Abstract
The macrolides-resistant Bordetella pertussis (MR-Bp) isolates in China evolved from the ptxP1/fhaB3 allele and rapidly became predominant, suggestive of an adaptive transmission ability. This was different from the global prevalent ptxP3 strains, in which MR-Bp was rarely reported. The study aimed to determine the underlying mechanism responsible for fitness and resistance in these two strains. We identify proteomic differences between ptxP1/fhaB3 and ptxP3/fhaB1 strains using tandem mass tag (TMT)-based proteomics. We then performed in-depth bioinformatic analysis to determine differentially expressed genes (DEGs), followed by gene ontology (GO), and protein–protein interaction (PPI) network analysis. Further parallel reaction monitoring (PRM) analysis confirmed the expression of four target proteins. Finally, the crystal violet method was used to determine biofilm-forming ability. The results showed that the main significantly different proteins between the two represent isolates were related to biofilm formation. Furthermore, we have confirmed that ptxP1/fhaB3 showed hyperbiofilm formation in comparison with ptxP3/fhaB1. It is suggested that the resistance and adaptability of ptxP1/fhaB3 strains may be related to the formation of biofilm through proteomics. In a word, we determined the significantly different proteins between the ptxP1/fhaB3 and ptxP3/fhaB1 strains through whole-cell proteome, which were related to biofilm formation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Bordetella pertussis (Bp) is the pathogen of pertussis, a severe respiratory infectious disease. Although pertussis has been well controlled globally through pertussis vaccination, the incidence of pertussis has increased significantly in recent years, showing the characteristics of a “resurgence” (Clark 2014). Around the 1980s, isolates carrying the ptxP1 allele became the predominant ptxP type in many countries (Mosiej et al. 2015). Since early 2000, PtxP3/fhaB1 Bp lineage has almost replaced the resident ptxP1-Bp lineage resulting in an almost worldwide selective sweep (Consortium 2019; Xu et al. 2019a, b). By contrast, the Chinese ptxP3 allele first emerged in 2000, but unlike trends elsewhere, ptxP1 remained predominant among the isolates (Wang and He 2015; Xu et al. 2015). Moreover, the macrolides-resistant (MR) ptxP3/fhaB1 Bp strains were occasionally reported without further dissemination (Guillot et al. 2012), which may be related to the fitness cost (greatly reduced pathogenicity) of Bp after MR.
However, there has become an explosive increase after the emergence of macrolides-resistant B. pertussis (MR-Bp) in China in 2011 (Wang et al. 2013, 2014; Liu et al. 2018). From our previous genomic results, we found that all MR-Bp strains in China have evolved from ptxP1/fhaB3 lineage (Xu et al. 2019). Recently, the MR-Bp strain in Japan and Vietnam was reported and related to the ptxP1/fhaB3 lineage, which raises the possibility that MR-Bp could spread all over the world (Kamachi et al. 2020; Yamaguchi et al. 2020; Feng et al. 2021). Thus, ptxP1/fhaB3 MR-Bp became the dominant strain in China, which is contrary to the phenomenon of the fitness cost of loss of virulence after macrolides resistance of Bp (Weiss and Falkow 1984). However, the mechanism of this phenomenon was under dispute.
Microbial biofilm is an organized aggregate of microorganisms, which is resistant to an extreme environment, such as increasing microbial resistance against various antibiotics (Yin et al. 2019). Bp is more resistant to macrolide antibiotics in the biofilm and biofilm spatial structure confers a consistent and robust fitness advantage (Dorji et al. 2016; Deschaine et al. 2018), suggesting that the adaptive transmission ability of ptxP1/fhaB3 MR-Bp may be related to the ability of biofilm formation.
Therefore, to reveal the possible mechanism of the fitness of prevalent MR-Bp, mass spectrometry-based proteomics of ptxP1/fhaB3 MR-Bp and the globally prevalent macrolides-sensitive (MS) ptxP3/fhaB1 Bp were performed in this study to screen the characteristic proteins of prevalent MR-Bp and detect the biofilm-forming ability of the two representative isolates.
Methods
Bacterial culture
Bordetella pertussis strains 19,147 (ptxP1/fhaB3) and 19,068 (ptxP3/fhaB1) were collected from the patients suffering from pertussis from China in 2019. These two strains from glycerol stocks were grown on Bordet–Gendou agar plates in parallel and incubated at 37 °C for 3 days. Pure cultures were obtained by sub-culturing a single clone onto second Bordet–Gendou agar plates and incubated again at 37 °C for 2–3 days. A loopful of pure colonies was inoculated into 40 ml of Stainer–Scholte (SS) with 1% Heptakis ((2,6-o-dimethyl) β-cyclodextrin) and incubated for 24 h with shaking at 37 °C with the starting OD600 adjusted to 0.1.
Sample preparation
After incubation, the whole-cell and supernatant were separated by centrifugation at 3000 × g at 4 °C for 15 min. The whole-cell pellet was washed three times with phosphate-buffered saline (PBS) and then frozen quickly with liquid nitrogen. The whole-cell pellet is stored at − 80 °C for subsequent tandem mass tag (TMT)-based proteomics analysis.
TMT-based proteomics analysis
The pellet was lysed with SDT (4% (w/v) SDS, 100 mM Tris/HCl pH7.6, 0.1 M DTT) and the number of proteins was quantified with the BCA Protein Assay Kit (Bio-Rad, USA). Subsequently, the proteins were digested with trypsin to obtain peptides (Chou and Schwartz 2011), which were further desalted by C18 Cartridge. After lyophilization, the peptides were reconstituted with 40 μL 0.1% formic acid solution and were quantified (OD280). TMT reagent (Thermo Fisher Scientific) was used to label the resulting 100 μg peptides, according to the manufacturer’s instructions. Labeled peptides were fractionated by the High pH Reversed-Phase Peptide Fractionation Kit (Thermo Scientific). Then, the samples were separated using an EASY-nLC liquid chromatography instrument (Thermo Fisher Scientific) and LC–MS/MS analysis was performed on a Q-Exactive mass spectrometer (Thermo Scientific). The protein identification and quantification were accomplished using Mascot2.2 and Proteome Discoverer1.4. We aligned the acquired protein sequence against the Bordetella pertussis sequence from Protein Sequence Database (Uniprot database) and the protein ratios were obtained by taking the median ratio of peptides that are unique for a protein group. Three biological replicates were performed for each strain.
Bioinformatics analysis
CELLO (http://cello.life.nctu.edu.tw/), which is a multi-class SVM classification system, was used to predict protein subcellular localization. The protein sequences of the selected differentially expressed proteins were locally searched using the NCBI BLAST + client software (NCBI-blast-2.2.28 + -win32.exe) and InterProScan to find homolog sequences, and then, gene ontology (GO) terms were mapped and sequences were annotated using the software program Blast2GO. The Fisher’s exact test is used to compare the distribution of each GO classification in the protein set and perform the enrichment analysis of GO annotations on the target protein set. Up-regulated and down-regulated proteins were defined as having fold changes (FC) > 1.2 and < 0.82, respectively. A two-tail Student’s t test was then performed with p < 0.05 assigned as significant. Three biological replicates were used for each strain. To explore the role of the functional link between different proteins of the two strains, we performed protein–protein interaction (PPI) network on the significantly different proteins. The PPI information of the studied proteins was based on the STRING database (http://string-db.org/) and Cytoscape software (version 3.8.2). The GO annotation results and volcano results were plotted by R scripts.
Parallel reaction monitoring (PRM)-based protein quantification
PRM is a liquid chromatography–mass spectrometry-based targeted peptide/protein quantification method. Proteins that were identified to be significantly different in TMT experiments were confirmed using PRM measurements. The proteins of whole-cell were trypsin digested, a liquid chromatography separation systems’ (HPLC) system was used for chromatographic separation, and Q-Exactive HF mass spectrometer (Thermo Scientific) was used for mass spectrometry analysis. Here, we selected 11 node proteins in the PPI network (Cpn10, RpoZ, RpsC, HscA, RpoD, RplB, RpsD, prn, fim2, fim3, and fimD) for PRM verification, of which four target proteins can monitor credible peptides, namely Prn, Fim2, Fim3, and RpoD. The list of target peptides and sub-ions for PRM quantification is shown in Supplementary Table 1.
Biofilm formation assay
To determine the biofilm formation of 19,147 (ptxP1/fhaB3) and 19,068 (ptxP3/fhaB1), the strains were grown for 4 days at 37 ℃ and stained with crystal violet every 24 h. The specific steps were as follows. Bp from overnight SS liquid cultures was diluted to OD600 = 0.1. 100 ul Bp culture were added to 96-well plates and incubated for 96 h at 37℃ at rest. The culture medium was discarded every 24 h and the cells were washed three times with PBS, supplemented with fresh culture medium. After incubation, 96-well plates were washed three times with PBS and left to dry. Crystal violet (1% w/v, 150 μL) was added to each well and the plate was incubated for 10 min at room temperature. The supernatant was then discarded and 200 µL of ethanol containing 10% acetic acid was added. After 10 min of incubation at room temperature, biofilm formation was then visualized by measuring the OD600. Five biological replicates were used at each time point. All statistical analyses were completed using GraphPad Prism 8.0.
Results
Overview of the significantly different proteins in whole-cell
TMT was performed to characterize and compare the whole-cell proteome of 19,147 (ptxP1/fhaB3) and 19,068 (ptxP3/fhaB1). There are a total of 68 significantly different proteins between the two strains (Supplementary Table 2). Compared with 19,068 (ptxP3/fhaB1), 40 proteins in 19,147 (ptxP1/fhaB3) are up-regulated and 28 proteins are down-regulated. Differentially expressed proteins were analyzed by volcano plot (Fig. 1a). The heat map shows the clustering of significantly differentially expressed proteins between the two strains (Fig. 1b).
Macrolide resistance is mainly caused by target modifications such as the methylation in a 23S ribosomal RNA (rRNA) adenine residue and the mutation in ribosomal protein L4 or L22 (Fyfe et al. 2016). To explore the key proteins that may be involved, we focused on macrolides-resistant related molecules. Although the Erm, an enzyme that catalyzes A2058 methylation to preclude antibiotic binding, was not detected in the two pertussis strain, the expression of RumA (A0A0N2IMR5) protein was found to be significantly different in the two strains (Fig. 1b). In addition, we found that the expression of ribosomal protein L2, also known as RplB (A0A171JW48), was significantly different in expression between the two strains.
Function analysis of significantly different proteins in whole-cell
The outer membrane acts as the first line of defense against the penetration of multiple toxic compounds, including several antimicrobial agents (Munita and Arias 2016). The differential characteristics of outer membrane proteins have been identified in antibiotic-resistant bacteria, which form a specific pattern of antibiotics (Peng et al. 2019). In addition, the loss of some outer membrane proteins reduced the virulence and fitness of bacteria (Smani et al. 2013). Therefore, we further analyzed the subcellular localization of significantly different proteins in the whole-cell (Fig. 2a). There are five kinds of proteins located in the outer membrane, namely FimD (A0A381A3A6), BN118_1423 (A0A0T7CMT4), HscA (Q7VXG7), AZ26_1060 (A0A171JW15), Prn1 (S5TUY6), and Prn1 (D2WF61).
Functional analysis of 19,147 (ptxP1/fhaB3) and 19,068 (ptxP3/fhaB1) proteins detected in the whole-cell a. The subcellular structure of significantly different proteins between the two groups. b. The top 20 enriched Gene Ontology (GO) terms of significantly different proteins. BP biological process, MF molecular function; CC cellular component
To further analyze the protein characteristics of MR-Bp, we performed a GO cluster analysis on the significantly different proteins in the whole-cell proteome. The top 20 enriched GO terms are presented in Fig. 2b. The most noteworthy is the iron–sulfur cluster and metallo-sulfur cluster assembly. The significant differences in metallo-sulfur cluster between the two strains are HscA (Q7VXG7), CyaY (Q7VT96), and IscA (A0A0U0VXP3).
Protein interaction analysis of the significantly different proteins in whole-cell
The functional connection of the significantly different proteins between 19,147 (ptxP1/fhaB3) and 19,068 (ptxP3/fhaB1) may be a breakthrough to answer the reason for the enhanced resistance and adaptability in MR-Bp. The PPI network was constructed using the String website and the figures were generated by Cytoscape (MCODE plug-in). The most significant module (MCODE score = 5.333) contained 7 nodes and 16 edges (Fig. 3a). In the whole-cell proteome in pertussis, Cpn10 (P0A339) and RpoZ (Q7VXZ4) were significantly higher in 19,147 (ptxP1/fhaB3) resistant strain compared to 19,068 (ptxP3/fhaB1) sensitive strains, and the other five proteins RpsC (Q7VTC7), HscA (Q7VXG7), RpoD (A0A0T7CNP5), RplB (A0A171JW48) and RpsD (P0A4C5) were significantly lower than 19,068 (ptxP3/fhaB1) indicating the role of key proteins in the macrolides resistance of Bp (Fig. 1b).
The next significant module (MCODE score = 4) contained four genes, which are prn, fim2, fim3, and fimD (Fig. 3b). Prn, fim2, and fim3 have internationally recognized pertussis virulence genes, and fim2/fim3 represents distinct serotypes of B. pertussis. In this experiment, compared with 19,068 (ptxP3/fhaB1), the expression of Fim3 and FimD increased in 19,147 (ptxP1/fhaB3), and the expression of Fim2 and Prn decreased.
PRM verifies significantly different protein expression levels in whole-cell
To confirm the reliability of the quantitative proteomics analyses, the 11 candidate proteins selected in PPI were evaluated by PRM analyses. The mass spectrometry identification results show that the four target proteins can be accurately identified namely Prn, Fim2, Fim3, and RpoD.
The fold changes of four proteins by PRM are listed in Table 1. Fim2, Prn, and RpoD were down-regulated in 19,147 (ptxP1/fhaB3), while Fim3 was up-regulated. The results of the relative quantification demonstrated that the target proteins displayed similar trends between the TMT and PRM analyses, thus supporting the reliability of the proteomics data.
The different proteins in whole-cell are related to the biofilm formation
In this study, many differential proteins related to macrolide resistance are closely related to the formation of biofilm, such as metallo-sulfur cluster. Moreover, it has been reported that biofilm-grown Bp confers increased tolerance to antimicrobial agents compared with planktonic cultures and biofilm formation provides a fitness advantage to bacteria (Dorji et al. 2016; Deschaine et al. 2018).
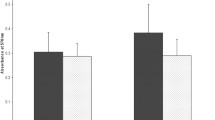
Crystal violet staining was used to evaluate whether biofilm formation changed in 19,147 (ptxP1/fhaB3) and 19,068 (ptxP3/fhaB1), of which ATCC strain 9797 was used as the standard strain. Interestingly, crystal violet staining revealed that 19,147 (ptxP1/fhaB3) had a significant increase in biofilm formation compared to 19,068 (ptxP3/fhaB1) (Fig. 4).
Discussion
In this study, we determined the significantly different proteins between the dominant lineage of Bp strains between China and other countries nearly all over the world through TMT-based proteomics and PRM-based protein quantification. The specific key proteins of MR-Bp strain include ribosome-associated proteins (RumA, RplB), outer membrane proteins (FimD, BN118_1423, HscA, AZ26_1060, Prn), Fe/S clusters proteins (HscA, CyaY, IscA), RNA polymerase proteins (RpoD, RpoZ), and virulence proteins (Prn, Fim2, Fim3, FimD). Interestingly, the deletion and mutation of multiple resistance genes encoding these proteins are closely related to the formation of biofilms in other bacterial species. Moreover, the biofilm formation of 19,147 (ptxP1/fhaB3) was increased compared with 19,068 (ptxP3/fhaB1) (Fig. 4). Based on our limited current knowledge, this is the first report screening the potential mechanisms of macrolides resistance and the fitness of prevalent MR-Bp through proteomics.
More and more evidence shows that defects in Fe/S proteins assembly and maturation are closely related to biofilm formation and antibiotic resistance (Mashruwala et al. 2016; Deshpande et al. 2020; Ellepola et al. 2021). Among the three significantly different proteins in the Fe/S cluster, HscA and IscA are involved in the iron–sulfur cluster assembly (Zeng et al. 2007; Mayer 2021), and either hscA mutation or loss of iscA will change the formation of bacterial biofilm (Vasil'eva and Strel'tsova 2013; Rondeau et al. 2019). In the PPI network, RNA polymerase related to the Fe/S cluster contributes to concern, and the RpoZ and RpoD in Bp strains may dynamically change macrolides resistance. RpoZ and RpoD, called ω and σ70 factors respectively, are the subunits of the RNA polymerase core enzyme. We found that the RpoZ expression of the 19,147 (ptxP1/fhaB3) resistant isolates was increased and the RpoD was decreased correspondingly. Although ω-encoding rpoZ evolves in the ecosystem during colder and drier periods, resulting in an increased rate of biofilm production of rpoZ variants in vitro (Cui et al. 2020), whether it is in Gram-positive or Gram-negative bacteria, defects of rpoZ have been confirmed to impair the ability to completely form biofilms and affect antibiotic resistance (Mukherjee and Chatterji 2008; Weiss et al. 2017; Bhardwaj et al. 2018). Another important function of the ω factor is involved in σ factor recruitment. RpoD mutation is associated with antibiotic resistance (Palace et al. 2020), and we have verified the low expression of RpoD in 19,147 (ptxP1/fhaB3) by PRM-based protein quantification.
In the PPI network, except for RNA polymerase (RpoZ and RpoD), both RpsC and RpsD are part of the 30S ribosomal subunit, and their changes will affect antibiotic resistance (Björkman et al. 1999; Gupta et al. 2016). Horizontal gene transfer (HGT) of rplB, also known as ribosomal protein L2, has been shown to play a key role in the genesis of antimicrobial resistance (Gentry and Holmes 2008; Manoharan-Basil et al. 2021). Cpn10, also known as GroES, and over-expression of Cpn10 promote streptomycin resistance (Goltermann et al. 2015).
As we all know, biofilms are communities of microorganisms attached to a surface that is significantly less susceptible to antimicrobial agents than non-adherent planktonic cells (Hall and Mah 2017). It has recently been discovered that biofilm formation was associated with Bp resistance. Compared with planktonic cultures, biofilm growing Bp conferred increased tolerance to antimicrobial agents (Dorji et al. 2016), which may be a possible reason for the emergence of macrolides resistance of the ptxP1/fhaB3 strain in China.
After antibiotic resistance, the adaptability of some bacteria will increase rather than decrease, which is usually related to a compensatory mutation in evolution (Durão et al. 2018). The mutation rate of ptxP1/fhaB3 strain in China is five times than the rest of the global ptxP1 isolates (Xu et al. 2019). Therefore, the adaptability of the Chinese MR-Bp strains may also be produced by compensation mutation, but it needs to be further verified.
Another aspect that deserves attention: the fitness advantage of bacteria is related to biofilm formation. When replication and virulence of drug-resistant bacteria were significantly attenuated, their biofilm formation decreased (Biot et al. 2020). Other studies have reported that hyperbiofilm formation is associated with enhanced virulence traits in Bp (Cattelan et al. 2017). The higher biofilm-forming strains will exhibit increased cellular adherence to epithelial cells and contribute to enhanced respiratory tract colonization. FimD (fimbrial subunits), highly expressed in 19,147 (ptxP1/fhaB3) strain, is critical in adherence to airway cells and can also serve as specific residues of the chaperone-usher pathway to mediate biofilm formation (Volkan et al. 2013; Guevara et al. 2016). Therefore, the link between the hyperbiofilm-forming ability of Bp and enhanced pathogenic phenotypes indirectly suggested that the significant difference in proteins related to biofilm formation may be a crucial factor for the adaptive transmission ability of ptxP1/fhaB3/MR-Bp.
In addition, the emergence and rapid growth of MR-Bp in China may also be related to the following reasons. First, it may be a result of selection pressure from vaccination. Acellular vaccine (ACV) was produced in the 1980s and subsequently replaced the whole-cell vaccine (WCV) in developed countries causing the transition from the ptxP1 allele to the ptxP3 allele (Mir-Cros et al. 2019; Safarchi et al. 2016). The replacement of WCV in China occurred a decade later than in developed countries which may be the reason why ptxP3 strains have not replaced ptxP1 strains in China (Du et al. 2016). Second, it may also be due to the selection pressure from antibiotics. Before China implemented the strict antibiotic stewardship policies in 2012, the problem of antibiotic abuse in China was very serious (Yin et al. 2013). The overuse of antibiotics has accelerated the rapid emergence of antibiotic resistance. Similarly, Vietnam, which reported ptxP1/fhaB3 MR-BP, also has serious issues with the irrational use of antibiotics (Mao et al. 2015).
However, the limitation of this study is that the proteomics from the culture supernatant was not performed. Besides the proteins from the cells, the outer membrane vesicles (OMVs) from the culture supernatant were also closely attributed to the biofilm formations. Therefore, it is impossible to rule out that some proteins related to the biofilm formations have not been discovered.
Conclusions
In summary, in the proteomics of 19,147 (ptxP1/fhaB3) and 19,068 (ptxP3/fhaB1) strains, the significantly different proteins are probably related to biofilm formation. The bacterium growing in biofilm is more resistant to macrolides and contributed to the persistence and transmission, which may be the reason for macrolides resistance and the fitness of ptxP1/fhaB3 MR-Bp strains in China. Further studies focused on biofilms need to be explored much more.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Bhardwaj N, Syal K, Chatterji D (2018) The role of ω-subunit of Escherichia coli RNA polymerase in stress response. Genes Cells 23:357–369. https://doi.org/10.1111/gtc.12577
Biot FV, Bachert BA, Mlynek KD et al (2020) Evolution of antibiotic resistance in surrogates of francisella tularensis (LVS and Francisella novicida): effects on biofilm formation and fitness. Front Microbiol 11:593542. https://doi.org/10.3389/fmicb.2020.593542
Björkman J, Samuelsson P, Andersson DI et al (1999) Novel ribosomal mutations affecting translational accuracy, antibiotic resistance and virulence of salmonella typhimurium. Mol Microbiol 31:53–58. https://doi.org/10.1046/j.1365-2958.1999.01142.x
Cattelan N, Jennings-Gee J, Dubey P et al (2017) Hyperbiofilm Formation by bordetella pertussis strains correlates with enhanced virulence traits. Infect Immun. https://doi.org/10.1128/iai.00373-17
Chou MF, Schwartz D (2011) Biological sequence motif discovery using motif-x. Curr Protoc Bioinformat 13:15–24. https://doi.org/10.1002/0471250953.bi1315s35
Clark TA (2014) Changing pertussis epidemiology: everything old is new again. J Infect Dis 209:978–981. https://doi.org/10.1093/infdis/jiu001
Consortium P (2019) PERISCOPE: road toward effective control of pertussis. Lancet Infect Dis 19:e179–e186. https://doi.org/10.1016/S1473-3099(18)30646-7
Cui Y, Schmid BV, Cao H et al (2020) Evolutionary selection of biofilm-mediated extended phenotypes in Yersinia pestis in response to a fluctuating environment. Nat Commun 11:281. https://doi.org/10.1038/s41467-019-14099-w
Deschaine BM, Heysel AR, Lenhart BA et al (2018) Biofilm formation and toxin production provide a fitness advantage in mixed colonies of environmental yeast isolates. Ecol Evol 8:5541–5550. https://doi.org/10.1002/ece3.4082
Deshpande A, Wu X, Huo W et al (2020) Chromosomal resistance to metronidazole in clostridioides difficile can be mediated by epistasis between iron homeostasis and oxidoreductases. Antimicrob Agent Chemot. https://doi.org/10.1128/aac.00415-20
Dorji D, Graham RM, Richmond P et al (2016) Biofilm forming potential and antimicrobial susceptibility of newly emerged Western Australian Bordetella pertussis clinical isolates. Biofouling 32:1141–1152. https://doi.org/10.1080/08927014.2016.1232715
Du Q, Wang X, Liu Y et al (2016) Direct molecular typing of Bordetella pertussis from nasopharyngeal specimens in China in 2012–2013. Eur J Clin Microbiol Infect Dis 35:1211–1214. https://doi.org/10.1007/s10096-016-2655-3
Durão P, Balbontín R, Gordo I (2018) Evolutionary mechanisms shaping the maintenance of antibiotic resistance. Trends Microbiol 26:677–691. https://doi.org/10.1016/j.tim.2018.01.005
Ellepola K, Huang X, Riley RP et al (2021) Streptococcus mutans lacking sufCDSUB Is Viable, but displays major defects in growth, stress tolerance responses and biofilm formation. Front Microbiol 12:671533. https://doi.org/10.3389/fmicb.2021.671533
Feng Y, Chiu CH, Heininger U et al (2021) Emerging macrolide resistance in Bordetella pertussis in mainland China: Findings and warning from the global pertussis initiative. Lancet Reg Health West Pac 8:100098. https://doi.org/10.1016/j.lanwpc.2021.100098
Fyfe C, Grossman TH, Kerstein K et al (2016) Resistance to macrolide antibiotics in public health pathogens. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a025395
Gentry DR, Holmes DJ (2008) Selection for high-level telithromycin resistance in staphylococcus aureus yields mutants resulting from an rplB-to-rplV gene conversion-like event. Antimicrob Agents Chemother 52:1156–1158. https://doi.org/10.1128/AAC.00923-07
Goltermann L, Sarusie MV, Bentin T (2015) Chaperonin GroEL/GroES over-expression promotes aminoglycoside resistance and reduces drug susceptibilities in escherichia coli following exposure to sublethal aminoglycoside doses. Front Microbiol 6:1572. https://doi.org/10.3389/fmicb.2015.01572
Guevara C, Zhang C, Gaddy JA et al (2016) Highly differentiated human airway epithelial cells: a model to study host cell-parasite interactions in pertussis. Infect Dis (lond). 48:177–188. https://doi.org/10.3109/23744235.2015.1100323
Guillot S, Descours G, Gillet Y et al (2012) Macrolide-resistant Bordetella pertussis infection in newborn girl, France. Emerg Infect Dis 18:966–968. https://doi.org/10.3201/eid1806.120091
Gupta V, Haider S, Sood U et al (2016) Comparative genomic analysis of novel acinetobacter symbionts: a combined systems biology and genomics approach. Sci Rep 6:29043. https://doi.org/10.1038/srep29043
Hall CW, Mah TF (2017) Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. FEMS Microbiol Rev 41:276–301. https://doi.org/10.1093/femsre/fux010
Kamachi K, Duong HT, Dang AD et al (2020) Macrolide-Resistant Bordetella pertussis, Vietnam, 2016–2017. Emerg Infect Dis 26:2511–2513. https://doi.org/10.3201/eid2610.201035
Liu X, Wang Z, Zhang J et al (2018) Pertussis Outbreak in a Primary School in China: Infection and Transmission of the Macrolide-resistant Bordetella pertussis. Pediatr Infect Dis J 37:e145–e148. https://doi.org/10.1097/INF.0000000000001814
Manoharan-Basil SS, Laumen JGE, Van Dijck C et al (2021) Evidence of Horizontal Gene Transfer of 50S Ribosomal Genes rplB, rplD, and rplY in Neisseria gonorrhoeae. Front Microbiol 12:683901. https://doi.org/10.3389/fmicb.2021.683901
Mao W, Vu H, Xie Z et al (2015) Systematic review on irrational use of medicines in China and Vietnam. PLoS ONE 10:e0117710. https://doi.org/10.1371/journal.pone.0117710
Mashruwala AA, Bhatt S, Poudel S et al (2016) The DUF59 Containing Protein SufT Is Involved in the Maturation of Iron-Sulfur (FeS) Proteins during Conditions of High FeS Cofactor Demand in Staphylococcus aureus. PLoS Genet 12:e1006233. https://doi.org/10.1371/journal.pgen.1006233
Mayer MP (2021) The Hsp70-Chaperone Machines in Bacteria. Front Mol Biosci 8:694012. https://doi.org/10.3389/fmolb.2021.694012
Mir-Cros A, Moreno-Mingorance A, Martín-Gómez MT et al (2019) Population dynamics and antigenic drift of Bordetella pertussis following whole cell vaccine replacement, Barcelona, Spain, 1986–2015. Emerg Microbes Infect. 8:1711–1720. https://doi.org/10.1080/22221751.2019.1694395
Mosiej E, Zawadka M, Krysztopa-Grzybowska K et al (2015) Sequence variation in virulence-related genes of Bordetella pertussis isolates from Poland in the period 1959–2013. Eur J Clin Microbiol Infect Dis 34:147–152. https://doi.org/10.1007/s10096-014-2216-6
Mukherjee R, Chatterji D (2008) Proteomics and mass spectrometric studies reveal planktonic growth of Mycobacterium smegmatis in biofilm cultures in the absence of rpoZ. J Chromatogr B Analyt Technol Biomed Life Sci 861:196–202. https://doi.org/10.1016/j.jchromb.2007.08.009
Munita JM, Arias CA (2016) Mechanisms of Antibiotic Resistance. Microbiol Spectr. https://doi.org/10.1128/microbiolspec.VMBF-0016-2015
Palace SG, Wang Y, Rubin DH et al (2020) RNA polymerase mutations cause cephalosporin resistance in clinical Neisseria gonorrhoeae isolates. Elife. https://doi.org/10.7554/eLife.51407
Peng B, Li H, Peng X (2019) Proteomics approach to understand bacterial antibiotic resistance strategies. Expert Rev Proteomics 16:829–839. https://doi.org/10.1080/14789450.2019.1681978
Rondeau M, Esmaeel Q, Crouzet J et al (2019) Biofilm-Constructing Variants of Paraburkholderia phytofirmans PsJN Outcompete the Wild-Type Form in Free-Living and Static Conditions but Not In Planta. Appl Environ Microbiol. https://doi.org/10.1128/AEM.02670-18
Safarchi A, Octavia S, Luu LD et al (2016) Better colonisation of newly emerged Bordetella pertussis in the co-infection mouse model study. Vaccine 34:3967–3971. https://doi.org/10.1016/j.vaccine.2016.06.052
Smani Y, Dominguez-Herrera J, Pachón J (2013) Association of the outer membrane protein Omp33 with fitness and virulence of Acinetobacter baumannii. J Infect Dis 208:1561–1570. https://doi.org/10.1093/infdis/jit386
Vasil’eva SV, Strel’tsova DA (2013) Reconstruction of Fe-S protein clusters in Escherichia coli and biofilm formation. Dokl Biochem Biophys 448:27–32. https://doi.org/10.1134/s1607672913010109
Volkan E, Kalas V, Pinkner JS et al (2013) Molecular basis of usher pore gating in Escherichia coli pilus biogenesis. Proc Natl Acad Sci U S A 110:20741–20746. https://doi.org/10.1073/pnas.1320528110
Wang Z, He Q (2015) Bordetella pertussis isolates circulating in china where whole cell vaccines have been used for 50 Years. Clin Infect Dis 61:1028–1029. https://doi.org/10.1093/cid/civ457
Wang Z, Li Y, Hou T et al (2013) Appearance of macrolide-resistant Bordetella pertussis strains in China. Antimicrob Agents Chemother 57:5193–5194. https://doi.org/10.1128/AAC.01081-13
Wang Z, Cui Z, Li Y et al (2014) High prevalence of erythromycin-resistant Bordetella pertussis in Xi’an, China. Clin Microbiol Infect 20:O825-830. https://doi.org/10.1111/1469-0691.12671
Weiss AA, Falkow S (1984) Genetic analysis of phase change in Bordetella pertussis. Infect Immun 43:263–269. https://doi.org/10.1128/iai.43.1.263-269.1984
Weiss A, Moore BD, Tremblay MHJ et al (2017) The ω Subunit Governs RNA Polymerase Stability and Transcriptional Specificity in Staphylococcus aureus. J Bacteriol. https://doi.org/10.1128/jb.00459-16
Xu Y, Zhang L, Tan Y et al (2015) Genetic diversity and population dynamics of Bordetella pertussis in China between 1950–2007. Vaccine 33:6327–6331. https://doi.org/10.1016/j.vaccine.2015.09.040
Xu Z, Octavia S, Luu LDW et al (2019a) Pertactin-negative and filamentous hemagglutinin-negative bordetella pertussis, Australia, 2013–2017. Emerg Infect Dis 25:1196–1199. https://doi.org/10.3201/eid2506.180240
Xu Z, Wang Z, Luan Y et al (2019b) Genomic epidemiology of erythromycin-resistant Bordetella pertussis in China. Emerg Microbes Infect 8:461–470. https://doi.org/10.1080/22221751.2019.1587315
Yamaguchi T, Kawasaki Y, Katsukawa C et al (2020) The First Report of Macrolide-Resistant Bordetella pertussis Isolation in Japan. Jpn J Infect Dis 73:361–362. https://doi.org/10.7883/yoken.JJID.2019.421
Yin X, Song F, Gong Y et al (2013) A systematic review of antibiotic utilization in China. J Antimicrob Chemother 68:2445–2452. https://doi.org/10.1093/jac/dkt223
Yin W, Wang Y, Liu L et al (2019) Biofilms: the microbial “protective clothing” in extreme environments. Int J Mol Sci. https://doi.org/10.3390/ijms20143423
Zeng J, Geng M, Jiang H et al (2007) The IscA from acidithiobacillus ferrooxidans is an iron-sulfur protein which assemble the [Fe4S4] cluster with intracellular iron and sulfur. Arch Biochem Biophys 463:237–244. https://doi.org/10.1016/j.abb.2007.03.024
Acknowledgements
We are grateful to Dr. Laurence Don Wai Luu and Ruiting Lan for their help. We would like to thank Shanghai Applied Protein Technology Co. Ltd. for providing technical support.
Funding
This work was supported by the Natural Science Foundation of China [Grant No. 82172312]; the Key Research and Development Program of Shaanxi Province [Grant No. 2021SF-003]; and the Xi'an Science and technology project [Grant No. 20YXYJ0006(1)].
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The study was designed by HY and ZW and mainly performed by ZL and SY. Material preparation was performed by KJ and YL. Data collection was performed by WW, SW and JS. Data analysis was performed by ZL and XM. The first draft of the manuscript was written by ZL and SY. And all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lv, Z., Yin, S., Jiang, K. et al. The whole-cell proteome shows the characteristics of macrolides-resistant Bordetella pertussis in China linked to the biofilm formation. Arch Microbiol 205, 219 (2023). https://doi.org/10.1007/s00203-023-03566-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03566-0