Abstract
Objectives
To assess the evolution of administered radiotracer activity for F-18-fluorodeoxyglucose (18F-FDG) PET/CT or PET/MR in pediatric patients (0–16 years) between years 2000 and 2021.
Methods
Pediatric patients (≤ 16 years) referred for 18F-FDG PET/CT or PET/MR imaging of the body during 2000 and 2021 were retrospectively included. The amount of administered radiotracer activity in megabecquerel (MBq) was recorded, and signal-to-noise ratio (SNR) was measured in the right liver lobe with a 4 cm3 volume of interest as an indicator for objective image quality. Descriptive statistics were computed.
Results
Two hundred forty-three children and adolescents underwent a total of 466 examinations. The median injected 18F-FDG activity in MBq decreased significantly from 296 MBq in 2000–2005 to 100 MBq in 2016–2021 (p < 0.001), equaling approximately one-third of the initial amount. The median SNR ratio was stable during all years with 11.7 (interquartile range [IQR] 10.7–12.9, p = 0.133).
Conclusions
Children have benefited from a massive reduction in the administered 18F-FDG dose over the past 20 years without compromising objective image quality.
Clinical relevance statement
Radiotracer dose was reduced considerably over the past two decades of pediatric F-18-fluorodeoxyglucose PET/CT and PET/MR imaging highlighting the success of technical innovations in pediatric PET imaging.
Key Points
• The evolution of administered radiotracer activity for F-18-fluorodeoxyglucose (18F-FDG) PET/CT or PET/MR in pediatric patients (0–16 years) between 2000 and 2021 was assessed.
• The injected tracer activity decreased by 66% during the study period from 296 megabecquerel (MBq) to 100 MBq (p < 0.001).
• The continuous implementation of technical innovations in pediatric hybrid 18F-FDG PET has led to a steady decrease in the amount of applied radiotracer, which is particularly beneficial for children who are more sensitive to radiation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
[18F]Fluorodeoxyglucose ([18F]FDG) positron emission tomography (PET) combined with computed tomography (CT) or magnetic resonance (MR) imaging plays an important role in the diagnosis, staging, response assessment, and management of metabolically active malignancies in children, including lymphoma, malignant soft tissue and bone sarcoma, melanoma, Langerhans cell histiocytosis, and MIBG-negative neuroblastoma, as well as non-malignant diseases such as chronic granulomatous disease or fever of unknown origin [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18]. For hybrid PET imaging, [18F]FDG is considered the most common radiopharmaceutical in clinical routine [19]. While hybrid [18F]FDG PET imaging has achieved widespread clinical adoption due to its merits in the diagnostic workup of children, its use is associated with radiation exposure. Especially for children, radiation exposure is of great concern: Firstly, recent studies suggest that children exhibit an increased radiation sensitivity for at least 25% of cancer sites, and secondly, the longer life expectancy of children increases the likelihood for secondary, radiation-induced cancers to emerge. Thus, a variety of epidemiological studies have linked childhood radiation exposure including that associated with medical imaging to the occurrence of certain cancers such as central nervous system (CNS) tumors or leukemia [20,21,22,23,24].
In hybrid PET, radiation exposure originates from the radiopharmaceutical [18F]FDG, and in the case of PET/CT also from the CT scanner [25, 26]. With PET/MR, all of the radiation exposure derives from the injected [18F]FDG [27,28,29]. Since the introduction of integrated PET/CT and later PET/MR, different technological developments in PET, such as detector and reconstruction algorithms as well as dose administration protocols, have facilitated reductions of the amount of administered radiotracer activity [4, 19].
In an effort to quantify the developments and improvements of the last two decades, we aimed to systematically analyze the evolution of administered radiotracer activity in pediatric patients who underwent [18F]FDG PET/CT or PET/MR in the period from 2000 to 2021 at a large tertiary medical center. Therein, we hypothesized that the technical improvements of hybrid PET imaging of the last two decades have facilitated considerable reductions in the amount of administered radiotracer activity without compromising image quality.
Materials and methods
Study design and population
In this retrospective single-center study, we included all pediatric patients aged 0–16 years who underwent a clinically indicated [18F]FDG PET/CT or PET/MR of the body between January 2000 and May 2021 at a large tertiary medical center. Clinical information including age, gender, body height and weight, International Classification of Diseases code (ICD-10), scanned body region, and amount of administered radiotracer activity in megabecquerel (MBq) were recorded. The study was approved by the local ethics committee (trial number BASEC 2020-03067) and was conducted in compliance with ICH-GCP-rules and the Declaration of Helsinki. The use for written informed consent was waived for patients scanned before January 2016. After 2016, only patients whose informed consent for further use of their medical data was available (due to a change in national regulatory rules) were included.
PET acquisition and image reconstruction
PET/CT and PET/MRI examinations were performed on 6 different scanners of the manufacturer General Electric (GE) Healthcare, all of which were up-to-date at the time of the scan. The following scanners were used in the course of time: Discovery LS, Discovery STE, Discovery RX, Discovery 690, Discovery MI, and Signa PET/MR. A detailed overview of the scanners’ characteristics is provided in the Supplementary Material. Exams were performed using a standardized clinical [18F]FDG dosage protocol, until 2016 with body weight–dependent amount of radiotracer activity, from 2017 with a body weight–dependent BMI-adapted protocol in accordance with the European Association of Nuclear Medicine (EANM) consensus guidelines and the 2016 update of the North American guidelines [30, 31]. Participants fasted for at least 4 h prior to [18F]FDG injection. Uptake time was set to 60 min by default.
CT or MR scans for attenuation correction and anatomical localization were performed as either whole-body examination (vertex to feet, e.g., in melanoma) or partial-body examination (vertex to thighs). Following the CT or MR acquisition, PET images were acquired, covering the same anatomical region. Since the first PET scanners have been introduced, several different PET reconstruction algorithms have been used [32]. In the early days, studies were reconstructed with simple and robust conventional filtered back-projection algorithms and later with iterative reconstruction algorithms [32,33,34]. In this context, it should be mentioned that the implementation of more modern reconstruction algorithms can also be associated with a decrease in the amount of administered radiotracer activity. More precisely, more modern algorithms can produce an image with acceptable image noise despite a lower applied amount of radiotracer activity [35].
Image quality
To assess objective image quality, one reader (A.G.G., 6 years of experience in radiology, and 1 year of experience in PET reading) carried out a signal-to-noise ratio (SNR) measurement in the right liver lobe in each examination, using a commercially available image analysis software (Advantage Workstation Version 4.7, GE Healthcare). Importantly, only liver parenchyma with normal appearance on both PET and CT was used for measurements. A volume of interest (VOI) with a standardized size of 4 cm3 was manually placed avoiding focal, non-physiologic uptake. SNR was defined as liver’s maximum standardized uptake value divided by the liver’s SUV standard deviation.
Statistical analysis
All statistical analyses were performed using the open-source statistics software R (version 4.1.0, R Foundation for Statistical Computing) [36]. Normality of the variables was assessed using the Shapiro-Wilk test. Categorical variables are expressed as frequency distribution and were compared using Fisher’s exact test. Continuous variables are given as median with interquartile range (IQR) in parentheses. Assessment of group differences was determined using the Kruskal-Wallis test for multiple groups and Wilcoxon test for two groups. For analysis of change in administered [18F]FDG activity over time, linear models were fitted, and relative changes were calculated between the beginning and end of the study period. For all comparisons, a p-value of < 0.05 was considered to be statistically significant.
Results
Study population characteristics
A detailed overview of the study population is provided in Table 1. In brief, 243 children and adolescents undergoing a total of 466 examinations, including 375 PET/CT scans (81%) and 91 PET/MR scans (19%), were included. The patients underwent [18F]FDG PET for hematologic neoplasms (mainly lymphoma) in 45%, osseous neoplasms (mainly osteosarcoma and Ewing sarcoma) in 11%, non-neoplastic hematologic diseases (mainly chronic granulomatous disease) in 8%, fever of unknown origin in 5%, soft tissue neoplasms (mainly rhabdomyosarcoma) in 5%, melanoma in 4%, osteomyelitis (infectious and non-infectious) in 3%, urogenital neoplasms in 3%, gastrointestinal neoplasms in 2%, and other diseases in 14% of cases, respectively. Sixty-three examinations were performed on a Discovery LS, 88 on a Discovery STE, 161 on a Discovery RX, 33 on a Discovery 690 VCT, 30 on a Discovery MI Gen 2 with digital detectors, and 91 on a Signa PET/MR.
Evolution of administered radiotracer activity
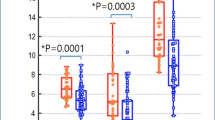
The median injected [18F]FDG activity was 218 MBq (range 32–402 MBq). From the time period 2000 to 2005 to the time period 2016–2021, the injected tracer activity decreased significantly (p < 0.001) by 66% from 296 MBq (IQR 253–372 MBq) to 100 MBq (IQR 79–161 MBq). Patient’s age-based, year-based, and scanner-based analysis of the study population are given in Tables 2, 3, and 4.
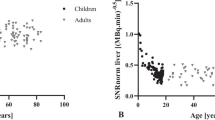
The overall evolution of the administered [18F]FDG radiotracer activity over time is given in Fig. 1. An age group–based analysis is presented in Fig. 2. The amount of [18F]FDG decreased continuously and substantially over time and for all age groups. Figure 3 illustrates the amount of injected activity between 2000 and 2021 divided into groups depending on the scanner used.
Signal-to-noise ratio of PET
The median SNR was 11.7 (IQR 10.7–12.9) and was stable during all years, as shown in Fig. 4. Thus, the decreasing amount of injected activity did not result in a lower SNR.
Discussion
In this study, we assessed the evolution of the amount of injected radiotracer activity of [18F]FDG in pediatric hybrid PET. The major findings of our study can be summarized as follows: First, we observed a major reduction in the amount of administered [18F]FDG activity in the last 20 years. Second, the reduction of [18F]FDG activity over time ranged from −57% up to −84%, depending on the age group, with the smallest reduction observed in the youngest patients. Third, the median administered [18F]FDG activity dropped from 7.3 MBq/kg in 2000–2005 to 2.7 MBq/kg in 2016–2021, respectively. Fourth, signal-to-noise ratio, an objective surrogate for image quality, remained stable during the same time.
Accurate staging and re-staging of oncological diseases in children and adolescents are crucial for appropriate treatment management and optimal outcomes. [18F]FDG PET/CT and PET/MR have evolved as important tools in the diagnosis, staging, and response assessment of various oncological diseases. Unfortunately, the use of PET/CT—and to a lesser extent PET/MR—is associated with considerable radiation exposure, which is particularly concerning for children given their increased sensitivity towards [37].
Concerning pediatric hybrid PET imaging, the EANM published a pediatric dosage card with respect to the amount of FDG in 2007 [38]. This guideline included recommendations for both “baseline activity” (for calculation purposes) and “minimum recommended activity” with multiplication factors. Between 2008 and 2016, these guidelines were updated repeatedly with the EANM further reducing the recommended value for the “minimum recommended activity” [31, 39]. These recommendations were also used at our institution, but with each hardware and software innovation implemented on our imaging systems the injected dose was reduced even further to fully harness the capabilities of these technological innovations.
These efforts were made in accordance with the ALARA principle (i.e., as low as reasonably achievable) to protect this radiation-sensitive patient cohort as much as possible. Nonetheless, care was given not to compromise image quality or the diagnostic evaluability of the acquired scans.
Specifically, the introduction of weight-adjusted tracer activity administration at our institution in 2016 marked an important milestone with it resulting in a significant reduction in patient’s exposure. Importantly, this dose regimens achieved widespread adoption at our institution beyond hybrid PET imaging. Notably, for cardiac SPECT imaging in adults, the weight-adjusted activity administration of radiotracer in combination with iterative reconstruction algorithms enabled dose reductions of 30% or even 40% [40].
Our data provides evidence that the continuous implementation of the latest hardware and software and the continuous optimization of injection protocols have enabled considerable dose reductions over the past two decades. This trend of ever decreasing radiopharmaceutical doses will continue in the next few years with exciting technologies such as artificial intelligence–powered image reconstructions entering the clinical market. Beyond these developments, scientists strive towards further improving pediatric dosage regimens. Notably, Cox et al recently investigated the relationship between patient-dependent parameters and [18F]FDG PET image quality in an effort to develop dedicated pediatric dosage protocols [41]. In their study of 102 children, they found that body weight was strongly associated with [18F]FDG PET image quality in children. Therefore, they proposed a nonlinear dosage regimen based on body mass, in order to provide a constant and clinically sufficient image quality while significantly reducing the effective dose beyond that recommended in current guidelines.
Importantly, with regard to hard- and software innovations for PET imaging, considerable advances have been made within the past 20 years. Optimizations in the scintillator crystal and photodetector combination, the acquisition electronics and advances in data processing, reconstruction, and image analysis together with increasing axial coverage of the scanner have facilitated considerable improvements in terms of the system sensitivity. The ultimate goal is always to maximize the number of counts acquired per unit of radiation dose to the patient and to achieve high spatial and temporal resolution.
These technical improvements can also be found in the scanner models used in this study [42]. For example, the earliest scanner model (Discovery LS) used 1st-generation bismuth germanate (BGO) detectors while the more recent scanner models (Discovery MI and Signa PET/MR system) use more advanced lutetium oxyorthosilicate detectors with small amounts of yttrium (LYSO) coupled with silicon photomultipliers (SiPMs). Moreover, the axial field of view increased from 15.2 cm for the oldest scanner model to ca. 25 cm for the most recent scanner models. Together with other technical improvements, these optimizations have facilitated an increase in sensitivity (in cps/kBq) from 6.41 to over 20 from the oldest to the most recent scanner models used in the current study.
Importantly, further advancements in PET instrumentation are already on the horizon as exemplified by the emergence of total body (TB) PET/CT systems. These revolutionary systems, with an extended axial field of view of up to 194 cm, enable the simultaneous imaging of the entire body. By encompassing the patient’s full anatomy, these imaging systems significantly improve signal detection efficiency, offering benefits for pediatric PET imaging. They allow for shorter acquisition times (less than a minute), reduced administered activity (as low as one-twentieth of the standard dose), and enhanced image quality due to improved signal-to-noise ratios. Moreover, this enables whole-body dynamic imaging with a temporal resolution of less than 1 s per imaging frame. Consequently, the integration of total body PET imaging into routine practice holds great potential for improving the safety and efficacy of pediatric PET procedures across a range of disorders affecting this vulnerable population [43].
Our study has some limitations. First, this study is a single-center retrospective analysis with a limited number of patients. Second, due to the long study period of over 20 years, we were not able to collect and report some data such as each PET system’s hardware and software configuration at the time of the acquisition, reconstruction methods, duration of the scan, and details on the CT and MR acquisition. Third, all examinations were performed on imaging systems of a single vendor. This may limit the generalizability of our results. Fourth, to quantify objective image quality, we only computed liver SNR. However, we acknowledge that there are many more metrics to objectify image quality. Fifth, radiation exposure from CT, which accounts for an important amount of the total radiation exposure, was not investigated.
Conclusion
The technical innovations of the past two decades have allowed for a continuous reduction in the amount of administered [18F]FDG (up to 84%) in pediatric PET/CT and PET/MR. Specifically, in pediatric patients, the average amount of administered activity in 2016–2021 is only approximately one-third of the amount administered between 2000 and 2005. In light of the increased sensitivity of children towards ionizing radiation, our findings are of special clinical significance and should promote further scientific efforts towards enabling further dose reductions for clinical pediatric hybrid PET imaging.
Abbreviations
- 18F-FDG:
-
[18F]fluorodeoxyglucose
- ALARA:
-
As low as reasonably achievable
- CT:
-
Computed tomography
- EANM:
-
European Association of Nuclear Medicine
- ICD:
-
International Classification of Diseases
- IQR:
-
Interquartile range
- MBq:
-
Megabecquerel
- MRI:
-
Magnetic resonance imaging
- PET:
-
Positron emission tomography
- SUVmax :
-
Maximum standardized uptake value
- TOF:
-
Time of flight
References
Freebody J, Wegner EA, Rossleigh MA (2014) 2-Deoxy-2-((18)F)fluoro-D-glucose positron emission tomography/computed tomography imaging in paediatric oncology. World J Radiol 6:741–755
Harrison DJ, Parisi MT, Shulkin BL (2017) The role of (18)F-FDG-PET/CT in pediatric sarcoma. Semin Nucl Med 47:229–241
Kluge R, Kurch L, Georgi T, Metzger M (2017) Current role of FDG-PET in pediatric Hodgkin’s lymphoma. Semin Nucl Med 47:242–257
Uslu L, Donig J, Link M, Rosenberg J, Quon A, Daldrup-Link HE (2015) Value of 18F-FDG PET and PET/CT for evaluation of pediatric malignancies. J Nucl Med 56:274–286
Gungor T, Engel-Bicik I, Eich G et al (2001) Diagnostic and therapeutic impact of whole body positron emission tomography using fluorine-18-fluoro-2-deoxy-D-glucose in children with chronic granulomatous disease. Arch Dis Child 85:341–345
Garg G, DaSilva R, Bhalakia A, Milstein DM (2016) Utility of fluorine-18-fluorodeoxyglucose positron emission tomography/computed tomography in a child with chronic granulomatous disease. Indian J Nucl Med 31:62–64
Theobald I, Fischbach R, Hulskamp G et al (2002) Pulmonary aspergillosis as initial manifestation of septic granulomatosis (chronic granulomatous disease, CGD) in a premature monozygotic female twin and FDG-PET diagnosis of spread of the disease. Radiologe 42:42–45
Meller J, Sahlmann CO, Scheel AK (2007) 18F-FDG PET and PET/CT in fever of unknown origin. J Nucl Med 48:35–45
Baum SH, Fruhwald M, Rahbar K, Wessling J, Schober O, Weckesser M (2011) Contribution of PET/CT to prediction of outcome in children and young adults with rhabdomyosarcoma. J Nucl Med 52:1535–1540
Ricard F, Cimarelli S, Deshayes E, Mognetti T, Thiesse P, Giammarile F (2011) Additional benefit of F-18 FDG PET/CT in the staging and follow-up of pediatric rhabdomyosarcoma. Clin Nucl Med 36:672–677
London K, Stege C, Cross S et al (2012) 18F-FDG PET/CT compared to conventional imaging modalities in pediatric primary bone tumors. Pediatr Radiol 42:418–430
Boktor RR, Omar WS, Mousa E et al (2012) A preliminary report on the impact of (1)(8)F-FDG PET/CT in the management of paediatric head and neck cancer. Nucl Med Commun 33:21–28
Halalsheh H, Kaste SC, Navid F et al (2018) The role of routine imaging in pediatric cutaneous melanoma. Pediatr Blood Cancer 65:e27412
Daldrup-Link HE, Theruvath AJ, Baratto L, Hawk KE (2021) One-stop local and whole-body staging of children with cancer. Pediatr Radiol. https://doi.org/10.1007/s00247-021-05076-x
Biassoni L, Easty M (2017) Paediatric nuclear medicine imaging. Br Med Bull 123:127–148
Cheng G, Servaes S, Zhuang H (2013) Value of (18)F-fluoro-2-deoxy-D-glucose positron emission tomography/computed tomography scan versus diagnostic contrast computed tomography in initial staging of pediatric patients with lymphoma. Leuk Lymphoma 54:737–742
Jadvar H, Connolly LP, Fahey FH, Shulkin BL (2007) PET and PET/CT in pediatric oncology. Semin Nucl Med 37:316–331
London K, Cross S, Onikul E, Dalla-Pozza L, Howman-Giles R (2011) 18F-FDG PET/CT in paediatric lymphoma: comparison with conventional imaging. Eur J Nucl Med Mol Imaging 38:274–284
Colleran GC, Kwatra N, Oberg L et al (2017) How we read pediatric PET/CT: indications and strategies for image acquisition, interpretation and reporting. Cancer Imaging 17:28
Foucault A, Ancelet S, Dreuil S et al (2022) Childhood cancer risks estimates following CT scans: an update of the French CT cohort study. Eur Radiol 32:5491–5498
Kutanzi KR, Lumen A, Koturbash I, Miousse IR (2016) Pediatric exposures to ionizing radiation: carcinogenic considerations. Int J Environ Res Public Health 13(11):1057. https://doi.org/10.3390/ijerph13111057
Mathews JD, Forsythe AV, Brady Z et al (2013) Cancer risk in 680,000 people exposed to computed tomography scans in childhood or adolescence: data linkage study of 11 million Australians. BMJ 346:f2360
Nikkila A, Raitanen J, Lohi O, Auvinen A (2018) Radiation exposure from computerized tomography and risk of childhood leukemia: Finnish register-based case-control study of childhood leukemia (FRECCLE). Haematologica 103:1873–1880
Pearce MS, Salotti JA, Little MP et al (2012) Radiation exposure from CT scans in childhood and subsequent risk of leukaemia and brain tumours: a retrospective cohort study. Lancet 380:499–505
Chawla SC, Federman N, Zhang D et al (2010) Estimated cumulative radiation dose from PET/CT in children with malignancies: a 5-year retrospective review. Pediatr Radiol 40:681–686
Nievelstein RA, Quarles van Ufford HM, Kwee TC et al (2012) Radiation exposure and mortality risk from CT and PET imaging of patients with malignant lymphoma. Eur Radiol 22:1946–1954
Schafer JF, Gatidis S, Schmidt H et al (2014) Simultaneous whole-body PET/MR imaging in comparison to PET/CT in pediatric oncology: initial results. Radiology 273:220–231
Gatidis S, Schmidt H, Gucke B et al (2016) Comprehensive oncologic imaging in infants and preschool children with substantially reduced radiation exposure using combined simultaneous (1)(8)F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging: a direct comparison to (1)(8)F-fluorodeoxyglucose positron emission tomography/computed tomography. Invest Radiol 51:7–14
Gatidis S, Bender B, Reimold M, Schafer JF (2017) PET/MRI in children. Eur J Radiol 94:A64–A70
Treves ST, Gelfand MJ, Fahey FH, Parisi MT (2016) 2016 Update of the North American Consensus Guidelines for Pediatric Administered Radiopharmaceutical Activities. J Nucl Med 57:15N-18N
Lassmann M, Treves ST (2014) Pediatric Radiopharmaceutical Administration: harmonization of the 2007 EANM Paediatric Dosage Card (Version 1.5.2008) and the 2010 North American Consensus guideline. Eur J Nucl Med Mol Imaging 41:1636
Sah BR, Stolzmann P, Delso G et al (2017) Clinical evaluation of a block sequential regularized expectation maximization reconstruction algorithm in 18F-FDG PET/CT studies. Nucl Med Commun 38:57–66
Vandenberghe S, D’Asseler Y, Van de Walle R et al (2001) Iterative reconstruction algorithms in nuclear medicine. Comput Med Imaging Graph 25:105–111
Riddell C, Carson RE, Carrasquillo JA et al (2001) Noise reduction in oncology FDG PET images by iterative reconstruction: a quantitative assessment. J Nucl Med 42:1316–1323
Gong K, Kim K, Cui J, Wu D, Li Q (2021) The evolution of image reconstruction in PET: from filtered back-projection to artificial intelligence. PET Clin 16:533–542
(2021) R: A language and environment for statistical computing. Vienna, Austria. Version 4.1.1, https://www.rproject.org
Brenner DJ, Doll R, Goodhead DT et al (2003) Cancer risks attributable to low doses of ionizing radiation: assessing what we really know. Proc Natl Acad Sci U S A 100:13761–13766
Lassmann M, Biassoni L, Monsieurs M et al (2007) The new EANM paediatric dosage card. Eur J Nucl Med Mol Imaging 34:796–798
Lassmann M, Biassoni L, Monsieurs M et al (2008) The new EANM paediatric dosage card. Eur J Nucl Med Mol Imaging 35:1748
Marcassa C, Zoccarato O, Calza P, Campini R (2013) Temporal evolution of administered activity in cardiac gated SPECT and patients’ effective dose: analysis of an historical series. Eur J Nucl Med Mol Imaging 40:325–330
Cox CPW, van Assema DME, Verburg FA, Brabander T, Konijnenberg M, Segbers M (2021) A dedicated paediatric [(18)F]FDG PET/CT dosage regimen. EJNMMI Res 11:65
Jones T, Townsend D (2017) History and future technical innovation in positron emission tomography. J Med Imaging (Bellingham) 4:011013
Nardo L, Schmall JP, Werner TJ, Malogolowkin M, Badawi RD, Alavi A (2020) Potential roles of total-body PET/computed tomography in pediatric imaging. PET Clin 15:271–279
Acknowledgements
The authors would like to thank Freya Klein, Josephine Trinckauf, Corina Weyermann, Marlena Hofbauer, Victoria Schober, Edlira Loga, Melanie Thüringer, Nina Bächle, Sabrina Epp, Ana-Mari Gaspar, Michèle Hug, Juliana Koller, Eirini Leivaditaki, and Danijel Tomic for their excellent support.
Furthermore, Dr. Michael Messerli would like to thank Prof. G. K. von Schulthess for his invaluable support in this work.
Funding
Open access funding provided by University of Zurich Dr. Moritz Schwyzer, Dr. Stephan Skawran, Dr. Michael Messerli, and Thomas Sartoretti are supported by a research grant from the Palatin Foundation, Switzerland. Dr. Antonio G. Gennari, Dr. Martin Huellner and Dr. Michael Messerli are supported by a grant from the CRPP “AI Oncological Imaging Network” of the University of Zurich, Switzerland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is PD Dr. Michael Messerli.
Conflict of interest
The University Hospital of Zurich holds a research agreement with GE healthcare (unrelated to the current study). Martin Huellner received speaker’s fees from GE Healthcare. Other than that, the authors declare that they have no competing interests.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
The use for written informed consent was waived for patients scanned before January 2016. After 2016, only patients whose informed consent for further use of their medical data was available (due to a change in national regulatory rules) were included.
Ethical approval
The study was approved by the local ethics committee (Kantonale Ethikkommission Zürich, Switzerland): trial number BASEC 2020-03067.
Study subjects or cohorts overlap
No study subjects or cohorts have been previously reported.
Methodology
• Retrospective
• Observational
• Performed at one institution
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Waelti, S., Skawran, S., Sartoretti, T. et al. A third of the radiotracer dose: two decades of progress in pediatric [18F]fluorodeoxyglucose PET/CT and PET/MR imaging. Eur Radiol 34, 3252–3259 (2024). https://doi.org/10.1007/s00330-023-10319-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-023-10319-6