Abstract
Exosomes are extracellular vesicles that are formed by two invaginations of the plasma membrane and can be released by all eukaryotic cells. Because of their bioactive contents, including nucleic acids and proteins, exosomes can activate a variety of functions in their recipient cells. Due to the plethora of physiological and pathophysiological functions, exosomes have received a lot of attention from researchers over the past few years. However, there is still no consensus regarding isolation and characterization protocols of exosomes and their subtypes. This heterogeneity poses a lot of methodical challenges but also offers new clinical opportunities simultaneously. So far, exosome-based research is still mostly limited to preclinical experiments and early-stage clinical trials since the translation of experimental findings remains difficult. Exosomes could potentially play an important role as future diagnostic and prognostic agents and might also be part of the development of new treatment strategies. Therefore, they have previously been investigated in a variety of nephrological and urological conditions such as acute kidney injury or prostate cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Extracellular vesicles (EVs) are membrane-enclosed, vesicular structures that can be subdivided into three major categories based on their size and biogenesis: apoptotic bodies, microvesicles, and exosomes [71]. Exosomes are the smallest EVs with a diameter of approximately 40 to 160 nm. They are formed by inward-budding of endosomes and are released from multivesicular bodies (MVB) by the vast majority of eukaryotic cells [45, 71, 110]. Like most EVs, exosomes play a major role for intercellular communication and contain bioactive contents composed of proteins, lipids, and nucleic acids [65, 66, 72, 84, 96]. Some proteins (CD9, CD63, CD81) are widely expressed among all exosomes, but the identification of specific exosomal marker proteins remains difficult because a majority of essential proteins for formation and secretion of exosomes (see Fig. 1) can be found among all EVs [47, 66, 75]. Additionally, the composition of exosomes is dependent on the cell of origin and its current state. Consequently, the secreting cell is able to elicit a variety of cell-specific functions in its target cells, including physiological and pathophysiological effects for example in different cancers or renal diseases [109, 110].
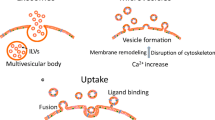
Biogenesis of exosomes: (1) ESCRT-dependent pathway: ESCRT 0-III work closely together to facilitate the second budding step and sort ubiquitin-tagged proteins into the ILVs. They are assisted by the syndecan-syntenin-ALIX adapter complex, which stabilizes ESCRT III at the neck of the vesicle. Finally, ESCRT III mediates the sequestration of the vesicles into the lumina of the endosomes. (2) ESCRT independent pathway: Budding is mainly mediated by a modified lipid composition of the vesicle membrane, with ceramides, cardiolipids, or cholesterol. Flotillins and Rab31 help to internalize tyrosine kinase receptors besides acting as scaffold proteins and preventing lysosomal degradation, respectively. (3) A third major contribution is provided by tetraspanins: they mediate the sorting of various proteins into the ILVs by forming of microdomains. The image was created with CorelDRAW Graphics Suite (Corel, Ottawa, ON, Canada)
The heterogeneous content of exosomes, their high stability, the protection of the cargo through a liquid bilayer, and their function in intercellular communication has led to a strong interest in pursuing their utilization for diagnosis, prognosis, carriers of vaccines, and therapy of different diseases with a large focus on cancer [56, 81, 109].
The procedures required for the isolation and proliferation of exosomes, along with their bioactive and cell-specific components are important steps for the characterization of exosomes and their potential diagnostic and prognostic value [109]. Several different methods and protocols for the isolation, separation, proliferation, and characterization of exosomes have led to insufficiently supported conclusions concerning the functions and applications of EVs. To improve reliability, reproducibility, and acceptance of EV research, the International Society for Extracellular Vesicles (ISEV) published updated Minimal Information for Studies of Extracellular Vesicles (MISEV) guidelines in 2018. These guidelines aim at implementing a methodical standard for the acquisition of EV- and exosome-related data to enable a sufficient interpretation of the acquired results [94].
In this review, we provide an overview on the current state of research on exosomes and their medical applications in nephrology and urology. Due to the afore mentioned difficulty in assigning specific functions and features to EV subtypes, ISEV has recommended to stop using terms such as exosomes which are associated with sometimes contradictory findings [78]. Despite these difficulties, the terms exosomes and EVs were used cautiously and on the basis of the descriptions of the cited works.
Background: structure and biogenesis
Exosomes are generated by two invaginations: First, endosomes are sequestered from the plasma membrane. Once matured to late endosomes, they release so-called intraluminal vesicles (ILVs) into the lumina, which become multivesicular endosomes or multivesicular bodies (MVBs). Subsequently, there are two different pathways for the further development: either they merge with lysosomes or autophagosomes, causing the degradation of their content, or they merge with the plasma membrane, releasing their entrapped ILVs into the extracellular space as exosomes (Fig. 1). This whole process is accompanied by the constant restructuring of both the membrane components (lipids, surface proteins) of the respective vesicles and their contents including proteins, nucleic acids, and metabolites. Overall, the lipid composition of the exosomal membrane largely consists of membrane lipids and has been described as relatively similar amongst exosomes of different cellular origin but varies from the composition of the parental cell.
Although the endosomal origin of exosomes has been generally acknowledged for several decades, the molecular mechanisms of exosomal biogenesis are still unclear. Even though many details have not been clarified yet, researchers have still agreed on two possible pathways of biogenesis: an ESCRT (endosomal sorting complex required for transport)-dependent pathway and an ESCRT-independent pathway, mediated by tetraspanins and several lipids (Fig. 1) [7, 75, 103]. The existence of the latter and its role in the case of dysfunction of the ESCRT machinery has been proven by siRNA-induced depletion of various ESCRT key elements by Stuffers et al. [92].
The coexistence of these two pathways at the cellular level and how much each mechanism contributes proportionally to ILV formation is likely cell type specific [37] and is supposed to generate pathway-depending MVB subtypes [92]. So far, it is believed that there is a close interplay of ESCRT-dependent and independent mechanisms, enabling cells to secrete exosomes under variable cellular conditions. The secretion and specific uptake of exosomes which are essential for the intercellular communication via exosomes are believed to be regulated by intersecting pathways as well.
Methods for the isolation of EVs, included exosomes
In accordance with the MISEV guidelines of the ISEV from 2018, the purification of exosomes from collected bio-fluids and the separation from non-EV components and other EV-types can be performed by various methods. These include ultracentrifugation (UC), precipitation methods (e.g. polyethylene glycol (PEG)-based), density gradient centrifugation (DGC), size exclusion chromatography (SEC), and immunomagnetism, among others [94]. According to two worldwide surveys from 2015 and 2019, separation via ultracentrifugation (UC) is the most frequently used method but alternative separation techniques increased from 2015 to 2019 (Table 1) [32, 78]. Recent findings revealed a strong influence of separation methods on subsequent RNA analyses and high method-to-method variations. Ideally, exosome separation methods are selected based on the target bio-fluid and molecular analyte [23, 70, 90]. In addition to the MISEV guidelines, the Urine Task Force of ISEV presents the current state of the art as well as current challenges in urinary EV analyses for clinical applications in great detail [25].
Besides total isolation of EVs and exosomes from biofluids, specific isolation methods for tumor EVs are investigated. Recent models of EV kinetics suggest that bulk EV measurements are limited in their sensitivity to detect small tumors due to low signal-to-noise ratios from small tumors [27]. To increase the signal from tumor derived exosomes, several approaches to enrich, deplete, and filter EVs are under active development. This may result in improved early diagnostic approaches in comparison to diagnostics based on bulk measurements [28, 71, 73].
Preparation of fluids
According to the current MISEV guidelines, extracted fluids containing the targeted EVs have to be pre-cleaned (centrifugation and/or filtration) to remove remaining cells and cellular debris [94]. After pre-cleaning of the supernatant, a protease inhibitor can be added to prevent protein degradation [20]. For EDTA-blood samples (or other body fluids like cerebrospinal fluid), a second centrifugation step should be implemented to remove platelets and remaining white blood cells [16]. Due to the high concentration of uromodulin (Tamm-Horsfall protein) in urine, some uromodulin-bound EVs are included in the low-speed pellet after centrifugation and not in the high-speed pellet. They can be released by the reduction of disulfide-bonds of uromodulin through treatment with reducing agents (e.g., Dithiothreitol) [29].
Isolation and purification methods
Centrifugation-based methods
UC can be used for the isolation of EVs from all fluids (cell culture supernatants and all body fluids), and control fractions can be used as negative controls [16]. The limitations of UC include the fact that it is a time-intensive procedure which is restricted to the processing of minor volumes and the impact of centrifugal force on the vesicles. Furthermore, portions of the sample can be lost or contaminated with particles of similar density and size with limited reproducibility. A combination with DGC, SEC, or filtration is recommended to avoid contamination [16, 26]. Brennan et al. reached the highest ratio of plasma-derived EVs with 61–150 nm/particles with 0–60 nm with a combination of DGC and UC. However, this combination also had the highest level of unwanted APO-B-containing lipoproteins [9]. Because of the above-mentioned disadvantages and the high costs of UC, other methods are needed, for example, DGC itself where EVs are isolated with a sucrose density gradient. This methodology is especially well suited for the extraction of EVs with low content [57], but contamination of the targeted fractions with particles of similar density is still possible.
Precipitation and size exclusion-based methods
Another method is the precipitation of less soluble components by introducing water-excluding polymers such as PEG or lectins to the samples and the isolation of precipitated EVs which are unable to solubilize with subsequent centrifugation or filtration. This enables the isolation of EVs from large sample sizes and represents the basis of a variety of commercial isolation kits (e.g., ExoQuick ULTRA by System Biosciences) [89]. Furthermore, this method can process around 2.5-fold higher concentrations than UC and uses no centrifugal forces while being easily reproducible [16, 26] and has been used increasingly in recent years (Table 1). Setbacks of this method include the number of impurities (e.g., IgG or Albumin which are likely bound to exosomes) [16] and the nonspecific isolation of vesicles (particles with sizes > 150 nm and < 60 nm) [9]. Alternatively size exclusion methods which rely on the separation of EVs based on their size can be used. SEC, which divides the particles by size using porous polymer microspheres, has gained a lot of attention [89]. It enables the division of the different vesicle classes into distinct fractions [16] and can be performed afterwards for the exclusion of remaining contaminants (e.g., in high-performance liquid chromatography (HPLC)).
Novel isolation methods
Immunoaffinity/-magnetic methods (e.g., MACS-Separation) are also promising because they are easy to use and EVs are not subjected to centrifugal or chemical forces (Miltenyi Biotec B.V. & Co. KG, Bergisch Gladbach, Germany). They require the definition of EV-specific membrane-bound biomarkers that can be targeted by antibodies and in the case of immunomagnetic isolation immune-magnetic beads that are coated with antibodies and can be separated from the structure of interest by a magnetic field [89]. Currently, there are only three antibodies available for this purpose which highlights the need for identification of more specific exosomal targets and tissue-specific EV targets. Other novel methods include microfluidics (combination of immunoaffinity via antibody-linked binding and membrane filtration) which are often costly and can lead to a loss of function in EVs [83] (Table 1) or EXODUS (exosome detection via the ultrafast-isolation system) which was established as a new method of ultracentrifugation. It is based on the coupling of two oscillators, which set the nanoporous membranes of the filter system into high-frequency vibration by generating transverse waves. This vibration prevents clogging of the filter, which gives EXODUS a considerable advantage over conventional filtration methods. Furthermore, EXODUS is significantly less time-consuming than other methods such as ultracentrifugation or PEG precipitation and provides better results in terms of yield and purity, as Chen et al. were able to demonstrate using urine samples [15]. A final isolation method that should be mentioned here is asymmetric flow field-flow fractionation (AF4). This method makes use of the different hydrodynamic properties of macromolecules for separation. A laminar flow is generated, which separates the analytes diffusing freely through a semi-permeable membrane according to their size. The major difference to conventional chromatographic methods is the absence of a stationary phase, which means that the analytes are not exposed to any of the known risks caused by binding to the stationary phase or shear forces. Although this method has been known for several decades, AF4 was only established for exosome isolation in 2019 by Zhang et al. AF4 is characterized by its good reproducibility, as well as its flexibility as the precise isolation is not dependent on specific surface molecules. It is also favored because of its shorter processing time and the lack of contamination by smaller molecules [107].
In conclusion, there is a mix of different isolation methods that display advantages and disadvantages with no standardized isolation procedure. Choosing the right method should be based on the type and volume of fluid that EVs are isolated from as well as on the design of the study. To utilize EVs in a clinical setting (e.g., as biomarkers), several challenges such as the isolation of different subtypes have yet to be overcome [97]. At the moment, methods for EV isolation are optimized accordingly, and new possibilities such as flow cytometry are being explored as alternative procedures [89].
Characterization and analysis
To ensure a successful isolation, the isolated vesicles should be visualized, quantified, and analyzed. For the visualization, identification of EVs and assessment of their integrity, either single EV particles or their content, can be analyzed.
Microscopic methods
Microscopic analyses such as transmission electron microscopy (TEM), cryo-EM, and other methods (scanning-probe microscopy (SPM) including atomic force microscopy (AFM)) [94] not only enable the visualization of EV morphology but can depict mechanisms of EV uptake and secretion as well. EV imaging also includes fluorescent imaging, which is useful for visualization within cells, cell groups and tissues (e.g. NanoImager, fluorescence microscopy) [25] (Keyence GmbH, Neu-Isenburg, Germany). Other clinical approaches include magnetic resonance imaging (MRI) in combination with nanoparticle-labelled exosomes [10], which can be used for the visualization of the distribution of exosomes. Some of these methods require the preparation or fixation of the samples, which has been argued to possibly change the morphology of EVs, which is why new microscopic technologies such as cryo-EM or atomic force microscopy (AFM) are supposed to be introduced for EV characterization [89]. Until then, nanoparticle tracking analysis (NTA) is amongst one of the most popular methods, which can be used for determining the size distribution (as it is very accurate when it comes to the measurement of size (Malvern Panalytical Ltd, Malvern, UK)) as well as the concentration of isolated EVs based on laser measurements of the scattered light of EVs in solution [16, 89]. However, NTA cannot distinguish between exosomes and other similar vesicles, which is why the concentration can be overestimated.
Identification of proteins, metabolites, and lipids
In comparison, flow cytometry, which enables the examination of the heterogeneity of the surface protein expression, is characterized by its ability to distinguish different (sub-)types of analytes. It allows the identification of single particles based on their fluorescent signals which are generated by a laser beam as they are passing through a small nozzle [89]. Here, no other vesicles are detected due to targeted immunostaining [16] which can be regarded as the biggest disadvantage, similar to immunomagnetics. Due to the low number of specific antibodies available as well as the low sensitivity caused by the small size of EVs [89], presumably not all subpopulations of EVs can be detected.
The most common, cost-, and time-saving method to identify isolated EVs and to analyze the different fractions of DGC or SEC is the western blot (WB) [33]. It can be used as an Immunoblot to identify unwanted contaminants, for example by targeting apolipoproteins (e.g., APO-E, APO-B) with antibodies to detect lipoprotein vesicles [9]. Another possible application is the so-called SDS-PAGE (sodium dodecyl sulfate polyacrylamide gel electrophoresis) to analyze the entire profile of the sample and determine the distribution of the components like the uromodulin-linked capture of exosomes in urine samples [29].
Mass spectrometry has also been used for the analysis of EV composition regarding proteomics (cytosolic and surface proteins) [77], metabolomics, and lipidomics. As an alternative, chromatography (e.g., HPLC) or Raman spectroscopy, which generates specific protein or metabolite profiles based on laser refraction [80], can be used for lipidomics and metabolomics [16]. To improve the outcome and to find more tissue-specific exosomal surface markers, it is possible to use a combination of existing methods as performed by Wu et al. who developed a proximity-dependent barcoding assay (PBA) which combines antibody-specific binding, rolling circle amplification, PCR, and sequencing of the products [20].
RNA analyses
Other approaches include RNA analyses such as microarrays, real-time PCR (RT-PCR), or sequencing of exosomal long-noncoding RNAs (exo-lncRNAs), micro-RNAs (exo-miRs), circular RNAs (exo-circRNAs), and mRNAs (exo-mRNAs) [12, 85, 99]. According to the ISEV guidelines, proteinase or RNAse treatments should be done before the isolation of exosomal RNA (exo-RNA) [89, 94] to remove nucleic acids that are bound on the outside of EVs as well as protein complexes [30]. So far, it is still unknown whether RNAs such as ribosomal RNA (rRNA) or lncRNA fragments which are associated with EVs are artifacts of the isolation process or of biological relevance [30]. There are a few databases such as the exoRBase 2.0 database [50] or the exRNA Atlas resource [69] that summarize exosomal RNA profiles as they can differ depending on the physiological state of the cell [89]. So far, the ISEV guidelines give no specific recommendations for the use of nucleic acids as markers for EVs and see a need for more studies regarding this aspect [94].
Even though there is a significant effort to enable the characterization of exosomes, there is still no effective protocol. This is not only due to the heterogeneity of EVs but also to the varieties within the exosome subclasses [45]. Outcomes in this aspect also depend on the technique used for the preceding isolation of EVs. Nevertheless, methods for EV characterization are essential for understanding EV-related processes and identifying biomarkers in physiological as well as pathophysiological conditions.
Pathophysiology in different urological and nephrological organ systems
General oncological and immunological mechanisms
So far, there is a consensus on the assumption that exosomes are involved in the modulation of the immune system, the activation of the complement cascade, and also in pathophysiological processes such as multi-drug resistance (MDR) in cancer [42, 112]. Hereby, the functional heterogeneity of exosomes depends on the cell or tissue of origin, which in part determines the different sets of their contents [45, 112]. For example, natural killer cells (NK cells) release EVs that contain cell-destructive proteins (FasL, perforins) and tumor-suppressing non-coding RNA (miR-186). On the contrary, EVs secreted by antigen presenting cells (APCs), such as dendritic cells (DCs) and proinflammatory M1-macrophages, contain immunostimulatory proteins like MHC-I and MHC-II [42].
Tumor-derived exosomes, also known as oncosomes, have been extensively studied and promote tumor progression through activation of tumor-promoting pathways, the downregulation of tumor suppressor genes, next to other possibilities [41, 104]. For example, Chen et al. showed that melanoma-derived EVs exhibit programmed death ligand 1 (PD-L1) that interacts with PD-1 of T-cells [34] and inhibits anti-tumor immunity [14].
By transporting bioactive non-coding RNAs (ncRNAs) that induce an enhanced expression of ATP-binding box (ABC) transporters or by direct transport of ABC transporters, tumor-derived exosomes can also induce MDR [104, 112]. ABC transporters lead to an increased efflux of chemotherapeutics in chemosensitive cells and are potentially involved in the biogenesis and spontaneous as well as antigen-triggered release of MDR-inducing EVs [6, 46, 104].
Besides tumor cells, other cells of the tumor microenvironment (TME) are also supposed to be involved in tumor progression by secretion of exosomes. Zhang et al. described the secretion of EVs by M2-subtype macrophages which are also known as tumor-associated macrophages (TAMs) and promote metastasis of different tumor entities by intercellular transmission of tumorigenic agents [111].
Multiple studies have investigated exosomes not only as therapeutic target structures but also as cancer vaccines in immunotherapy due to their biocompatibility, as well as the high stability. Most studies investigating this topic have deployed exosomes derived from dendritic cells (DEXs) either as carriers for anti-tumor agents such as peptides or as effectors after stimulation of DCs with specific cancer biomarkers. There are still challenges in the manufacturing of exosomes, such as the lack of a cell-free platform for EV production and of sufficient quality controls [81]. Multiple reviews highlighted the use of EVs as drug delivery systems and summarized current findings and challenges in detail [38].
Benign nephrological and urological conditions
Because exosomes mediate a variety of functions, they have been investigated for multiple diseases and conditions using different isolation and characterization procedures. These include nephrological and urological conditions.
Early detection of kidney diseases is still a pressing issue today, as most common biomolecular markers for kidney disease have been described as insensitive and ineffective for an early diagnosis [44]. As methods for early diagnosis and therapy are challenging, end-stage renal disease (ESRD) is still associated with high morbidity and mortality. In recent years, exosomes have emerged as a new approach to solving this problem. Exo-mRNA offers a valuable alternative to RNA extracted from cells out of urine because of its high stability [24]. The close link between EVs and the development and progression of renal diseases could also offer a new therapeutic approach. Due to the size of EVs, it is assumed that urinary EVs originate from tissue that is part of the urinary tract such as the nephron or the urothelium of the bladder and ureter.
Diagnostic and prognostic value of exosomes in nephro- and urological conditions
As described above, exosomes play an important role in mediating the immune response and can also contribute to dysregulation of inflammatory reactions. Inflammation which can be triggered by many mechanisms (e.g., ischemia/reperfusion (I/R), proteinuria, toxins) is a central component of acute kidney injury (AKI). Lv et al. demonstrated that increased levels of miR-19b-3p, a negative regulator of a NFκB pathway suppressor, in exosomes derived from injured tubular epithelial cells (TECs) triggered the polarization of macrophages into the M1 proinflammatory phenotype. They investigated kidney and tubular exosomes derived from an AKI mouse model, which they isolated and identified using differential centrifugation as well as UC and a combination of TEM and NTA respectively, as well as exosomes of immortalized mouse TECs in in vitro experiments [62].
So far, the diagnosis of AKI comes along with an increase in serum creatinine or a decrease in urine output, but several exosomal biomarkers have already been identified in urine samples. These include exosomal fetuin-A [113], the transcription factor AFT3 and its mRNA, and a decrease in AQP-1 [5, 85, 86]. Sonoda et al. conducted a longitudinal study, where they identified a strong increase of the expression of miR-200 family members (e.g., miR-141-3p, miR-200a-3p) 3 days after AKI injury. They extracted urinary exosomes and RNAs of IR mouse models using the Urine exosome purification and RNA isolation (Norgen Biotek) as well as a total exosome isolation reagent from urine (Thermo Fisher) and the miRNeasy (Qiagen). Sonoda et al. performed RT (reverse transcription)-PCR for the identification of extracted exosomes. Most of the identified miRNAs regulated an effector of the TGF-β1 pathway which has a direct impact on renal fibrosis [85].
Fibrosis is another pathomechanism that commonly occurs in kidney diseases. Wang et al. studied the change of composition of secreted exosomes under physiological, inflammatory, and fibrotic conditions in primary human proximal TECs. Interestingly, miRNAs (e.g., miR-200a, miR-204), that showed the most alterations under pathophysiological conditions like renal fibrosis, were already known to be elements of renal disease such as progressive CKD. Similarly, many of the abnormally expressed exosomal proteins are involved in pathways of renal fibrosis (e.g., laminin, plectin, fibronectin) or counter-mechanisms such as complement factor C3 or HSPG2 (counteract hypoxia by increasing angiogenesis and vascular permeability) [101]. Other studies defined urinary exo-miR-21, miR-29c, and miR-200b as biomarkers for renal fibrosis [61, 106].
Besides biomolecular markers that indicate renal fibrosis or inflammation, exosomal contents have also been studied regarding their correlation with the progression of CKD. Ding et al. used cell culture and mouse models to show that treatment with isolated EVs of PKD1-cells and urinary EVs from autosomal dominant polycystic kidney disease (ADPKD) patients which were extracted using UC and filtration methods as well as characterized via WB analyses activated the proliferation and formation of cyst-like structures. The promotion of cyst growth was attributed to downregulated PKD1-mRNA and increased levels of miRNAs of the miR-200 family. Also, an increase in fibrosis and the recruitment of macrophages due to increased expression of several factors like fibronectin, collagen 1, or cytokines was observed. Upon treatment of cells and mice with an inhibitor of a key enzyme for EV biogenesis, a delay of the disease progression was seen (decreased cyst growth, renal fibrosis, and macrophage populations) [22].
Other studies utilized exo-mRNA as well as exosomal surface proteins to assess the success of kidney transplantations. El Fekhi et al. identified an exo-mRNA signature to discriminate biopsy samples from patients with any-cause rejection of kidney transplants and those with no rejection with an AUC of 0.93 showing a diagnostic improvement compared to the AUC of 0.57 of the current standards of care. They used a urine exosome isolation kit for exosome extraction [24]. Dimuccio et al. analyzed urinary exosomes of healthy volunteers and transplanted patients which they isolated using centrifugation and identified through cytofluorimetric and WB analyses as well as NTA. They observed that levels of the exosomal surface protein CD133, which is decreased in end-stage renal disease, were elevated upon successful kidney transplantation with decreased graft-versus-host-reactions [21].
Exosomes as treatment option in nephro- and urological conditions
Exosomal contents that promote kidney injury could be used as targets for delaying the progression of renal diseases or even prevent their formation [22, 44, 53]. Alternatively, protective EVs such as EVs derived from mesenchymal stem cells (MSCs), which are characterized by their regenerative potential could be used for therapy of nephrological diseases. Multiple preclinical studies have shown that EVs (including MSC-derived EVs) display protective effects in kidney injury by promoting regeneration of epithelia, angiogenesis, and reduction of inflammation and apoptosis [8]. Studies on renal fibrosis showed that human umbilical cord MSC-derived exosomes ameliorated the fibrotic phenotype in unilateral ureteral obstruction (UUO) rat models by inhibiting profibrotic pathways and proteins (YAP, p38MAP/ERK) [43, 58]. Even when EVs are derived from other sources, such as endothelial colony forming cells (ECFCs), these have also shown protective effects in ischemic kidney injury (e.g. by exo-miR-486-5p) [98].
The translation of these findings into clinical practice remains a challenge due to a lack of standardized protocols and the difficult distinction of EV-subtypes, their respective functions and characteristics [8].
Urooncology
Oncosomes, which are present in body fluids at high concentrations [114], have been described extensively as structures of prognostic and diagnostic value [14, 34]. They can be extracted and analyzed regarding their embedded nucleic acids and proteins by performing liquid biopsies which have previously been used as a less invasive tool for the detection of biomarkers like circulating tumor cells (CTCs) [56].
Typically, different diagnostic approaches compete, although the integration of different tests may improve diagnostic performances in cases where combined approaches complement each other. For instance, de la Calle et al. tested a prostate cancer screening algorithm based on liquid biopsies followed by multiparametric MRI (mpMRI) prior to tissue biopsy which aimed at reducing unnecessary biopsies, mpMRI, and over-detection of ISUP (International Society of Urological Pathology) 2014 grade group (GG) 1 prostate cancer (PC) [49].
Renal cell carcinoma
Renal cell carcinoma (RCC) makes up about 3% of all cancers worldwide [11]. Late detection in an advanced stage is associated with poor prognosis and stresses the need for identification of biomarkers for early diagnosis [82]. EVs and miRNAs, especially exo-miRs, are the subjects of current research that aims at implementing liquid biopsies as a noninvasive tool for diagnosis, analysis, and monitoring of cancer [74], including clear cell RCC (ccRCC).
Recently, some independent studies demonstrated that specific exo-miR might serve as promising agents for the diagnosis and monitoring of RCC [56, 100, 108] (NCT04053855). Wang et al. and Zhang et al. found that the expression of serum-derived circulating exo-miR-210 in patients with ccRCC was significantly elevated which was associated with metastasis and poor prognosis, and in turn levels significantly decreased within the first week after surgical tumor resection. They isolated exosomes using the Total Exosome Isolation Reagent (Invitrogen) and the Total Exosome Isolation Reagent (Invitrogen) in combination with the EpCAM isolation beads (Invitrogen) to isolate exosomes [100] and EpCAM (epithelial cell adhesion molecule) positive exosomes [108] respectively. Also, Li et al. are currently performing a pilot feasibility study (PEP-C-study, NCT04053855) for molecular detection of RCC via urinary exosomal carbo anhydrase 9 (CA9) [55]. A well-established commercial isolation kit for the isolation and characterization of exosomes is supposed to be used, as well as TEM, flow cytometry and RT-qPCR. If the pilot study is successful, a multicenter study will be conducted.
In addition to the potential significance of exosomes for early diagnosis of RCC, there is evidence that exosomes might help elucidate mechanisms of disease progression and metastasis.
When it comes to systemic therapy, immune checkpoint inhibition (ICI) with tyrosine kinase-inhibitors (TKI) as well as multikinase inhibitors (MKI) [59], such as sunitinib, primary or secondary drug resistance is an issue [31]. A recent study identified lncARSR as an inductor of sunitinib resistance in initially sensitive RCC cells. This was partly due to transport through EVs and could be reversed upon targeting lncARSR [52]. Several substances have already been investigated regarding their inhibiting effects on exosome biogenesis and secretion in drug resistant cancers. These include ketoconazole (KTZ) and tipifarnib (tipi) which have been tested in RCC cell lines or metastatic PC cell lines [18, 34, 35].
Other treatment options targeting EVs or EV-related processes have been suggested upon investigation of RCC progression, especially of metastasis. Zhang et al. found that exo-miR-21-5p derived from M2 macrophages has pro-metastatic effects (in vitro and in vivo) in RCC by activation of the PTEN/Akt pathway. Inhibition of miR-21-5p in M2 exosomes led to a reduction of the metastatic potential of RCC cells. Exosomes were isolated via centrifugation and UC and subsequently identified using TEM and Dynamic Light Scattering (DLS) [111]. In contrast, Li et al. observed that metastatic spread and tumor growth was decreased in an orthotopic mouse model of ccRCC after injection of MSC-derived exosomes which were extracted using UC and identified via NTA, WB analysis and TEM. These effects were attributed to the MSC-derived exo-miR-182 which appeared to promote the T-cell modulated immune response and led to a reduced expression of VEGF-A and overall tumor progression [54].
Urothelial carcinoma
The urothelial carcinoma of the bladder (BC) is the 10th most diagnosed cancer among adults [79]. The 5-year progression rate of initially non-muscle-invasive cancer (NMIBC) to muscle-invasive disease (MIBC) with poor prognosis ranges up to 45% [3].
Furthermore, cystoscopy is mandatory for diagnosis of BC. With a sensitivity of 84% for high-grade tumors, urinary cytology serves as an adjunct with insufficient sensitivity for low-grade tumors [105] and negative urinary cytology does not exclude tumor presence. Several potential urinary tests have been investigated [88], but none have been established in the routine clinical workflow which is why the search for molecular biomarkers is ongoing. For instance, Wen et al. performed a study to investigate the potential of exo-CA9 mRNA, which has previously been described as a diagnostic tool for BC [64]. Exosomes were extracted from urine samples of BC patients and controls using a combination of centrifugation, and a urinary exosome isolation solution by Hope Tech Biotechnology and TEM and flow cytometry was performed for the characterization of isolated exosomes. The average level of urinary exosomal CA9 was not only significantly increased in BC patients compared to controls, but Wen et al. also suggest that the detection of exo-CA9 mRNA might have a sufficient sensitivity and specificity to be used as a marker for the diagnosis of BC [102].
Until now, therapy of BC remains challenging due to its heterogeneous tumor biology. Radical cystectomy (RC) is the standard treatment for organ confined MIBC [91], but there is a recurrence rate of up to 40% after RC [87] which is possibly due to early micro-metastatic dissemination or “metabolic rewiring” after proceeding secretion of bladder derived exosomes. Hiltbrunner et al. detected overexpression of urinary exosomal proteins and tumor metabolism-related oncogenes in BC patients’ urinary samples, despite complete histopathological downstaging due to RC and neoadjuvant chemotherapy (NAC). They used UC to isolate exosomes from urine which they subsequently characterized via flow cytometry, NTA, EM, and mass spectrometry. Contrary to ureteral derived exosomes (without prior tumor contact) which did not exhibit an overexpression of these components, urinary exosomes derived from the bladder revealed a potentially malignant “memory” phenotype, despite histopathological downstaging and could promote cancer dissemination [39].
Prostate cancer
PC is the 2nd most frequently diagnosed cancer in men worldwide and is the most frequently diagnosed cancer overall in more than 50% of countries [93]. Differentiating between clinical indolent PC and aggressive organ confined PC is crucial. Due to the risk of overdiagnosis and overtreatment on the one hand and lack of clinically significant localized PC eligible for curative treatment on the other hand, the use of prostate specific antigen (PSA) as a marker for early detection must be chosen wisely and recommendations differ between guidelines [2, 40, 51]. Therefore, several new tests have been introduced to extend diagnostic options.
Currently, only one exosome-based diagnostic test for PC detection has been commercialized. The ExoDx Prostate (IntelliScore) (EPI) test discriminates indolent (benign and GG1) from clinically significant PC (≥ GG2) of men with more than 50 years of age with PSA values in a gray-zone of 2–10 ng/ml presenting for biopsy [68]. The test is based on qPCR signals of the three mRNAs of PCA3, ERG, and SPDEF derived from urinary exosomes. Its clinical performance has been validated in three independent, multi-site, prospective clinical validation studies in the US [63, 67, 68, 95]. Furthermore, a European study utilized the EPI-CE test, a CE-marked in vitro diagnostic (IVD) version specifically developed for the use in European laboratories [48]. Two other exosome-based tests are currently under clinical validation (NCT04100811, NCT03957252). The Sentinel test platform consisting of three different tests (Sentinel PCa, Sentinel CS and Sentinel HG) quantifies the expression levels of hundreds of urinary exo-sncRNAs using Affymetrix GeneChips [99]. The ClarityDx test is based on the prostate-specific membrane antigen, polysialic acid, and ghrelin-growth hormone receptor as detected by microflow cytometry data of plasma-derived exosomes [27].
Despite these multi-center validation trials, various smaller studies have been conducted to find potential biomarkers for PC. Recently, miRNAs isolated from urinary EVs by differential centrifugation have been examined in a cohort of 70 patients for their potential to distinguish between ISUP GG1, GG2, and GG3 patients to allow for active surveillance of PC patients as an alternative to radical treatment. While the NGS analysis of miRNA data showed promising classification performances, results could not be reproduced when NGS quantification was replaced by qPCR quantification [76]. A study by Logozzi et al. showed that PSA-expressing exosomes in plasma, that were extracted via centrifugation, were increased in PC patients compared to BPH and control groups by performing NTA, WB, ELISA and flow cytometry [60]. Almeida et al. extracted urinary EVs of PC patients using centrifugation, which they identified with NTA, EM, WB and RNA analyses. The comparison of total RNA-sequencing of FFPE PC tumor tissue and their paired urinary EV measurements highlighted strong differences in circRNA signals between tumor and EVs and suggests an important role of circRNAs in PC detection and treatment [4].
Treatment of advanced PC is another challenging aspect, especially when it comes to castration resistant PC (CRPC), where initially effective androgen deprivation therapy (ADT) becomes insufficient for disease control [13]. Metastatic CRPC (mCRPC) is associated with poor survival and treatment options are limited, although new agents are continuously emerging on the market. Exosomes derived from PC cells and other cells of the TME can promote metastatic spread [1]. Recently, Guo et al. demonstrated that the upregulated lncRNA LINC01213 in exosomes derived from androgen-independent PC (AIPC) cells via UC could induce androgen independency by uptake of former dependent PC cells (ADPC) through activation of the Wnt/β-catenin signaling pathway [36]. These findings introduce exosome contents as well as exosome biogenesis and secretion as new structures of interest for treatment of mCRPC. For instance, Datta et al. have identified Manumycin-A (MA) as a suppressor of exosomal biogenesis in CRPC cells [17]. Del Re et al. demonstrated the detection of androgen receptor splice variant 7 (AR-V7) in plasma-derived exosome RNA as a reliable predictor for hormonal therapy in mCRPC patients. Overall, they view exosome-derived RNA as a more promising diagnostic agent than more invasive and complex procedures such as the extraction of CTCs [19].
Concluding remarks
Exosome-based research has seen a large increase in interest over the last few years. This is not only due to the previously underestimated importance of exosomes in biological and pathological functions but also attributed to the therapeutic and diagnostic possibilities that arise from the exosomal structure and stability. So far, exosome-based research is still mostly limited to preclinical experiments and early-stage clinical trials, because the translation of experimental findings remains difficult. Even though there are attempts at implementing a standardized protocol for the isolation, purification, and characterization of exosomes by the ISEV, the commonly used methods display disadvantages that make it challenging to utilize them in clinical practice. Although the application of exosomes in diagnostics and clinical therapy is still in its early stages, their importance in physiological and pathophysiological processes appears to be significant and has been observed in a variety of studies.
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed in this review.
References
Akoto T, Saini S (2021) Role of exosomes in prostate cancer metastasis. Int J Mol Sci 22(7):3528. https://doi.org/10.3390/ijms22073528
Albertsen PC (2020) Prostate cancer screening and treatment: where have we come from and where are we going? BJU Int 126:218–224. https://doi.org/10.1111/bju.15153
Ali-El-Dein B, Al-Marhoon MS, Abdel-Latif M et al (2011) Survival after primary and deferred cystectomy for stage T1 transitional cell carcinoma of the bladder. Urol Ann 3:127–132. https://doi.org/10.4103/0974-7796.84960
Almeida A, Gabriel M, Firlej V et al (2022) Urinary extracellular vesicles contain mature transcriptome enriched in circular and long noncoding RNAs with functional significance in prostate cancer. J Extracell Vesicles 11:e12210. https://doi.org/10.1002/jev2.12210
Asvapromtada S, Sonoda H, Kinouchi M et al (2018) Characterization of urinary exosomal release of aquaporin-1 and -2 after renal ischemia-reperfusion in rats. Am J Physiol Renal Physiol 314:F584–F601. https://doi.org/10.1152/ajprenal.00184.2017
Aung T, Chapuy B, Vogel D et al (2011) Exosomal evasion of humoral immunotherapy in aggressive B-cell lymphoma modulated by ATP-binding cassette transporter A3. Proc Natl Acad Sci U S A 108:15336–15341. https://doi.org/10.1073/pnas.1102855108
Baietti MF, Zhang Z, Mortier E et al (2012) Syndecan-syntenin-ALIX regulates the biogenesis of exosomes. Nat Cell Biol 14:677–685. https://doi.org/10.1038/ncb2502
Birtwistle L, Chen X-M, Pollock C (2021) Mesenchymal stem cell-derived extracellular vesicles to the rescue of renal injury. Int J Mol Sci 22(12):6596. https://doi.org/10.3390/ijms22126596
Brennan K, Martin K, FitzGerald SP et al (2020) A comparison of methods for the isolation and separation of extracellular vesicles from protein and lipid particles in human serum. Sci Rep 10:1039. https://doi.org/10.1038/s41598-020-57497-7
Busato A, Bonafede R, Bontempi P et al (2016) Magnetic resonance imaging of ultrasmall superparamagnetic iron oxide-labeled exosomes from stem cells: a new method to obtain labeled exosomes. Int J Nanomedicine 11:2481–2490. https://doi.org/10.2147/ijn.s104152
Capitanio U, Bensalah K, Bex A et al (2019) Epidemiology of renal cell carcinoma. Eur Urol 75:74–84. https://doi.org/10.1016/j.eururo.2018.08.036
Chakrabortty SK, Khodor YL, Kitchen RR et al (2022) Exosome based analysis for space associated neuro-ocular syndrome and health risks in space exploration. NPJ Microgravity 8:40. https://doi.org/10.1038/s41526-022-00225-4
Chandrasekar T, Yang JC, Gao AC et al (2015) Mechanisms of resistance in castration-resistant prostate cancer (CRPC). Transl Androl Urol 4:365–380. https://doi.org/10.3978/j.issn.2223-4683.2015.05.02
Chen G, Huang AC, Zhang W et al (2018) Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 560:382–386. https://doi.org/10.1038/s41586-018-0392-8
Chen Y, Zhu Q, Cheng L et al (2021) Exosome detection via the ultrafast-isolation system: EXODUS. Nat Methods 18:212–218. https://doi.org/10.1038/s41592-020-01034-x
Coughlan C, Bruce KD, Burgy O et al (2020) Exosome isolation by ultracentrifugation and precipitation and techniques for downstream analyses. Curr Protoc Cell Biol 88:e110. https://doi.org/10.1002/cpcb.110
Datta A, Kim H, Lal M et al (2017) Manumycin A suppresses exosome biogenesis and secretion via targeted inhibition of Ras/Raf/ERK1/2 signaling and hnRNP H1 in castration-resistant prostate cancer cells. Cancer Lett 408:73–81. https://doi.org/10.1016/j.canlet.2017.08.020
Datta A, Kim H, McGee L et al (2018) High-throughput screening identified selective inhibitors of exosome biogenesis and secretion: a drug repurposing strategy for advanced cancer. Sci Rep 8:8161. https://doi.org/10.1038/s41598-018-26411-7
Del Re M, Biasco E, Crucitta S et al (2017) The detection of androgen receptor splice variant 7 in plasma-derived exosomal RNA strongly predicts resistance to hormonal therapy in metastatic prostate cancer patients. Eur Urol 71:680–687. https://doi.org/10.1016/j.eururo.2016.08.012
Di Wu, Yan J, Shen X et al (2019) Profiling surface proteins on individual exosomes using a proximity barcoding assay. Nat Commun 10:3854. https://doi.org/10.1038/s41467-019-11486-1
Dimuccio V, Ranghino A, PraticòBarbato L et al (2014) Urinary CD133+ extracellular vesicles are decreased in kidney transplanted patients with slow graft function and vascular damage. PLoS ONE 9:e104490. https://doi.org/10.1371/journal.pone.0104490
Ding H, Li LX, Harris PC et al (2021) Extracellular vesicles and exosomes generated from cystic renal epithelial cells promote cyst growth in autosomal dominant polycystic kidney disease. Nat Commun 12:4548. https://doi.org/10.1038/s41467-021-24799-x
Drula R, Ott LF, Berindan-Neagoe I et al (2020) MicroRNAs from liquid biopsy derived extracellular vesicles: recent advances in detection and characterization methods. Cancers (Basel) 12(8):2009. https://doi.org/10.3390/cancers12082009
El Fekih R, Hurley J, Tadigotla V et al (2021) Discovery and validation of a urinary exosome mRNA signature for the diagnosis of human kidney transplant rejection. JJ Am Soc Nephrol 32(4):994–1004. https://doi.org/10.1681/asn.2020060850
Erdbrügger U, Blijdorp CJ, Bijnsdorp IV et al (2021) Urinary extracellular vesicles: a position paper by the Urine Task Force of the International Society for Extracellular Vesicles. J Extracell Vesicles 10:e12093. https://doi.org/10.1002/jev2.12093
Erozenci LA, Böttger F, Bijnsdorp IV et al (2019) Urinary exosomal proteins as (pan-)cancer biomarkers: insights from the proteome. FEBS Lett 593:1580–1597. https://doi.org/10.1002/1873-3468.13487
Fairey AS, Paproski RJ, Pink D et al (2020) Clinical analysis of the extracellular vesicle-fingerprint score blood test to refine the prediction of clinically significant prostate cancer and avoid prostate biopsy. JCO 38:5530. https://doi.org/10.1200/JCO.2020.38.15_suppl.5530
Ferguson S, Weissleder R (2020) Modeling EV kinetics for use in early cancer detection. Adv Biosyst 4:e1900305. https://doi.org/10.1002/adbi.201900305
Fernández-Llama P, Khositseth S, Gonzales PA et al (2010) Tamm-Horsfall protein and urinary exosome isolation. Kidney Int 77:736–742. https://doi.org/10.1038/ki.2009.550
Fischer S, Deindl E (2021) Characterization of RNA in extracellular vesicles. Appl Sci 11:7520. https://doi.org/10.3390/app11167520
Fisher R, Gore M, Larkin J (2013) Current and future systemic treatments for renal cell carcinoma. Semin Cancer Biol 23:38–45. https://doi.org/10.1016/j.semcancer.2012.06.004
Gardiner C, Di Vizio D, Sahoo S et al (2016) Techniques used for the isolation and characterization of extracellular vesicles: results of a worldwide survey. J Extracell Vesicles 5:32945. https://doi.org/10.3402/jev.v5.32945
Gibbings DJ, Ciaudo C, Erhardt M et al (2009) Multivesicular bodies associate with components of miRNA effector complexes and modulate miRNA activity. Nat Cell Biol 11:1143–1149. https://doi.org/10.1038/ncb1929
Greenberg JW, Kim H, Ahn M et al (2022) Combination of tipifarnib and sunitinib overcomes renal cell carcinoma resistance to tyrosine kinase inhibitors via tumor-derived exosome and T cell modulation. Cancers (Basel) 14(4):903. https://doi.org/10.3390/cancers14040903
Greenberg JW, Kim H, Moustafa AA et al (2021) Repurposing ketoconazole as an exosome directed adjunct to sunitinib in treating renal cell carcinoma. Sci Rep 11:10200. https://doi.org/10.1038/s41598-021-89655-w
Guo Z, Lu X, Yang F et al (2022) Exosomal LINC01213 plays a role in the transition of androgen-dependent prostate cancer cells into androgen-independent manners. J Oncol 2022:8058770. https://doi.org/10.1155/2022/8058770
Gurung S, Perocheau D, Touramanidou L et al (2021) The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal 19:47. https://doi.org/10.1186/s12964-021-00730-1
Hernandez-Oller L, Seras-Franzoso J, Andrade F et al (2020) Extracellular Vesicles as Drug Delivery Systems in Cancer. Pharmaceutics 12(12):1146. https://doi.org/10.3390/pharmaceutics12121146
Hiltbrunner S, Mints M, Eldh M et al (2020) Urinary exosomes from bladder cancer patients show a residual cancer phenotype despite complete pathological downstaging. Sci Rep 10:5960. https://doi.org/10.1038/s41598-020-62753-x
Hoffman RM (2022) Striking the right balance with prostate cancer screening. JAMA Netw Open 5:e222174. https://doi.org/10.1001/jamanetworkopen.2022.2174
Hoshino A, Costa-Silva B, Shen T-L et al (2015) Tumour exosome integrins determine organotropic metastasis. Nature 527:329–335. https://doi.org/10.1038/nature15756
Hussain MWA, Jahangir S, Ghosh B et al (2022) Exosomes for regulation of immune responses and immunotherapy. JNT 3:55–85. https://doi.org/10.3390/jnt3010005
Ji C, Zhang J, Zhu Y et al (2020) Exosomes derived from hucMSC attenuate renal fibrosis through CK1δ/β-TRCP-mediated YAP degradation. Cell Death Dis 11:327. https://doi.org/10.1038/s41419-020-2510-4
Jin C, Wu P, Li L et al (2021) Exosomes: emerging therapy delivery tools and biomarkers for kidney diseases. Stem Cells Int 2021:7844455. https://doi.org/10.1155/2021/7844455
Kalluri R, LeBleu VS (2020) The biology, function, and biomedical applications of exosomes. Science 367(6478):eaau6977. https://doi.org/10.1126/science.aau6977
Koch R, Aung T, Vogel D et al (2016) Nuclear trapping through inhibition of exosomal export by indomethacin increases cytostatic efficacy of doxorubicin and pixantrone. Clin Cancer Res 22:395–404. https://doi.org/10.1158/1078-0432.ccr-15-0577
Kowal J, Arras G, Colombo M et al (2016) Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc Natl Acad Sci U S A 113:E968–E977. https://doi.org/10.1073/pnas.1521230113
Kretschmer A, Kajau H, Margolis E et al (2022) Validation of a CE-IVD, urine exosomal RNA expression assay for risk assessment of prostate cancer prior to biopsy. Sci Rep 12:4777. https://doi.org/10.1038/s41598-022-08608-z
de La Calle CM, Fasulo V, Cowan JE et al (2021) Clinical utility of 4Kscore®, ExosomeDx™ and magnetic resonance imaging for the early detection of high grade prostate cancer. J Urol 205:452–460. https://doi.org/10.1097/ju.0000000000001361
Lai H, Li Y, Zhang H et al (2021) exoRBase 2.0: an atlas of mRNA, lncRNA and circRNA in extracellular vesicles from human biofluids. Nucleic Acids Res 50:D118–D128. https://doi.org/10.1093/nar/gkab1085
Lakes J, Arsov C (2019) PSA-screening und molekulare marker (PSA screening and molecular markers). Urologe A 58:486–493. https://doi.org/10.1007/s00120-019-0900-y
Le Qu, Ding J, Chen C et al (2016) Exosome-transmitted lncARSR promotes sunitinib resistance in renal cancer by acting as a competing endogenous RNA. Cancer Cell 29:653–668. https://doi.org/10.1016/j.ccell.2016.03.004
Lee SA, Yoo TH (2022) Therapeutic application of extracellular vesicles for various kidney diseases: a brief review. BMB Rep 55:3–10. https://doi.org/10.5483/BMBRep.2022.55.1.141
Li D, Lin F, Li G et al (2021) Exosomes derived from mesenchymal stem cells curbs the progression of clear cell renal cell carcinoma through T-cell immune response. Cytotechnology 73:593–604. https://doi.org/10.1007/s10616-021-00480-5
Li G, Cuilleron M, Cottier M et al (2006) The use of MN/CA9 gene expression in identifying malignant solid renal tumors. Eur Urol 49:401–405. https://doi.org/10.1016/j.eururo.2005.10.025
Li G, Mallouk N, Flandrin P et al (2021) Presence of urinary exosomes for liquid biopsy of clear cell renal cell carcinoma: orotocol for a pilot feasibility study. JMIR Res Protoc 10:e24423. https://doi.org/10.2196/24423
Li K, Wong DK, Hong KY et al (2018) Cushioned-density gradient ultracentrifugation (C-DGUC): a refined and high performance method for the isolation, characterization, and use of exosomes. Methods Mol Biol 1740:69–83. https://doi.org/10.1007/978-1-4939-7652-2_7
Liu B, Hu D, Zhou Y et al (2020) Exosomes released by human umbilical cord mesenchymal stem cells protect against renal interstitial fibrosis through ROS-mediated P38MAPK/ERK signaling pathway. Am J Transl Res 12:4998–5014
Ljungberg B, Albiges L, Abu-Ghanem Y et al (2022) European Association of Urology Guidelines on Renal Cell Carcinoma: the 2022 update. Eur Urol 82(4):399–410. https://doi.org/10.1016/j.eururo.2022.03.006
Logozzi M, Angelini DF, Giuliani A et al (2019) Increased plasmatic levels of PSA-expressing exosomes distinguish prostate cancer patients from benign prostatic hyperplasia: a prospective study. Cancers (Basel) 11(10):1449. https://doi.org/10.3390/cancers11101449
Lv C-Y, Ding W-J, Wang Y-L et al (2018) A PEG-based method for the isolation of urinary exosomes and its application in renal fibrosis diagnostics using cargo miR-29c and miR-21 analysis. Int Urol Nephrol 50:973–982. https://doi.org/10.1007/s11255-017-1779-4
Lv L-L, Feng Y, Wu M et al (2020) Exosomal miRNA-19b-3p of tubular epithelial cells promotes M1 macrophage activation in kidney injury. Cell Death Differ 27:210–226. https://doi.org/10.1038/s41418-019-0349-y
Margolis E, Brown G, Partin A et al (2022) Predicting high-grade prostate cancer at initial biopsy: clinical performance of the ExoDx (EPI) Prostate Intelliscore test in three independent prospective studies. Prostate Cancer Prostatic Dis 25:296–301. https://doi.org/10.1038/s41391-021-00456-8
de Martino M, Lucca I, Mbeutcha A et al (2015) Carbonic anhydrase IX as a diagnostic urinary marker for urothelial bladder cancer. Eur Urol 68:552–554. https://doi.org/10.1016/j.eururo.2015.06.015
Mathieu M, Martin-Jaular L, Lavieu G et al (2019) Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol 21:9–17. https://doi.org/10.1038/s41556-018-0250-9
Mathieu M, Névo N, Jouve M et al (2021) Specificities of exosome versus small ectosome secretion revealed by live intracellular tracking of CD63 and CD9. Nat Commun 12:4389. https://doi.org/10.1038/s41467-021-24384-2
McKiernan J, Donovan MJ, Margolis E et al (2018) A prospective adaptive utility trial to validate performance of a novel urine exosome gene expression assay to predict high-grade prostate cancer in patients with prostate-specific antigen 2–10 ng/ml at initial biopsy. Eur Urol 74:731–738. https://doi.org/10.1016/j.eururo.2018.08.019
McKiernan J, Donovan MJ, O’Neill V et al (2016) A novel urine exosome gene expression assay to predict high-grade prostate cancer at initial biopsy. JAMA Oncol 2:882–889. https://doi.org/10.1001/jamaoncol.2016.0097
Murillo OD, Thistlethwaite W, Rozowsky J et al (2019) exRNA Atlas analysis reveals distinct extracellular RNA cargo types and their carriers present across human biofluids. Cell 177:463-477.e15. https://doi.org/10.1016/j.cell.2019.02.018
Mussack V, Wittmann G, Pfaffl MW (2019) Comparing small urinary extracellular vesicle purification methods with a view to RNA sequencing-Enabling robust and non-invasive biomarker research. Biomol Detect Quantif 17:100089. https://doi.org/10.1016/j.bdq.2019.100089
Nederveen JP, Warnier G, Di Carlo A et al (2020) Extracellular vesicles and exosomes: insights from exercise science. Front Physiol 11:604274. https://doi.org/10.3389/fphys.2020.604274
O’Brien K, Breyne K, Ughetto S et al (2020) RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat Rev Mol Cell Biol 21:585–606. https://doi.org/10.1038/s41580-020-0251-y
Park J, Park JS, Huang C-H et al (2021) An integrated magneto-electrochemical device for the rapid profiling of tumour extracellular vesicles from blood plasma. Nat Biomed Eng 5:678–689. https://doi.org/10.1038/s41551-021-00752-7
Poulet G, Massias J, Taly V (2019) Liquid biopsy: general concepts. Acta Cytol 63:449–455. https://doi.org/10.1159/000499337
Raiborg C, Stenmark H (2009) The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature 458:445–452. https://doi.org/10.1038/nature07961
Ramirez-Garrastacho M, Berge V, Linē A et al (2022) Potential of miRNAs in urinary extracellular vesicles for management of active surveillance in prostate cancer patients. Br J Cancer 126:492–501. https://doi.org/10.1038/s41416-021-01598-1
Rosa-Fernandes L, Rocha VB, Carregari VC et al (2017) A perspective on extracellular Vesicles Proteomics. Front Chem 5:102. https://doi.org/10.3389/fchem.2017.00102
Royo F, Théry C, Falcón-Pérez JM et al (2020) Methods for separation and characterization of extracellular vesicles: results of a worldwide survey performed by the ISEV rigor and standardization subcommittee. Cells 9(9):1955. https://doi.org/10.3390/cells9091955
Saginala K, Barsouk A, Aluru JS et al (2020) Epidemiology of bladder cancer. Med Sci (Basel) 8(1):15. https://doi.org/10.3390/medsci8010015
Samoylenko A, Kögler M, Zhyvolozhnyi A et al (2021) Time-gated Raman spectroscopy and proteomics analyses of hypoxic and normoxic renal carcinoma extracellular vesicles. Sci Rep 11:19594. https://doi.org/10.1038/s41598-021-99004-6
Santos P, Almeida F (2021) Exosome-based vaccines: history, current state, and clinical trials. Front Immunol 12:711565. https://doi.org/10.3389/fimmu.2021.711565
Sequeira JP, Constâncio V, Lobo J et al (2021) Unveiling the world of circulating and exosomal microRNAs in renal cell carcinoma. Cancers (Basel) 13(21):5252. https://doi.org/10.3390/cancers13215252
Sidhom K, Obi PO, Saleem A (2020) A review of exosomal isolation methods: is size exclusion chromatography the best option? Int J Mol Sci 21(18):6466. https://doi.org/10.3390/ijms21186466
Skog J, Würdinger T, van Rijn S et al (2008) Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat Cell Biol 10:1470–1476. https://doi.org/10.1038/ncb1800
Sonoda H, Lee BR, Park K-H et al (2019) miRNA profiling of urinary exosomes to assess the progression of acute kidney injury. Sci Rep 9:4692. https://doi.org/10.1038/s41598-019-40747-8
Sonoda H, Yokota-Ikeda N, Oshikawa S et al (2009) Decreased abundance of urinary exosomal aquaporin-1 in renal ischemia-reperfusion injury. Am J Physiol Renal Physiol 297:F1006–F1016. https://doi.org/10.1152/ajprenal.00200.2009
Sonpavde G, Khan MM, Svatek RS et al (2011) Prognostic risk stratification of pathological stage T2N0 bladder cancer after radical cystectomy. BJU Int 108:687–692. https://doi.org/10.1111/j.1464-410x.2010.09902.x
Soria F, Droller MJ, Lotan Y et al (2018) An up-to-date catalog of available urinary biomarkers for the surveillance of non-muscle invasive bladder cancer. World J Urol 36:1981–1995. https://doi.org/10.1007/s00345-018-2380-x
Sousa KP de, Rossi I, Abdullahi M et al (2022) Isolation and characterization of extracellular vesicles and future directions in diagnosis and therapy. Wiley Interdiscip Rev Nanomed Nanobiotechnol e1835. https://doi.org/10.1002/wnan.1835
Srinivasan S, Yeri A, Cheah PS et al (2019) Small RNA Sequencing across diverse biofluids identifies optimal methods for exRNA isolation. Cell 177:446-462.e16. https://doi.org/10.1016/j.cell.2019.03.024
Stein JP, Lieskovsky G, Cote R et al (2001) Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. JCO 19:666–675. https://doi.org/10.1200/JCO.2001.19.3.666
Stuffers S, Sem Wegner C, Stenmark H et al (2009) Multivesicular endosome biogenesis in the absence of ESCRTs. Traffic 10:925–937. https://doi.org/10.1111/j.1600-0854.2009.00920.x
Sung H, Ferlay J, Siegel RL et al (2021) Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Théry C, Witwer KW, Aikawa E et al (2018) Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles 7:1535750. https://doi.org/10.1080/20013078.2018.1535750
Tutrone R, Donovan MJ, Torkler P et al (2020) Clinical utility of the exosome based ExoDx Prostate(IntelliScore) EPI test in men presenting for initial Biopsy with a PSA 2–10 ng/mL. Prostate Cancer Prostatic Dis 23:607–614. https://doi.org/10.1038/s41391-020-0237-z
Valadi H, Ekström K, Bossios A et al (2007) Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 9:654–659. https://doi.org/10.1038/ncb1596
Veerman RE, Teeuwen L, Czarnewski P et al (2021) Molecular evaluation of five different isolation methods for extracellular vesicles reveals different clinical applicability and subcellular origin. J Extracell Vesicles 10(9):e12128. https://doi.org/10.1002/jev2.12128
Viñas JL, Spence M, Gutsol A et al (2018) Receptor-ligand interaction mediates targeting of endothelial colony forming cell-derived exosomes to the kidney after ischemic injury. Sci Rep 8:16320. https://doi.org/10.1038/s41598-018-34557-7
Wang W-LW, Sorokin I, Aleksic I et al (2020) Expression of small noncoding RNAs in urinary exosomes classifies prostate cancer into indolent and aggressive disease. J Urol 204:466–475. https://doi.org/10.1097/ju.0000000000001020
Wang X, Wang T, Chen C et al (2018) Serum exosomal miR-210 as a potential biomarker for clear cell renal cell carcinoma. J Cell Biochem. https://doi.org/10.1002/jcb.27347
Wang X, Wilkinson R, Kildey K et al (2017) Unique molecular profile of exosomes derived from primary human proximal tubular epithelial cells under diseased conditions. J Extracell Vesicles 6:1314073. https://doi.org/10.1080/20013078.2017.1314073
Wen J, Yang T, Mallouk N et al (2021) Urinary exosomal CA9 mRNA as a novel liquid biopsy for molecular diagnosis of bladder cancer. Int J Nanomedicine 16:4805–4811. https://doi.org/10.2147/ijn.s312322
Xie S, Zhang Q, Jiang L (2022) Current knowledge on exosome biogenesis, cargo-sorting mechanism and therapeutic implications. Membranes (Basel) 12(5). https://doi.org/10.3390/membranes12050498
Xue D, Han J, Liang Z et al (2022) Current perspectives on the unique roles of exosomes in drug resistance of hepatocellular carcinoma. J Hepatocell Carcinoma 9:99–112. https://doi.org/10.2147/JHC.S351038
Yafi FA, Brimo F, Steinberg J et al (2015) Prospective analysis of sensitivity and specificity of urinary cytology and other urinary biomarkers for bladder cancer. Urol Oncol 33:66.e25–31. https://doi.org/10.1016/j.urolonc.2014.06.008
Yu Y, Bai F, Qin N et al (2018) Non-proximal renal tubule-derived urinary exosomal miR-200b as a biomarker of renal fibrosis. Nephron 139:269–282. https://doi.org/10.1159/000487104
Zhang H, Lyden D (2019) Asymmetric-flow field-flow fractionation technology for exomere and small extracellular vesicle separation and characterization. Nat Protoc 14:1027–1053. https://doi.org/10.1038/s41596-019-0126-x
Zhang W, Ni M, Su Y et al (2018) MicroRNAs in serum exosomes as potential biomarkers in clear-cell renal cell carcinoma. Eur Urol Focus 4:412–419. https://doi.org/10.1016/j.euf.2016.09.007
Zhang Y, Bi J, Huang J et al (2020) Exosome: a review of its classification, isolation techniques, storage, diagnostic and targeted therapy applications. Int J Nanomedicine 15:6917–6934. https://doi.org/10.2147/ijn.s264498
Zhang Y, Liu Y, Liu H et al (2019) Exosomes: biogenesis, biologic function and clinical potential. Cell Biosci 9:19. https://doi.org/10.1186/s13578-019-0282-2
Zhang Z, Hu J, Ishihara M et al. (2022) The miRNA-21-5p payload in exosomes from M2 macrophages drives tumor cell aggression via PTEN/Akt signaling in renal cell carcinoma. Int J Mol Sci 23(6):3005. https://doi.org/10.3390/ijms23063005
Zhong Y, Li H, Li P et al (2021) Exosomes: a new pathway for cancer drug resistance. Front Oncol 11:743556. https://doi.org/10.3389/fonc.2021.743556
Zhou H, Pisitkun T, Aponte A et al (2006) Exosomal fetuin—a identified by proteomics: a novel urinary biomarker for detecting acute kidney injury. Kidney Int 70:1847–1857. https://doi.org/10.1038/sj.ki.5001874
Zhou X, Xie F, Wang L et al (2020) The function and clinical application of extracellular vesicles in innate immune regulation. Cell Mol Immunol 17:323–334. https://doi.org/10.1038/s41423-020-0391-1
Acknowledgements
The authors thank Lucia Denk for her help with figure illustration and Eric Pion for proof reading.
Author information
Authors and Affiliations
Contributions
Idea: Phillipp Torkler, Silke Haerteis, Thiha Aung; literature search and data analysis: Lina Mahl, Johanna Ollig, Verena Schweihofer, Lara Wiegand, Phillipp Torkler; draft: Lina Mahl, Johanna Ollig, Verena Schweihofer, Lara Wiegand, Phillipp Torkler; Revision: Lina Mahl, Johanna Ollig, Verena Schweihofer, Lara Wiegand, Phillipp Torkler, Silke Haerteis, Thiha Aung.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent for publication
Not applicable.
Conflict of interests
PT was an employee of Exosome Diagnostics from 2016 to 2020. The authors state no other conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mahl, L., Ollig, J., Schweihofer, V. et al. Importance and implications of exosomes in nephrology and urology. Pflugers Arch - Eur J Physiol 475, 153–166 (2023). https://doi.org/10.1007/s00424-022-02771-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-022-02771-y