Abstract
Postoperative cognitive dysfunction (POCD) is a kind of serious postoperative complication in surgery with general anesthesia and it may affect patients’ normal lives. Activated microglia are thought to be one of the key factors in the regulation of POCD process. Once activated, resident microglia change their phenotype and secrete kinds of cytokines to regulate inflammatory response in tissues. Among these secretory factors, brain-derived neurotrophic factor (BDNF) is considered to be able to inhibit inflammation response and protect nervous system. Therefore, the enhancement of BDNF expression derived from resident microglia is suggested to be potential treatment for POCD. In our study, we focused on the role of C8-ceramide (a kind of interventional drug) and assessed its regulatory effect on improving the expression of BDNF secreted from microglia to treat POCD. According to the results of our study, we observed that C8-ceramide stimulated primary microglia to up-regulate the expression of BDNF mRNA after being treated with lipopolysaccharide (LPS) in vitro. We proved that C8-ceramide had ability to effectively improve POCD of mice after being accepted carotid artery exposure and their abnormal behavior recovered better than that of mice from the surgery group. Furthermore, we also demonstrated that C8-ceramide enhanced the cognitive function of mice via the PKCδ/NF-κB signaling pathway. In general, our study has confirmed a potential molecular mechanism that led to the occurrence of POCD caused by surgery and provided a new clinical strategy to treat POCD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
POCD is a kind of serious complication after surgery with general anesthesia. People who suffer from POCD always have persistent disturbances in memory, delirium, poor comprehension and inattention in social activities (Krenk et al. 2010; Monk and Price 2011; Yang et al. 2022). Millions of patients worldwide accept surgery with general anesthesia every year, so the potential population with surgery-induced POCD cannot be ignored. The previous studies have showed that the incidence of POCD was as high as 25.8% on the 1st week after surgery and it was still 10% on the 3rd month after surgery (Moller et al. 1998). Severe POCD may prolong patients’ hospital stay, seriously affect their life quality, and also lead to high mortality rate (Steinmetz et al. 2009). POCD will cause new pain to patients after surgery and bring a heavy burden to society and medical insurance (Fondeur et al. 2022). How to prevent and treat POCD has gradually become important and necessary to academic circles in recent years.
In previous studies, scholars have thought that the occurrence of POCD was induced by an extremely complex molecular network. Among them, inflammatory response, oxidative stress and neurotrophic factor deprivation are important to the process of POCD (Golia et al. 2019; Saxena et al. 2019). It should be pointed out that the neuroinflammation caused by surgery has been widely accepted to be able to induce POCD (Alam et al. 2018; Kawano et al. 2018). Many previous studies have showed that the occurrence of POCD was often accompanied by a significant increase in the expression of inflammatory factors in the nervous system, which has suggested that neuro-inflammation regulatory should receive broader attention in the treatment of POCD (Lin et al. 2020; Su et al. 2020). The activation of the peripheral immune system after inhalation general anesthesia can enhance the expression of inflammatory factors and the accumulation of reactive oxygen species in the central nervous system (CNS), which will further lead to the activation of microglia (Saxena et al. 2019; Hovens et al. 2015; Goehler et al. 2000).
Microglia are the resident immune cells originating in the yolk sac in the CNS and they have ability to migrate into the brain at the early stage of life (Degos et al. 2013). Microglia are widely distributed in the CNS and they regulate the stress response in certain circumstances (Askew et al. 2017). In the previous studies, activated microglia had ability to release pro-inflammatory factors and reactive oxygen compounds, which led to the occurrence of POCD (Saxena et al. 2019). During the inflammatory process, inflammatory related signaling pathways are activated, which improves the secretion of pro-inflammatory factors derived from immune cells and induces the process of tissue inflammatory. The PKCδ/NF-κB signaling pathway has been considered as a classical pro-inflammatory signaling pathway. The activation of NF-κB up-regulate the expression of tumor necrosis factor ɑ (TNFɑ), interleukin-1 (IL-1), IL-6, IL-8 and adhesion molecules in the tissues (Hoesel and Schmid 2013). The pro-inflammatory factors are able to accelerate the aggregation of leukocytes and further enhance tissue inflammation. The activation of NF-κB is a phosphorylation process under the regulation of kinds of kinases, such as mitogen-activated protein kinase family (MAPK), protein kinase C (PKC) and Akt (Chen et al. 2023; Dan et al. 2008; Moscat et al. 2003). Among them, PKCδ in the PKC family is important to effectively phosphorylate NF-κB (the signal to activate the PKCδ/NF-κB signaling pathway) and stimulate immune cascade reactions.
The role of microglia in the stress response depends on the fact that they not only secrete inflammatory factors to damage tissues, but also secrete neurotrophic factors (such as BDNF) to repair injured tissues (Parkhurst et al. 2013). BDNF belongs to neurotrophin family, which is able to induce the neural differentiation of stem cells and has anti-inflammatory effects to improve the microenvironment in the CNS (Ge et al. 2019; Wang et al. 2019). BDNF determines the neuron-glia communication and maintain the homeostasis in the nervous system (Fields and Burnstock 2006; Takasaki et al. 2008). In the inflammatory tissues, pro-inflammatory factors directly damage neurons’ function and synaptic plasticity, and then microglia receive the signals from injured neurons to up-regulate the expression of BDNF (Gómez-Casati et al. 2010). In this process, the role of the BDNF/TrkB signaling pathway is thought to be important in the secretion of BDNF, and several previous studies have found the activation of the BNDF/TrkB signaling pathway was able to enhance the expression level of BDNF derived from the microglia (Lee et al. 2010). Some other studies have also showed that the improving expression of BDNF had positive impact on patients’ cognitive function in a certain degree after surgery (Fan et al. 2016; Calabrese et al. 2014). In general, how to enhance the concentration of BDNF in the CNS may become a potential therapeutic strategy to POCD.
Ceramide family contains kinds of drugs acting as lipid second messengers and they are involved in various cellular activities, including cell growth, differentiation and inflammation (Singh et al. 2012). C8-ceramide is a synthetic cell-permeable short chain ceramide analog and it is used in studies to explore the pharmacological effect of nature ceramide (Hamanaka et al. 2002). A previous study has demonstrated that ceramide could decrease the concentration of NO and PGE2 induced by LPS via the inhibition of NF-κB, AP-1and PKC (Hsu et al. 2001). C8-ceramide has been proved to have ability to reduce the expression of IL-6 and TNF-α derived from activated macrophages (Chiba et al. 2007). Some scholars pointed out in their studies that the anti-inflammatory effect of exogenous ceramide on microglia may be the result of their inhibitory regulation on the MAPKs, PI3K/Akt and Jak/STAT signaling pathways (Jung et al. 2013). Furthermore, Nakajima et al. have demonstrated that C8-ceramide could enhance the expression of BDNF derived from microglia (Nakajima et al. 2002).
Therefore, we hypothesized that C8-ceramide had ability to play protective effect on cognitive function via promoting the expression of BDNF and suppressing neuro-inflammation. In this study, primary microglia were stimulated by LPS and C57BL/6J mice were subjected to right carotid artery exposure. The expression of BDNF and pro-inflammatory cytokines (IL-6, TNF-α) was assessed at different time after surgery. The learning and memory of mice in all groups were evaluated by the Morris Water Maze (MWM) test and fear conditioning test. The expression level of related signaling pathway proteins in hippocampus of mice were tested by western blot and the polarization of microglia was detected by immunofluorescence staining.
Methods
Cell culture
Microglia were harvested from C57BL/6J mice as previously described (Chhor et al. 2013). In brief, C57BL/6J mice (within one day after birth) were soaked into 75% ethanol approximately 10 s and then dissected their cortices in D-Hank’s balanced salt solution with 2% penicillin–streptomycin (PS, Gibco, USA). The cortices were quickly moved into centrifuge tube containing pre-cooled high glucose Dulbecco’s modified Eagle’s medium (DMEM, Gibco, USA) supplemented with 10% fetal bovine serum (FBS, Gibco, USA) and 1% PS, and then dissociated them into small pieces for later using. The cell suspension was keep in culture incubator (37 ℃, 5% CO2). Microglia were isolated from mixed glial cells on the 12th to 14th day. Microglia were collected after centrifugation (1000 rpm × 15 min) and replated in culture dishes containing DEME with 10% FBS at a density of 7.5 × 105 cells/ml. The purity of microglia was revealed by immunofluorescence staining by testing the expression of Iba-1 (the specific marker to microglia) (n = 3). The harvested microglia were be divided into the following four groups: the control group, the LPS treated group, the LPS + C8-ceramide treated group and the C8-ceramide treated group. The mice would accept C8-ceramide (25 μM) treatment 30 min before LPS intervention (1 μg/ml). The total RNA was collected at the 24th hour after treatment to test the expression of BDNF mRNA in all four groups.
Animals and surgery
The eight-weeks-male C57BL/6J mice (24–28 g) were purchased from Chengdu Dossy experimental animal company, LTD. Before the surgery, all mice were given sufficient food and water in the comfortable and stable environment (12 h light/dark cycle; 24 ± 1 ℃ room temperature; 55 ± 10% relative humidity) to adapt to the new environment. All mice were randomly divided into four groups: the control group, the surgery group, the surgery + C8-ceramide group and the C8-ceramide group. There were 43 mice in each group. The mice in the surgery + C8-ceramide group and the C8-ceramide group were treated with C8-ceramide at the dosage of 10 mg/kg (0.1 ml per mouse) through intraperitoneal injection for 4 consecutive days before being subjected to surgery (Ponnapakkam et al. 2018; Balakrishnan et al. 2016).
In our previous study, the right carotid artery exposure surgery was used to construct animal POCD disease model (Fan et al. 2016). In the operation, the mice were monitored and stayed on a heating blanket at a stable temperature of 37 ℃. A 1.5 cm-length incision was made in the central of neck after 30-min sevoflurane inhalation. The soft tissue covering the trachea was gently removed to expose the common carotid artery. The common carotid artery was completely separated and explored without injuring the vagus nerve. Then, the wound was disinfected and sutured carefully. The operation was performed in a sterile environment and lasted approximately 10 min. The mice were finally anesthetized by 3% sevoflurane inhalation for 2 h. All animals were put back into their own cages after awakening.
Behavioral tests
MWM test
The mice in all groups were tested by the MWM test to assess their spatial memory ability as previously described (Vorhees and Williams 2006). The MWM test chamber was a circular tank with a 122 cm diameter and a 50 cm height, and there was a submerged platform placed 1 cm below the water surface. The temperature and humidity in the MWM test room were constant and the water temperature was keep in 24 ± 1℃. It must be pointed out that all things in the test room were stayed in the same place without any movement throughout the test period.
The MWM test chamber was equally divided into four quadrants and the submerged platform quadrant located in one of quadrants, which was called the target quadrant. The swimming path of mice was recorded by the WMT-100s water maze video analysis system. In the training period, mice in all groups were given a five-day training, which contained 4 trials per day and 60 s per trial (the time interval between two trials was 30 min). Once the mice stayed on the platform for at least 15 s, the system would stop timing and it was defined as the escape latency period. In the testing period, the mice were placed at the opposite side of the target quadrant and the hidden platform was removed. The results were shown as how many times mice passed through the platform and how long they stayed in the target quadrant within 60 s.
Fear conditioning test
The fear conditioning test included the context-related fear test and tone-related fear test, and these two parts were able to test hippocampal-dependent and non-hippocampal-dependent cognitive function respectively. The previous studies about the fear conditioning test were referred in our experiment design (Zhang et al. 2014). In our study, the training began on the 13th day after surgery and test at 24 h after the training (Wang et al. 2023.) The mice in all groups were placed in a dark test chamber which was wiped with 70% alcohol in advance, and then they were stimulated by three tone-foot shock pairings (tone 2000 Hz, 85 db, 30 s; foot shock 0.7 mA, 2 s) with 1-min time interval. The context-related test started at the 24th hour after training. Then, mice were put back to the same test chamber for 8 min without any shock and recorded their freezing behavior. Mice were placed in a relatively light chamber, where was wiped with H2O2 in advance and the tone stimulation (2000 Hz, 85 db, 30 s) was turned on for three cycles with 1-min time interval. All mouse freezing behavior was recorded by a camera and their freezing time was used to evaluate their memory function.
Blood and brain tissue harvesting
Mice in all groups were anesthetized with sevoflurane and their thoracic cavity was opened to expose the heart. The blood samples of mice in each group were collected directly from their hearts. All mice were perfused with ice-cold normal saline. The hippocampus of mice in all groups were isolated immediately and stored in liquid nitrogen. The brain tissues were fixed in 4% paraformaldehyde and coronary artery was take out for immunofluorescence staining. The blood samples were centrifuged for 15 min at the speed of 1500 rpm and the plasma was collected. The hippocampus of mice were homogenized in cold RIPA buffer containing enzyme inhibitor and then the solution was centrifuged at the speed of 12,000 rpm for 15 min. Finally, the supernates were collected and stored at − 80 ℃ for future use.
Enzyme-linked immunosorbent assay (ELISA) analysis
The concentration of TNF-α and IL-6 were tested at the 6th and 24th hours after surgery. After being homogenized and centrifuged, the concentration of protein collected from mouse hippocampus in each group was calculated by bicinchoninic acid (BCA) assay. The expression level of TNF-α and IL-6 in plasma and hippocampus were measured by TNF-α ELISA kits (R&D systems, USA) and IL-6 ELISA kits (R&D systems, USA) according to the operation instruction.
Western blot
The hippocampus of mice in each group was collected on the 3rd and 7th days after surgery for western blot. The protein samples were boiled in 5* loading buffer and separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). Then the protein on SDS-PAGE was transferred to poly-(vinylidene fluoride) (PVDF) membranes. PVDF membranes were blocked with 5% milk for 90 min at room temperature and incubated with primary antibodies at 4 ℃ overnight. PVDF membranes were washed with TBST buffer for three times and then incubated with secondary antibodies for 2 h at room temperature on the next day. The protein bands on PVDF membranes were visualized in chemiluminescence machine (Bio-Rad, USA) with using enhanced chemiluminescence kit (Solarbio, China).
The primary antibodies used in this study were showed as follows: anti-BDNF antibody (1:1000 dilution, Abacm, UK); anti-phospho-TrkB (Y705) antibody (1:1000 dilution, Abacm, UK); anti-TrkB antibody (1:1000 dilution, Abacm, UK); anti-PKCδ antibody (1:1000 dilution, Cell Signaling Technology, USA); anti-phospho-PKCδ antibody (1:1000 dilution, Cell Signaling Technology, USA); anti-NF-κB p65 antibody (1:1000 dilution, Cell Signaling Technology, USA); anti-iNOS antibody (1:1000 dilution, Cell Signaling Technology, USA); anti-Arg-1 antibody (1:1000 dilution, Cell Signaling Technology, USA); anti-Tubulin antibody (1:2000 dilution, Cell Signaling Technology, USA); anti-β-actin antibody (1:2000 dilution, Cell Signaling Technology, USA). The β-actin and tubulin were used as the reference proteins.
Immunofluorescent staining
To observe the activation of microglia, mice were sacrificed to harvest brain tissues at the 24th hour after surgery. Brain tissues were fixed in 4% paraformaldehyde at room temperature for three days and embedded in paraffin. After being repaired by EDTA, the sections were washed with PBS buffer and blocked with 5% bovine serum albumin at room temperature for 1 h. The sections were incubated with primary antibody overnight at 4 ℃ and then washed with PBS buffer for three times. The secondary antibodies were added to cover the sections completely at room temperature for 1.5 h in the dark. After being washed with PBS buffer, sections were stained with 4′,6-diamidino-2-phenylindole (DAPI) to locate nucleus for 5 min and immediately sealed with anti-fluorescence quenching agent. Stained images were obtained through the LSM700 confocal microscopy system (ZEISS, Germany).
The primary antibodies used in this study are shown as follows: anti-Iba-1 antibody (1:200 dilution, Abacm, UK); anti-iNOS antibody (1:200 dilution, Cell Signaling Technology, USA); anti-Arg-1 antibody (1:200 dilution, Cell Signaling Technology, USA).
Reverse transcription polymerase chain reaction (RT-PCR)
The total RNA of microglia with different intervention was collected with Trizol reagent (Sigma-Aldrich, USA) according to the operation instruction. The concentration and purity of total RNA were measured by using Microvolume UV–Vis Spectrophotometer (Thermo, Germany). Then, the reverse transcription of the total RNA was completed by using the HiScript II Q RT SuperMix for qPCR kit (Vazyme Biotech, China). The synthesized cDNA templates were used to do quantitative real-time PCR by using SYBR Green PCR reagents (Bio-Rad, USA). The ΔΔCt (the threshold cycle) values were calculated and the results were expressed as the ratio of the mRNA copies of BDNF gene to that of 18s RNA gene (reference gene). All data was presented in the fold change compared to the control group.
The primers involved in our study were showed as follows: 5′-TTACCTGGATGCCGCAAACAT-3′ (forward) and 5′-TGACCCACTCGCTAATACTGTC-3′ (reverse) for BDNF; 5′-CGGACAGGATTGACAGATT-3′ (forward) and 5′-CAAATCGCTCCAACCAACTAA-3′ (reverse) for 18s RNA.
Statistic analysis
Data was showed as mean ± standard deviation (n ≥ 3). The homogeneity variance was tested and all data was analyzed by one-way analysis of variance (ANOVA using the SPSS 22.0 software). Fisher’s Least Significant Difference (LSD) test was used for post-hoc analysis. All statistical graphs in this paper were performed by Graphpad prism 6.0 system. The p-value less than 0.05 was considered to be significant difference.
Results
C8-ceramide promoted BDNF expression in microglia and attenuated learning and memory impairment after surgical trauma
To investigate the effect of C8-ceramide on the BDNF expression derived from microglia in inflammatory tissues, primary microglia were treated with C8-ceramide before LPS exposure and then assessed the expression of BDNF mRNA by RT-PCR. As the results showed, the expression of BDNF mRNA in the LPS-treated group were significantly down-regulated than that in the control group (Fig. 1A). However, when primary microglia were pre-treated with C8-ceramide, the reduction of BDNF mRNA expression mRNA induced by LPS would be reversed and return to the same expression level with the control group. Furthermore, C8-ceramide used alone was able to significantly increased the expression of BDNF mRNA compared to those of mice from other groups. The above data strongly indicated that C8-ceramide could stimulate primary microglia to secrete BDNF and avoid damage to the nervous system caused by inflammation response.
The results of the RT-PCR and behavioral tests. (A) The expression of BDNF mRNA of primary microglia in each group. (B) The escape latency time of mice during the 5-days training. (C) The percentage of time spent in target quadrant of mice in each group. (D) The number of platform crossings of mice in each group. (E) The swimming path of mice recorded by a video camera. (F) The freezing time in contextual test. (G) The freezing time in tone test. (* p < 0.05, compared with the control group; # p < 0.05, compared with the surgery + C8-ceramide group; ^ p < 0.05, the C8 group compared with the control group, n = 43)
In order to explore whether C8-ceramide had effect on surgery-induced cognitive dysfunction, the MWM test and the Fear conditioning test were used to evaluated mice’ learning and memory ability. At first, all mice were subjected to train for five days on the 7th day after surgery. Compared to the mice in the control group, mice accepting surgery were significantly increased their latency to find the hidden platform on the 2nd and 3rd day in the training process (Fig. 1B). However, mice pre-treated with C8-ceramide have effectively restored the latency to find hidden platform and no significant differences could be found when compared to that in the control group. As the training time increasing, the escape latency period of the mice in each group gradually shortened, which indicated that the mice in all groups had ability to find the hidden platform and could further conduct the MWM test (Fig. 1B). On the 6th day in the spatial probe test, the swimming path recorded by computer showed that mice in the surgery group spent the least time in the target quadrant and cross the original hidden platform infrequently when compared to the mice in the other groups (Fig. 1C–E). Furthermore, when mice were pre-treated with C8-ceramide before the surgery, their learning and memory ability was same with the mice in the control group, which demonstrated that C8-ceramide had ability to protect the hippocampus of mice and prevent the process of POCD induced by surgery.
Similar to the MWM test, the context-related freezing time of mice in the surgery group was significantly decreased than those of other groups, and their cognitive impairment could be attenuated by the pre-treatment of C8-ceramide (Fig. 1F). It must be pointed out that there had no significant difference among all four groups in the tone-related fear test (Fig. 1G). It indicated that the surgery-induced impairment of hippocampus-dependent cognitive function could be restored by using C8-ceramide and surgery could not cause damage to the non-hippocampus-dependent cognitive function.
C8-ceramide reversed the down-regulation of BDNF expression induced by surgery
In the previous study, we verified that C8-ceramide enhanced the secretion of BDNF derived from primary microglia, which could reverse the effect of LPS in vitro. To explore whether C8-ceramide have the same effect on microglia in vivo and the potential molecular mechanism in these process, the hippocampus in mice from all groups was collected on the 3rd and 7th day after surgery. The results of western blot showed that surgical procedures had negative effect on the expression of BDNF in the hippocampus of mice on the 3rd day, but this inhibition effect would be reversed when C8-ceramide was used in advance (Fig. 2A, B). However, using C8-ceramide alone did not improve the expression of BDNF in the hippocampus of mice when compared to the control group.
The expression of the BNDF/TrkB signaling pathway in the hippocampus of mice in all groups at the 3rd day after surgery. (A, B) The expression of BDNF in the hippocampus of mice in all groups at the 3rd day after surgery. (C–E) The expression of total TrkB and p-TrkB in the hippocampus of mice in all groups at the 3rd day after surgery. (* p < 0.05, compared with the control group; # p < 0.05, compared with the surgery + C8-ceramide group, n = 3)
Due to the fact that the BNDF/TrkB signaling pathway has been considered as the classical signaling pathway to regulate the secretion of BDNF, we assumed that C8-ceramide could up-regulate the expression of BDNF via activating the BNDF/TrkB signaling pathway. In our study, the total expression of TrkB did not have any significant difference in all four groups, but the p-TrkB expression level in the surgery group was the lowest compared to those of other groups (Fig. 2C–E). Consistent with the trend of BDNF expression, using C8-ceramide alone did not significantly increase the expression of p-TrkB, which indicated that C8-ceramide, as a neurotrophic substance, may only promote the secretion of BDNF under stress condition to protect the nervous system. The above data has demonstrated that C8-ceramide promoted the secretion of BDNF via up-regulating the BNDF/TrkB signaling pathway in surgery-induced cognitive dysfunction.
However, we also found that there had no significant difference in both the expression of BDNF and p-TrkB in the hippocampus of mice from all the groups on the 7th day after surgery (Fig. 3A–D). It suggested that C8-ceramide could treat surgery-induced cognitive impairment at the early stage after surgery and shorten the duration of POCD to enhance the life quality of patients.
The expression of the BNDF/TrkB signaling pathway in the hippocampus of mice in all groups at the 7th day after surgery. (A, B) The expression of BDNF in the hippocampus of mice in all groups at the 7th day after surgery. (C, D) The expression of p-TrkB in the hippocampus of mice in all groups at the 3rd day after surgery (n = 3)
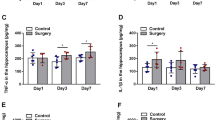
C8-ceramide down-regulated the expression of surgery-induced pro-inflammatory factors in the hippocampus of mice
Although the entire surgical process in our study was in a sterile environment, other predispositions could cause tissue inflammation, including surgical trauma. To evaluate the relationship between POCD and tissue inflammation, as well as the role of C8-ceramide on the inhibition of tissue inflammation, the blood serum and hippocampus of mice in all groups were harvested to test the concentration of TNF-α and IL-6. Compared with the control group, the expression of IL-6 and TNF-α were significantly increased in the hippocampus of mice on the 6th hour after surgery (Fig. 4A, B). Meanwhile, the similar expression change of these two pro-inflammatory factors was also found in the collected serum in the surgery group (Fig. 4C, D). However, when mice were pre-treated with C8-ceramide, the concentration of TNF-α and IL-6 in the hippocampus and serum of mice was close to that in the control group, which suggested that the increasing expression of BDNF induced by C8-ceramide inhibited the surgery-induced inflammatory response in the hippocampus of mice. Furthermore, it must be pointed out that the expression of IL-6 and TNF-α in the hippocampus of mice from the surgery group was still the highest in all four groups at the 24th hour after surgery and it meant that even for a long period of time after surgery, the hippocampus of mice in the surgery group was still in the inflammatory state and it was clearly detrimental to the development of POCD (Fig. 4E, F).
The inflammation level in the hippocampus and serum of mice in all groups at the 6th and 24th hour after surgery. (A, B) The expression of inflammatory factors in the hippocampus of mice in all groups at the 6th hour after surgery. (C, D) The expression of inflammatory factors in the serum of mice in all groups at the 6th hour after surgery. (E, F) The expression of inflammatory factors in the hippocampus of mice in all groups at the 24th hour after surgery. (* p < 0.05, compared with the control group; # p < 0.05, compared with the surgery + C8-ceramide group; ** p < 0.01, compared with the control group; ## p < 0.01, compared the surgery + C8-ceramide group, n = 3)
C8-ceramide inhibited the PKCδ/NF-κB signaling pathway
NF-κB is an important transcription factor to regulate the expression of various inflammatory cytokines. The activation of NF-κB requires the phosphorylation process and it can be stimulated by kinds of proteins and transcription factors, including PKCδ. As the previous studies showed, the PKCδ/NF-κB signaling pathway was important in inflammatory response. Therefore, to test whether the PKCδ/NF-κB signaling pathway was activated in the hippocampus of mice to induce the inflammatory response, we detected the expression of key proteins in this signaling pathway.
In our study, we found that the total expression of PKCδ in the hippocampus of mice from all four groups was close and there were no significant difference among them, but the phosphorylation of PKCδ was significantly enhanced in the hippocampus of mice from the surgery group (Fig. 5A–C). As the phosphorylation level of PKCδ increased, we also found that the phosphorylation of NF-κB (p65) also enhanced in the hippocampus of mice from the surgery group, which indicated that the PKCδ/NF-κB signaling pathway was activated in the hippocampus (Fig. 5D, E). Interestingly, when mice were pre-treated with C8-ceramide before surgery, the phosphorylation process of PKCδ and NF-κB in their hippocampus was inhibited. The above data and the previous Elisa tests on TNF-α and IL-6 confirmed with each other, which indicated that the cognitive dysfunction caused by surgery was related to the activation of the PKCδ/NF-κB signaling pathway and the enhancement of BDNF expression derived from microglia had ability to suppress the surgery-induced inflammation in the hippocampus of mice.
C8-ceramide inhibited the the activation of the PKCδ/NF-κB pathway in the hippocampus of mice. (A–C) The expression of total PKCδ and p-PKCδ in the hippocampus of mice in all groups. (D, E) The expression of p-p65 in the hippocampus of mice in all groups. (* p < 0.05, compared with the control group; # p < 0.05, compared with the surgery + C8-ceramide group, n = 3)
C8-ceramide had ability to promote the activation of microglia to M2 phenotype
The process of tissue inflammation is determined by the polarization of macrophages. As macrophages in the CNS, the polarization of microglia in the hippocampus plays a decisive role on the process of POCD. To test the polarization of microglia in the hippocampus of mice, we used immunofluorescence staining to detect the expression of specific antigen of microglia. iNOS and Arg-1 were recognized as the specific markers to M1/M2 microglia respectively. The results showed that there were more M1 microglia (iNOS positive cells) in the hippocampus of mice in the surgery group than those of other groups (Fig. 6A, B). On the contrast, M2 microglia (Arg-1 positive cells) mainly existed in the hippocampus of mice from the C8-ceramide group (Fig. 6C, D). The density of M1 and M2 phenotype of microglia regulate the process of inflammatory response in the hippocampus of mice and it was also consistent with the previous study findings.
The immunofluorescence of the polarization of microglia in the hippocampus of mice in all groups. (A, B) The density of M1 microglia in the hippocampus of mice in all groups. (C, D) The density of M2 microglia in the hippocampus of mice in all groups. Scale bars were 200 μm. (**/^^ p < 0.01, compared with the control group; ## p < 0.01, compared with the surgery + C8-ceramide group, n = 3)
Furthermore, we found that the expression of iNOS and Arg-1 in the hippocampus of mice were different in all four groups tested by western blot. In the surgery group, the expression of iNOS was significantly higher than that of other groups (Fig. 7A, B). Once mice were pre-treated with C8-ceramide, the expression of Arg-1 would be enhanced and showed that the increasing number of M2 microglia (Fig. 7C, D). The above data suggested that C8-ceramide had ability to regulate the polarization of microglia to surppress the inflammatory response.
The expression of specific protein to identify the polarization of microglia. (A, B) The expression of iNOS in the microglia from the hippocampus. (C, D) The expression of Arg-1 in the microglia from the hippocampus. (* p < 0.05, compared with the control group; # p < 0.05, compared with the surgery + C8-ceramide group; ^ p < 0.05, the C8 group compared with the control group, n = 3)
Discussion
In our study, surgery process could cause the cognitive dysfunction and down-regulate the expression of BDNF in the hippocampus of mice. Meanwhile, we also found that the hippocampus of mice in the surgery group was in a high inflammatory state, which was supported by the enhancement concentration of pro-inflammatory cytokines and the density of M1 microglia in the mice’ hippocampus. The application of C8-ceramide reversed the inflammatory response in the hippocampus of mice and stimulated the polarization of microglia towards M2 phenotype in our study. LPS is a kind of gram-negative cell wall component to induce the strong inflammatory response through TLR4 signaling pathway and significantly decrease the expression of BDNF mRNA in primary microglia in vitro (Rahimifard et al. 2017). LPS is often used in vitro to simulate the inflammatory response in vivo. Our research findings have provided strong evidences to show that surgery-induced cognitive dysfunction was the outcome of uncontrolled inflammation response and the decreasing expression of BDNF in the hippocampus of mice.
Many previous studies have demonstrated that surgery-induced neuro-inflammation was an independent risk factor in the development of POCD (Su et al. 2020; Huang et al. 2018). In our study, TNF-α and IL-6 were used as the representative cytokines to assess the inflammatory response. The concentration of TNF-α and IL-6 were significantly increased in the serum and hippocampus of mice in the surgery group at the 6th and 24th hours after surgery. We measured the concentration of pro-inflammatory factors in serum and hippocampus at the 6th hours post surgery, while only pro-inflammatory factor concentration in hippocampus was measured at the 24th hours post surgery. The reason was that we thought that surgical procedures tor mice were limited to brain tissues. Therefore, compared to other methods, pro-inflammatory factors in the hippocampus had a greater impact on cognitive function, and pro-inflammatory factors in the serum could be cleared in a short period of time or had limited impact on the hippocampus due to the presence of the circulatory system.
The NF-κB signaling pathway is considered as a classical signaling pathway to regulate the inflammation and this signaling pathway is conserved in all mammals (Lin et al. 2020). PKCδ is a multifunctional enzyme to participate in the activation of various signaling pathways. The phosphorylation of PKCδ can stimulate the activation of NF-κB signaling pathway and initiate the downstream inflammatory cascade reactions (Hsu et al. 2001; Li et al. 2019). After being phosphorylated, the p65 would release NF-κB and transfer it into the nucleus (Kawai and Akira 2007). Intranuclear NF-κB would bind to the promoter of the target DNA sequence and enhance the synthesis of various cytokines, including pro-inflammatory factors (Kawai and Akira 2007; Lawrence 2009). In our study, we found a significant increase in the phosphorylation of PKCδ and NF-κB in the hippocampus of mice from the surgery group, which proved the activation of the PKCδ/NF-κB signaling pathway. However, C8-ceramide had ability to inhibit the PKCδ/NF-κB signaling pathway and suppress the inflammatory response induced by surgery. It has showed that C8-ceramide had a bright future in the field of neuro-inflammation treatment.
The bridge of BDNF and inflammatory pathway determines the polarization of microglia. After the exposure of common carotid artery surgery, the immune cells-microglia crosstalk in the CNS induce the polarization of M1 microglia, and the releasing of reactive oxygen species and pro-inflammatory factors, which decrease the synthesis and secretion of BDNF in the hippocampus of mice. We detected the phosphorylation of proteins related to BDNF/TrkB signaling pathway and PKCδ/NF-κB signaling pathway after surgery with/without C8-ceramide treatment. The basis of our study was that microglia were immune cells in the CNS and they could detect changes in the microenvironment in the brain and respond quickly. As we all known, excessive neuroinflammation caused by surgery would damage normal neurons and resulted in the decreasing expression of BDNF and POCD.
Neuro-inflammation only exists in the early stage after surgery, but it may has long-term effect on learning and memory function, due to the fact that excessive inflammatory factors secreted by microglia can damage neurons and the plasticity of synapses and the process of neural regeneration is very slow (Saxena et al. 2019; Fan et al. 2016). Therefore, it will always take a long time to restore damaged neurological function and some patients may even have a lifelong illness. It must be pointed out that the chronic neuro-inflammation significantly inhibit the expression of BDNF which is harmful to the neural regeneration and accelerate the process of POCD (Golia et al. 2019; Lima Giacobbo et al. 2019). However, the BDNF/TrkB signaling pathway, as one of the main molecular mechanisms in the learning and memory process, is important to the synaptic plasticity and the formation of long-term potentiation (LTP) (Parkhurst et al. 2013). The increasing expression of BDNF is beneficial to the anti-inflammation and neural regeneration (Yin et al. 2020). Parkhurst et al. have proved that learning and memory impairment would occur once microglia were completely cleared from the brain (Parkhurst et al. 2013), and it meant that BDNF derived from microglia was important to the cognitive function. Our study has also proved that the increasing expression of BDNF secreted by microglia had ability to improve surgery-induced cognitive impairment. The previous studies have showed that C8-ceramide increased the expression of BDNF in microglia at the dose of 25 μM (Nakajima et al. 2002), which was the same with our study. However, in our study, there was no significant different in the expression of BDNF between the C8-ceramide group and the control group. The possible reason may be that the expression of BDNF was strictly controlled and depended on the microenvironment in the CNS. It is worthy to further study.
Microglia are the resident immune cells in the CNS and can polarize toward pro-inflammatory phenotype (M1 microglia) and anti-inflammatory phenotype (M2 microglia) under specific circumstances (Tang and Le 2016). According to the current theory, the microglia just polarize towards M2 phenotype in a very short period of time and M1 phenotype of microglia are common in plenty of animal neural disease models (Yin et al. 2020). The immunofluorescence staining in our study showed that the microglia in the hippocampus of mice from the surgery group were mainly M1 microglia, while M2 microglia existed in the hippocampus of mice from the C8-ceramide group. Therefore, the application of C8-ceramide may be an effective method to inhibit the neuroinflammation in the CNS and it is an effective method to treat POCD.
Our research found that surgery process significantly down-regulated the BDNF expression and reduced the phosphorylation of TrkB on the 3rd day after surgery. Interestingly, on the 7th day after surgery, the expression of BNDF in the hippocampus of mice was not significantly different among all four groups. We speculated that it might be temporarily up-regulated to the expression of BDNF as compensatory, due to the fact that the behavioral tests on the 8th to 14th days after operation showed that the cognitive function of the mice indeed decreased. Our previous study also found that the expression of BDNF was still significantly lower than that of healthy mice on the 5th day after surgery, and it indicated that the recovery of BDNF secretion started on the 7th day after surgery rather than at the early stage and the recovery of cognitive function was even later. It must be pointed out that we did not measure the concentration of BDNF in the hippocampus of mice on the 14th day after surgery, which provided guidance for our future research. The using of C8-ceramide significantly inhibited or shortened the duration of inflammation in the hippocampus of mice after surgery, which played an important role on the prevention and treatment of POCD.
Conclusion
In our study, we have proved that surgery could cause neuroinflammation in the CNS to induce POCD and the application of C8-ceramide could enhance the expression of BDNF to suppress neuroinflammation via the regulation of PKCδ/NF-κB signaling pathway for the first time. It has revealed a potential molecular mechanism of surgery-induced POCD and proves a promising treatment method. According to our study, we strongly believe that C8-ceramide will become a star drug to treat surgery-induced POCD and reduce the risk of suffering from POCD in a large population.
Data Availability
The data set analysed in this study are available from the corresponding author on reasonable request.
Abbreviations
- BCA:
-
Bicinchoninic acid
- BDNF:
-
Brain-derived neurotrophic factor
- CNS:
-
Central nervous system
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- ELISA:
-
Enzyme-linked immunosorbent assay
- FBS:
-
Fetal bovine serum
- IL-1:
-
Interleukin-1
- LPS:
-
Lipopolysaccharide
- LSD:
-
Least significant difference
- LTP:
-
Long-term potentiation
- MAPK:
-
Mitogen-activated protein kinase
- MWM:
-
Morris Water Maze
- PKC:
-
Protein kinase C
- POCD:
-
Postoperative cognitive dysfunction
- PS:
-
Penicillin–streptomycin
- PVDF:
-
Poly-(vinylidene fluoride)
- RT-PCR:
-
Reverse transcription polymerase chain reaction
- SDS-PAGE:
-
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
- TNF-ɑ:
-
Tumor necrosis factor-ɑ
References
Alam A, Hana Z, Jin Z, Suen KC, Ma D (2018) Surgery, neuroinflammation and cognitive impairment. EBioMedicine 37:547–556. https://doi.org/10.1016/j.ebiom.2018.10.021
Askew K, Li K, Olmos-Alonso A, Garcia-Moreno F, Liang Y, Richardson P, Tipton T, Chapman MA, Riecken K, Beccari S, Sierra A, Molnár Z, Cragg MS, Garaschuk O, Perry VH, Gomez-Nicola D (2017) Coupled proliferation and apoptosis maintain the rapid turnover of microglia in the adult brain. Cell Rep 18(2):391–405. https://doi.org/10.1016/j.celrep.2016.12.041
Balakrishnan P, Song CK, Jahn A, Cho HJ (2016) Ceramide and N, N, N-Trimethylphytosphingosine-Iodide (TMP-I)-based lipid nanoparticles for cancer therapy. Pharm Res 33(1):206–216. https://doi.org/10.1007/s11095-015-1780-5
Calabrese F, Rossetti AC, Racagni G, Gass P, Riva MA, Molteni R (2014) Brain-derived neurotrophic factor: a bridge between inflammation and neuroplasticity. Front Cell Neurosci 8:430. https://doi.org/10.3389/fncel.2014.00430
Chen Y, Fang ZM, Yi X, Wei X, Jiang DS (2023) The interaction between ferroptosis and inflammatory signaling pathways. Cell Death Dis 14(3):205. https://doi.org/10.1038/s41419-023-05716-0
Chhor V, Le Charpentier T, Lebon S, Oré MV, Celador IL, Josserand J, Degos V, Jacotot E, Hagberg H, Sävman K, Mallard C, Gressens P, Fleiss B (2013) Characterization of phenotype markers and neuronotoxic potential of polarised primary microglia in vitro. Brain Behav Immun 32:70–85. https://doi.org/10.1016/j.bbi.2013.02.005
Chiba N, Masuda A, Yoshikai Y, Matsuguchi T (2007) Ceramide inhibits LPS-induced production of IL-5, IL-10, and IL-13 from mast cells. J Cell Physiol 213(1):126–136. https://doi.org/10.1002/jcp.21101
Dan HC, Cooper MJ, Cogswell PC, Duncan JA, Ting JP, Baldwin AS (2008) Akt-dependent regulation of NF-{kappa}B is controlled by mTOR and Raptor in association with IKK. Genes Dev 22(11):1490–1500. https://doi.org/10.1101/gad.1662308
Degos V, Vacas S, Han Z, van Rooijen N, Gressens P, Su H, Young WL, Maze M (2013) Depletion of bone marrow-derived macrophages perturbs the innate immune response to surgery and reduces postoperative memory dysfunction. Anesthesiology 118(3):527–536. https://doi.org/10.1097/ALN.0b013e3182834d94
Fan D, Li J, Zheng B, Hua L, Zuo Z (2016) Enriched environment attenuates surgery-induced impairment of learning, memory, and neurogenesis possibly by preserving BDNF expression. Mol Neurobiol 53(1):344–354. https://doi.org/10.1007/s12035-014-9013-1
Fields RD, Burnstock G (2006) Purinergic signalling in neuron-glia interactions. Nat Rev Neurosci 7(6):423–436. https://doi.org/10.1038/nrn1928
Fondeur J, Escudero Mendez L, Srinivasan M, Hamouda RK, Ambedkar B, Arzoun H, Sahib I, Mohammed L (2022) Dexmedetomidine in prevention of postoperative delirium: a systematic review. Cureus 14(6):e25639. https://doi.org/10.7759/cureus.25639
Ge Y, Li Q, Nie Y, Gao J, Luo K, Fang X, Wang C (2019) Dexmedetomidine improves cognition after carotid endarterectomy by inhibiting cerebral inflammation and enhancing brain-derived neurotrophic factor expression. J Int Med Res 47(6):2471–2482. https://doi.org/10.1177/0300060519843738
Goehler LE, Gaykema RP, Hansen MK, Anderson K, Maier SF, Watkins LR (2000) Vagal immune-to-brain communication: a visceral chemosensory pathway. Autonom Neurosci Basic Clin 85(1–3):49–59. https://doi.org/10.1016/S1566-0702(00)00219-8
Golia MT, Poggini S, Alboni S, Garofalo S, Ciano Albanese N, Viglione A, Ajmone-Cat MA, St-Pierre A, Brunello N, Limatola C, Branchi I, Maggi L (2019) Interplay between inflammation and neural plasticity: both immune activation and suppression impair LTP and BDNF expression. Brain Behav Immun 81:484–494. https://doi.org/10.1016/j.bbi.2019.07.003
Gómez-Casati ME, Murtie JC, Rio C, Stankovic K, Liberman MC, Corfas G (2010) Nonneuronal cells regulate synapse formation in the vestibular sensory epithelium via erbB-dependent BDNF expression. Proc Natl Acad Sci USA 107(39):17005–17010. https://doi.org/10.1073/pnas.1008938107
Hamanaka S, Hara M, Nishio H, Otsuka F, Suzuki A, Uchida Y (2002) Human epidermal glucosylceramides are major precursors of stratum corneum ceramides. J Invest Dermatol 119(2):416–423. https://doi.org/10.1046/j.1523-1747.2002.01836.x
Hoesel B, Schmid JA (2013) The complexity of NF-κB signaling in inflammation and cancer. Mol Cancer 12:86. https://doi.org/10.1186/1476-4598-12-86
Hovens IB, van Leeuwen BL, Nyakas C, Heineman E, van der Zee EA, Schoemaker RG (2015) Postoperative cognitive dysfunction and microglial activation in associated brain regions in old rats. Neurobiol Learn Mem 118:74–79. https://doi.org/10.1016/j.nlm.2014.11.009
Hsu YW, Chi KH, Huang WC, Lin WW (2001) Ceramide inhibits lipopolysaccharide-mediated nitric oxide synthase and cyclooxygenase-2 induction in macrophages: effects on protein kinases and transcription factors. J Immunol (Baltimore, MD, 1950) 166(9):5388–5397. https://doi.org/10.4049/jimmunol.166.9.5388
Huang C, Irwin MG, Wong GTC, Chang RCC (2018) Evidence of the impact of systemic inflammation on neuroinflammation from a non-bacterial endotoxin animal model. J Neuroinflammation 15(1):147. https://doi.org/10.1186/s12974-018-1163-z
Jung JS, Shin KO, Lee YM, Shin JA, Park EM, Jeong J, Kim DH, Choi JW, Kim HS (2013) Anti-inflammatory mechanism of exogenous C2 ceramide in lipopolysaccharide-stimulated microglia. Biochem Biophys Acta 1831(6):1016–1026. https://doi.org/10.1016/j.bbalip.2013.01.020
Kawai T, Akira S (2007) Signaling to NF-kappaB by Toll-like receptors. Trends Mol Med 13(11):460–469. https://doi.org/10.1016/j.molmed.2007.09.002
Kawano T, Yamanaka D, Aoyama B, Tateiwa H, Shigematsu-Locatelli M, Nishigaki A, Iwata H, Locatelli FM, Yokoyama M (2018) Involvement of acute neuroinflammation in postoperative delirium-like cognitive deficits in rats. J Anesth 32(4):506–517. https://doi.org/10.1007/s00540-018-2504-x
Krenk L, Rasmussen LS, Kehlet H (2010) New insights into the pathophysiology of postoperative cognitive dysfunction. Acta Anaesthesiol Scand 54(8):951–956. https://doi.org/10.1111/j.1399-6576.2010.02268.x
Lawrence T (2009) The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol 1(6):a001651. https://doi.org/10.1101/cshperspect.a001651
Lee MJ, Kim J, Kim MY, Bae YS, Ryu SH, Lee TG, Kim JH (2010) Proteomic analysis of tumor necrosis factor-alpha-induced secretome of human adipose tissue-derived mesenchymal stem cells. J Proteome Res 9(4):1754–1762. https://doi.org/10.1021/pr900898n
Li X, Zhao Z, Kuang P, Shi X, Wang Z, Guo L (2019) Regulation of lipid metabolism in diabetic rats by Arctium lappa L. polysaccharide through the PKC/NF-κB pathway. Int J Biol Macromol 136:115–122. https://doi.org/10.1016/j.ijbiomac.2019.06.057
Lima Giacobbo B, Doorduin J, Klein HC, Dierckx RAJO, Bromberg E, de Vries EFJ (2019) Brain-derived neurotrophic factor in brain disorders: focus on neuroinflammation. Mol Neurobiol 56(5):3295–3312. https://doi.org/10.1007/s12035-018-1283-6
Lin X, Chen Y, Zhang P, Chen G, Zhou Y, Yu X (2020) The potential mechanism of postoperative cognitive dysfunction in older people. Exp Gerontol 130:110791. https://doi.org/10.1016/j.exger.2019.110791
Moller JT, Cluitmans P, Rasmussen LS, Houx P, Rasmussen H, Canet J, Rabbitt P, Jolles J, Larsen K, Hanning CD, Langeron O, Johnson T, Lauven PM, Kristensen PA, Biedler A, van Beem H, Fraidakis O, Silverstein JH, Beneken JE, Gravenstein JS (1998) Long-term postoperative cognitive dysfunction in the elderly ISPOCD1 study. ISPOCD investigators. International Study of Post-Operative Cognitive Dysfunction. Lancet (London, England) 351(9106):857–861. https://doi.org/10.1016/s0140-6736(97)07382-0
Monk TG, Price CC (2011) Postoperative cognitive disorders. Curr Opin Crit Care 17(4):376–381. https://doi.org/10.1097/MCC.0b013e328348bece
Moscat J, Diaz-Meco MT, Rennert P (2003) NF-kappaB activation by protein kinase C isoforms and B-cell function. EMBO Rep 4(1):31–36. https://doi.org/10.1038/sj.embor.embor704
Nakajima K, Tohyama Y, Kohsaka S, Kurihara T (2002) Ceramide activates microglia to enhance the production/secretion of brain-derived neurotrophic factor (BDNF) without induction of deleterious factors in vitro. J Neurochem 80(4):697–705. https://doi.org/10.1046/j.0022-3042.2001.00752.x
Parkhurst CN, Yang G, Ninan I, Savas JN, Yates JR 3rd, Lafaille JJ, Hempstead BL, Littman DR, Gan WB (2013) Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor. Cell 155(7):1596–1609. https://doi.org/10.1016/j.cell.2013.11.030
Ponnapakkam T, Saulsberry T, Hill T, Hill-Odom M, Goyal N, Anbalagan M, Liu J, Foroozesh M (2018) Inhibition of breast tumor growth in mice after treatment with ceramide analog 315. Anticancer Drugs 29(9):898–903. https://doi.org/10.1097/CAD.0000000000000675
Rahimifard M, Maqbool F, Moeini-Nodeh S, Niaz K, Abdollahi M, Braidy N, Nabavi SM, Nabavi SF (2017) Targeting the TLR4 signaling pathway by polyphenols: a novel therapeutic strategy for neuroinflammation. Ageing Res Rev 36:11–19. https://doi.org/10.1016/j.arr.2017.02.004
Saxena S, Lai IK, Li R, Maze M (2019) Neuroinflammation is a putative target for the prevention and treatment of perioperative neurocognitive disorders. Br Med Bull 130(1):125–135. https://doi.org/10.1093/bmb/ldz010
Singh AT, Dharmarajan A, Aye IL, Keelan JA (2012) Ceramide biosynthesis and metabolism in trophoblast syncytialization. Mol Cell Endocrinol 362(1–2):48–59. https://doi.org/10.1016/j.mce.2012.05.009
Steinmetz J, Christensen KB, Lund T, Lohse N, Rasmussen LS, ISPOCD Group (2009) Long-term consequences of postoperative cognitive dysfunction. Anesthesiology 110(3):548–555.https://doi.org/10.1097/ALN.0b013e318195b569
Su Y, Pu Y, Zhao Z, Yang X (2020) Influence of combined epidural anesthesia on cognitive function, inflammation and stress response in elderly liver cancer patients undergoing surgery. Oncol Lett 19(4):2733–2738. https://doi.org/10.3892/ol.2020.11395
Takasaki I, Takarada S, Tatsumi S, Azegami A, Yasuda M, Fukuchi M, Tabuchi A, Kondo T, Tabuchi Y, Tsuda M (2008) Extracellular adenosine 5′-triphosphate elicits the expression of brain-derived neurotrophic factor exon IV mRNA in rat astrocytes. Glia 56(13):1369–1379. https://doi.org/10.1002/glia.20704
Tang Y, Le W (2016) Differential roles of M1 and M2 microglia in neurodegenerative diseases. Mol Neurobiol 53(2):1181–1194. https://doi.org/10.1007/s12035-014-9070-5
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1(2):848–858. https://doi.org/10.1038/nprot.2006.116
Wang YH, Guo YC, Wang DR, Liu JY, Pan J (2019) Adipose stem cell-based clinical strategy for neural regeneration: a review of current opinion. Stem Cells Int 2019:8502370. https://doi.org/10.1155/2019/8502370
Wang Y, Wu D, Li D, Zhou X, Fan D, Pan J (2023) The role of PERK-eIF2α-ATF4-CHOP pathway in sevoflurane induced neuroapoptosis and cognitive dysfunction in aged mice. Cell Signal 110:110841. https://doi.org/10.1016/j.cellsig.2023.110841
Yang X, Huang X, Li M, Jiang Y, Zhang H (2022) Identification of individuals at risk for postoperative cognitive dysfunction (POCD). Therapeut Adv Neurol Disord 15:17562864221114356. https://doi.org/10.1177/17562864221114356
Yin R, Zhao S, Qiu C (2020) Brain-derived neurotrophic factor fused with a collagen-binding domain inhibits neuroinflammation and promotes neurological recovery of traumatic brain injury mice via TrkB signalling. J Pharm Pharmacol 72(4):539–550. https://doi.org/10.1111/jphp.13233
Zhang J, Tan H, Jiang W, Zuo Z (2014) Amantadine alleviates postoperative cognitive dysfunction possibly by increasing glial cell line-derived neurotrophic factor in rats. Anesthesiology 121(4):773–785. https://doi.org/10.1097/ALN.0000000000000352
Acknowledgements
We acknowledge the colleagues of West China Hospital of Stomatology, Sichuan University, Chengdu, PR China and also acknowledge the colleagues of Sichuan Provincial People’s Hospital, Chengdu, PR China.
Funding
This study was supported by grants from National Natural Science Foundation of China (81500933) and Sichuan Science and Technology Program (2022YFS0302, 2021YFS0375).
Author information
Authors and Affiliations
Contributions
Guangqian Li: Conceptualization, Methodology, Investigation, Formal analysis, Writing – original draft. Yuhao Wang: Conceptualization, Methodology, Formal analysis, Writing – original draft. Lei Qian: Investigation, Visualization. Pan Gu: Visualization, Formal analysis, Investigation. Jian Pan: Conceptualization, Supervision, Project administration, Writing - review & editing. Dan Fan: Conceptualization, Supervision, Project administration, Writing - review & editing, Funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Ethical approval was obtained from the Sichuan Provincial People’s Hospital Research Ethics Committee.
Additional information
Communicated by Sreedharan Sajikumar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, G., Wang, Y., Qian, L. et al. C8-ceramide modulates microglia BDNF expression to alleviate postoperative cognition dysfunction via PKCδ/NF-κB signaling pathway. Exp Brain Res 242, 1543–1559 (2024). https://doi.org/10.1007/s00221-024-06847-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-024-06847-2