Abstract
As humans accelerate global environmental change, extreme climatic events are increasing in frequency, duration, and intensity. In marine environments, extreme events of particular concern are marine heatwaves (MHWs). Previous reviews synthesising subsets of field-based research examining the ecological effects of these short-term warming events have found they could drive important changes to ecosystems. Here, we reviewed the traits of published literature that has used field, laboratory, and modelling approaches to examine the biotic effects of MHWs, and highlight some of the key findings. Since its first use in this context in 2013, the term “marine heatwave” has generally been used in field-based literature primarily documenting changes in abundance, biodiversity, species distributions, and mortality, primarily in fish and molluscs. Research is increasingly also conducted in laboratory settings, with these investigations focussing on changes in mechanistic processes such as growth and biochemical responses, often in smaller or less motile organisms including seagrasses, algae, molluscs, and crustaceans. Given their different applications, these complementary approaches will provide a more complete understanding when used to consider the same taxonomic groups. Moreover, those manipulating MHWs in laboratory experiments could benefit from the consideration of a range of MHW traits (intensity, duration, and their combination), for species in both isolation and combination, and in the presence of additional abiotic stressors. As such, where future research into MHWs use varied approaches and treatment settings, they will contribute to a more holistic understanding of the biological and ecological effects of future short-term warming events in our ocean.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change is one of the greatest threats to global biodiversity and ecosystem services provided by the natural world. In the ocean, short-term elevated temperatures—and their consequences—have been gaining increased attention (Oliver et al. 2018, 2021). These marine heatwaves (MHWs) can be defined as prolonged, discrete, anomalously warm events and described by their duration, intensity, rate of evolution, and spatial extent (Hobday et al. 2016). Although extreme climatic events occur naturally, the increased frequency and duration of MHWs over the past century (Oliver et al. 2018) are suggested to reflect large influences of anthropogenic activities (Laufkötter et al. 2020; Jacox et al. 2022). Further increases in the frequency and intensity of MHWs are forecast to occur throughout the twenty-first century (Oliver et al. 2019). Such increases will lead to biological and ecological changes including large-scale mortality, species distribution shifts, and genetic tropicalisation of temperate ecosystems (Smale & Wernberg 2013; Wernberg et al. 2016; Coleman et al. 2020), as well as drive economic implications such as the disruption and closure of fisheries (e.g., Caputi et al. 2019; Barbeaux et al. 2020; Dudley et al. 2021; Smith et al. 2021). It is worth noting, however, that the biological responses observed—which shape such ecological and economic effects—can be location-, species-, and process-specific (Vinagre et al. 2016; Bernal et al. 2020).
With recent and forecasted MHW events gaining increased attention, the body of literature investigating their abiotic and biotic aspects has rapidly developed. A number of key investigations have defined what a MHW is, focussing on traits such as duration and intensity (Hobday et al. 2016, 2018; Oliver et al. 2018, 2021; Holbrook et al. 2020). Now that our understanding of the abiotic aspects of MHWs has improved, research regarding the biological and ecological impacts of MHWs is expanding. That is, while we have a rich history of understanding the effects of temperature on physiology, biology, and occasionally ecology from neighbouring research areas (e.g., acute stress events in ecophysiology and ecotoxicology (Monteiro et al. 2021); degree heating days in coral reefs (e.g., Kayanne 2017; McClanahan and Azali 2021); SSTa in marine biogeography (e.g., Deser et al. 2010)), there is an emerging body of research specifically exploring the biological and ecological effects of these recently defined MHWs. Where the growing body of research on MHWs and their ecological implications has allowed, some notable research has brought together sub-sets of the published literature. For example, a recent meta-analysis focussed on the field-based effects of eight well-described periods of extreme warming and identified that the effects of these MHWs were spatially variable, although they were consistently deleterious across biological processes and taxa (Smale et al. 2019). Similarly, a synthesis of in situ effects of MHWs on algal species found that native, canopy-forming species were adversely affected whereas turf-forming species, especially those classed as invasive, benefitted from such events (Straub et al. 2019).
Owing to the recent definition of the term “marine heatwave”, and the focus of previous synthesis papers on the field-based effects of key MHWs (e.g., Smale et al. 2019; Straub et al. 2019), there is yet to be a comprehensive synthesis of the field-, laboratory-, and modelling-based literature on biotic effects of these events. Moreover, although laboratory-based research represents an expanding proportion of the literature, it has been overlooked in previous syntheses, which have focussed on the large-scale impacts observed in natural systems (Straub et al. 2019; Smith et al. 2023). Such controlled laboratory experiments are typically used to allow a greater disentangling of the effects and interactions between drivers on mechanistic processes (De Boeck et al. 2015; Cooke et al. 2017). Thus, synthesizing the literature surrounding these investigations will help gain a fuller picture of the current state of MHW research, and indicate research areas that need to be further explored.
Here, we have systematically reviewed the current literature regarding the biological and ecological impacts of MHWs for papers using field-, laboratory-, and modelling- methods to quantify: (i) publication patterns—when and where research has taken place; (ii) biotic context—the species and responses investigated; and (iii) details regarding experimental laboratory work including the traits of MHWs manipulated (intensity, duration, frequency), and biotic and abiotic traits considered (number of species, presence of other stressors). We also use a narrative synthesis framework to highlight some of the key findings in terms of biological and ecological impacts. Considering all of the literature together allows for a more comprehensive understanding of the research considering the biological and ecological impacts of MHWs, and the areas that could be further explored in the future.
Methods
We searched the online databases Web of Knowledge, Science Direct, and Scopus for literature associated with marine heatwaves (MHWs) using the search string: (“marine” OR “ocean*) AND (“heatwave” OR “heat wave” OR “heat-wave”). The search was conducted on the 11th of March 2021 and literature published until the end of 2020 was selected. The search returned 4819 articles. The articles were screened for duplicates and relevant content, removing any that were unrelated to MHWs. After initial screening, the remaining articles were read and assessed for inclusion in our final database based on their design (i.e., considering biological or ecological effects) and relation to MHWs. To be included in our database, articles had to make clear that the investigation was regarding the effects of a MHW (specifically, through use of the term “marine heatwave”). Only primary research articles were included (i.e., review articles and meta-analyses were removed) to reduce dual counts. The final database included 120 articles.
For each of the retained articles, we recorded key information related to the article that addressed key questions we were interested in exploring. These questions and information included: How have the approaches used, specifically the techniques (field, laboratory, modelling) and definition of MHWs, changed over time? To address this question, we extracted the year each paper was published, the techniques used (i.e., field observation, laboratory experiment, modelling; time series and remote sensing data were grouped under field methods owing to their results being inherently based on field data) and if a definition of MHW was used, and if so, what it was. We were also interested in understanding where research into MHWs has been conducted, and whether the approaches used differed across locations. In addition to the methodological detail extracted (noted above), we also identified where the research had been done within each paper (that is, where the research had been completed/observations made, rather than the location of the author’s affiliation). We extracted all of this information at the “paper” level, rather than extracting information at lower levels (i.e., each individual study or experiment), owing to the ambiguity in distinguishing whether components within a single publication were distinct study units or one complete study. Relatedly, to assess how the taxa considered and the response variables used varied within the published literature (only from papers using field and laboratory techniques as the amount of research employing modelling methods was too small), we recorded each species that was considered within a paper under broad categorical groups based on commonly used terms (e.g., seagrass, coral; kelp are as a group separate from “algae” given the large number of papers which have focussed on them specifically), as well as the response variables investigated which were also sorted into broad categorical groups based on commonly used terms (e.g., abundance, photosynthesis).
Given the focus that has been placed on using laboratory-based experiments to consider the effects of MHWs on biota, we were interested in better understanding how these manipulations had been done. Specifically, to understand which attributes of MHWs have been the focus we extracted the traits of MHWs considered in manipulative laboratory experiments (intensity, duration, number of MHWs, recovery period). This information is reported at the “treatment” level (i.e., in the 29 papers using laboratory experiments, there were 50 MHW treatments) rather than the paper level. Further, we explored whether each paper has manipulated such traits of the MHWs individually or simultaneously (i.e., orthogonally crossed). In addition, to identify if MHW effects had been considered in the context of other environmental changes, we noted any other biotic and abiotic conditions also considered in manipulative laboratory experiments (i.e., the number of taxonomic groups included, consideration of other stressors). This is reported at the paper level. During the data extraction process we noted any key literature for inclusion in the qualitative narrative synthesis in the Discussion below (sensu Falkenberg et al. 2018).
Results
Research approaches, publication patterns, & MHW definition use
Within the literature on the ecological effects of MHWs, most of the published papers have used field-based methods (73%), with a smaller proportion using laboratory settings (24%), and the least using modelling approaches (3%) (note that two papers used multiple methods, with these papers counted once for each method category). This literature surrounding MHWs has rapidly grown within the past decade, with the majority of papers published from 2016 onwards (95%; Fig. 1A). While the early literature was primarily field based, which continues to form the majority, a growing proportion of the literature employs laboratory-based components (since 2017), with modelling approaches introduced in 2020. The number of papers considering biological or ecological effects of MHWs that provide a definition of this event increased from 0% in 2014 to 51% in 2020 (Fig. 1B). The most commonly used definition we identified was published in 2016 (Hobday et al. 2016), which was used in 89% of papers that stated a definition, followed by papers citing Hobday et al. (2018) (in 7% of papers with a definition).
Geographic location of research
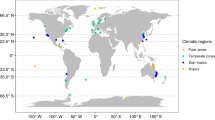
When broken down by country, the majority of papers have examined MHW impacts in Australia (~ 43%) and the USA (~ 23%) (Fig. 2A). Comparatively little research has been conducted in European countries, with a small number of papers exploring MHW impacts in Germany (n = 4), Portugal (n = 3), Spain (n = 3), the UK (n = 2), and Italy (n = 1) (Fig. 2A). A distinct lack of research conducted in Africa, Asia, and South America was observed (Fig. 2A). Where the method used in the research is considered (i.e., field, laboratory, or modelling methods), most papers published using field approaches were from Australia (n = 44) and the USA (n = 27), with the few others from Mexico (n = 6), Brazil (n = 2), and New Zealand (n = 2) (Fig. 2B). Papers using laboratory methods were also predominantly conducted in Australia (n = 9), followed by Germany (n = 4), Vietnam (n = 4), and Portugal (n = 3) (Fig. 2C). Only two papers used location-specific modelling approaches, which were conducted in Australia and the USA (n = 1 each) (Fig. 2D). One paper modelled the effects of MHWs on plankton communities in 23 locations worldwide (Hayashida et al. 2020), however, the effects were not tied to specific countries but rather general regions, so this is not reflected in Fig. 2.
Taxonomic groups and response variables
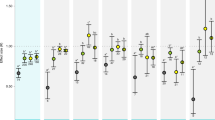
Regarding the taxa considered in research measuring the biological and ecological effects of MHWs, fish have received the greatest attention in the literature, followed by molluscs, algae, seagrasses, corals, kelp, crustaceans, and plankton (Fig. 3). Here, we have included kelp as a separate group given the large number of papers focussed on them specifically; when kelp are considered with the other algae, this becomes the taxonomic group that has received the most attention. There was overlap across methods used for each taxonomic group, with most of the groups considered in papers using field-based methods also considered in the laboratory (except for birds and mammals as they were not considered in the laboratory; Fig. 3). However, there were some notable differences. That is, for papers using field-based methods, fish were by far the most commonly considered group (n = 53), followed by molluscs (n = 32), coral (n = 29), algae (n = 26), and kelp (n = 25) which all received a similar amount of attention that was greater than experienced by the other groups. In the laboratory, a focus was placed on seagrasses (n = 27), which were examined far more often than algae (n = 16), molluscs (n = 15), and crustaceans (n = 13) (Fig. 3). Importantly, there were large differences in the relative ranking in papers using field or laboratory methods for seagrass (equal least and most number of papers in the field and laboratory, respectively), fish (most and fourth most), coral (third most and least—of those that were considered in the laboratory), and kelp (fifth most and third least—again, of those that were considered in the laboratory).
Heatmap and count of the number of categorical response variables considered for each taxonomic group within papers using field and laboratory methods. n is the number of times a response was observed for each taxa. Total row and column include taxa and response variables not shown for brevity (response variables measured across < 4 taxa, and taxa with < 4 response variables measured or non-specific taxa (i.e., other) being removed)
While taxa observed in papers using field and laboratory methods overlapped, there was a disparity in the response variables considered between the two methods of investigation. Field data tended to focus on large-scale observational responses such as abundance, biodiversity, species distributions, and mortality (Fig. 3), whereas laboratory data had more of a focus on mechanistic responses including growth, biochemical changes, and photosynthesis (Fig. 3).
Research methods in laboratory experiments
Within the laboratory experiments, the traits of MHWs assessed varied in terms of intensity, duration, and frequency. The majority (~ 80%) of MHW treatments employed elevated temperatures ranging from 3 ˚C to 8 ˚C above control temperatures. The durations of MHWs used in experiments ranged from 3 h to 108 days, although ~ 95% of treatments had a duration of > 5 days (as per Hobday et al. 2016). In terms of MHW frequency, almost 90% of papers considered a single MHW event, with few laboratory experiments investigating the effects of multiple MHWs. Finally, only ~ 23% of papers assessed the impacts of MHWs after a recovery period, when elevated temperatures had reduced to control temperatures. The majority of papers (~ 79%) investigated the impact of MHW intensity (either a single or multiple elevated temperatures), whereas examination of the impacts of MHW duration only occurred in two papers (~ 7%). Four papers (~ 14%) investigated how MHW frequency and intensity combined to affect organisms, however, the use of near-natural mesocosms led to naturally varying temperatures and not a fully orthogonal combination (Pansch et al. 2018; Guan et al. 2020; Nguyen et al. 2020; Saha et al. 2020). Interestingly, no papers investigated frequency × duration, intensity × duration, or frequency × intensity × duration interactions of MHWs and their potential combined effects on marine organisms.
In the current laboratory-based literature, almost 70% of papers investigated responses of a single species, 15% used 2 species, and 6% investigated the effects of MHWs on ecological communities (Fig. 4). In addition to species being considered in isolation, the literature often reports about a single abiotic stressor. Less than 35% of laboratory-based papers included additional stressors in combination with MHWs, which most often included light availability (n = 3), with other stressors including acidification, grazing, heavy metal exposure, and nutrient enrichment (all n = 1). All additional stressors were manipulated in addition to the MHW events, rather than investigated as components of MHWs (i.e., the influence of MHWs on nutrient availability was not investigated, rather MHW temperature as well and nutrient availability were each manipulated).
Discussion
The emergence of “marine heatwave” research and the need to incorporate perspectives from different “academic languages”
The increase of literature investigating the biological and ecological effects of MHWs over the past decade likely reflects increasing interest in this topic, given the growing awareness of global change broadly, as well as the occurrence of large, high-profile MHW events driving notable biotic impacts and consequences for the provisioning of ecosystem services (e.g., The Blob, Ningaloo Niño) (Pearce and Feng 2013; Bond et al. 2015; Beas-Luna et al. 2020; McPherson et al. 2021). For example, The Blob caused significant declines in stocks of commercially important fish species and the fisheries that rely upon them (Barbeaux et al. 2020), and Ningaloo Niño caused severe losses of seagrass meadows, potentially releasing up to 9 Tg of CO2 previously locked in the sediments stabilised by the seagrass meadows (Arias-Ortiz et al. 2018). These two events contributed almost 75% of the field research observed in the literature search here, highlighting their significance in the field.
In addition to recognition of the occurrence and consequences of these MHW events, the increase in publications may also reflect development of terminology in the field. The term “marine heatwave” was first used by Pearce et al. (2011) to describe the development of an anomalous warm water event. The term was first applied with regards to the biological or ecological implications of these warm water events in 2013 and has since become increasingly widespread. Perhaps given the origins, it is unsurprising this term is largely used to discuss field observations. Moreover, the origins of its definition may also play a part in the regions where MHWs are most often considered. The most commonly used definition of MHW we identified is from Hobday et al. (2016) with the author team of the paper including researchers primarily from temperate regions (southern Australia, the UK, and north-west USA), which may have contributed to this definition being taken up by research communities who work in these and other similar locations.
Conversely, etymological differences may have led to an under-representation of MHW research conducted by researchers who use different terminology or “academic languages” (Falkenberg and Tubb 2017). For example, although it is widely acknowledged that increased temperature is a direct cause of biological responses such as coral bleaching and mortality (Hughes et al. 2018), coral researchers often refer to warm thermal anomalies in terms of “degree-heating days” (or a variation thereof), potentially meaning such research is not included in the MHW literature. Moreover, there is a long history of considering the biological effects of temperature in other fields of research (e.g., ecophysiology and ecotoxicology; Monteiro et al. 2021) which, although not conducted in the context of climate change or MHWs, could contribute to our understanding of the effects of such events. Consequently, knowledge surrounding the biological and ecological impacts of MHWs may be even greater, and increasing even more rapidly, than identified here due to etymological differences. Thus, a greater standardisation of terminology surrounding these events will allow comparisons of effects across sub-disciplines and ultimately improve our understanding of the broad biotic impacts of MHWs, extreme climatic events, or elevated thermal anomalies.
Geographic location of research and the need to expand the areas considered
Literature focussing on the ecological effects of MHWs shows a strong geographic trend, with more research in temperate regions (i.e., Australia, North America). In addition to potential etymological influences (discussed in Sect. "The emergence of “marine heatwave” research and the need to incorporate perspectives from different “academic languages”"), this pattern is likely influenced by the locations large, field-based MHWs have occurred and therefore been studied (i.e., the Ningaloo Niño in Australia and The Blob in the USA, respectively). In regions where such large events have not occurred, field observations are not possible and researchers can only perform laboratory experiments or undertake modelling simulations, with the potential to predict outcomes for species and communities should such an event occur.
Whilst investigations of MHW events have shown that their effects in temperate systems can be large and devastating (e.g., Smale & Wernberg 2013; Santora et al. 2020), MHWs in tropical regions may have greater consequences. Tropical organisms live closer to their upper thermal limits and have reduced warming tolerance and thermal safety margins relative to their temperate counterparts (Vinagre et al. 2016; Pinksy et al. 2019). Thus, even short-term intense increases in temperature are more likely to exceed these limits compared to similar environmental changes when experienced by temperate organisms (Tewksbury et al. 2008). Further, the thermal displacement by MHWs in the tropics is larger than in temperate regions owing to weak spatial sea surface temperature gradients (Jacox et al. 2020), therefore, the impacts on species in such regions are of crucial interest. Despite the greater susceptibility to extreme temperatures, little literature regarding the ecological impacts of MHWs has been published from tropical regions, although a few field and lab articles are noted, with a distinct lack of data from Africa, as well as most of Asia, and South America. While some research may not be possible in certain areas (e.g., field observations where MHWs have not yet occurred), other approaches can be used to gain a more complete, global understanding of the potential effects of MHWs.
Taxonomic groups and response variables examined with contrasting approaches and the need to consider them in combination
Taxonomic groups
Research surrounding certain taxonomic groups is emphasized in the current literature on the biological and ecological impacts of MHWs. Specifically, focus has been placed on taxa that are charismatic and easy to document in the field (with the greatest number of papers including fish), which is where the majority of MHW research has been done to date. Such taxonomic bias is well recognized (Donaldson et al. 2016; Titley et al. 2017), and may be driven by the broad understanding of importance of charismatic organisms (e.g., fisheries resources and biogenic habitat biodiversity), as well as the relative ease of their monitoring (Troudet et al. 2017). Such taxonomic patterns can lead to skewed conclusions and the potential to wrongly estimate ecosystem effects (Feeley et al. 2017; Troudet et al. 2017). Consequently, this taxonomic bias should be addressed in future research. This future work could expand the body of research from considering foundation species (e.g., seagrass, corals, kelp), to also incorporate organisms representative of other functional groups such as detritivores, herbivores, and planktivores that also play important trophic roles and effects on whom could drive notable ecosystem-wide consequences.
The response variables considered for each taxa also differ. For example, while those that use field methods often document the changes in abundance for fish, molluscs, and algae, relatively little is known about the direct impacts of MHWs on their mortality, while the inverse pattern was found for investigations of corals (i.e., a focus on mortality, with less consideration of abundance); although this is likely an artefact of the sessile and persistent nature of corals in contrast to the other mentioned taxa (that is coral skeletons allow for the identification of mortality in the field, whereas the others are simply not present following mortality). In contrast, for the three most considered taxa in laboratory investigations of seagrass, algae, and molluscs, there were fairly similar patterns in response variables with a key gap for all being consideration of reproduction. Filling this gap and quantifying changes in reproductive processes will provide knowledge necessary in identifying how MHWs may alter recruitment and ultimately the continued persistence of populations in the face of climate change (Shanks et al. 2020). Together, such patterns indicate there are response variables needing to be included in more research efforts, but these are different among the taxa investigated. Importantly, where the relevant field and laboratory approaches are used to quantify the range of response variables (from, for example, abundance and distribution observed in the field, to laboratory quantification of biochemical alterations) we will better be able to relate changes in large-scale patterns (such as abundance) with the underlying mechanisms (discussed further below).
Response variables in the field and laboratory
Some response variables have been considered more than others with this reflecting, in part, the methods used to conduct the investigation (i.e., field or laboratory). Field observations (including time series and remote sensing), which are used in the majority of papers published to date, primarily document changes in abundance, biodiversity, and distribution in response to MHWs, meaning we have an insight as to the scale and ecosystem impacts of these events. For example, some of these field observations have identified MHW-driven mortality of populations, which are particularly evident in sessile species (Smale and Wernberg 2013; Garrabou et al. 2022). Further, field research has revealed MHWs can cause shifts in community structure by decreasing the abundance of cool-affinity species while increasing the abundance of warm-affinity species (community tropicalisation), which can be sustained for extended periods (Wernberg et al. 2016; Suryan et al. 2021; Michaud et al. 2022), thus altering the ecological function of the ecosystem (Smale et al. 2019).
The quantification of large-scale responses in the field requires researchers to already be conducting long-term monitoring including observing organisms at the site and documenting traits that easily enable comparisons of abundance, mortality, and biodiversity. These long-term datasets are necessary to identify changes in community-level responses (e.g., MchPherson et al. 2021; Beas-Luna et al. 2020). While changes in abundance and biodiversity can be apparent, many field investigations use before-after data making it difficult to establish whether MHWs directly or indirectly impact species as they have the capacity to do both within an ecosystem (e.g., Smale et al. 2017). For example, the direct loss of habitat forming species or changes in phytoplankton abundance can indirectly translate to changes in biodiversity as associated species are affected (Nowicki et al. 2019; Thomsen et al. 2019; Michaud et al. 2022). Timely field observations can, however, provide greater detail as to both direct and indirect impacts of MHWs (Magel et al. 2020). For example, monitoring before and after a MHW allowed identification that where rabbitfish were able to establish, rates of herbivory by these organisms on kelp increased 30-fold driving an indirect effect (Zarco-Perello et al. 2017).
There has been a recent increase in the number of laboratory experiments that complement field observations and offer the capacity to investigate small-scale mechanisms (notably biochemical responses and photosynthesis) that underlie biological processes ranging from sub-cellular levels to biotic interactions. Responses considered more often in papers using laboratory methods include those that require frequent or fine-scale measurement, generally considering the direct impact of MHWs on organisms (Madeira et al. 2018; Atkinson et al. 2020; Joyce et al. 2022). Laboratory experiments also offer a stable platform for studying organisms that are rare or difficult to observe in field settings (that is, fish are the most considered organisms in the field, while seagrasses dominate laboratory settings), and thus their inclusion could result in a more holistic understanding of the biotic effects of MHWs. Such experimental approaches have shown that MHWs can induce biochemical changes (e.g., abundance of fatty acids, antioxidant compounds, protective proteins) in organisms including algae (Gouvea et al. 2017; Britton et al. 2020), crustaceans (Madeira et al. 2018), and diatoms (Feijao et al. 2020), which have implications for their continued role within food webs. Further, the photosynthetic capacity of habitat forming seaweeds is largely negatively affected by MHWs and this can affect their ecosystem role (Gouvea et al. 2017; Rendina et al. 2019; Britton et al. 2020; Sanchez-Barredo et al. 2020), however, responses may be species specific (Atkinson et al. 2020). Data collected from laboratory-based experimental investigations can, then, allow more precise investigations that may uncover subtle responses of organisms to MHWs.
The differences in the type of data collected, and biological insight obtained, through field and laboratory investigations highlight the need to combine data obtained using multiple approaches to provide a holistic understanding of the direct and indirect impacts of MHWs. Whilst it is commonplace for a developing field of research to document changes in a narrow range of species for a narrow range of responses, ensuring links can be made across field and laboratory research would allow a more complete understanding of impacts. Field surveys of abundance, biodiversity, and distribution, which document the outcome of an event are complemented by laboratory-based investigations of growth, biochemical responses, and photosynthesis that provide a mechanistic understanding of the channels through which organisms are affected. Other, less frequently reported responses include reproduction, feeding, genetics, organism behaviour, and ecosystem function. Documenting effects of MHWs on these responses in the field or laboratory (as appropriate) would provide a greater understanding of overall biological and ecosystem effects. For instance, effects on genetic variability can have implications for population persistence (Gaggiotti 2003), and behavioural changes can have repercussions for entire ecosystem functioning (Allgeier et al. 2020). Thus, greater inclusion of these processes in the published literature would further our understanding of resistance, population persistence, energy transfer, and changes in ecosystem service provision under MHWs.
Manipulations of MHWs in laboratory experiments and the need to enhance their relevance
MHW traits
Laboratory-based experiments allow manipulation and investigation of the effects of the traits of stressors—in the case of MHWs traits of particular interest are their intensity, duration, and frequency. In the most common definition of MHWs adopted in the literature, Hobday et al. (2016) characterized MHWs as discrete events with temperatures warmer than the 90th percentile based on historical data and lasting at least 5 days. Despite the increasing prevalence of this definition, and focus on considering intensity (80% of the papers employing a single MHWs of a single duration with multiple magnitudes to quantify their impacts), many investigations do not apply this characterisation of location-specific intensity, instead arbitrarily increasing temperatures in comparison to a control to simulate a MHW event. In terms of duration, while the majority of investigations that simulated MHWs employed durations of at least five days (as per Hobday et al. 2016), real-world events are often much longer. Only two of the papers found in this review examined responses of organisms after > 50 days at MHW temperatures, and thus our understanding of how extended events can impact organisms is limited. Moreover, only 7% of papers considered the role of MHW duration, and none of the identified papers conducted fully orthogonal experiments to investigate interactions of MHW intensity, frequency, and duration. While investigations simulating MHWs with individually differing traits is a starting point, real world events vary in both intensity and duration, and the occurrence of two events with the same characteristics is unlikely. Further, combinations of such traits may lead to additive or synergistic effects on organisms, as has been found by combining different stressors in experiments (Crain et al. 2008). Thus, understanding how combinations of these traits affect organisms will give a greater understanding how long species can persist under stressful conditions induced by MHWs with various intensities, and will enhance predictions of limits of survival during extended events.
The temporal patterns of MHWs—specifically their frequency and recurrence—can also be manipulated in laboratory experiments. Researchers are yet to make the most of this capacity, with only 14% of papers manipulating MHW frequency. This trait is important to understand owing to concepts such as thermal priming. For instance, species can upregulate genes related to thermal stress during a MHW, the effects of which can carry over to provide protection from subsequent events (Nguyen et al. 2020; Pazzaglia et al. 2022; Xu Et al. 2022). In contrast, species may show recovery from single MHWs, but subsequent events can cause downregulation of genes related to energy metabolism and molecular defence (He et al. 2023), leading to more severe impacts in subsequent events.
Relatedly, central to the definition of MHWs is that temperature returns to the pre-heatwave condition following the event. However, this is not reflected in the reviewed literature, with less than a quarter of papers using laboratory-based methods observing responses after a recovery period. As MHW onset occurs rapidly and the event can be brief, resistant organisms may be able to persist following a return to baseline temperatures without substantial long-term impacts (Britton et al. 2020). In contrast, organisms that are susceptible to extreme events may show latent responses (Minuti et al. 2021). The potential for these delayed and persistent organism responses highlight the need to consider the long-term impacts of complex MHWs characterised by differing intensities, durations, and frequencies, even after the event has ended.
The presence of interacting species, ecosystem states, indirect effects, and multiple stressors
Marine heatwaves manifest in ecosystems that are comprised of multiple species and are undergoing complex changes to a range of different abiotic parameters. The experimental laboratory research is, however, still dominated by considerations of single species and stressors. Promisingly, some papers have included multiple species, and these often quantify whole organism responses across multiple species. Such understanding can be important for understanding why species, particularly those that are similar, differ in their responses to MHWs. In the future, it will be useful to further investigate interactions among species. For example, experimental manipulations have been used to examine the effect of a MHW on the regulation of chemical defence against grazers by quantifying feeding of isopod grazers on rockweed, which indicated that this was not disrupted (Saha et al. 2020). Such understanding is particularly important given that the interactions between species can modify the direct responses observed through indirect effects (e.g., the response of a primary producer can be altered by indirect effects of its herbivore consumer; Falkenberg et al. 2013). Here, two of the laboratory-based papers investigated how MHWs can impact communities, with both finding shifts in biodiversity that could translate to alterations in ecosystem functioning (Remy et al. 2017; Pansch et al. 2018). Such phenomena have been observed in the field, and show that such alternative stable states can remain for extended periods after MHW events have finished (Suryan et al. 2021). Such protracted impacts of MHWs can be reinforced as the system demonstrates hysteresis, whereby there are ecological interactions persisting even after environmental conditions have returned to pre-MHW conditions (Beisner et al. 2003). These extended alterations in ecosystem states then lead to impacts on the associated provisioning and regulating services (Smith et al. 2023). With such studies able to quantify how ecosystem function may shift under MHWs, the body of literature investigating MHW impacts would benefit from moves towards understanding more complex systems by incorporating multiple species in combination.
While considering the isolated responses to single stressors, such as MHWs, often provides the foundation for a field of research, the reality in natural systems is likely to be more complex as they are affected by multiple changes due to both global climate impacts (e.g., long-term warming, ocean acidification, sea level rise, etc.; IPCC, 2022), and other localised human activities (e.g., nutrient pollution, species introduction, etc.; Howarth et al. 2000; Molnar et al. 2008). While multiple stressors can interact in complex ways, leading to additive, synergistic, or antagonistic effects (Crain et al. 2008), < 35% of the observed literature reviewed here simulating MHWs considered manipulations of additional stressors. Where multiple stressors were observed, these were artificially manipulated along with the MHW traits (Gouvêa et al. 2017; Bass et al. 2023). As MHWs themselves can drive other changes in the environment, for example by modifying nutrient availability (Hayashida et al. 2020), experiments manipulating multiple stressors will enhance their real world applicability and allow development of a greater understanding of cause–effect relationships (Seebacher & Franklin 2012; De Boeck et al. 2015; Cooke et al. 2017).
Conclusions
This synthesis has revealed that the body of literature surrounding research into the ecological effects of MHWs is rapidly increasing, and incorporates complementary field, laboratory, and modelling approaches. With the increasing intensity, duration, and frequency of MHWs, it will be important that we can understand their effects across a range of geographic locations, species, biotic processes, communities, and ecosystems. The literature reviewed here revealed the variability that can be observed in biological and ecological responses to such events. To broaden our understanding of the ecological effects that MHWs can drive, incorporating a wider geographic range of studies, as well as a wider range of mechanistic responses such as genetics, reproduction, behaviour, and trophic interactions, would be particularly useful as these can drive changes in abundance, mortality, and ecosystem functioning. At present, laboratory investigations considering MHWs tend to focus on single traits of MHW events (i.e., intensity or duration), and use single species-single stressor experiments. Incorporating the effects of different MHW traits, multiple MHW events, how organisms recover from MHWs, and additional stressors will further enhance our understanding of how MHWs impact individual organisms; considering multiple species will enhance understanding of ecosystem responses. As we recognize the biological and ecological effects caused by MHWs across diverse regions, ecosystems, species, and processes, we will be better able to identify those that are most at risk. Improved understanding of the effects caused by MHWs will further our capacity to forecast how these events will shape ecosystems in the future, and prepare human communities to mitigate or adapt to induced changes.
Data availability
The database of reviewed literature is available in Supplementary Information.
References
Allgeier JE, Cline TJ, Walsworth TE, Wathen G, Layman CA, Schindler DE (2020) Individual behavior drives ecosystem function and the impacts of harvest. Sci Adv 6(9):eaax8329. https://doi.org/10.1126/sciadv.aax8329
Arias-Ortiz A, Serrano O, Masqué P, Lavery PS, Mueller U, Kendrick GA, Rozaimi M, Esteban A, Fourqurean JW, Marbà N, Mateo MA, Murray K, Rule MJ, Duarte CM (2018) A marine heatwave drives massive losses from the world’s largest seagrass carbon stocks. Nat Clim Chang 8(4):4. https://doi.org/10.1038/s41558-018-0096-y
Atkinson J, King NG, Wilmes SB, Moore PJ (2020) Summer and winter marine heatwaves favor an invasive over native seaweeds. J Phycol 56(6):1591–1600. https://doi.org/10.1111/jpy.13051
Barbeaux SJ, Holsman K, Zador S (2020) Marine heatwave stress test of ecosystem-based fisheries management in the Gulf of Alaska Pacific cod fishery. Front Mar Sci. https://doi.org/10.3389/fmars.2020.00703
Bass AV, Smith KE, Smale DA (2023) Marine heatwaves and decreased light availability interact to erode the ecophysiological performance of habitat-forming kelp species. J Phycol. https://doi.org/10.1111/jpy.13332
Beas-Luna R, Micheli F, Woodson CB, Carr M, Malone D, Torre J, Boch C, Caselle JE, Edwards M, Freiwald J, Hamilton SL (2020) Geographic variation in responses of kelp forest communities of the California current to recent climatic changes. Glob Chang Biol 26(11):6457–6473. https://doi.org/10.1111/gcb.15273
Beisner BE, Haydon DT, Cuddington K (2003) Alternative stable states in ecology. Front Ecol Environ 1(7):376–382. https://doi.org/10.1890/1540-9295(2003)001[0376:ASSIE]2.0.CO;2
Bernal MA, Schunter C, Lehmann R, Lightfoot DJ, Allan BJM, Veilleux HD, Rummer JL, Munday PL, Ravasi T (2020) Species-specific molecular responses of wild coral reef fishes during a marine heatwave. Sci Adv 6(12):eaay3423. https://doi.org/10.1126/sciadv.aay3423
Bond NA, Cronin MF, Freeland H, Mantua N (2015) Causes and impacts of the 2014 warm anomaly in the NE Pacific. Geophys Res Lett 42(9):3414–3420. https://doi.org/10.1002/2015GL063306
Britton D, Schmid M, Noisette F, Havenhand JN, Paine ER, McGraw CM, Revill AT, Virtue P, Nichols PD, Mundy CN, Hurd CL (2020) Adjustments in fatty acid composition is a mechanism that can explain resilience to marine heatwaves and future ocean conditions in the habitat-forming seaweed Phyllospora comosa (Labillardière) C. Agardh. Global Chang Biol 26(6):3512–3524. https://doi.org/10.1111/gcb.15052
Caputi N, Kangas M, Chandrapavan A, Hart A, Feng M, Marin M, de Lestang S (2019) Factors affecting the recovery of invertebrate stocks from the 2011 Western Australian extreme marine heatwave. Front Mar Sci. https://doi.org/10.3389/fmars.2019.00484
Coleman MA, Minne AJP, Vranken S, Wernberg T (2020) Genetic tropicalisation following a marine heatwave. Sci Rep 10(1):12726. https://doi.org/10.1038/s41598-020-69665-w
Cooke SJ, Birnie-Gauvin K, Lennox RJ, Taylor JJ, Rytwinski T, Rummer JL, Franklin CE, Bennett JR, Haddaway NR (2017) How experimental biology and ecology can support evidence-based decision-making in conservation: avoiding pitfalls and enabling application. Conserv Physiol. https://doi.org/10.1093/conphys/cox043
Crain CM, Kroeker K, Halpern BS (2008) Interactive and cumulative effects of multiple human stressors in marine systems. Ecol Lett 11(12):1304–1315. https://doi.org/10.1111/j.1461-0248.2008.01253.x
De Boeck HJ, Vicca S, Roy J, Nijs I, Milcu A, Kreyling J, Jentsch A, Chabbi A, Campioli M, Callaghan T, Beierkuhnlein C, Beier C (2015) Global change experiments: challenges and opportunities. Bioscience 65(9):922–931. https://doi.org/10.1093/biosci/biv099
Deser C, Alexander MA, Xie SP, Phillips AS (2010) Sea surface temperature variability: patterns and mechanisms. Ann Rev Mar Sci 2:115–143. https://doi.org/10.1146/annurev-marine-120408-151453
Donaldson MR, Burnett NJ, Braun DC, Suski CD, Hinch SG, Cooke SJ, Kerr JT (2016) Taxonomic bias and international biodiversity conservation research. FACETS. https://doi.org/10.1139/facets-2016-0011
Dudley PN, Rogers TL, Morales MM, Stoltz AD, Sheridan CJ, Beulke AK, Pomeroy C, Carr MH (2021) A more comprehensive climate vulnerability assessment framework for fisheries social-ecological systems. Front Mar Sci. https://doi.org/10.3389/fmars.2021.678099
Falkenberg LJ, Tubb A (2017) Economic effects of ocean acidification: publication patterns and directions for future research. Ambio 46(5):543–553. https://doi.org/10.1007/s13280-017-0895-9
Falkenberg LJ, Russell BD, Connell SD (2013) Future herbivory: the indirect effects of enriched CO2 may rival its direct effects. Mar Ecol Prog Ser 492:85–95. https://doi.org/10.3354/meps10491
Falkenberg LJ, Dupont S, Bellerby RGJ (2018) Approaches to reconsider literature on physiological effects of environmental change: examples from ocean acidification research. Front Mar Sci 5:453. https://doi.org/10.3389/fmars.2018.00453
Feeley KJ, Stroud JT, Perez TM (2017) Most ‘global’ reviews of species’ responses to climate change are not truly global. Divers Distrib 23(3):231–234. https://doi.org/10.1111/ddi.12517
Feijão E, Franzitta M, Cabrita MT, Caçador I, Duarte B, Gameiro C, Matos AR (2020) Marine heat waves alter gene expression of key enzymes of membrane and storage lipids metabolism in Phaeodactylum tricornutum. Plant Physiol Biochem 156:357–368. https://doi.org/10.1016/j.plaphy.2020.09.022
Gaggiotti OE (2003) Genetic threats to population persistence. Ann Zool Fenn 40(2):155–168
Garrabou J, Gómez-Gras D, Medrano A, Cerrano C, Ponti M, Schlegel R, Bensoussan N, Turicchia E, Sini M, Gerovasileiou V, Teixido N, Mirasole A, Tamburello L, Cebrian E, Rilov G, Ledoux J-B, Souissi JB, Khamassi F, Ghanem R, Harmelin J-G (2022) Marine heatwaves drive recurrent mass mortalities in the Mediterranean Sea. Global Chang Biol 28(19):5708–5725. https://doi.org/10.1111/gcb.16301
Gouvêa LP, Schubert N, Martins CDL, Sissini M, Ramlov F, de Rodrigues ER, Bastos EO, Freire VC, Maraschin M, Carlos Simonassi J, Varela DA, Franco D, Cassano V, Fonseca AL, Barufi J, Horta PA (2017) Interactive effects of marine heatwaves and eutrophication on the ecophysiology of a widespread and ecologically important macroalga: temperature and nutrient effect on macroalgae. Limnol Oceanogr 62(5):2056–2075. https://doi.org/10.1002/lno.10551
Guan C, Saha M, Weinberger F (2020) Simulated heatwaves lead to upregulated chemical defense of a marine foundation macrophyte against microbial colonizers. Front Mar Sci 7:463. https://doi.org/10.3389/fmars.2020.00463
Hayashida H, Matear RJ, Strutton PG (2020) Background nutrient concentration determines phytoplankton bloom response to marine heatwaves. Glob Chang Biol 26:4800–4811. https://doi.org/10.1111/gcb.15255
He G, Xiong X, Peng Y, Yang C, Xu Y, Liu X, Liang J, Masanja F, Yang K, Xu X, Zheng Z, Deng Y, Leung JYS, Zhao L (2023) Transcriptomic responses reveal impaired physiological performance of the pearl oyster following repeated exposure to marine heatwaves. Sci Total Environ 854:158726. https://doi.org/10.1016/j.scitotenv.2022.158726
Hobday AJ, Alexander LV, Perkins SE, Smale DA, Straub SC, Oliver ECJ, Benthuysen JA, Burrows MT, Donat MG, Feng M, Holbrook NJ, Moore PJ, Scannell HA, Sen Gupta A, Wernberg T (2016) A hierarchical approach to defining marine heatwaves. Prog Oceanogr 141:227–238. https://doi.org/10.1016/j.pocean.2015.12.014
Hobday AJ, Oliver E, Sen Gupta A, Benthuysen J, Burrows M, Donat M, Holbrook N, Moore P, Thomsen M, Wernberg T, Smale D (2018) Categorizing and naming marine heatwaves. Oceanography. https://doi.org/10.5670/oceanog.2018.205
Holbrook NJ, Sen Gupta A, Oliver ECJ, Hobday AJ, Benthuysen JA, Scannell HA, Smale DA, Wernberg T (2020) Keeping pace with marine heatwaves. Nat Rev Earth Environ 1(9):482–493. https://doi.org/10.1038/s43017-020-0068-4
Howarth RW, Anderson DB, Cloern JE, Elfring C, Hopkinson CS, Lapointe B, Walker D (2000) Nutrient pollution of coastal rivers, bays, and seas. Issues Ecol 7:1–16
Hughes TP, Kerry JT, Baird AH, Connolly SR, Dietzel A, Eakin CM, Heron SF, Hoey AS, Hoogenboom MO, Liu G, McWilliam MJ, Pears RJ, Pratchett MS, Skirving WJ, Stella JS, Torda G (2018) Global warming transforms coral reef assemblages. Nature 556(7702):492–496. https://doi.org/10.1038/s41586-018-0041-2
IPCC (2022) Climate change 2022: impacts, adaptation, and vulnerability. In: Pörtner H-O, Roberts DC, Tignor M, Poloczanska ES, Mintenbeck K, Alegría A, Craig M, Langsdorf S, Löschke S, Möller V, Okem A, Rama B (eds) Contribution of working group II to the sixth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, p 3056. https://doi.org/10.1017/9781009325844
Jacox MG, Alexander MA, Bograd SJ, Scott JD (2020) Thermal displacement by marine heatwaves. Nature 584(7819):7819. https://doi.org/10.1038/s41586-020-2534-z
Jacox MG, Alexander MA, Amaya D, Becker E, Bograd SJ, Brodie S, Hazen EL, Pozo Buil M, Tommasi D (2022) Global seasonal forecasts of marine heatwaves. Nature 604(7906):7906. https://doi.org/10.1038/s41586-022-04573-9
Joyce PWS, Tang WY, Falkenberg LJ (2022) Marine heatwaves of different magnitudes have contrasting effects on herbivore behaviour. Sci Rep 12(1):17309. https://doi.org/10.1038/s41598-022-21567-9
Kayanne H (2017) Validation of degree heating weeks as a coral bleaching index in the northwestern Pacific. Coral Reefs 36:63–70. https://doi.org/10.1007/s00338-016-1524-y
Laufkötter C, Zscheischler J, Frölicher TL (2020) High-impact marine heatwaves attributable to human-induced global warming. Science 369(6511):1621–1625. https://doi.org/10.1126/science.aba0690
Madeira C, Leal MC, Diniz MS, Cabral HN, Vinagre C (2018) Thermal stress and energy metabolism in two circumtropical decapod crustaceans: responses to acute temperature events. Mar Environ Res 141:148–158. https://doi.org/10.1016/j.marenvres.2018.08.015
Magel JMT, Dimoff SA, Baum JK (2020) Direct and indirect effects of climate change-amplified pulse heat stress events on coral reef fish communities. Ecol Appl 30(6):e02124. https://doi.org/10.1002/eap.2124
McClanahan TR, Azali MK (2021) Environmental variability and threshold model’s predictions for coral reefs. Front Mar Sci. https://doi.org/10.3389/fmars.2021.778121
McPherson ML, Finger DJ, Houskeeper HF, Bell TW, Carr MH, Rogers-Bennett L, Kudela RM (2021) Large-scale shift in the structure of a kelp forest ecosystem co-occurs with an epizootic and marine heatwave. Commun Biol 4(1):298. https://doi.org/10.1038/s42003-021-01827-6
Michaud KM, Reed DC, Miller RJ (2022) The Blob marine heatwave transforms California kelp forest ecosystems. Commun Biol 5:1143. https://doi.org/10.1038/s42003-022-04107-z
Minuti JJ, Byrne M, Hemraj DA, Russell BD (2021) Capacity of an ecologically key urchin to recover from extreme events: Physiological impacts of heatwaves and the road to recovery. Sci Total Environ 785:147281. https://doi.org/10.1016/j.scitotenv.2021.147281
Molnar JL, Gamboa RL, Revenga C, Spalding MD (2008) Assessing the global threat of invasive species to marine biodiversity. Front Ecol Environ 6(9):485–492
Monteiro DA, Kalinin AL, Rantin FT, McKenzie DJ (2021) Use of complex physiological traits as ecotoxicological biomarkers in tropical freshwater fishes. J Exp Zool 335:745–760. https://doi.org/10.1002/jez.2540
Nguyen HM, Kim M, Ralph PJ, Marín-Guirao L, Pernice M, Procaccini G (2020) Stress memory in seagrasses: first insight into the effects of thermal priming and the role of epigenetic modifications. Front Plant Sci 11:494. https://doi.org/10.3389/fpls.2020.00494
Nowicki R, Heithaus M, Thomson J, Burkholder D, Gastrich K, Wirsing A (2019) Indirect legacy effects of an extreme climatic event on a marine megafaunal community. Ecol Monogr 89(3):e01365. https://doi.org/10.1002/ecm.1365
Oliver ECJ, Donat MG, Burrows MT, Moore PJ, Smale DA, Alexander LV, Benthuysen JA, Feng M, Sen Gupta A, Hobday AJ, Holbrook NJ, Perkins-Kirkpatrick SE, Scannell HA, Straub SC, Wernberg T (2018) Longer and more frequent marine heatwaves over the past century. Nat Commun 9(1):1324. https://doi.org/10.1038/s41467-018-03732-9
Oliver ECJ, Burrows MT, Donat MG, Sen Gupta A, Alexander LV, Perkins-Kirkpatrick SE, Benthuysen JA, Hobday AJ, Holbrook NJ, Moore PJ, Thomsen MS, Wernberg T, Smale DA (2019) Projected marine heatwaves in the 21st century and the potential for ecological impact. Front Mar Sci. https://doi.org/10.3389/fmars.2019.00734
Oliver ECJ, Benthuysen JA, Darmaraki S, Donat MG, Hobday AJ, Holbrook NJ, Schlegel RW, Sen Gupta A (2021) Marine heatwaves. Ann Rev Mar Sci 13(1):313–342. https://doi.org/10.1146/annurev-marine-032720-095144
Pansch C, Scotti M, Barboza FR, Al-Janabi B, Brakel J, Briski E, Bucholz B, Franz M, Ito M, Paiva F, Saha M, Sawall Y, Weinberger F, Wahl M (2018) Heat waves and their significance for a temperate benthic community: a near-natural experimental approach. Glob Chang Biol 24(9):4357–4367. https://doi.org/10.1111/gcb.14282
Pazzaglia J, Badalamenti F, Bernardeau-Esteller J, Ruiz JM, Giacalone VM, Procaccini G, Marín-Guirao L (2022) Thermo-priming increases heat-stress tolerance in seedlings of the Mediterranean seagrass P. oceanica. Mar Pollut Bull 174:113164. https://doi.org/10.1016/j.marpolbul.2021.113164
Pearce AF, Feng M (2013) The rise and fall of the “marine heat wave” off Western Australia during the summer of 2010/2011. J Mar Syst 111–112:139–156. https://doi.org/10.1016/j.jmarsys.2012.10.009
Pearce A, Lenanton R, Jackson G, Moore J, & Feng M (2011) The ‘marine heat wave’ off Western Australia during the summer of 2010/11 (Fisheries research report No. 222.). Department of Fisheries, Western Australia.
Pinsky ML, Eikeset AM, McCauley DJ, Payne JL, Sunday JM (2019) Greater vulnerability to warming of marine versus terrestrial ectotherms. Nature 569:108–111. https://doi.org/10.1038/s41586-019-1132-4
Remy M, Hillebrand H, Flöder S (2017) Stability of marine phytoplankton communities facing stress related to global change: interactive effects of heat waves and turbidity. J Exp Mar Biol Ecol 497:219–229. https://doi.org/10.1016/j.jembe.2017.10.002
Rendina F, Bouchet PJ, Appolloni L, Russo GF, Sandulli R, Kolzenburg R, Putra A, Ragazzola F (2019) Physiological response of the coralline alga Corallina officinalis L. to both predicted long-term increases in temperature and short-term heatwave events. Mar Environ Res 150:104764. https://doi.org/10.1016/j.marenvres.2019.104764
Saha M, Barboza FR, Somerfield PJ, Al-Janabi B, Beck M, Brakel J, Ito M, Pansch C, Nascimento-Schulze JC, Jakobsson Thor S, Weinberger F, Sawall Y (2020) Response of foundation macrophytes to near-natural simulated marine heatwaves. Glob Chang Biol 26(2):417–430. https://doi.org/10.1111/gcb.14801
Sánchez-Barredo M, Sandoval-Gil JM, Zertuche-González JA, Ladah LB, Belando-Torrentes MD, Beas-Luna R, Cabello-Pasini A (2020) Effects of heat waves and light deprivation on giant kelp juveniles (Macrocystis pyrifera, Laminariales, Phaeophyceae). J Phycol 56(4):880–894. https://doi.org/10.1111/jpy.13000
Santora JA, Mantua NJ, Schroeder ID, Field JC, Hazen EL, Bograd SJ, Sydeman WJ, Wells BK, Calambokidis J, Saez L, Lawson D, Forney KA (2020) Habitat compression and ecosystem shifts as potential links between marine heatwave and record whale entanglements. Nat Commun 11(1):536. https://doi.org/10.1038/s41467-019-14215-w
Seebacher F, Franklin CE (2012) Determining environmental causes of biological effects: the need for a mechanistic physiological dimension in conservation biology. Philos Trans R Soc b: Biol Sci 367(1596):1607–1614. https://doi.org/10.1098/rstb.2012.0036
Shanks AL, Rasmuson LK, Valley JR, Jarvis MA, Salant C, Sutherland DA, Lamont EI, Hainey MAH, Emlet RB (2020) Marine heat waves, climate change, and failed spawning by coastal invertebrates. Limnol Oceanogr 65:627–636. https://doi.org/10.1002/lno.11331
Smale DA, Wernberg T (2013) Extreme climatic event drives range contraction of a habitat-forming species. Proc Royal Soc b: Biol Sci 280(1754):20122829. https://doi.org/10.1098/rspb.2012.2829
Smale DA, Wernberg T, Vanderklift MA (2017) Regional-scale variability in the response of benthic macroinvertebrate assemblages to a marine heatwave. Mar Ecol Prog Ser 568:17–30. https://doi.org/10.3354/meps12080
Smale DA, Wernberg T, Oliver ECJ, Thomsen M, Harvey BP, Straub SC, Burrows MT, Alexander LV, Benthuysen JA, Donat MG, Feng M, Hobday AJ, Holbrook NJ, Perkins-Kirkpatrick SE, Scannell HA, Sen Gupta A, Payne BL, Moore PJ (2019) Marine heatwaves threaten global biodiversity and the provision of ecosystem services. Nat Clim Chang. https://doi.org/10.1038/s41558-019-0412-1
Smith KE, Burrows MT, Hobday AJ, Gupta AS, Moore PJ, Thomsen M, Wernberg T, Smale DA (2021) Socioeconomic impacts of marine heatwaves: global issues and opportunities. Science 374:eabj3593. https://doi.org/10.1126/science.abj3593
Smith KE, Burrows MT, Hobday AJ, King NG, Moore PJ, Sen Gupta A, Thomsen MS, Wernberg T, Smale DA (2023) Biological impacts of marine heatwaves. Ann Rev Mar Sci 15(1):119–145. https://doi.org/10.1146/annurev-marine-032122-121437
Straub SC, Wernberg T, Thomsen MS, Moore PJ, Burrows MT, Harvey BP, Smale DA (2019) Resistance, extinction, and everything in between – the diverse responses of seaweeds to marine heatwaves. Front Mar Sci. https://doi.org/10.3389/fmars.2019.00763
Suryan RM, Arimitsu ML, Coletti HA, Hopcroft RR, Lindeberg MR, Barbeaux SJ, Batten SD, Burt WJ, Bishop MA, Bodkin JL, Brenner R, Campbell RW, Cushing DA, Danielson SL, Dorn MW, Drummond B, Esler D, Gelatt T, Hanselman DH, Zador SG (2021) Ecosystem response persists after a prolonged marine heatwave. Sci Rep 11(1):6235. https://doi.org/10.1038/s41598-021-83818-5
Tewksbury JJ, Huey RB, Deutsch CA (2008) Putting the heat on tropical animals. Science 320(5881):1296–1297. https://doi.org/10.1126/science.1159328
Thomsen MS, Mondardini L, Alestra T, Gerrity S, Tait L, South PM, Lilley SA, Schiel DR (2019) Local extinction of bull kelp (Durvillaea spp.) due to a marine heatwave. Front Mar Sci 6:84. https://doi.org/10.3389/fmars.2019.00084
Titley MA, Snaddon JL, Turner EC (2017) Scientific research on animal biodiversity is systematically biased towards vertebrates and temperate regions. PLoS ONE 12(12):e0189577. https://doi.org/10.1371/journal.pone.0189577
Troudet J, Grandcolas P, Blin A, Vignes-Lebbe R, Legendre F (2017) Taxonomic bias in biodiversity data and societal preferences. Sci Rep 7(1):9132. https://doi.org/10.1038/s41598-017-09084-6
Vinagre C, Leal I, Mendonça V, Madeira D, Narciso L, Diniz MS, Flores AAV (2016) Vulnerability to climate warming and acclimation capacity of tropical and temperate coastal organisms. Ecol Ind 62:317–327. https://doi.org/10.1016/j.ecolind.2015.11.010
Wernberg T, Bennett S, Babcock RC, de Bettignies T, Cure K, Depczynski M, Dufois F, Fromont J, Fulton CJ, Hovey RK, Harvey ES, Holmes TH, Kendrick GA, Radford B, Santana-Garcon J, Saunders BJ, Smale DA, Thomsen MS, Tuckett CA, Wilson S (2016) Climate-driven regime shift of a temperate marine ecosystem. Science 353(6295):169–172. https://doi.org/10.1126/science.aad8745
Xu Y, Wang Z, Zhang Y, Liang J, He G, Liu X, Zheng Z, Deng Y, Zhao L (2022) Transcriptome analysis reveals acclimation responses of pearl oysters to marine heatwaves. Sci Total Environ 810:151189. https://doi.org/10.1016/j.scitotenv.2021.151189
Zarco-Perello S, Wernberg T, Langlois TJ, Vanderklift MA (2017) Tropicalization strengthens consumer pressure on habitat-forming seaweeds. Sci Rep 7:820. https://doi.org/10.1038/s41598-017-00991-2
Funding
The authors of this manuscript received no funding for the work reported in their manuscript.
Author information
Authors and Affiliations
Contributions
LJF and PWSJ conceived the study and conducted the literature search. PWSJ, CBT, YLY collated data from literature. PWSJ analyzed and visualised data. PWSJ and LJF wrote the original draft of the manuscript, which was revised by all authors.
Corresponding author
Ethics declarations
Conflict of interest
LJF is an Associate Editor with Marine Biology. The authors declare that they have no other competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Additional information
Responsible Editor: Sandra Shumway.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Joyce, P.W.S., Tong, C.B., Yip, Y.L. et al. Marine heatwaves as drivers of biological and ecological change: implications of current research patterns and future opportunities. Mar Biol 171, 20 (2024). https://doi.org/10.1007/s00227-023-04340-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-023-04340-y