. https://doi.org/10.1126/science.abl7122
Banks WA, Kovac A, Majerova P, Bullock KM, Shi M, Zhang J (2017) Tau proteins cross the blood-brain barrier. J Alzheimers Dis 55(1):411–419. https://doi.org/10.3233/JAD-160542
Zou W, Pu T, Feng W, Lu M, Zheng Y, Du R et al (2019) Blocking meningeal lymphatic drainage aggravates Parkinson’s disease-like pathology in mice overexpressing mutated alpha-synuclein. Transl Neurodegener 8:7. https://doi.org/10.1186/s40035-019-0147-y
Roberts KF, Elbert DL, Kasten TP, Patterson BW, Sigurdson WC, Connors RE et al (2014) Amyloid-beta efflux from the central nervous system into the plasma. Ann Neurol 76(6):837–844. https://doi.org/10.1002/ana.24270
Feng W, Zhang Y, Wang Z, Xu H, Wu T, Marshall C et al (2020) Microglia prevent beta-amyloid plaque formation in the early stage of an Alzheimer’s disease mouse model with suppression of glymphatic clearance. Alzheimers Res Ther 12(1):125. https://doi.org/10.1186/s13195-020-00688-1
Eide PK, Mariussen E, Uggerud H, Pripp AH, Lashkarivand A, Hassel B et al (2021) Clinical application of intrathecal gadobutrol for assessment of cerebrospinal fluid tracer clearance to blood. JCI Insight. https://doi.org/10.1172/jci.insight.147063
Edeklev CS, Halvorsen M, Lovland G, Vatnehol SAS, Gjertsen O, Nedregaard B et al (2019) Intrathecal use of gadobutrol for glymphatic MR imaging: prospective safety study of 100 patients. AJNR Am J Neuroradiol 40(8):1257–1264. https://doi.org/10.3174/ajnr.A6136
Halvorsen M, Edeklev CS, Fraser-Green J, Lovland G, Vatnehol SAS, Gjertsen O et al (2021) Off-label intrathecal use of gadobutrol: safety study and comparison of administration protocols. Neuroradiology 63(1):51–61. https://doi.org/10.1007/s00234-020-02519-4
Sperre A, Karsrud I, Rodum AHS, Lashkarivand A, Valnes LM, Ringstad G, Eide PK (2023) Prospective safety study of intrathecal gadobutrol in different doses. AJNR Am J Neuroradiol 44(5):511–516. https://doi.org/10.3174/ajnr.A7841
Patel M, Atyani A, Salameh JP, McInnes M, Chakraborty S (2020) Safety of intrathecal administration of gadolinium-based contrast agents: a systematic review and meta-analysis. Radiology 297(1):75–83. https://doi.org/10.1148/radiol.2020191373
Hovd MH, Mariussen E, Uggerud H, Lashkarivand A, Christensen H, Ringstad G, Eide PK (2022) Population pharmacokinetic modeling of CSF to blood clearance: prospective tracer study of 161 patients under work-up for CSF disorders. Fluids Barriers CNS 19(1):55. https://doi.org/10.1186/s12987-022-00352-w
Xiang J, Hua Y, Xi G, Keep RF (2023) Mechanisms of cerebrospinal fluid and brain interstitial fluid production. Neurobiol Dis 183:106159. https://doi.org/10.1016/j.nbd.2023.106159
Eide PK, Lashkarivand A, Pripp A, Valnes LM, Hovd MH, Ringstad G et al (2023) Plasma neurodegeneration biomarker concentrations associate with glymphatic and meningeal lymphatic measures in neurological disorders. Nat Commun 14(1):2084. https://doi.org/10.1038/s41467-023-37685-5
Eide PK, Lashkarivand A, Pripp AH, Valnes LM, Hovd M, Ringstad G et al (2023) Mechanisms behind changes of neurodegeneration biomarkers in plasma induced by sleep deprivation. Brain Commun. https://doi.org/10.1093/braincomms/fcad343
Funding
None.
Author information
Authors and Affiliations
Contributions
Geir Ringstad provided the first version of the manuscript. Both authors (Geir Ringstad and Per Kristian Eide) made revisions to the draft and have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Geir Ringstad and Per Kristian Eide are shareholders in BrainWideSolutions AS, Oslo, Norway, which is a holder of patent US 11,272,841.
Ethical approval
Not applicable (review manuscript).
Consent to participate
Not applicable (review manuscript).
Consent for publication
Not applicable (review manuscript).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ringstad, G., Eide, P.K. Glymphatic-lymphatic coupling: assessment of the evidence from magnetic resonance imaging of humans. Cell. Mol. Life Sci. 81, 131 (2024). https://doi.org/10.1007/s00018-024-05141-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-024-05141-2