Abstract
Purpose
Early and accurate assessment of distal radius fractures (DRFs) is crucial for optimal prognosis. Identifying fractures likely to lose threshold alignment (instability) in a cast is vital for treatment decisions, yet prediction tools’ accuracy and reliability remain challenging. Artificial intelligence (AI), particularly Convolutional Neural Networks (CNNs), can evaluate radiographic images with high performance. This systematic review aims to summarize studies utilizing CNNs to detect, classify, or predict loss of threshold alignment of DRFs.
Methods
A literature search was performed according to the PRISMA. Studies were eligible when the use of AI for the detection, classification, or prediction of loss of threshold alignment was analyzed. Quality assessment was done with a modified version of the methodologic index for non-randomized studies (MINORS).
Results
Of the 576 identified studies, 15 were included. On fracture detection, studies reported sensitivity and specificity ranging from 80 to 99% and 73–100%, respectively; the AUC ranged from 0.87 to 0.99; the accuracy varied from 82 to 99%. The accuracy of fracture classification ranged from 60 to 81% and the AUC from 0.59 to 0.84. No studies focused on predicting loss of thresholds alignement of DRFs.
Conclusion
AI models for DRF detection show promising performance, indicating the potential of algorithms to assist clinicians in the assessment of radiographs. In addition, AI models showed similar performance compared to clinicians. No algorithms for predicting the loss of threshold alignment were identified in our literature search despite the clinical relevance of such algorithms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The use of Artificial Intelligence (AI) to perfectly detect and classify fractures on radiographic images and to predict the best treatment option is considered a holy grail. This is also true for distal radius fractures (DRFs), where surgery aims to prevent losing threshold alignment (also known as a fracture being ‘unstable’) after closed reduction. The terminology might be confusing, as “fracture instability” and “fracture redisplacement” are often used interchangeably with “loss of threshold fracture alignment”; they are, however, insufficient and should be avoided where possible.
Detection of DRFs is most often not an issue, but non-displaced fractures or more subtle fracture lines, such as a radial styloid fracture, can be missed [1]. It has been noted that four out of five diagnostic errors made in the emergency department are missed fractures, and about 13–17% of missed fractures are located in the wrist [2, 3]. AI could be of great help here in aiding physicians.
DRF classification should (1) enable a standardized method to describe fractures and give guidance in the proper treatment per classification, (2) provide a consistent method of recording in the electronic patient system for evaluation of the patient in research, and (3) help compare studies using the same classifications and therefore optimize the treatment protocols. Considering this, a reliable fracture classification system can provide insight into clinical decision-making [4]. Therefore, a fracture classification tool without inherent surgeon bias is of interest.
When a DRF is displaced, closed reduction and cast immobilization are traditionally chosen [5]. However, secondary displacement occurs in up to 64% of the patients [6]. Identifying fractures likely to lose threshold alignment could greatly help clinical decision-making between nonoperative and surgical treatment. However, the accuracy and reliability of current fracture loss of threshold alignment prediction tools still need to be improved [7,8,9,10].
AI can execute tasks that humans previously performed. Specifically, Convolutional Neural Networks (CNN), which can evaluate visual input, have been of interest [11]. While earlier AI methods have led to applications with subhuman performance, recent CNNs can match and even surpass the capacity of humans to detect certain fractures on radiographs, focusing on isolated fracture types per model [12,13,14,15,16]. The strength of computers and algorithms is their ability to perform many calculations rapidly, consistently and without exhaustion. CNNs can be used to implement automated fracture detection, classification, and prediction algorithms to guide clinicians in clinical and emergency settings. There has been less focus on using CNNs as a prediction tool, even though this might be the most valuable attribution for treatment decisions. Given the above-mentioned challenges within the care for DRFs and the promising development of AI, we conducted a systematic review to give an overview of studies using CNNs with radiographs to detect, classify, and/or predict loss of threshold alignment of DRFs. This study aimed to answer two questions: (1) What is the accuracy of current CNNs in detecting and classifying DRFs and predicting their loss of threshold alignment on radiographs? (2) Does the use of CNNs outperform the diagnostic performance of clinicians?
Methods
Article selection, quality assessment, and data extraction
The systematic literature search was performed according to the PRISMA statement [17] and conducted in Medline ALL, Embase, Web of Science Core Collection, Cochrane Central Register of Controlled Trials and Google Scholar (100 top-ranked) in January 2024. The search strategy can be found in Appendix 1. This review was not registered online.
After removing duplicities, two authors (LHMD and KDON) independently screened the title and abstract for potential inclusion. Subsequently, a full-text review was done on the remaining articles with the defined inclusion and exclusion criteria. Articles were included if they described the use of CNNs to detect or classify DRFs or to predict loss of threshold alignment of DRFs on plain radiographs. Papers describing studies in children, reviews, letters, conference abstracts, surgical techniques, studies using robots, animal and cadaveric studies, non-orthopaedic fractures, and studies not published in English or Dutch were excluded. The inconclusive inclusion of articles was discussed afterward by the two reviewers. Covidence (Veritas Health Innovation, Melbourne, Australia) was used for the screening process and full-text review.
To assess the quality of the included articles, two reviewers (KDON, JW) independently used a modified version of the methodologic index for non-randomized studies (MINORS). A third reviewer was consulted if the scoring was inconsistent (LHMD). Studies with low scores on three or more items were excluded. Standardized forms were used to extract and record data (Microsoft Excel Version 16.21; Microsoft Inc, Redmond, WA, USA).
Outcome measures
The primary outcome was the performance of the AI model used, given in sensitivity, specificity, accuracy, Area Under the Receiver Operator Characteristics Curve (AUC), F-1 score, and average precision when present. The secondary outcome was comparing the AI models’ performance to clinicians’ performance. The highest possible F1-score is 1.0, indicating perfect precision and recall, and the lowest possible value is 0. The AUC is a score to measure the ability of a classifier to distinguish between classes. Scores lie between 0.5 (classifier equal to chance) and 1 (a perfect classifier), scores < 0.5 are not reported as they predict the wrong result. Average precision 50 (AP50) is a metric for localizing objects, meaning there is a 50% overlap between the object predicted by the algorithm versus the golden standard.
From each included article, the following data points were collected: author, year of publication, type of CNN model used, radiographic views, output classes, ground truth label assignment, number of patients or radiographs, performance metric (e.g. sensitivity, specificity, accuracy), comparison of CNN versus radiologist or reports, whether external validation was performed and potential open access availability of the model (Table 1). The reported output classes include DRF detection (fracture yes/no), localization and classification.
Quality appraisal
In this study, the MINOR Criteria included the following items: disclosure, input features, ground truth, external validation, performance metric, and AI model (Table 2). Disclosure was reported in almost all the studies except Suzuki et al. [18]. All studies clearly described the study aim. Eight studies did not describe the input features used [15, 19,20,21,22,23,24,25]. Five studies [19, 22,23,24,25] did not specify the ground truth used as a reference standard for the AI model. The external validation method was described only in six studies [13, 15, 26,27,28,29]. Two studies [23, 24] did not describe the performance metric assessed in the studies. All studies described which AI model was used. According to the outcomes of the MINORS criteria, five studies were excluded because three or more criteria were missing.
Statistical analysis
If possible, a meta-analysis will be performed. If not possible due to the variance in utilized algorithms, an overview will be given, describing the number of patients or radiographs used in training and (internal or external) validation, accuracy, sensitivity, specificity, AUC, F-1 score, average precision, and Youden index when present.
Results
Included studies
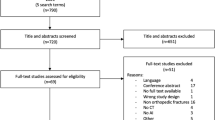
The literature search resulted in a total of 576 articles; after removal of duplicates, 365 abstracts were screened. Forty-six studies were full-text screened, and after quality assessment, eighteen studies were included in this review. (Fig. 1). No new eligible studies were identified through reference lists.
Description of studies
Of the included studies, fourteen studies described detection [15, 18, 20, 26, 28, 30,31,32,33,34,35,36,37,38], one study both detection and classification [29], two studies both detection and localization [13, 21] and one study localization and classification [39] of DRFs. No studies on the prediction of loss of threshold alignment were found. Four studies used posterior-anterior (PA) and lateral radiographs [15, 32, 36, 40], in five studies anterior-posterior (AP) and lateral [18, 28,29,30, 38], and in three studies [26, 34, 37] an extra oblique projection was used. Three studies only used lateral [33], AP [31], or PA [39] radiographs, and in three studies [20, 21, 35], the projection was not clearly described. As the ground truth, fifteen [15, 18, 20, 21, 26, 28, 29, 31,32,33, 36,37,38,39,40] studies used one or more radiologists’ or surgeons’ expertise to detect DRF. In addition, one study [34] used the radiological reports, checked and verified by a radiology registrar competent, and one study [30] used the clinical diagnosis of orthopaedic surgeons. In one study [35], the ground truth was not reported. The number of included radiographs ranged from 221 [21] to 31,490 [15] and from 32 [21] to 3500 [15] for training and testing sets, respectively. Validation sets were used in six studies [15, 20, 21, 26, 28, 30], ranging from 54 [20] to 1461 [28] radiographs. The total number of fractures on the radiographs used in the studies ranged from 221 [21] to 4452 [34] DRFs.
Detection
The sensitivity of fracture detection was reported in fourteen studies [15, 18, 22, 26, 28, 30,31,32,33,34,35, 37, 38, 40], ranging from 80% [13] to 99% [18]. Specificity was also reported, from 73% [28] to 100% [13, 18]. The AUC was reported in twelve studies [15, 18, 27, 28, 30,31,32,33, 36, 37, 40, 29] ranging from 0.87 [13] to 0.99 [30]. The accuracy was reported in nine studies [18, 29,30,31,32, 34, 35, 37, 38] ranging from 82% [22] to 99% [18]. In addition, Raisuddin et al. [36] reported a balanced accuracy of 76%. See Table 1.
Two CNN models were compared by Kim et al. [34], where the sensitivity, specificity, AUC and accuracy were similar for both models. Lindsey et al. [15] reported the performance of different test sets separately, where the AUC was 0.97, 0.98, and 0.99 for the internal, external, and clinical data test sets, respectively.
Classification
Two studies reported the performance of the classification of DRFs [29, 39]. The AUC assessed separately by Tobler et al. [29] on fragment displacement, joint involvement, and detection of multiple fractures was 0.59, 0.68, and 0.84, respectively. The accuracy was 60%, 64% and 78%, respectively [29]. Min et al. reported an AUC of 0.82, accuracy of 81%, sensitivity of 83%, specificity of 72% and a F1-score of 0.86.
AI versus clinicians
Among the included studies, eight [15, 18, 26, 29, 31, 36, 37, 40] compared the performance of AI and clinicians’ performance. According to Blüthgen et al. [40], radiologists’ performance was comparable to internal data and better on external data. Cohen et al. [26] found AI sensitivity significantly higher than initial radiology reports (IRR), with combined AI and IRR showing even greater sensitivity. Gan et al. [31] demonstrated that AI outperforms radiologists in accuracy, sensitivity, specificity, and Youden index. Comparisons with orthopaedic surgeons showed similar results. Lindsey et al. [15] revealed comparable sensitivity and AUC of aided and unaided emergency medicine clinicians by CNN. Notably, the model showed higher specificity compared to unaided clinicians. Raisuddin et al. [36] showed higher radiologist performance in normal cases and similar performance in hard cases.
Suzuki et al. [18] showed equal to better accuracy, sensitivity and specificity of CNN versus orthopaedic surgeons, though without statistically significant differences.
In Lee et al. [37], the sensitivity, specificity, accuracy, and AUC of two reviewers aided by AI increased in all fields compared to unaided. In addition, this study showed a decrease in mean interpretation time when aided by AI. Lastly, Tobler et al. [29] reported higher AUC for radiology residents than AI’s assessment of DRFs without osteosynthetic material or cast.
Discussion
This systematic review provides an overview of various computer vision algorithms for detecting and classifying DRFs on plain radiographs. Overall, the included studies showed that the performance of DRF detection is excellent, with accuracies and AUC up to 100% and 0.99, respectively. Compared with clinicians’ performance, AI had at least comparable and often better results. The development of a DRF classification model of DRF reported accuracies and AUC of 60–81% and 0.59–0.84, respectively [29, 39]. No studies describing algorithms predicting the loss of threshold alignment of DRFs were found.
This current study has several limitations. First, the comparability of the studies was limited. The studies were not consistent in the reported performance metrics. In addition, the studies used various types of DL and CNN models. However, the results of the studies show comparable performances of the different types of AI used, and the heterogenicity of the models did not affect our research questions. Secondly, the role of AI in the classification of DRF was only reported in two studies with different assessments of classifications. Therefore, evaluating AI’s overall ability to classify DRFs is difficult. Thirdly, the ground truth was differently defined between studies or even not reported at all. Lastly, only six out of 18 studies performed an external validation of the AI model. To use AI in clinical practice, a model must be trained, tested, externally validated, and preferably prospectively validated. This validation is crucial to explore transportability and bias [41]. The lack of commonplace external validation shows that most algorithms cannot be used for daily practice yet.
The strengths of this review include the broad search in different databases and the quality assessment according to the modified MINORS criteria with AI-specific factors.
The included studies reported a sensitivity and specificity between 80 and 100% in detecting DRFs. There was a significant decrease in performance between the internal and external validation set on the separate assessment of the performance on AP and lateral views. This showed the necessity of training a DL model on data comparable to the intended target data. On the other hand, to eventually build a model capable of being used on an outside institution, further improving the AI model’s performance on external validation data sets is necessary. When AP and lateral views were combined, they showed similar performance on both internal and external sets. The reported AUC and accuracy were good to excellent across the included studies. The F1-score reported in the included studies showed poor to good precision.
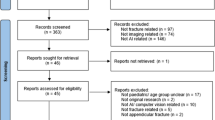
Three studies used localization in addition to detection. This helps clinicians look into the black box of the algorithm, allowing them to check for any mistakes the algorithm might make. See Fig. 2 for different options for presenting localisations. Future studies might choose to implement similar visualizations to help clinicians implement this in their daily practice.
Some of the included studies used the same CNN architecture backbone. For instance, Inception version 3 and version 4 were used in two studies [31, 33], both show comparable sensitivity, specificity, and AUC. In addition, one study [28] used a combined Inception- Resnet-Faster R-CNN and showed lower specificity and AUC. The ResNet algorithm or backbone was used in five studies [21, 27,28,29, 35], all showing comparable performances of the algorithms.
In conclusion, AI models for detecting DRFs demonstrate promising performances across various metrics. However, results may vary depending on each study’s dataset, model architecture, and evaluation methods. From a clinical perspective, DL and CNN algorithms have the potential to aid clinicians in medical imaging tasks and improve diagnostic accuracy in recognizing and consistently recording DRFs. Furthermore, we recommend focusing on diligent AI research, which involves presenting extensive outcomes, a comprehensive explanation of the dataset and the ground truth, and proper external validation.
Data availability
No datasets were generated or analysed during the current study.
References
Tyson S, Hatem SF. Easily missed fractures of the Upper Extremity. Radiol Clin North Am. 2015;53(4):717–36. viii.
Mattijssen-Horstink L, Langeraar JJ, Mauritz GJ, van der Stappen W, Baggelaar M, Tan E. Radiologic discrepancies in diagnosis of fractures in a Dutch teaching emergency department: a retrospective analysis. Scand J Trauma Resusc Emerg Med. 2020;28(1):38.
Guly HR. Diagnostic errors in an accident and emergency department. Emerg Med J. 2001;18(4):263–9.
Ilyas AM, Jupiter JB. Distal radius fractures–classification of treatment and indications for surgery. Orthop Clin North Am. 2007;38(2):167–73.
Alluri RK, Hill JR, Ghiassi A. Distal Radius fractures: approaches, indications, and techniques. J Hand Surg Am. 2016;41(8):845–54.
Makhni EC, Ewald TJ, Kelly S, Day CS. Effect of patient age on the radiographic outcomes of distal radius fractures subject to nonoperative treatment. J Hand Surg Am. 2008;33(8):1301–8.
Walenkamp MM, Aydin S, Mulders MA, Goslings JC, Schep NW. Predictors of unstable distal radius fractures: a systematic review and meta-analysis. J Hand Surg Eur Vol. 2016;41(5):501–15.
Gray RRL, Foremny G, Lee S, Greditzer T. Radiography of the distal Radius. J Orthop Trauma. 2021;35(Suppl 3):s21–6.
Jung HW, Hong H, Jung HJ, Kim JS, Park HY, Bae KH, et al. Redisplacement of distal Radius fracture after initial closed reduction: analysis of prognostic factors. Clin Orthop Surg. 2015;7(3):377–82.
LaMartina J, Jawa A, Stucken C, Merlin G, Tornetta P. 3rd. Predicting alignment after closed reduction and casting of distal radius fractures. J Hand Surg Am. 2015;40(5):934–9.
Shen D, Wu G, Suk HI. Deep learning in Medical Image Analysis. Annu Rev Biomed Eng. 2017;19:221–48.
Adams M, Chen W, Holcdorf D, McCusker MW, Howe PD, Gaillard F. Computer vs human: deep learning versus perceptual training for the detection of neck of femur fractures. J Med Imaging Radiat Oncol. 2019;63(1):27–32.
Bluthgen C, Becker AS, Vittoria de Martini I, Meier A, Martini K, Frauenfelder T. Detection and localization of distal radius fractures: deep learning system versus radiologists. Eur J Radiol. 2020;126:108925.
Chung SW, Han SS, Lee JW, Oh KS, Kim NR, Yoon JP, et al. Automated detection and classification of the proximal humerus fracture by using deep learning algorithm. Acta Orthop. 2018;89(4):468–73.
Lindsey R, Daluiski A, Chopra S, Lachapelle A, Mozer M, Sicular S, et al. Deep neural network improves fracture detection by clinicians. Proc Natl Acad Sci U S A. 2018;115(45):11591–6.
Olczak J, Fahlberg N, Maki A, Razavian AS, Jilert A, Stark A, et al. Artificial intelligence for analyzing orthopedic trauma radiographs. Acta Orthop. 2017;88(6):581–6.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010;8(5):336–41.
Suzuki T, Maki S, Yamazaki T, Wakita H, Toguchi Y, Horii M, et al. Detecting Distal Radial fractures from wrist radiographs using a deep convolutional neural network with an accuracy comparable to Hand Orthopedic surgeons. J Digit Imaging. 2022;35(1):39–46.
Ebsim R, Naqvi J, Cootes TF, editors. Automatic detection of wrist fractures from Posteroanterior and lateral radiographs: a deep learning-based Approach. Computational methods and clinical applications in Musculoskeletal Imaging; 2019 2019//; Cham: Springer International Publishing.
Hardalaç F, Uysal F, Peker O, Çiçeklidağ M, Tolunay T, Tokgöz N et al. Fracture detection in wrist X-ray images using deep learning-based object detection models. Sens (Basel). 2022;22(3).
Joshi D, Singh TP, Joshi AK. Deep learning-based localization and segmentation of wrist fractures on X-ray radiographs. Neural Comput Appl. 2022;34(21):19061–77.
Rashid T, Zia MS, Najam Ur R, Meraj T, Rauf HT, Kadry S. A minority class Balanced Approach using the DCNN-LSTM Method to Detect Human wrist fracture. Life (Basel). 2023;13(1).
Yahalomi E, Chernofsky M, Werman M, editors. Detection of distal Radius fractures trained by a small set of X-Ray images and faster R-CNN. Intelligent Computing; 2019 2019//; Cham: Springer International Publishing.
Yang F, Cong R, Xing M, Ding B. Study on AO classification of distal radius fractures based on multi-feature fusion. Journal of Physics: Conference Series. 2021;1800(1):012006.
Javed R, Khan TA, Janjua JI, Muhammad MA, Ramay SA, Basit MK. WRIST FRACTURE PREDICTION USING TRANSFER LEARNING, A CASE STUDY. J Popul Ther Clin Pharmacol. 2023;30(18):1050–62.
Cohen M, Puntonet J, Sanchez J, Kierszbaum E, Crema M, Soyer P et al. Artificial intelligence vs. radiologist: accuracy of wrist fracture detection on radiographs. Eur Radiol. 2022.
Kim JE, Yoo HJ, Chae HD, Choi JY, Hong SH, Kang JH, et al. Dual-layer detector CT with virtual noncalcium imaging: diagnostic performance in patients with suspected wrist fractures. AJR Am J Roentgenol. 2021;216(4):1003–13.
Thian YL, Li Y, Jagmohan P, Sia D, Chan VEY, Tan RT. Convolutional Neural Networks for Automated Fracture Detection and localization on wrist radiographs. Radiol Artif Intell. 2019;1(1):e180001.
Tobler P, Cyriac J, Kovacs BK, Hofmann V, Sexauer R, Paciolla F, et al. AI-based detection and classification of distal radius fractures using low-effort data labeling: evaluation of applicability and effect of training set size. Eur Radiol. 2021;31(9):6816–24.
Oka K, Shiode R, Yoshii Y, Tanaka H, Iwahashi T, Murase T. Artificial intelligence to diagnosis distal radius fracture using biplane plain X-rays. J Orthop Surg Res. 2021;16(1):694.
Gan K, Xu D, Lin Y, Shen Y, Zhang T, Hu K, et al. Artificial intelligence detection of distal radius fractures: a comparison between the convolutional neural network and professional assessments. Acta Orthop. 2019;90(4):394–400.
Anttila TT, Karjalainen TV, Mäkelä TO, Waris EM, Lindfors NC, Leminen MM et al. Detecting Distal Radius fractures using a segmentation-based Deep Learning Model. J Digit Imaging. 2022.
Kim DH, MacKinnon T. Artificial intelligence in fracture detection: transfer learning from deep convolutional neural networks. Clin Radiol. 2018;73(5):439–45.
Kim MW, Jung J, Park SJ, Park YS, Yi JH, Yang WS, et al. Application of convolutional neural networks for distal radio-ulnar fracture detection on plain radiographs in the emergency room. Clin Exp Emerg Med. 2021;8(2):120–7.
Üreten K, Sevinç HF, İğdeli U, Onay A, Maraş Y. Use of deep learning methods for hand fracture detection from plain hand radiographs. Ulus Travma Acil Cerrahi Derg. 2022;28(2):196–201.
Raisuddin AM, Vaattovaara E, Nevalainen M, Nikki M, Järvenpää E, Makkonen K, et al. Critical evaluation of deep neural networks for wrist fracture detection. Sci Rep. 2021;11(1):6006.
Lee KC, Choi IC, Kang CH, Ahn KS, Yoon H, Lee JJ et al. Clinical validation of an Artificial Intelligence Model for detecting distal Radius, Ulnar Styloid, and scaphoid fractures on conventional wrist radiographs. Diagnostics. 2023;13(9).
Zhang J, Li Z, Lin H, Xue M, Wang H, Fang Y, et al. Deep learning assisted diagnosis system: improving the diagnostic accuracy of distal radius fractures. Front Med. 2023;10:1224489.
Min H, Rabi Y, Wadhawan A, Bourgeat P, Dowling J, White J, et al. Automatic classification of distal radius fracture using a two-stage ensemble deep learning framework. Phys Eng Sci Med. 2023;46(2):877–86.
Blüthgen C, Becker AS, Vittoria de Martini I, Meier A, Martini K, Frauenfelder T. Detection and localization of distal radius fractures: deep learning system versus radiologists. Eur J Radiol. 2020;126:108925.
Oliveira ECL, van den Merkhof A, Olczak J, Gordon M, Jutte PC, Jaarsma RL, et al. An increasing number of convolutional neural networks for fracture recognition and classification in orthopaedics: are these externally validated and ready for clinical application? Bone Jt Open. 2021;2(10):879–85.
Acknowledgements
We acknowledge Maarten Engel of the Medical Library of Erasmus MC for his help in developing the final search strategy.
Funding
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article and did not receive funding for the writing of this article.
Author information
Authors and Affiliations
Consortia
Contributions
For this article the following contributions were made: All authors were part of the design and conception of this work in varying degrees. KDON, LHMD, JPW performed the data collection, with supervision of the other authors in varying degrees.KDON, LHMD, JPW created the tables and figures, supervised by other authors in varying degrees.KDON and LHMD created the first draft of the manuscript text, and this text was revised multiple times by the other authors. All authors have contributed substantially to the article enough to qualify for authorship, as according to ICMJE guidelines.
Corresponding authors
Ethics declarations
Ethical approval
This study was performed at the Trauma Research Unit Department of Surgery, Erasmus University Medical Center Rotterdam, the Netherlands and at the Orthopedic Department, the University of Groningen, University Medical Center Groningen, the Netherlands.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oude Nijhuis, K., Dankelman, L.H.M., Wiersma, J.P. et al. AI for detection, classification and prediction of loss of alignment of distal radius fractures; a systematic review. Eur J Trauma Emerg Surg (2024). https://doi.org/10.1007/s00068-024-02557-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00068-024-02557-0