Abstract
Dirofilariosis is a vector-borne disease mainly caused by Dirofilaria immitis and Dirofilaria repens. In contrast to the known endemicity of dirofilariosis in southern and south-eastern Europe, information on the distribution of D. repens in Central-Europe is fragmentary. We tested 8877 serum samples from dogs from Austria, Denmark, Germany, Italy, Lithuania, Poland, Switzerland and the UK using an ELISA detecting filarial-specific antibodies, hypothesising higher occurrence of D. repens. Based on two overlapping frequency distributions, presumed negative samples had a mean optical density (OD) value of 0.097, representing 97.45% of all samples. Presumed positive samples, representing 2.55% of all sera, had a mean OD value of 0.287. Test prevalence based on the calculated cut-off was 3.51% for all sera (4.36% for Austria, 1.94% for Denmark, 1.39% for Germany, 3.37% for Italy, 6.90% for Lithuania, 6.99% for Poland, 0.77% for Switzerland and 0.0% for the UK, respectively). The bimodal distribution, representing overlapping distributions of OD values from positive and negative dogs, enabled the assignment of a probability of true infection status to each dog. Mean probabilities of true infection status across groups, based on the postal codes of origin, allowed us to estimate and map true prevalences. For all countries, except the UK, the true prevalence was lower than the test prevalence. The large number of serum samples and the use of a non-gold standard analytical method allowed us to create a more realistic picture of the distribution of D. repens in Central Europe and the UK.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In Europe, dogs and wild carnivores are hosts of a variety of filarial species. Canine filariae belong to the family of the Onchocercidae, whereof the clinically relevant representatives are Dirofilaria immitis and D. repens, whilst Acanthocheilonema reconditum, Dipetalonema (A.) dracunculoides and Cercopithifilaria spp. are mostly apathogenic (Ionică et al. 2015; Sonnberger et al. 2021). Mosquito species of the genera Culex, Aedes and Anopheles serve as vectors and intermediate hosts, transmitting infectious Dirofilaria third-stage larvae to a susceptible host (Cancrini et al. 2007; Morchon et al. 2012; Silaghi et al. 2017). Dirofilaria immitis causes ‘heartworm disease’ in dogs and occasionally cats and ferrets; D. repens is at the origin of subcutaneous/ocular ‘skinworm’ disease (Genchi et al. 2011). Both Dirofilaria spp. can cause subcutaneous, ocular and pulmonary dirofilariosis in humans which act as dead-end hosts (Pampiglione and Rivasi 2000; Simon et al. 2012).
Dirofilaria immitis and D. repens predominantly occur in southern and south-eastern European areas, respectively. For D. immitis, a trend to spread in north-eastern Europe has been observed in recent decades (Genchi et al. 2011). The northern border of transmission of D. immitis is not well documented to date, but cases have been identified as north as Central France, Southern Switzerland, Northern Italy, Slovakia, Bulgaria, Hungary and Ukraine (Deplazes et al. 1995; Farkas et al. 2020; Hermosilla et al. 2006; Laidoudi et al. 2019; Mendoza-Roldan et al. 2020; Panayotova-Pencheva et al. 2020; Petruschke et al. 2001; Sassnau et al. 2014a; Széll et al. 2020).
The endemic areas of D. repens mostly overlap with those of D. immitis but also extend significantly northwards. In particular, in the north-eastern parts of Central Europe, including Poland, Lithuania, Ukraine and the western and south-western as well as eastern parts of Russia, this species has become an important zoonotic disease in recent decades, causing thousands of infections in humans (Kondrashin et al. 2020; Rossi et al. 2015). For instance, in Ukraine D. repens was confirmed in 1′465 human cases between the years 1996 and 2012, based on a Europe-wide unique nationwide mandatory reporting for human cases since 1975 (Salamatin et al. 2013). Surveillance of human subcutaneous and ocular dirofilariosis in the Russian Federation and Belarus revealed overall 1′272 cases between 1997 and 2013, mainly from south-western regions of the Russian Federation, but with a northwards spread (Kartashev et al. 2015; Moskvina and Ermolenko 2018). Data collected between 1981 and 2011 from the Russian Federation, Ukraine, Belarus and Kazakhstan were applied to a climatic prediction model and regional warming was found to increase the annual generations of Dirofilaria spp. and thus increase suitable transmission areas by 18.5% in 2030 (Kartashev et al. 2014). Previous autochthonous cases of D. repens in humans have been described in Poland (Cielecka et al. 2012), Italy (Pampiglione et al. 2001), nine countries of the Balkan Peninsula (Tasić-Otašević et al. 2015), Hungary (Dóczi et al. 2015) and Slovakia (Babal et al. 2008; Boldiš et al. 2020). More recently, between 2017 and 2022, single cases of human D. repens infections were reported again from the Balkan area, in Serbia (Krstic et al. 2017), Croatia (Skrinjar et al. 2022), Bulgaria (Velev et al. 2019) and Romania (Ciuca et al. 2018), and further cases from Greece (Bozidis et al. 2021) and Italy (Ahmed et al. 2022), Interestingly, also in Austria, overall 39 human cases of dirofilariosis were identified between 1978 and 2020, with a general increasing course since 1998 (Riebenbauer et al. 2021). In addition, single human cases were reported from south-eastern France (Hennocq et al. 2020) and Germany (Uslu et al. 2017), confirming a north- and westwards trend in Europe.
Dirofilaria repens infections in dogs are also well documented in north-eastern, northern and southern Europe (Alsarraf et al. 2021; Genchi et al. 2011; Pantchev et al. 2009; Tarello 2011). However, in Central Europe, the epidemiological situation of D. repens north of the Alps is not fully known and only fragmentary data are available. For decades, in most cases of D. repens infections diagnosed in dogs in Central Europe and Great Britain the dogs originated from classical endemic areas or had a travel history with exposure in these areas. In Austria, for instance, most of previously detected cases in dogs were suspected to be imported. However, single cases detected in humans and dogs indicated autochthonous transmission of D. repens in eastern Austria (Fuehrer et al. 2016). Interestingly, in a surveillance programme that included 7632 mosquitoes, DNA from D. repens was amplified in two of 437 pools: the positive pools were from eastern Austria (Silbermayr et al. 2014), close to Hungary. Accordingly, the almost triplication of documented cases between 2014 and 2017 suggested endemic establishment in the country (Sonnberger et al. 2020). Also of interest is the so-called ´stable´ transmission of D. repens in north-eastern Germany and positive cases in dogs in the same region (Czajka et al. 2014; Sassnau et al. 2013). On the other side, D. repens was shown to be the most frequently imported filarial infection in Germany already in 2008–2010: in more than 8000 dogs with travel history or imported from endemic countries, D. repens was identified in 42 dogs, with Slovenia and Hungary being the most frequent mentioned countries (Pantchev et al. 2011). In fact, the endemic situation in these and surrounding countries such as the Czech Republik and Slovakia is confirmed by recent studies (Farkas et al. 2020; Jurankova et al. 2022; Martina et al. 2021). Furthermore, increasing reported cases of canine dirofilariosis due to D. repens in Lithuania, Latvia, Poland and Belarus confirm the trend for spreading northeastern (Alsarraf et al. 2021; Sabūnas et al. 2019).
Suspected reasons for the increasing number of reports of D. repens in northern Europe are the organised import of infected dogs from animal shelters protecting stray and unwanted dogs in the endemic countries, as well as the concurrent changing travel habits of humans taking their pets more frequently on trips. Furthermore, global trade can lead to the spread of infective vectors, allowing them to infect susceptible hosts in non-endemic regions (Genchi et al. 2011).
However, for instance, although predicted, the establishment of D. repens in the southern parts of Switzerland in areas where D. repens has been found several times in individual dogs has not been progressed in the last 30 years (Fuehrer et al. 2021). Therefore, some further unknown epidemiological factors may be associated with transmission of the parasite.
Diagnosis of filarial infections in dogs is achieved by concentration of blood microfilariae using the Knott Test (Knott 1939) or a filter method (Bell 1967). Morphometric measurements of microfilariae fixed with the Knott Test allow the differentiation of D. immitis and D. repens from the other smaller filarial species (Magnis et al. 2013). Another, older approach is the use of acid phosphatase staining with isolated microfilariae for morphological differentiation (Chalifoux and Hunt 1971; Peribáñez et al. 2001). Today, genetic identification of individual microfilariae at species level can easily be achieved by PCR (Rishniw et al. 2006). However, all microfilaria-related diagnostic methods lack high sensitivity due to long lasting prepatent infections, intermittent microfilaremia due to microfilarial periodicity, same-sex infections, anthelmintic-induced adult sterility and infections in which microfilariae have been destroyed by anthelmintics or by an immune response (Bowman and Mannella 2011; Rawlings et al. 1982).
For the identification of heartworm infections, detection of D. immitis antigen produced by adult females in the definitive host’s blood is the most common diagnostic procedure today (Weil 1987). There are commercially available ELISA kits that detect infections with at least one female worm and are therefore described as highly sensitive and nearly 100% specific (Atkins 2003; Lee et al. 2011). However, cross-reactions with sera from dogs infected with Angiostrongylus vasorum have been documented in 3/6 commercially available test kits (Schnyder and Deplazes 2012). Moreover, it has been suggested that the use of slow-kill heartworm treatments can induce immune complexes that lead to misleading false-negative results (Drake et al. 2015). If those complexes are destroyed by heat treatment, samples convert from negative to positive, as it has been shown for 7% of samples in a study from the USA (Velasquez et al. 2014).
Serology for the detection of specific antibodies has previously been considered to have low specificity, because of cross-reactivity with many other non-filarial nematodes (Grieve et al. 1981; Grieve and Knight 1985; Sisson et al. 1985). Recently, a monoclonal antibody based on-plate affinity purification of a crude D. immitis antigen was shown to not cross-react with sera from dogs experimentally infected with non-filarial nematodes such as Angiostrongylus vasorum, Toxocara canis, Ancylostoma caninum and Trichuris vulpis (Joekel et al. 2017). Furthermore, few positive reactions were found in dogs with documented natural infections with Crenosoma vulpis, A. vasorum and Capillaria aerophila (syn. Eucoleus aerophilus), but in these dogs previous exposure to filarial species could not be excluded. On the other hand, more than 50% of the dogs with low-pathogenic filarial infections with Acanthocheilonema, Dipetalonema spp. (Joekel et al. 2017) and Cercopithifilaria spp. (Deplazes, personal communication) had positive antibody reactions. Therefore, this test was defined as filarial-specific with a sensitivity of 93.8% for D. immitis patent infections and 100% for D. repens patent infections. Seroconversion of dogs experimentally infected with D. repens occurred between 24- and 80-day post inoculation (dpi) with third-stage larvae, much earlier than the beginning of patency (161–238 dpi) (Petry et al. 2015). Due to these test characteristics, the presented ELISA represents a suitable epidemiological tool, especially for the study of Dirofilaria spp. in low or non-endemic areas, where transmission of the apathogenic species is scarce (Joekel et al. 2017). To date, there have been no large-scale studies with high numbers of tested dogs outside Southern Europe. The aim of this work was to determine the distribution patterns of canine filarial infections in dogs with overall 8877 blood samples from Austria, Denmark, Germany, Italy, Lithuania, Poland, Switzerland and the UK.
Material and methods
Dog blood samples
The samples from Austria, Denmark, Germany, Italy, Poland and the UK were previously collected as part of other studies (Guardone et al. 2013; Schnyder et al. 2013a, 2013b). The samples from Italy were used as a positive known endemic area for a variety of filarial species (Cringoli et al. 2001; Magi et al. 2008, 2012; Otranto et al. 2013; Traversa et al. 2010). The samples from Lithuania originated from Kaunas (Central Lithuania; pet and sheltered dogs) and Klaipeda (Western Lithuania; pet dogs). The samples from Switzerland were collected for a sero-epidemiological survey on Angiostrongylus vasorum (Lurati et al. 2015). All sera were collected from dogs presented at veterinary clinics for different reasons and were complemented by corresponding data on the owner’s postal code. Due to data protection, no further information about the animal or the animal owner was available.
ELISA for detection of filarial-specific antibodies
The ELISA was performed as previously described (Joekel et al. 2017), with following modifications: a large batch of monoclonal antibodies (mAb Di36/1) was prepared. All ELISA plates included three positive control sera from dogs with experimental D. repens infections (Joekel et al. 2017), two negative controls from healthy dogs to adapt the plate-to-plate variation, a background and a conjugate control. The cut-off value was calculated for each country as follows: mean plus three standard deviations of the ELISA optical density (OD) values (measured at 405 nm) of sera from 300 dogs per country (n = 228 for Denmark, year of sampling: 2017). As an additional procedure, all sera that resulted positive (above the cut-off value calculated for each country) were retested in an ELISA without D. immitis somatic antigen (defined as control ELISA) to exclude false-positive reactions between the murine monoclonal antibodies and the dog sera. For the positive sera in the control ELISA, we included a restriction criterion: if the OD value of the retested sample without antigen was higher than the value of the test ELISA with somatic antigen, the sample was considered uninterpretable and excluded from further analyses. Furthermore, 53 samples from Lithuania were re-examined by Knott test and in each positive sample (n = 8) 10 microfilariae were measured morphometrically (Magnis et al. 2013). Eventually, in 3 cases the diagnosis was confirmed by PCR (Cafarelli et al. 2019).
Statistical and data analysis
The frequency distribution of the OD values was assumed to be a mixture distribution with two or more modes. The probability distribution with the highest mode represents the OD values of the assumed positive dogs and the probability distribution(s) of the lower mode(s) represents the assumed negative dogs. As a first stage mixture distribution was analysed as a mixture of normal distributions using the R package mclust (Scrucca et al. 2016). This optimised the number of mixture distributions using BIC. As a second step the number of distributions was further reduced by combining mixture components for clustering (Baudry et al. 2010). This resulted in the most likely probability distributions to which negative and positive dogs belonged and thus enabled a probability of true infection status to be assigned to each dog. Mean probabilities of the true infection status across groups of dogs represent an estimate of true prevalence.
Mapping, geographical distribution
Based on the postal codes of the addresses of origin of the samples, the mean probabilities were mapped into 1st or 2nd level administrative districts. First or second level was chosen so that districts of similar area could be readily compared across different countries. Mean prevalences were calculated when there were 5 or more samples available in a district. Districts with fewer than 5 samples were excluded to avoid bias created by the chance finding of one high OD value in a very small sample size. The mean prevalences were plotted into each district using 1st or 2nd administrative level shape files using R library ggplot2 (Wickham 2016).
Results
Table 1 shows the total number of tested dog samples for each country, the number of sera above the cut-off OD values, the calculated prevalence of each country and the corresponding true prevalence.
Analysis of the OD values using the Gaussian finite mixture models showed that the density distribution could be described by three normal distributions with a mean OD of 0.089, 0.120 and 0.287. Further analysis demonstrated that the two distributions with the lower mean ODs could be combined into a single distribution with 0.097. The presumed positive samples which were in the upper distribution with mean OD of 0.287 consisted of 2.55% of the samples, whilst 97.45% of samples were in the lower distribution and represented the presumed negative samples (Fig. 1).
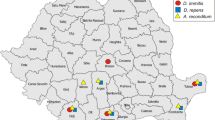
The estimated prevalence of canine filariosis across various central and northern European countries is illustrated in Fig. 2.
In an animal shelter in Lithuania, the ELISA survey revealed a high prevalence, identifying it as hotspot of infection. Further investigation of blood samples from dogs at this shelter identified microfilaremia in 8 of 53 dogs. Morphometric analyses of 10 microfilariae per positive sample yielded a mean length of 362.2–388.8 µm, suggestive of D. repens. Furthermore, the D. repens diagnosis was performed and confirmed in 3 cases by PCR.
Discussion
Identification of the European transmission areas of D. repens is of great importance due to its zoonotic threat. In this study, the application of a recently developed serological test (Joekel et al. 2017) based on detection of specific antibodies directed against filarial antigens allowed the estimation of the occurrence of canine filariosis based on a large number of dog sera. In contrast to southern Europe, being endemic for several filarial species, only autochthonous transmission of D. repens is expected in central Europe, and especially in the northern areas. This assumption was confirmed in this study by finding higher true prevalences in Poland and Lithuania, known endemic areas of D. repens (Alsarraf et al. 2021; Sabūnas et al. 2019), supporting also the hypothesised minor role of other filarial infections in the investigated areas.
The statistical approach was based on the assumption that there is no gold standard. The OD values were modelled to follow a bimodal distribution, with the upper distribution mode representing positive samples, whilst the lower distribution mode comprised the negative samples. Each individual OD value was assigned a probability of belonging to the upper modal distribution (i.e. a positive sample). The mean of these probabilities thus represents an estimate of the prevalence. The same procedure was used previously in a study on feline toxoplasmosis, where a gold standard diagnostic was not available (Schreiber et al. 2021). This approach differs from the assumption that a sample is positive if it is above a certain threshold in that no sample is classified as positive with probability 1, or vice versa. This avoids the problem of false negatives and false positives, but at the individual level there is no absolute certainty that the sample is positive or negative. However, at a population level, inferences can be made for prevalences. The mean of the probabilities that samples are positive provides an estimate of the prevalence. In our study, this allowed us to map the estimated true prevalences for seven countries in Europe.
Despite this, the method may lead to some counterintuitive results. For example, in Switzerland the estimated prevalence was 0.58, which was not dissimilar to the test prevalence of 0.77. In contrast, the UK had an estimated prevalence of 0.44 and a test prevalence of 0. This can be explained by the fact that the UK data include some false negatives based on the defined cut-off, but they belong to the positive rather than the negative model distribution (i.e. on the lower tail). The Swiss data would therefore show fewer false negatives, with the positives belonging to the upper part of the positive distribution. The high number of serum samples testing negative in the UK suggests that filarial infections in dogs remain very rare. Single positive case reports, i.e. one dog originally from Romania (Agapito et al. 2018) and one dog imported from Corfu (Wright 2017), suggest that both had a history of foreign travel.
The findings of the presented sero-prevalence of Dirofilaria spp. in Austria and the distribution pattern with positive foci in the eastern parts of the country are consistent with the recently reported detection of D. repens in military dogs in the Kaisersteinbruch region. However, the relative prevalence was only 1.4% including 94 samples with 2 positive findings (Sonnberger et al. 2021). The higher prevalence in the present study might be due to the higher number of samples examined. To the authors’ knowledge, there are no similar reports for the cluster in Western Austria. However, eggs of potential invasive mosquitoes that are competent vectors for D. repens transmission (Aedes spp.) were found along highways and in urban areas in both Eastern and Northern Tyrol (Fuehrer et al. 2020), additionally supported by autochthonous human infections (Geissler et al. 2022; Riebenbauer et al. 2021).
The distribution pattern for Germany is directed westwards in the present study, with a single cluster around the area where the federal states of Bavaria, Saxony-Anhalt and Thuringia border the Czech Republic. The first autochthonous case of D. repens infection was diagnosed in a southwestern region of Germany (Baden-Württemberg) (Hermosilla et al. 2006) and in three of 44 southwestern German hunting dogs that had no travel history (Pantchev et al. 2009). Additional suspected autochthonous D. repens infections were diagnosed in dogs from a sledge-dog kennel in Northeastern Germany, with limited travel history to Poland in winter (Sassnau et al. 2009). Furthermore, D. repens and D. immitis have been identified in mosquitoes from Southwestern and Northeastern Germany, covering the same geographic parts where autochthonous D. repens infections have been reported in dogs. The conclusion for this phenomenon was the suitability of the climate conditions for dirofilarial development in the mosquito vector, designating these regions at potential risk for stable endemicity (Sassnau et al. 2014a, 2014b; Sassnau and Genchi 2013). Finally, the endemicity of D. repens in Germany remains questionable, as most current data refer to imported or traveling pets (Pantchev et al. 2011; Schäfer et al. 2019a, b).
The test prevalence of almost 7% for Poland calculated in the present study with positive foci throughout the country confirms the results from previous studies showing that D. repens has become endemic in all districts of Poland (Fuehrer et al. 2021). Our finding of comparatively lower prevalence reflects the recent reported decline in the prevalence of D. repens in dogs in Poland (Alsarraf et al. 2021). Reasons for the decline in prevalence are thought to be increased awareness for the disease among dog owners and veterinarians, as well as preventive measures taken during the season of mosquito activity.
The low calculated test prevalence of 0.77% for Switzerland with three positively tested dog samples from Western Switzerland and one positive sample from Eastern Switzerland does not allow a statement about the origin of occurrence of D. repens within the country. Based on these findings, neither an autochthonous occurrence of the parasite nor imported cases could be confirmed. Moreover, most of the cases of dirofilariosis reported to date have a confirmed history of import or residence abroad (Fuehrer et al. 2021; Glaus et al. 2019), including positive dogs from southern Switzerland (Ticino), considered to be the border of the endemic area for both Dirofilaria spp. (Fuehrer et al. 2021).
To the authors’ knowledge, there are no published reports of Dirofilaria spp. in animals in Denmark. Our findings of 28 positive samples out of 1440 remain new. However, the measured OD value was only slightly above the cut-off value and no further tests were performed to confirm positivity. Therefore, false-positive test results are still possible.
Compared to previously published data on 8.3% prevalence percentages (25 positives out of 300 dog sera samples) in the Abruzzo region of central Italy (Traversa et al. 2010), the present test prevalence of 3.37% appears low. For D. immitis infections, data from more than 10′000 serological assays performed between 2009 and 2019 identified changing patterns between northern, central and southern Italy, with an overall gradual increase over time (Mendoza-Roldan et al. 2020). Recent studies and a questionnaire study showed that clinical infections with D. immitis and D. repens remain frequent diagnoses in veterinary clinics in the country (Ferrara et al. 2022Genchi et al. 2019; Macchioni et al. 2020).
Our comprehensive investigation of samples from Lithuania (ELISA, Knott, morphometric measurements and PCR) clearly confirms the endemic occurrence of D. repens in the Kaunas region of Central Lithuania. The samples were from dogs that lived in animal shelters that had not travelled before. Previously reported positive cases of D. repens in dogs (61 positives from 2280 blood samples) and seven confirmed cases of human infections support these findings. Moreover, a significantly higher infection rate was found in dogs from animal shelters than in pet dogs (Sabūnas et al. 2019).
Overall, the present study represents a novel approach to illustrate the putative occurence of filarial infections in Central Europe by combining ELISA with the statistical approach using OD density curves, and finally mapping the mean probabilities on a sample level.
Data availability
Data are available on request from the authors.
References
Agapito D, Aziz NA, Wang T, Morgan E, Wright I (2018) Subconjunctival Dirofilaria repens infection in a dog resident in the UK. J Small Anim Pract 59:50–52. https://doi.org/10.1111/jsap.12795
Ahmed N, Tonelli L, Labagnara G, Doglioni C, Pedica F (2022) Subcutaneous dirofilariosis in Italy a diagnostic hypothesis to remind when the anamnesis is misleading. Pathologica 114:174–177. https://doi.org/10.32074/1591-951X-297
Alsarraf M, Levytska V, Mierzejewska EJ, Poliukhovych V, Rodo A, Alsarraf M, Kavalevich D, Dwużnik-Szarek D, Behnke JM, Bajer A (2021) Emerging risk of Dirofilaria spp. infection in Northeastern Europe: high prevalence of Dirofilaria repens in sled dog kennels from the Baltic countries. Sci Rep 11:1–8. https://doi.org/10.1038/s41598-020-80208-1
Atkins CE (2003) Comparison of results of three commercial heartworm antigen test kits in dogs with low heartworm burdens. J Am Vet Med Assoc 222:1221–1223. https://doi.org/10.2460/javma.2003.222.1221
Babal P, Kobzova D, Novak I, Dubinsky P, Jalili N (2008) First case of cutaneous human dirofilariosis in Slovak Republic. Bratisl Lek Listy 109:486–488
Baudry J-P, Raftery AE, Celeux G, Lo K, Gottardo R (2010) Combining mixture components for clustering. J Comput Graph Stat 19:332–353. https://doi.org/10.1198/jcgs.2010.08111
Bell D (1967) Membrane filters and microfilariae: a new diagnostic technique. Ann Trop Med Parasitol 61:220–223. https://doi.org/10.1080/00034983.1967.11720805
Boldiš V, Ondriska F, Bošák V, Hajdúk O, Antolová D, Miterpáková M (2020) Pseudo-tumor of the epididymis, a rare clinical presentation of human Dirofilaria repens infection: a report of autochthonous case of dirofilariasis in Southwestern Slovakia. Acta Parasitol 65:550–553. https://doi.org/10.2478/s11686-020-00170-w
Bowman DD, Mannella C (2011) Macrocyclic lactones and Dirofilaria immitis microfilariae. Top Companion Anim Med 26:160–172. https://doi.org/10.1053/j.tcam.2011.07.001
Bozidis P, Sakkas H, Pertsalis A, Christodoulou A, Kalogeropoulos CD, Papadopoulou C (2021) Molecular analysis of Dirofilaria repens isolates from eye-care patients in Greece. Acta Parasitol 66:271–276. https://doi.org/10.1007/s11686-020-00257-4
Cafarelli C, Russo G, Mathis A, Silaghi C (2019) De novo genome sequencing and comparative stage-specific transcriptomic analysis of Dirofilaria repens. Int J Parasitol 49:911–919. https://doi.org/10.1016/j.ijpara.2019.04.008
Cancrini G, Scaramozzino P, Gabrielli S, Di Paolo M, Toma L, Romi R (2007) Aedes albopictus and Culex pipiens implicated as natural vectors of Dirofilaria repens in central Italy. J Med Entomol 44:1064–1066. https://doi.org/10.1603/0022-2585(2007)44[1064:aaacpi]2.0.co;2
Chalifoux L, Hunt RD (1971) Histochemical differentiation of Dirofilaria immitis and Dipetalonema reconditum. J Am Vet Med Assoc 158:601–605. https://doi.org/10.1111/j.1751-0813.1973.tb14671.x
Cielecka D, Zarnowska-Prymek H, Masny A, Salamatin R, Wesolowska M, Golab E (2012) Human dirofilariosis in Poland: the first cases of autochthonous infections with Dirofilaria repens. Ann Agric Environ Med 19:6
Ciuca L, Simon F, Rinaldi L, Kramer L, Genchi M, Cringoli G, Acatrinei D, Miron L, Morchon R (2018) Seroepidemiological survey of human exposure to Dirofilaria spp. in Romania and Moldova. Acta Trop 187:169–174. https://doi.org/10.1016/j.actatropica.2018.07.012
Cringoli G, Rinaldi L, Veneziano V, Capelli G (2001) A prevalence survey and risk analysis of filariosis in dogs from the Mt. Vesuvius area of southern Italy. Vet Parasitol 102:243–252. https://doi.org/10.1016/s0304-4017(01)00529-5
Czajka C, Becker N, Jost H, Poppert S, Schmidt-Chanasit J, Kruger A, Tannich E (2014) Stable transmission of Dirofilaria repens nematodes, northern Germany. Emerg Infect Dis 20:328–331. https://doi.org/10.3201/eid2002.131003
Deplazes P, Guscetti F, Wunderlin E, Bucklar H, Skaggs J, Wolff K (1995) Endoparasite infection in stray and abandoned dogs in southern Switzerland. Schweiz Arch Tierheilkd 137:172–179
Dóczi I, Bereczki L, Gyetvai T, Fejes I, Skribek Á, Szabó Á, Berkes S, Tiszlavicz L, Bartha N, Bende B (2015) Description of five dirofilariasis cases in South Hungary and review epidemiology of this disease for the country. Wien Klin Wochenschr 127:696–702. https://doi.org/10.1007/s00508-015-0825-4
Drake J, Gruntmeir J, Merritt H, Allen L, Little SE (2015) False negative antigen tests in dogs infected with heartworm and placed on macrocyclic lactone preventives. Parasit Vectors 8:1–5. https://doi.org/10.1186/s13071-015-0698-4
Farkas R, Mag V, Gyurkovszky M, Takács N, Vörös K, Solymosi N (2020) The current situation of canine dirofilariosis in Hungary. Parasitol Res 119:129–135. https://doi.org/10.1007/s00436-019-06478-5
Fuehrer HP, Auer H, Leschnik M, Silbermayr K, Duscher G, Joachim A (2016) Dirofilaria in humans, dogs, and vectors in Austria (1978–2014)-from imported pathogens to the endemicity of Dirofilaria repens. PLoS Negl Trop Dis 10:e0004547. https://doi.org/10.1371/journal.pntd.0004547
Fuehrer HP, Schoener E, Weiler S, Barogh BS, Zittra C, Walder G (2020) Monitoring of alien mosquitoes in Western Austria (Tyrol, Austria, 2018). PLoS Negl Trop Dis 14:e0008433. https://doi.org/10.1371/journal.pntd.0008433
Fuehrer HP, Morelli S, Unterköfler MS, Bajer A, Bakran-Lebl K, Dwużnik-Szarek D, Farkas R, Grandi G, Heddergott M, Jokelainen P (2021) Dirofilaria spp. and Angiostrongylus vasorum: current risk of spreading in Central and Northern Europe. Pathogens 10:1268. https://doi.org/10.3390/pathogens10101268
Geissler N, Ruff J, Walochnik J, Ludwig W, Auer H, Wiedermann U, Geissler W (2022) Autochthonous human Dirofilaria repens infection in Austria. Acta Parasitol 67:1039–1043. https://doi.org/10.1007/s11686-021-00506-0
Genchi C, Kramer LH, Rivasi F (2011) Dirofilarial infections in Europe. Vector-Borne Zoonotic Dis 11:1307–1317. https://doi.org/10.1089/vbz.2010.0247
Genchi M, Rinaldi L, Venco L, Cringoli G, Vismarra A, Kramer L (2019) Dirofilaria immitis and D. repens in dog and cat: a questionnaire study in Italy. Vet Parasitol 267:26–31. https://doi.org/10.1016/j.vetpar.2019.01.014
Glaus TM, Ivasovic F, Jörger FB, Schreiber N, Baron Toaldo M (2019) Minimal invasive Extraktion von adulten Dirofilaria immitis bei einem Hund mit Kavalsyndrom. Schweiz Arch Tierheilkd 161:749–754. https://doi.org/10.17236/sat00234
Grieve RB, Mika-Johnson M, Jacobson R, Cypess R (1981) Enzyme-linked immunosorbent assay for measurement of antibody responses to Dirofilaria immitis in experimentally infected dogs. Am J Vet Res 42:66–69
Grieve RB, Knight DH (1985) Anti-Dirofilaria immitis antibody levels before and after anthelmintic treatment of experimentally infected dogs. J Parasitol 71(1):56–61
Guardone L, Schnyder M, Macchioni F, Deplazes P, Magi M (2013) Serological detection of circulating Angiostrongylus vasorum antigen and specific antibodies in dogs from central and northern Italy. Vet Parasitol 192:192–198. https://doi.org/10.1016/j.vetpar.2012.10.016
Hennocq Q, Helary A, Debelmas A, Monsel G, Labat A, Bertolus C, Martin C, Caumes E (2020) Oral migration of Dirofilaria repens after creeping dermatitis. Parasite 27:16. https://doi.org/10.1051/parasite/2020015
Hermosilla C, Pantchev N, Dyachenko V, Gutmann M, Bauer C (2006) First autochthonous case of canine ocular Dirofilaria repens infection in Germany. Vet Rec 158:134. https://doi.org/10.1136/vr.158.4.134
Ionică AM, Matei IA, Mircean V, Dumitrache MO, D’Amico G, Győrke A, Pantchev N, Annoscia G, Albrechtová K, Otranto D, Modrý D, Mihalca AD (2015) Current surveys on the prevalence and distribution of Dirofilaria spp. and Acanthocheilonema reconditum infections in dogs in Romania. Parasitol Res 114:975–982. https://doi.org/10.1007/s00436-014-4263-4
Joekel DE, Maier S, Huggel K, Schaper R, Deplazes P (2017) Specific Antibody Detection in Dogs with Filarial Infections. Parasitol Res 116:81–90. https://doi.org/10.1007/s00436-017-5494-y
Jurankova J, Senkyrikova Mitkova B, Novotna M, Hofmannova L, Cervena B, Bowman DD, Modry D (2022) Further data on the distribution of Dirofilaria spp. in the Czech Republic in dogs. Folia Parasitol (Praha) 69:2022.007. https://doi.org/10.14411/fp.2022.007
Kartashev V, Afonin A, González-Miguel J, Sepúlveda R, Simón L, Morchón R, Simón F (2014) Regional warming and emerging vector-borne zoonotic dirofilariosis in the Russian Federation, Ukraine, and other post-Soviet states from 1981 to 2011 and projection by 2030. BioMed Res Int 2014:858936. https://doi.org/10.1155/2014/858936
Kartashev V, Tverdokhlebova T, Korzan A, Vedenkov A, Simón L, González-Miguel J, Morchón R, Siles-Lucas M, Simón F (2015) Human subcutaneous/ocular dirofilariasis in the Russian Federation and Belarus, 1997–2013. Int J Infect Dis 33:209–211. https://doi.org/10.1016/j.ijid.2015.02.017
Knott J (1939) A method for making microfilarial surveys on day blood. J Trans R Soc Trop Med Hyg 33(2):191–196. https://doi.org/10.1016/s0035-9203(39)90101-x
Kondrashin AV, Morozova LF, Stepanova EV, Turbabina NA, Maksimova MS, Morozov EN (2020) Anthology of dirofilariasis in Russia (1915–2017). Pathogens 9:275. https://doi.org/10.3390/pathogens9040275
Krstic M, Gabrielli S, Ignjatovic M, Savic S, Cancrini G, Randelovic G, Momcilovic S, Stojnev S, Otasevic S (2017) An appraisal of canine and human cases reveals an endemic status of dirofilariosis in parts of Serbia. Mol Cell Probes 31:37–41. https://doi.org/10.1016/j.mcp.2016.08.005
Laidoudi Y, Ringot D, Watier-Grillot S, Davoust B, Mediannikov O (2019) A cardiac and subcutaneous canine dirofilariosis outbreak in a kennel in central France. Parasite 26:72. https://doi.org/10.1051/parasite/2019073
Lee ACY, Bowman DD, Lucio-Forster A, Beall MJ, Liotta JL, Dillon R (2011) Evaluation of a new in-clinic method for the detection of canine heartworm antigen. Vet Parasitol 177:387–391. https://doi.org/10.1016/j.vetpar.2010.11.050
Lurati L, Deplazes P, Hegglin D, Schnyder M (2015) Seroepidemiological survey and spatial analysis of the occurrence of Angiostrongylus vasorum in Swiss dogs in relation to biogeographic aspects. Vet Parasitol 212:219–226. https://doi.org/10.1016/j.vetpar.2015.08.017
Macchioni F, Sed G, Cecchi F (2020) Canine filarial infections in an area of Central Italy (Tuscany-Latium border) historically free from the disease. Vet Parasitol Reg Stud Reports 20:100404. https://doi.org/10.1016/j.vprsr.2020.100404
Magi M, Calderini P, Gabrielli S, Dell’Omodarme M, Macchioni F, Prati MC, Cancrini G (2008) Vulpes vulpes: a possible wild reservoir for zoonotic filariae. Vector-Borne and Zoonotic Dis 8:249–252. https://doi.org/10.1089/vbz.2007.0207
Magi M, Guardone L, Prati M, Tozzini G, Torracca B, Monni G, Macchioni F (2012) Canine filarial infections in Tuscany, central Italy. J Helminthol 86:113–116. https://doi.org/10.1017/S0022149X11000113
Magnis J, Lorentz S, Guardone L, Grimm F, Magi M, Naucke TJ, Deplazes P (2013) Morphometric analyses of canine blood microfilariae isolated by the Knott’s test enables Dirofilaria immitis and D. repens species-specific and Acanthocheilonema (syn. Dipetalonema) genus-specific diagnosis. Parasit Vectors 6:48. https://doi.org/10.1186/1756-3305-6-48
Martina M, Zuzana H, Daniela V, Lenka B (2021) Different epidemiological pattern of canine dirofilariosis in two neighboring countries in Central Europe-the Czech Republic and Slovakia. Parasitol Res 120:547–552. https://doi.org/10.1007/s00436-020-06995-8
Mendoza-Roldan J, Benelli G, Panarese R, Iatta R, Furlanello T, Beugnet F, Zatelli A, Otranto D (2020) Leishmania infantum and Dirofilaria immitis infections in Italy, 2009–2019: changing distribution patterns. Parasit Vectors 13:1–8. https://doi.org/10.1186/s13071-020-04063-9
Morchon R, Carreton E, Gonzalez-Miguel J, Mellado-Hernandez I (2012) Heartworm Disease (Dirofilaria immitis) and their vectors in Europe - new distribution trends. Front Physiol 3:196. https://doi.org/10.3389/fphys.2012.00196
Moskvina TV, Ermolenko AV (2018) Dirofilariasis in Russian Federation: a big problem with large distribution. Russ Open Med J 7:102–102. https://doi.org/10.15275/rusomj.2018/0102
Otranto D, Brianti E, Dantas-Torres F, Miro G, Latrofa MS, Mutafchiev Y, Bain O (2013) Species diversity of dermal microfilariae of the genus Cercopithifilaria infesting dogs in the Mediterranean region. Parasitology 140:99–108. https://doi.org/10.1017/S0031182012001357
Pampiglione S, Rivasi F (2000) Human dirofilariasis due to Dirofilaria (Nochtiella) repens: an update of world literature from 1995 to 2000. Parassitologia 42:231–254
Pampiglione S, Rivasi F, Angeli G, Boldorini R, Incensati R, Pastormerlo M, Pavesi M, Ramponi A (2001) Dirofilariasis due to Dirofilaria repens in Italy, an emergent zoonosis: report of 60 new cases. Histopathology 38:344–354. https://doi.org/10.1046/j.1365-2559.2001.01099.x
Panayotova-Pencheva M, Šnábel V, Dakova V, Čabanová V, Cavallero S, Trifonova A, Mirchev R, Hurníková Z, Vasilková Z, Miterpáková M (2020) Dirofilaria immitis in Bulgaria: the first genetic baseline data and an overview of the current status. Helminthologia 57:211–218. https://doi.org/10.2478/helm-2020-0026
Pantchev N, Norden N, Lorentzen L, Rossi M, Rossi U, Brand B, Dyachenko V (2009) Current surveys on the prevalence and distribution of Dirofilaria spp. in dogs in Germany. Parasitol Res 105:63–74. https://doi.org/10.1007/s00436-009-1497-7
Pantchev N, Etzold M, Daugschies A, Dyachenko V (2011) Diagnosis of imported canine filarial infections in Germany 2008–2010. Parasitol Res 109:61–76. https://doi.org/10.1007/s00436-011-2403-7
Peribáñez MA, Lucientes J, Arce S, Morales M, Castillo JA, MaJ G (2001) Histochemical differentiation of Dirofilaria immitis, Dirofilaria repens and Acanthocheilonema dracunculoides microfilariae by staining with a commercial kit, Leucognost-SP®. Vet Parasitol 102:173–175. https://doi.org/10.1016/s0304-4017(01)00516-7
Petruschke G, Rossi L, Genchi C, Pollono F (2001) Canine dirofilariasis in the canton of Ticino and in the neighboring areas of northern Italy. Schweiz Arch Tierheilkd 143:141–147
Petry G, Genchi M, Schmidt H, Schaper R, Lawrenz B, Genchi C (2015) Evaluation of the adulticidal efficacy of imidacloprid 10%/moxidectin 2.5%(w/v) spot-on (Advocate®, Advantage® Multi) against Dirofilaria repens in experimentally infected dogs. Parasitol Res 114:131–144. https://doi.org/10.1007/s00436-015-4519-7
Rawlings C, Dawe D, McCall J, Keith J, Prestwood A (1982) Four types of occult Dirofilaria immitis infection in dogs. J Am Vet Med Assoc 180:1323–1326
Riebenbauer K, Weber PB, Walochnik J, Karlhofer F, Winkler S, Dorfer S, Auer H, Valencak J, Laimer M, Handisurya A (2021) Human dirofilariosis in Austria: the past, the present, the future. Parasit Vectors 14:227. https://doi.org/10.1186/s13071-021-04696-4
Rishniw M, Barr SC, Simpson KW, Frongillo MF, Franz M, Alpizar JLD (2006) Discrimination between six species of canine microfilariae by a single polymerase chain reaction. Vet Parasitol 135:303–314. https://doi.org/10.1016/j.vetpar.2005.10.013
Rossi A, Peix Á, Pavlikovskaya T, Sagach O, Nikolaenko S, Chizh N, Kartashev V, Simón F, Siles-Lucas M (2015) Genetic diversity of Dirofilaria spp. isolated from subcutaneous and ocular lesions of human patients in Ukraine. Acta Trop 142:1–4. https://doi.org/10.1016/j.actatropica.2014.10.021
Sabūnas V, Radzijevskaja J, Sakalauskas P, Petkevičius S, Karvelienė B, Žiliukienė J, Lipatova I, Paulauskas A (2019) Dirofilaria repens in dogs and humans in Lithuania. Parasit Vectors 12:1–10. https://doi.org/10.1186/s13071-019-3406-y
Salamatin RV, Pavlikovska TM, Sagach OS, Nikolayenko SM, Kornyushin VV, Kharchenko VO, Masny A, Cielecka D, Konieczna-Salamatin J, Conn DB, Golab E (2013) Human dirofilariasis due to Dirofilaria repens in Ukraine, an emergent zoonosis: epidemiological report of 1465 cases. Acta Parasitol 58:592–598. https://doi.org/10.2478/s11686-013-0187-x
Sassnau R, Genchi C (2013) Qualitative risk assessment for the endemisation of Dirofilaria repens in the state of Brandenburg (Germany) based on temperature-dependent vector competence. Parasitol Res 112:2647–2652. https://doi.org/10.1007/s00436-013-3431-2
Sassnau R, Dyachenko V, Pantchev N, Stockel F, Dittmar K, Daugschies A (2009) Dirofilaria-repens-Befall in einem Schlittenhunde-Rudel im Land Brandenburg. Diagnose und Therapie der kaninen kutanen Dirofilariose. Tierarztl Prax Ausgabe K, Kleintiere/Heimtiere 37:95. https://doi.org/10.1055/s-0038-1622777
Sassnau R, Kohn M, Demeler J, Kohn B, Muller E, Krucken J, von Samson-Himmelstjerna G (2013) Is Dirofilaria repens endemic in the Havelland district in Brandenburg, Germany? Vector Borne Zoonotic Dis 13:888–891. https://doi.org/10.1089/vbz.2012.1293
Sassnau R, Czajka C, Kronefeld M, Werner D, Genchi C, Tannich E, Kampen H (2014a) Dirofilaria repens and Dirofilaria immitis DNA findings in mosquitoes in Germany: temperature data allow autochthonous extrinsic development. Parasitol Res 113:3057–3061. https://doi.org/10.1007/s00436-014-3970-1
Sassnau R, Daugschies A, Lendner M, Genchi C (2014b) Climate suitability for the transmission of Dirofilaria immitis and D. repens in Germany. Vet Parasitol 205:239–245. https://doi.org/10.1016/j.vetpar.2014.06.034
Schäfer I, Volkmann M, Beelitz P, Merle R, Müller E, Kohn B (2019a) Retrospective analysis of vector-borne infections in dogs after travelling to endemic areas (2007–2018). Vet Parasitol: X 2:100015. https://doi.org/10.1016/j.vpoa.2019.100015
Schäfer I, Volkmann M, Beelitz P, Merle R, Müller E, Kohn B (2019b) Retrospective evaluation of vector-borne infections in dogs imported from the Mediterranean region and southeastern Europe (2007–2015). Parasit Vectors 12:1–10. https://doi.org/10.1186/s13071-018-3284-8
Schnyder M, Deplazes P (2012) Cross-reactions of sera from dogs infected with Angiostrongylus vasorum in commercially available Dirofilaria immitis test kits. Parasit Vectors 5:258. https://doi.org/10.1186/1756-3305-5-258
Schnyder M, Schaper R, Bilbrough G, Morgan E, Deplazes P (2013a) Seroepidemiological survey for canine angiostrongylosis in dogs from Germany and the UK using combined detection of Angiostrongylus vasorum antigen and specific antibodies. Parasitology 140:1442–1450. https://doi.org/10.1017/S0031182013001091
Schnyder M, Schaper R, Pantchev N, Kowalska D, Szwedko A, Deplazes P (2013b) Serological detection of circulating Angiostrongylus vasorum antigen-and parasite-specific antibodies in dogs from Poland. Parasitol Res 112:109–117. https://doi.org/10.1007/s00436-013-3285-7
Schreiber N, Basso W, Riond B, Willi B, Torgerson PR, Deplazes P (2021) Antibody kinetics and exposure to Toxoplasma gondii in cats: a seroepidemiological study. Int J Parasitol 51:291–299. https://doi.org/10.1016/j.ijpara.2020.09.011
Scrucca L, Fop M, Murphy TB, Raftery AE (2016) mclust 5: clustering, classification and density estimation using Gaussian finite mixture models. The R Journal 8:289
Silaghi C, Beck R, Capelli G, Montarsi F, Mathis A (2017) Development of Dirofilaria immitis and Dirofilaria repens in Aedes japonicus and Aedes geniculatus. Parasit Vectors 10:94. https://doi.org/10.1186/s13071-017-2015-x
Silbermayr K, Eigner B, Joachim A, Duscher GG, Seidel B, Allerberger F, Indra A, Hufnagl P, Fuehrer HP (2014) Autochthonous Dirofilaria repens in Austria. Parasit Vectors 7:1–3. https://doi.org/10.1186/1756-3305-7-226
Simon F, Siles-Lucas M, Morchon R, Gonzalez-Miguel J, Mellado I, Carreton E, Montoya-Alonso JA (2012) Human and animal dirofilariasis: the emergence of a zoonotic mosaic. Clin Microbiol Rev 25:507–544. https://doi.org/10.1128/CMR.00012-12
Sisson D, Dilling G, Wong M, Thomas W (1985) Sensitivity and specificity of the indirect-fluorescent antibody test and two enzyme-linked immunosorbent assays in canine dirofilariasis. Am J Vet Res 46:1529–1533
Skrinjar I, Brailo V, Loncar Brzak B, Lozic Erent J, Bukovski S, Juras DV (2022) Live intraoral Dirofilaria repens of lower lip mimicking mucocele-First reported case from Croatia. Int J Environ Res Public Health 19:4330. https://doi.org/10.3390/ijerph19074330
Sonnberger BW, Graf B, Straubinger RK, Rackl D, Obwaller AG, Peschke R, Barogh BS, Joachim A, Fuehrer HP (2021) Vector-borne pathogens in clinically healthy military working dogs in eastern Austria. Parasitol Int 84:102410. https://doi.org/10.1016/j.parint.2021.102410
Széll Z, Bacsadi Á, Szeredi L, Nemes C, Fézer B, Bakcsa E, Kalla H, Tolnai Z, Sréter T (2020) Rapid spread and emergence of heartworm resulting from climate and climate-driven ecological changes in Hungary. Vet Parasitol 280:109067. https://doi.org/10.1016/j.vetpar.2020.109067
Tarello W (2011) Clinical aspects of dermatitis associated with Dirofilaria repens in pets: a review of 100 canine and 31 feline cases (1990–2010) and a report of a new clinic case imported from Italy to Dubai. J Parasitol Res 2011:578385. https://doi.org/10.1155/2011/578385
Tasić-Otašević SA, Božinović MST, Gabrielli SV, Genchi C (2015) Canine and human Dirofilaria infections in the Balkan Peninsula. Vet Parasitol 209:151–156. https://doi.org/10.1016/j.vetpar.2015.02.016
Traversa D, Aste G, Milillo P, Capelli G, Pampurini F, Tunesi C, Santori D, Paoletti B, Boari A (2010) Autochthonous foci of canine and feline infections by Dirofilaria immitis and Dirofilaria repens in central Italy. Vet Parasitol 169:128–132. https://doi.org/10.1016/j.vetpar2009.12.034
Uslu U, Jahn J, Erdmann M (2017) Delayed diagnosis of subcutaneous dirofilariasis following a mosquito bite sustained in Germany. J Dtsch Dermatol Ges 15:727–728. https://doi.org/10.1111/ddg.13257
Velasquez L, Blagburn BL, Duncan-Decoq R, Johnson EM, Allen KE, Meinkoth J, Gruntmeir J, Little SE (2014) Increased prevalence of Dirofilaria immitis antigen in canine samples after heat treatment. Vet Parasitol 206:67–70. https://doi.org/10.1016/j.vetpar.2014.03.021
Velev V, Pelov T, Garev T, Peev S, Kaftandjiev I, Harizanov R (2019) Epididymal dirofilariasis in a child: First case report from Bulgaria. Med Princ Pract 28:96–98. https://doi.org/10.1159/000494619
Weil GJ (1987) Dirofilaria immitis: identification and partial characterization of parasite antigens in the serum of infected dogs. Exp Parasitol 64:244–251. https://doi.org/10.1016/0014-4894(87)90149-4
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York. https://doi.org/10.1007/978-0-387-98141-3
Wright I (2017) Case report: Dirofilaria repens in a canine castrate incision. Companion Animal 22:316–318. https://doi.org/10.12968/coan.2017.22.6.316
Funding
Open access funding provided by University of Zurich The study was partially supported by a grant from Bayer Animal Health GmbH, coordinated by Dr. Roland Schaper, and represents the doctoral dissertation of Jeannine Ellen Fehr.
Author information
Authors and Affiliations
Contributions
JEF: performed research, analysed data, writing – original draft. MaS: resources, methodology, fund acquisition, writing – original draft, review and editing. DEJ: methodology, resources, writing – review and editing. NP: resources, writing – review and editing. MiS: resources, organisation. PT: methodology, contributed new methods or models, writing – review and editing. PD: conceptualisation, supervision, methodology, contributed new methods or models, writing – review and editing.
Corresponding author
Ethics declarations
Ethics approval
All applicable international and institutional guidelines for the care and use of animals were followed. Part of the study was conducted in compliance with Lithuanian animal welfare regulations (No. B1-866, 2012; No. XI-2271, 2012).
Consent to participate
Not applicable.
Consent for publication
All authors read and approved the final version of the manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Julia Walochnik
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fehr, J.E., Schnyder, M., Joekel, D.E. et al. Estimated specific antibody-based true sero-prevalences of canine filariosis in dogs in Central Europe and the UK. Parasitol Res 121, 3671–3680 (2022). https://doi.org/10.1007/s00436-022-07695-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07695-1