Abstract
Measurement of upper limb function is critical for tracking clinical severity in amyotrophic lateral sclerosis (ALS). The Amyotrophic Lateral Sclerosis Rating Scale-revised (ALSFRS-r) is the primary outcome measure utilised in clinical trials and research in ALS. This scale is limited by floor and ceiling effects within subscales, such that clinically meaningful changes for subjects are often missed, impacting upon the evaluation of new drugs and treatments. Technology has the potential to provide sensitive, objective outcome measurement. This paper is a structured review of current methods and future trends in the measurement of upper limb function with a particular focus on ALS. Technologies that have the potential to radically change the upper limb measurement field and explore the limitations of current technological sensors and solutions in terms of costs and user suitability are discussed. The field is expanding but there remains an unmet need for simple, sensitive and clinically meaningful tests of upper limb function in ALS along with identifying consensus on the direction technology must take to meet this need.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Amyotrophic lateral sclerosis (ALS), also known as motor neurone disease (MND), is a rapidly progressive and ultimately fatal neurodegenerative disease characterized by degeneration of upper and lower motor neurons, with extra motor involvement increasingly recognised [1]. People with ALS experience muscle weakness and spasticity, which results in loss of limb function, respiratory impairment, loss of speech and swallow and in 20–50% cognitive and behavioural change [2]. In about two-thirds of cases, first symptoms appear in the limbs [3], which manifest in problems such as inability to raise the arms, loss of hand dexterity, foot drop, and difficulty walking [4]. A recent study [5] on disease progression reported that symptom development in ALS appeared to be an organised process, with onset in the arm occurring more than bulbar and leg onset, respectively. Among arm-onset patients, involvement of the contralateral arm developed significantly faster compared to other sites.
Currently, there are two drugs approved for ALS: Riluzole, which provides a modest benefit of slowing disease progression; and Edaravone, which has shown limited efficacy in a highly selected cohort of patients [6]. The primary endpoint in the trials for these drugs and in the majority of ALS clinical trials to date has been the Amyotrophic Lateral Sclerosis Rating Scale Revised (ALSFRS-r) [7]. This multi-item ordinal scale relies on reproducible scoring by a trained rater in consultation with the patient, assigning a level of functioning from zero to four for each of twelve domains. It includes specific upper limb items: handwriting, cutting food and handling cutlery and dressing and washing. However, problems with construct validity have been reported and the slope shows a non-linear longitudinal decline [8, 9]. Moreover, analysis of the subgroups within the ALSFRS-r demonstrates floor and ceiling effects, which limit sensitivity and significantly increases the risk of failure to identify a real effect of an intervention under investigation [9, 10].
The measurement of patient outcomes could be improved using additional technology-assisted outcomes [11], such as Inertial Measurement Units (IMUs), activity monitors and motion analysis systems. Such technologies, if widely used, have the potential to address the subjectivity of current measures such as the ALSFRS-r. Additionally, the integration of technology in assessment provides opportunities for remote monitoring and remote data collection in clinical trials [12].
The aim of this paper is to present a structured review of the literature pertaining to both traditional, low tech, measurement tools currently used for assessment of upper limb function and hand dexterity with a specific focus on their application to ALS; and novel technology-enabled devices that will in future provide quantitative measurement of upper limb function and dexterity. Improved measurement of motor function of the upper limb confers an increased power to detect changes for novel therapeutic approaches. Challenges and opportunities in devising and implementing technology are discussed.
Methodology
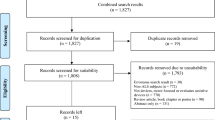
The authors reviewed the literature available on Google Scholar, PubMed, Scopus and general search engines. This structured review includes representative papers in each of the traditional and technology sections as defined by the authors. The following main keywords were used to identify papers of interest which were then assessed by the authors: (1) ALS, amyotrophic lateral sclerosis, MND, motor neurone disease; (2) upper limb, finger tapping test; (3) medical device; (4) neurology, neuromuscular diseases. Inclusion criteria were not limited to ALS focused devices. Any novel device that focused on upper limb impairment was included if there was not a specific ALS equivalent. Exclusion criteria was as follows: posters, technology-based devices developed for healthy participants and multiple papers that used the same technology-based sensors. From this, a representative sample of 43 traditional upper limb measurement papers and 47 technology-based papers were chosen that provide a structured review of the overall field.
Traditional upper limb measurement
Forty-three papers were reviewed which employed traditional upper limb measurement. Assessment of upper limb measurement purports to examine both gross and fine motor control. In ALS this is currently assessed by three questions of the ALSFRS-r, which score handwriting, using utensils or feeding tube fastenings and managing dressing and hygiene. Limitations on detecting impairment resulting from hand dominance versus the affected limb have been recognised, as well as the inability to accommodate for cultural differences [13, 14]. A limited number of trials incorporate objective outcomes by addition of objective measures such as manually picking up objects. Traditional measurement tools include questionnaires, objective functional grading scales such as the Action Research arm Test (ARAT) [15] and Motor Assessment Scale (MAS) [16], and objective tests of impairment including dynamometry for strength measurement, pinch and grip strength testing, gross motors tests such as the box and block test and fine motor tests like the finger tapping test and nine-hole peg test (NHPT). These traditional tests are outlined in Table 1.
At present, there is no consensus between specific questions and the rating system used. The subjective nature of these questionnaires has led to the incorporation of additional objective instruments, as is the case with the ARAT and Jebsen Hand Function Test. These hybrid evaluation tools include sections on tasks related to fine motor control which can be objectively recorded, usually with a stopwatch. However, all inherent subjective biases remain, for example, a delay in a tester starting a stopwatch. Moreover, there has been no cross validation with disease specific scales such as the ALSFRS-r. To the authors’ knowledge, only the NHPT has seen limited use in ALS-specific studies [40].
Due to the subjective nature of the neurological questionnaires, several performance-based tests have been included as part of clinical evaluation (see Table 2). A commonly used instrument is the nine-hole peg test (NHPT), which measures hand dexterity. This has been validated in all age groups, has high interrater validity and is sensitive to patients with neuromuscular or musculoskeletal conditions [41]. It is commercially available, quick, easy to administer and has a minimal ceiling effect. Limitations include the complexity of the task, which can be challenging for patients with cognitive impairment, and the early floor effect for moderate to severe hand impairment, where some useful function of the hand remains but the test cannot be completed.
The Finger Tapping Test (FTT) is one of the most widely used measures of motor function in neurological practice [50, 51]. It involves tapping the index finger against the thumb rapidly while the clinician judges whether the movement is normal or abnormal by visually evaluating amplitude, frequency and accuracy. Visual grading is subjective and for non-expert evaluators, is insensitive to small but meaningful changes. There are currently two main methods used to evaluate the FTT; tip of index finger to tip of thumb or tip of index finger to distal crease of thumb with the distal crease of the thumb suggested as a more sensitive measure [52]. Commercial objective versions of the FTT are limited to simple tapping devices, as these are integral to the Halstead-Reitan Neuropsychological Battery (HRNB), a widely used battery that contains a finger tapping test. This instrument uses a tapping lever mounted with a key-driven mechanical counter [53]. Other devices include the light beam finger tapping test [54, 55], which has limited utility as it is cumbersome and has limited benefits when compared with the current visual assessment used by expert clinicians.
Technology based solutions for upper limb measurement
There has been a substantial increase in the number of novel sensor devices available which have been broadly classified into 4 categories, direct measurement, indirect measurement, keyboard surrogates and mobile applications. These classifications have been synthesised by the authors to distinguish the main differences in measurement methodology. Table 3 provides a summary of the main devices in these four categories including mechanical and clinical advantages. Forty-five papers were found that evaluate these different technology categories. Figure 1 displays a selection of images of a selection of the technology-based sensors.
A Typical example of a glove-based device [56], B accelerometers can be attached to various positions on the hand and wrist to capture movement in terms of acceleration, seen here placed on index finger [59], C gyroscope sensors measure orientation and angular velocity, can be positioned anywhere, seen here with device that fits on thumb and index finger [78], D image of the inertial measurement unit (IMU) developed PD-Monitor, a commercial PD device that focuses on a finger tapping test [66], E magnetometers offer a counterpoint to accelerometer and gyroscopes but are not used much on their own, image shows a device that relies on two magnetometers [65], F Leap Motion Controller (Leap Motion Inc., San Francisco, USA.), a commercial system that detects the motion and portion of the hand using infrared (IR) sensors, G A 3D Marker-based camera setup where position is determined through the use of reflective markers [71], H a digital pen (Manus Neurodynamica Ltd.) that aims to quantify handwriting, along with tablet stylus’ they are bracketed into mobile application devices [79], I example of a mobile app interface designed to measure a tapping test [80]
Direct measurement devices encompass accelerometers, gyroscopes, magnetometers, and inertial measurement units (IMUs). Accelerometer devices which are placed on the index finger and record the acceleration as a finger tap have been developed [59, 60, 81]. Gyroscopes have been used to measure bradykinesia or tremors in Parkinson’s disease (PD) patients [63, 78, 82]. Inertial measurement units (IMUs) combine the input from several different sensors to give a more accurate output of movement. A range of studies [66,67,68, 83, 84] have examined different IMUs for use in hand and finger tracking, most associated with the finger tapping test.
Glove-based systems provide quantitative analysis of hand function, which can be used to guide rehabilitation and improve the patient’s recovery, [57, 85,86,87,88]. However, these devices interfere with normal movement as they cover the hand and pose difficulties with respect to hygiene. Although each sensor has strengths (Table 3), a common issue most with most direct measurement devices is noise, and sensor placement can be extremely varied which limits consensus between researchers.
Indirect measurement devices focus on optical sensor systems that offer an alternative to physical devices placed on a subject’s hand or fingers. There are a number of commercially available systems, such as Vicon (Vicon, Oxford, UK), which use a high-resolution camera setup and strategically placed reflective markers placed on the body. Motion capture systems are more accurate when markers are placed on the participant’s body and used for positioning. Most other marker-based optical systems use either passive or active markers to determine position, but some used a combined camera-based approach with IMUs used as the markers substitute [70]. Systems that record motion capture without the use of markers based on algorithms and pattern recognition. Most systems are expensive and unvalidated in a clinical setting. The Microsoft Kinect and Leap Motion Controller (Leap Motion Inc., San Francisco, USA)) are relatively inexpensive motion capture-based systems. The Kinect has been used [89, 90] to examine reachable workspace as a potential outcome measure in neurological conditions. This system correlated findings with gross motor sub scores of the ALSFRS-r; however, currently available systems are limited in resolution when measuring fine motor movements [91]. The size and space needed for most of the systems also render them unsuitable to clinical settings.
Keyboard typing negates the need for additional sensors and the equipment is readily available. Combinations of keyboard and sensors have been used to quantify upper limb impairment in ALS patients, and to determining a sensitive marker that could be used to monitor disease progression. Other methods such as tapping specific keys [92], calculating an interkeystroke interval (IKI) parameter [75], and determining motor speed from tapping a gaming mouse [93] have also been developed. Although this type of measurement is easy to set up, it is limited as data can only be gathered when tapping the key.
Mobile applications allow for remote monitoring and provide feedback on disease progression. These offer remote monitoring combined with objective testing. Due to the advances in smartphone technology, most phones are now equipped with accelerometers and gyroscopes that can be utilised to provide an accuracy similar to laboratory settings, depending on the measurement aims. Smartphone screens are sensitive to touch and also offer an alternative to the keyboard systems. Most mobile applications use a modified version of the Finger Tapping Test but similar to the keyboard devices, they are limited in their ability to record with data gathered mostly surrounding index finger amplitude and velocity [94,95,96,97,98,99,100,101]. Berry et al. [102] have reported on the benefits of using a mobile app for a self-administered ALSFRS-r, PD applications have been developed that gather hand function information in PD. There is a further additional to this category with the development of other novel tools such as digital pens, for example, the NeuroMotor Pen (Manus Neurodynamica Ltd), that aim to quantify handwriting ability. These are used in conjunction with mobile platforms with the aim of easily integrating them into current commercially available devices (i.e., iPad (Apple Inc.)).
Discussion and conclusion
This review summarised the current literature in relation to the measurement of upper limb function in ALS and included forty-three papers on traditional and forty-five on novel technology-based assessment solutions. There is a paucity of ALS-specific research in this area and the majority of the studies discussed are not ALS specific, as most of the scales and measurement devices developed have focused on other neurological conditions such as PD. Nonetheless, the identified strengths and limitatio ns of these scales and devices and the learnings from these studies are applicable to ALS. The advantages and disadvantages outlined in Tables 1, 2, 3 are universal across neurological conditions and highlight an unmet need for novel, technology-based solutions for assessment of upper limb function.
Sensors such as accelerometers or motion capture systems are cheap, and available with software that supports their use in clinical settings. However, all current systems have limitations, and there is no clear leader in the field. While integration with currently validated questionnaires is important, care must be taken not to limit the potential of an objective sensor by tying it too closely to the subjective questionnaires.
For technology to be effectively used for measurement of hand function or dexterity, it must provide an objective measure of hand function, which is clinically meaningful and sensitive to small but meaningful changes and designed with the patient and clinician in mind (Fig. 2). The rapidly progressive nature of symptoms in ALS provides an additional challenge as assessment tools must be suitable for frequent use and ideally for remote monitoring. Many currently available novel measurements are limited by issues such as cost or complexity of assessment setup and are not amenable to frequent use or suitable for remote monitoring. Simple and widely used measurement tools such as hand grip dynamometry are limited in ALS by rapidly progressive weakness and presence of a floor effect, while some meaningful hand function (e.g., tapping a tablet screen) is preserved.
Data privacy and CE marking of novel devices or algorithms must also be taken into consideration [103]. Adoption of any new device is dependent on the strategies surrounding the CE mark and operational aspects, which reflect decisions that need to be taken early in the development of a device. Clinicians must be satisfied a novel device will give precise, reliable and continuous information about patient limb position and function [104] especially if the information will be used to inform clinical decisions. A thoughtf ully designed sensitive device has the potential to provide enhanced information, which in turn improves the efficiency of clinical trial evaluations [105].
The benefits of technology are clearly recognized. In ALS, the challenge is to develop assessment devices that will adequately address the current limitations of current measurement instruments such as the ALSFRS-R in a reproducible, user-friendly and inexpensive manner. While no currently available device has met all of the necessary criteria to ensure universal acceptance in clinical practice (Fig. 2), there is clearly a demand for technological innovation which will be best achieved by ongoing collaboration between bioengineers and expert clinical professionals.
Change history
17 June 2022
The incorrect word 'structure' was left in instead of 'structured' in the article’s title
References
Andersen PM, Abrahams S, Borasio GD et al (2012) EFNS guidelines on the clinical management of amyotrophic lateral sclerosis (MALS)–revised report of an EFNS task force. Eur J Neurol 19(3):360–375
Phukan J, Elamin M, Bede P et al (2012) The syndrome of cognitive impairment in amyotrophic lateral sclerosis: a population-based study. J Neurol Neurosurg Psychiatry 83(1):102–108
Wijesekera LC, Leigh PN (2009) Amyotrophic lateral sclerosis. Orphanet J Rare Dis 4:3
Gordon PH (2013) Amyotrophic lateral sclerosis: an update for 2013 clinical features, pathophysiology, management and therapeutic trials. Aging Dis 4(5):295–310
Walhout R, Verstraete E, van den Heuvel MP et al (2018) Patterns of symptom development in patients with motor neuron disease. Amyotroph Lateral Scler Frontotemporal Degener 19(1–2):21–28
Oskarsson B, Gendron TF, Staff NP (2018) Amyotrophic lateral sclerosis: an update for 2018. Mayo Clin Proc 93(11):1617–1628
Cedarbaum JM, Stambler N, Malta E et al (1999) The ALSFRS-R: a revised ALS functional rating scale that incorporates assessments of respiratory function. J Neurol Sci 169(1):13–21
Mandrioli J, Biguzzi S, Guidi C et al (2015) Heterogeneity in ALSFRS-R decline and survival: a population-based study in Italy. Neurol Sci 36(12):2243–2252
Rooney J, Burke T, Vajda A et al (2017) What does the ALSFRS-R really measure? A longitudinal and survival analysis of functional dimension subscores in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry 88(5):381–385
Franchignoni F, Mandrioli J, Giordano A et al (2015) A further Rasch study confirms that ALSFRS-R does not conform to fundamental measurement requirements. Amyotroph Lateral Scler Frontotemporal Degener 16(5–6):331–337
van den Berg LH, Sorenson E, Gronseth G et al (2019) Revised Airlie House consensus guidelines for design and implementation of ALS clinical trials. Neurology 92(14):e1610–e1623
van Eijk RPA, de Jongh AD, Nikolakopoulos S et al (2021) An old friend who has overstayed their welcome: the ALSFRS-R total score as primary endpoint for ALS clinical trials. Amyotroph Lateral Scler Frontotemporal Degener. https://doi.org/10.1080/21678421.2021.1879865
Pinto S, Gromicho M, de Carvalho M (2019) Assessing upper limb function with ALSFRS-R in amyotrophic lateral sclerosis patients. Amyotroph Lateral Scler Frontotemporal Degener 20(5–6):445–448
Hu F, Jin J, Jia R et al (2017) Measuring the validation of assessing the non-dominant-hand function by ALSFRS-r in Chinese ALS patients. J Clin Neurosci 46:17–20
Lyle RC (1981) A performance test for assessment of upper limb function in physical rehabilitation treatment and research. Int J Rehabil Res 4(4):483–492
Carr JH, Shepherd RB, Nordholm L et al (1985) Investigation of a new motor assessment scale for stroke patients. Phys Ther 65(2):175–180
Dowrick AS, Gabbe BJ, Williamson OD et al (2006) Does the disabilities of the arm, shoulder and hand (DASH) scoring system only measure disability due to injuries to the upper limb? J Bone Jt Surg Br 88-B(4):524–527
Gummesson C, Atroshi I, Ekdahl C (2003) The disabilities of the arm, shoulder and hand (DASH) outcome questionnaire: longitudinal construct validity and measuring self-rated health change after surgery. BMC Musculoskelet Disord 4:11–11
Chesworth BM, Hamilton CB, Walton DM et al (2014) Reliability and validity of two versions of the upper extremity functional index. Physiother Can 66(3):243–253
Hefford C, Abbott JH, Arnold R et al (2012) The patient-specific functional scale: validity, reliability, and responsiveness in patients with upper extremity musculoskeletal problems. J Orthopaed Sports Phys Therapy. 42(2):56–65
Stratford P, Gill C, Westaway M et al (1995) Assessing disability and change on individual patients: a report of a patient specific measure. Physiother Can 47(4):258–263
Simone A, Rota V, Tesio L et al (2011) Generic ABILHAND questionnaire can measure manual ability across a variety of motor impairments. Int J Rehabil Res 34(2):131–140
Penta M, Tesio L, Arnould C et al (2001) The ABILHAND Questionnaire as a measure of manual ability in chronic stroke patients. Stroke 32(7):1627–1634
Chung KC, Hamill JB, Walters MR et al (1999) The Michigan Hand Outcomes Questionnaire (MHQ): assessment of responsiveness to clinical change. Ann Plast Surg 42(6):619–622
Shauver MJ, Chung KC (2013) The Michigan hand outcomes questionnaire after 15 years of field trial. Plast Reconstr Surg 131(5):779e–787e
Ashford S, Slade M, Turner-Stokes L (2013) Conceptualisation and development of the arm activity measure (ArmA) for assessment of activity in the hemiparetic arm. Disabil Rehabil 35(18):1513–1518
Platz T, Pinkowski C, van Wijck F et al (2005) Reliability and validity of arm function assessment with standardized guidelines for the Fugl-Meyer Test, Action Research Arm Test and Box and Block Test: a multicentre study. Clin Rehabil 19(4):404–411
Carpinella I, Cattaneo D, Ferrarin M (2014) Quantitative assessment of upper limb motor function in Multiple Sclerosis using an instrumented Action Research Arm Test. J Neuroeng Rehabil 18(11):67
Disease MDSTFORSFPS (2003) The Unified Parkinson’s Disease Rating Scale (UPDRS): status and recommendations. Mov Disord 18(7):738–750
Goetz CG, Fahn S, Martinez-Martin P et al (2007) Movement Disorder society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): process, format, and clinimetric testing plan. Mov Disord 22(1):41–47
Ferrucci L, Koh C, Bandinelli S et al (2007) Disability, functional status, and activities of daily living. In: Birren JE (ed) Encyclopedia of gerontology, 2nd edn. Elsevier, New York, pp 427–436
Cech DJ, Martin ST (2012) Chapter 5-evaluation of function, activity, and participation. In: Cech DJ, Martin ST (eds) Functional movement development across the life span, 3rd edn. W.B. Saunders, Saint Louis, pp 88–104
Dodds TA, Martin DP, Stolov WC et al (1993) A validation of the Functional Independence Measurement and its performance among rehabilitation inpatients. Arch Phys Med Rehabil 74(5):531–536
van der Putten JJMF, Hobart JC, Freeman JA et al (1999) Measuring change in disability after inpatient rehabilitation: comparison of the responsiveness of the Barthel Index and the Functional Independence Measure. J Neurol Neurosurg Psychiatry 66(4):480
Uswatte G, Taub E, Morris D et al (2005) Reliability and validity of the upper-extremity Motor Activity Log-14 for measuring real-world arm use. Stroke 36(11):2493–2496
Poole JL, Whitney SL (1988) Motor assessment scale for stroke patients: concurrent validity and interrater reliability. Arch Phys Med Rehabil 69(3 Pt 1):195–197
Wolf SL, Catlin PA, Ellis M et al (2001) Assessing Wolf motor function test as outcome measure for research in patients after stroke. Stroke 32(7):1635–1639
Van de Winckel A, Feys H, Lincoln N et al (2007) Assessment of arm function in stroke patients: Rivermead Motor Assessment arm section revised with Rasch analysis. Clin Rehabil 21(5):471–479
Côté R, Battista RN, Wolfson C et al (1989) The Canadian Neurological Scale. Valid Reliab Assess 39(5):638–638
Czell D, Neuwirth C, Weber M et al (2019) Nine hole peg test and transcranial magnetic stimulation: useful to evaluate dexterity of the hand and disease progression in amyotrophic lateral sclerosis. Neurol Res Int. https://doi.org/10.1155/2019/7397491
Joyce NC, McDonald CM (2012) Neuromuscular disease management and rehabilitation, part I: diagnostic and therapy issues. Phys Med Rehab Clin N Am. https://doi.org/10.1016/j.pmr.2012.06.013
Oxford Grice K, Vogel KA, Le V et al (2003) Adult norms for a commercially available Nine Hole Peg Test for finger dexterity. Am J Occup Ther 57(5):570–573
Mathiowetz V, Weber K, Kashman N et al (1985) Adult norms for the nine hole peg test of finger dexterity. Occup Therapy J Res 5(1):24–38
Amirjani N, Ashworth NL, Olson JL et al (2011) Validity and reliability of the Purdue Pegboard Test in carpal tunnel syndrome. Muscle Nerve 43(2):171–177
Kirby TJ (1979) Dexterity testing and residents’ surgical performance. Trans Am Ophthalmol Soc 77:294–307
Desrosiers J, Rochette A, Hébert R et al (1997) The Minnesota manual dexterity test: reliability, validity and reference values studies with healthy elderly people. Can J Occup Ther 64(5):270–276
Desrosiers J, Bravo G, Hébert R et al (1994) Validation of the box and block test as a measure of dexterity of elderly people: reliability, validity, and norms studies. Arch Phys Med Rehabil 75(7):751–755
Mathiowetz V, Wiemer DM, Federman SM (1986) Grip and pinch strength: norms for 6- to 19-year-olds. Am J Occup Ther 40(10):705–711
Jebsen RH, Taylor N, Trieschmann RB et al (1969) An objective and standardized test of hand function. Arch Phys Med Rehabil 50(6):311–319
Reitan RM, Wolfson D (1993) The Halstead-Reitan neuropsychological test battery theory and clinical interpretation. Neuropsychology Press, Tucson (English)
Ashendorf L, Vanderslice-Barr JL, McCaffrey RJ (2009) Motor tests and cognition in healthy older adults. Appl Neuropsychol 16(3):171–176
Shirani A, Newton BD, Okuda DT (2017) Finger tapping impairments are highly sensitive for evaluating upper motor neuron lesions. BMC Neurol 17(1):55
Christianson M, Leathem J (2004) Development and standardisation of the computerised finger tapping test: Comparison with other finger tapping instruments. N Z J Psychol 33:44–49
Roalf DR, Rupert P, Mechanic-Hamilton D et al (2018) Quantitative assessment of finger tapping characteristics in mild cognitive impairment, Alzheimer’s disease, and Parkinson’s disease. J Neurol 265(6):1365–1375
Coleman AR, Moberg PJ, Ragland JD et al (1997) Comparison of the halstead-reitan and infrared light beam finger tappers. Assessment 4(3):277–286
Grandez K, Solas G, Bustamante P et al (2010) Sensor device for testing activities in Parkinson and ALS patients. In: 2010 4th International Conference on Pervasive Computing Technologies for Healthcare, pp 22–25
Halic T, Kockara S, Demirel D et al (2014) MoMiReS: Mobile mixed reality system for physical and occupational therapies for hand and wrist ailments. In: 2014 IEEE Innovations in Technology Conference, pp 16–16
Dai H, Lin H, Lueth TC (2015) Quantitative assessment of parkinsonian bradykinesia based on an inertial measurement unit. Biomed Eng Online 14:68–68
Stamatakis J, Ambroise J, Crémers J et al (2013) Finger tapping clinimetric score prediction in Parkinson’s disease using low-cost accelerometers. Comput Intell Neurosci 2013(04/16):717853
Okuno R, Yokoe M, Akazawa K et al (2006) Finger taps movement acceleration measurement system for quantitative diagnosis of Parkinson's disease. In: Conference proceedings : Annual International Conference of the IEEE Engineering in Medicine and Biology Society IEEE Engineering in Medicine and Biology Society Conference, pp 6623–6626
Prätorius M, Valkov D, Burgbacher U et al (2014) DigiTap: an eyes-free VR/AR symbolic input device. Proc ACM Symp Virtual Real Softw Technol 11(11):9–18
Bobić V, Djurić-Jovičić M, Dragašević N et al (2019) An expert system for quantification of bradykinesia based on wearable inertial sensors. Sensors (Basel, Switzerland) 19(11):2644
Salarian A, Russmann H, Wider C et al (2007) Quantification of tremor and bradykinesia in Parkinson’s disease using a novel ambulatory monitoring system. IEEE Trans Biomed Eng 54(2):313–322
Kim JW, Lee JH, Kwon Y et al (2011) Quantification of bradykinesia during clinical finger taps using a gyrosensor in patients with Parkinson’s disease. Med Biol Eng Comput 49(3):365–371
Sano Y, Kandori A, Shima K et al (2016) Quantifying Parkinson’s disease finger-tapping severity by extracting and synthesizing finger motion properties. Med Biol Eng Comput 54(6):953–965
Gao C, Smith S, Lones M et al (2018) Objective assessment of bradykinesia in Parkinson’s disease using evolutionary algorithms: clinical validation. Transl Neurodegener 7:18
Rovini E, Esposito D, Fabbri L, Pancani S, Vannetti F, Cavallo F (2019) Vision Optical-Based Evaluation of Senshand Accuracy for Parkinson’s Disease Motor Assessment. In: 2019 IEEE International Symposium on Measurements & Networking (M&N) 2019: pp. 1–6
Martinez-Manzanera O, Roosma E, Beudel M et al (2016) A method for automatic and objective scoring of bradykinesia using orientation sensors and classification algorithms. IEEE Trans Biomed Eng 63(5):1016–1024
Krupicka R, Viteckova S, Cejka V et al (2017) BradykAn: a motion capture system for objectification of hand motor tests in Parkinson Disease. In: 2017 E-Health and Bioengineering Conference (EHB), pp 22–24
di Biase L, Summa S, Tosi J et al (2018) Quantitative analysis of bradykinesia and rigidity in Parkinson’s disease. Front Neurol 9:121
Jobbagy A, Harcos P, Karoly R et al (2005) Analysis of finger-tapping movement. J Neurosci Methods 141(1):29–39
Lee WL, Sinclair NC, Jones M et al (2019) Objective evaluation of bradykinesia in Parkinson’s disease using an inexpensive marker-less motion tracking system. Physiol Meas 40(1):014004
Mitsi G, Mendoza EU, Wissel BD et al (2017) Biometric digital health technology for measuring motor function in parkinson’s disease: results from a feasibility and patient satisfaction study. Front Neurol 8:273–273
Bot BM, Suver C, Neto EC et al (2016) The mPower study, Parkinson disease mobile data collected using ResearchKit. Sci Data 3(1):160011
Austin D, Jimison H, Hayes T et al (2011) Measuring motor speed through typing: a surrogate for the finger tapping test. Behav Res Methods 43(4):903–909
Da Silva FN, Irani F, Richard J et al (2012) More than just tapping: index finger-tapping measures procedural learning in schizophrenia. Schizophr Res 137(1–3):234–240
Gur RC, Richard J, Hughett P et al (2010) A cognitive neuroscience-based computerized battery for efficient measurement of individual differences: standardization and initial construct validation. J Neurosci Methods 187(2):254–262
Djurić-Jovičić M, Jovičić NS, Roby-Brami A et al (2017) Quantification of finger-tapping angle based on wearable sensors. Sensors (Basel, Switzerland) 17(2):203
Ltd. MN. 2021 [15th December 2021]. Available from: https://www.manusneuro.com/
Wissel BD, Mitsi G, Dwivedi AK et al (2018) Tablet-based application for objective measurement of motor fluctuations in Parkinson disease. Digit Biomark 1(2):126–135
Patel S, Sherrill D, Hughes R et al (20006) Analysis of the severity of dyskinesia in patients with Parkinson's disease via wearable sensors. In: International Workshop on Wearable and Implantable Body Sensor Networks (BSN'06), pp 3–5
Seok HY, Kim JW, Kim YH et al (2019) Quantitative evaluation of hand motor function using a gyrosensor in mild and moderate carpal tunnel syndrome. Muscle Nerve 59(4):465–469
Salchow-Hommen C, Callies L, Laidig D et al (2019) A tangible solution for hand motion tracking in clinical applications. Sensors (Basel) 19(1):208
Akhbardeh A, Arjona JK, Krysko KM et al (2020) Novel MS vital sign: multi-sensor captures upper and lower limb dysfunction. Ann Clin Transl Neurol 7(3):288–295
Wang Q, Markopoulos P, Yu B et al (2017) Interactive wearable systems for upper body rehabilitation: a systematic review. J Neuroeng Rehabil 14(1):20
Hsiao P, Yang S, Lin B, Lee I, Chou W (2015) Data glove embedded with 9-axis IMU and force sensing sensors for evaluation of hand function. In: 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), 2015, pp. 4631–4634
Simone LK, Sundarrajan N, Luo X et al (2007) A low cost instrumented glove for extended monitoring and functional hand assessment. J Neurosci Methods 160(2):335–348
van Ommeren AL, Sawaryn B, Prange-Lasonder GB et al (2019) Detection of the intention to grasp during reaching in stroke using inertial sensing. IEEE Trans Neural Syst Rehabil Eng 27(10):2128–2134
Han JJ, Kurillo G, Abresch RT et al (2015) Reachable workspace in facioscapulohumeral muscular dystrophy (FSHD) by Kinect. Muscle Nerve 51(2):168–175
Oskarsson B, Joyce NC, De Bie E et al (2016) Upper extremity 3-dimensional reachable workspace assessment in amyotrophic lateral sclerosis by Kinect sensor. Muscle Nerve 53(2):234–241
Butt AH, Rovini E, Dolciotti C et al (2018) Objective and automatic classification of Parkinson disease with Leap Motion controller. Biomed Eng Online 17(1):168
Giovannoni G, van Schalkwyk J, Fritz VU et al (1999) Bradykinesia akinesia inco-ordination test (BRAIN TEST): an objective computerised assessment of upper limb motor function. J Neurol Neurosurg Psychiatry 67(5):624–629
Hubel K, Yund E, Herron T et al (2013) Computerized measures of finger tapping: Reliability, malingering and traumatic brain injury. J Clin Exp Neuropsychol 35:745–758
Arora S, Venkataraman V, Zhan A et al (2015) Detecting and monitoring the symptoms of Parkinson’s disease using smartphones: a pilot study. Parkinsonism Relat Disord 21(6):650–653
Kassavetis P, Saifee TA, Roussos G et al (2015) Developing a tool for remote digital assessment of Parkinson’s disease. Mov Disord Clin Pract 3(1):59–64
Lee CY, Kang SJ, Hong S-K et al (2016) A validation study of a smartphone-based finger tapping application for quantitative assessment of bradykinesia in Parkinson’s disease. PLoS ONE 11(7):e0158852–e0158852
Lee W, Evans A, Williams DR (2016) Validation of a smartphone application measuring motor function in Parkinson’s disease. J Parkinsons Dis 6(2):371–382
Printy BP, Renken LM, Herrmann JP et al (2014) Smartphone application for classification of motor impairment severity in Parkinson’s disease. Conf Proc IEEE Eng Med Biol Soc 2014:2686–2689
Memedi M, Sadikov A, Groznik V et al (2015) Automatic spiral analysis for objective assessment of motor symptoms in Parkinson’s disease. Sensors (Basel, Switzerland) 15(9):23727–23744
Memedi M, Khan T, Grenholm P et al (2013) Automatic and objective assessment of alternating tapping performance in Parkinson’s disease. Sensors (Basel, Switzerland) 13(12):16965–16984
Graça R, e Castro RS, Cevada J (2014) ParkDetect: Early diagnosing Parkinson's Disease. In: 2014 IEEE International Symposium on Medical Measurements and Applications (MeMeA), 2014:pp. 1–6
Berry JD, Paganoni S, Carlson K et al (2019) Design and results of a smartphone-based digital phenotyping study to quantify ALS progression. Ann Clin Transl Neurol 6(5):873–881
Van Eijk RP, Beelen A, Kruitwagen ET, Murray D, Radakovic R, Hobson E, Knox L, Helleman J, Burke T, Pérez MÁ, Reviers E (2021) A road map for remote digital health technology for motor neuron disease. J Med Internet Res 23(9):e28766
Ravizza A, De Maria C, Di Pietro L et al (2019) Comprehensive review on current and future regulatory requirements on wearable sensors in preclinical and clinical testing [review]. Front Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2019.00313
Gresham G, Schrack J, Gresham LM et al (2018) Wearable activity monitors in oncology trials: current use of an emerging technology. Contemp Clin Trials 64:13–21
Acknowledgements
This work was supported by the Health Research Board (HRB), Grant number MRCG-2018-03 and the Irish Motor Neurone Disease Foundation (no grant number).
Funding
Open Access funding provided by the IReL Consortium.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hayden, C.D., Murphy, B.P., Hardiman, O. et al. Measurement of upper limb function in ALS: a structured review of current methods and future directions. J Neurol 269, 4089–4101 (2022). https://doi.org/10.1007/s00415-022-11179-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-022-11179-8