Abstract
Objectives
This retrospective study aimed to compare the average glandular dose (AGD) per acquisition in breast biopsies guided by contrast-enhanced mammography (CEM), conventional stereotactic breast biopsy (SBB), and digital breast tomosynthesis (DBT). The study also investigated the influence of compressed breast thickness (CBT) and density on AGD. Furthermore, the study aimed to estimate the AGD per procedure for each guidance modality.
Methods
The study included 163 female patients (mean age 57 ± 10 years) who underwent mammography-guided biopsies using SBB (9%), DBT (65%), or CEM (26%) guidance. AGD and CBT data were extracted from DICOM headers, and breast density was visually assessed. Statistical analyses included two-sample t-tests and descriptive statistics.
Results
Mean AGD per acquisition varied slightly among CEM (1.48 ± 0.22 mGy), SBB (1.49 ± 0.40 mGy), and DBT (1.55 ± 0.47 mGy), with CEM presenting higher AGD at lower CBTs and less dose escalation at higher CBTs. For CBT > 55 mm, CEM showed reduced AGD compared to SBB and DBT (p < 0.001). Breast density had minimal impact on AGD, except for category A. The estimated AGD per procedure was approximately 11.84 mGy for CEM, 11.92 mGy for SBB, and 6.2 mGy for DBT.
Conclusion
The study found mean AGD per acquisition to be similar for CEM and SBB, with DBT slightly higher. CEM demonstrated higher AGD at lower CBT but lower AGD at higher CBT, indicating reduced dose escalation with increasing thickness. While breast density had minimal overall impact, variations were noted in category A. DBT was more dose-efficient per procedure due to fewer acquisitions required.
Clinical relevance statement
CEM guidance provides effective lesion visualization within safe radiation limits, improving the precision of percutaneous image-guided breast interventions and supporting its potential consideration in a wider range of breast diagnostic procedures.
Key Points
-
Limited data exist on the AGD using CEM guidance for breast biopsies.
-
CEM and SBB exhibit similar AGD per acquisition; DBT demonstrated the lowest AGD per procedure.
-
Radiation from CEM guidance fits within safe limits for percutaneous image-guided breast interventions.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Contrast-enhanced mammography (CEM) is an emerging imaging technique based on the acquisition of a dual-energy mammogram, which is acquired approximately two minutes after the intravenous injection of iodinated contrast media [1]. CEM consists of a low-energy (LE) image that resembles a conventional 2D mammogram, providing morphologic details, and a recombined image (RC) that displays the distribution of the contrast media, revealing changes in breast perfusion often associated with neoplastic angiogenesis.
Suspicious non-palpable findings typically require histological evaluation, usually performed using breast imaging guidance through techniques such as core-needle or vacuum-assisted biopsies. CEM guidance introduces a novel approach in breast interventional radiology, which employs dual-energy stereotactic acquisitions post-iodinated contrast media administration, enhancing the visualization of suspicious abnormalities. Initial reports confirm the technical feasibility and high success rate of CEM-guided biopsies [2, 3].
Alternative contemporary mammographic methods employed for percutaneous breast interventions include conventional stereotactic breast biopsy (SBB) and digital breast tomosynthesis (DBT) [4, 5]. While both SBB and CEM-guided procedures typically require a minimum of three acquisitions to determine the target coordinates, DBT-guided interventions usually necessitate only a single acquisition [5].
The average glandular dose (AGD), typically measured in milligrays (mGy), is the key metric for evaluating exposure doses in mammographic techniques, reflecting the dose absorbed by mammary tissue [6]. This metric, which can be estimated from technical parameters using the method proposed by Dance et al [7,8,9], is widely recognized in medical imaging and radiotherapy [9,10,11]. Lower AGD values are generally associated with a reduced risk of radiation-induced breast cancer [10, 11].
Comparative studies have analyzed AGD across various mammographic techniques. Routine DBT shows a slight AGD increase over digital mammography (DM) [12, 13], while CEM often exhibits higher radiation levels than DM but may be comparable to or less than DBT [14,15,16]. These differences are associated with factors like breast thickness and density, which correlate with rising AGD [17]. However, such studies typically focus on routine diagnostics, with less emphasis on interventional applications. Early reports on CEM-guided biopsy have revealed a gap in detailed AGD data [2, 3, 18], with recent publications beginning to fill this void, though comprehensive data are still needed [19, 20].
The primary aim of this study was to compare the AGD obtained using CEM guidance with that from other mammography-guided techniques, such as SBB or DBT, exploring its relationship with compressed breast thickness (CBT) and density. We analyzed the AGD for each image acquisition and extended our evaluation to estimate AGD per procedure, based on the typical number of acquisitions by each technique according to our institution’s protocol. Additionally, we assessed the contributions of LE and high-energy (HE) images to the overall radiation dose in CEM guidance.
Methods
Study design and ethics
This retrospective study compared the AGD in patients undergoing breast biopsies through different mammography-guided techniques, including SBB, DBT-guided, and CEM-guided procedures. Informed consent was secured for all procedures, with additional consent for contrast administration in CEM. Patients who underwent CEM-guided biopsy were part of a prospective study [2], approved by the hospital ethics committee (protocol number 2019/8890), for which they provided additional written informed consent for research. For retrospective analyses involving SBB and DBT-guided biopsies, ethical approval was obtained, and the need for additional consent was appropriately waived.
Study population
The study included all consecutive patients who underwent mammography-guided procedures between June 2018 and September 2021, using the same mammographic equipment, specifically the Senographe Pristina™ (GE Healthcare), equipped with automatic optimization of parameters (AOP). Only cases with a complete image dataset available were included. We excluded any cases presenting incomplete datasets, as well as those acquisitions involving spot-magnified views or specimen images, to maintain the data’s integrity and reliability in our analysis.
Interventional mammographic guidance techniques
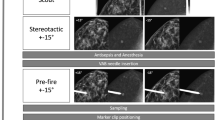
SBB and CEM-guided procedures commence with the acquisition of a scout image, which is captured perpendicular to the image detector to provide an initial overview of the lesion. After this, additional images are acquired at 15-degree intervals, both positive and negative relative to the original scout image plane, to ascertain the three-dimensional coordinates of the lesion, which are essential for precise targeting. After injecting local anesthesia, and especially in cases of subtle findings, we may need to take extra images to compensate for any shifts caused by the anesthesia itself. This check is repeated after inserting the biopsy needle, in a pre-fire mode, to confirm its correct alignment with the lesion. A final image after the biopsy verifies both the successful retrieval of the lesion and the accurate placement of the clip marker. Our prior research on CEM-guided biopsies indicated that, typically, an average of two scout images are required for precise initial alignment [2]. According to our standard protocol, at least eight images are needed per procedure, as shown in Supplementary Fig. 1.
DBT-guided procedures, however, adopt a different strategy by using a single multiplanar scout image to directly estimate the lesion’s depth (z-coordinate), thereby reducing the need for multiple image pairs. This approach usually involves about four images, potentially resulting in a lower overall AGD due to fewer steps and acquisitions [21]. Nonetheless, the actual number of images taken can vary with each modality, influenced by factors like the initial clarity of the lesion and any necessary adjustments for patient movement or lesion shift.
Equipment and AGD assessment
Interventional acquisitions for SBB, DBT, and CEM in this study were performed using the same equipment—the Senographe Pristina™, along with the Serena™ and Serena Bright™ addons (GE HealthCare), all operating under AOP mode. The AOP dynamically adjusts technical factors such as kVp and mA based on the detected properties of breast tissue during an initial scout-view exposure. For instance, the AOP uses lower settings for polymethylmethacrylate (PMMA) phantoms representing breasts with a CBT under 40 mm, while it increases settings for phantoms over 50 mm to effectively image thicker tissues [22, 23]. Notably, for SBB and DBT-guided interventions, the only AOP setting available is an upscaled dose level designated as standard plus (STD +) mode, distinct from the standard (STD) mode used for routine and interventional CEM, routine DM, and routine DBT. Moreover, kVp and filtration settings remain consistent across SBB and DBT modalities. This tailored approach ensures that each modality receives an optimized radiation dose, adjusting for factors such as breast composition and thickness, thus accommodating procedural needs and variations in patient anatomy.
Data collection
Metadata was retrieved from the anonymized DICOM headers of all images acquired during the interventional procedures and transferred to a structured spreadsheet. This dataset encompassed key parameters, including AGD and CBT, facilitating a nuanced analysis of the radiographic exposure and imaging conditions.
Breast density was determined visually in accordance with the breast composition categories of the American College of Radiology (ACR) BI-RADS® mammography lexicon [24], employing routine images corresponding to each interventional case for consistency. In the context of CEM-guided interventions, the low-energy image (LE) was used for density assessment, due to its analogous characteristics to conventional mammography [25]. The evaluations were carried out by a dedicated breast radiologist with 10 years of experience in mammographic imaging and breast density assessment (R.A.).
Statistical analysis
The extracted data were organized according to the biopsy guidance modality used: SBB, DBT, and CEM. Data obtained from each acquisition facilitated a comparative evaluation of the AGD per single acquisition across different modalities. AGD has also been compared per procedure by considering the estimated mean number of acquisitions for each modality. Additionally, AGD was evaluated as a function of breast thickness and within various breast density categories, comparing different guidance modalities. CBT was classified in 5 mm intervals (e.g. the 20 mm category includes thicknesses from 15 mm to 25 mm) and further divided into ‘thin’ (< 35 mm), ‘thick’ (> 55 mm), and ‘mid’ (all other measurements). This categorization was applied to assess the influence of breast thickness on AGD. The AGD repartition between LE and HE exposures was also assessed in the subgroup of patients who underwent CEM guidance.
Statistical analysis was carried out using Minitab v20.3 (Minitab, Inc. 2020. http://www.minitab.com), a commercial computer software. Two-sample Student’s t-tests were employed to compare AGD across different biopsy guidance techniques, with an alpha set at 0.05 for statistical significance. This choice was predicated on the data’s suitability for parametric testing, given the assumption of normal distribution for AGD values and the aim to assess mean differences between modalities. Grouping SBB and DBT together increased the statistical power, and facilitated a direct comparison against CEM, performing the test between two populations, reflecting our interest in evaluating the novel CEM’s performance relative to established techniques. The analysis was conducted on a per-acquisition basis to standardize the comparison and mitigate the impact of procedural variability, thereby providing a more direct assessment of the inherent radiation dose characteristics of each guidance modality.
Results
Included patients
Between June 2018 and September 2021, 177 patients underwent mammography-guided procedures. After excluding 14 patients with incomplete data, the study comprised 163 patients, accounting for 163 biopsies and 1212 mammographic acquisitions. A flowchart outlining the patient and procedure selection is provided in Fig. 1.
Table 1 summarizes the information on the included population. The average patient age was 57 years old and varied little with different guidance techniques (p = 0.292). The overall average CBT for the studied population was 42 mm. While the average CBT for SBB and CEM-guided procedures was consistent with this average, DBT-guided procedures had a slightly but significantly higher average CBT (p < 0.001).
Most patients had breast density classified as either ACR category B or C for all modalities; SBB showed an increased proportion of patients with ACR category A compared to other modalities, while there was no ACR category A in the CEM guidance cohort. In our study, CEM guidance was typically reserved for RC-only findings, which are less commonly observed in category A breasts, known for their less dense tissue. Additionally, category A breasts had a higher mean CBT compared to other categories.
AGD comparisons
Tables 2–4 provide a descriptive analysis of AGD, overall and as a function of breast thickness (binned at 10 mm steps) and density. The overall mean AGD for a single CEM-biopsy acquisition was 1.48 ± 0.22 mGy, which aligns closely with SBB (1.49 ± 0.40 mGy) and DBT (1.55 ± 0.47 mGy) guidance. The marginally higher AGD noted in DBT acquisitions likely stems from the increased CBT observed in this group, as outlined in Table 1.
For all guidance modalities, the AGD increased with increasing breast thickness, though this increase was less pronounced for CEM-guidance (Table 3 and Fig. 2). CEM demonstrated a slightly higher mean AGD in ‘thin’ breasts (CBT below 35 mm): 1.34 mGy (95% CI = 1.29–1.40) compared to 1.05 mGy (95% CI = 1.02–1.09) for the combination SBB + DBT (p < 0.001). Conversely, in ‘thick’ breasts (CBT above 55 mm), CEM showed a slightly lower mean AGD: 1.81 mGy (95% CI = 1.71–1.92) versus 2.13 mGy (95% CI = 2.07–2.20) for the combination SBB + DBT (p < 0.001).
Overall, AGD was consistent across ACR breast density categories for all guidance modalities (Table 4 and Fig. 3), except for breast density category A. This category showed an approximate increase of 0.30 mGy in AGD (p < 0.001), an observation that correlates with a higher mean CBT for this group, as detailed in Table 1.
Per-procedure AGD estimation
Utilising AGD data from Table 2 and adhering to our institution’s biopsy protocols (see Supplementary Fig. 1), we estimated the average AGD for each procedure type. For conventional SBB and CEM-guided biopsies, which typically require a minimum of eight images, the estimated AGDs per procedure were approximately 11.92 mGy and 11.84 mGy, respectively. By comparison, DBT-guided biopsies, needing fewer images at an average of 4 per procedure, yielded a lower estimated AGD per procedure, approximately 6.2 mGy.
AGD repartition in CEM-guided biopsy
In the subgroup of CEM-guided procedures, the contributions of LE and HE acquisitions to the radiation dose were evaluated. On average, a higher percentage of AGD values is assigned to LE images compared to HE images across all CBT categories (Fig. 4). The proportion of the total dose attributed to LE images varies from 54% to 71%, while the remaining 29% to 46% is associated with HE images.
AGD distribution between LE and HE images for CEM-guided biopsies. The left graph displays AGD for LE and HE exposures across breast thickness intervals. The right graph shows the percentage contribution of LE and HE to total AGD. Breast thickness is categorized by ± 5 mm intervals around the central measurement (e.g. 20 mm includes 15–25 mm)
Discussion
Mammography-guided biopsies, traditionally reliant on stereotactic methods, have evolved significantly with the introduction of DBT and CEM. CEM has expanded from an MRI alternative to a promising biopsy guidance tool [26], effectively targeting lesions only visible on RC images [2, 3, 27, 28]. Studies show that up to 40% of such lesions are malignant [2, 28, 29], underlining CEM’s essential role in identifying potentially cancerous abnormalities. Despite these technological advances, comprehensive comparisons of AGD across different biopsy techniques are still lacking.
This retrospective study assessed the AGD per acquisition across three mammographic-guided biopsy techniques: SBB, DBT, and CEM. To mitigate variations in the number of acquisitions and procedures, our analysis focused on per-acquisition data rather than entire procedures due to differences in equipment performance and operator learning curves. Additionally, instances of multiple biopsies in a single session increased image counts, deviating from standard procedural workflows. While overall mean AGD per acquisition values were comparable, detailed analysis revealed that AGD for DBT was higher compared to SBB and CEM, particularly when not adjusted for CBT differences. Importantly, all measured AGDs per acquisition remained below the 3 mGy limit set by the Mammography Quality Standards Act [30].
AGD consistently increased with CBT across all modalities. To address potential biases from varying CBT distributions, we stratified AGD per acquisition by CBT levels. At lower CBT levels (20 mm), CEM biopsies showed higher mean AGD than SBB and DBT, but this difference diminished with increased CBT, showing lower AGD for CEM at thicknesses above 50 mm. This consistent dosing profile within CEM across varying thicknesses can be attributed to its AOP settings. Unlike SBB and DBT, which use ‘STD +‘ mode to enhance image quality at a slightly increased dose, CEM operates in ‘STD’ mode, which requires less dose adjustment for thicker breast tissue. These mode differences likely support the specific AGD trends seen in CEM, suggesting a technical basis for its differential dose responses. AGD was relatively consistent across different ACR breast density categories, irrespective of the guidance modality used, except for a slight increase in category A. This increase could be attributed to the higher mean CBT in this category, necessitating greater radiation doses for effective imaging.
The retrospective nature of this study also constrained a comprehensive per-procedure analysis. This focus on per-acquisition data allowed us to standardize comparisons across modalities, considering procedural complexities. While the estimated AGDs per procedure for SBB and CEM were similar, reflecting comparable acquisition counts, DBT-guided biopsies showed lower AGD but lacked detailed data on lesion enhancement—a key factor that CEM guidance confronts, potentially enhancing biopsy accuracy. Our findings, though specific to the equipment used, enrich the ongoing discourse on AGD in CEM-guided biopsies. Our study estimated the procedural AGD for CEM at approximately 11.84 mGy, which is lower than the findings reported by Tang et al [19] at 14.3 mGy and Sammara et al [20] at 14.8 mGy. While these comparisons provide important benchmarks, the small sample size in Tang’s study [19] and the lack of detailed methodology in both points out the need for cautious interpretation. Our research offers a comprehensive analysis of AGD per acquisition and estimated per procedure, enabling direct comparisons with SBB and DBT guidance. Nevertheless, actual AGD values in clinical practice may vary, reflecting the complexity of real-world scenarios.
Furthermore, the increase in AGD for CEM-guided image acquisition was mostly due to an increase in LE dose, aligning with findings by Genaro et al [14] in routine CEM scenarios. This nuanced understanding of AGD variations reinforces the importance of considering the specific contributions of LE doses in CEM’s radiation profile.
Our study underscores the potential of CEM for decision-making in mammography-guided biopsies. Echoing Amir et al [21], who suggest not limiting DBT biopsies to DBT-specific findings, the application of CEM guidance could broaden to encompass more indications, thus enabling more precise targeting and richer morpho-functional insights. Demonstrating that CEM guidance does not lead to higher radiation doses is vital for its wider implementation, highlighting the need for additional, and extensive prospective studies.
This study provides important insights but is limited by its retrospective design and single-centre scope, using specific equipment. Not all consecutive cases were included due to incomplete datasets. AGD calculations, crucial for assessing radiation dose, are based on ideal conditions not always met in biopsy scenarios, hence the results are indicative rather than definitive. Moreover, relying solely on AGD to assess radiation risk might be inadequate, as it does not account for other influential factors such as glandular tissue volume exposed to radiation and varying patient characteristics, including age. Furthermore, our analysis excludes AGD data for breast density category A in CEM-guided biopsies, as this category was not represented due to the absence of RC-only findings in less dense breasts. This limitation might have influenced our overall AGD results, reinforcing the need for further studies to include all breast densities to ensure comprehensive radiation safety evaluations. Future research should utilise a prospective design, expand across multiple centres, include all densities and ensure comprehensive data collection, and consider additional metrics beyond AGD to enhance radiation risk assessment
Conclusions
In this series, CEM single acquisitions presented AGD values that were on par with those from SBB, and lower than DBT, as demonstrated by the mean AGD metrics obtained using our current imaging equipment. Interestingly, while CEM exhibits higher AGDs in thinner breasts, the increase in AGD with increased CBT is less steep for CEM compared to SBB and DBT, resulting in relatively lower AGD values in thicker breasts. DBT guidance was shown to be more dose-efficient in procedural terms, attributed to fewer image acquisitions. Nevertheless, CEM’s ability to offer enhanced visualization for breast interventions, while adhering to safe radiation parameters, solidifies its potential for expanded clinical application and warrants additional investigative efforts.
Abbreviations
- ACR:
-
American College of Radiology
- AGD:
-
Average glandular dose
- AOP:
-
Automatic optimization of parameters
- CBT:
-
Compressed breast thickness
- CEM:
-
Contrast-enhanced mammography
- CI:
-
Confidence interval
- DBT:
-
Digital breast tomosynthesis
- DM:
-
Digital mammography
- HE:
-
High-energy image
- LE:
-
Low-energy image
- RC:
-
Recombined image
- SBB:
-
Stereotactic breast biopsy
- STD:
-
Standard AOP mode (GE HealthcareTM)
- STD +:
-
Standard plus AOP mode (GE HealthcareTM)
References
Neeter LMFH, Raat HPJF, Alcantara R et al (2021) Contrast-enhanced mammography: what the radiologist needs to know. BJR Open 3:20210034
Alcantara R, Posso M, Pitarch M et al (2023) Contrast-enhanced mammography-guided biopsy: technical feasibility and first outcomes. Eur Radiol 33:417–428 https://doi.org/10.1007/s00330-022-09021-w/Published
Weaver OO, Yang WT, Scoggins ME et al (2022) Challenging contrast-enhanced mammography-guided biopsies: practical approach using real-time multimodality imaging and a proposed procedural algorithm. AJR Am J Roentgenol. https://doi.org/10.2214/AJR.22.28572
Ames V, Britton PD (2011) Stereotactically guided breast biopsy: a review. Insights Imaging. https://doi.org/10.1007/s13244-010-0064-1
Schrading S, Distelmaier M, Dirrichs T et al (2015) Digital breast tomosynthesis-guided vacuum-assisted breast biopsy: initial experiences and comparison with prone stereotactic vacuum-assisted biopsy. Radiology. https://doi.org/10.1148/radiol.14141397
Salomon E, Homolka P, Semturs F et al (2019) Comparison of a personalized breast dosimetry method with standard dosimetry protocols. Sci Rep 9:5866. https://doi.org/10.1038/s41598-019-42144-7
Dance DR, Skinner CL, Young KC et al (2000) Additional factors for the estimation of mean glandular breast dose using the UK mammography dosimetry protocol. Phys Med Biol 45:3225–3240
Dance DR (1990) Monte-Carlo calculation of conversion factors for the estimation of mean glandular breast dose. Phys Med Biol 35:1211–1219
van Engen RE, Bosmans H, Bouwman RW et al (2018) Protocol for the quality control of the physical and technical aspects of digital breast tomosynthesis systems version 1.03. European Reference Organisation for Quality Assured Breast Screening and Diagnostic Services (EUREF), Germany
Hendrick RE (2010) Radiation doses and cancer risks from breast imaging studies. Radiology 257:246–253. https://doi.org/10.1148/radiol.10100570
Yaffe MJ, Mainprize JG (2011) Risk of radiation-induced breast cancer from mammographic screening. Radiology 258:98–105. https://doi.org/10.1148/radiol.10100655/-/DC1
Dance DR, Young KC, Van Engen RE (2011) Estimation of mean glandular dose for breast tomosynthesis: factors for use with the UK, European and IAEA breast dosimetry protocols. Phys Med Biol 56:453–471. https://doi.org/10.1088/0031-9155/56/2/011
Ui Shin S, Min Chang J, Sun Bae M et al (2015) Comparative evaluation of average glandular dose and breast cancer detection between single-view digital breast tomosynthesis (DBT) plus single-view digital mammography (DM) and two-view DM: correlation with breast thickness and density. Eur Radiol 25:1–8. https://doi.org/10.1007/s00330-014-3399-z
Gennaro G, Cozzi A, Schiaffino S et al (2022) Radiation dose of contrast-enhanced mammography: a two-center prospective comparison. Cancers. https://doi.org/10.3390/cancers14071774
Phillips J, Mihai G, Hassonjee SE et al (2018) Comparative dose of contrast-enhanced spectral mammography (CESM), digital mammography, and digital breast tomosynthesis. AJR Am J Roentgenol 211:839–846. https://doi.org/10.2214/AJR.17.19036
James JR, Pavlicek W, Hanson JA et al (2017) Breast radiation dose with CESM compared with 2D FFDM and 3D tomosynthesis mammography. AJR Am J Roentgenol 208:362–372. https://doi.org/10.2214/AJR.16.16743
Fusco R, Raiano N, Raiano C et al (2020) Evaluation of average glandular dose and investigation of the relationship with compressed breast thickness in dual energy contrast enhanced digital mammography and digital breast tomosynthesis. Eur J Radiol 126:108912. https://doi.org/10.1016/j.ejrad.2020.108912
James J (2022) Contrast-enhanced spectral mammography (CESM)-guided breast biopsy as an alternative to MRI-guided biopsy. Br J Radiol. https://doi.org/10.1259/bjr.20211287
Tang YC, Cheung YC (2023) Contrast-enhanced mammography-guided biopsy: technique and initial outcomes. Quant Imaging Med Surg 13:5349354–5345354. https://doi.org/10.21037/QIMS-23-137
Sammarra M, Piccolo CL, Sarli M et al (2024) Contrast-enhanced mammography-guided biopsy: preliminary results of a single-center retrospective experience. J Clin Med 13:933
Amir T, Zuckerman SP, Barufaldi B et al (2021) Comparison of radiation dose between 2D digital stereotactic versus digital breast tomosynthesis-guided breast biopsies. Eur J Radiol 134:109407. https://doi.org/10.1016/j.ejrad.2020.109407
GE Healthcare. Pristina Serena™ Operator Manual, 5845701-8EN, Rev-4. Published July 31, 2023. Available at: https://www.gehealthcare.com/support/manuals?search=eyJzZWFyY2hUZXJtIjoiNTg0NTcwMS04RU4iLCJsYW5ndWFnZU5hbWUiOiJFbmdsaXNoIChFTikifQ%3D%3D. Accessed 16 April 2024. Specific sections referenced: Chapter 11, Acquiring Biopsy Images, Sections 1-2, pp. 103–104.
GE Healthcare. Pristina Serena™ Bright Operator Manual, 5845678-8EN, Rev-3. Published December 07, 2022. Available at: https://www.gehealthcare.com/support/manuals?search=eyJzZWFyY2hUZXJtIjoiNTg0NTY3OC04RU4iLCJsYW5ndWFnZU5hbWUiOiJFbmdsaXNoIChFTikifQ%3D%3D. Accessed 16 April 2024. Specific sections referenced: Chapter 7 Acquiring CESM Biopsy Images pp. 35–38.
Sickles, EA, D’Orsi CJ, Bassett LW et al (2013) ACR BI-RADS® mammography. In: ACR BI-RADS® Atlas, breast imaging reporting and data system, 5th edn. American College of Radiology, Reston
Lalji UC, Jeukens CRLPN, Houben I et al (2015) Evaluation of low-energy contrast-enhanced spectral mammography images by comparing them to full-field digital mammography using EUREF image quality criteria. Eur Radiol 25:2813–2820. https://doi.org/10.1007/s00330-015-3695-2
van Nijnatten TJA, Morscheid S, Baltzer PAT et al (2024) Contrast-enhanced breast imaging: current status and future challenges. Eur J Radiol 171:111312. https://doi.org/10.1016/j.ejrad.2024.111312
Kornecki A, Bhaduri M, Khan N et al (2023) Contrast-enhanced mammography-guided breast biopsy: single center experience. AJR Am J Roentgenol. https://doi.org/10.2214/AJR.22.28780
Aribal E, Guldogan N, Seker ME et al (2024) MRI only detected lesions: Can contrast enhanced mammography guided biopsy be an alternative method: Initial clinical findings. Eur J Radiol 173:111373. https://doi.org/10.1016/j.ejrad.2024.111373
Kim G, Mehta TS, Brook A et al (2022) Enhancement type at contrast-enhanced mammography and association with malignancy. Radiology 305:299–306. https://doi.org/10.1148/radiol.210966
MQSA (2001) The Mammography Quality Standards Act final regulations: preparing for MQSA inspections; final guidance for industry and FDA. FDA, Washington
Acknowledgements
We would like to thank all the women participating in the study, as well as the radiology team and radiographers from the Hospital del Mar, Barcelona, Spain. This article is part of Rodrigo Alcantara Souza’s doctoral thesis in the Doctoral Programme in Medicine at the Department of Medicine, Universitat Autònoma de Barcelona, Spain.
Funding
Open Access Funding provided by Universitat Autonoma de Barcelona.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. R. Alcantara.
Conflict of Interest
The authors of this manuscript declare relationships with the following companies: R.A. declares to be a member of the speakers’ bureau for Bard BD and GE Healthcare. V.I. declares to be a member of the speakers’ bureau for GE Healthcare. P.M. is a GE Healthcare employee. All other authors declare that they have no competing interests.
Statistics and biometry
One of the authors (X.C.) has significant statistical expertise.
Informed consent
Written informed consent was obtained from all prospectively enroled subjects (patients) in this study. For retrospective analyses involving SBB and DBT-guided biopsies, ethical approval was obtained, and the need for additional consent was appropriately waived.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
The CEM guidance cohort was previously analyzed for a different purpose and published at https://doi.org/10.1007/s00330-022-09021-w. The current analysis was not previously performed. The retrospectively assessed cohort has not been previously reported.
Methodology
-
Retrospective review of prospectively enroled cohort
-
Performed at one institution, single equipment
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alcantara, R., Azcona, J., Pitarch, M. et al. Breast radiation dose with contrast-enhanced mammography-guided biopsy: a retrospective comparison with stereotactic and tomosynthesis guidance. Eur Radiol (2024). https://doi.org/10.1007/s00330-024-10920-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00330-024-10920-3