Abstract
The multi-elemental composition of 4 edible wild-growing mushroom species that commonly occur in Polish forests was compared to 13 cultivated mushroom species available in trade. A considerable variation in the macroelements content was revealed with cultivated species containing higher amounts of macroelements. The mean content of B, Co, Cr, Fe, Pb, Pr, Pt, Sb, Sm, Sr, Te, and Tm was higher in cultivated mushroom species, while the opposite was noted for Ba, Cd, Cu, Hg, La, Mo, Sc, and Zn. Selected cultivated forms exhibited increased content of Al (F. velutipes), As (H. marmoreus, F. velutipes), Ni (P. ostreatus, A. polytricha, H. marmoreus), and Pb (P. ostreatus, A. polytricha, F. velupites, and L. edodes). Wild-growing species, B. boletus, I. badia, and S. bovinus contained high Hg levels, close to or exceeding tolerable intakes. Compared to cultivated mushrooms, they also generally revealed a significantly increased content of Al (with the highest content in B. edulis and I. badia), As and Cd (with the highest content in B. edulis and S. bovinus in both cases). In turn, the cultivated mushrooms were characterized by a higher content of Ni (particularly in A. bisporus) and Pb (with the highest content in P. eryngii). The exposure risks may, however, differ between wild and cultivated mushrooms since the former are consumed seasonally (although in some regions at a high level), while the latter are available throughout the year. Both cultivated and wild-growing mushrooms were found to be a poor source of Ca and Mg, and only a supplemental source of K, Cu, Fe, and Zn in the human diet. These results suggest that mushrooms collected from the wild or cultivated, should be consumed sparingly. The study advocates for more strict monitoring measures of the content of toxic metals/metalloids in mushrooms distributed as food, preferentially through the establishment of maximum allowance levels not limited only to a few elements and mushroom species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
There is considerable interest in mushrooms due to their taste, nutritional value as well as potential risks associated with their consumption [1,2,3,4]. The vast majority of these studies focus on one or several years, during which sporocarps were collected for the purpose of investigating the content of macro- and/or trace elements [5]. Attention has been directed to selected or all detectable elements in fruiting bodies, depending on the site of their collection, species, and availability [6, 7]. These studies have also included the analysis of both cultivated and wild-growing mushroom species, the latter being divided into wood-growing and aboveground species [8, 9].
Although analysis of elements has been the subject of numerous papers for the last 20 years [10, 11], in the case of selected elements such an as Hf, Nb, Ta, Tm or W, literature data are still highly limited [12]. The development of analytical chemistry has allowed the content of these elements to be determined in different mushroom species [13, 14]. The content of elements in particular mushroom species is highly diverse and is usually mushroom species-dependent [12]. Mushrooms are able to accumulate elements more effectively than the majority of vascular plant species (excluding hyperaccumulators). Therefore, the assessment of the content, especially of trace elements with detrimental health effects such as Ag, As, Be, Cd, Pb or Tl, is highly relevant in the case of human consumption [12].
The global production of cultivated mushrooms is continuously increasing [15, 16]. The total worldwide production of cultivated edible mushroom species, including truffles, in 2016 was up to 11 billion metric tons, with the highest output in China, Italy, the USA, the Netherlands and Poland (72.3; 6.34; 3.89; 2.78, and 2.41%, respectively of global production) [12]. A variety of cultivated mushroom species available in the trade have been over the last 15 years. The majority of these studies focused on different species or species of the same genus [5, 7, 17, 18]. The mineral composition of cultivated mushroom species described in literature data is diverse, as in the case of, for example, Agaricus bisporus fruit bodies, which have been clearly explained by Bosiacki et al. [19].
The aim of the present study was to compare the mineral composition of 4 of the most popular wild-growing mushroom species marketed near Polish roads and 13 cultivated mushroom species available in markets in Poland and selected countries in 2018 and 2019. It was assumed that because cultivated mushrooms originate from processes carried out under controlled conditions, they should have a lower content of elements than wild-growing mushroom species.
Materials and methods
Characteristics of experimental materials
Cultivated mushrooms
The cultivated mushroom species were: Agaricus bisporus (Lange) Imbach (bs); Agaricus bisporus (Lange) Imbach (bs, portobello); Agrocybe cylindracea (DC.) Maire; Auricularia polytricha (Mont.) Sacc.; Flammulina velutipes (Curtis) Singer; Hypsizygus marmoreus (Peck) Bigelow (ws); Hypsizygus marmoreus (Peck) Bigelow (bs); Lentinula edodes (Berk.) Pegler; Pleurotus citrinopileatus Singer; Pleurotus djamor (Rumph. ex Fr.) Boedijn; Pleurotus eryngii (DC.) Quèl.; Pleurotus ostreatus (Jacq.) Kumm. and Tremella fuciformis Berk. The studied mushrooms were purchased in shops and on the markets of the largest cities in particular countries in 2017, 2018, and 2019. The mass of fresh fruit bodies ranged from 100 to 500 g, while dry samples ranged from 30 to 100 g, depending on mushroom species and package size. Prior to transport to Poland, the purchased fruit bodies were dried at a temperature of 45 °C for 96 h using laboratory ovens available in universities or laboratories in certain cities to determine the content of dry matter and protect the experimental material during transport to the laboratory. The number of purchased packages together with information about the place of mushroom production and sale are described in Table 1.
Wild mushrooms
The wild-growing mushroom species were: Boletus edulis Bull., Imleria badia (Fr.), Leccinum scabrum (Bull.) Gray and Suillus bovinus (L.) Roussel. Particular wild-growing mushroom species were marketed in the vicinity of the national routes (DK3, DK24, DK32 and DK11) localized in Poland. Mushrooms were purchased in 2017, 2018, and 2019 from 10 different locations as 250 g of fresh mass samples each time. All samples were directly transported to the laboratory. The species of the purchased mushrooms was confirmed by the qualified and certified mycologist on the basis on their morphological features.
Procedure
All fruit bodies were carefully cleaned with distilled water from the rest of underlying soil or substrate to prevent external contamination. All samples were dried in an electric oven (at 45 ± 1 °C for 96 h; SLW 53 STD, Pol-Eko, Poland) and the fruit bodies were ground in a laboratory mill PM 200 (Retsch, Haan, Germany). The mass of 0.300 ± 0.001 g of a dry sample was digested with 6 mL of concentrated nitric acid (65%; Sigma-Aldrich, St. Louis, MO, USA) in Teflon containers using a closed microwave sample preparation system (Mars 6 Xpress, CEM USA). After digestion, the samples were filtered (Qualitative Filter Papers Whatman, Grade 595, previously washed in 200 mL of water) and diluted with water from Milli-Q (resistivity 18.2 MΩ cm, Merck Millipore, Darmstadt, Germany) to a final volume of 15.0 mL. Each of the samples was analyzed in three repetitions. All samples were analyzed in one analytical procedure using tools of quality assurance (the control samples).
Instruments
The inductively coupled plasma optical emission spectrometry (Agilent 5110 ICP-OES, Agilent USA) was used for the determination of 68 elements. The following conditions of the analytical procedure were applied: Radio Frequency (RF) power 1.2 kW, 0.7, 1.0, and 12.0 L min−1, respectively for nebulizer gas, auxiliary gas, and plasma gas flows, detector Charge Coupled Device (CCD) temperature −40 °C, the time of signal accusation 5 s for 3 replicates. The range of calibration was LOQ-10 mg/L. The detection limits for all elements were determined (as 3-sigma criteria, based on the standard deviation (sigma) obtained in the multiple blank analysis) at the level of 0.01–0.09 mg kg−1 dry weight (DW). Detailed information is given in Table S1 in Supplementary data. The following elements: Ca, K, Mg, Na, S, Al, As, B, Ba, Cd, Co, Cr, Cu, Fe, Hg, La, Mn, Mo, Ni, Pb, Pr, Sb, Sc, Sm, Sr, Te, Tm, and Zn were determined above the level of the detection limit.
The uncertainty for the total analytical procedure (including sample preparation, for uncertainty budget calculation the coverage factor k = 2 was used) was at the level of 20%. The recovery for the analysis of certified reference materials (CRM NCSDC (73,349)—bush branches and leaves; CRM S-1—loess soil; CRM 2709—soil) was acceptable (80–120%) for most of the determined elements (Table S2 in Supplementary data). For elements without certified values, the standard addition method was adopted. The content of sulfur was checked by a FLASH 2000 analyzer with an FPD detector (Thermo Scientific).
Statistical analysis
Statistical analyses were performed using both STATISTICA 12.0 software (StatSoft, USA) and the Agricolae package (R, Bell Laboratories). For a general comparison of the mean content of macro- and trace elements in all wild-growing and cultivated mushroom species, the one-way multivariate analysis of variance (MANOVA) with the Hotelling-Lawley procedure was used. To indicate uniform groups of objects (α = 0.05), one-dimensional analysis of variance (ANOVA), and finally, the multiple comparison Tukey’s HSD test were performed. The heatmaps were prepared individually for all groups of elements (macro-, trace and all detectable elements, separately) to show diversity between all the studied mushroom species and to show the grouping of similar mushroom species with regard to the content of macro-, trace and all elements jointly, the Hierarchical Cluster Dendrograms were determined [20].
Particular homogenous groups were determined using the ward.D2 agglomeration method (hclust {stats}) with Euclidean Distance. Additionally, according to the calculation of the synthetic Perkal index, the rank-sum was performed to compare all studied mushroom species with respect to their enrichment of macro-, trace and all elements jointly [21].
Results
Content of macroelements
Analysis of macroelements in both groups of mushrooms revealed significant differences between particular species as well as within specific groups (Table 2). The fruit bodies of T. fuciformis and A. bisporus (1870 and 36,300 mg kg−1, respectively) were most enriched with Ca and K The highest content of Mg was determined in A. polytricha and P. citrinopileatus (1720 and 1610 mg kg−1, respectively), while Na and S, were most abundant in P. eryngii and H. mormoreus (ws), (428 and 5270 mg kg−1, respectively). It is important to stress that content of Mg, Na, and S in the majority cultivated mushroom species was significantly higher than for wild-growing mushrooms.
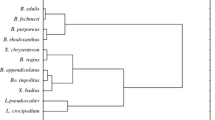
A heatmap, as the graphical presentation of diversity between all the studied mushrooms, allowed differences between studied mushrooms to be shown, and together with the Hierarchical Cluster Dendrograms enabled the grouping of similar mushroom species as regards the content of macroelements. Thus, it is clear that wild-growing mushroom species create a separate group, despite significantly higher contents of K in B. edulis, L. scabrum and S. bovinus than those determined for the majority of cultivated mushroom species (Table 2, Fig. 1a). Additionally, a heatmap allowed two additional groups to be distinguished for cultivated mushroom species including: A.bisporus (bs), A. bisporus (portobello), A. polytricha, H. marmoreus (ws), H. marmoreus (bs), P. citrinopileatus (first group); A. cylindracea, F. velutipes, L. edodes, P. djamor, P. eryngii, P. ostreatus and T. fuciformis (second group).
It is worth underlining that the mean content of Ca, Mg, Na, and S calculated for all cultivated mushroom species (673; 1007; 259 and 2380 mg kg−1, respectively) was higher than the mean content of these metals for wild-growing mushroom species (324; 424; 81.2 and 264 mg kg−1, respectively). The opposite situation was recorded for K, whose mean content in wild-growing mushroom species (26,900 mg kg−1) was higher than that calculated for all cultivated mushroom species (21,400 mg kg−1). It is also significant that the ranges of Ca, Mg, Na, and S content determined for cultivated mushrooms (123–2207; 178–5839; 26.8–833 and 882–9670 mg kg−1, respectively) were wider than for wild mushrooms (95.3–574; 207–603; 20.1–179 and 122–365, respectively).
The rank-sum calculated to compare particular mushroom species as regards their enrichment of macro-, trace or all determined elements allowed the differences between them to be shown. The diversity of the studied mushroom species was revealed as follows: A. polytricha > H. mormoreus (ws) > (H. mormoreus (bs) = A. cylindracea) > A. bisporus > (P. eryngii > P. cirinopileatus = F. velutipes) > A. bisporus (portobello) > (L. edodes = P. djamor = T. fuciformis) > P. ostreatus > S. bovinus > B. edulis > L. scabrum > I. badia with respect to the content of all macroelements. This indicates a generally lower content of macroelements in wild-growing than in cultivated mushroom species.
Content of trace elements
The content of trace elements in all the studied mushroom species was significantly diverse (Table 3). Flamulina velutipes was the most enriched with Al (mean 110 mg kg−1) and also Sm, together with A. cylindracea and T. fuciformis (means 2.22; 2.15 and 2.10 mg kg−1, respectively). Similarly, in H. marmoreus (ws) the highest mean content of As (3.71 mg kg−1) was recorded; however, in this mushroom species, and also A. polytricha, F. velutipes and H. marmoreus (bs), the highest mean content of Cr was also determined (50.9; 49.4; 49.3 and 52.3 mg kg−1, respectively). Lentinula edodes and P. eryngii were the most enriched with B (108 and 95.1 mg kg−1, respectively), while F. velutipes and H. marmoreus (bs) had the highest content of Fe (199 and 144 mg kg−1, respectively). The latter species was also the most effective accumulator of Mn (31.1 mg kg−1). The highest mean content of Pr was determined in A. bisporus, and A. bisporus (portobello) fruit bodies (4.10 and 4.82 mg kg−1). In comparison, P. djamor and P. eryngii were able to accumulate the highest amounts of Pb (2.49 and 3.53 mg kg−1, respectively). The fruit bodies of A. cylindracea were the most enriched with Sc (0.473 mg kg−1), the same as P. ostreatus with Te (1.17 mg kg−1), A. bisporus (portobello) with Ni (3.78 mg kg−1) and A. polytricha and T. fuciformis with Sr (10.3 and 8.26 mg kg−1, respectively). The highest contents of Ba, Cd, Cu, Hg, La, Mo, and Zn, were determined in wild-growing mushroom species. Imleria badia was characterized with the highest mean content of Ba and La (32.2 and 0.241 mg kg−1, respectively), and L. scabrum with the highest level of Cd (3.64 mg kg−1) and a high content of Zn (132 mg kg−1). Boletus edulis was the wild-growing mushroom species with the highest mean contents of Hg, Mo, Sc and Zn (2.85; 0.549; 0.318 and 149 mg kg−1, respectively).
Differences between selected mushroom species were clearly confirmed by a heatmap for trace elements (Fig. 1b). Furthermore, with respect to the accumulation of detectable trace elements, wild-growing species create a separate group of mushrooms. Among the cultivated mushroom species, a clear separate groups of species were composed of: A. polytricha, H. marmoreus (ws), H. marmoreus (bs) and P. citrinopileatus (the first group); L. edodes, P. djamor and P. eryngii (the second group); A. bisporus (bs), A. bisporus (portobello), A. cylindracea, F. velutipes, P. ostreatus, T. fuciformis (the third group). The last fourth group was created by all wild-growing mushroom species.
The mean content of B, Co, Cr, Fe, Pb, Pr, Sb, Sm, Sr, Te, and Tm calculated for all cultivated mushroom species (48.5; 2.20; 43.2; 72.1; 1.96; 1.26; 26.2; 1.36; 4.34; 0.470 and 0.328 mg kg−1, respectively) was higher than the mean content for all wild-growing mushroom species (2.39; 0.156; 0.657; 47.2; 0.981; 0.150; 0.091; 0.027; 0.679; 0.164 and 0.035 mg kg−1, respectively). The opposite situation was observed for Ba, Cd, Cu, Hg, La, Mo, Sc, and Zn. The mean content of the above mentioned metals in wild-growing mushroom species was as follows: 13.2; 1.80; 19.9; 1.68; 0.100; 0.366; 0.139 and 119 mg kg−1, respectively) and above the mean for all cultivated mushroom species (1.23; 0.553; 13.2; 0.237; 0.040; 0.210; 0.059 and 83.6 mg kg−1, respectively. It is worth emphasizing that the ranges in the contents of B, Ba, Co, Cr, Fe, Ni, Pb, Pr, Sb, Sm, Sr, Te, and Tm were different for cultivated (0.010–366; 0.224–5.25; 0.154–4.40; 11.3–59.5; 11.0–515; 0.010–14.1; 0,010–11.5; 0.003–9.92; 0.010–59.8; 0.010–2.90; 0.740–16.4; 0.003–7.30 and 0.003–3.22 mg kg−1, respectively) and wild-growing (0.039–5.93; 0.210–50.1; 0.020–0.92; 0.120–1.54; 22.2–92.3; 0.450–2.67; 0.227–2.21; 0.020–0.420; 0.010–0.210; 0.013–0.051; 0.113–1.68; 0.040–0.429 and 0.010–0.132 mg kg−1, respectively) mushroom species.
In the case of all detectable trace elements, the rank-sum was as follows: A. cylindracea > L. edodes > P. djamor > P. cirinopileatus > H. mormoreus (bs) > B. edulis > P. eryngii > P. ostreatus > A. bisporus > A. polytricha > F. velutipes > I. badia > H. marmoreus (ws) > A. bisporus (portobello) > T. fuciformis > L. scabrum > S. bovinus. Here too the content of all trace elements was lower in wild-growing mushrooms than in cultivated mushroom species, with the exception of B. edulis.
Content of all elements in cultivated and wild-growing mushroom species
The aim of dividing the elements into two groups was to show their effect on the differentiation of fungal species. Nevertheless, the fruit bodies occurred together. Hence, it is first and foremost necessary to evaluate the content of all elements to show the actual differences between species. A heatmap visible in Fig. 1c allowed to show almost the same groups of mushroom species as in Fig. 1b and different to those in Fig. 1a. The rank-sum calculated for all detectable elements was as follows: A. cylindracea > P. cirinopileatus > H. mormoreus (bs) > L. edodes > P. djamor > A. polytricha > P. eryngii > A. bisporus > F. velutipes > P. ostreatus > B. edulis > H. marmoreus (ws) > A. bisporus (portobello) > T. fuciformis > I. badia > L. scabrum > S. bovinus. This confirms that the wild-growing mushroom species marketed in the vicinity of roads were less enriched with elements than the cultivated mushroom species available in world trade.
Discussion
Mushrooms form an important part of the diet in many world regions [22,23,24,25]. Historically, various species have been collected from the wild, although the development of modern fungi-culture techniques have allowed them to be produced under controlled conditions and in large quantities that can be distributed worldwide. This has contributed to the global demand for mushrooms as food. Additionally, various cultivated mushrooms have been suggested as potent functional foods due to the biological properties of their components—as shown using experimental models and clinical trials [26,27,28,29,30,31]. Considering the conditions under which cultivated mushrooms are obtained, one could assume that their quality—regarding the nutritional value and level of contamination—is superior to specimens collected from the wild. However, such an assumption requires a direct comparison, which was the aim of the present study, which examined the content of macro- and trace elements in the most frequently consumed species of both groups in Poland.
Numerous previous studies have separately explored element content in fruiting bodies of wild and cultivated mushrooms [7, 32,33,34,35]. In both cases, increased levels of toxic or potentially toxic elements were reported [12]. It is well established that contamination of mushrooms for human consumption varies between species, most likely due to interspecies differences in biological features, but also because it is largely driven by abiotic conditions, predominantly the pollution of forest soil, in the case of wild mushrooms, or overgrown substrate, as regards cultivated forms. The present study employed the ICP-OES, frequently utilized in studies on elemental content in mushrooms [5, 7, 8, 18, 35], and demonstrated that in terms of metal contamination, wild edible mushrooms in Poland pose a greater risk than popular cultivated species available on the market. Comparison of mean element content in wild and cultivated mushrooms indicated that the former reveal increased levels of the majority of relevant trace elements toxic to humans: Al by 19% (with the highest content in B. edulis and I. badia), As by 20% (with the highest content in B. edulis and S. bovinus), Cd by 225% (with the highest content in B. edulis and L. scabrum), Hg by 609% (with the highest content in B. edulis and S. bovinus). Previous studies have also shown an increased content of As, Cd and Hg in B. edulis [36,37,38]. Moreover, Cd-binding phytochelatins, a family of cysteine-rich oligopeptides, have been identified in this mushroom species [39]. All in all, these and previous findings highlight that B. edulis, which is one of the most culinary-valued and frequently collected mushroom species in various regions of Europe, can be a more significant source of dietary exposure to toxic elements than cultivated fungi.
To at least partially understand the health risks arising from the consumption of the studied cultivated and wild mushrooms, the determined levels of toxic elements were compared to the existing tolerable intake levels. Since all the products were obtained from Poland, the following tolerable daily intakes (TDI; µg kg−1 body weight) proposed by European Food Safety Authority (EFSA) were used to measure the contents in fruiting bodies: Al (143), As (0.21), Cd (0.36), Ni (2.8), Hg (4.0; inorganic) and Pb (3.6) [40,41,42]. Considering these values and assuming a single serving of 25 g of dry mushrooms, their consumption by a 70 kg adult would, in the worse scenario (maximum noted level), account for 88.2 and 31.0% of TDI of Al in the case of F. velutipes and B. edulis. Similarly, consumption of H. marmoreus would maximally account for as much as 166% of TDI of As in the case of H. marmoreus and 67% for F. velutipes. One should, however, note that the present study assessed only the total As content in mushrooms while the toxicity of As is highly associated with forms under which this metalloid is present with the greatest threat posed by inorganic and methylated forms [43, 44]. For Ni, the maximum content, noted in P. ostreatus, A. polytricha and H. marmoreus, would account for 91.8, 61.2 and 56.1%, respectively. Consumption of a single 25 g serving would maximally constitute 99.2% of the TDI of Pb for P. ostreatus and 43% for A. polytricha, F. velupites and L. edodes. However, one should note that the mean contents of Al, As, Cd, Ni, and Pb in the majority of the studied mushrooms fall much below the TDI to consider them as a significant dietary source of exposure. The consumption of all cultivated mushrooms would also not contribute significantly to the TDI of Hg, if one considers the maximum contents noted in their fruiting bodies. However, wild-growing mushrooms, with the exception of I. badia, revealed worrying levels of this metal: the consumption of B. edulis would minimally and maximally account for 177.5 and 232.5% of TDI, in the case of L. scabrum—82.5 and 127.5%, respectively while for S. bovinus—107.5 and 135%, respectively. One should note that the TDI set by the EFSA considers total inorganic content and provides a separate guideline for methylated Hg while in the present study the speciation analysis was not conducted and only total levels of Hg in fruit bodies were reported. Nevertheless, the present study advocates the inclusion of species such as B. edulis, I. badia and L. scabrum, whose fruiting bodies, originating from forests, are available on the market in various European countries, within maximum allowance limits—particularly the level of Hg. In the present study, the total Cr was determined, while from the food safety perspective the content of Cr (VI) is of the highest concern. For Cr (III) the EFSA sets the TDI at the level of 0.3 mg kg−1 body weight [45]. However, the mean Cr content in the cultivated mushrooms was over 65-fold higher than in wild specimen, the consumption of a single serving would constitute only 5.1% of TDI (and 0.08% for wild mushrooms). It can therefore be assumed that Cr in studied mushrooms did not constitute any relevant risk to the consumers.
Importantly, the consumption of cultivated and wild mushrooms is different. The former are often consumed all year round, while the latter are mostly consumed seasonally. This must be taken into account in the assessment of health risks. However, there are regions in which the consumption of wild mushrooms, although limited mostly to autumn seasons, is very high, resulting in the potential high intake of toxic metals and metalloids [24, 25, 46].
All in all, the increased levels in some specimens of the selected mushroom species underline the necessity for the regular monitoring of both cultivated and wild mushrooms available for consumers. A feasible solution would also be to introduce maximum allowance levels for other elements than just Cd and Pb in mushrooms and to apply these levels to all edible wild and cultivated species.
One should note that consumption of edible species, growing in the wild as well as cultivated forms, is known to occasionally cause idiosyncratic reactions. These are mostly manifested through mild gastrointestinal symptoms, although more serious effects, including rhabdomyolysis, have been reported [47,48,49,50,51]. The contamination of fruiting bodies, e.g., with toxic elements, is one of the potential explanations of this phenomenon since the ingestion of increased amounts of metals and metalloids with foodstuffs can be associated with different adverse reactions, including gastrointestinal effects [52,53,54,55].
The present study also addresses the content of macro- and trace elements of nutritional value in wild and cultivated mushrooms. In general, the latter was much more nutritional with respect to their macroelemental composition and revealed a higher content of Ca (by twofold), Mg (by 2.4-fold), Na (by 3.2-fold) and S (by ninefold). Moreover, the cultivated mushrooms were also superior in terms of Cr and Fe content, which compared to wild mushrooms was by 65.7-fold and 1.5-fold higher, respectively.
Considering that mushrooms are increasingly consumed and marketed for their nutritional value [12], the macro- and trace elements observed in the present study were compared to Adequate Intakes (AIs) for adults established by the European Food Safety Authority [56] at the level of Ca—950 mg, Mg—350 mg, K—3500 mg, Cu—1.6 mg, Fe—11 mg, Mn—3.0 mg and Zn—11.7 mg. Assuming a consumption of 25 g by a 70-kg adult, the cultivated and wild mushrooms would meet these requirements at the following respective levels: Ca—1.9% (max. 5.8% for T. fuciformis) and 0.8% (max. 1.5% for B. edulis), Mg—7.4% (max. 41.7% for A. polytricha) and 3.0% (max. 4.3% for S. bovinus), K—16.2% (max. 34.0% for A. bisporus) and 19.2% (max. 31.2% for S. bovinus), Cu—18.7% (max. 100% for P. ostreatus) and 31.6% (max. 97.4% for S. bovinus), Fe—18.2% (max. 117% for F. velutipes) and 11.0% (max. 20.9% for I. badia), Mn—10% (max. 35.6% for H. marmoreus) and 10% (max. 20.4% for B. edulis), Zn—17.1% (max. 46.6% for A. bisporus) and 25.6% (max. 39.0% for B. edulis). Such juxtaposition clearly shows that the studied mushrooms, both cultivated and wild-growing, can only be considered as an additional, supplemental source of minerals such as K, Cu, Fe, and Zn, but cannot serve as the primary source of their delivery in the human diet.
Conclusion
The results of the present study generally indicate a higher elemental content in cultivated than wild mushroom species. Moreover, the higher contents of elements in cultivated mushroom species and high ranges of the determined content suggest that in cultivation processes, substrates with insufficient purity are used, especially by producers from selected countries in the world. The present study shows that both cultivated and wild mushrooms pose some risks to human health, mainly if consumed more often. In the case of cultivated forms worrying levels of Al, As, Ni, and Pb were found while wild-growing mushrooms exhibited high Hg content. At the same time, both groups of mushrooms were demonstrated to be a poor source of various minerals and in the case of K, Cu, Fe, and Zn they could only serve as a secondary source in the human diet. This considered we argue that mushrooms should only be consumed occasionally and that more strict measures are required to ensure the safety and quality of marketed cultivated and wild mushrooms.
Declaration
Conflict of interest
The authors declare that there is no conflict of interest.
Compliance with ethics requirement
This article does not contain any studies with human participants or animals performed by any of the authors.
References
Falandysz J, Borovička J (2013) Macro and trace mineral constituents and radionuclides in mushrooms: health benefits and risks. Appl Microbiol Biotechnol 97:477–501
Kalač P (2010) Trace element contents in European species of wild growing edible mushrooms: a review for the period 2000–2009. Food Chem 122:2–15
Kalač P (2016) Proximate composition and nutrients. In: Kalač P (ed) Edible mushrooms. Elsevier, Boston, pp 7–69
Świsłowski P, Dołhańczuk-Śródka A, Rajfur M (2020) Bibliometric analysis of European publications between 2001 and 2016 on concentrations of selected elements in mushrooms. Environ Sci Pollut Res 27:22235–22250
Niedzielski P, Mleczek M, Budka A, Rzymski P, Siwulski M, Jasińska A, Gąsecka M, Budzyńska S (2017) A screening study of elemental composition in 12 marketable mushroom species accessible in Poland. Eur Food Res Technol 243:1759–1771
Falandysz J, Szymczyk K, Ichihashi H, Bielawski L, Gucia M, Frankowska A, Yamasaki S-I (2001) ICP/MS and ICP/AES elemental analysis (38 elements) of edible wild mushrooms growing in Poland. Food Addit Contam A 18:503–513
Siwulski M, Mleczek M, Rzymski P, Budka A, Jasińska A, Niedzielski P, Kalač P, Gąsecka M, Budzyńska S, Mikołajczak P (2017) Screening the multi-element content of pleurotus mushroom species using inductively coupled plasma optical emission spectrometer (ICP-OES). Food Anal Method 10:487–496
Mleczek M, Niedzielski P, Kalač P, Budka A, Siwulski M, Gąsecka M, Rzymski P, Magdziak Z, Sobieralski K (2016) Multielemental analysis of 20 mushroom species growing near a heavily trafficked road in Poland. Environ Sci Pollut Res 23:16280–16295
Kokkoris V, Massas I, Polemis E, Koutrotsios G, Zervakis GI (2019) Accumulation of heavy metals by wild edible mushrooms with respect to soil substrates in the Athens metropolitan area (Greece). Sci Total Environ 685:280–296
Wang X-M, Zhang J, Li T, Wang Y-Z, Liu H-G (2015) Content and bioaccumulation of nine mineral elements in ten mushroom species of the genus Boletus. J Anal Method Chem 2015:165412–165412
Mirończuk-Chodakowska I, Socha K, Zujko ME, Terlikowska KM, Borawska MH, Witkowska AM (2019) Copper, manganese, selenium and zinc in wild-growing edible mushrooms from the eastern territory of “green lungs of poland”: nutritional and toxicological implications. Int J Environ Res Public Health 16:3614
Kalač P (2019) Mineral composition and radioactivity of edible mushrooms. Academic Press, Elsevier Inc
Falandysz J, Chudzińska M, Barałkiewicz D, Drewnowska M, Hanć A (2017) Toxic elements and bio-metals in Cantharellus mushrooms from Poland and China. Environ Sci Pollut Res 24:11472–11482
Koutrotsios G, Danezis GP, Georgiou CA, Zervakis GI (2018) Rare earth elements concentration in mushroom cultivation substrates affects the production process and fruit-bodies content of Pleurotus ostreatus and Cyclocybe cylindracea. J Sci Food Agric 98:5418–5427
FAOSTAT (2016) http://faostat3.fao.org/browse/Q/QC/E
USDA (2018) Mushrooms. The National Agricultural Statistics Service (NASS), Agricultural Statistics Board
Vetter J (1994) Mineral elements in the important cultivated mushrooms Agaricus bisporus and Pleurotus ostreatus. Food Chem 50:277–279
Mleczek M, Siwulski M, Rzymski P, Budka A, Kalač P, Jasińska A, Gąsecka M, Budzyńska S, Niedzielski P (2018) Comparison of elemental composition of mushroom Hypsizygus marmoreus originating from commercial production and experimental cultivation. Sci Hortic Amsterdam 236:30–35
Bosiacki M, Krzebietke S, Bosiacki M, Siwulski M, Sobieralski K (2018) The content of selected heavy metals in fruiting bodies of Agaricus bisporus (Lange) Imbach. wild growing in Poland. J Elementol 23:875–886
Falniowski A (2003) The numerical methods in taxonomy. [Metody numeryczne w taksonomii]. WUJ, Kraków [in Polish]
Smith DM (1972) Geography and social indicators. S Afr Geogr J 54:43–57
Mortimer PE, Karunarathna SC, Li Q, Gui H, Yang X, Yang X, He J, Ye L, Guo J, Li H, Sysouphanthong P, Zhou D, Xu J, Hyde KD (2012) Prized edible Asian mushrooms: ecology, conservation and sustainability. Fungal Divers 56:31–47
Klimaszyk P, Rzymski P (2018) The yellow knight fights back: toxicological, epidemiological, and survey studies defend edibility of Tricholoma equestre. Toxins 10:468
Kotowski MA, Pietras M, Łuczaj Ł (2019) Extreme levels of mycophilia documented in Mazovia, a region of Poland. J Ethnobiol Ethnomed 15:12–12
Svanberg I, Lindh H (2019) Mushroom hunting and consumption in twenty-first century post-industrial Sweden. J Ethnobiol Ethnomed 15:42–42
Abrams DI, Couey P, Shade SB, Kelly ME, Kamanu-Elias N, Stamets P (2011) Antihyperlipidemic effects of Pleurotus ostreatus (oyster mushrooms) in HIV-infected individuals taking antiretroviral therapy. BMC Complement Altern Med 11:60–60
Dai X, Stanilka JM, Rowe CA, Esteves EA, Nieves C, Spaiser SJ, Christman MC, Langkamp-Henken B, Percival SS (2015) Consuming Lentinula edodes (Shiitake) mushrooms daily improves human immunity: a randomized dietary intervention in healthy young adults. J Am Coll Nutr 34:478–487
Tang C, Hoo PC-X, Tan LT-H, Pusparajah P, Khan TM, Lee L-H, Goh B-H, Chan K-G (2016) Golden needle mushroom: a culinary medicine with evidenced-based biological activities and health promoting properties. Front Pharmacol 7:474–474
Hess J, Wang Q, Gould T, Slavin J (2018) Impact of Agaricus bisporus mushroom consumption on gut health markers in healthy adults. Nutrients 10:1402
Hess JM, Wang Q, Kraft C, Slavin JL (2017) Impact of Agaricus bisporus mushroom consumption on satiety and food intake. Appetite 117:179–185
Poniedziałek B, Siwulski M, Wiater A, Komaniecka I, Komosa A, Gąsecka M, Magdziak Z, Mleczek M, Niedzielski P, Proch J, Ropacka-Lesiak M, Lesiak M, Henao E, Rzymski P (2019) The effect of mushroom extracts on human platelet and blood coagulation: in vitro screening of eight edible species. Nutrients 11:3040
Borovička J, Řanda Z (2007) Distribution of iron, cobalt, zinc and selenium in macrofungi. Mycol Prog 6:249–259
Falandysz J, Rizal LM (2016) Arsenic and its compounds in mushrooms: a review. J Environ Sci Healt C 34:217–232
Siobud-dorocant E, Doré JC, Michelot D, Poirier F, Viela C (1999) Multivariate analysis of metal concentration profiles in mushrooms. SAR QSAR Environ Res 10:315–370
Siwulski M, Budka A, Rzymski P, Gąsecka M, Kalač P, Budzyńska S, Magdziak Z, Niedzielski P, Mleczek P, Mleczek M (2020) Worldwide basket survey of multielemental composition of white button mushroom Agaricus bisporus. Chemosphere 239:124718
Falandysz J, Zhang J, Wang Y-Z, Saba M, Krasińska G, Wiejak A, Li T (2015) Evaluation of mercury contamination in fungi Boletus species from latosols, lateritic red earths, and red and yellow earths in the circum-pacific mercuriferous belt of Southwestern China. PLoS ONE 10:e0143608–e0143608
Frankowska A, Ziółkowska J, Bielawski L, Falandysz J (2010) Profile and bioconcentration of minerals by King Bolete (Boletus edulis) from the Płocka Dale in Poland. Food Addit Contam B 3:1–6
Mleczek M, Niedzielski P, Rzymski P, Siwulski M, Gąsecka M, Kozak L (2016) Variations of arsenic species content in edible Boletus badius growing at polluted sites over four years. J Environ Sci Healt B 51:469–476
Collin-Hansen C, Pedersen SA, Andersen RA, Steinnes E (2007) First report of phytochelatins in a mushroom: induction of phytochelatins by metal exposure in Boletus edulis. Mycologia 99:161–174
EFSA (2009) Cadmium in food—scientific opinion of the panel on contaminants in the food chain. EFSA J 980:1–139
EFSA (2009) Scientific opinion on arsenic in food. EFSA J 7:1351
EFSA (2012) Scientific Opinion on the risk for public health related to the presence of mercury and methylmercury in food. EFSA J 10:2985
Ventura-Lima J, Bogo MR, Monserrat JM (2011) Arsenic toxicity in mammals and aquatic animals: a comparative biochemical approach. Ecotoxicol Environ Saf 74:211–218
Braeuer S, Goessler W (2019) Arsenic species in mushrooms, with a focus on analytical methods for their determination—a critical review. Anal Chim Acta 1073:1–21
EFSA (2014) Scientific opinion on the risks to public health related to the presence of chromium in food and drinking water. EFSA J 12:3595
Peintner U, Schwarz S, Mešić A, Moreau PA, Moreno G, Saviuc P (2013) Mycophilic or mycophobic? Legislation and guidelines on wild mushroom commerce reveal different consumption behaviour in European countries. PLoS ONE 8:e63926
Akilli NB, Dundar ZD, Koylu R, Gunaydin YK, Cander B (2014) Rhabdomyolysis induced by Agaricus Bisporus. J Acad Emerg Med 13:212–213
Benjamin DR (1995) Mushrooms: poisons and panaceas: a handbook for naturalists, mycologists, and physicians. W.H. Freeman, New York
Chwaluk P (2013) Rhabdomyolysis as an unspecyfic symptom of mushroom poisoning—a case report. Przegla̧d Lekarski 70:684–686
Gawlikowski T, Romek M, Satora L (2014) Edible mushroom-related poisoning. Human Exp Toxicol 34:718–724
Rzymski P, Klimaszyk P (2018) Is the yellow knight mushroom edible or not? A systematic review and critical viewpoints on the toxicity of Tricholoma equestre. Compr Rev Food Sci Food Saf 17:1309–1324
Ibrahim D, Froberg B, Wolf A, Rusyniak DE (2006) Heavy metal poisoning: clinical presentations and pathophysiology. Clin Lab Med 26:67–97
Govorushko S, Rezaee R, Dumanov J, Tsatsakis A (2019) Poisoning associated with the use of mushrooms: a review of the global pattern and main characteristics. Food Chem Toxicol 128:267–279
Rai PK, Lee SS, Zhang M, Tsang YF, Kim K-H (2019) Heavy metals in food crops: health risks, fate, mechanisms, and management. Environ Int 125:365–385
Rzymski P, Niedzielski P, Kaczmarek N, Jurczak T, Klimaszyk P (2015) The multidisciplinary approach to safety and toxicity assessment of microalgae-based food supplements following clinical cases of poisoning. Harmful Algae 46:34–42
EFSA (2017) Dietary reference values for nutrients summary report. EFSA Support Publ e15121:98
Acknowledgement
This research was financially supported by the framework of the Ministry of Science and Higher Education program "Regional Initiative of Excellence" in 2019-2022, Project No. 005/RID/2018/19.
Author information
Authors and Affiliations
Contributions
Conceptualization: M.M. and M.S.; Formal analysis: M.M., M.S., P.M. and P.R.; Investigation and methodology: S.B., J.P., P.N.; Supervision: M.M., M.S.; Statistical analysis: A.B. and M.G.; Visualization: M.M. and A.B.; Writing—original draft: M.M., M.G., P.M, P.N., S.B., P.R; Writing—review & editing: M.M. and P.R.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mleczek, M., Budka, A., Siwulski, M. et al. A comparison of toxic and essential elements in edible wild and cultivated mushroom species. Eur Food Res Technol 247, 1249–1262 (2021). https://doi.org/10.1007/s00217-021-03706-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-021-03706-0