Abstract
Purpose
The aim of this study was to investigate a first-site-metastasis pattern (FSMP) in unresectable stage III NSCLC after concurrent chemoradiotherapy (cCRT) with or without immune checkpoint inhibition (ICI).
Methods
We defined three patient subgroups according to the year of initial multimodal treatment: A (2011–2014), B (2015–2017) and C (2018–2020). Different treatment-related parameters were analyzed. Observed outcome parameters were brain metastasis-free survival (BMFS), extracranial distant metastasis-free survival (ecDMFS) and distant metastasis-free survival (DMFS).
Results
136 patients treated between 2011 and 2020 were included with ≥ 60.0 Gy total dose and concurrent chemotherapy (cCRT); thirty-six (26%) received ICI. Median follow-up was 49.7 (range:0.7–126.1), median OS 31.2 (95% CI:16.4–30.3) months (23.4 for non-ICI vs not reached for ICI patients, p = 0.001).
Median BMFS/ecDMFS/DMFS in subgroups A, B and C was 14.9/16.3/14.7 months, 20.6/12.9/12.7 months and not reached (NR)/NR/36.4 months (p = 0.004/0.001/0.016).
For cCRT+ICI median BMFS was 53.1 vs. 19.1 months for cCRT alone (p = 0.005). Median ecDMFS achieved 55.2 vs. 17.9 (p = 0.003) and median DMFS 29.5 (95% CI: 1.4–57.6) vs 14.93 (95% CI:10.8–19.0) months (p = 0.031), respectively.
Multivariate analysis showed that age over 65 (HR:1.629; p = 0.036), GTV ≥ 78 cc (HR: 2.100; p = 0.002) and V20 ≥ 30 (HR: 2.400; p = 0.002) were negative prognosticators for BMFS and GTV ≥ 78 cc for ecDMFS (HR: 1.739; p = 0.027).
After onset of brain metastasis (BM), patients survived 13.3 (95% CI: 6.4–20.2) months and 8.6 months (95% CI: 1.6–15.5) after extracranial-distant-metastasis (ecDM). Patients with ecDM as FSMP reached significantly worse overall survival of 22.1 (range:14.4–29.8) vs. 40.1 (range:18.7–61.3) months (p = 0.034) in the rest of cohort. In contrast, BM as FSMP had no impact on OS.
Conclusion
This retrospective analysis of inoperable stage III NSCLC patients revealed that age over 65, V20 ≥ 30 and GTV ≥ 78 cc were prognosticators for BMFS and GTV ≥ 78 cc for ecDMFS. ICI treatment led to a significant improvement of BMFS, ecDMFS and DMFS. ecDM as FSMP was associated with significant deterioration of OS, whereas BM as FSMP was not.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Regarding cancer-related mortality, lung cancer is the most common cause worldwide [1]. Inoperable stage III NSCLC is a highly heterogeneous disease with varying macroscopic tumor extensions, and therefore, patients’ prognosis depends heavily on multiple patient and treatment factors. After multimodality treatment, historically, the five-year survival rate of patients has been as low as ten to thirty percent [2,3,4]. The standard of care for patients with unresectable stage III non-small cell lung cancer (NSCLC) is concurrent chemoradiotherapy [5] followed by maintenance therapy with the programmed cell death ligand 1 (PD-L1) inhibitor durvalumab [6]. The addition of immune checkpoint inhibition (ICI) to the multimodality treatment strategy has led to a significant improvement in patient outcomes, as demonstated by the unprecedented results of the PACIFIC trial [7, 8].
However, distant failure remains a burden for patients with unresectable NSCLC: Over time 30% of affected patients develop brain metastases (BM) [9], but there is encouraging evidence that anti-PD-1/PD-L1 agents may have a beneficial impact on their development [10]. Knowledge of the effects of chemoradioimmunotherapy on the development of distant metastases (DM) is limited. This retrospective analysis evaluates the impact of treatment patterns and patient-related factors on brain metastasis-free (BMFS), extracranial distant metastasis-free (ecDMFS), and distant metastasis-free survival (DMFS) after concurrent chemoradiotherapy (cCRT) with and without immune checkpoint inhibitors (ICI).
Patients and methods
A total of 189 consecutive patients with stage III NSCLC were assessed for eligibility in 2021. The retrospective study and analysis of individual patient data were approved by the local ethics committee (reference number: 17-230). Informed consent to the treatment and data collection for research purposes was obtained from all patients.
Out of the 189 patients treated between February 2011 and November 2020, 136 (71.9%) were included. All selected patients had been diagnosed with unresectable stage III A–C (UICC 8th edition) NSCLC. They received concurrent cCRT with and without ICI. This included either simultaneous and maintenance treatment with the PD‑1 inhibitor nivolumab as part of the phase II ETOP 6–14 NICOLAS study [11, 12], or maintenance treatment with the PD-L1 inhibitor durvalumab in accordance with the PACIFIC trial [7].
The cCRT consisted of conventional fractionated thoracic radiotherapy (TRT) with a total dose of ≥ 60 Gy and platinum-based chemotherapy (cisplatin/carboplatin in combination with vinorelbine/pemetrexed). To assess improvements over time, three subgroups of patients were classified according to the year of the first TRT: Subgroup A (2011–2014), Subgroup B (2015–2017), and Subgroup C (2018–2020).
All survival parameters were calculated from the last day of TRT only considering the first site of failure (FSMP), including brain metastasis-free survival (BMFS), extracranial distant metastasis-free survival (ecDMFS) and distant metastasis-free survival (DMFS). Patients with local recurrence prior to distant failure or with simultaneous multifocal progression were not included in the BM, ecDM or DM subgroup, Patient and treatment-related characteristics are displayed in Table 1.
Baseline positron emission tomography (PET) and computed tomography (CT) staging were performed in 133 (97.8%) patients before initiation of multimodality treatment to improve contouring quality [13, 14] and cranial contrast-enhanced MRI was acquired in 88 (64.7%) patients, while all other patients obtained cranial contrast-enhanced CT.
cCRT was recommended in a multidisciplinary tumor board for each patient. An Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0–1 and adequate lung function (diffusing capacity of the lung for carbon monoxide corrected for hemoglobin (DLCO) ≥ 40%, forced expiratory volume in 1 s (FEV1) ≥ 1 L) were required.
Patients were positioned supine with their arms held overhead by WingSTEP™ (Innovative Technologie Voelp, Innsbruck, Austria). Target volumes were defined according to an internal protocol closely following the later published guidelines of the European Society for Radiotherapy and Oncology-Advisory Committee on Radiation Oncology Practice (ESTRO-ACROP) [15] and based conventional planning CT and PET-CT.
Gross tumor volume (GTV) and planning target volume (PTV) were transcribed from the original treatment plans. To differentiate small vs. large volumes, we chose median dichotomization for GTV (78.0 cc) and split PTV in < vs ≥ 700 cc based on literature research of previously published data [16,17,18,19].
Prior to the introduction of intensity-modulated radiation therapy (IMRT), TRT with 50 Gy in 2 Gy single-dose fractions, followed by a sequential 16-Gy boost, was used. After implementation of IMRT, TRT consisted of 30 fractions with simultaneous integrated boost (SIB) of 2.0/2.12 Gy to the lymph nodes (LN)/GTV of the primary tumor (PT) with a total dose of 60.0/63.6 Gy.
Follow-up was performed every three months for the first two years after treatment, every six months for the next two years, and annually thereafter. Routine blood tests, pulmonary function tests, clinical examinations, and imaging such as PET-CT or CT scans were analyzed, response was assessed according to RECIST 1.1. Additional diagnostic measures such as contrast-enhanced magnetic resonance imaging (MRI) and bone scintigraphy, were performed when deemed necessary.
Brain metastases (BM), extracranial distant metastases (ecDM) and distant metastases (DM) were documented with PET-CT, CT or MRI scans. Histological confirmation of progressive disease was not obligatory.
The impact of each parameter on BMFS, ecDMFS and DMFS was analyzed by means of Kaplan-Meier analysis using the log-rank test. Multivariable Cox-regression analysis was performed with all parameters which had shown to be significant (p < 0.05) in univariate analysis. All analyses including univariate and multivariate analysis were performed using SPSS version 28 (IBM; Armonk, New York, USA).
Results
A total of 136 consecutive patients with unresectable stage IIIA–C NSCLC (UICC 8th edition) received initial treatment between 2011 and 2020 and were eligible for this analysis. A summary of patient- and tumor-characteristics is shown in Table 1.
In the entire cohort, the median follow-up was 49.7 (range: 0.7–126.1) months. Seventy-eight (57%) patients were older than 65 years and the mean age was 66.9 (range 33.6–82.5) years. Table 2 shows OS, BMFS, ecDMFS and DMFS for the entire cohort and each subgroup.
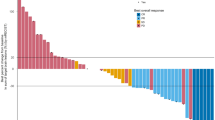
The median overall survival for patients with DM as FSMP vs. without DM was 25.5 (95% CI: 20.1–31.0) vs. 64.9 (95% CI: 34.3–95.4) months (p = 0.037). Median survival after onset of DM as FSMP was 11.9 months (range: 1.0–121). The overall survival of patients with isolated BM as FSMP was 27.4 (95% CI: 20.6–34.1) vs. 32.9 (95% CI: 11.7–54.0) months without BM (p = 0.657). Patients survived a median of 13.3 months after the diagnosis of BM (range: 1.3–121). Overall survival in patients presenting with ecDM as FSMP vs. without ecDM was 22.1 (95% CI: 14.4–29.8) vs. 40.1 (95% CI: 18.7–61.3) months (p = 0.034). These patients survived a median of 8.6 months (range: 1–98months). See also Fig. 1.
In univariate analysis for the entire cohort, stratification into treatment groups A, B, and C was significantly associated with BMFS (p = 0.004), ecDMFS (p = 0.001), and DMFS (p = 0.016). Median BMFS was 14.9 (95% CI: 9.9–19.9) months in subgroup A, 20.6 (95% CI: 9.6–31.3) months in subgroup B and was not reached in subgroup C. The median ecDMFS was 16.3 (95% CI: 11.1–21.6), 12.9 (95% CI: 6.4–19.5) months and not reached, for groups A, B and C, respectively. Median DMFS for subgroups A and B was 14.7 (95% CI: 9.2–20.6) and 12.7 (95% CI: 5.6–19.9) months, and for subgroup C was 36.4 (95% CI: 21.8–51.1) months (see also supplementary figures 1–3).
Both, patients treated with simultaneous + maintenance nivolumab and patients treated with maintenance durvalumab were included in this retrospective cohort. We found no significant differences for OS (p = 0.841), PFS (p = 0.764), DMFS (p = 0.919), ecDMFS (p = 0.628) and BMFS (p = 0.668) between these subgroups. Therefore, we pooled them as an ICI cohort versus patients treated without ICI.
In patients who received ICI, median BMFS was 20.6 (95% CI: 14.4–26.9) months vs. 17.6 (95% CI: 12.9–22.3) for those without ICI (p = 0.005). Median ecDMFS was 16.9 (95% CI: 10.2–23.6) months vs. NR, respectively (p = 0.003). Median DMFS was 14.9 (95% CI 10.7–19.2) months vs. NR (p = 0.031). Median BMFS of patients over 65 was 20.1 (95% CI: 12.6–27.5) months vs. 25.6 (95% CI: 8.7–42.4) months in patients under the age of 65 (p = 0.016).
Patients who had a cMRI scan prior to treatment showed no significant difference in DMFS (p = 0.295) or BMFS (p = 0.189).
We analyzed the use of VMAT and observed significantly longer metastasis free survival in patients that received VMAT vs. patients with 3D-TRT. Median BMFS was 52.2 (95% CI: 22.1–82.3) vs. 14.9 (95% CI: 9.4–20.4) months (p = 0.001). Median ecDMFS was not reached vs. 14.9 (95% CI: 7.9–21.9) months (p = 0.001) and median DMFS was 25.6 (95% CI: 9.1–42.0) vs. 11.9 (95% CI: 6.7–17.2) months, respectively (p = 0.006).
GTV had significant impact on outcome in univariate analysis: DMFS was 26.3 (95% CI: 12.6–40.0) vs 10.8 (95% CI: 8.6–13.0) months (p = < 0.001), BMFS was 52.2 (95% CI: 20.2–84.2) vs 14.2 (95% CI: 10.5–17.8) months (p < 0.001) and ecDMFS was 64.9 (95% CI: 23.6–106.2) vs 12.7 (95% CI: 8.9–16.6) months (p < 0.001) for patients with GTV < 78 cc vs ≥ 78 cc, respectively. In the non-ICI cohort GTV had a significant negative impact on DMFS (p = 0.006), BMFS (p = 0.004) and ecDMFS (p = 0.007). In patients treated with ICI only BMFS (p = 0.030) was significantly impacted by GTV whereas DMFS (p = 0.074) and ecDMFS (p = 0.078) were not.
PTV ≥ 700 cc was significantly associated with shorter BMFS and ecDMFS. The median BMFS for patients with PTV ≥ 700 cc was 16.3 (95% CI: 11.0–21.7) compared to 50.3 (95% CI: 23.5–77.1) months for patients with PTV < 700 cc (p = 0.021). The median ecDMFS was 12.7 (95% CI: 8.6–16.8) vs. 55.2 (95% CI: 18.0–92.4) months (p = 0.008).
For the entire cohort, patients with a total lung V20 ≥ 30% had a median BMFS of 10.9 months (95% CI: 2.1–19.8) compared to 26.5 months (95% CI: 10.9–42.2) for patients with V20 < 30% (p = 0.001). The median ecDMFS was 10.6 months (95% CI: 6.2–14.9) vs. 27.2 months (95% CI: 15.1–39.3) for patients with V20 ≥ 30% and V20 < 30%, respectively (p = 0.005), and the median DMFS was 5.6 months (95% CI: 0.0–13.7) vs. 17.9 months (95% CI: 11.6–24.3) (p = 0.004).
In patients treated without ICI, stratification by year of treatment showed significant association with ecDMFS (p = 0.004) and DMFS (p = 0.047) and a trend toward longer BMFS (p = 0.061). In the non-ICI cohort, a significant association was found between the use of VMAT and BMFS (p = 0.021) and ecDMFS (0.013). For DMFS there was a trend (p = 0.054). The median BMFS was 14.1 months (95% CI: 10.1–18.3) for patients with PTV ≥ 700 cc and 24.7 months (95% CI: 12.1–37.4) for patients with PTV < 700 cc (p = 0.036). The median ecDMFS was 11.9 months (95% CI: 10.2–13.6) and 27.4 months (95% CI: 20.8–33.9), respectively (p = 0.006).
The median BMFS amounted to 11.2 months (95% CI: 4.6–17.8) for patients with SUVmax ≥ 13.75 vs. 21.1 months (95% CI: 13.9–28.3), for patients with SUVmax < 13.75 (p = 0.006). The median ecDMFS was found to be significantly longer in patients with a lower V20 (< 30) than those with a higher V20 (≥ 30). The same was observed for median DMFS and BMFS. Patients with higher SUVmax (≥ 13.75) also showed a trend towards shorter median DMFS. The p-values for these associations were 0.009 and 0.063, respectively.
V20 ≥ 30 was significantly associated with DMFS, ecDMFS and BMFS in the non-ICI cohort: Median BMFS was not reached for V20 ≥ 30 (p = 0.002) and V20 < 30. In Non-ICI patients, the median ecDMFS (p = 0.009) and the median DMFS (p = 0.020) were also significantly linked to V20 ≥ 30. The detailed results of the univariate analyses can be found in Table 3.
The multivariate analysis for the entire cohort included use of immune checkpoint inhibitors (ICI), PD-L1-status ≥ 1%, stratification by treatment year, age over 65, T‑stage, V20 ≥ 30, GTV ≥ 78 cc PTV ≥ 700 cc and use of VMAT.
V20 ≥ 30 was significantly associated with BMFS [HR: 2.400 (95% CI: 1.396–4.126, p = 0.002)], and DMFS [HR: 1.784 (95% CI: 1.028–3.097, p = 0.040)].
GTV ≥ 78 cc had a significant negative impact on BMFS [HR: 2.100 (95% CI: 1.396–3.358, p = 0.002)], ecDMFS [HR: 1.739 (95% CI: 1.006–2.835, p = 0.027)] and DMFS [HR: 2.394 (95% CI: 1.485–3.858, p < 0.001)].
BMFS was significantly associated with Age ≥ 65 [HR: 1.629 (95% CI: 1.031–2.572, p = 0.036)].
In patients treated without ICI, a GTV ≥ 78 cc was also associated with shorter BMFS [HR: 1.976 (95% CI: 1.181–3.308, p = 0.010)] and DMFS [HR: 2.049 (95% CI: 1.181–3.554, p = 0.011)].
SUVmax ≥ 13.75 remained a predictor of short ecDMFS [HR: 2.420 (95% CI: 1.379–4.248, p = 0.002)] V20 ≥ 30 was significantly associated with shorter ecDMFS [HR: 2.883 (95% CI: 1.512–5.497, p = 0.001)] and BMFS [HR: 2.273 (95% CI: 1.301–3.970, p = 0.004)].
For detailed results of the multivariate analysis refer to Table 4.
The median post-progression survival in the entire cohort after onset of DM was 13.3 (95% CI: 6.0–20.6) months. Patients initially treated with ICI sowed a clear trend towards longer post-DM-survival of 20.7 (95% CI: 13.9–27.6) vs 7.8 (95% CI: 4.4–11.5) months (p = 0.052). The median survival after onset of DM in subgroup A, B and C were 7.2 (95% CI: 3.1–11.3), 7.8 (95% CI: 0.0–24.2) and 18.5 (95% CI 12.1–25.0) months, respectively (p = 0.243).
After BM patients in the entire cohort survived a median of 13.3 (95% CI: 6.4–20.2) months. Median post-BM-survival was not reached in patients who received ICI in the course of their initial treatment compared to 9.5 (95% CI: 2.0–16.9) months in those without ICI (p = 0.177). For subgroups A, B and C median post-BM-survival was 9.5 (95% CI: 0.0–22.6), 7.8 (0.0–17.9) and 13.3 (95% CI: 10.7–6.0) months, respectively (p = 0.787).
The median post-ecDM-survival was 8.6 (95% CI: 1.6–15.5) months in the entire cohort and 20.7 (96% CI: 15.4–26.1) vs 6.4 (95% CI: 3.5–9.4) months for patients initially treated with vs without ICI, respectively. In subgroup A it was 4.8 (95% CI: 1.1–8.5) months, in subgroup B it was 6.8 (95% CI: 0.0–27.7) months and in subgroup C it was 18.6 (95% CI: 2.1–35.1) months (p = 0.357).
Discussion
In the past decade, important advances have been achieved in the multimodal treatment of inoperable stage III NSCLC. The introduction of VMAT has decreased the toxicity of TRT [20,21,22] and routine concurrent chemo-radiotherapy has constantly been improved [23]. The PACIFIC trial has changed the landscape of thoracic oncology with unprecedented improvements in OS and PFS [7, 8, 24]. In this comprehensive analysis of patients treated in the past decade we depict patterns of first distant failure after cCRT for Stage III NSCLC. We identified factors during initial treatment that predict patients’ risk for later onset of metastasis and analyzed impact of metastasis on OS of affected patients.
We observed no significant differences for OS, PFS, DMFS, ecDMFS and BMFS between patients treated with durvalumab vs. those treated with nivolumab, therefore we decided pool both cohorts together against patients treated without ICI to raise patient numbers.
In our cohort the median DMFS for patients treated with CRT+ICI was 29.5 months vs 14.93 months (p = 0.031) for those treated with CRT alone. This is in close accordance with the data of the PACIFIC-trial, were the median time to death or distant metastasis was reported as 28.3 months for patients treated with durvalumab vs.16.2 months for patients in the placebo arm [25]. In the LUN 14–179 trial, a time to metastatic disease or death of 30.7 months (95% CI: 18.7 to NR) was reported for patients receiving pembrolizumab after cCRT [26]. In contrast to Kishi et al., we did not observe female sex as a negative prognosticator for the onset of DM [27].
As previously described, the implementation of Durvalumab maintenance drastically improves intrathoracic control after cCRT [7, 8, 24, 28,29,30,31,32,33]. In univariate analysis for BMFS, ecDMFS and DMFS we observed vast improvements in patients treated with cCRT+ICI vs those treated with cCRT alone (p = 0.005; p = 0.003 and p = 0.031) as shown in Table 3 and Fig. 1. Kishi et al. recently reported significantly lower rates of distant metastasis in patients treated with cCRT + durvalumab vs. without durvalumab in a real-world setting [30].
BMFS and ecDMFS had a significant negative correlation with larger GTV, PTV (≥ 700 cc) and with total lung V20 ≥ 30%. This suggests that patients with initially larger initial tumor burden are more likely to experience distant failure after cCRT. The association of larger PTVs with diminished regional control has been reported frequently reported in literature [18, 19, 34, 35]. However, the impact of initial GTV and its’ shrinkage during CRT in patients that afterwards receive IO maintenance is currently unknown and should be considered for future investigations [36, 37]. Also, underdosage of large tumours or reduced margins at the discretion of the performing physicians could be an explanation of this strong negative correlation.
The study split patients into three subgroups (A 20112014, B 2015–2017 and C 2018–2020) and showed constant improvements of DMFS, BMFS and ecDMFS over the years, especially in subgroup C, which coincided with the implementation of ICItreatment Interestingly, the study also observed numerical improvements in post-DM-survival from 7.2 months in subgroup A to 15.1 months in subgroup B to 18.5 in subgroup C. Although these numerical benefits were not statistically significant, they hint towards improved post-progression therapy and we are planning further analyses on this subject [4, 38].
Patients treated with cCRT alone, who initially presented with PD-L1 expressing (≥ 1%) tumors had significantly worse DMFS in univariate analysis (p = 0.033) and showed a clear negative trend in multivariate analysis (HR: 1.666; p = 0.091). This indicates a vast improvement in outcomes for those patients after implementation of durvalumab maintenance, considering IO was almost exclusively administered in patients with PD-L1 on ≥ 1% of tumor cells due to regulations by the European medicines agency (EMA).
The importance of post progression survival (PPS) has been highlighted by Imai et al. [39]. In this study’s patients initially treated with cCRT+ICI showed numerically improved post-DM-survival compared to those who were initially treated with cCRT alone. Interestingly, BM as FSMP did not significantly impact survival, whereas ecDM lead to impaired OS. Also PPS after BM as FSMP was significantly longer than after the onset of ecDM. This could be due to the highly efficient treatment options of BM via stereotactic radiosurgery (SRS) [40]. Overall the median post progression survival achieved in our cohort (13.3 months) is 2.7 months longer compared to the 10.6 months reported by Delasos et al., but nevertheless unsatisfying [41]. It is of utmost interest to further improve treatment for patients with progression after cCRT (+/−) ICI for stage III NSCLC, especially those with ecDM as FSMP. Further trials are needed to tailor post progression therapy aiming to improve PPS.
We are planning to analyse factors for PPS, like second line TKI-therapy, ICI-therapy or chemotherapy in the near future.
Given the limitations of this retrospective study we want to acknowledge the low number of patients with PD-L1 assessment prior to treatment, lack of Next Generation Sequencing and subtotal MRI staging prior to treatment. These points should be included in all prospective trials concerning lung cancer.
Conclusion
In conclusion, this retrospective analysis of unresectable stage III NSCLC patients identified V20 ≥ 30%, GTV ≥ 78 cc, and age over 65 as strong prognosticators for BMFS and GTV ≥ 78 cc for ecDMFS. Additionally, T‑stage, V20 ≥ 30 and GTV ≥ were found to be prognosticators for DMFS. Implementation of ICI treatment resulted in a significant improvement of BMFS, ecDMFS, and DMFS. Patients with ecDM had shorter overall survival, while BM did not impact survival of affected patients compared to the entire cohort.
References
Siegel RL, Miller KD, Fuchs HE et al (2021) Statistics, 2021. CA: a cancer journal for clinicians. Cancer 71(1):7–33. https://doi.org/10.3322/caac.21654
Käsmann L, Taugner J, Eze C et al (2019) Performance Status and Its Changes Predict Outcome for Patients With Inoperable Stage III NSCLC Undergoing Multimodal Treatment. Anticancer Res 39(9):5077–5081. https://doi.org/10.21873/anticanres.13701
Taugner J, Käsmann L, Eze C et al (2019) Survival score to characterize prognosis in inoperable stage III NSCLC after chemoradiotherapy. Transl Lung Cancer Res 8(5):593–604. https://doi.org/10.21037/tlcr.2019.09.19
Taugner J, Eze C, Käsmann L et al (2020) Pattern-of-failure and salvage treatment analysis after chemoradiotherapy for inoperable stage III non-small cell lung cancer. Radiat Oncol 15(1):148. https://doi.org/10.1186/s13014-020-01590-8
(1995) Chemotherapy in non-small cell lung cancer: a meta-analysis using updated data on individual patients from 52 randomised clinical trials. Non-small Cell Lung Cancer Collaborative Group. BMJ : British Medical Journal 311(7010): 899–909
Remon J, Soria J‑C, Peters S (2021) Early and locally advanced non-small-cell lung cancer: an update of the ESMO Clinical Practice Guidelines focusing on diagnosis, staging, systemic and local therapy. Annals of oncology : official journal of the European Society for Medical Oncology 32(12): 1637–1642. https://doi.org/10.1016/j.annonc.2021.08.1994
Antonia SJ, Villegas A, Daniel D et al (2017) Durvalumab after Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. N Engl J Med 377(20):1919–1929. https://doi.org/10.1056/NEJMoa1709937
Spigel DR, Faivre-Finn C, Gray JE et al (2022) Five-Year Survival Outcomes From the PACIFIC Trial: Durvalumab After Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. J Clin Oncol 40(12):1301–1311. https://doi.org/10.1200/JCO.21.01308
Rosset M, Reifegerste D, Baumann E et al. (2019) Langzeittrends beim Krebsinformationsdienst (KID) des Deutschen Krebsforschungszentrums (DKFZ) : Eine Analyse der Anfragen von Patientinnen und Patienten sowie Angehörigen von 1992 bis 2016 (Trends in cancer information services over 25 years : An analysis of inquiries from patients and relatives made to the Cancer Information Service of the German Cancer Research Center from 1992to 2016). Bundesgesundheitsblatt, Gesundheitsforschung, Gesundheitsschutz 62(9): 1120–1128. https://doi.org/10.1007/s00103-019-02996-w
Li W, Jiang J, Huang L et al (2022) Efficacy of PD-1/L1 inhibitors in brain metastases of non-small-cell lung cancer: pooled analysis from seven randomized controlled trials. Future Oncol 18(3):403–412. https://doi.org/10.2217/fon-2021-0795
Peters S, Felip E, Dafni U et al (2019) Safety evaluation of nivolumab added concurrently to radiotherapy in a standard first line chemo-radiotherapy regimen in stage III non-small cell lung cancer-The ETOP NICOLAS trial. Lung Cancer 133:83–87. https://doi.org/10.1016/j.lungcan.2019.05.001
Peters S, Felip E, Dafni U et al (2021) Progression-Free and Overall Survival for Concurrent Nivolumab With Standard Concurrent Chemoradiotherapy in Locally Advanced Stage IIIA‑B NSCLC: Results From the European Thoracic Oncology Platform NICOLAS Phase II Trial (European Thoracic Oncology Platform 6–14). J Thorac Oncol 16(2):278–288. https://doi.org/10.1016/j.jtho.2020.10.129
Eze C, Schmidt-Hegemann N‑S, Sawicki LM et al (2021) PET/CT imaging for evaluation of multimodal treatment efficacy and toxicity in advanced NSCLC-current state and future directions. Eur J Nucl Med Mol Imaging 48(12):3975–3989. https://doi.org/10.1007/s00259-021-05211-8
Manapov F, Eze C, Holzgreve A et al (2022) PET/CT for Target Delineation of Lung Cancer Before Radiation Therapy. Semin Nucl Med 52(6):673–680. https://doi.org/10.1053/j.semnuclmed.2022.05.003
Nestle U, de Ruysscher D, Ricardi U et al (2018) ESTRO ACROP guidelines for target volume definition in the treatment of locally advanced non-small cell lung cancer. Radiother Oncol 127(1):1–5. https://doi.org/10.1016/j.radonc.2018.02.023
Wiersma TG, Dahele M, Verbakel WFAR et al (2013) Concurrent chemoradiotherapy for large-volume locally-advanced non-small cell lung cancer. Lung Cancer 80(1):62–67. https://doi.org/10.1016/j.lungcan.2013.01.006
van Reij EJF, Dahele M, van de Ven PM et al (2014) Changes in non-surgical management of stage III non-small cell lung cancer at a single institution between 2003 and 2010. Acta Oncol 53(3):316–323. https://doi.org/10.3109/0284186X.2013.819995
Taugner J, Käsmann L, Karin M et al (2022) Planning target volume as a predictor of disease progression in inoperable stage III non-small cell lung cancer patients treated with chemoradiotherapy and concurrent and/or sequential immune checkpoint inhibition. Invest New Drugs 40(1):163–171. https://doi.org/10.1007/s10637-021-01143-0
Karin M, Taugner J, Käsmann L et al (2020) Association of Planning Target Volume with Patient Outcome in Inoperable Stage III NSCLC Treated with Chemoradiotherapy: A Comprehensive Single-Center Analysis. Cancers. https://doi.org/10.3390/cancers12103035
Dubaere E, Goffaux M, Wanet M et al (2019) Long term outcome after 48 Gy stereotactic ablative body radiotherapy for peripheral stage I non-small cell lung cancer. Bmc Cancer 19(1):639. https://doi.org/10.1186/s12885-019-5863-2
Peng J, Pond G, Donovan E et al (2020) A Comparison of Radiation Techniques in Patients Treated With Concurrent Chemoradiation for Stage III Non-Small Cell Lung Cancer. Int J Radiat Oncol Biol Phys 106(5):985–992. https://doi.org/10.1016/j.ijrobp.2019.12.027
Merrow CE, Wang IZ, Podgorsak MB (2012) A dosimetric evaluation of VMAT for the treatment of non-small cell lung cancer. J Applied Clin Med Phys 14(1):4110. https://doi.org/10.1120/jacmp.v14i1.4110
Flentje M, Huber RM, Engel-Riedel W et al. (2016) GILT—A randomised phase III study of oral vinorelbine and cisplatin with concomitant radiotherapy followed by either consolidation therapy with oral vinorelbine and cisplatin or best supportive care alone in stage III non-small cell lung cancer. Strahlentherapie und Onkologie : Organ der Deutschen Rontgengesellschaft … [et al] 192(4): 216–222. https://doi.org/10.1007/s00066-016-0941-8
Gray JE, Villegas A, Daniel D et al (2020) Three-Year Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC-Update from PACIFIC. J Thorac Oncol 15(2):288–293. https://doi.org/10.1016/j.jtho.2019.10.002
Antonia SJ, Villegas A, Daniel D et al (2018) Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC. N Engl J Med 379(24):2342–2350. https://doi.org/10.1056/NEJMoa1809697
Durm GA, Jabbour SK, Althouse SK et al (2020) A phase 2 trial of consolidation pembrolizumab following concurrent chemoradiation for patients with unresectable stage III non-small cell lung cancer: Hoosier Cancer Research Network LUN 14-179. Cancer 126(19): 4353–4361. https://doi.org/10.1002/cncr.33083
Kishi N, Matsuo Y, Shintani T et al (2023) Recurrence patterns and progression-free survival after chemoradiotherapy with or without consolidation durvalumab for stage III non-small cell lung cancer. JRR 64(1):142–153. https://doi.org/10.1093/jrr/rrac057
Offin M, Shaverdian N, Rimner A et al (2020) Clinical outcomes, local-regional control and the role for metastasis-directed therapies in stage III non-small cell lung cancers treated with chemoradiation and durvalumab. Radiother Oncol 149:205–211. https://doi.org/10.1016/j.radonc.2020.04.047
Jung HA, Noh JM, Sun J‑M et al (2020) Real world data of durvalumab consolidation after chemoradiotherapy in stage III non-small-cell lung cancer. Lung Cancer 146:23–29. https://doi.org/10.1016/j.lungcan.2020.05.035
Käsmann L, Eze C, Taugner J et al (2020) Implementation of durvalumab maintenance treatment after concurrent chemoradiotherapy in inoperable stage III non-small cell lung cancer (NSCLC)—a German radiation oncology survey. Transl Lung Cancer Res 9(2):288–293. https://doi.org/10.21037/tlcr.2020.03.25
Taugner J, Käsmann L, Eze C et al (2021) Real-world prospective analysis of treatment patterns in durvalumab maintenance after chemoradiotherapy in unresectable, locally advanced NSCLC patients. Invest New Drugs 39(4):1189–1196. https://doi.org/10.1007/s10637-021-01091-9
Taugner J, Käsmann L, Eze C et al (2021) Durvalumab after Chemoradiotherapy for PD-L1 Expressing Inoperable Stage III NSCLC Leads to Significant Improvement of Local-Regional Control and Overall Survival in the Real-World Setting. Cancers. https://doi.org/10.3390/cancers13071613
Flörsch B, Taugner J, Käsmann L et al (2022) Treatment patterns and prognosis of patients with inoperable stage III NSCLC after completion of concurrent chemoradiotherapy ± immune checkpoint inhibition: a decade-long single-center historical analysis. J Cancer Res Clin Oncol. https://doi.org/10.1007/s00432-022-04174-z
Morgensztern D, Waqar S, Subramanian J et al (2012) Prognostic significance of tumor size in patients with stage III non-small-cell lung cancer: a surveillance, epidemiology, and end results (SEER) survey from 1998 to 2003. J Thorac Oncol 7(10):1479–1484. https://doi.org/10.1097/JTO.0b013e318267d032
Nagasaka M, Abdallah N, Crosby M et al. (2019) A retrospective study evaluating the pretreatment tumor volume (PTV) in non-small cell lung cancer (NSCLC) as a predictor of response to program death‑1 (PD-1) inhibitors. Lung Cancer: Targets and Therapy 10: 95–105. https://doi.org/10.2147/LCTT.S219886
Käsmann L, Niyazi M, Blanck O et al. (2018) Prädiktiver und prognostischer Wert des Tumorvolumens und seiner Veränderungen während radikaler Strahlentherapie beim nicht-kleinzelligen Bronchialkarzinom im Stadium III : Ein systematischer Review (Predictive and prognostic value of tumor volume and its changes during radical radiotherapy of stage III non-small cell lung cancer : A systematic review). Strahlentherapie und Onkologie : Organ der Deutschen Rontgengesellschaft … [et al] 194(2): 79–90. https://doi.org/10.1007/s00066-017-1221-y
Ostheimer C, Mäurer M, Ebert N et al. (2021) Prognostic impact of gross tumor volume during radical radiochemotherapy of locally advanced non-small cell lung cancer-results from the NCT03055715 multicenter cohort study of the Young DEGRO Trial Group. Strahlentherapie und Onkologie : Organ der Deutschen Rontgengesellschaft … [et al] 197(5): 385–395. https://doi.org/10.1007/s00066-020-01727-4
Garg S, Gielda BT, Kiel K et al (2014) Patterns of locoregional failure in stage III non-small cell lung cancer treated with definitive chemoradiation therapy. Pract Radiat Oncol 4(5):342–348. https://doi.org/10.1016/j.prro.2013.12.002
Imai H, Kaira K, Minato K (2017) Clinical significance of post-progression survival in lung cancer. Thorac Cancer 8(5):379–386. https://doi.org/10.1111/1759-7714.12463
Ernani V, Stinchcombe TE (2019) Management of Brain Metastases in Non-Small-Cell Lung Cancer. JOP 15(11):563–570. https://doi.org/10.1200/JOP.19.00357
Delasos L, Wei W, Hassan KA et al (2023) Clinical outcomes with pembrolizumab-based therapies in recurrent/refractory NSCLC after chemoradiation and consolidative durvalumab. Clin Lung Cancer. https://doi.org/10.1016/j.cllc.2023.04.008
Acknowledgements
The piece has not been previously published and is not under consideration elsewhere. The persons listed as authors have given their approval for the submission.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
KH, JT and FM analyzed and interpreted the data, performed the statistical analysis and wrote the manuscript. SM, LK and BF helped with the statistical analysis and editing the manuscript. All authors helped in drafting the manuscript. All authors read and gave their stamp of approval for the submission of the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
A. Tufman receives travel support from Pfizer and Amgen. C. Belka eceives grants or contracts from any entity but not related to this manuscript from Viewray, Brainlab and ELEKTA. C. Belka receives honoraria from BMS, ROCHE, MERCK, Astrazeneca and Viewray. C. Belka receives support for attending meetings and/or travel from BMS, ROCHE, MERCK, Astrazeneca and Viewray. C. Belka receives BMS, ROCHE, MERCK, Astrazeneca and Viewray. C. Belka serves in a fiduciary role in the ESTRO. C. Eze receives grants or contracts from any entity but not related to this manuscript from German Cancer Aid. C. Eze receives consulting fees from Novartis. C. Eze receives support for attending meetings and/or travel from Novartis. F. Manapov receives an unrestricted Research Institutional Grant from AstraZeneca. F. Manapov receives honoraria from AstraZeneca, Novartis, Roche, Lilly, Elekta and Brainlab. F. Manapov serves in the advisory board of AstraZeneca, Novartis. L. Käsmann receives honoraria from AMGEN. N. Reinmuth reports receiving honoraria for speaker activities and participation in advisory boards for AbbVie, AstraZeneca, Bristol-Myers Squibb, Boehringer Ingelheim, Merck Sharp & Dohme, Pfizer, Roche, and Takeda. K. Hofstetter, J. Taugner, S. Mansoorian, B. Flörsch and T. Duell declare that they have no competing interests.
Ethical standards
All patients gave express written informed consent. This retrospective analysis is in compliance with the principles of the Declaration of Helsinki and its subsequent amendments. This work was approved by the Ethics Committee of the Ludwig Maximilian University of Munich (reference number: 17-230).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
K. Hofstetter und J. Taugner contributed equally to the manuscript.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplementary Information
66_2023_2175_MOESM1_ESM.tif
sFigure 1: Kaplan–Meier curves of brain metastasis-free survival (BMFS), for all patients (left) vs. patients without ICI (right) stratified by treatment year groups
66_2023_2175_MOESM2_ESM.tif
sFigure 2: Kaplan–Meier curves of extracranial distant metastasis-free survival (ecDMFS) for all patients (left) vs. patients without ICI (right) stratified by treatment year groups
66_2023_2175_MOESM3_ESM.tif
sFigure 3: Kaplan–Meier curves of distant metastasis-free survival (DMFS) for all patients (left) vs. patients without ICI (right) stratified by treatment year groups
66_2023_2175_MOESM4_ESM.tif
sFigure 4: Kaplan–Meier curves of extracranial distant metastasis-free survival (ecDMFS) for all patients with and without ICI
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hofstetter, K., Taugner, J., Käsmann, L. et al. First-site-metastasis pattern in patients with inoperable stage III NSCLC treated with concurrent chemoradiotherapy with or without immune check-point inhibition: a retrospective analysis. Strahlenther Onkol 200, 614–623 (2024). https://doi.org/10.1007/s00066-023-02175-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-023-02175-6