Abstract
Maintaining homeothermy may be a major challenge when species are confronted with ambient temperatures beyond their thermoneutral zone. Bird species occupying open landscapes are inherently exposed to acute heat loss and heat gain, which force them to adopt a suite of behavioural and physiological strategies to maintain homeothermy. Both types of responses could be influenced by their relative bill and leg sizes, but experimental data are lacking. Here, we evaluated how variation in body postural adjustments, panting, and locomotor activity in the dunlin Calidris alpina can be explained by experimental ambient temperature and relative bill and leg sizes. Additionally, we estimated resting metabolic rate and evaporative water loss to assess potential links between both physiological traits and relative bill and leg sizes. Temperatures below the thermoneutral zone were counteracted by enhancing metabolic heat production through increased locomotor activity, while body postural adjustments were used less than expected. Within the thermoneutral zone, back rest (tucking the bill under body feathers) and unipedal (standing on one leg) were preferred by dunlins, probably as being more comfortable for resting. At temperatures above the thermoneutral zone, dunlins were inactive and increased the time of bill exposure and wet-sitting and ultimately panting when challenged with temperatures above 37 °C. Interestingly, above the thermoneutral zone, but below their body temperature, birds with relatively longer bills and legs spent more time exposing them, probably to increase dry heat transfer into the environment. Our findings also highlighted the importance of the availability of wet substrates for minimising heat stress in wetland species.
Significance statement
Recent correlational field studies found support for a relationship between relative bill and leg sizes and thermoregulatory behaviour in birds inhabiting open landscapes. However, experimental data are lacking, and the mechanisms underlying this relationship remain poorly understood. Here, we performed an experiment to model behavioural and physiological responses to ambient temperature change and relative bill and leg sizes in the dunlin Calidris alpina, a long-billed and long-legged shorebird. Additionally, we also examined potential links among metabolic rates, evaporative water loss and relative appendage sizes. Our findings showed a strong experimental relationship between behavioural and physiological responses and ambient temperature, as well as a link between appendage size and resting body postures. Our findings also have a conservation message by highlighting that the type of substrate available for roosting is important for minimising heat stress in wetland species.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Maintaining a high body temperature (Tb) within a very narrow range may be a major challenge for endothermic vertebrates when they are confronted with environmental temperatures beyond their thermoneutral zone, i.e., range of ambient temperatures within which endotherms are able to maintain their Tb without increasing metabolic rate and water loss (Boyles et al. 2011; McKechnie et al. 2017). Many birds live in open landscapes where they are regularly exposed to acute external cold and heat loads and as a result, these have evolved specific behavioural and physiological responses to cope with these thermal challenges (Luskick et al. 1978; Brodsky and Weatherhead 1984; Tieleman et al. 2002; Amat and Masero 2004; Gutiérrez et al. 2015; González-Medina et al. 2020). Groups such as shorebirds, waterfowl and gulls rely on exposed intertidal areas for foraging. However, at high tide, they are forced to move to alternative sites, which are usually open, non-vegetated beaches or mudflats near the tide line (e.g., Rogers et al. 2006), where they rest for long periods of time while waiting for the next low-tide period (e.g., Rosa et al. 2006). To minimise the high metabolic costs associated with resting in these open habitats and adjust the pattern of heat transfer between their bodies and the environment, they can alter both the unfeathered appendage area exposed to the surrounding air and their surface-to-volume ratio (Brodsky and Weatherhead 1984; Wiersma and Piersma 1994; Ryeland et al. 2017, 2019, 2021). Other (non-mutually exclusive) behaviours include selecting microhabitats that maximise metabolic efficiency at high temperatures, such as wet substrates (Walsberg 1993; Wolf and Walsberg 1996; van de Ven et al. 2019; Ryeland et al. 2021). In addition to these behavioural adjustments, these species may rely on metabolically costly processes such as shivering, panting or gular fluttering to regulate their thermal balance in challenging environmental temperatures (Bartholomew et al. 1968; Amat and Masero 2004; du Plessis et al. 2012).
Unfeathered bills, legs and feet are bird appendages that are highly vascularized, and their potential roles in regulating thermal balance are well recognised (e.g., Steen and Steen 1965; Hagan and Heath 1980; Tattersall et al. 2009, 2017; Carr and Lima 2012; Cabello-Vergel et al. 2021; Playà-Montmany et al. 2021). Appendage size may vary among populations of the same bird species according to the thermal environment, and it has been suggested, for example, that a larger bill surface area could be an adaptation to increase dry heat loss under hot climatic conditions (Greenberg et al. 2012). However, these appendages may also be a source of heat gain for birds living in extremely hot environments, where the environmental temperature can approach or surpass the Tb (Greenberg and Danner 2012; Gardner et al. 2016; Ryeland et al. 2017), or of heat loss when exposed to cold (Hagan and Heath 1980; Tattersall et al. 2009; Greenberg and Danner 2012; Gardner et al. 2016), although these aspects of bird thermoregulation are less-known (Gardner et al. 2016; van de Ven et al. 2016; Ryeland et al. 2017, 2019; Benham and Bowie 2021; Playà-Montmany et al. 2021).
In the case of shorebirds (Charadrii), the role of these appendages as ‘thermal windows’ for dry heat exchange may be particularly important, since these birds generally have large relative bill and leg sizes. Thus, for example, shorebirds with smaller legs expend less energy to maintain homeothermy in cold environments than individuals with longer leg lengths (Cartar and Guy Morrison 2005). Within this context, Ryeland et al. (2017, 2019, 2021) found that resting body postures of shorebirds can be mediated by bill and leg sizes. These studies supported back rest (bill tucked within the back plumage) and unipedal (standing on one leg) postures at high tide as effective strategies for reducing heat loss at low temperatures, while sitting in wet substrates favouring body heat loss in hot conditions. They also found evidence for the use of back rest as a strategy to minimise heat gain through the bill at the examined upper-temperature limits (Ryeland et al. 2017, 2019). In contrast, they did not find support for the use of unipedal posture as a strategy to insulate against heat gain, although its use at extremely high environmental temperature values cannot be disregarded (Ryeland et al. 2017, 2019). In this framework, experimental data are needed to test the correlations that predict bird postural adjustments according to ambient temperature (Ta).
Here, we experimentally exposed a migratory shorebird with a relatively large bill, and leg size, the dunlin Calidris alpina, to a wide range of Ta values, in order to model behavioural and physiological responses to environmental temperature change in relation to relative bill and leg size while resting. As birds adjust body posture, they adopt heat-dissipating/conservating behaviours to maintain thermal homeostasis (Ryeland et al. 2017, 2019, 2021; Pavlovic et al. 2019). We predicted that (i) dunlins would reduce the exposed unfeathered area to minimise body heat loss and body heat gain under cold and extremely hot conditions (where Ta approaches Tb), respectively; and (ii) dunlins would initiate metabolically and hygric costly processes, such as panting, only in extremely hot environments. Furthermore, it is well established that one of the strategies used to dissipate body heat and minimise evaporative water loss (EWL) in endotherms is to reduce locomotor activity (Wolf 2000; Thompson et al. 2018). We also predicted, therefore, that (iii) dunlins would decrease their locomotor activity as Ta increased to decrease body heat production. Last, we measured the resting metabolic rate (RMR) and EWL of dunlins at different Ta values to assess the potential role of relative bill and tarsi sizes in explaining variations in both rates. EWL is a metabolically costly process, so if the appendage surface area can be used as a radiator to expel “dry” heat under thermally stressful conditions (Greenberg et al. 2012), we expected that individuals with larger relative bill and tarsi sizes would consume less oxygen to maintain homeothermy when Ta exceeds their thermoneutral zone but below their Tb (Playà-Montmany et al. 2021).
Materials and methods
Bird capture and maintenance
Wintering adult dunlins were caught using mist nets in Extremadura’s rice fields, in southwest Spain (39°01′N, 5°58′W), which is a key area for many nonbreeding waterbirds (Masero et al. 2011; Navedo et al. 2012). After capture, birds were individually colour-ringed to facilitate further identification and were weighed (± 0.01 g). The bill length, width and depth (both measures taken at the anterior edge of the nostrils), tarsus length, width and depth (both taken at the middle of the tarsus) were measured with a digital calliper (± 0.01 mm), while the wing length (flattened and straightened length) was measured using a wing ruler (± 0.5 mm). The 11 individuals showing the highest variability in appendage size (among the 40 individuals captured) were selected for this study (bill length CV = 8.29%; tarsus length CV = 4.27%) and transported to the bird facilities at the University of Extremadura (e.g., Gutiérrez et al. 2011). There, the birds were housed in an environmentally controlled room (320 cm × 260 cm × 255 cm), where they were exposed to fluorescent lighting in a 14 h light:10 h dark cycle starting at 7:00 AM. The daylight period was divided into high-tide (from 2:30 to 7:30 PM) and low-tide (the remaining light hours) periods simulating a diurnal tide through food availability. Live fly larvae Protophormia terraenovae and commercial pellets (Dibaq-Diproteq) were available ad libitum in several trays during the “low tide” period, and all food was removed during the “high tide” period (see details below).
The room temperature was set at 10 °C and 8 °C during the day and night, respectively, throughout the acclimation period (5 weeks), based on the average environmental temperature in the Extremadura’s rice fields during catching. A large pool (80 cm × 260 cm, 3 cm deep) provided fresh water ad libitum for bathing and drinking, and the rest of the ground surface was covered with wet sand. The room was equipped with a one-way window to allow vision through the window only by the observer.
All birds completed the experiment, and all of them were released in good health in the area of capture.
Metabolic measurement protocol
Following the acclimation period, the RMR and EWL were measured at 10 °C, 25 °C and 35 °C during the “high tide” simulation period after 4 h of fasting to ensure that the birds were in a postabsorptive state (see González-Medina et al. 2020). The dunlins were weighed (± 0.1 g) before and after the metabolic measurements, and the mean body mass (Mb) was used in the analyses. Tb was measured prior to and following metabolic trials by using a calibrated laboratory thermometer (P700, Dostmann electronic GmbH, Wertheim, Germany) attached to a copper–constantan thermocouple inserted 1 cm into the cloaca. A maximum of six individuals were measured at a time. Individuals were randomly exposed to 10 °C or 25 °C using a flow rate of 1000 ml min−1 during three trials of 110 min each. Birds were exposed at 35 °C during two trials of 65 min each, and the flow rate was increased up to 3000 ml min−1 to ensure that individuals remained calm under the hot conditions (Whitfield et al. 2015), which was verified by the use of infrared cameras.
Gas exchange measurements
Oxygen consumption (VO2) and EWL were measured using an open flow-through respirometry system (Sable Systems) following González-Medina et al. (2020). Briefly, each post-absorptive individual was placed in a polypropylene metabolic chamber (effective volume = 3.9 L) inside a compressor-cooled incubator (Memmerts GmbH, Germany). An identical, but empty, chamber was used for baseline measurements. The floor of each chamber was covered with a 1 cm mineral oil layer to avoid evaporation from excreta and equipped with a wire mesh platform suspended 3 cm above this oil layer to allow individuals to rest without touching the oil. Dry air (< 1 kPa WVP) was pumped into the metabolic chamber at a rate of 1000 or 3000 ml min−1 using a mass flow controller (MFS, Sable Systems International) (95% washout time: 4–12 min). Low humidity levels were maintained during trials (0.7–0.9 kPa at the highest Ta). The water vapour density (μg ml−1) and O2 concentration (%) in the excurrent airstream were analysed using an H2O analyser (RH300, Sable Systems) and an O2 analyser (FC-10 Oxygen Analyser, Sable Systems), respectively. Before each trial, both gas analysers were zeroed using nitrogen and spanned using the O2 dilution technique for the H2O analyser (Lighton 2008) and incoming air (20.95% O2) for the O2 analyser. The RMR and EWL were estimated as the 2 min the most stable values at each temperature using Eqs. 10.2 and 10.9, respectively, from Lighton (2008) with a respiratory quotient of 0.70, assuming lipid and protein catabolism (Kvist and Lindström 2001). Expedata software (version 1.9.14, Sable Systems, Las Vegas, NV, USA) was used to control the multiplexer, record data and process data for analyses.
Experimental temperature treatment
Behavioural observations were conducted 2 weeks after the respirometry measurements to ensure that individuals were re-habituated to captivity and stayed calm. Birds were exposed to each experimental Ta (10 °C, 15 °C, 20 °C, 25 °C, 30 °C, 35 °C and 38 °C) during a single daylight period and on consecutive days (a steady 5–3 °C in the last step–stepwise increase of experimental Ta starting at 10 °C and ending at 38 °C). Each experimental Ta was set at 7:00 AM, i.e., 7.5 h before the resting period, and ended at 9:00 PM. The room temperature at night was kept 5 °C lower than the experimental temperature tested the following daylight period. There were variations between the experimental temperature set for the room and the temperature that the dunlins experienced near the ground surface. Thus, as an approximation of the temperature experienced by birds on the ground, we measured the air temperature at dunlin height, i.e., 2 cm above water and 5 cm above ground level, every 2 min using HOBO Micro Station Data Loggers H21-002 (Onset Computer Corporation, Bourne, Massachusetts, USA). We also recorded the water temperature (°C; 1 cm upper layer) and wet sand temperature (°C; 1 cm upper layer). The mean Ta above water and wet sand differed by 0.57 ± 0.15 °C (mean ± SE) on average; thus, the mean value was used in successive analyses (see below). The relative humidity ranged from 75 to 90% throughout the observational trials, and the birds did not experience air movement.
Behavioural data collection
Focal behavioural observations were conducted during the high-tide period. These observations started at 3:30 PM, i.e., 1 h after the food was removed, to allow the birds time to recover from the disturbance caused by entering the room. Each individual was observed continuously during a period of 5 min every 20 min. This leaded to a total of four observation bouts of 5 min each per individual at each Ta, and the total proportion of each observation bout representing each body posture was calculated. Observations were conducted by a single researcher (NPM) through the one-way window using 10 × 42 binoculars. The order of observation of focal individuals was randomly chosen at the beginning of the observation period and was maintained during the following observations. It was not possible to record data blind because our study involved following a focal individual.
Although shorebirds usually rest during the high-tide period (no food available), individuals can be active, showing locomotion (walking) and preening behaviours. We considered that an individual was resting once it remained inactive for > 60 s. In each resting posture, we noted whether the bird was standing on one leg, standing on both legs or sitting. The bill position was noted as back rest or not. We also noted panting behaviour (defined as gaping when breathing; Smit et al. 2016) (bill open for ≥ 5 s, see Oswald et al. 2019), as well as the substrate on which the dunlins were resting (wet sand or water). We quantified the times that each individual was engaged in each behaviour and located on each substrate.
Data analysis
Bill and tarsus measurements were converted to surface area estimates following Greenberg et al. (2012). We used equations to approximate the bill surface area as an elliptical cone and the tarsi surface area as an elliptical cylinder (see equations in Greenberg et al. 2012). The coefficients of variation for bill surface area and tarsi surface area were 13.13% and 6.32%, respectively.
We used the proportion of time per observation period spent resting on each body posture as the response variables. Factors influencing these response variables were assessed by fitting generalised linear mixed models (GLMMs) with a binomial response and logit-link function (fitted by the maximum likelihood with Laplace approximation). This calculation treated each minute of observation as a “presence” (for example, a bird in a back rest posture) or “absence” (a bird with the bill exposed) (see Ryeland et al. 2017, 2019, 2021). We modelled each resting body posture by including Ta as a covariate, appendage size, and the interaction between Ta and appendage size as fixed factors, body mass (Mb) to control for body size across individuals, and bird identity as a random factor to account for repeat observations of birds. The interaction term was included in the models to assess whether dunlins with different appendage size might show different behavioural responses across the range of temperatures. The quadratic term of Ta was also included in models where preliminary analyses and previous studies (Ferns 1992; Ryeland et al. 2021) showed a quadratic relation between Ta and body posture. To limit model overfitting, and for comparative purposes with previous studies (Ryeland et al. 2017, 2019, 2021), the back rest model only included as appendage size the bill surface area, whereas unipedal and sitting models only included tarsi surface area. Appendage sizes and Mb were log-10 transformed and all explanatory variables were standardised prior to analysis.
We checked model assumptions using the DHARMa package in R (Hartig 2020). Observation bout was also included in the models as a random factor to absorb additional variance in the response variable (Harrison 2014), as there was some evidence of overdispersion (dispersion parameter > 1.5).
The influence of Ta on the proportion of time that birds were active during the high tide period was also examined by performing a GLMM with a binomial response and logit-link function. We included the same predictors, excluding appendage surface areas. Mb was included in this analysis because previous studies showed that it can influence behavioural thermoregulation (Ryeland et al. 2017, 2019, 2021; Pattinson et al. 2020). Panting was exclusively performed at 38 °C and occurred rarely (see results), so this behaviour was not modelled.
The potential relationships between physiological thermoregulatory and morphological traits at 10 °C, 25 °C and 35 °C were analysed by fitting GLMMs, where RMR (ml O2 min−1) or EWL (mg h−1) was the response variables, and Ta, appendage size and the interaction between Ta and appendage size were included as covariates. Individual identity was included as a random factor and Mb as a fixed factor. We ran separately (for the reasons stated above) a model with bill surface area and a model with tarsi surface area as predictors.
Potential collinearity among predictor variables in the global models was evaluated by calculating the variance inflation factor (VIF) using the “car” package (Fox and Weisberg 2019). Variables were only removed if VIF were > 5 (Zuur et al. 2010). We first built global models using the ‘lme4’ package (Bates et al. 2014) for each analysis. Model selection was performed based on the Akaike information criterion for small sample sizes (AICc) to identify the top model(s) (all models with ΔAICc < 2) and AICc weights (wi) to further distinguish between the top models (Burnham and Anderson 2002). We used the “dredge” function in the R package MuMIn (Barton 2018) for this procedure. For cases in which more than one model had ΔAICc < 2 but wi < 0.9 (Burnham and Anderson 2002), we performed model averaging of parameter estimates (Grueber et al. 2011). A predictor was considered significant when the 95% confidence interval (CI) for the estimated coefficient did not overlap zero. Statistical analyses were conducted in R 4.0.2 (R Core Team 2014), and all figures were produced using the R package “ggplot2” (Wickham 2016). Data are presented as mean ± SE.
Results
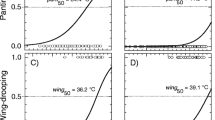
Overall, Ta significantly predicted all behaviours (Table 1). Birds increased their time spent in back rest and standing on one leg with increasing Ta values until they were exposed from 23 to 26 °C. From this temperature range upwards, the use of both resting body postures decreased (Fig. 1). Back rest and unipedal behaviours across the range of temperatures also were influenced by bill surface area and tarsi surface area, respectively (Tables 1, 2). Individuals with relatively smaller bill sizes displayed back rest behaviour more frequently than those with larger bill sizes at high Ta, whereas dunlins with smaller tarsi sizes, relative to body size, tended to display a greater use of unipedal posture than individuals with larger tarsi sizes at such high Ta (Fig. 1).
Model predictions for the percentage (%) of the resting time (simulated high tide) that dunlins spent in a back rest (tucking the bill under body feathers), b unipedal (standing on one leg), c sitting in wet sand and d locomotor activity in relation to environmental temperatures (°C). The shaded areas show the 95% confidence interval (CI) around the predicted values. Bill and tarsi data were back-log transformed, and grouped according to quartiles (lower 25%, median 50%, and upper 25%) for representation purposes only
Sitting and locomotor activity were only predicted by both the linear and quadratic terms of Ta (Tables 1, 2). Sitting was mainly used above 30 °C (30.67 ± 0.03 °C at the ground level), after which point its use increased linearly, achieving 100% at 37 °C (36.52 ± 0.23 °C at the ground level) (Fig. 1). Locomotor activity during the resting period strongly decreased from 20 °C (22.03 ± 0.04 °C at the ground level) upwards (Fig. 1), with birds remaining active less than 5% of time above 35 °C (34.81 ± 0.14 °C at the ground level). Panting occurred rarely (it was only observed in a few individuals and in a few occasions) and only when the room temperature was set to the maximum temperature (37 °C; 36.52 ± 0.23 °C at the ground level) and while sitting.
Variations in RMR and EWL were not explained by the bill or tarsi surface area. The best model for RMR included Ta and Mb, and both predictors had a significant effect on RMR (Tables 1, 2). The best-fitting model explaining EWL variation only included Mb (Table 1), but it was not a significant predictor (Table 2).
Discussion
Thermoregulation plays a major role in organism homeostasis. When exposed to extreme Ta, endotherms adjust body heat production and heat interchange through behavioural and physiological mechanisms. Among the behavioural strategies used to dissipate body heat at high Ta, the experimental data showed that dunlins dealt with high Ta values during the daily resting period by using specific body postures. Our study also suggested that dunlins minimised body heat production at high Ta values by decreasing locomotor activity. However, during cold exposure, dunlins only seemed to rely on heat production by maintaining high levels of locomotor activity (our model predicted dunlins to spend > 60% of time active in cold conditions). We also found a role of unfeathered appendage size in explaining body posture adjustments, but not in RMR and EWL variation across temperatures.
Dunlins, like other birds inhabiting open habitats at temperate latitudes, experience cold winters with commonly high wind speeds, the latter of which greatly contributes to increased body heat loss by forced convection (Wiersma and Piersma 1994). Wintering in temperate latitudes should, therefore, impose the need to implement behavioural and physiological strategies to maintain Tb (e.g., Wolf 2000; Ryeland et al. 2017, 2019, 2021; Cooper et al. 2019). Shorebirds have previously been shown to consistently display body postural adjustments to minimise body heat loss at low Ta values (see Ryeland et al. 2017, 2019). However, we found that dunlins used back rest and unipedal resting less than expected when exposed to temperatures below the lower critical value of the thermoneutral zone (19.8 °C according to Kelly and Weathers 2002). In the absence of wind near the ground level, captive dunlins did not experience heat loss by forced convection (wind), and they seemed to be able to cope with lower temperatures (12 °C on average) mainly by increasing heat production through locomotor activity. Most shorebirds roost communally in flocks ranging from tens to thousands of birds, and in windy cold environments they can reduce heat loss by flocking and facing the wind (Wiersma and Piersma 1994). However, it has also been shown that shorebirds such as the red knot Calidris canutus can compensate for thermoregulation costs at low temperatures by using heat generated as a by-product of walking or running (Bruinzeel and Piersma 1998), and presumably, flight would have a similar or stronger effect (Rogers et al. 2006). Our findings confirmed this large compensation to maintaining homeothermy by using locomotor activity in cold and windless environments. Last, when remaining inactive in cold conditions, dunlins barely used unipedal resting or sitting, suggesting bipedal standing as the main body posture adopted under cold conditions.
At temperatures within thermoneutrality (e.g., from 20 to 30 °C), no additional energy or water is allocated for Tb maintenance (Wiersma and Piersma 1994); thus, non-thermoregulatory responses should be expected within this range of Ta. We found dunlins standing on one leg and tucking the bill while exposed to thermoneutral conditions. Back rest is commonly adopted by several shorebird species, including dunlins, while resting or sleeping at high tide (e.g., Klaassen 1990; Dominguez 2003). Previous studies conducted with the flamingo Phoenicopterus ruber, for example, showed unipedal resting to be a less energetically demanding posture that favours muscle relaxation and body balance (see Chang and Ting 2017). Thus, within the thermoneutral zone, a back rest posture and unipedal resting could have been selected by dunlins as being more comfortable for resting (Klaassen 1990; Dominguez 2003; Chang and Ting 2017).
At high Ta values (> 30 °C), dunlins increased the time of bill exposure, as this may favour body heat dissipation by radiation. This result is analogous to that found by Ryeland et al. (2017), in which shorebirds reduced their back rest behaviour at high Ta values to favour heat flux into the environment. At hot conditions, but below their Tb (39–40 °C; see also González-Medina et al. 2020), we also found that the proportion of time spent resting on back rest, and to lesser extent on unipedal, was related to the relative size of bill and legs, respectively. Dunlins with the largest relative bill sizes clearly spent more time with their bills exposed compared to dunlins with the smallest relative bills, supporting dunlins adjusted their bill postures to increase dry heat transfer into the environment at high temperatures.
In addition to adjusting their bill position, as Ta increased, dunlins remained sitting in wet sand most of the time, which is a previously observed behaviour known as ‘wet-sitting’ (see Ryeland et al. 2021). Previous studies with wild shorebirds highlighted the importance of sitting in wet substrates under heat stress, as this is an effective mechanism to avoid hyperthermia (Battley et al. 2003; Rogers et al. 2006; Ryeland et al. 2021). Ryeland et al. (2021), for example, showed that several coastal shorebird species spent more time wet-sitting at high tide when roosting at ~ 30 °C. By selecting this microhabitat and body posture, shorebirds may favour body heat loss by conduction across the leg surface and the lower body, as both surfaces directly contact the wet substrate (e.g., Ferns 1992; Ryeland et al. 2021). Sitting can also be less energetically expensive than standing, contributing to the balance of energy budgets under these conditions (Tickle et al. 2012; Pavlovic et al. 2019). The maximum temperature registered on the surface of the wet sand inside the room was ~ 37 °C, which was lower than the core Tb (~ 39 °C) measured in dunlins when exposed to 35 °C inside the metabolic chambers. Therefore, the thermal gradient (Ta-Tb) between the wet substrate and the body would allow dry heat transfer from the body to the wet sand when sitting, reducing the body heat burden of the birds (Rogers et al. 2006; Ryeland et al. 2021). At the highest Ta value, this gradient should have been minimised, and a few dunlins additionally dissipated heat by using panting (forced evaporative water loss) (Amat and Masero 2004; McKechnie and Wolf 2019) while wet-sitting. Models predicting panting/gular flutter behaviours in arid-zone bird species found that panting (behavioural response present in 50% of observations) occurred at ~ 37 °C and ~ 41 °C in drinking and nondrinking species, respectively (Smit et al. 2016). Our experimental birds had access to freshwater ad libitum, and they started to use this avian respiratory heat dissipation behaviour at ~ 37 °C, supporting model predictions for drinking species. Finally, we did not observe heat-avoidance behaviours such as ptiloerection (raising of the ‘back’ feathers) and wing-dropping (defined as holding the wings away from the body), which have both been described in small-sized wild shorebirds (Battley et al. 2003; Amat and Masero 2004). Feather erecting may increase convective or evaporative cooling (Battley et al. 2003), while wing-dropping could be a mechanism to increase the surface area of the body to enhance radiative and convective dry heat loss by exposing thermal windows under the wing (Smit et al. 2016). The absence of ground-level wind inside the room could explain why dunlins did not use both heat-avoidance behaviours.
Inactivity is a response shared among a large fraction of the world’s avifauna to minimise the impacts of heat stress (e.g., Davies 1982; Wolf 2000; Silva et al. 2015; Cooper et al. 2019), which we know mainly from studies performed in passerine species from arid habitats and Mediterranean climates (Kelly et al. 2004; Thompson et al. 2018; Cooper et al. 2019). However, although this thermoregulatory response to thermal stress has been largely overlooked in non-passerine species such as shorebirds, our results indicated that changes in locomotor activity are a common response used to overcome thermoregulatory challenges, as dunlins adjust locomotor activity to increase or minimise metabolic heat production at low and high Ta values, respectively. Inevitably, some of these thermoregulatory behaviours involve trade-offs with other fitness-improving activities, such as antipredator behaviours or foraging (du Plessis et al. 2012). For example, the time allocated to sitting cannot be spent being alert to predators (Ryeland et al. 2017, 2019; Timmis et al. 2022), which makes sitting individuals much more vulnerable (Ryeland et al. 2017, 2019; Timmis et al. 2022). These constraints associated with thermal behaviours can be accentuated by the current global warming, which forces birds to spend longer periods of time adopting behavioural strategies to alleviate thermal stress.
We found no significant influence of relative appendage sizes on either RMR or EWL. The low intraspecific variation in bill and tarsi sizes might explain why they did not influence both physiological traits. The studies that found large effects on bill and tarsus lengths on shorebirds thermoregulation involved several species that displayed enormous variation in morphological trait sizes (Ryeland et al. 2017, 2019, 2021). For example, the CV obtained by Ryeland et al. (2019) for tarsus length using many shorebird species was approximately 53%, while in our dunlin sample it was ~ 6%. Therefore, this circumstance could explain the lack of explanatory power of the appendage sizes in explaining variation in RMR and EWL.
In conclusion, we provided a better understanding of the physiological and behavioural responses to thermal stress in a small bird species inhabiting open landscapes. Our findings indicated that captive dunlins adopted for behavioural strategies that included body postural adjustments and locomotor activity to defend Tb under challenging Ta values. In hot environmental conditions, dunlins used thermoregulatory behaviours that strongly favoured conductive (sitting) and radiative (bill exposition) dry heat transfer from the body to the surroundings, while in cold environments, individuals remained active for longer periods of time to favour metabolic heat production. Our study showed that panting was only performed for a few individuals at the highest Ta values, supporting that the birds favoured behavioural responses over this costly physiological response. Lastly, previous studies (Ryeland et al. 2021) supported the importance of wetland margin substrate in the thermoregulation of several shorebird species, as the time spent sitting was significantly mediated by the substrate upon which they roosted. Our experimental findings support this positive role of the substrate type (in our case wet sand) in minimising heat stress in shorebirds, which is remarkably relevant to shorebirds, as they face a reduction in wetland habitat availability along with increasing Ta values due to global warming (Ryeland et al. 2021).
Data availability
Data are available as supplementary material.
References
Amat JA, Masero JA (2004) How Kentish plovers, Charadrius alexandrinus, cope with heat stress during incubation. Behav Ecol Sociobiol 56:26–33
Bartholomew GA, Lasiewski RC, Crawford EC (1968) Patterns of panting and gular flutter in cormorants, pelicans, owls, and doves. Condor 70:31–34
Barton K (2018) MuMIn: multi-model inference. R package version 1.40.4. https://CRAN.R-project.org/package=MuMIn
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. arXiv prepint arXiv 1406.5823
Battley PF, Rogers DI, Piersma T, Koolhaas A (2003) Behavioural evidence for heat-load problems in great knots in tropical Australia fuelling for long-distance flight. Emu 103:97–103
Benham PM, Bowie RC (2021) The influence of spatially heterogeneous anthropogenic change on bill size evolution in a coastal songbird. Evol Appl 14:607–624
Boyles JG, Seebacher F, Smit B, McKechnie AE (2011) Adaptive thermoregulation in endotherms may alter responses to climate change. Integr Comp Biol 51:676–690
Brodsky LM, Weatherhead PJ (1984) Behavioural thermoregulation in wintering black ducks: roosting and resting. Can J Zool 62:1223–1226
Bruinzeel LW, Piersma T (1998) Cost reduction in the cold: heat generated by terrestrial locomotion partly substitutes for thermoregulation costs in Knot Calidris canutus. Ibis 140:323–328
Burnham KP, Anderson DR (2002) Model selection and inference: a practical information-theoretic approach, 2nd edn. Springer-Verlag, New York
Cabello-Vergel J, Soriano-Redondo A, Villegas A, Masero JA, Sánchez-Guzmán JM, Gutiérrez JS (2021) Urohidrosis as an overlooked cooling mechanism in long-legged birds. Sci Rep 11:20018
Carr JM, Lima SL (2012) Heat-conserving postures hinder escape: a thermoregulation–predation trade-off in wintering birds. Behav Ecol 23:434–441
Cartar RV, Guy Morrison RI (2005) Metabolic correlates of leg length in breeding arctic shorebirds: the cost of getting high. J Biogeogr 32:377–382
Chang YH, Ting LH (2017) Mechanical evidence that flamingos can support their body on one leg with little active muscular force. Biol Lett 13:20160948
Cooper CE, Withers PC, Hurley LL, Griffith SC (2019) The field metabolic rate, water turnover, and feeding and drinking behavior of a small avian desert granivore during a summer heatwave. Front Physiol 10:1405
Davies SJJF (1982) Behavioural adaptations of birds to environments where evaporation is high and water is in short supply. Comp Biochem Physiol A 71:557–566
Dominguez J (2003) Sleeping and vigilance in black-tailed godwit. J Ethol 21:57–60
du Plessis KL, Martin RO, Hockey PA, Cunningham SJ, Ridley AR (2012) The costs of keeping cool in a warming world: implications of high temperatures for foraging, thermoregulation and body condition of an arid-zone bird. Glob Change Biol 18:3063–3070
Ferns PN (1992) Thermoregulatory behavior of rock doves roosting in the Negev Desert. J Field Ornithol 63:57–65
Fox J, Weisberg S (2019) An R Companion to Applied Regression, 3rd edn. Sage, Thousand Oaks
Gardner JL, Symonds MR, Joseph L, Ikin K, Stein J, Kruuk LE (2016) Spatial variation in avian bill size is associated with humidity in summer among Australian passerines. Clim Change Resp 3:11
González-Medina E, Cabello-Vergel J, Playà-Montmany N, Villegas A, Parejo M, Abad-Gómez JM, Sánchez-Guzmán JM, Masero JA (2020) Going to sleep with a full belly: thermal substitution by specific dynamic action in shorebirds. Comp Biochem Physiol A 244:110689
Greenberg R, Danner RM (2012) The influence of the California marine layer on bill size in a generalist songbird. Evolution 66:3825–3835
Greenberg R, Cadena V, Danner RM, Tattersall G (2012) Heat loss may explain bill size differences between birds occupying different habitats. PLoS ONE 7:e40933
Grueber CE, Nakagawa S, Laws RJ, Jamieson IG (2011) Multimodel inference in ecology and evolution: challenges and solutions. J Evol Biol 24:699–711
Gutiérrez JS, Masero JA, Abad-Gómez JM, Villegas A, Sánchez-Guzmán JM (2011) Metabolic consequences of overlapping food restriction and cell-mediated immune response in a long-distance migratory shorebird, the little ringed plover Charadrius dubius. J Avian Biol 42:259–265
Gutiérrez JS, Soriano-Redondo A, Dekinga A, Villegas A, Masero JA, Piersma T (2015) How salinity and temperature combine to affect physiological state and performance in red knots with contrasting non-breeding environments. Oecologia 178:1077–1091
Hagan AA, Heath JE (1980) Regulation of heat loss in the duck by vasomotion in the bill. J Therm Biol 5:95–101
Harrison XA (2014) Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ 2:e616
Hartig MF (2020) DHARMa: residual diagnotics for hierarchical (multi-level/mixed) regression models. R package v. 0.2.0. https://www.CRAN.R-project.org/package=DHARMa
Kelly A, Godley BJ, Furness RW (2004) Magpies, Pica pica, at the southern limit of their range actively select their thermal environment at high ambient temperatures. Zool Middle East 32:13–26
Kelly JP, Weathers WW (2002) Effects of feeding time constraints on body mass regulation and energy expenditure in wintering dunlin (Calidris alpina). Behav Ecol 13:766–775
Klaassen M (1990) Short note on the possible occurrence of heat stress in roosting waders on the Banc d’Arguin, Mauritania. Ardea 78:63–65
Kvist A, Lindström Å (2001) Basal metabolic rate in migratory waders: intra-individual, intraspecific, interspecific and seasonal variation. Funct Ecol 15:465–473
Lighton JRB (2008) Measuring metabolic rates: a manual for scientists. Oxford University Press, New York
Luskick S, Battersby B, Kelty M (1978) Behavioral thermoregulation: orientation toward the sun in herring gulls. Science 200:81–83
Masero JA, Santiago-Quesada F, Sánchez-Guzmán JM, Villegas A, Abad-Gómez JM, Lopes RJ, Encarnação V, Corbacho C, Morán R (2011) Long lengths of stay, large numbers, and trends of the black-tailed godwit Limosa limosa in rice fields during spring migration. Bird Conserv Int 21:12–24
McKechnie AE, Gerson AR, McWhorter TJ, Smith EK, Talbot WA, Wolf BO (2017) Avian thermoregulation in the heat: evaporative cooling in five Australian passerines reveals within-order biogeographic variation in heat tolerance. J Exp Biol 220:2436–2444
McKechnie AE, Wolf BO (2019) The physiology of heat tolerance in small endotherms. Physiology 34:302–313
Navedo JG, Masero JA, Sánchez-Guzmán JM, Abad-Gómez JM, Gutiérrez JS, Sansón EG, Villegas A, Costillo E, Corbacho C, Morán R (2012) International importance of Extremadura, Spain, for overwintering migratory dabbling ducks: a role for reservoirs. Bird Conserv Int 22:316–327
Oswald KN, Smit B, Lee AT, Cunningham SJ (2019) Behaviour of an alpine range-restricted species is described by interactions between microsite use and temperature. Anim Behav 157:177–187
Pattinson NB, Thompson ML, Griego M et al (2020) Heat dissipation behaviour of birds in seasonally hot arid-zones: are there global patterns? J Avian Biol 51:e02350
Pavlovic G, Weston MA, Symonds MRE (2019) Morphology and geography predict the use of heat conservation behaviours across birds. Funct Ecol 33:286–296
Playà-Montmany N, González-Medina E, Cabello-Vergel J, Parejo M, Abad-Gómez JM, Sánchez-Guzmán JM, Villegas A, Masero JA (2021) The thermoregulatory role of relative bill and leg surface areas in a Mediterranean population of great tit (Parus major). Ecol Evol 11:15936–15946
Rogers DI, Piersma T, Hassell CJ (2006) Roost availability may constrain shorebird distribution: exploring the energetic costs of roosting and disturbance around a tropical bay. Biol Conserv 133:225–235
Rosa S, Encarnaçao AL, Granadeiro JP, Palmeirim JM (2006) High water roost selection by waders: maximizing feeding opportunities or avoiding predation? Ibis 148:88–97
Ryeland J, Weston MA, Symonds MRE (2017) Bill size mediates behavioural thermoregulation in birds. Funct Ecol 31:885–893
Ryeland J, Weston MA, Symonds MRE (2019) Leg length and temperature determine the use of unipedal roosting in birds. J Avian Biol 50:e02008
Ryeland J, Weston MA, Symonds MRE (2021) The importance of wetland margin microhabitat mosaics; the case of shorebirds and thermoregulation. J Appl Ecol 58:382–391
Silva JP, Catry I, Palmeirim JM, Moreira F (2015) Freezing heat: thermally imposed constraints on the daily activity patterns of a free-ranging grassland bird. Ecosphere 6:1–13
Smit B, Zietsman G, Martin RO, Cunningham SJ, McKechnie AE, Hockey PAR (2016) Behavioural responses to heat in desert birds: implications for predicting vulnerability to climate warming. Clim Change Resp 3:9
Steen I, Steen JB (1965) The importance of the legs in the thermoregulation of birds. Acta Physiol Scand 63:285–291
Tattersall GJ, Andrade DV, Abe AS (2009) Heat exchange from the toucan bill reveals a controllable vascular thermal radiator. Science 325:468–470
Tattersall GJ, Arnaout B, Symonds MRE (2017) The evolution of the avian bill as a thermoregulatory organ. Biol Rev 92:1630–1656
Thompson ML, Cunningham SJ, McKechnie AE (2018) Interspecific variation in avian thermoregulatory patterns and heat dissipation behaviours in a subtropical desert. Physiol Behav 188:311–323
Tickle PG, Nudds RL, Codd JR (2012) Barnacle geese achieve significant energetic savings by changing posture. PLoS ONE 7:e46950
Tieleman BI, Williams JB, Buschur ME (2002) Physiological adjustments to arid and mesic environments in larks (Alaudidae). Physiol Biochem Zool 75:305–313
Timmis AD, Symonds MR, Weston MA (2022) The influence of resting posture and orientation on alertness and escape in shorebirds. J Ornithol 163:509–521
van de Ven TMFN, Martin RO, Vink TJF, McKechnie AE, Cunningham SJ (2016) Regulation of heat exchange across the hornbill beak: functional similarities with toucans? PLoS ONE 11:e0154768
van de Ven TM, McKechnie AE, Cunningham SJ (2019) The costs of keeping cool: behavioural trade-offs between foraging and thermoregulation are associated with significant mass losses in an arid-zone bird. Oecologia 191:205–215
Walsberg GE (1993) Thermal consequences of diurnal microhabitat selection in a small bird. Ornis Scand 24:174–182
Whitfield MC, Smit B, McKechnie AE, Wolf BO (2015) Avian thermoregulation in the heat: scaling of heat tolerance and evaporative cooling capacity in three southern African arid-zone passerines. J Exp Biol 218:1705–1714
Wickham H (2016) Programming with ggplot2. ggplot2. Springer, Cham, pp 241–253
Wiersma P, Piersma IT (1994) Effects of microhabitat, flocking, climate and migratory goal on energy expenditure in the annual cycle of red knots. Condor 96:257–279
Wolf B (2000) Global warming and avian occupancy of hot deserts; a physiological and behavioral perspective. Rev Chil Hist Nat 73:395–400
Wolf BO, Walsberg GE (1996) Thermal effects of radiation and wind on a small bird and implications for microsite selection. Ecology 77:2228–2236
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14
Acknowledgements
We thank the State Agency of Meteorology for giving us access to climate data, the Junta de Extremadura for permissions and Jorge S. Gutiérrez and Andrea Soriano for suggestions in data analysis, and two anonymous reviewers and Associated Editor who improved an earlier version of the manuscript.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. Research funding was provided by the IB16183 project (Junta de Extremadura and European Regional Development Fund), a predoctoral fellowship from Junta de Extremadura and European Social Fund (PD16099) to NPM, grant PEJ2018-003697-P (Ministerio de Ciencia e Innovación, Agencia Estatal de Investigación and European Social Fund) to JCV, and Junta de Extremadura (Consejería de Economía, Ciencia y Agenda Digital) and ERDF through grant GR21081.
Author information
Authors and Affiliations
Contributions
NPM, AV and JAM conceived the idea and designed the study. JMAG, MP and JAM captured the dunlins. NPM and EGM collected the data, and NPM analysed the data. NPM led the writing of the manuscript with the support of JAM and AV. All authors contributed to the revisions and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
All procedures were approved by the bioethical committee of the University of Extremadura, Spain (108/2016), and were conducted under the governmental license CN0032/18/ACA. All applicable national and institutional guidelines for the use of animals were followed.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by K. van Oers
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Playà-Montmany, N., González-Medina, E., Cabello-Vergel, J. et al. Behavioural and physiological responses to experimental temperature changes in a long-billed and long-legged bird: a role for relative appendage size?. Behav Ecol Sociobiol 77, 7 (2023). https://doi.org/10.1007/s00265-022-03280-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03280-9