Abstract
The Great Dismal Swamp, a freshwater forested peatland, has accumulated massive amounts of soil carbon since the postglacial period. Logging and draining have severely altered the hydrology and forest composition, leading to drier soils, accelerated oxidation, and vulnerability to disturbance. The once dominant Atlantic white cedar, cypress, and pocosin forest types are now fragmented, resulting in maple-gum forest communities replacing over half the remaining area. In order to determine the effect of environmental variabes on carbon emissions, this study observes 2 years of CO2 and CH4 soil flux, which will also help inform future management decisions. Soil emissions were measured using opaque, non-permanent chambers set into the soil. As soil moisture increased by 1 unit of soil moisture content, CH4 flux increased by 457 μg CH4–C/m2/h. As soil temperature increased by 1 °C, CO2 emissions increased by 5109 μg CO2–C/m2/h. The area of Atlantic white cedar in the study boundary has an average yearly flux of 8.6 metric tons (t) of carbon from CH4 and 3270 t of carbon from CO2; maple-gum has an average yearly flux of 923 t of carbon from CH4 and 59,843 t of carbon from CO2; pocosin has an average yearly flux of 431 t of carbon from CH4 and 15,899 t of carbon from CO2. Total Cha−1year−1 ranged from 1845 kg of Cha−1year−1 in maple-gum to 2024 kg Cha−1year−1 for Atlantic white cedar. These results show that soil carbon gas flux depends on soil moisture, temperature and forest type, which are affected by anthropogenic activities.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forested peat wetlands store large quantities of carbon in the form of organic biomass (Anderson et al. 2016), largely in the root and soil pool (Powell and Day 1991). Human alteration of wetland hydrology can lead to drying of the soil, which leads to oxidation, changes in plant species composition, altered ecosystem health, increased fire risk, and potentially large emissions of greenhouse gases (GHG) (Reddy et al. 2015). Forest degradation and land use change are important contributors to climate change globally. This study aims to quantify the differential GHG fluxes of carbon from the soil matrices occurring in maple-gum (Acer rubrum and Nyssa sylvatica), Atlantic white cedar (Chamaecyparis thyoides), and pocosin (Pinus serotina) habitats at the Great Dismal Swamp. The main drivers of carbon flux are generally soil and vegetation characteristics, including soil moisture and flooding, wetland or forest type, and soil chemistry; hence, we tested the hypotheses that the GHG flux of carbon differs between soils under maple-gum, Atlantic white cedar, and pocosin habitats, and that carbon flux is dependent on soil temperature and soil moisture. We also explore other possible variables contributing to differences in flux rates in the Great Dismal Swamp.
Some soil respiration has been measured in the Great Dismal Swamp, for example CH4 flux in 1980–1981, where waterlogged soil was found to be a CH4 source and soils during drought acted as a sink, while normally-dry forest soil did not act as a sink; temperature, season, and soil water content were used to determine CH4 flux within a maple-gum site in the Great Dismal Swamp (Harriss et al. 1982). Another study in Pocosin Lakes National Wildlife Refuge, a pocosin habitat relatively near the Great Dismal Swamp, consisting of natural, restored and degraded pocosins, found that phenolics present in the peats, which are found in higher concentrations in shrubs than herbaceous vegetation, protect against peat oxidation during short term droughts, mitigating the increase in CO2 emissions found in sphagnum wetland areas. Phenolics were found to be inversely related to soil respiration, protecting peat in shrub communities during droughts (Wang et al. 2015).
Many studies have quantified carbon flux in forest, peat and wetland soils (Bubier et al. 2003). Different variables affect carbon flux in different ecosystems. In subalpine forests of the Rocky Mountains in the US, leaf area index, soil nitrogen and tree height were found to account for much of the variability in positive total below ground carbon flux (Berryman et al. 2016). In a drained forested peatland in Finland, GHG flux (carbon uptake) was found to depend on season (irradiance and temperature), vapor pressure deficit and water table, but did not correlate with plant community composition or soil micro topography (Lohila et al. 2011). In the wet-dry topics of Australia, GHG soil flux was found to be controlled by soil moisture (Beringer et al. 2013). On a floodplain in the mid-Atlantic region of the US, carbon flux was found to depend on water-filled pore space and mass of deposited mineral sediment, clay fraction and particle size, temperature, pH, and soil redox (Batson et al. 2014). In open water and emergent vegetation, ebullition and diffusion of CH4 and CO2 flux were found to depend on season and wetland structure (McNicol et al. 2017). Different forests and wetlands can also act as a source or a sink. Snow-covered northern wetlands in China were found to act as a source or sink at different times of the year, depending on snow pack density, temperature, and type of wetland (Miao et al. 2012).
In this study, we measured carbon-based GHG emissions (CO2 and CH4) from soils present within three forested wetland habitat types that differ in peat chemistry, carbon density, and peat accretion (Drexler et al. 2017), and hydrologic patterns. We found CO2 and CH4 flux respond to changes in soil temperature and soil moisture as well as forest community type. The Great Dismal Swamp has experienced massive peat loss facilitated not only by chronic oxidation from perennially lower water tables, but also from dry-condition-induced fires that burn through thousands of years of peat deposition in relative short periods of time to reduce elevations, and further promote forest habitat shifts. The objectives of this study are to evaluate the consequence of these shifts on the fluxes of carbon-based GHGs and inform future management decisions.
Methods
Study design and site description
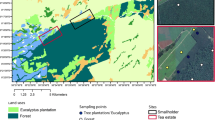
The Great Dismal Swamp is a freshwater forested wetland of over 54,000 ha located in southeastern Virginia and northeastern North Carolina, less than 64 km from the Atlantic coast. Currently, the US Fish and Wildlife Service manages the Great Dismal Swamp National Wildlife Refuge, while the small section in the southeast is managed by North Carolina Dismal Swamp State Park (Fig. 1). Before European settlement, the forested wetland was estimated to occupy over 404,000 ha in extent but has been reduced to its current size through anthropogenic pressures for development and clearing for agriculture (Laderman et al. 1989; Oaks and Whitehead 1979). There are still ~250 km of ditches and roads running through the Great Dismal Swamp (Barrd 2006), making hydrologic restoration a challenge. The native forest types once dominating the Great Dismal Swamp were baldcypress (Taxodium distichum) and Atlantic white cedar (C. thyoides) (Barrd 2006), with baldcypress in the wetter areas and Atlantic white cedar in the slightly higher areas (Brown and Atkinson 1999). Today, Atlantic white cedar and baldcypress still remain (Laing et al. 2011) in remnant populations along with pond pine (P. serotina), but red maple (A. rubrum), black gum (N. sylvatica) and sweet bay (Liquidambar styraciflua) (referred to as “maple-gum”) have become a major part of the contemporary forest composition, comprising over 60% of the current Great Dismal Swamp extent (Laderman et al. 1989) due to their resilience and ability to compete with other species in the new, drained conditions. Pine pocosin (hereafter, pocosin) is a type of fire-adapted wetland (Parthum et al. 2017) of the Atlantic coastal plain characterized by nutrient poor, often saturated (Kim et al. 2017) peat soils inhabited by pond pine (P. serotina) and loblolly pine (Pinus taeda), and a mix of dense shrubs (e.g., sweet pepperbush [Clethra alnifolia], inkberry [Ilex glabra] and greenbriar [Smilax rotundifolia]). Here, we focus on Atlantic white cedar and pocosin which are vulnerable to alternative succession by maple-gum.
Map showing the Great Dismal Swamp with forest types as determined by The Natural Communities of Virginia, the locations of the nine study sites, and the location within the mid-Atlantic of the US (Fleming et al. 2001)
Data were collected at three sites in each of the three forest types, using four sampling plots within each site, for a total of 9 sites and 36 sampling plots. Sites were chosen as good representatives of the target forest types in the Great Dismal Swamp, within accessibility constraints, based on mix of species within each forest type (maple-gum, Atlantic white cedar, and pocosin) (Jenkins et al. 2001), canopy cover, inundation and moisture regime, and disturbance. To determine the rate of soil CO2 and CH4 flux in the Great Dismal Swamp and the driving factors for these rates, two years of monthly CO2 and CH4 measurements were taken, as well as soil temperature at 10 cm and ambient air temperature. Gas fluxes (CO2 and CH4) were measured from chambers (adapted from Krauss and Whitbeck (2012) for use with tubes instead of syringes) set onto permanent bases, which were installed in each sampling plot, for a total of 12 chamber bases per forest type. Each site was visited once per month, with all sites visited within a 3–4 day period, and each base was sampled for 10 min each time. We also took monthly site-level moisture measurements of litter, soil of 0–5 cm depth, and soil of 5–10 cm depths within each site. Soil temperature at 10 cm was recorded from all sites over 2+ years using continuous loggers (model HOBO Pro v.2, Onset Computer Corp., Bourne, MA, USA) as well as manually during each sampling.
We minimized impact on the study site as much as possible by not walking near the chambers except to place the equipment, and by placing equipment from a bench to distribute the weight of the researcher. Any ebullition events under the chambers during the sample time were captured but none were noted. We also used an in situ portable cavity ring-down spectroscopy analyzer (Los Gatos Research Ultra Portable Greenhouse Gas Analyzer [UGGA], San Jose, California) to detect smaller concentrations of CH4 than the traditional gas chromatography technique (Christiansen et al. 2015), since CH4 is found in much lower concentrations than CO2 at these sites. Using the spectroscopy analyzer also allows for a short sampling time, which reduces the buildup of pressure inside the chamber which can reduce the diffusive flux of the system (Parkin et al. 2012) but still provides hundreds of data points at the sampling rate of over one measurement per second, enough to establish the flux rate.
Gas flux measurements and ancillary data
Ancillary data
Each gas flux measuring chamber base was set and left for the duration of the study. Chamber bases measure 29.4 cm by 29.4 cm (864 cm2 area), and are 12.7 cm deep composed of straight sides forming an open top with a square trough in which to set the chamber top, which added 30.5 cm to the chamber height during sampling. The volume of the chamber when set into the base is 27.22 liters and was adjusted for reductions in volume due to surface water when flooded as necessary. This trough was filled with water before sampling so that the opaque chamber top and base form an air tight seal. Gas exchange was also blocked from below the chamber base by insertion to 12 cm into the soil, which was deep enough to avoid leakage during the sampling time (Rochette et al. 2008). While the chamber bases were permanent, the chamber tops were placed in the bases by hand during sampling only.
The UGGA intakes gas from a tube connected to the chamber top and returns it to the chamber after it is run through a cavity enhanced laser spectrometer. The UGGA sampling rate is once every 0.975 s and measurements are recorded in parts per million for both CO2 and CH4. The chamber volume was calculated by measuring the inner sides and top of the chamber, accounting for compaction and water volume. With no standing water, the chamber volume is ~27 liters. Water depth at each chamber was measured if applicable and used to adjust chamber volume. Tree age and diameter at breast height were measured once using tree cores and circumference.
The volume of gas in each chamber was sampled during the daytime each month by placing the chamber top into the bottom trough with the UGGA running. The chambers were left in place for 10 min and then removed and the analyzer was allowed to return to baseline by remaining open to the air for 4 min in between each sample. Air temperature sometimes varied by several degrees over the sampling period at each site. All measurements were during the day, between sun rise and sun set.
In order to minimize disturbance of the soil caused by the sampling process (Winton and Richardson 2016), wide footed stools were placed near each point before sampling and the chamber top was lowered onto each base from a plank set between the stools. This mitigates against negative measurement impacts (c.f., Winton et al. 2017). Leaving the chambers for one month—or greater in our case—before sampling also avoids errors based on soil disturbance from insertion (Muñoz et al. 2011). The analyzer was calibrated and maintained according to the manufacturer’s instructions.
Statistical analysis
Statistical analysis includes basic statistics such as average emissions over time and by site, as well as linear regression which shows the relationship between the measured variables and gas flux, and a paired two sample t-test to show whether the forest classes statistically represent the same population based on measured variables. Ecosystem respiration is determined by calculating the slope of the increasing concentration of gas inside the chamber in ppm/0.975 s and converting that into CO2 or CH4 (and then into carbon) per square meter of ground surface. Unless otherwise noted, yearly data from this study is made up of measurements throughout all months of the year sampled, averaged and added together so that seasonal fluctuations are represented. Regression shows the relationships between the measured variables and the gas flux.
The paired two sample t-test of CO2 and CH4 fluxes in each forest type is used to show if two means come from the same statistical population. Linear regression is used to show the relationships between measured variables and gas flux.
Results
Results include descriptive statistics showing the site characteristics and differences, linear regression showing variable effects on emissions, and a paired t-test to determine whether sample means indicate the same or different populations. CO2 net emissions, both averaged across all measurements and individually at the different sites, varied in response to the changing seasons and temperatures, as well as by forest type. Average CO2 net emissions in the peak growing season (April–September over 120.95 mg CO2–C/m2/h) was almost three times higher than CO2 flux in October–December (42.84 mg CO2–C/m2/h) and January–March (34.76 mg CO2–C/m2/h). CH4 flux also varied throughout the year but in response to soil moisture and other variables as well as season and temperature (Tables 1 and 2). See Gutenberg and Sleeter 2018.
The CO2 net emissions measurement distribution is positively skewed, with the greatest number of values between 7000 and 200,000 μg CO2–C/m2/h, with values ranging up to 567,897 μg CO2–C/m2/h. The distribution of CH4 flux measurements is also positively skewed, but to a greater extent with many very high outliers, with the greatest number of flux values between 0 and 50 μg CH4–C/m2/h, and a right tail of values ranging up to 56,686 μg CH4–C/m2/h. CO2 net emissions showed a more consistent range of values, whereas range of CH4 flux measurements varied by site (Table 1).
Linear regression shows that much of the variation in CO2 emissions is explained by changes in air and soil temperature and soil moisture from 0 to 5 cm. See Table 2 for actual results from all sites. Much of the variation in CH4 flux is explained by variation in water depth, soil moisture from 0 to 5 cm, and soil moisture from 5 to 10 cm, but less of the overall variation in CH4 flux is accounted for.
CO2 emissions across all the sites show a statistically significant relationship with air temperature (P < 0.001) and soil temperature (P < 0.001), as well as a relationship with soil moisture content (SMC) in the top 5 cm of soil (P = 0.074) and in the litter layer (P = 0.081) that is significant at a 90% confidence level. The slopes and R2 values for CO2 net emissions plotted against soil temperature varied by forest type. Atlantic white cedar sites had the best fit (R2 = 0.6419) followed by pocosin (R2 = 0.5588) and then maple-gum (R2 = 0.488). Increasing soil temperature caused a generally greater increase in CO2 net emissions on Atlantic white cedar sites than in pocosin or maple-gum sites, and a generally greater increase in CO2 net emissions in pocosin sites than in maple-gum sites (Atlantic white cedar slope 10,320 μg C/m2/h/°C, pocosin slope 8183 μg C/m2/h/°C, maple-gum slope 6693 μg C/m2/h/°C).
CH4 flux shows a statistically significant relationship with SMC in the top 5 cm of soil (P = 0.027), and a relationship with water depth (P = 0.090) which is statistically significant at a 90% confidence level. Average age of trees and average diameter of trees at breast height sampled at the sites do not show a significant relationship to CH4 (P-values 0.209 and 0.279, respectively) or CO2 (P-values 0.225 and 0.901, respectively) flux. Measuring 0–10 cm SMC was less helpful in a predictive sense than measuring 0–5 in both cases, suggesting a tight connection between gaseous carbon fluxes from the soil and condition in very upper soil horizon only. The relationship between CH4 flux and 0–5 cm or 5–10 cm soil moisture is not linear. The differences between the forest types are less pronounced when possible statistical outliers are removed, with outliers determined as values above the 3rd quartile value plus 1.5× the middle quartile range, and less than the 1st quartile value minus 1.5× the middle quartile range, in this case −83 to 120 μg CH4–C/m2/h.
Soil moisture varied by time and by site during this study, including fully saturated or flooded soil at most of the sites at some point during the year. Precipitation accounted for some of the water depth and soil moisture variation, but not all; the highest water depth measurements occurred after a high precipitation event, but some of the other higher water depth measurements appear to be unrelated to precipitation and may be due to water management. SMC varies spatially more than temporally, although soil moisture and water depth were generally higher in winter. SMC is defined as (wet weight–dry weight)/dry weight.
CO2 net emissions decreased as soil moisture increased (Fig. 2). CH4 flux increased with increasing soil moisture. Temperature varied by season during the duration of this study. CO2 net emissions increased with increasing temperature, and CH4 flux showed little relationship with temperature (Fig. 3). Over the year, CO2 net emissions vary with temperature but CH4 responded to soil moisture and water depth as well as temperature.
Statistical analysis showed that the three different forest communities studied have different rates of carbon gas flux and are therefore statistically separate populations in this regard (Table 3 and Fig. 4). The differences between Atlantic white cedar and the other two forest types in terms of CH4 flux were the only statistically significant values at a 95% confidence level, but all tests tended toward separation between the three populations in both measurements. Atlantic white cedar is the forest type with flux measurements most different from the other two, and maple-gum and pocosin are the populations with the most similar measurements.
Discussion
Drivers of flux
The results show that forest type has an effect on flux, in addition to the effects of moisture and temperature. Drivers behind soil respiration and carbon gas flux in other studies include ground water or surface water level and source, vegetation type, water-filled pore space, restoration status, temperature and pH. In this study, soil moisture and temperature were the main drivers for CH4 and CO2 flux, respectively, with some influence from forest type (Figs 2, 3 and Table 3).
In this study, soil moisture from 0–5 cm deep was more highly correlated to CH4 flux than soil moisture 5–10 cm deep, indicating the relationship to depth to ground water in other studies may be a measure of field capacity or water retention of the soil and time since last rewetting. CO2 net emissions decreasing as soil moisture increases is expected and may be related to the biogeochemical chain of electron acceptors as oxygen is depleted in flooded conditions. This may also be related to reduced metabolic activity and increased soil moisture in the colder months. Refuge management activity affects water level in the ditches and therefore depth of the water table, especially closer to the roads and ditches. This was taken into account by the water depth variable. Of the sites sampled in this study, two were flooded frequently, the northernmost pocosin site and the northernmost maple-gum site. The Atlantic white cedar and pocosin sites that are closest to the highest concentration of ditches were least often flooded.
Comparison with other studies—CO2
Net CO2 emissions in the Great Dismal Swamp in this study was 740 g C/m2/year for cedar, 684 g C/m2/year for maple-gum, and 711 g C/m2/year for pocosin, or 7397 kg C/ha/year for cedar, 6844 kg C/ha/year for maple-gum, and 7113 kg C/ha/year for pocosin. The individual sites ranged from the lowest CO2 fluxes in the wettest, most frequently inundated sites (low of 575 g C/m2/year at the wettest maple-gum site), to the highest in the dryer sites (817 g C/m2/year at a dry pocosin site). These CO2 measurements are within the ranges reported in the studies below.
Other studies of soil respiration in forests and wetlands as well as in laboratory incubation experiments have reported a wide range of values, mainly higher than the values found in this study. These measurements were either reported in, or were converted to, grams of carbon, per square meter, per year (g C/m2/year). For CO2 net emissions, high values were seen in a Canadian peatland, with very high values when the water table was 70 cm below the surface (239,805.00–341,540.45 g C–CO2/m2/year), and still fairly high when the water table was 10 cm above the surface (10,900–18,167 g C–CO2/m2/year) (Moore and Knowles 1989). Moderate levels of CO2 flux were seen in North Carolina peatlands and pocosins, with short pocosin ranging from 438 to 1314 g C/m2/year, tall pocosin (which is more similar to the pocosin in the Great Dismal Swamp) ranging from 788 to 1095 g C/m2/year, and maple-gum ranging from 657 to 2190 g C/m2/year (Bridgham and Richardson 1992). Peak soil respiration in the subalpine Rocky Mountains was also moderately high at 1654 g C/m2/year (Berryman et al. 2016). A floodplain in Virginia saw similar levels of soil respiration, with 1091 g C–CO2/m2/year (Batson et al. 2014). Chambers in the water of a restored wetland in California also showed similar levels of 915 g C–CO2/m2/year (McNicol et al. 2017). Low levels of CO2 soil flux were seen in peatlands during snow cover in the winter in China, with 3–12 g C–CO2/m2/year in the peat and 20 g C–CO2/m2/year in a marsh (Miao et al. 2012). A natural site in Pocosin Lakes National Wildlife Refuge measured only 14 g C–CO2/m2/year (Wang et al. 2015). Peatland in Australia and forestry drained peatland in Finland showed net uptake of CO2, −308 g C–CO2/m2/year (Beringer et al. 2013) and −237 g C–CO2/m2/year (Lohila et al. 2011) respectively.
Comparison with other studies - CH4
CH4 flux in the Great Dismal Swamp in this study was 0.05 g C/m2/year for the cedar forest type, 1.29 g C/m2/year for maple-gum, and 3.81 g C/m2/year for pocosin. This is lower than the 1982 CH4 measurements shown below, which were taken over a 17-month period in maple-gum forest cover, but more in line with the low to moderate results in other study areas.
For CH4, Great Dismal Swamp measurements in the maple-gum forest type from 1982 showed high flux rates of 130 g C–CH4/m2/year to 1968 g C–CH4/m2/year (Harriss et al. 1982). The high CH4 values may be due to ebullition from pockets in the soil air space or below the water table or water surface in the case of flooded conditions. Other measurements are relatively low; 2.9 g C–CH4/m2/year in restored California wetland water chambers (McNicol et al. 2017), 0.01–0.04 g C–CH4/m2/year in snow-covered peatlands in China (Miao et al. 2012), 7.67 g C–CH4/m2/year in an inundated fen in Canada, and 0.19 g C–CH4/m2/year in an inundated bog in Canada (Moore and Knowles 1989).
Total area
The forest cover of the Great Dismal Swamp is 61% maple-gum, 15% pocosin, 12% cypress-gum, 3% Atlantic white cedar and 9% other with a total study area of 54,000 ha (Fleming et al. 2001). Using these figures as a guideline, the total area of Atlantic white cedar in the study area (1620 ha) has an average yearly flux of 0.75 metric tons carbon from CH4 and 11,983 metric tons of carbon from CO2. The total area of maple-gum in the study area (32,940 ha) has an average yearly flux of 425 metric tons of carbon from CH4 and 225,457 metric tons of carbon from CO2. The total area of pocosin in the study area (8100 ha) has an average yearly flux of 309 metric tons of carbon from CH4 and 57,617 metric tons of carbon from CO2. The total yearly carbon loss (not including uptake due to plant productivity and carbon burial) from the study area made up of these three forest types (54,000 ha minus the 21% that is cypress-gum or other is 42,660 ha) would then be 295,792 metric tons of carbon per year, from soil flux alone.
Increased sampling efficiencies
In an attempt to minimize error due to sampling, the techniques were designed to minimize researcher impact on the system: keeping the bases in place over the duration of the study; not stepping on the ground near the chambers when avoidable; leaving the sites as natural as possible; and keeping field equipment away from the chambers by using 12 foot long fluorinated ethylene propylene tubes. Despite these precautions, it is possible that motion above the ground could have caused some ebullition of CH4 from below ground during sampling. For example, bubbles rising through the standing water were often visible when approaching flooded sites. However, these small bubbles seemed to be restricted to the paths used for walking and were not seen in the chamber areas.
Hutchinson and Livingston (2001) provide several recommendations for reducing error during chamber sampling—an air tight chamber base, in our case achieved using the water trough seal; sufficient chamber base installation depth, based on soil porosity and sampling time. Our depth of 12.7 cm deep is easily sufficient according to that study’s calculations, given a short sampling time of 10 min, even at the highest calculated porosity (requiring at least 8.6 cm depth). Their recommendation that all chambers include a vent near ground level aims to reduce error due to sudden changes in pressure during sampling—for example, when the chamber top is placed and when gas is extracted as a sample. However, since we used continuous sampling, there were no disturbances to pressure leading to the disturbances that they observed in their study. Also, the short sampling time and reflective white chamber tops eliminate risk of pressure change due to increasing temperature inside the chamber as compared with the outside temperature. Additionally, our continuous sampling allows us to see if any large disturbances occur in real time. Some small deviation from the overall linear trends at the very beginning of sampling may have been due to the pressure change of placing the chamber top.
Smith and Dobbie (2001), although studying N2O soil emissions in agricultural land, found mostly statistically insignificant differences between sampling several days apart interpolating, and sampling every 8 h, as well as between samplings at different times of the day. Rochette and Eriksen-Hamel (2008) found that many methods were sufficient for N2O treatment comparison, but insufficient for comparison with other studies at other sites due to the limitations of their physical techniques. However, our study avoids many of these common pitfalls; although our chambers are unvented and uninsulated, our deployment duration was only 10 min. In addition, we had sufficient insertion depth, chamber height greater than 10 cm, no sample handling or storage, no use of plastic syringes, and no delay between sampling and analysis. We did not, however, use quality control gas standards as suggested in Rochette and Eriksen-Hamel (2008) but did follow the manufacturer’s instructions for maintenance at a greater than 99% accuracy level (Los Gatos Research, Inc. specifications 2014).
Implications of the study
Since soil moisture is responsive in part to manageable conditions, there is the possibility of management activities influencing future carbon gas flux. Prior to the 1970’s, when the Great Dismal Swamp National Wildlife Refuge was established, land use decisions (i.e. ditch construction and forestry) led to drier soil conditions and ecosystem vulnerability, which, given these findings, could have not only reduced CH4 flux, but also changed the characteristics of the soil and plant communities in addition to the changes caused directly by harvesting select species of timber. For example, fire susceptibility in terms of ignition success, burn depth and total combustion depends on factors including soil moisture, bulk density, organic matter component and species composition (Benscoter et al. 2011). In the Great Dismal Swamp, wetter areas with higher mean water levels were found to have thicker peat and higher species richness than drier areas, and while conditions in wetter areas did not meet fire risk conditions, drier areas were found to be always at risk of burning (Schulte 2017). CH4 flux, however, is a small fraction of net carbon flux. Future rewetting of the swamp may change plant and microbial communities once again, favoring wetland species that are tolerant of frequent flooding, overall moister conditions, and anoxic root zones (Ausec et al. 2009; Hartman et al. 2008). This may lead to increased CH4 flux and decreased CO2 net emissions. Rewetting may also cool the soil through evapotranspiration, further reducing CO2 net emissions and possibly mitigating some temperature increase due to changing global temperatures.
The limitations of this project suggest opportunities for future study such as sampling over greater temporal resolution or sampling over the length of a 24-h period to determine the effect of sunlight hours to see whether the same patterns persist. Such measurement disparity can be seen when eddy covariance measurements occurring over 24-h periods are compared with chamber methods; CH4 fluxes were 2–4 times higher from chambers (Krauss et al. 2016). Sampling differences from one day to the next with minimal changes in temperature or moisture would also be informative, since sampling each chamber only once a month means we do not have a full seasonal picture either. Sampling before, during and after a weather event would also be useful and could show the effect of rising ground water on flux as air is replaced with water in soil air pockets. Since surface soil moisture and temperature can be measured remotely using satellite data, it may be possible to model GHG flux throughout the refuge.
Conclusion
CH4 flux increased as temperature increased for pocosin, but decreased with temperature for cedar and maple. All of the CH4 fluxes increased as soil moisture increased. On average, as soil moisture increased by one unit of SMC, CH4 flux increased by 457 μg C–CH4/m2/h (Fig. 2). On average, as temperature increased by 1 °C, CO2 flux increased by 5109 μg C–CO2 m2/h (Fig. 3). Cedar average CH4 flux was significantly different from both maple and pocosin. These results show that soil carbon gas flux depends on soil moisture, temperature, and forest type, all as affected by anthropogenic activities in these peatlands.
Overall, CO2 net emissions occurred at much higher concentrations than CH4 flux in the Great Dismal Swamp. CH4 uptake sometimes outpaced production, but the soil was usually a net source, while CO2 net emissions always showed a net source. Different forest types showed somewhat different trends. CO2 was primarily associated with soil temperature, and CH4 flux was primarily associated with soil moisture in the top 5 cm (surface soil moisture). This information is relevant to assessing the implications of changing management decisions as habitat in the refuge is being restored. Although the US Fish and Wildlife Service does not manage for carbon sequestration, it is valuable to understand how changing hydrologic regimes that increase or decrease soil moisture may impact carbon balance in addition to habitat and other management goals (Sleeter et al. 2017).
This study shows the relationship between surface soil moisture and temperature, and gas flux. More variables could be studied to determine their relationship with soil carbon gas flux. The forests in the Great Dismal Swamp have been managed for centuries, which has very likely influenced current conditions including hydrology and soil conditions, since the peat soil is composed of organic matter accumulated over thousands of years. Topography, water flow, and soil nutrients would also play a role. While this study looked at sites situated in representative examples of three different forest types, more replication of these sites in different conditions could provide more information on gas flux in different hydrologic regimes, disturbed conditions, growth stages and tree maturities, and combinations of these factors.
References
Anderson FE, Bergamaschi B, Sturtevant C, Knox S, Hastings L, Windham-Myers L, Detto M, Hestir EL, Drexler J, Miller RL, Matthes JH, Verfaillie J, Baldocchi D, Snyer RL, Fujii R (2016) Variation of energy and carbon fluxes from a restored temperate freshwater wetland and implications for carbon market verification protocols. J Geophys Res 121:777–795. https://doi.org/10.1002/2015JG003083
Ausec L, Kraigher B, MandiC–Mulec I (2009) Differences in the activity and bacterial community structure of drained grassland and forest peat soils. Soil Biol Biogeochem 41:1874–1881. https://doi.org/10.1016/j.soilbio.2009.06.010
Barrd SC (2006) Great Dismal Swamp National Wildlife Refuge and Nansemond National Wildlife Refuge Final Comprehensive Conservation Plan July 2006. U.S. Fish and Wildlife Service Comprehensive Conservation Plan
Batson J, Noe GB, Hupp CR, Krauss KW, Rybicki NB, Schenk ER (2014) Soil greenhouse gas emissions and carbon budgeting in a short-hydroperiod floodplain wetland. J Geophys Res 120:77–95. https://doi.org/10.1002/2014JG002817
Benscoter BW, Thompson DK, Waddington JM, Flannigan MD, Wotton BM, de Groot WJ, Turetsky MR (2011) Interactive effects of vegetation, soil moisture and bulk density on depth of burning of thick organic soils. Int J Wildland Fire 20:418–429. https://doi.org/10.1071/WF08183
Beringer J, Livesley SJ, Randle J, Hutley L (2013) Carbon dioxide fluxes dominate the greenhouse gas exchanges of a seasonal wetland in the wet-dry tropics of northern Australia. Agric For Meteorol 182-183:239–247. https://doi.org/10.1016/j.agrformet.2013.06.008
Berryman E, Ryan MG, Bradford JB, Hawbaker TJ, Birdsey R (2016) Total belowground carbon flux in subalpine forests is related to leaf area index, soil nitrogen, and tree height. Ecosphere 7:8(16):article e01418
Bridgham SD, Richardson CJ (1992) Mechanisms controlling soil respiration (CO2 and CH4) in southern peatlands. Soil Biol Biochem 24(11):1089–1099
Brown DA, Atkinson RB (1999) Assessing survivability and growth of Atlantic white cedar [Chamaecyparis thyoides (L.) BSP] in the Great Dismal Swamp National Wildlife Refuge. In Atlantic White Cedar. Ecology and Management Symposium, USDA Forest Service GTR SRS-27, Google Scholar, p 1–7
Bubier J, Crill P, Mosedale A, Frolking S, Linder E (2003) Peatland responses to varying interannual moisture conditions as measured by automatic CO2 chambers. Glob Biogeochem Cycles 17(2):15. https://doi.org/10.1029/2002GB001946
Christiansen JR, Outhwaite J, Smukler SM (2015) Comparison of CO2, CH4 and N2O soil-atmosphere exchange measured in static chambers with cavity ring-down spectroscopy and gas chromatography. Agric For Meteorol 211-212:48–57. https://doi.org/10.1016/j.agrformet.2015/06/004
Drexler JZ, Fuller CC, Orlando J, Salas A, Wurster FC, Duberstein JA (2017) Estimation and uncertainty of recent carbon accumulation and vertical accretion in drained and undrained forested peatlands of the southeastern USA. J Geophys Res Biogeosci 122(10):2563–2579. https://doi.org/10.1002/2017JG003950
Fleming G, Coulling P, Walton D, McCoy K, Parrish M (2001) The natural communities of Virginia: classification of ecological community groups. First approximation. Natural Heritage Technical Report 01-1. Virginia Department of Conservation and Recreation, Division of Natural Heritage, Richmond, Unpublished report
Gutenberg L, Sleeter R (2018) Soil flux (CO2, CH4), soil temperature, and soil moisture measurements at the Great Dismal Swamp National Wildlife Refuge (2015–2017). U.S. Geological Survey data release. https://doi.org/10.5066/P9KBRSO4
Harriss RC, Sebacher DI, Day Jr. FP (1982) Methane flux in the Great Dismal Swamp. Nature 297:673–674
Hartman WH, Richardson CJ, Vilgalys R, Bruland GL (2008) Environmental and anthropogenic controls over bacterial communities in wetland soils. Proc Natl Acad Sci USA 105(46):17842–17847. https://doi.org/10.1073/pnas.0808254105
Hutchinson GL, Livingston GP (2001) Vents and seals in non-steady-state chambers used for measuring gas exchange between soils and the atmosphere. Eur J Soil Sci 52:675–682
Jenkins JC, Birdsey RA, Pan Y (2001) Biomass and NPP estimation for the Mid-Atlantic region (USA) using plot-level forest inventory data. Ecol Appl 11(4):1174–1193. https://doi.org/10.2307/3061020
Kim J-W, Lu Z, Gutenberg L, Zhu Z (2017) Characterizing hydrologic changes of the Great Dismal Swamp using SAR/InSAR. Remote Sens Environ 198:187–202. https://doi.org/10.1016/j.rse.2017.06.009
Krauss KW, Holm Jr. GO, Perez BC, McWhorter DE, Cormier N, Moss RF, Johnson DJ, Neubauer SC, Raynie RC (2016) Component greenhouse gas fluxes and radiative balance from two deltaic marshes in Louisiana: pairing chamber techniques and eddy covariance. J Geophys Res Biogeosci 121:1503–1521. https://doi.org/10.1002/2015JG003224
Krauss KW, Whitbeck Jl (2012) Soil greenhouse gas fluxes during wetland forest retreat along the lower Savannah River, Georgia (USA). Wetlands 32(1):73–81. https://doi.org/10.1007/s13157-011-0246-8
Laderman AD, Brody M, Pendleton E (1989) The ecology of Atlantic white cedar wetlands- a community profile. U.S. Department of Interior, Fish and Wildlife Service, National Wetlands Research Center. Biolo Rep 85(7.21):114
Laing JM, Shear TH, Blazich FA (2011) How management strategies have affected Atlantic White-cedar forest recovery after massive wind damage in the Great Dismal Swamp. For Ecol Manag 262(8):1337–1344. https://doi.org/10.1016/j.foreco.2011.06.026
Lohila A, Minkkinen K, Aurela M, Tuovinen J-P, Penttila T, Ojanen P, Laurila T (2011) Greenhouse gas flux measurements in a forestry-drained peatland indicate a large carbon sink. Biogeosciences 8:3203–3218. https://doi.org/10.5194/bg-8-3203-2011
McNicol G, Sturtevant CS, Knox SH, Dronova I, Baldocchi DD, Silver WL (2017) Effects of seasonality, transport pathway, and spatial structure on greenhouse gas fluxes in a restore wetland. Glob Change Biol. 15. https://doi.org/10.1111/gcb.13580
Miao Y, Song C, Wang X, Sun X, Meng H, Sun L (2012) Greenhouse gas emissions from different wetlands during the snow-covered season in Northeast China. Atmos Environ 62:328–335. https://doi.org/10.1016/j.atmosenv.2012.08.036
Moore TR, Knowles R (1989) The influence of water table levels on methane and carbon dioxide emissions from peatland soils. Can J Soil Sci 69(1):33–38. https://doi.org/10.4141/cjss89-004
Muñoz C, Saggar S, Berben P, Giltrap D, Jha N (2011) Influence of waiting time after insertion of base chamber into soil on produced greenhouse gas fluxes. Chil J Agric Res 71(4):610–614
Oaks RQ, Whitehead DR (1979) Geologic setting and origin of the Dismal Swamp, southeastern Virginia and northeastern North Carolina. In: Kirk PW (ed) The Great Dismal Swamp. University Press of Virginia, Charlottesville, p 1–24
Parkin TB, Venterea RT, Hargreaves SK (2012) Calculating the detection limits of chamber-based soil greenhouse gas flux measurements. J Environm Qual 41:705–715. https://doi.org/10.2134/jeq2011.0394
Parthum B, Pindilli E, Hogan D (2017) Benefits of the fire mitigation ecosystem service in The Great Dismal Swamp National Wildlife Refuge, Virginia, USA. J Environ Manag 203(1):375–382. https://doi.org/10.1016/j.jenvman.2017.08.018
Powell SW, Day Jr. FP (1991) Root Production in four communities in the Great Dismal Swamp. Am J Bot 78(2):288–297
Reddy AD, Hawbaker TJ, Wurster F, Zhu Z, Ward S, Newcomb D, Murray R (2015) Quantifying soil carbon loss and uncertainty from a peatland wildfire using multi-temporal LiDAR. Remote Sens Environ 170:306–316. https://doi.org/10.1016/j.rse.2015.09.017
Rochette P, Eriksen-Hamel NS (2008) Chamber measurements of soil nitrous oxide flux- are absolute values reliable? Soil Sci Soc Am J 72(2):331–342
Schulte ML (2017) Hydrologic controls on ecosystem structure and function in the Great Dismal Swamp. Master’s thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA
Sleeter R, Sleeter BM, Williams B, Hogan D, Hawbaker T, Zhu Z (2017) A carbon balance model for the Great Dismal Swamp ecosystem. Carbon Balance Manag 12(2):20. https://doi.org/10.1186/s13021-017-0070-4
Smith KA, Dobbie KE (2001) The impact of sampling frequency and sampling times on chamber-based measurements of N2O emissions from fertilized soils. Glob Change Biol 7:933–945
Wang H, Richardson CJ, Ho M (2015) Dual controls on carbon loss during drought in peatlands. Nat Climate Change 5(6):584–587
Winton S, Flanagan N, Richardson CJ (2017) Neotropical peatland methane emissions along a vegetation and biogeochemical gradient. PLOS ONE 12(10):12. https://doi.org/10.1371/journal.pone.0187019
Winton S, Richardson CJ (2016) A cost-effective method for reducing soil disturbance-induced errors in static chamber measurement of wetland methane emissions. Wetlands Ecol Manag 24:419–425. https://doi.org/10.1007/s11273-015-9468-5
Acknowledgements
This work was funded by the U.S. Geological Survey through the National Assessment of Ecosystem Carbon Sequestration and Greenhouse Gas Fluxes (LandCarbon) under the Land Change Science Program of the Climate and Land Use Mission Area. This study is also supported by George Mason University, College of Science, Department of Geography and Geoinformation Sciences. The staff of GDSNWR and DSSP also provided advice to the study design and assistance accessing field plots. Thanks to USGS Wetland and Aquatic Research Center, USGS Eastern Geographic Science Center and George Mason Global Environment and Natural Resources Institute. Thanks to Rachel Sleeter for help with data and review. Thanks to Christopher Wright, Joshua Simon, Christina Musser, Timothy Larson, and Alexander Jonesi for assistance with data collection. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. Data generated during this study are available at https://doi.org/10.5066/P9KBRSO4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Gutenberg, L., Krauss, K.W., Qu, J.J. et al. Carbon Dioxide Emissions and Methane Flux from Forested Wetland Soils of the Great Dismal Swamp, USA. Environmental Management 64, 190–200 (2019). https://doi.org/10.1007/s00267-019-01177-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00267-019-01177-4