Abstract
Purpose
Radiation pneumonitis is a serious complication of radioembolization. In holmium-166 ([166Ho]) radioembolization, the lung mean dose (LMD) can be estimated (eLMD) using a scout dose with either technetium-99 m-macroaggregated albumin ([99mTc]MAA) or [166Ho]-microspheres. The accuracy of eLMD based on [99mTc]MAA (eLMDMAA) was compared to eLMD based on [166Ho]-scout dose (eLMDHo-scout) in two prospective clinical studies.
Materials and Methods
Patients were included if they received both scout doses ([99mTc]MAA and [166Ho]-scout), had a posttreatment [166Ho]-SPECT/CT (gold standard) and were scanned on the same hybrid SPECT/CT system. The correlation between eLMDMAA/eLMDHo-scout and LMDHo-treatment was assessed by Spearman’s rank correlation coefficient (r). Wilcoxon signed rank test was used to analyze paired data.
Results
Thirty-seven patients with unresectable liver metastases were included. During follow-up, none developed symptoms of radiation pneumonitis. Median eLMDMAA (1.53 Gy, range 0.09–21.33 Gy) was significantly higher than median LMDHo-treatment (0.00 Gy, range 0.00–1.20 Gy; p < 0.01). Median eLMDHo-scout (median 0.00 Gy, range 0.00–1.21 Gy) was not significantly different compared to LMDHo-treatment (p > 0.05). In all cases, eLMDMAA was higher than LMDHo-treatment (p < 0.01). While a significant correlation was found between eLMDHo-scout and LMDHo-treatment (r = 0.43, p < 0.01), there was no correlation between eLMDMAA and LMDHo-treatment (r = 0.02, p = 0.90).
Conclusion
[166Ho]-scout dose is superior in predicting LMD over [99mTc]MAA, in [166Ho]-radioembolization. Consequently, [166Ho]-scout may limit unnecessary patient exclusions and avoid unnecessary therapeutic activity reductions in patients eligible for radioembolization.
Trail registration: NCT01031784, registered December 2009. NCT01612325, registered June 2012.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
During hepatic radioembolization, microspheres with beta-emitting isotopes of either yttrium-90 (90Y) or holmium-166 ([166Ho]) are injected via catheterization of the hepatic artery [1]. Treatment is preceded by injection of a scout dose to simulate distribution, most commonly using technetium-99 m-macroaggregated albumin ([99mTc]MAA) [2]. The purpose of this scout procedure is threefold: (1) to analyze the anticipated intrahepatic distribution of activity after treatment; (2) to exclude unacceptable extrahepatic abdominal activity caused by hepato-gastro-intestinal collaterals; (3) to estimate the anticipated radiation absorbed dose in the lungs caused by shunting. The latter is of importance to avoid radiation pneumonitis, a rare but serious complication.
Arteriovenous anastomoses in liver parenchyma or in tumors allow for shunting of particles and may cause depositions of activity in the lungs. This can severely affect respiratory function [2]. Radiation pneumonitis typically occurs 1–6 months posttreatment and is clinically characterized by dry cough and progressive exertional dyspnea, potentially becoming life-threatening [3].
Patients are generally excluded from radioembolization if predicted lung mean dose (LMD) exceeds 30 Gy for a single treatment and/or 50 Gy for multiple treatments [1]. In a survey among radioembolization centers, 48% of respondents answered that up to a quarter of their patients were considered ineligible for therapy, based on lung shunting as assessed with [99mTc]MAA [4]. While this finding highlights the substantial impact of lung shunting on clinical practice, there is scientific evidence suggesting that lung shunting is largely overestimated by [99mTc]MAA, especially when using planar imaging [2,3,4,5]. Several explanations for the poor predictive value of [99mTc]MAA have been identified, including inaccurate quantification of [99mTc]MAA, particle size reduction by fragmentation of the albumin aggregates and differences in biodistribution of [99mTc]MAA compared to the treatment particle [2,3,4,5,6].
Treatment with [166Ho]-microspheres can be preceded by a scout dose consisting of the same microspheres, instead of [99mTc]MAA. For [166Ho], beta-decay is accompanied by the emission of gamma photons (81 keV, 6.2% abundance), enabling the use of quantitative SPECT/CT to predict distribution of [166Ho]-microspheres [2]. Braat et al. showed that use of [166Ho]-scout dose is safe, even if significant extrahepatic depositions occur [7].
Previously, Elschot et al. compared the performance of [99mTc]MAA and [166Ho]-scout for estimation of LMD prior to [166Ho]-radioembolization in 14 patients with unresectable liver metastases [2]. In that clinical phase I study, [166Ho]-scout proved to be more accurate than [99mTc]MAA in predicting LMD, with [99mTc]MAA significantly overestimating LMD compared to posttreatment [166Ho]-SPECT/CT [2]. Although significant, differences were only validated in a limited number of patients. In the present study, the clinical value of [166Ho]-scout versus [99mTc]MAA-scout for LMD prediction was investigated in an expanded patient population, consisting of both the initial phase I study and a subsequent phase II within-patient comparison study.
Materials and Methods
Patients
All patients from the prospective phase I and II Holmium Embolization Particles for Arterial Radiotherapy (HEPAR) studies were included (Clinicaltrials.gov numbers NCT01031784 and NCT01612325) [7, 8]. Each patient had unresectable liver metastases treated with [166Ho]-microspheres. The institutional review board approved the studies and all patients provided written informed consent before enrollment [6]. Patients were included in the present analysis if they received both scout doses ([99mTc]MAA and [166Ho]-scout), had a posttreatment [166Ho]-SPECT/CT (defined as gold standard) and were all scanned on the same hybrid SPECT/CT system.
Between December 2009 and March 2015, 53 patients were included in the phase I and II HEPAR studies. All patients received [99mTc]MAA, [166Ho]-scout and subsequent [166Ho]-treatment dose. Of these, sixteen patients were excluded from the analysis due to scanning on a non-hybrid SPECT system (10 patients) or unavailability of a posttreatment scan (6 patients), resulting in a total of 37 patients for analysis. The majority of patients presented with colorectal carcinoma (19/37, 51.4%) (Table 1).
Procedure
Several days before treatment a preparatory angiography was performed. An aimed total activity of 150 MBq [99mTc]MAA (0.8 mg, approximately 1.8 million particles, TechneScan LyoMAA; Mallinckrodt Medical B.V., Petten, The Netherlands) was injected at one or more injection positions, followed by SPECT/CT [6]. The median injected activity was 142 MBq, range 65–491 MBq. To avoid degradation of [99mTc]MAA, activity was prepared on demand, immediately before use and imaging was performed immediately after angiography. On the day of treatment, exact injection positions were reproduced, and patients first received an aimed scout dose of 250 MBq [166Ho]-microspheres in the morning. The scout dose consisted of approximately 60 mg; 3 million microspheres, with a median injected activity of 261 MBq (range 147–292 MBq). A vascular sheath was left in the common femoral artery to facilitate repeat catheterization in the afternoon. If subsequent SPECT/CT revealed no contra-indications for radioembolization, catheterization was repeated and the [166Ho]-microspheres treatment dose was administrated in the afternoon. The [166Ho]-microspheres were produced on site (University Medical Center Utrecht, Utrecht, the Netherlands) [9, 10]. Median administered treatment activity of [166Ho]-microspheres per procedure was 6.159 MBq (range 2.207–12.897 MBq). In all patients, the injection positions in the three procedures were assessed as being adequately matched. Provided that catheters were situated within the same vessel, any positional variance was considered inconsequential to the magnitude of the lung shunt. In the majority of treatments (25/37, 67.6%), injections were performed sequentially in the left and right hepatic artery. Follow-up consisted of physical examinations, blood work and imaging during a period of at least 3 months after [166Ho]-treatment [8]. Adverse events were scored according to the Common Toxicity Criteria for Adverse Events version 3.0 [8].
Imaging
All SPECT/CT images were acquired on the same dual headed SPECT/CT camera (Symbia T16, Siemens Health Care). [99mTc]MAA-SPECT images were acquired using a low energy collimator, 128 × 128 matrix, 120 angles (20 s. per projection) over a noncircular 360° orbit and a 140-keV ± 7.5% photopeak energy window. [166Ho]-SPECT data were acquired using a medium energy collimator, 128 × 128 matrix with 120 angles over a noncircular 360° orbit and a 81-keV ± 7.5% photopeak window. Low-dose CT data were acquired and used to create a CT-derived attenuation map (Syngo MI Applications; Siemens Healthcare). All SPECT/CT images enclosed the entire liver and the basal lung fields. [99mTc]MAA and [166Ho]-SPECT were reconstructed using clinical reconstructions, applying previous protocols [2].
Quantitative Analysis
Using SPECT/CT images, volumes of interest (VOIs) were segmented on corresponding co-registered abdominal low-dose CT scans, using ITK-snap (version 3.8.0) [11]. The liver VOI was manually delineated. To minimize intraobserver differences, the lungs were automatically delineated using a freely available pre-trained convolutional neural network, lung mask, using a U-net model (R231) [12]. The body contour was obtained by threshold-based segmentation of the low-dose CT in order to obtain total body counts in the co-registered SPECT image. All images were visually checked to ensure correct segmentation and registration. Erroneous registration of liver activity in lungs was expected, due to co-registration errors, partial volume effect and/or patient breathing. Therefore, a 3D 2 cm margin was automatically added around the liver VOI. The voxels in the 3D liver + 2 cm were excluded from the lung VOI [2].
To maximize accuracy, estimated LMD (eLMD) was based on measured activity in the left lung alone, as it was less prone to erroneous registration of liver activity in the lung VOI [13]. The LMD was assumed to be equal in both lungs. The eLMD on all SPECT/CT’s was calculated using the following formula,
in which Anet is the net administered activity (calibrated activity for [166Ho]-microspheres treatment–measured residual activity in the administration system after [166Ho]-microspheres treatment), 15.87 J/GBq the conversion factor of energy deposition and Mleft lung VOI the calculated mass of the left lung VOI (volume left lung VOI multiplied by an assumed lung density of 0.3 g/mL) [2].
Statistical Analysis
The LMDHo-treatment was assessed by posttreatment [166Ho]-SPECT/CT (i.e., the gold standard). The correlation between eLMDMAA/eLMDHo-scout and LMDHo-treatment was assessed by calculating the Spearman’s rank correlation coefficient (r). Absolute differences in eLMDMAA/Ho-scout minus LMDHo-treatment (ΔeLMDMAA/Ho-scout) were calculated to compare the predictive value of both methods. Bland–Altman analyses to assess the correlation between eLMDMAA/eLMDHo-scout and LMDHo-treatment were not conducted, given the median LMDHo-treatment was (near) zero (see results section), indicating that differences were explained by observed lung shunting during scout procedures. Descriptive parameters are presented as medians and range. Statistical data analysis was performed using a commercial statistical software package (SPSS for Windows, version 21.0; SPSS Inc.). Wilcoxon signed rank test was used to analyze paired data (significance level 0.05), since normal distribution could not be assumed.
Results
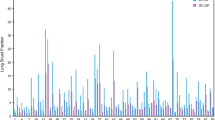
Median follow-up was 4 months (range 1–14 months). During follow-up, none of the included patients showed symptoms of radiation pneumonitis. Median LMDHo-treatment was 0.00 Gy (range 0.00–1.20 Gy). eLMDMAA was significantly higher with a median of 1.53 Gy (range 0.09–21.33 Gy) (p < 0.01). The eLMDHo-scout was not significantly different from LMDHo-treatment (median 0.00 Gy, range 0.00–1.21 Gy) (p > 0.05) (Fig. 1).
Planar images of two patients illustrating the difference in activity distribution. The eLMDMAA for the first patient was 21.3 Gy, while both eLMDHo-scout and LMDHo-treatment were 0.0 Gy (A–C). The second patient had an eLMDMAA of 21.1 Gy, with the eLMDHo-scout and LMDHo-treatment both measured as 0.0 Gy (D–F). From left to right; (A/D) pretreatment [99mTc]MAA scintigraphy, (B/E) pretreatment [166Ho]-scout scintigraphy and (C/F) posttreatment [166Ho]-scintigraphy
In all cases, eLMDMAA was higher than the LMDHo-treatment (Fig. 2). While a significant, positive correlation was found between eLMDHo-scout and LMDHo-treatment (r = 0.43, p < 0.01), there was no correlation between eLMDMAA and LMDHo-treatment (r = 0.02, p = 0.90). The median ΔeLMDMAA of 1.52 Gy (range 0.09–21.33 Gy) was significantly higher than median ΔeLMDHo-scout of 0.00 Gy (range 0.00–1.21 Gy) (p < 0.01) (Fig. 3).
Regarding the ΔeLMDMAA, eight out of 37 patients (21.6%) demonstrated a difference greater than 5 Gy. Two out of 37 (5.4%) showed an absolute difference exceeding 20 Gy. These two patients were diagnosed and treated for colorectal carcinoma and neuroendocrine tumor liver metastases, respectively. Interestingly, intrahepatic cholangiocarcinoma (ICC) patients constituted half of the cases with differences exceeding 5 Gy. Among the five ICC patients included in this study, four out of five (80%) displayed a difference greater than 5 Gy. The median eLMDHo-scout for ICC patients was 0.0 Gy (range 0.00–0.00 Gy), while the median eLMDMAA was 6.11 Gy (range 0.09–16.3 Gy). Median time interval between [99mTc]MAA and [166Ho]-scout was seven days (range 2–20 days). No (serious) adverse events possibly, probably or definitively related to the [166Ho]-scout were registered.
Discussion
The lung absorbed dose based on posttreatment [166Ho]-SPECT/CT and estimated by [166Ho]-scout were both significantly lower than estimations based on [99mTc]MAA. None of the patients developed signs of radiation pneumonitis.
As highlighted by van Elschot et al., the differences between [99mTc]MAA and [166Ho]-scout are primarily attributed to the distinct particle characteristics and biodistribution patterns of [99mTc]MAA and [166Ho]-microspheres [2].
The higher accuracy of [166Ho]-scout for LMD prediction confirms previous phase I findings by Elschot et al. [2]. The methods used in the current study and the phase I study by Elschot et al. differed slightly. The eLMD calculated by Elschot et al. was based on the registered activity in both lungs. In the current study, the right lung was excluded to minimize erroneous capture of liver activity. As the lung perfusion between the right and left lung was assumed to be nearly symmetrical, the left lung was considered representative for eLMD [14].
The [99mTc]MAA dose deposition in the lungs observed in this study, 1.53 Gy (range 0.09–21.33 Gy), is in line with prior reports. A study on predictive lung dosimetry in 90Y-radioembolization, using [99mTc]MAA-SPECT/CT, reported a median eLMDMAA of 4.51 (range 0.85–18.87) [15]. Recently, Stella et al. investigated the occurrence of radiation pneumonitis after 90Y-radioembolization in relation to LMD. The eLMD was calculated on [99mTc]MAA planar scintigraphy by multiplying LSF with administered therapeutic activity. The actual LMD was determined on posttreatment 90Y-PET. In line with this study, a median eLMDMAA of 3.5 Gy (range 0.2–89.0 Gy) and an actual median LMD of 1 Gy (range 0.0–22.1 Gy) were reported.[5] However, eLMDMAA derived from planar scintigraphy is known to overestimate LMD compared to SPECT/CT measurements [3, 16].
Likewise, in the context of resin [90Y] radioembolization, the potential advantages of using the same particle for scout and treatment have been investigated. In a single-arm clinical trial, involving 30 patients with HCC, the efficacy and safety of 0.56 GBq resin [90Y] microspheres (scout90Y) were compared with [99mTc]MAA for predicting the therapeutic resin [90Y] dose [17]. The mapping procedures using both [99mTc]MAA and scout90Y were performed on the same day, with treatment activity administered after three days. Scout90Y, using attenuation corrected SPECT/CT images, outperformed [99mTc]MAA SPECT/CT in predicting lung shunt fraction (LSF). In the case of LSF, scout90Y demonstrated a strong linear correlation with the therapeutic dose (r = 0.76, p < 0.001), in contrast to [99mTc]MAA’s weak correlation (r = 0.39, p = 0.032). These findings underscore the potential advantages of using a surrogate scout over [99mTc]MAA for LMD prediction in glass [90Y] radioembolization as well.
Accurate eLMD is important, not only to prevent radiation pneumonitis, but even more to avoid unnecessary dose reduction and/or patient exclusion [6]. LMD predictions are typically made by quantification of [99mTc]MAA distribution on planar scintigraphy [3]. The LSF is determined by dividing the counts in the lung area by the total counts in both the lung and liver regions [5]. The resulting LSF may then be multiplied by the planned therapeutic activity to acquire an eLMD. For all commercially available radioembolization particles, the upper dose limit to the lungs is set at 30 Gy for single radioembolization treatment [1, 18]. To date, this is also the case for [166Ho]-microspheres; however, the rationale for this maximum is based on limited research and adopted from 90Y data. Moreover, in the above-mentioned study by Stella et al., only two out of 14 patients with an eLMDMAA above 30 Gy developed radiation pneumonitis after 90Y-treatment [5]. These results suggest that treatment adjustments or exclusion based on eLMDMAA seem to be unjustified in numerous cases. In the prospective SARAH- and EPOCH-trial, 6.2% (14/226) and 1.8% (4/215) of patients, respectively, were excluded based on eLMD by planar [99mTc]MAA imaging. This stresses the need for a more accurate prediction method for LMD. At the same time, the 30 Gy eLMD threshold will be difficult to validate as the number of reported radiation pneumonitis cases in clinic is very low (< 1%) [5].
In line with a previous report by our group, no (serious) adverse events related to [166Ho]-scout were registered during follow-up [7]. Moreover, in the recently completed SIM and HEPAR PLuS studies, [166Ho]-scout was used instead of [99mTc]MAA, further confirming its safety [19, 20].
Regarding the quantification method, the used lung dosimetry model was based on commonly applied assumptions, including minimal lung absorbed dose from extra-pneumonic tissue, complete local energy absorption and similar lung density for all patients. This impacts the accuracy of the LMD calculations, since lung density depends on the presence of lung pathologies, scanning position and inclusion of lung vasculature [13, 18]. Since the same model was applied for [99mTc]MAA and [166Ho]-scout analysis, the expected effect of these factors on the comparison was also limited.
Lastly, patterns of vascularization differ per tumor type. Our study was primarily based on metastatic colorectal tumors (51.4%). More hypervascular tumor types, such as hepatocellular carcinoma (HCC), are more susceptible to arteriovenous shunting, which consequently leads to a higher LMD [18, 21, 22]. HCC patients were not part of the present study. However, five patients with ICC, another hypervascular tumor, were included. The LMD was overestimated in four out of five ICC patients when using [99mTc]MAA, while the eLMD from [166Ho]-scout was in line with the actual LMD. It is therefore likely that [166Ho]-scout superiority in estimating LMD will hold in hypervascular tumors due to its inherent physical benefits over [99mTc]MAA. With the increase in use of [166Ho]-scout dose, it is expected that definitive data in hypervascular tumors, including HCC, will become available within the coming years. This study has several limitations regarding the administration technique used. First, the [99mTc]MAA-scout procedure and [166Ho]-treatment were performed on different days, while [166Ho]-scout and [166Ho]-treatment were performed on the same day. Second, [99mTc]MAA and [166Ho]-scout administration methods were different, bolus syringe injection for [99mTc]MAA versus a dedicated administration box for both [166Ho]-scout and [166Ho]-treatment. The administration pressure, volume and velocity may influence intravascular flow dynamics of the particles and thus particle distribution.[23] Third, even slight differences in injection position may lead to different flow dynamics for [99mTc]MAA and [166Ho]-scout. Fortunately, these factors are less likely to influence the assessment of lung shunting compared to the known influence on intrahepatic distribution [6]. Other limitations relate to the imaging techniques used. Due to its narrow field of view, SPECT imaging did not always include the upper lung regions. This limited the accuracy of LMD estimation to a certain extent since quantification depended on a specific area of the left lung only. Even though commonly assumed in the literature, distribution of microspheres in the lungs is not homogenous. Gravitational dependence of alveolar and vascular pressure results in preferential perfusion of the lower dorsal lung regions compared to the apex [24]. Nevertheless, missing upper regions on SPECT/CT images are expected to have a small effect on the current comparison. Furthermore, the emission spectrum of [166Ho] is not ideal for SPECT imaging, due to the high-energy gamma emissions which cause a significant down-scatter contribution in the 80.6 keV photopeak window. Accurate scatter correction methods relying on Monte Carlo simulations are often not available in clinical practice. Using conventional energy-window-based scatter correction, low count regions are more prone to inaccurate quantification due to under- or over correction.
Conclusion
[166Ho]-scout is superior in predicting lung mean dose over [99mTc]MAA. Using [166Ho]-scout may avoid unnecessary patient exclusions and therapeutic activity reductions in patients eligible for radioembolization.
References
Weber M, Lam M, Chiesa C, Konijnenberg M, Cremonesi M, Flamen P, et al. EANM procedure guideline for the treatment of liver cancer and liver metastases with intra-arterial radioactive compounds. Eur J Nucl Med Mol Imaging. 2022;49:1682–99.
Elschot M, Nijsen JFW, Lam MGEH, Smits MLJ, Prince JF, Viergever MA, et al. 99mTc-MAA overestimates the absorbed dose to the lungs in radioembolization: a quantitative evaluation in patients treated with 166Ho-microspheres. Eur J Nucl Med Mol Imaging. 2014;41:1965–75.
Kappadath CS, Lopez BP, Salem R, Lam MG. Lung shunt and lung dose calculation methods for radioembolization treatment planning. Q J Nucl Med Mol Imaging. 2021;65:32–42.
Reinders MTM, Mees E, Powerski MJ, Bruijnen RCG, van den Bosch MAAJ, Lam MGEH, et al. Radioembolisation in Europe: a survey amongst CIRSE members. Cardiovasc Intervent Radiol. 2018;41:1579–89.
Stella M, van Rooij R, Lam MGEH, de Jong HWAM, Braat AJAT. Lung dose measured on postradioembolization 90Y PET/CT and incidence of radiation pneumonitis. J Nucl Med. 2022;63(7):1075–80.
Smits M, Dassen M, Prince J, Braat A, Beijst C, Bruijnen R, et al. The superior predictive value of 166 Ho-scout compared with 99m Tc-macroaggregated albumin prior to 166 Ho-microspheres radioembolization in patients with liver metastases. Eur J Nucl Med Mol Imaging. 2020;47:798–806.
Braat AJAT, Prince JF, van Rooij R, Bruijnen RCG, van den Bosch MAAJ, Lam MGEH. Safety analysis of holmium-166 microsphere scout dose imaging during radioembolisation work-up: a cohort study. Eur Radiol. 2018;28:920–8.
Smits MLJ, Nijsen JFW, van den Bosch MAAJ, Lam MGEH, Vente MAD, Mali WPTM, et al. Holmium-166 radioembolisation in patients with unresectable, chemorefractory liver metastases (HEPAR trial): a phase 1, dose-escalation study. Lancet Oncol. 2012;13:1025–34.
Nijsen JFW, Zonnenberg BA, Woittiez JRW, Rook DW, Swildens-Van Woudenberg IA, Van Rijk PP, et al. Holmium-166 poly lactic acid microspheres applicable for intra-arterial radionuclide therapy of hepatic malignancies: effects of preparation and neutron activation techniques. Eur J Nucl Med. 1999;26:699–704.
Nijsen JFW, Van Steenbergen MJ, Kooijman H, Talsma H, Kroon-Batenburg LMJ, Van De Weert M, et al. Characterization of poly(l-lactic acid) microspheres loaded with holmium acetylacetonate. Biomaterials. 2001;22:3073–81.
Yushkevich PA, Piven J, Hazlett HC, Smith RG, Ho S, Gee JC, et al. User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. Neuroimage. 2006;31:1116–28.
Hofmanninger J, Prayer F, Pan J, Röhrich S, Prosch H, Langs G. Automatic lung segmentation in routine imaging is primarily a data diversity problem, not a methodology problem. Eur Radiol Exp. 2020;4:1–13.
Yu N, Srinivas SM, Difilippo FP, Shrikanthan S, Levitin A, McLennan G, et al. Lung dose calculation with SPECT/CT for 90Yittrium radioembolization of liver cancer. Int J Radiat Oncol Biol Phys. 2013;85:834–9.
Cheng CP, Taur AS, Lee GS, Goris ML, Feinstein JA. Relative lung perfusion distribution in normal lung scans: observations and clinical implications. Congnit Heart Dis. 2006;1:210–6.
Kao YH, Magsombol BM, Toh Y, Tay KH, Chow PKH, Goh ASW, et al. Personalized predictive lung dosimetry by technetium-99m macroaggregated albumin SPECT/CT for yttrium-90 radioembolization. EJNMMI Res. 2014;4:33.
Elsayed M, Cheng B, Xing M, Sethi I, Brandon D, Schuster DM, Bercu Z, Galt J, Barron B, Kokabi N. Comparison of Tc-99m MAA planar versus SPECT/CT imaging for lung shunt fraction evaluation prior to Y-90 radioembolization: are we overestimating lung shunt fraction? Cardiovasc Intervent Radiol. 2021;44(2):254–60.
Kokabi N, Webster LA, Elsayed M, Switchenko JM, Chen B, Brandon D, Galt J, Sethi I, Cristescu M, Kappadath SC, Schuster DM. Accuracy and safety of scout dose resin yttrium-90 microspheres for radioembolization therapy treatment planning: a prospective single-arm clinical trial. J Vasc Interv Radiol. 2022;33(12):1578–87.
Kappadath SC, Lopez BP, Salem R, Lam MGEH. Reassessment of the lung dose limits for radioembolization. Nucl Med Commun 2021:1064–75.
van Roekel C, van den Hoven AF, Bastiaannet R, Bruijnen RCG, Braat AJAT, de Keizer B, Lam MGEH, Smits MLJ. Use of an anti-reflux catheter to improve tumor targeting for holmium-166 radioembolization-a prospective, within-patient randomized study. Eur J Nucl Med Mol Imaging. 2021;48(5):1658–68.
Braat AJAT, Bruijnen RCG, van Rooij R, Braat MNGJA, Wessels FJ, van Leeuwaarde RS, van Treijen MJC, de Herder WW, Hofland J, Tesselaar MET, de Jong HWAM, Lam MGEH. Additional holmium-166 radioembolisation after lutetium-177-dotatate in patients with neuroendocrine tumour liver metastases (HEPAR PLuS): a single-centre, single-arm, open-label, phase 2 study. Lancet Oncol. 2020;21(4):561–70.
Gaba RC, Zivin SP, Dikopf MS, Parvinian A, Casadaban LC, Lu Y, et al. Characteristics of primary and secondary hepatic malignancies associated with hepatopulmonary shunting. Radiology. 2014;271:602–12.
Smits MLJ, van den Hoven AF, Rosenbaum CENM, Zonnenberg BA, Lam MGEH, Nijsen JFW, et al. Clinical and laboratory toxicity after intra-arterial radioembolization with 90Y-microspheres for unresectable liver metastases. PLoS ONE. 2013;8:1–9.
Miller SR, Jernigan SR, Abraham RJ, Buckner GD. Comparison of bolus versus dual-syringe administration systems on glass yttrium-90 microsphere deposition in an in vitro microvascular hepatic tumor model. J Vasc Interv Radiol. 2023;34:11–20.
Suresh K, Shimoda LA. Lung circulation. Compr Physiol. 2016;6:897.
Funding
The Department of Radiology and Nuclear Medicine of the UMC Utrecht receives royalties and research support from Quirem Medical and Terumo.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Marnix Lam is a consultant for Boston Scientific, Terumo/Quirem Medical, and receives research support from Boston Scientific and AAA/Novartis. Maarten Smits has served as a speaker for BTG and Terumo Medical. Arthur Braat is consultant for Boston Scientific/BTG and Terumo/Quirem Medical. All other authors declare to have no conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The Institutional Review Board granted approval for the Phase I and II Holmium Embolization Particles for Arterial Radiotherapy (HEPAR) studies, which are incorporated in this research. These studies are registered under Clinicaltrials.gov with the identifiers NCT01031784 and NCT01612325.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
For this type of study, consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wagemans, M.E.H.M., Braat, A.J.A.T., van Rooij, R. et al. Lung Mean Dose Prediction in Transarterial Radioembolization (TARE): Superiority of [166Ho]-Scout Over [99mTc]MAA in a Prospective Cohort Study. Cardiovasc Intervent Radiol 47, 443–450 (2024). https://doi.org/10.1007/s00270-023-03656-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-023-03656-y