Abstract
Context
Radical nephroureterectomy (RNU) with bladder cuff resection is the standard treatment in patients with high-risk upper tract urothelial cancer (UTUC). However, it is unclear which specific surgical technique may lead to improve oncological outcomes in term of intravesical recurrence (IVR) in patients with UTUC.
Objective
To evaluate the efficacy of surgical techniques and approaches of RNU in reducing IVR in UTUC patients.
Evidence Acquisition
Three databases were queried in January 2024 for studies analyzing UTUC patients who underwent RNU. The primary outcome of interest was the rate of IVR among various types of surgical techniques and approaches of RNU.
Evidence Synthesis
Thirty-one studies, comprising 1 randomized controlled trial and 1 prospective study, were included for a systematic review and meta-analysis. The rate of IVR was significantly lower in RNU patients who had an early ligation (EL) of the ureter compared to those who did not (HR: 0.64, 95% CI: 0.44–0.94, p = 0.02). Laparoscopic RNU significantly increased the IVR compared to open RNU (HR: 1.28, 95% CI: 1.06–1.54, p < 0.001). Intravesical bladder cuff removal significantly reduced the IVR compared to both extravesical and transurethral bladder cuff removal (HR: 0.65, 95% CI: 0.51–0.83, p = 0.02 and HR: 1.64, 95% CI: 1.15–2.34, p = 0.006, respectively).
Conclusions
EL of the affected upper tract system, ureteral management, open RNU, and intravesical bladder cuff removal seem to yield the lowest IVR rate in patients with UTUC. Well-designed prospective studies are needed to conclusively elucidate the optimal surgical technique in the setting of single post-operative intravesical chemotherapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Open radical nephroureterectomy (RNU) with complete resection of the ipsilateral bladder cuff is the treatment of choice for high-risk clinically non-metastatic upper tract urothelial cancer (UTUC) [1, 2]. Approximately 30–40% of patients experience intravesical cancer recurrence (IVR) following RNU [3], presumably due to seeding in the majority of cases [4]. Single dose post-operative intravesical chemotherapy is given to lower the risk of IVR based on two prospective trials [5, 6]. Preventing IVR would allow for less intense follow-up, possibly reduce the need for intravesical single dose post-operative chemotherapy and, thereby, lower the cost and burden of care associated with RNU.
To date, several studies [7,8,9,10,11,12] have reported on the differences in oncological outcomes, including IVR, due to surgical methods or approaches. Grossmann et al. [7] reported there was significant difference in IVR between laparoscopic and open RNU, while Correia et al. [8] showed there was no significant difference between the two groups. As these topics remain controversial, in this systematic review and meta-analysis, we summarize the data on the available surgical methods, and clarify the impact of interventional approach on the risk of IVR of the RNU in patients with UTUC.
Evidence acquisition
We registered the study with the International Prospective Register of Systematic Reviews (PROSPERO: registration number: CRD42024504011). This systematic review and meta-analysis was conducted in line with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement (PRISMA 2020 checklist, Supplementary Table 1).
Search strategy
On January 2024, the Medline, Scopus, and Web of Science databases were searched to identify studies investigating the impact of surgical technique during RNU on the risk of IVR. The search terms included: “ureteric neoplasms”, “recurrence”, “nephroureterectomy”. The detailed search strategy for each database is shown in the Supplementary Appendix 1. Two investigators independently performed an initial screening based on the titles and abstracts and noted the cause of the exclusion of ineligible reports. Full texts were retrieved and evaluated for eligibility. In addition, searches of reference lists were performed to identify additional studies of interest. In the case of discrepancies, they were solved by consensus among the authors.
Inclusion and exclusion criteria
We incorporated studies that evaluated IVR following RNU in patients with UTUC. The studies were required to report the surgical technique and the hazard ratio (HR) of IVR following RNU. We excluded studies that lacked original patient data, along with reviews, letters, editorial remarks, responses from authors, case reports, and articles not written in English. When encountering duplicate studies from the same cohorts, we selected either the more recent or the higher-quality publication.
Data extraction
After duplicate removal, two authors independently screened the titles and abstracts of retrieved records using a standard form. All eligible studies were assessed in full text. The extracted data included: first author, publication year, study region and design, numbers of patients undergoing RNU, and median follow-up time. Additionally, we collected RNU approach, bladder cuff excision method, tumor stage and grade. The endpoint of interest was the rate of IVR reported as hazard ratio (HR) and 95% confidence interval (CI). If the IVR rate was not available in the text, Kaplan-Meier curves were digitized using WebPlotDigitizer software (version 4.6) to extract survival estimates with corresponding 95% CIs [13, 14]. In cases where available graphs did not include 95% CIs, IPDfromKM software was used to reconstruct individual patient data (IPD) based on digitized Kaplan-Meier curves and calculate estimates with corresponding 95% CIs [15].
Quality assessment & risk of bias
Study quality and risk of bias were evaluated using the Risk-of-Bias (ROB version 2) tool as outlined in the Cochrane Handbook for Systematic Reviews of Interventions [16]. We used the ROBINS-I tool to evaluate bias in non-randomized studies [17]. The RoB2 and ROBINS-I assessment of each study was performed by two authors independently. Finally, we evaluated potential publication bias by using funnel plot and Peters’ linear regression test for funnel plot asymmetry was performed when at least ten studies were included in the meta-analysis.
Statistical analysis
All statistical analyses were performed using R Version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria, 2023; meta). To evaluate the effect of the surgical technique on the rate of intravesical recurrence in patients with UTUC who underwent RNU, we generated and analyzed forest plots with HR and 95%CI. For evaluating iRFS, analyses based on contrasts were conducted to estimate the differences in the logarithm of the HR. The standard error was determined using the published HR and CI. Cochrane’s Q test and the I-square test were used to evaluate the heterogeneity with I2 statistics greater than 50% considered significant. When significant heterogeneity was observed, we attempted to investigate the causes of heterogeneity [18]. We performed sensitivity analyses to increase homogeneity and confirm the reliability of our results. P-values at < 0.05 were considered significant.
Evidence synthesis
Study selection and characteristics
Our initial search identified 3,039 records. After we removed duplicates, 1,882 records remained for screening of titles and abstracts which led to 1,251 articles being excluded (Supplementary Fig. 1). According to our inclusion criteria, we identified 1 RCT, 1 prospective study, and 29 retrospective studies comprising 20,048 eligible patients for meta-analyses [7, 8, 19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47]. The absolute number of IVR was 5066 patients (25.3%). The median follow-up duration of all patients was 36.1 months ranging from 11.4 to 104.3. The detailed characteristics of the included studies are summarized in Supplementary Table 2.
We found that the surgical relevant steps were generally categorized into three relevant steps for our endpoint of interest: timing of ureter ligation, surgical approach, and technique used for the management of the distal ureter. The timing of ureter ligation was divided into two groups: early ligation (EL) and non-EL (NEL). The definition of EL was generally the ligation of the ureter as soon as possible after starting the surgery or before manipulation of the kidney [20, 25, 31, 38]. The surgical approach was divided into open RNU (ORNU), laparoscopic RNU (LRNU), robot assisted RNU (RANU), and hand-assisted LRNU (H-LRNU). The removal of the distal ureter was divided into an extravesical bladder cuff (EVBC), intravesical bladder cuff (IVBC), transurethral incision bladder cuff (TUBC), and intracorporeal EVBC removal. EVBC was defined as the dissection of the distal ureter and bladder cuff extravesically with lower midline abdominal incision. IVBC was performed generally through a cystotomy procedure with intravesical complete visual excision of the complete intravesical ureter under visual inspection. Intracorporeal EVBC was performed solely with laparoscopic approach, without lower midline abdominal incision.
Risk of bias assessment
Authors’ judgments about each domain for each included study are graphed in Supplementary Fig. 2 and Supplementary Table 3. Funnel plots and Peter’s Linear Regression analysis are depicted in Supplementary Fig. 3.
Meta-analysis
The results of the meta-analysis are described in Fig. 1.
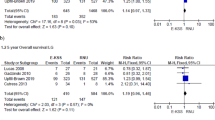
Forest plots showing the effect of surgical technique, approaches, and distal ureter management in preventing intravesical recurrence of UTUC patients who underwent radical nephroureterectomy: (A) NEL vs. EL, (B) ORNU vs. LRNU, (C) ORNU vs. RANU, (D) LRNU vs. RANU, (E) LRNU vs. H-LRNU, (F) EVBC vs. IVBC, (G) EVBC vs. TUBC, (H) IVBC vs. TUBC, (I) Open EVBC vs. intracorporeal EVBC
Early ligation (EL) vs no early ligation (NEL) of the ureter
Our analysis comprised four studies: 1 RCT, 1 prospective study, and 2 retrospective studies, with a total of 958 patients [20, 25, 31, 38]. Overall, 390 patients (40.7%) had EL and 568 patients (58.5%) did not. Out of 958 patients, 301 (31.4%) experienced IVR with median follow-up 35 months. The rate of IVR was significantly lower in patients who had an EL compared to those who did not (pooled HR: 0.64, 95% CI: 0.44–0.94, p = 0.021; Fig. 1A). The Cochrane’s Q tests and I2 statistic indicated heterogeneity (p = 0.07, I2 = 58%). We conducted sensitivity analysis and detected the cause of heterogeneity (Supplementary Fig. 4A).
Open RNU (ORNU) vs laparoscopic RNU (LRNU)
Nineteen retrospective studies with a total of 13,243 patients, assessed the impact of LRNU compared to ORNU on IVR. Overall, 2997 of the 13,243 patients (22.6%) experienced IVR. The median follow-up time was 37.7 months. The rate of IVR was significantly higher in patients treated by LRNU compared those treated by ORNU (pooled HR: 1.28, 95% CI: 1.06–1.54, p = 0.009; Fig. 1B). The Cochrane’s Q tests and I2 statistic indicated significant heterogeneity (p < 0.001, I2 = 71%). Despite conducting a sensitivity analysis, the cause of heterogeneity remained undetected (Supplementary Fig. 4B,4 C,4D). Peter’s Linear Regression analysis did not show the significant difference (p = 0.059, Supplementary Fig. 3B).
ORNU vs Robot assisted NU (RANU)
Two retrospective studies, comprising 1,144 patients, were analyzed to compare the incidence of IVR between ORNU and RANU [7, 36]. There was no significant difference in the rate of IVR between two groups (pooled HR: 1.41, 95% CIs: 0.91–2.18, p = 0.12; Fig. 1C). The Cochrane’s Q tests and I2 statistic indicated heterogeneity (p = 0.08, I2 = 68%). Despite conducting a sensitivity analysis, the cause of heterogeneity remained undetected (Supplementary Fig. 4).
LRNU vs RANU
An analysis of three studies, comprising 1,588 patients, was conducted to compare the incidence of IVR between LRNU and RANU [7, 39, 40]. LRNU included 799 patients (50.3%) and RANU comprised 789 (49.7%). There was no significant difference in the rate of IVR between the two approaches (pooled HR: 1.10, 95% CI: 0.83–1.47, p = 0.5; Fig. 1D). The Cochrane’s Q tests and I2 statistic revealed no significant heterogeneity (p = 0.9, I2 = 0%).
LRNU vs Hand-assisted LRNU (H-LRNU)
An analysis of two studies [40, 44], comprising 1,521 patients, was conducted to compare the rate of IVR between LRNU and H-LRNU. LRNU included 639 patients (42%), while H-LRNU had 882 (58%). There was no significant difference in the rate of IVR between two groups (pooled HR: 0.98, 95% CI: 0.80–1.21, p = 0.9; Fig. 1E). The Cochrane’s Q tests and I2 statistic revealed no significant heterogeneity (p = 0.9, I2 = 0%).
Extravesical bladder cuff (EVBC) vs intravesical bladder cuff (IVBC)
Six retrospective studies, comprising 5,409 patients, were analyzed to compare the rate of IVR between EVBC and IVBC removal [23, 26, 29, 37, 41, 45]. The rate of IVR was significantly lower in patients who had an IVBC removal compared to those who had an EVBC removal (pooled HR: 0.65, 95% CI: 0.51–0.83, p < 0.001; Fig. 1F). The Cochrane’s Q tests and I2 statistic indicated heterogeneity (p = 0.015, I2 = 65%). We conducted sensitivity analysis, and the cause of heterogeneity was detected (Supplementary Fig. 4E).
EVBC vs TUBC
Five retrospective studies, comprising 1,702 patients, were analyzed to compare the incidence of IVR between EVBC and TUBC removal [23, 24, 26, 29, 39]. There was no significant difference in the rate of IVR between two approaches (pooled HR: 0.85, 95% CI: 0.27–2.64, p = 0.8; Fig. 1G). The Cochrane’s Q tests and I2 statistic indicated heterogeneity (p < 0.001, I2 = 84%). Despite conducting a sensitivity analysis, the cause of heterogeneity remained undetected (Supplementary Fig. 4F, 4G, 4 H).
IVBC vs TUBC
Two retrospective studies, comprising 2,249 patients, were analyzed to compare the incidence of IVR between IVBC and TUBC removal [26, 45]. The rate of IVR was significantly lower in patients who had an IVBC removal compared to those who had a TUBC removal (pooled HR: 1.64, 95% CI: 1.15–2.34, p = 0.006; Fig. 1H). The Cochrane’s Q tests and I2 statistic indicated no significant heterogeneity (p = 0.6, I2 = 0%).
Open EVBC vs Intracorporeal EVBC
Two retrospective studies, comprising 392 patients, were analyzed to compare the incidence of IVR between open and intracorporeal EVBC removal [42, 46]. There was no significant difference in the rate of IVR between two approaches (pooled HR: 1.69, 95% CI: 0.74–3.86, p = 0.2; Fig. 1I). The Cochrane’s Q tests and I2 statistic indicated heterogeneity (p = 0.06, I2 = 71%).
Discussion
In this systematic review and meta-analysis, we showed the effect of various surgical techniques during RNU on the rate of IVR in patients treated with RNU for UTUC. Our study revealed several critical findings. First, EL of the ureter during RNU significantly reduces the rate of IVR compared to NEL. Second, LRNU was associated with an increase in the rate of IVR compared to ORNU. Third, IVBC removal was associated with a significant lower rate of IVR in comparison to EVBC or TUBC.
To the best of our knowledge, our meta-analysis is the first to analyze the association between EL and NEL regarding the rate of IVR. Our study indicates that EL of the ureter during RNU with a relative risk reduction of the rate of IVR compared to NEL. Although the pathogenesis of IVR following RNU is still unclear and highly multi factional, one hypothesized mechanism is the downstream seeding of tumor cells from the upper urinary tract into the bladder eventually setting in the bladder mucosa [48]. Therefore, EL of the ureter as soon as possible is plausible to prevent seeding the tumor cells distally. On the other hands, investigator have raised concerns regarding the potential adverse effects of EL of the ureter prior to renovascular ligation on local recurrence and survival rates, due to the risk of direct tumor handling and a significant rise in intrarenal pelvic pressure. Inokuchi et al., for example, revealed that the patients who had an EL had significantly worse in both overall survival (OS) and cancer-specific survival (CSS) compared to those who did not (OS: HR: 1.88, 95%CI 1.24–2.85, p = 0.003, CSS: HR: 1.93, 95%CI 1.14–3.25, p = 0.014) [38]. Chen et al. reported that EL of the ureter was significantly associated with a lower IVR rate compared to NEL (HR: 0.43, 95%CI 0.29–0.64, p = 0.04) [20]. One prospective and two retrospective studies did not a difference, however [25, 31, 38], our analysis supports that EL of the ureter seems to prevent spreading the UTUC to the bladder reducing thereby the IVR rate following RNU, compared to NEL. However, the impact of EL on OS and CSS remains unclear and poorly investigated.
We found that LRNU was associated with a significantly higher risk of IVR compared to ORNU. Seisen et al. [3] reporting that LRNU is indeed associated with an increased rate of IVR compared to ORNU (pooled HR: 1.62, 95%CI: 1.18–2.22, p = 0.001). The biology underlying this reality is unclear, but Rouprêt et al. [49] hypothesized that manipulating on UTUC under high intra-abdominal pressure could increases the risk of gravitational seeding with deposition of cancer cells in regions such as the bladder or retroperitoneal space, especially in the context of locally advanced tumors [10, 49]. While RANU should lead to some effect, this has not been shown. This could be due to many other covariates such as EL, differences in the approach to the bladder cuff, and more widely use of intravesical single post-operative chemotherapy in the age of RANU.
We found that IVBC removal leads to lower IVR compared to EVBC and TUBC removal. Complete resection of the distal ureter and the bladder orifice is an integral part of RNU decreasing the risk of IVR [3, 12]. There are several techniques for bladder cuff management, with two previous meta-analyses revealing that EVBC removal is associated with an increased rate of IVR compared to IVBC removal [3, 12]. While an EVBC can obtain a complete removal such as an IVBC, the higher IVR rate is likely due to an incomplete removal in some cases, voluntary or involuntary [12]. TUBC removal has been, similarly, shown to lead to higher IVR rate compared to IVBC removal (pooled HR:1.63. 95%CI: 1.28–2.08, p < 0.001). Here again, it is likely that not all the urothelial moiety that is to be removed has eventually been removed. Alternatively or additionally, seeding can have been caused by the TUR process as well [12].
Limitations
Our study has certain limitations. Firstly, the majority of included studies were retrospective. Additionally, there was significant heterogeneity in some of our analyses. Secondly, our analysis did not perform an analysis regarding the difference in IVR between transperitoneal and retroperitoneal approach. Thirdly, other crucial clinicopathological factors, such as ureteroscopy for definitive diagnosis before RNU, adjuvant intravesical chemotherapy, tumor multiplicity, location, stage, sex, and gender, were not always adjusted for while being considered to be also independent predictors of IVR. Utilizing adjusted HR would have enabled a more accurate interpretation of the results. However, some trials did not conduct or report multivariable analyses. As a result, it was not feasible to conduct an adjusted meta-analysis.
Conclusions
We found that EL of the ureter significantly reduced the risk of IVR in patients with UTUC following RNU. LRNU lead to significantly increased of the risk of IVR compared to ORNU. IVBC removal significantly decreased the rate of IVR compared to both EVBC or TUBC removal. Our analyses mostly included retrospective studies, further studies are needed to clarify the benefit of surgical techniques and approaches to reduce the IVR rate in patients with UTUC following RNU in the age of simple post-operative intravesical chemotherapy. These three concrete and valid steps can serve as a basis for quality indicators in the management of high-risk UTUC.
Figure 1. Forest plots showing the effect of surgical technique, approaches, and distal ureter management in preventing intravesical recurrence of UTUC patients who underwent radical nephroureterectomy: (A) NEL vs. EL, (B) ORNU vs. LRNU, (C) ORNU vs. RANU, (D) LRNU vs. RANU, (E)LRNU vs. H-LRNU, (F) EVBC vs. IVBC, (G) EVBC vs. TUBC, (H) IVBC vs. TUBC, (I) Open EVBC vs. intracorporeal EVBC.
EL: early ligation, EVBC: extravesical bladder cuff, IVBC: intravesical bladder cuff, H-LRNU = hand-assisted laparoscopic radical nephroureterectomy, LRNU: laparoscopic radical nephroureterectomy, ORNU: open radical nephroureterectomy, RANU: robot assisted radical nephroureterectomy, RNU: radical nephroureterectomy, TUBC: transurethral incision of the bladder cuff, UTUC: upper tract urothelial cancer.
References
Margulis V, Shariat SF, Matin SF, Kamat AM, Zigeuner R, Kikuchi E et al (2009) Outcomes of radical nephroureterectomy: a series from the Upper Tract Urothelial Carcinoma collaboration. Cancer 115:1224–1233
Lughezzani G, Jeldres C, Isbarn H, Sun M, Shariat SF, Alasker A et al (2009) Nephroureterectomy and segmental ureterectomy in the treatment of invasive upper tract urothelial carcinoma: a population-based study of 2299 patients. Eur J Cancer 45:3291–3297
Seisen T, Granger B, Colin P, Léon P, Utard G, Renard-Penna R et al (2015) A systematic review and Meta-analysis of clinicopathologic factors linked to Intravesical Recurrence after Radical Nephroureterectomy to treat Upper Tract Urothelial Carcinoma. Eur Urol 67:1122–1133
Xylinas E, Kluth L, Passoni N, Trinh QD, Rieken M, Lee RK et al (2014) Prediction of intravesical recurrence after radical nephroureterectomy: development of a clinical decision-making tool. Eur Urol 65:650–658
O’Brien T, Ray E, Singh R, Coker B, Beard R (2011) Prevention of bladder tumours after nephroureterectomy for primary upper urinary tract urothelial carcinoma: a prospective, multicentre, randomised clinical trial of a single postoperative intravesical dose of mitomycin C (the ODMIT-C trial). Eur Urol 60:703–710
Ito A, Shintaku I, Satoh M, Ioritani N, Aizawa M, Tochigi T et al (2013) Prospective randomized phase II trial of a single early intravesical instillation of pirarubicin (THP) in the prevention of bladder recurrence after nephroureterectomy for upper urinary tract urothelial carcinoma: the THP Monotherapy Study Group Trial. J Clin Oncol 31:1422–1427
Grossmann NC, Soria F, Juvet T, Potretzke AM, Djaladat H, Ghoreifi A et al (2023) Comparing Oncological and Perioperative outcomes of Open versus laparoscopic versus robotic radical nephroureterectomy for the Treatment of Upper Tract Urothelial Carcinoma: a Multicenter, multinational, Propensity score-matched analysis. Cancers (Basel). ;15
Correia J, Mendes G, Texeira B, Madanelo M, Fraga A, Silva-Ramos M (2022) Perioperative and oncological outcomes of laparoscopic and open radical nephroureterectomy for locally advanced upper tract urothelial carcinoma: a single-center cohort study. Cent Eur J Urol 75:257–264
Peng L, Mehmud I, Meng C, Tang D, Li K, Gan L et al (2023) Comparison of Perioperative outcomes and complications of laparoscopic and robotic nephroureterectomy approaches in patients with Upper-Tract Urothelial Carcinoma. Ann Surg Oncol 30:3805–3816
Piszczek R, Nowak Ł, Krajewski W, Chorbińska J, Poletajew S, Moschini M et al (2021) Oncological outcomes of laparoscopic versus open nephroureterectomy for the treatment of upper tract urothelial carcinoma: an updated meta-analysis. World J Surg Oncol 19:129
Veccia A, Antonelli A, Francavilla S, Simeone C, Guruli G, Zargar H et al (2020) Robotic versus other nephroureterectomy techniques: a systematic review and meta-analysis of over 87,000 cases. World J Urol 38:845–852
Lai S, Guo R, Seery S, Wu P, Liu J, Zhang Y et al (2020) Assessing the impact of different distal ureter management techniques during radical nephroureterectomy for primary upper urinary tract urothelial carcinoma on oncological outcomes: a systematic review and meta-analysis. Int J Surg 75:165–173
Shim S, Lim Y, Hong M, Song G, Han H (2021) Statistical data extraction and validation from graph for data integration and meta-analysis. Korea J Big Data 6:61–70
Rohatgi A WebPlotDigitizer. 4.6 ed
Liu N, Zhou Y, Lee JJ (2021) IPDfromKM: reconstruct individual patient data from published Kaplan-Meier survival curves. BMC Med Res Methodol 21:111
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD et al (2011) The Cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919
Assel M, Sjoberg D, Elders A, Wang X, Huo D, Botchway A et al (2019) Guidelines for reporting of statistics for Clinical Research in Urology. Eur Urol 75:358–367
Favaretto RL, Shariat SF, Chade DC, Godoy G, Kaag M, Cronin AM et al (2010) Comparison between laparoscopic and open radical nephroureterectomy in a contemporary group of patients: are recurrence and disease-specific survival associated with surgical technique? Eur Urol 58:645–651
Chen MK, Ye YL, Zhou FJ, Liu JY, Lu KS, Han H et al (2012) Clipping the extremity of ureter prior to nephroureterectomy is effective in preventing subsequent bladder recurrence after upper urinary tract urothelial carcinoma. Chin Med J (Engl) 125:3821–3826
Kobayashi Y, Saika T, Miyaji Y, Saegusa M, Arata R, Akebi N et al (2012) Preoperative positive urine cytology is a risk factor for subsequent development of bladder cancer after nephroureterectomy in patients with upper urinary tract urothelial carcinoma. World J Urol 30:271–275
Xylinas E, Colin P, Audenet F, Phe V, Cormier L, Cussenot O et al (2013) Intravesical recurrence after radical nephroureterectomy for upper tract urothelial carcinomas: predictors and impact on subsequent oncological outcomes from a national multicenter study. World J Urol 31:61–68
Fradet V, Mauermann J, Kassouf W, Rendon R, Jacobsen N, Fairey A et al (2014) Risk factors for bladder cancer recurrence after nephroureterectomy for upper tract urothelial tumors: results from the Canadian Upper Tract collaboration. Urol Oncol 32:839–845
Luo HL, Kang CH, Chen YT, Chuang YC, Cheng YT, Lee WC et al (2014) Oncological impact of endoscopic bladder cuff management during nephroureterectomy varies according to upper urinary tract tumor location. Int J Urol 21:366–369
Ou CH, Yang WH (2014) Impact of earlier ureteral ligation on intravesical recurrence during hand-assisted retroperitoneoscopic nephroureterectomy. Urol Int 92:68–73
Xylinas E, Rink M, Cha EK, Clozel T, Lee RK, Fajkovic H et al (2014) Impact of distal ureter management on oncologic outcomes following radical nephroureterectomy for upper tract urothelial carcinoma. Eur Urol 65:210–217
Yamashita S, Ito A, Mitsuzuka K, Tochigi T, Namima T, Soma F et al (2016) Clinical implications of intravesical recurrence after radical nephroureterectomy for upper urinary tract urothelial carcinoma. Int J Urol 23:378–384
Lee CH, Ku JY, Jeong CW, Ku JH, Kwak C, Kim HH et al (2017) Predictors for Intravesical Recurrence Following Radical Nephroureterectomy for Upper Tract Urothelial Carcinoma: A National Multicenter Analysis. Clin Genitourin Cancer 15:e1055–e61
Liu JY, Dai YB, Zhou FJ, Long Z, Li YH, Xie D et al (2017) Laparoscopic versus open nephroureterectomy to treat localized and/or locally advanced upper tract urothelial carcinoma: oncological outcomes from a multicenter study. BMC Surg 17:8
Long X, Qi L, Zu X, Li Y, He W, Pi S et al (2017) Prevention of recurrent bladder tumors after Nephroureterectomy for Primary Upper urinary tract Urothelial Carcinoma: an Individualized Selection Strategy based on patient risk stratification is needed. Urol Int 98:312–319
Yamashita S, Ito A, Mitsuzuka K, Ioritani N, Ishidoya S, Ikeda Y et al (2017) Efficacy of early ureteral ligation on prevention of intravesical recurrence after radical nephroureterectomy for upper urinary tract urothelial carcinoma: a prospective single-arm multicenter clinical trial. Jpn J Clin Oncol 47:870–875
Kido K, Hatakeyama S, Fujita N, Yamamoto H, Tobisawa Y, Yoneyama T et al (2018) Oncologic outcomes for open and laparoscopic radical nephroureterectomy in patients with upper tract urothelial carcinoma. Int J Clin Oncol 23:726–733
Kim SH, Song MK, Kim JK, Hong B, Kang SH, Ku JH et al (2019) Laparoscopy versus Open Nephroureterectomy in Prognostic Outcome of patients with Advanced Upper Tract Urothelial Cancer: a retrospective, Multicenter, propensity-score matching analysis. Cancer Res Treat 51:963–972
Shigeta K, Kikuchi E, Abe T, Hagiwara M, Ogihara K, Anno T et al (2019) Long-term oncologic outcomes of laparoscopic Versus Open Radical Nephroureterectomy for patients with T3N0M0 Upper Tract Urothelial Carcinoma: a Multicenter Cohort Study with Adjustment by Propensity score matching. Ann Surg Oncol 26:3774–3781
Azawi NH, Næraa SH, Subhi Y, Vásquez JL, Norus T, Dahl C et al (2020) Oncological outcomes of radical nephroureterectomy for upper urinary tract urothelial neoplasia in Denmark. Scand J Urol 54:58–64
Chen IA, Chang CH, Huang CP, Wu WJ, Li CC, Chen CH et al (2021) Factors Predicting Oncological outcomes of Radical Nephroureterectomy for Upper Tract Urothelial Carcinoma in Taiwan. Front Oncol 11:766576
Chung JH, Song W, Kang M, Jeon HG, Jeong BC, Seo SI et al (2021) Conditional Intravesical recurrence-free Survival Rate after Radical Nephroureterectomy with bladder cuff excision for Upper Tract Urothelial Carcinoma. Front Oncol 11:730114
Inokuchi J, Kuroiwa K, Nishiyama H, Kojima T, Kakehi Y, Sugimoto M et al (2021) Significance of the timing of ureteral ligation on prognosis during radical nephroureterectomy for upper urinary tract urothelial cancer. Int J Urol 28:208–214
Katims AB, Say R, Derweesh I, Uzzo R, Minervini A, Wu Z et al (2021) Risk factors for Intravesical recurrence after minimally invasive nephroureterectomy for Upper Tract Urothelial Cancer (ROBUUST collaboration). J Urol 206:568–576
Li CC, Chang CH, Huang CP, Hong JH, Huang CY, Chen IA et al (2021) Comparing Oncological outcomes and Surgical complications of Hand-Assisted, laparoscopic and robotic nephroureterectomy for Upper Tract Urothelial Carcinoma. Front Oncol 11:731460
Ryoo H, Kim J, Kim T, Kang M, Jeon HG, Jeong BC et al (2021) Effects of complete bladder cuff removal on oncological outcomes following radical nephroureterectomy for Upper Tract Urothelial Carcinoma. Cancer Res Treat 53:795–802
Shigeta K, Matsumoto K, Takeda T, Hattori S, Kaneko G, Matsushima M et al (2021) Evaluating the oncological outcomes of pure laparoscopic radical Nephroureterectomy Performed for Upper-Tract Urothelial Carcinoma patients: a Multicenter Cohort Study adjusted by propensity score matching. Ann Surg Oncol 28:465–473
Huang YC, Wang HJ, Sung MT, Chuang YC, Chen YT, Cheng YT et al (2022) The lowest level of tumor involvement is a significant prognostic factor for upper tract urothelial carcinoma after radical nephroureterectomy: a large retrospective cohort study. Front Oncol 12:1031774
Kuo CC, Chen GH, Chang CH, Huang CY, Chen CH, Li CC et al (2022) Surgical outcome predictor analysis following hand-assisted or pure laparoscopic transperitoneal nephroureterectomy using the Taiwan upper urinary tract urothelial carcinoma database. Front Surg 9:934355
Blackmur JP, Chew E, Trail M, Brodie K, Santoni N, Rodger F et al (2023) Assessment of association between lower ureteric excision technique and oncological outcomes for upper urinary tract urothelial carcinoma: retrospective analysis from the Scottish Renal Cancer Consortium. World J Urol 41:757–765
Huang EY, Tai MC, Chung HJ, Chang YH, Huang WJ (2023) Effects of different combinations of radical nephroureterectomy and bladder cuff excision procedures for upper tract urothelial carcinoma on bladder recurrence. Int Braz J Urol 49:469–478
Shimura S, Matsumoto K, Ikeda M, Moroo S, Koguchi D, Taoka Y et al (2023) A multi-institutional retrospective study of open versus laparoscopic nephroureterectomy focused on the intravesical recurrence. Asia Pac J Clin Oncol 19:71–78
Hafner C, Knuechel R, Zanardo L, Dietmaier W, Blaszyk H, Cheville J et al (2001) Evidence for oligoclonality and tumor spread by intraluminal seeding in multifocal urothelial carcinomas of the upper and lower urinary tract. Oncogene 20:4910–4915
Rouprêt M, Smyth G, Irani J, Guy L, Davin JL, Saint F et al (2009) Oncological risk of laparoscopic surgery in urothelial carcinomas. World J Urol 27:81–88
Funding
Open access funding provided by Medical University of Vienna.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has conflicts of interest to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tsuboi, I., Matsukawa, A., Kardoust Parizi, M. et al. Differential effect of surgical technique on intravesical recurrence after radical nephroureterectomy in patients with upper tract urothelial cancer: a systematic review and Meta-analysis. World J Urol 42, 488 (2024). https://doi.org/10.1007/s00345-024-05185-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00345-024-05185-w