Abstract
Purpose
Cerebrospinal fluid (CSF) leakage is a challenging complication of intradural cranial surgery, and children are particularly at risk. The use of dural sealants confers protection in adults, but pediatric studies are scarce. We evaluated the safety and efficacy of Evicel® fibrin sealant as an adjunct to primary dural suturing in children undergoing cranial surgery.
Methods
A multicenter trial prospectively enrolled pediatric subjects (< 18 years) undergoing cranial neurosurgery who, upon completion of primary sutured dural repair, experienced CSF leakage. As agreed by the EMA Evicel® Pediatric Investigation Plan, 40 subjects were intra-operatively randomized 2:1 to Evicel® or additional sutures (‘Sutures’). Data analysis was descriptive. The efficacy endpoint was treatment success rate, with success defined as intra-operative watertight closure after provocative Valsalva maneuver (primary endpoint). Safety endpoints were postoperative CSF leakage (incisional CSF leakage, pseudomeningocele or both) and surgical site complications (secondary endpoints).
Results
Forty subjects (0.6–17 years) were randomized to Evicel® (N = 25) or Sutures (N = 15) (intention-to-treat). Intracranial tumor was the most common indication and procedures were mostly supratentorial craniotomies. Success rates were 92.0% for Evicel® and 33.3% for Sutures, with a 2.76 estimated ratio of success rates (Farrington-Manning 95% CI [1.53, 6.16]). Sensitivity analyses in per-protocol and safety sets showed similar results. Despite a higher rescue treatment rate, the frequencies of postoperative CSF leakage and wound complications were higher for Sutures than for Evicel®.
Conclusion
This small-scale prospective study shows Evicel® treatment to be safe and effective as an adjunct to primary sutured dura mater closure in a pediatric population. Compared to additional sutures, Evicel® was associated with reduced postoperative CSF leakage and surgical site complications. (Trial registration: The trial was registered as NCT02309645 and EudraCT 2013-003558-26).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Postoperative leakage of cerebrospinal fluid (CSF) is a challenging complication of intradural cranial surgery, as it may interfere with wound healing and lead to pseudomeningocele, surgical site infection, meningitis and fistula. Risk factors for CSF leakage include younger age and infratentorial or intraventricular approaches [1,2,3]. These place children at an increased risk, but because pediatric studies are heterogeneous and often of low quality, the incidence and risk profiles in children remain incompletely understood. Interpretation of published rates, which vary between 0 and 38%, is further hindered by the variable use of adjunctive and rescue treatment, and by inconsistency in the definitions for CSF leakage [3, 4]. A meta-analysis of good-quality studies estimated the overall incidence at 7.4% [3], concurring with a more recent large retrospective study [4]. Aside from causing patient morbidity, CSF leakage and its complications lead to significant health care expenditure [5, 6]. Pediatric series have reported rates of invasive measures of 49.1% and 70.0% [3, 7].
According to the longstanding tenet in neurosurgery, the most important measure to prevent CSF leakage is a meticulous repair of the anatomical dural barrier [8]. Techniques include primary and additional suturing of the native dura, adjunctive use of dural sealants, and, in case of dural defect, dural graft augmentation with biologic or synthetic material. In a recent retrospective multicenter study of 2,310 subjects, multivariate regression analysis showed dural sealant use to be associated with reduced incisional CSF leakage and reduced wound infection [1], and this is supported by a body of prior randomized and non-randomized adult studies [9,10,11,12,13,14,15,16,17,18,19]. Duraplasty was associated with an increased risk of incisional CSF leak [1]. However, the authors of the study caution that this result may be confounded by the large variability in dural substitutes, because individual studies have provided evidence to the contrary [20, 21]. Because pediatric series are limited and retrospective, the effects of dural sealants in children are difficult to estimate [3, 4], and those of duraplasty have been inconsistent [3, 4, 20, 21].
High-level evidence for the value of dural sealants was derived from a randomized controlled trial (RCT) which demonstrated that Evicel®, a human-derived fibrin sealant, was superior to additional suturing in providing intra-operative watertight dura mater closure [9]. More recently, a RCT confirmed this for BioSeal®, a porcine-derived fibrin sealant [22]. Originally developed as topical hemostats, fibrin sealants are mainly composed of thrombin and fibrinogen [14, 23]. Their safety and hemostatic efficacy were demonstrated in clinical trials of several surgical disciplines [24,25,26,27], supporting their clinical indication in the United States (US) and the European Union (EU) [28, 29]. As adjuncts to dura repair [14, 23], fibrin sealants present the advantages over synthetic sealants that they do not swell and thus avoid causing additional intracranial pressure, and that they resorb physiologically through fibrinolysis and thus do not lead to foreign body reactions. In the EU, Evicel® is indicated for suture line sealing in dura mater closure in adults [28, 29].
To address the need for effective measures to prevent CSF leakage in pediatric neurosurgery, we conducted a RCT evaluating Evicel® fibrin sealant versus additional sutures, as adjunct treatments to primary sutured dural closure in children undergoing intradural cranial surgery. The study aimed to evaluate the safety and efficacy of Evicel® as an adjunct to primary sutures to obtain intra-operative watertight closure.
Methods
Trial design
This prospective, open-label, multicenter, randomized controlled clinical trial evaluated the safety and effectiveness of Evicel® as an adjunct to sutured dural repair in children undergoing cranial neurosurgery, in comparison to additional suturing. The trial was conducted at 7 institutions in the United Kingdom (UK). Ethical considerations prevented the use of a sham control group and required that rescue treatment be provided when study treatment fails to achieve dural closure. The design was approved by the European Medicines Agency (EMA) as part of a Paediatric Investigation Plan (PIP) for Evicel® (EMEA-001149-PIP01-11-M07), and aligned with EU Guidance on clinical investigation of plasma-derived fibrin sealants (CPMP/BPWG/1089/00, 29 July 2004) (control group receiving treatment without fibrin sealant, that is approved for the pediatric indication, and surgical situations representing regular clinical practice). The design described in the PIP required a minimum of 40 subjects to be randomized in a 2:1 ratio to Evicel® or additional suturing (‘Sutures’), with stratification according to infra- and supratentorial approach. The trial was conducted in accordance with the International Conference on Harmonization (ICH) Harmonized Tripartite Guideline for Good Clinical Practice (2016); the Declaration of Helsinki (2013); the European Union Clinical Trial Directive (2001/20/EC, May 2001); and the EU GCP Directive (2005/28/EC). Approval from the Office for Research Ethics Committees Northern Ireland, Lisburn, UK, was obtained prior to commencing the trial. Original clinical trial application approval was obtained from UK MHRA on 12 June 2014. The trial was registered as NCT02309645 and EudraCT 2013-003558-26.
Study subjects and procedure
Subjects younger than 18 years requiring elective or emergent craniotomy or craniectomy for an infra- or supratentorial procedure were considered. Per local regulations, the subject’s parent or legal representative provided informed consent; assent was obtained from subjects who possessed the intellectual and emotional ability to comprehend the trial’s concepts. Patients were enrolled intra-operatively if -upon completion of primary sutured dural repair- a CSF leak was apparent (spontaneously or after Valsalva maneuver) in a Class I surgical wound with dural cuffs sufficiently wide to allow for coverage by fibrin sealant or placement of additional sutures. Surgeons used their institution’s standard clinical practice for the type and technique of primary suture closure of the dura. Exclusion criteria were existing surgical dural lesions with potential CSF leakage, hydrocephalus, existing CSF drainage or burr holes with damage to the dura, or known hypersensitivity to Evicel® component(s). Inclusion and exclusion criteria are fully listed in Supplementary Information 1.
Upon enrollment, subjects were randomized in a 2:1 ratio to Evicel® or Sutures. The Sponsor provided 2 sets (supra- and infratentorial) of computer-generated randomization envelopes containing randomization number and treatment allocation. For Evicel®-randomized subjects, 1 or 2 thin layers were applied (1- to 2-min cure time between layers), covering the suture line and ≥5 mm adjacent area including all suture holes. Evicel® was applied by drip or spray using the applicator device and accessory tips, according to surgeon preference [28]. The amount depended on the suture area and application method, with no maximum dose. If a second Valsalva maneuver showed persistent CSF leakage, a second treatment (1 or 2 layers) could be applied. If a subject was randomized to Sutures, additional suturing was performed per the investigator’s institutional standard of care.
Following randomized treatment, if final provocative Valsalva maneuver showed watertight closure, the treatment was deemed successful. For Evicel®-randomized subjects, no further adjunct was allowed. For Sutures-randomized subjects, adjunctive treatments (except fibrin sealants) were allowed to assure closure durability. If, after final Valsalva, CSF leakage was still apparent, the subject was deemed a treatment failure. For rescue treatment, the surgeon was to revert to their institutional standard of care for closure, which could include on-lay dural patch or fibrin sealants (except Evicel®). Post-operatively, subjects were followed until hospital discharge and, on post-operative days 5 (±2) and 30 (±3), with an office visit.
Primary endpoint
The primary endpoint related to efficacy. It was defined as the proportion of subjects with treatment success, considered as the achievement of intra-operative watertight closure after completion of the randomized treatment as assessed by provocative testing with Valsalva maneuver.
Secondary endpoints
Secondary endpoints related to safety. Adverse events (AE) and serious adverse events (SAE) definitions complied with the EU Clinical Trial Directive, [30, 31] were collected from randomization until postoperative day 30 (±3) and were adjudicated for relationship with study product (Evicel® arm only, as no study product was used in the Sutures arm) and procedure. Specific safety endpoints were the incidence of postoperative CSF leakage, and surgical site complications including infection, seroma and hematoma [32]. Under postoperative CSF leak, a distinction was made between incisional CSF (iCSF) leak, pseudomeningocele, or a combination of both. The iCSF leak and pseudomeningocele were defined by clinical observation, diagnostic testing, or the need for surgical intervention. Because they were considered important medical events (as potential treatment failures), any events of postoperative CSF leakage were to be categorized as SAEs.
Statistical analysis
The proportions of treatment success were summarized descriptively by treatment group, including the estimated ratio of success proportions with 2-sided 95% confidence interval (CI) (Farrington-Manning). The primary analysis was based on the intention-to-treat (ITT) set and included summaries of success proportions within the infra- and supratentorial stratum. Sensitivity analyses were performed in the Per-protocol (PP) and Safety sets. Safety endpoints were summarized descriptively. Software SAS® Version 9.1 [EG] was used.
Results
Study subjects
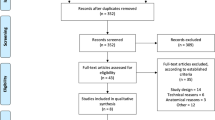
Between 9 October 2014 and 17 September 2021, 40 of 63 screened subjects were randomized to Evicel® (‘Evicel’) (N = 25) or Sutures (N = 15) (ITT set) (Fig. 1). The baseline patient characteristics (Table 1) were similar across groups. There were 8 major protocol deviations affecting randomization or primary endpoint (3 Evicel® and 5 Sutures). The PP set comprised 22 Evicel® and 10 Sutures subjects. Five major protocol deviations (3 Evicel® and 2 Sutures) occurred at one site and concerned the exclusion criteria ‘existing CSF drainage’ (Fig. 1). These were reported to MHRA under the serious breach category and reviewed by the trial’s safety lead and biostatistician. All subject data were re-monitored. It was confirmed that the safety of enrolled subjects had not been compromised, and a sensitivity analysis was performed on the PP set. The Safety set counted 26 Evicel® and 14 Sutures subjects because 1 Sutures-allocated subject received Evicel® (major protocol deviation). This subject was included under Sutures for the ITT analysis and included under Evicel® for sensitivity analysis in the Safety set.
Consort flow diagram with disposition of study subjects. Shown are the Intention-to-treat set (ITT), Per protocol set (PP) and Safety set. * Exclusions due to existing endoscopic third ventriculostomy (n = 1), ventriculoperitoneal drain (n = 1), and external ventricular drain (n = 1). # Exclusions due to misrandomization (n = 2), procedure deviation (Valsalva not done after additional sutures) (n = 1), existing ventriculoperitoneal drain (n = 1) and external ventricular drain (n = 1). † Subject received Evicel® instead of Sutures and is therefore included in Evicel® Safety Analysis Set
Surgical procedures and operative parameters
In both groups, most procedures were craniotomies and supratentorial approaches (Table 2, ITT set); the most frequent surgical indication was intracranial tumor, and more than half of intra-operative CSF leaks were spontaneous. The surgical and operation room times were similar between groups; hospitalization time was slightly longer for Sutures than for Evicel®. All but 3 Evicel®-treated subjects received 1 treatment, mostly of 1 product layer (Table 3). In Sutures-treated subjects, the median number of sutures was low (2.0), albeit with a wide range (1.0–12.0); 1 subject received additional treatment for suture durability. After completion of study treatment, 3 Evicel®-treated subjects (11.5%) and 9 Sutures-treated subjects (64.3%) received rescue treatment for persistent CSF leak. The quantitative use of Evicel® product is described in Supplementary Information 2.
Primary endpoint: efficacy
In the ITT analysis, success rates were 92.0% (23/25) for Evicel® and 33.3% (5/15) for Sutures, establishing an estimated PE/PS ratio of 2.76 (95% CI: [1.53, 6.16]) (Table 4). Sensitivity analyses in the PP and Safety sets showed similar rate differences: 90.9% (20/22) for Evicel® and 40.0% (4/10) for Sutures with an estimated PE/PS ratio of 2.27 (95% CI: [1.27, 5.53]) in the PP set, and 88.5% (23/26) for Evicel and 35.7% (5/14) for Sutures with an estimated ratio PE/PS of 2.48 (95% CI: [1.39, 5.52]) in the Safety set. Although subjects undergoing infratentorial surgery were few, analysis by surgical approach (ITT set) showed that, within both the supra- and infratentorial stratum, the success rates were markedly higher for Evicel® than for Sutures, while for both Evicel® and Sutures, success rates were higher for supratentorial than for infratentorial procedures.
Secondary endpoints: safety
All Sutures-treated subjects and most Evicel®-treated subjects (84.6%) experienced at least 1 AE (Table 5). Seven AEs in 5 Evicel®-treated subjects (19.2%) and 16 AEs in 8 Sutures-treated subjects (57.1%) were considered serious (Table 5). One SAE of pseudomeningocele in an Evicel®-treated subject was considered related to study product by the Sponsor, while most other SAEs in both groups were considered related or possibly related to the procedure (85.7% and 87.5% for Evicel® and Sutures, respectively) (Table 5).
Events of postoperative CSF leak were categorized as SAEs: 1 pseudomeningocele for Evicel®, 1 pseudomeningocele with iCSF leak and 4 pseudomeningoceles without iCSF leak for Sutures (Table 5).
Overall, 38 AEs (16.7%) were categorized as nervous system disorders. These occurred in 13 Evicel®-treated (50.0%) and 10 Sutures-treated subjects (71.4%) and included headache (9 Evicel®-treated (34.6%) and 5 Sutures-treated (35.7%) subjects), and seizures (1 Evicel®-treated (3.8%) and 1 Sutures-treated subject (7.1%)).
Surgical site complications were overall less frequent in Evicel®- than in Sutures-treated subjects (Table 6). Specifically, the frequency of pseudomeningocele, and to a lesser degree, those of iCSF leak, infection and hematoma, were lower after treatment with Evicel® than Sutures.
Discussion
This trial supports the safety and efficacy of Evicel® as an adjunct to primary sutured dural repair in children undergoing cranial neurosurgery. The trial was conducted as part of a PIP for Evicel®, following approval for this indication in adults. The data mirrors the findings from an earlier clinical trial in adults, by showing that Evicel® performed better than additional sutures in establishing watertight closure of a primary sutured incision, with no safety signals identified [9].
This trial is the first to prospectively investigate fibrin sealant use to prevent CSF leakage in children after cranial neurosurgery, with an intra-operative primary efficacy endpoint and additional suturing as a comparator [9]. The observed 2.76 success rate ratio of Evicel® to Sutures was accompanied by lower rates of postoperative CSF leakage, consisting of iCSF leakage and/or pseudomeningocele, for Evicel®, despite a higher rescue treatment rate in the Sutures group. This supports the adage in neurosurgery that intra-operative watertight dural repair protects against postoperative CSF leakage and its complications, and should be aimed for [8]. In addition to a scarcity of data, current literature on the use of fibrin sealants in pediatric patients is exclusively retrospective and shows high variability in study design, type of dural incision, primary treatment, definition of CSF leak and other outcome measures, and duration of follow-up. As suggested by Slot et al., this may explain why their recent meta-analysis and large retrospective series of pediatric populations failed to show a significant effect of watertight closure on the incidence of postoperative CSF leakage [3, 4]. The authors specifically indicated that most studies were of fair-to-low quality and failed to clearly define CSF leak as a (primary) outcome [3, 4]. In accordance with Evicel®’s current clinical indication [28, 29], the current trial included only dural incisions that allowed for primary suturing of the dural cuff. When such is not possible, e.g. due to dural defects, today’s advanced neurosurgical techniques and dural substitutes help prevent postoperative complications [3, 20, 21, 33]. Recent studies questioning the true need for watertight closure report mostly on supratentorial procedures (with lower hydrostatic pressure) and dural incisions that were systematically augmented by duraplasty or dural overlay [21, 33, 34]. The current trial supports that in scenarios where sutured closure is possible, fibrin sealants such as Evicel® are an effective, readily available, and non-invasive adjunct to obtain watertight closure [9]. Larger controlled trials are needed to demonstrate cost/effectiveness of the currently available adjunctive treatments for dural closure in the pediatric population.
In addition to lower rates of pseudomeningocele and iCSF leak, surgical site complications such as infection, bleeding and other complications were less frequent following Evicel® than additional suturing. Originally developed as hemostats, fibrin sealants may help hemostasis and were shown in multiple randomized and non-randomized studies to support the prevention of surgical site infections [9,10,11,12,13,14,15,16,17,18,19]. Additional sutures, in contrast, may inflict further dural damage through needle holes and dural traction. As a potential treatment failure, the single pseudomeningocele in the Evicel® group was considered an SAE possibly related to Evicel® product. The Sutures group counted 5 SAE of pseudomeningocele, but causal relatedness was not part of the trial’s safety evaluation since no adjunct product was used in that group. Comorbidities were highly prevalent in the study population; these related mostly to serious conditions underlying the surgical indications and, together with the major cranial procedure, may help explain why most subjects experienced one or more AEs. Expectedly, the incidence of AEs was similar in the two groups, as most AEs were considered related or possibly related to the procedure. For Evicel®, however, no new safety signals were identified. In contrast to its predecessor product Quixil®/Crosseal™, Evicel® does not contain the clot-stabilizing agent tranexamic acid (TA). Because preclinical in vivo studies showed that TA could cause seizures and direct effects on neuronal cells when applied to cortex and spinal cord [35,36,37,38,39,40,41], Quixil®/Crosseal™ was contraindicated in procedures where contact with CSF could occur [24]. In the current trial, most AEs in the nervous system disorder category were single events, except for headache, which occurred in circa one third of patients in each group. Seizures were noted in 1 Evicel® subject and in 1 Sutures subject, who had prior histories of seizures.
Although small, the study population was a representative sample of the pediatric neurosurgery population. Intracranial tumor was the most frequent surgical indication, followed by epilepsy. The surgeries involved almost exclusively craniotomies, mostly with a supratentorial approach. These distributions resemble those of other large pediatric series [4, 21]. Infratentorial tumors are more frequent in children than in adults but occur mostly in the first decade of life [42, 43]. The low proportion of infratentorial approaches in this trial is likely due to the wide age range of subjects, half of them being 10 years or older. Moreover, the exclusion of children with hydrocephalus or CSF drainage resulted in the exclusion of patients with Chiari malformation, a pediatric neurosurgical indication that typically requires an infratentorial approach. Surgeries for epilepsy were all supratentorial, consistent with epilepsy surgery commonly being performed on the cerebral lobes [44].
This study presents certain limitations. The study size was small, and data analyses were descriptive. However, designed within the MHRA and EMA PIP frameworks, it is the first to prospectively investigate a dural sealant in the pediatric population, with a randomized controlled design and intra-operative efficacy endpoint. Exclusion of study subjects with major protocol deviations further reduced sample size. However, the PP and Safety sets maintained allocation ratios close to the intended 2:1 ratio, and sensitivity analyses showed consistent efficacy results with ratios of success proportions larger than 2. The exclusions from the PP set concerned 3 Evicel® subjects and 1 Sutures subject with success, and 4 Sutures subjects with failure, placing the Evicel® group at a disadvantage for efficacy analysis. Infratentorial procedures, known to confer a higher risk for CSF leak, were underrepresented. This is due to the relatively small sample size in this study and the fact that the proportion of these patients is a priori expected to be low [4, 21]. In the prior adult trial, however, the success rate of Evicel® was significantly superior to that of sutured dural closure in the infratentorial as well as the supratentorial stratum [9], and, despite the low numbers, the current trial showed a similar trend.
In conclusion, the findings from this trial support the rationale that Evicel® is safe and effective as an adjunct to primary sutured closure of the dura mater in the pediatric population and helps in preventing postoperative CSF leakage and associated surgical site complications. These results closely mirror those from a similar trial with Evicel® conducted previously in an adult population [9].
Data availability
All data generated or analyzed during this study are included in this published article and its Supplementary Material online, and at https://clinicaltrials.gov/ct2/show/study/NCT02309645.
Change history
12 July 2024
A Correction to this paper has been published: https://doi.org/10.1007/s00381-024-06503-8
References
Kinaci A, Slot EMH, Kollen M, Germans MR, Amin-Hanjani S, Carlson AP, Majeed K, Depauw P, Robe PA, Regli L, Charbel FT, van Doormaal TPC (2023) Risk factors and management of incisional cerebrospinal fluid leakage after craniotomy: a retrospective international multicenter study. Neurosurgery 92:1177–1182
Behling F, Kaltenstadler M, Noell S, Schittenhelm J, Bender B, Eckert F, Tabatabai G, Tatagiba M, Skardelly M (2017) The prognostic impact of ventricular opening in glioblastoma surgery: a retrospective single center analysis. World Neurosurg 106:615–624
Slot EMH, van Baarsen KM, Hoving EW, Zuithoff NPA, van Doormaal TPC (2021) Cerebrospinal fluid leakage after cranial surgery in the pediatric population-a systematic review and meta-analysis. Childs Nerv Syst 37:1439–1447
Slot EMH, van Doormaal TPC, van Baarsen KM, Krayenbühl N, Regli L, Germans MR, Hoving EW (2023) Incisional CSF leakage after intradural cranial surgery in children: incidence, risk factors, and complications. J Neurosurg Pediatr 31:313–320
Grotenhuis JA (2005) Costs of postoperative cerebrospinal fluid leakage: 1-year, retrospective analysis of 412 consecutive nontrauma cases. Surg Neurol 64:490–493
van Lieshout C, Slot EMH, Kinaci A, Kollen MH, Hoving EW, Frederix GWJ, van Doormaal TPC (2021) Cerebrospinal fluid leakage costs after craniotomy and health economic assessment of incidence reduction from a hospital perspective in the Netherlands. BMJ Open 11:e052553
Lassen B, Helseth E, Egge A, Due-Tonnessen BJ, Ronning P, Meling TR (2012) Surgical mortality and selected complications in 273 consecutive craniotomies for intracranial tumors in pediatric patients. Neurosurgery 70:936–943
Cushing H (1908) Surgery of the head. In: Keen WW (ed) Surgery, its principles and practice. W.B. Saunders Co, Philadelphia, pp 17–276
Green AL, Arnaud A, Batiller J, Eljamel S, Gauld J, Jones P, Martin D, Mehdorn M, Ohman J, Weyns F (2015) A multicentre, prospective, randomized, controlled study to evaluate the use of a fibrin sealant as an adjunct to sutured dural repair. Br J Neurosurg 29:11–17
Nakamura H, Matsuyama Y, Yoshihara H, Sakai Y, Katayama Y, Nakashima S, Takamatsu J, Ishiguro N (2005) The effect of autologous fibrin tissue adhesive on postoperative cerebrospinal fluid leak in spinal cord surgery: a randomized controlled trial. Spine (Phila Pa 1976) 30:E347–351
(2008) Fibrin sealant for the sealing of dura sutures. NCT00681824. Baxter healthcare corporation-sponsored study. https://clinicaltrials.gov/ct2/show/results/NCT00681824?term=00681824&rank=1
Hutter G, von Felten S, Sailer MH, Schulz M, Mariani L (2014) Risk factors for postoperative CSF leakage after elective craniotomy and the efficacy of fleece-bound tissue sealing against dural suturing alone: a randomized controlled trial. J Neurosurg 121:735–744
Kinaci A, Algra A, Heuts S, O’Donnell D, van der Zwan A, van Doormaal T (2018) Effectiveness of dural sealants in prevention of cerebrospinal fluid leakage after craniotomy: a systematic review. World Neurosurg 118:368–376 e361
Esposito F, Angileri FF, Kruse P, Cavallo LM, Solari D, Esposito V, Tomasello F, Cappabianca P (2016) Fibrin sealants in dura sealing: a systematic literature review. PLoS ONE 11:e0151533
Gazzeri R, Fiore C, Galarza M (2014) (EVICEL) for augmentation of dural closure: results of a preliminary clinical study. Surg Technol Int 25:265–270
Van Doormaal T, Germans MR, Sie M, Brouwers B, Carlson A, Dankbaar JW, Fierstra J, Depauw P, Robe P, Regli L (2021) Single-arm, open-label, multicentre first in human study to evaluate the safety and performance of dural sealant patch in reducing CSF leakage following elective cranial surgery: the ENCASE trial. BMJ Open 11:e049098
Strong MJ, West GA, Woo H, Couture DE, Wilson JA, Munoz LF, Rosen CL, Greenlee JD, van Loveren HR, Iantosca M, Baird CJ, Smith M, McGirt M, Parish J, Asher AL (2017) A pivotal randomized clinical trial evaluating the safety and effectiveness of a novel hydrogel dural sealant as an adjunct to dural repair. Oper Neurosurg (Hagerstown) 13:204–212
Osbun JW, Ellenbogen RG, Chesnut RM, Chin LS, Connolly PJ, Cosgrove GR, Delashaw JB Jr., Golfinos JG, Greenlee JD, Haines SJ, Jallo J, Muizelaar JP, Nanda A, Shaffrey M, Shah MV, Tew JM Jr., van Loveren HR, Weinand ME, White JA, Wilberger JE (2012) A multicenter, single-blind, prospective randomized trial to evaluate the safety of a polyethylene glycol hydrogel (duraseal dural sealant system) as a dural sealant in cranial surgery. World Neurosurg 78:498–504
Than KD, Baird CJ, Olivi A (2008) Polyethylene glycol hydrogel dural sealant may reduce incisional cerebrospinal fluid leak after posterior fossa surgery. Neurosurgery 63:ONS182-186; discussion ONS186-187
Hale AT, Gannon SR, Zhao S, Dewan MC, Bhatia R, Bezzerides M, Stanton AN, Naftel RP, Shannon CN, Pruthi S, Wellons JC (2020) Graft dural closure is associated with a reduction in CSF leak and hydrocephalus in pediatric patients undergoing posterior fossa brain tumor resection. J Neurosurg Pediatr 228–234
Roth J, Benvenisti H, Constantini S (2018) Watertight dural closure in pediatric craniotomies-is it really necessary? World Neurosurg 114:e743–e746
Yu R, Zhu W, Kocharian R, Ilie B, Wang Z, Kang D, Zhao G, Yang H, Shu K, Liu X, Ke Y, Qu S, Mao Y (2022) A multicenter, prospective, randomized clinical study to evaluate the efficacy and safety of fibrin sealant as an adjunct to sutured dural repair. Chin Med J (Engl) 135:2506–2508
Spotnitz WD (2014) Fibrin sealant: the only approved hemostat, sealant, and adhesive-a laboratory and clinical perspective. ISRN Surg 2014:203943
Chalmers RT, Darling Iii RC, Wingard JT, Chetter I, Cutler B, Kern JA, Hart JC (2010) Randomized clinical trial of tranexamic acid-free fibrin sealant during vascular surgical procedures. Br J Surg 97:1784–1789
Dhillon S (2011) Fibrin sealant (evicel® [quixil®/crosseal™]): a review of its use as supportive treatment for haemostasis in surgery. Drugs 71:1893–1915
Fischer CP, Wood CG, Shen J, Batiller J, Hart JC, Patel B, Albala DM (2011) A randomized trial of aprotinin-free fibrin sealant versus absorbable hemostat. Clin Appl Thromb Hemost 17:572–577
Schwartz M, Madariaga J, Hirose R, Shaver TR, Sher L, Chari R, Colonna JO 2nd, Heaton N, Mirza D, Adams R, Rees M, Lloyd D (2004) Comparison of a new fibrin sealant with standard topical hemostatic agents. Arch Surg 139:1148–1154
EVICEL® Fibrin Sealant (Human) - Prescribing information. https://fda.report/DailyMed/57f91587-566c-4a6f-90c7-5050f3d4aaa6
Evicel™ Product information - European medicines agency. https://www.ema.europa.eu/en/medicines/human/EPAR/evicel#product-information-section
EU Clinical Trial Directive. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:02001L0020-20220101
CT3 - Detailed guidance on the collection, verification and presentation of adverse event/reaction reports arising from clinical trials on medicinal products for human use. https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:52011XC0611(01)
CDC/NHSN surveillance definitions for specific types of infections. http://www.socinorte.com/wp-content/uploads/2014/06/CDC-SSI.pdf
Barth M, Tuettenberg J, Thomé C, Weiss C, Vajkoczy P, Schmiedek P (2008) Watertight dural closure: is it necessary? A prospective randomized trial in patients with supratentorial craniotomies. Neurosurgery 63:352–358
Krieger MD, McComb JG, Levy ML (1999) Toward a simpler surgical management of Chiari I malformation in a pediatric population. Pediatr Neurosurg 30:113–121
Schlag MG, Hopf R, Redl H (2000) Convulsive seizures following subdural application of fibrin sealant containing tranexamic acid in a rat model. Neurosurgery 47:1463–1467
Schlag MG, Hopf R, Zifko U, Redl H (2002) Epileptic seizures following cortical application of fibrin sealants containing tranexamic acid in rats. Acta Neurochir (Wien) 144:63–69
Pellegrini A, Giaretta D, Chemello R, Zanotto L, Testa G (1982) Feline generalized epilepsy induced by tranexamic acid (AMCA). Epilepsia 23:35–45
Cox S, Cole M, Mankarious S, Tawil N (2003) Effect of tranexamic acid incorporated in fibrin sealant clots on the cell behavior of neuronal and nonneuronal cells. J Neurosci Res 72:734–746
Furtmüller R, Schlag MG, Berger M, Hopf R, Huck S, Sieghart W, Redl H (2002) Tranexamic acid, a widely used antifibrinolytic agent, causes convulsions by a gamma-aminobutyric acid(A) receptor antagonistic effect. J Pharmacol Exp Ther 301:168–173
Lecker I, Wang DS, Whissell PD, Avramescu S, Mazer CD, Orser BA (2016) Tranexamic acid-associated seizures: causes and treatment. Ann Neurol 79:18–26
Hutchinson RW, Mendenhall V, Abutin RM, Muench T, Hart J (2011) Evaluation of fibrin sealants for central nervous system sealing in the mongrel dog durotomy model. Neurosurgery 69:921–929
Mengide JP, Berros MF, Turza ME, Linares JM (2023) Posterior fossa tumors in children: an update and new concepts. Surg Neurol Int 14:114
Prasad KSV, Ravi D, Pallikonda V, Raman BVS (2017) Clinicopathological study of pediatric posterior fossa tumors. J Pediatr Neurosci 12:245–250
Galan FN, Beier AD, Sheth RD (2021) Advances in epilepsy surgery. Pediatr Neurol 122:89–97
Acknowledgements
We gratefully acknowledge investigator Samantha Hettige (St George's University Hospitals NHS Foundation Trust, London, UK) for participating in the study. Medical writing support, in the form of literature, medical writing and editorial services was provided by An Billiau, MD PhD, Celsus Medical Writing, and funded by Ethicon Inc.
Funding
Ethicon, Inc. provided financial support for the study; Ethicon, Inc. contributed to the design of the study, analysis, and interpretation of data and reviewed the manuscript.
Author information
Authors and Affiliations
Contributions
BI and RK designed the study. GS, SM, KA, JK, VJ and BP collected data. GS, SM, KA, JK, VJ, BI, EB, RK and BP analyzed and interpreted data. GS, SM, KA, JK, VJ, BI, EB, RK and BP provided input, reviewed and approved the final manuscript for submission.
Corresponding author
Ethics declarations
Ethics approval
The trial was conducted in accordance with the International Conference on Harmonization (ICH) Harmonized Tripartite Guideline for Good Clinical Practice (2016); the Declaration of Helsinki (2013); the European Union Clinical Trial Directive (2001/20/EC, May 2001); and the EU GCP Directive (2005/28/EC). Approval from the Office for Research Ethics Committees Northern Ireland, Lisburn, UK, was obtained prior to commencing the trial. Original clinical trial application approval was obtained from UK MHRA on 12 June 2014.
Consent
Study subjects were children younger than 18 years requiring elective or emergent craniotomy or craniectomy for an infra- or supratentorial procedure. Per local regulations, the subject’s parent or legal representative provided informed consent; assent was obtained from subjects who possessed the intellectual and emotional ability to comprehend the trial’s concepts.
Conflict of interest
All participating institutions were in receipt of financial compensation from Ethicon, Inc. for the work conducted for this research study. All investigators were required to complete a Financial Disclosure. B Ilie, E Barnett, R Kocharian are employees of Ethicon, Inc. E Barnett and R Kocharian receive stock options as part of employment with ETHICON. The authors do not have other competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: During the production process, there are some headers within Tables 1, 2, 3, 4, 5 and 6 that were not indented, similar to the submitted version. This makes the understanding and clarity of the table more difficult for the readers. Tables have been updated.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sivakumar, G., Magdum, S., Aquilina, K. et al. Safety and effectiveness of Evicel® fibrin sealant as an adjunct to sutured dural repair in children undergoing cranial neurosurgery. Childs Nerv Syst 40, 2735–2745 (2024). https://doi.org/10.1007/s00381-024-06434-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-024-06434-4