Abstract
Modern coral reefs face substantial threats that endanger their capacity to function and provide resources for human populations. Chronic human pressure through exploitation and land-based pollution has led to reef degradation and lower productivity. More recently, this pressure is compounded with successive episodes of coral bleaching across the tropical oceans. However, response trajectories of reef resources to exploitation, habitat degradation, and local management may not be straightforward, as these ecosystems are incredibly complex. We employed a comprehensive and standardized survey of parrotfish assemblages within reef systems in Guam to examine trajectories of biomass and function across a decade of change. Parrotfishes represent one of the most commercially and ecologically important coastal resources throughout Micronesia. We found that parrotfish biomass declined by nearly 30% over the course of ten years, with concomitant decreases in grazing and bioerosion rates at a similar magnitude. There was evidence of fishery exploitation playing a role, but overall patterns were not always aligned with the hypothesis of fishery-induced decline. For example, two-thirds of marine-protected area sites declined in biomass and mean body length was stable for several highly targeted species. Further, most biomass decline stemmed from small species with higher resilience to fishery exploitation, whereby three of the most vulnerable fishery target species either maintained or increased total biomass. The ultimate drivers of parrotfish assemblage trajectories in Guam are likely a complex mixture of exploitation, habitat change from multiple stressors, and responses to management measures. However, the potential future decline in fishery production and ecological function is substantial and merits continuous monitoring and proactive management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the Pacific Islands, coral reefs have provided coastal fishery resources to human populations for thousands of years (Kirch and Hunt 1997). However, human populations have increased dramatically in this region over the past century, and this growth has been accompanied by modernization of fishing practices, collectively straining the sustainability of marine resources. In more recent history, environmental degradation from land development and the effects of climate change have placed the sustainability of coastal coral reef resources in jeopardy in many regions (Maragos 1993; Munday et al. 2008; Hughes et al. 2017).
The coral reef ecosystems surrounding the US Pacific Island Territory of Guam have a long history of documented disturbance, largely driven by increasing pressure from coastal development and human population increase (Maragos 1993). Military construction before, during, and after World War II (1939–1945) represented an unprecedented period of coastal change through dredging, filling, and other land modifications. Continued development since that period has led to high rates of sedimentation in Guam’s southern reefs where highly erodible soils are discharged via river systems and coastal run off (Richmond 1993), leading to substantially reduced coral cover in many areas. The use of motorboats and modern fishing techniques emerged in the 1960s, and night-time spearfishing with SCUBA became the primary source of commercially harvested reef fish in the 1980s (Myers 1993), disproportionally affecting large-bodied reef fish. However, the recent decade has been particularly dynamic in new ways, as Guam’s coral reefs have experienced successive heatwaves leading to mass bleaching and subsequent mortality of heat-intolerant corals (Reynolds et al. 2014; Raymundo et al. 2019), as well as an increased participation in commercial fish harvesting (Western Pacific Regional Fishery Management Council 2016). Conversely, after decades of debate, recent legislation (March 2020) banned the use of SCUBA spearfishing in Guam. Further, the marine-protected area (MPA) network, established in 2001 and covering approximately 35 km2 of reef habitat, has existed for a 20-year duration, suggesting the MPAs should now show considerable returns in fish production (Claudet et al. 2008). These contrasting environmental and management drivers spell an uncertain future for Guam’s coral reef health and productivity.
Coral reef fishes, in particular, represent a culturally and commercially important resource in Guam. Despite this, there is limited understanding of fishery production and patterns of fish functional diversity across Guam’s reefs through space or time. Despite a long history of marine research in Guam, including several long-standing fishery-dependent surveys of coastal fish catches (e.g., Zeller et al. 2007; Sundberg et al. 2015), there have been no fishery-independent monitoring programs designed to inform managers on the temporal or spatial trends of commercially important species. Such monitoring efforts over long timescales represent the most valuable method for documenting change in the marine environment and for understanding the potential drivers of change (Flower et al. 2017).
Parrotfishes (Labridae: Scarinae) are among the most important harvested reef fish families in Guam for literally thousands of years (Amesbury & Hunter-Anderson 2008), with the combined harvested weight of parrotfish species representing approximately 30% of the coastal fishery (Houk et al. 2012). Parrotfishes are also ubiquitous and diverse members of coral reef fauna across the tropical Indo-Pacific and are ecologically important because of their ability to scrape and bioerode the reef benthos via feeding on euendolithic microalgae and microbes (Clements et al. 2017). Their feeding habits elicit unique population-level responses to habitat disturbances, whereby short cycles of disturbance to coral communities can generate positive responses in parrotfish numbers by creating short-term successional environments that bolster nutritional resources (Russ et al. 2015; Graham et al. 2020; Fidler et al. 2021).
Given the dynamic changes that have occurred in Guam across the recent decade, this study employs a comprehensive fish and benthic survey of the outer reef slopes surrounding Guam, following standardized protocols from an identical survey conducted a decade prior in 2011–2012 (Taylor et al. 2015), to examine trajectories of parrotfish assemblages across recent history. Our objectives were to (1) determine the trajectories and spatial patterns of parrotfish biomass and assemblage structure in recent years, (2) determine the relative magnitude of anthropogenic or environmental factors structuring parrotfish assemblages, (3) examine trends in mean body size for common species, and (4) integrate these data streams to estimate change in parrotfish functional rates (i.e., grazing and bioerosion) through time. This study is intended to examine variation at the within-island scale and, in doing so, both inform regional fisheries managers and policy makers as well as to advance our understanding of the influence of various factors on parrotfish assemblages.
Methods
Study area and surveys
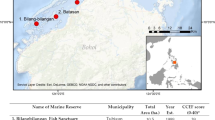
We conducted a survey of parrotfish assemblages on the outer reef slopes of the island of Guam, replicated across a ten-year interval. Sites were initially selected across a gradient of environmental and anthropogenic pressures, including within and outside of four marine-protected areas. A total of 17 sites (Fig. 1) was surveyed using diver-operated stereo-video technology. At each site, we surveyed fish assemblages stratified across two depths (6–10 m and 18–20 m). Surveys were originally completed in the summers of 2011–2012 (Taylor et al. 2014, 2015), and the sites were resurveyed in summer months of 2021. Each survey conducted represented a timed swim transect at depth (24-min surveys in 2011–2012, 30-min surveys in 2021) whereby the diver towed a surface GPS to calculate distance traveled during the survey, which was later converted to reef area based on a 5-m survey width (average area of 0.513 ha in 2011/2012, 0.592 ha in 2021). Individual fish within 8 m in front and 2.5 m either side of the camera trajectory were identified down to the species level, annotated by color phase (where applicable), and measured to the nearest millimeter (fork length) using the EventMeasure software (www.seagis.com.au). The number of parrotfishes at each depth within site was converted to biomass densities per hectare. Body mass (g) for individuals was calculated based on known length–weight relationships for species from Guam (Taylor and Choat 2014, Taylor unpublished data).
To identify drivers of parrotfish biomass and assemblage structure, we quantified environmental variables from video data. This included reef slope (0–90 degrees, mean of multiple measurements per survey using video frames at random intervals), cover of live coral (mean of 10 estimates per survey on a 1–5 scale), and structural rugosity (mean of 10 estimates per survey on a 1–5 scale). Classifications of coral cover and substrate rugosity followed Johnson et al. (2019). A site-level fishing pressure index (fpi) for parrotfish harvests was derived from creel survey data (a standardized shoreline and boat basin survey of fishing effort and catch composition) collected by the Guam Division of Aquatic and Wildlife Resources, following Taylor (2014). The index used the total fishing trips targeting parrotfishes by geographic sector and combined shore-based and boat-based surveys using a scaling factor to account for relative differences in catch between the two fisheries. Site-level values were divided by associated reef area and log-transformed. Wave exposure at each site was estimated using a time series of wind-speed records and angles of exposure, following Houk et al. (2015). Finally, we quantified adjacent reef type (e.g., coastal habitat that separates the land from the outer reef slope) categorically as either lagoon, reef flat, or fringing landmass.
Analyses
Biomass
We examined the influence of explanatory factors on the total biomass (summed across all species; kg ha−1) of parrotfishes in Guam across sites, depths, and through time using model selection in a linear mixed effects model framework. Explanatory factors were scaled and centered and checked for distributions. Being heavily skewed to smaller values, coral cover was natural log-transformed. Because we expected the influence of fishing pressure to vary across depth in a primarily spear-based fishery, we included an interaction between fpi and depth. In the original model, square-root-transformed parrotfish total biomass was predicted by fixed factors year, fpi, depth, exposure, slope, coral, rugosity, and adjacent reef type. Depth and year were nested within random factor site to account for non-independence.
As the change across time periods was an important factor, we followed this analysis with another model to examine temporal drivers of change in biomass. Past values were subtracted from present values for all continuous variables that differed over time (Δ parrotfish biomass, Δ coral, and Δ rugosity) and considered along with fpi (data resolution was not high enough to detect change in the distribution of fishing effort across the time period), exposure, slope, and adjacent habitat. For this model, depth was nested within random factor site.
Assemblage structure
We investigated variation in parrotfish assemblage structure (biomass densities; i.e., the percent contribution of each species to the whole) in Guam across sites and through time using redundancy analysis (RDA). This analysis was performed using the “vegan” package in R version 4.2.1 (Dixon 2003). A reduced species assemblage using the principle 12 species identified in Taylor et al. (2014) was square-root-transformed and standardized by the total. Explanatory variables were treated as before for univariate models and were tested for multicollinearity using variance inflation factors (vif). No variables were excessively collinear (vif > 5), and thus, all were used in the model formulation.
We complemented this analysis with a coarser view of the hierarchical structuring of parrotfish assemblages using multivariate regression trees (MRT; De’ath 2002). MRT repeatedly splits the data into sites clustered by similarity across breakpoint thresholds in environmental factors, and the optimal number of splits is determined by cross-validating the relative errors. MRT was carried out using the same set of predictor variables as RDA above.
Body size
To examine decadal trends in parrotfish body size, we fit linear models to species-specific changes in body size over time (i.e., against factor year) using individual body size measurements derived from stereo-video surveys. The number of observations varied widely among species and between years and statistical significance is influenced by sample size. Hence, for species with significant trends through time, we ran 10,000 simulations whereby the annual sample sizes were bootstrapped with the lowest sample size for that species. We then examined what proportion of simulations resulted in significant differences.
Functional extrapolation
Grazing end erosion rates for each individual parrotfish > 10 cm were calculated using species-specific functions of the form:
FL is the fork length of the parrotfish as measured during stereo-video analysis. If the video analysis could not determine the exact length of a fish, average length for the same species, site, and (if available) depth was used. Coefficients a and b were calculated from species- and size-specific feeding metrics (bite rates [bpm], percentage of bites leaving scars [%scars], and bite area [area] or volume [vol]) collected in the central Indian Ocean (Lange et al. 2020), the Red Sea, Indonesia, and the Great Barrier Reef (Robinson et al. 2020) following procedures described in Lange et al. (2020). In short, allometric models were fitted for each species and feeding metric (power functions for bpm, area, and vol, logarithmic functions for %scars). Then, 1000 possible regression lines were computed within the error probability distribution of each fitted model, and their coefficients were extracted (package “mass”). Using random coefficients from each metric, the calculation of grazing and bioerosion rates for fish sizes 10 to 60 cm following formulas (ii) and (iii) was repeated 1000 times and a power function was fitted to data from each iteration.
To average period of daylight over the year, 12 h was used for calculations. Proportion of daytime feeding was adapted from Bellwood (1995): 83.3% for large parrotfish (Cetoscarus ocellatus, Chlorurus microrhinos, Scarus rubroviolaceus) and 87.7% for smaller species (Chlorurus sordidus and other Scarus spp.). For (iii), an average substrate density of 1.52 ± 0.19 g cm−3 was used.
The resulting distribution of model coefficients (n = 1000) was used to extract the mean and standard deviation for the final grazing/bioerosion regression coefficients used in formula (i) (ESM Table S1). Total annual grazing and erosion functions (m2 or kg m−2 reef yr−1) at each site, depth, and year were then calculated by summing rates of individual fish and dividing by the area surveyed.
Results
Trends in total parrotfish biomass
Across 17 sites and two depths, we surveyed 7978 individual parrotfishes within 8.73 hectares in 2011/2012 and 7307 individual parrotfishes within 10.06 hectares in 2021. These values equate to 159 kg ha−1 in 2011/2012 and 113 kg ha−1 in 2021. A total of 19 species were observed during the 2011/2012 survey period compared to 17 during 2021. The most common species observed were Chlorurus spilurus, Scarus psittacus, and Scarus schlegeli, whereas the species with the highest biomass densities were C. spilurus, S. psittacus, and Scarus altipinnis. The optimal model predicting variability in biomass included factors year, coral, depth, fpi, and an interaction between depth and fpi (Electronic Supplementary Material Fig. S1). Year was the most influential factor, whereby mean parrotfish biomass decreased by 28.8% from 2011/2012 to 2021 (Fig. 2a). Beyond this temporal decline, we found that biomass was lower by 22.4% in deep (18–20 m; lower biomass) versus shallow (6–10 m; higher biomass) sites (Fig. 2a). Variation in ratios of biomass between depths varied across decadal time points did not follow a predictable pattern. The influences of coral and fpi were not statistically significant; however, there was a significant interaction between depth and fpi, whereby parrotfish biomass was negatively correlated with fishing pressure across shallow sites but not across deep sites (Fig. 2b). At the species level, all but four species decreased in biomass. Hipposcarus longiceps (a large-bodied, schooling species common in fishery harvests) and Scarus fuscocaudalis (a small-bodied, rare species generally found at greater depths) increased by approximately 60%, whereas Chlorurus frontalis (a large-bodied, schooling species common at shallow depths and in fishery harvests) and Chlorurus microrhinos (a large-bodied species common in fishery harvests) increased marginally by 10 and 2%, respectively.
Patterns of parrotfish biomass on outer reef slopes of Guam across time and depth (top panel). On average, total biomass decreased by 28% from 2011/2012 to 2021 and by 22% from shallow to deep sites. Relationships between fishing pressure index and parrotfish biomass (bottom panel) demonstrated an interaction between fishing pressure and depth
Only the change in coral cover (Δ coral) correlated with the change in site-specific parrotfish biomass over time (χ21,16 = 4.73, P = 0.0296). We found that Δ parrotfish biomass decreased with increasing Δ coral (Fig. 3). In total, 6 sites decreased in coral cover by more than 0.3 relative points (on a scale of 1–5), and 13 sites increased by more than 0.3 relative points. All six sites with decreased coral cover were shallow. Eleven of the thirteen sites with increased coral cover were dominated by the fast-growing species Porites rus. Examination of parrotfish biomass trends at the site level reveals interesting patterns. The two most inaccessible marine-protected area sites (based on ease of surveillance by conservation officers or inaccessibility of sites) predictably had the highest rates of biomass increase. However, among the highest rates of biomass decline occurred at two other marine-protected area sites potentially associated with a greater frequency of poaching activity.
Trends in parrotfish assemblages
Despite a nearly 30% decline in biomass between 2011/2012 and 2021, parrotfish assemblage structure remained largely constant across the decade (Fig. 4). The combined decadal trajectories in deep sites were negligible (Fig. 4a), whereas shallow sites overall showed slight trajectory aligned with higher fishing pressure. However, year was a highly insignificant factor in the RDA model (F1,59 = 0.56, P = 0.768). The overall RDA model explained 27.9% of the variation in parrotfish assemblage structure, with the first two axes highly significant. Factors exposure (F1,59 = 8.95, P < 0.001) and depth (F1,59 = 8.83, P < 0.001) were the most influential, while fpi (F1,59 = 2.83, P = 0.008), rugosity (F1,59 = 2.46, P = 0.027), and adjacent habitat (fringe [F1,59 = 5.26, P < 0.001] and lagoon [F1,59 = 3.13, P = 0.007]) were also significant drivers of parrotfish assemblage structure. The most influential parrotfish species delineating assemblages in Guam were Scarus rubroviolaceus (associated with sites of low fishing pressure and high exposure), Scarus forsteni, Scarus schlegeli (associated with deep depths and adjacent lagoons), and Chlorurus spilurus (associated with sites of high rugosity).
Redundancy analysis (RDA) demonstrating patterns and drivers of biomass-weighted parrotfish assemblage structure across 17 sites and two time periods on outer reef slopes of Guam, Micronesia. Vectors demonstrate the strength (length), direction, and statistical significance (black, P < 0.05; gray, P > 0.05) of explanatory factors. Fish images depict the four most important species defining differences across assemblages (Scarus rubroviolaceus [S.rub], Scarus forsteni [S.for], Chlorurus spilurus [C.spi], and Scarus schlegeli [S.sch]). Plots are split by deep and shallow sites for clarity, and site trajectories across time are displayed with dashed lines from 2011/2012 (yellow dots) to 2021 (blue dots)
Results of the MRT analysis were highly reflective of the RDA, with factors exposure and depth hierarchically accounting for most of the structure in the data set (ESM Fig. S2). Sites were initially split based on high and low exposure values (accounting for 10.4% of the variation), and then high and low exposure subsets were split by depth categories (5.9 and 5.6% of variation on either side). Beyond this, fringe habitat and fpi were important factors. The total model explained 30.8% of the variability in parrotfish assemblages. At low exposure, shallow depths sites, the influence of fpi on the life-history trait variation of parrotfish assemblages is evident, whereby high fishing pressure leads to assemblages with a low proportion of larger-bodied, later-maturing species.
Trends in parrotfish mean body size
We examined change in parrotfish size distributions across the decade for 13 species with adequate size observations (Fig. S3). Despite considerable declines in biomass for most species, most species’ size distributions remained unchanged. We identified significant increases in mean size for C. microrhinos and S. forsteni, and significant decreases in mean size for H. longiceps, S. altipinnis, and S. psittacus. Iterations of bootstrapped simulations using equal sample size across years (with n set to the year with less observations) demonstrated that P < 0.05 for 100% of simulations for all species except C. microrhinos (72% of simulations). Species-specific patterns of change were largely corroborated with trends observed in the Guam Commercial Fisheries Biosampling database (Sundberg et al. 2015) across a similar period (2009–2017).
Trends in parrotfish functional metrics
Predicted annual rates of parrotfish grazing and bioerosion declined by 26 and 27%, respectively, between 2011/2012 and 2021. Patterns of decline were similar across shallow and deep sites (Fig. 5). Because parrotfish assemblage structure and most species-specific body size distributions did not change considerably across the time period, reductions were driven by overall decreases in parrotfish biomass rather than shifts in species composition or biomass contributions stemming from size relationships. Hence, site-specific temporal trends in functional rates largely mirrored those of parrotfish biomass.
Discussion
This study demonstrates the factors influencing parrotfish assemblages at the island scale through space and time, as well as the relative strength of these factors and the level at which assemblages vary. However, perhaps the most important result of this study is that modern parrotfish assemblages in Guam have reduced in biomass by nearly 30%, leading to considerable consequences for the magnitude of important functions that parrotfishes provide in Guam’s reefs. Interestingly, patterns of change in parrotfish biomass and mean size do not manifest a straightforward story of habitat decline or overfishing as the only causes. Rather, the patterns identified likely reflect the dynamic recent history of Guam’s reef systems regarding increased fishing pressure (Western Pacific Regional Fishery Management Council 2016) followed by new management strategies (e.g., the cessation of SCUBA spearfishing), repeated coral bleaching (Raymundo et al. 2019), and the duration and variable poaching history of the MPA network.
Specifically, analyses of total biomass over time did not appear to follow a strong site-specific pattern associated with fishing pressure, as the fishing pressure index was a good indicator of total biomass only in shallow sites. However, this depth interaction masked some simple patterns across the protection status of sites. While 65% of the sites were unprotected, 70% of the sites that had a decline of > 10% were unprotected; conversely, while only 35% of the sites were protected, 63% of the sites with a > 10% increase in total biomass were within protected areas. Hence, clearly there is a signature of effective protection from fishing on the biomass trajectories of reefs surrounding Guam. Further, the two sites with the greatest increase in biomass over time represented the most well-protected MPAs (Pati Point and Piti), while the two more heavily poached MPAs showed considerable declines in parrotfish biomass (Tumon and Achang). Although the fishing pressure index was a significant predictor of biomass decline only for shallow sites, there were nearly equal numbers (12 deep, 11 shallow) of sites, including four deep MPA sites, that reduced in biomass by > 10%. Overall, only 3 of 17 sites (shallow and deep combined; 2 of these representing MPAs) increased in parrotfish biomass over the time period. This demonstrates that other environmental factors are facilitating the ongoing decline of parrotfish biomass in Guam. Despite the negative trend, it is important to place parrotfish biomass into biogeographic perspective, as estimates vary widely across the tropical Pacific region. Comparing with reef systems using identical survey protocols, recent estimates of parrotfish biomass (\(\overline{x}\) = 113 kg/ha ± 63 SD; range 13–310 kg/ha at site level) are relatively average among most Micronesian islands (Taylor et al. 2015; range 16–308 kg/ha at site level), but considerably lower than nearby Yap (Taylor et al. 2015; \(\overline{x}\) = 254 kg/ha ± 132 SD; range 148–524 kg/ha at site level) and the northern Great Barrier Reef (Johnson et al. 2019; \(\overline{x}\) = 220 kg/ha ± 226 SD; range 4–1175 kg/ha at site level). Compared with other studies using belt transects, recent Guam biomass estimates are considerably higher than those of US Pacific islands of Oahu, Hawaii (Howard et al. 2009; ~ 4–38 kg/ha at habitat level), and Tutuila, American Samoa (Page 1998; ~ 63–90 kg/ha at habitat level).
When we focused on site-specific trends in biomass across the decade, only changes in coral cover were correlated with changes in biomass. This relationship was negative (i.e., lower coral cover yielded higher parrotfish densities), which reflects patterns observed elsewhere (Gilmour et al. 2013; Russ et al. 2015; Emslie and Pratchett 2018; Questel and Russ 2018; Graham et al. 2020) associated with short-term disturbance to coral communities. Parrotfishes target euendolithic microalgae that often proliferate during early successional change after the death of corals (Clements et al. 2017), and therefore, the enhanced nutritional yield after disturbance can benefit parrotfish growth rates (Taylor et al. 2020). Estimates of coral cover decreased slightly on average in shallow sites (also see Raymundo et al. 2019) but increased in deep sites. Disentangling these patterns suggests different drivers of parrotfish biomass in shallow versus deep sites. In shallow areas, despite decreases in coral cover associated with bleaching and other stressors, higher fishing pressure may have led to decreased parrotfish biomass (e.g., Fig. 2b). At deep sites, increases in coral cover (largely attributed to monotypic reef builders like Porites rus) were associated with declines in total parrotfish biomass, while the impact of fishing pressure was less pronounced. However, causal relationships are tenuous to determine here, and trajectories of parrotfish biomass associated with benthic communities are complex across time. The generally positive response of parrotfishes to benthic disturbance is a short-term and cyclical effect and will depend on the scale, frequency, and type of disturbance (Emslie and Pratchett 2018; Taylor et al. 2020). Sustained degradation of habitats will not bode well for parrotfish populations in the long term without effective fishery management policies (Houk et al. 2022).
A nearly 30% decline in parrotfish biomass on outer reef slopes of Guam translates to declines of 26 and 27% in parrotfish grazing and bioerosion rates on benthic substrates. Ultimately, the decadal trend in Guam likely reflects negative consequences for most core functional processes on local reefs (Brandl et al. 2019), and reversal of this trend should be a management priority. Fortunately, the management measures likely most beneficial for reef fish communities went into place in 2020, when Guam’s legislature passed a bill banning the use of night-time SCUBA spearfishing. This practice was common in Guam for at least 35 years and represented the primary source of reef fish in the commercial markets (Myers 1993; Houk et al. 2012; Lindfield et al. 2014). In other regions, baseline monitoring of fish stocks at the onset of a SCUBA-based fishery quickly demonstrated negative impacts to coastal fishery resources, thereby mustering the political will to ban this practice (Green 2003). However, this baseline monitoring for fishery resources has not existed in Guam and therefore has not been available to local resource managers. Given the recent ban on SCUBA spearfishing, we expect that implementing our survey design at a higher frequency (e.g., every 2–3 years) will enable us to monitor the recovery of parrotfish populations across the next one or two decades. Standardized monitoring programs are important to inform resource managers and politicians about the trends of biological resources and the impacts of activities or management measures that are implemented. Importantly, fish populations are natural resources that do not operate on political timescales: the life histories of coral reef fishes facilitate a mismatch between potentially rapid rates of extraction and much slower rates of recovery (Taylor et al. 2012).
While parrotfish assemblages in Guam were relatively stable across the survey time periods, many factors were found to hierarchically influence assemblage structure across space. While depth, fishing pressure, and adjacent habitat type were all important, wave exposure was the most important factor delineating populations at the within-island site scale. Wave exposure is an important environmental factor that not only physically structures coral reef environments, but also influences the behavioral aspects of herbivore communities (Bejarano et al. 2017), the detrital and microbial composition of benthic substrates (Crossman et al. 2001, Tribollet et al. 2008), and their trajectories of recovery after disturbance (Lange et al. 2021). High-wave exposure limits the diversity of corals and the structural complexity of shallow water environments (Dollar 1982), and this influences fish populations at the island scale. Northeast facing coasts of Guam experience high wave energy for much of the year and, as a result, contain a reduced diversity of parrotfishes dominated by important fishery targets Scarus rubroviolaceus and Chlorurus frontalis. Wave exposure shapes fish communities in the Main Hawaiian Islands (Friedlander et al. 2003), it is the most considerable factor structuring parrotfish assemblages across the entire northern Great Barrier Reef (Gust et al. 2001; Gust 2002; Johnson et al. 2019), and yet its relative influence is low across islands of Micronesia where island geomorphology differs substantially (Taylor et al. 2015). Hence, the magnitude of influence of wave exposure is dependent on the scale of observation.
Species-specific patterns of mean size and biomass densities across the past decade did not reflect an expected biomass decline caused by overexploitation. Parrotfish assemblages have a clear gradient of life-history characteristics across species that can reflect and predict their vulnerability to overexploitation (Jennings et al. 1999; Taylor et al. 2014). Two characteristics of exploited fisheries are decreases in mean harvested size, especially among larger-bodied species, and ‘species replacements’ whereby the relative biomass of vulnerable larger-bodied species decreases and smaller-bodied, more resilient species become proportionally more common. Both of these trends have been observed historically in Guam (Lindfield et al. 2014; Taylor et al. 2014). However, only one species showed a pattern clearly reflective of overharvesting (Scarus altipinnis: 4th highest vulnerability score, significant decrease in body size, and 35% decrease in biomass density). Chlorurus microrhinos—the second most vulnerable species in Guam based on life-history traits—increased in mean body size and remained stable in biomass, despite historical declines in both variables prior to the survey period. Hipposcarus longiceps—the third most vulnerable species (Taylor and Cruz 2017; Moore 2022)—had a significant decrease in body size but a 60% increase in biomass across time, suggesting that heavy recruitment of juveniles is driving population recovery. Conversely, the five species considered least vulnerable (S. forsteni, S. schlegeli, Chl. spilurus, Calotomus carolinus, and S. psittacus) decreased in biomass density by an average of 32% across the decade. Changes in H. longiceps demography were based on only 33 surveyed individuals, so the variability surrounding these estimates is likely high, but the patterns were stark. Notably, H. longiceps fishery catches in Guam have shown patterns that support density-dependent patterns of increased recruitment (Houk et al. 2018). However, the drivers of these unexpected patterns across species are clearly complex and likely reflect the interactions of multiple environmental and anthropogenic influences across the past decade.
In conclusion, parrotfishes are important fishery resources in Guam and throughout the tropical regions of the world, they are functionally important to coral reefs, and they are inherently vulnerable as conspicuous coastal species that are intricately linked to the dynamics of benthic communities. The previous decade was one of substantial change for coral reef ecosystems not only in Guam, but across the Indo-Pacific. The next decade promises more environmental change alongside additional human population growth (Hughes et al. 2017). Natural disturbances are becoming more severe and more frequent, fostering an uncertain future for the trajectories of coral reefs and associated fish populations in Guam. Armed with a modern understanding of complex ecological interactions within coral reef ecosystems, monitoring to facilitate adaptive management measures has never been more important, especially in areas like Guam where large human populations depend on reef resources.
References
Amesbury JR, Hunter-Anderson RL (2008) An analysis of archaeological and historical data on fisheries for pelagic species in Guam and the Northern Mariana Islands. Report prepared for Pelagic fisheries research program, University of Hawaii. Micronesian Archaeological research services, Mangilao, Guam. http://www.soest.hawaii.edu/PFRP/pdf/MARSreport.pdf
Bejarano S, Jouffrey J-B, Chollett I, Allen R, Roff G, Marshell A, Steneck R, Ferse SCA, Mumby PJ (2017) The shape of success in a turbulent world: wave exposure filtering of coral reef herbivory. Funct Ecol 31:1312–1324. https://doi.org/10.1111/1365-2435.12828
Bellwood DR (1995) Direct estimate of bioerosion by two parrotfish species, Chlorurus gibbus and C. sordidus, on the Great Barrier Reef. Australia Mar Biol 121:419–429. https://doi.org/10.1007/BF00349451
Brandl SJ, Rasher DB, Cóte IM, Casey JM, Darling ES, Lefcheck JS, Duffy JE (2019) Coral reef ecosystem functioning: eight core processes and the role of biodiversity. Front Ecol Environ 17:445–454. https://doi.org/10.1002/fee.2088
Claudet J, Osenberg CW, Benedetti-Cecchi L, Domenici P, García-Charton J-A, Pérez-Ruzafa A, Badalamenti F, Bayle-Sempere J, Brito A, Bulleri F, Culioli J-M, Dimech M, Falcón JM, Guala I, Milazzo M, Sáchez-Meca J, Somerfield PJ, Stobart B, Vandeperre F, Valle C, Planes S (2008) Marine reserves: size and age do matter. Ecol Lett 11:481–489. https://doi.org/10.1111/j.1461-0248.2008.01166.x
Clements KD, German DP, Piché J, Tribollet A, Choat JH (2017) Integrating ecological roles and trophic diversification on coral reefs: multiple lines of evidence identify parrotfishes as microphages. Biol J Linn Soc 120:729–751. https://doi.org/10.1111/bij.12914
Crossman DJ, Choat JH, Clements KD, Hardy T, McConochie J (2001) Detritus as food for grazing fishes on coral reefs. Limnol Oceanogr 46:1596–1605
De’ath G (2002) Multivariate regression trees: a new technique for modelling species-environment relationships. Ecology 83:1105–1117
Dixon P (2003) VEGAN, a package of R functions for community ecology. J Veg Sci 14:927–930
Dollar S (1982) Wave stress and coral community structure in Hawaii. Coral Reef 1:71–81
Emslie MJ, Pratchett MS (2018) Differential vulnerabilities of parrotfishes to habitat degradation. In: Hoey AS, Bonaldo RM (eds) Biology of parrotfishes. CRC Press, Boca Raton, pp 355–382
Fidler RY, Andradi-Brown DA, Awaludinnoer PD, Purwanto HNI, Ahmadia GN, Harborne AR (2021) The importance of biophysical context in understanding marine protected area outcomes for coral reef fish populations. Coral Reef 40:791–805
Flower J, Ortiz JC, Chollett I, Abdullah S, Castro-Sanguino C, Hock K, Lam V, Mumby PJ (2017) Interpreting coral reef monitoring data: a guide for improved management decisions. Ecol Ind 72:848–869
Friedlander AM, Brown EK, Jokiel PL, Smith WR, Rodgers KS (2003) Effects of habitat, wave exposure, and marine protected area status on coral reef fish assemblages in the Hawaiian archipelago. Coral Reef 22:291–305
Gilmour JP, Smith LD, Heyward AJ, Baird AH, Pratchett MS (2013) Recovery of an isolated coral reef system following severe disturbance. Science 340:69–71. https://doi.org/10.1126/science.1232310
Graham NAJ, Robinson JPW, Smith SE, Govinden R, Gendron G, Wilson SK (2020) Changing role of coral reef marine reserves in a warming climate. Nat Commun 11:2000
Green A (2003) American Samoa bans destructive SCUBA fishery: the role of monitoring in management. In: Wilkinson C, Green A, Almany J, Dionne S (eds) Monitoring coral reef marine protected areas: a practical guide on how monitoring can support effective management of MPAs. Townsville, Australian Institute of Marine Science IUCN Marine Program, pp 38–39
Gust N (2002) Scarid biomass on the northern great Barrier reef: the influence of exposure, depth and substrata. Environ Biol Fishes 64:353–366
Gust N, Choat JH, McCormick MI (2001) Spatial variability in reef fish distribution, abundance, size and biomass: a multi-scale analysis. Mar Ecol Prog Ser 214:237–251
Houk P, Rhodes K, Cuetos-Bueno J, Lindfield S, Fread V, McIlwain JL (2012) Commercial coral-reef fisheries across Micronesia: a need for improving management. Coral Reef 31:13–26
Houk P, Camacho R, Johnson S, McLean M, Maxin S, Anson J, Joseph E, Nedlic O, Luckymis M, Adams K, Hess D, Lemari L, Kabua E, Buthung E, Yalon A, Graham C, Benavente D, Leberer T, Taylor B, van Woesik R (2015) The Micronesia Challenge: assessing the relative contribution of stressors on coral reefs to facilitate science-to-management feedback. PLoS One 10(6):e0130823
Houk P, Cuetos-Bueno J, Tibbatts B, Gutierrez J (2018) Variable density dependence and the restructuring of coral-reef fisheries across 25 years of exploitation. Sci Rep 8:5725
Houk P, McInnis A, Benavente D, Gaag M, Maxin S, McLean M, Nam J, Segal B, Skilling T, Teriso C, Hemminger J (2022) Climate change disturbances contextualize the outcomes of coral-reef fisheries management across Micronesia. PLoS Clim 1:e0000040
Howard KG, Schumacher BD, Parrish JD (2009) Community structure and habitat associations of parrotfishes on Oahu. Hawaii Environ Biol Fishes 85:175–186
Hughes TP, Barnes ML, Bellwood DR, Cinner JE, Cumming GS, Jackson JBC, Kleypas J, Van De Leemput IA, Lough JM, Morrison TH, Palumbi SR, Van Nes EH, Scheffer M (2017) Coral reefs in the Anthropocene. Nature 546:82–90
Jennings S, Reynolds JD, Polunin NVC (1999) Predicting the vulnerability of tropical reef fishes to exploitation with phylogenies and life histories. Conserv Biol 13:1466–1475. https://doi.org/10.1046/j.1523-1739.1999.98324.x
Johnson GB, Taylor BM, Robbins WD, Franklin EC, Toonen R, Bowen B, Choat JH (2019) Diversity and structure of parrotfish assemblages across the northern Great Barrier Reef. Diversity 11:14. https://doi.org/10.3390/d11010014
Kirch PV, Hunt TL (1997) Historical ecology in the Pacific Islands: prehistoric environmental and landscape change. Yale University Press, New Haven
Lange ID, Perry CT, Morgan KM, Roche R, Benkwitt C, Graham NAJ (2020) Site-level variation in parrotfish grazing and bioerosion as a function of species-specific feeding metrics. Diversity 12:379. https://doi.org/10.3390/d12100379
Lange ID, Benkwitt CE, McDevitt-Irwin JM, Tietjan KL, Taylor B, Chinkin M, Gunn RL, Palmisciano M, Steyaert M, Wilson B, East HK, Turner J, Graham NAJ, Perry CT (2021) Wave exposure shapes reef community composition and recovery trajectories at a remote coral atoll. Coral Reef 40:1819–1829
Lindfield SJ, McIlwain JL, Harvey ES (2014) Depth refuge and the impacts of SCUBA spearfishing on coral reef fishes. PLoS One 9:e92628. https://doi.org/10.1371/journal.pone.0092628
Maragos JE (1993) Impact of coastal construction on coral reefs in the U.S.-affiliated Pacific Islands. Coastal Manag 21:235–269. https://doi.org/10.1080/08920759309362207
Moore BR (2022) Age-based life history of Pacific longnose parrotfish Hipposcarus longiceps from New Caledonia. J Fish Biol 100:997–1008
Munday PL, Jones GP, Pratchett MS, Williams AJ (2008) Climate change and the future for coral reef fishes. Fish Fish 9:261–285. https://doi.org/10.1111/j.1467-2979.2008.00281.x
Myers RF (1993) Guam’s small-boat-based fisheries. Mar Fish Rev 55:117–128
Page M (1998) The biology, community structure, growth and artisanal catch of parrotfishes of American Samoa. Department of Marine and Wildlife Resources, American Samoa. Available at www.botany.hawaii.edu/basch/uhnpscesu/pdfs/sam/Page1998parrotfishesAS.pdf
Questel S-LA, Russ GR (2018) No-take marine reserve effects on parrotfish and parrotfish-benthos interactions. In: Hoey AS, Bonaldo RM (eds) Biology of parrotfishes. CRC Press, Boca Raton, pp 329–354
Raymundo LJ, Burdick D, Hoot WC, Miller RM, Brown V, Reynolds T, Gault J, Idechong J, Fifer J, Williams A (2019) Successive bleaching events cause mass coral mortality in Guam, Micronesia. Coral Reef 38:677–700
Reynolds T, Burdick D, Houk P, Raymundo L, Johnson S (2014) Unprecedented coral bleaching across the Marianas Archipelago. Coral Reef 33:499
Richmond RH (1993) Coral reefs: present problems and future concerns resulting from anthropogenic disturbance. Am Zool 33:524–536. https://doi.org/10.1093/icb/33/6/524
Robinson JPW, McDevitt-Irwin JM, Dajka J-C, Hadj-Hammou J, Howlett S, Graba-Landry A, Hoey AS, Nash KL, Wilson SK, Graham NAJ (2020) Habitat and fishing control grazing potential on coral reefs. Funct Ecol 34:240–251. https://doi.org/10.1111/1365-2435.13457
Russ GR, Questel SA, Rizzari JR, Alcala AC (2015) The parrotfish-coral relationship: refuting the ubiquity of a prevailing paradigm. Mar Biol 162:2029–2045
Sundberg M, Humphreys R, Lowe MK, Cruz E, Gourley J, Ochavillo D (2015) Status of life history sampling conducted through the commercial fisheries bio-sampling programs in the Western Pacific Territories of American Samoa and Guam and in the Commonwealth of the Northern Mariana Islands. NOAA Administrative Report H-15–08, 56 p https://repository.library.noaa.gov/view/noaa/9054
Taylor BM (2014) Drivers of protogynous sex change differ across spatial scales. Proc R Soc London Ser B 281:20132423
Taylor BM, Choat JH (2014) Comparative demography of commercially important parrotfish species from Micronesia. J Fish Biol 84:383–402
Taylor BM, Cruz E (2017) Age-based and reproductive biology of the Pacific Longnose Parrotfish Hipposcarus longiceps from Guam. PeerJ 5:e4079
Taylor BM, McIlwain JL, Kerr AM (2012) Marine reserves and reproductive biomass: a case study of a heavily targeted reef fish. PLoS one 7:e39599
Taylor BM, Houk P, Russ GR, Choat JH (2014) Life histories predict vulnerability to overexploitation in parrotfishes. Coral Reef 33:869–878
Taylor BM, Lindfield SJ, Choat JH (2015) Hierarchical and scale-dependent effects of fishing pressure and environment on the structure and size distribution of parrotfish communities. Ecography 38:520–530
Taylor BM, Benkwitt CE, Choat JH, Clements KD, Graham NAJ, Meekan MG (2020) Synchronous biological feedbacks in parrotfishes associated with pantropical coral bleaching. Global Chang Biol 26:1285–1294
Tribollet A (2008) Dissolution of dead corals by euendolithic microorganisms across the northern Great Barrier Reef (Australia). Microb Ecol 55:569–580
Western Pacific Regional Fishery Management Council (2016) Fishing Conflicts on Guam: a report of meetings and interviews with fishermen. 15 http://www.wpcouncil.org/wp-content/uploads/2019/05/WPRFMC.-January-2016.-Fishing-Conflicts-on-Guam-A-Report-of-Meetings-and-Interviews-with-Fishermen.-Honolulu.pdf
Zeller D, Booth S, Davis G, Pauly D (2007) Re-estimation of small-scale fishery catches for U.S. flag-associated island areas in the western Pacific: the last 50 years. Fish Bull 105:266–277
Acknowledgements
This paper is funded by a cooperative agreement with the National Oceanic and Atmospheric Administration, Project NA18OAR4170077, which is sponsored by the University of Guam Sea Grant from NOAA Office of Sea Grant, Department of Commerce. The views expressed herein are those of the authors and do not necessarily reflect the views of NOAA or any of its sub-agencies. The authors thank A McInnis, M Deinhart, J Hartup, M Cruz, J Miller, J Perez, and T Genereaux for field assistance, and P Houk and G Roff for helpful discussions. We thank two anonymous reviewers for their comments on an earlier draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Taylor, B.M., Duenas, A.E.K. & Lange, I.D. Decadal changes in parrotfish assemblages around reefs of Guam, Micronesia. Coral Reefs 41, 1693–1703 (2022). https://doi.org/10.1007/s00338-022-02315-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-022-02315-x