Abstract
Background
Bariatric surgery in patients with BMI over 50 kg/m2 is a challenging task. The aim of this study was to address main issues regarding perioperative management of these patients by using a worldwide survey.
Methods
An online 48-item questionnaire-based survey on perioperative management of patients with a BMI superior to 50 kg/m2 was ideated by 15 bariatric surgeons from 9 different countries. The questionnaire was emailed to all members of the International Federation of Surgery for Obesity (IFSO). Responses were collected and analyzed by the authors.
Results
789 bariatric surgeons from 73 countries participated in the survey. Most surgeons (89.9%) believed that metabolic/bariatric surgery (MBS) on patients with BMI over 50 kg/m2 should only be performed by expert bariatric surgeons. Half of the participants (55.3%) believed that weight loss must be encouraged before surgery and 42.6% of surgeons recommended an excess weight loss of at least 10%. However, only 3.6% of surgeons recommended the insertion of an Intragastric Balloon as bridge therapy before surgery. Sleeve Gastrectomy (SG) was considered the best choice for patients younger than 18 or older than 65 years old. SG and One Anastomosis Gastric Bypass were the most common procedures for individuals between 18 and 65 years. Half of the surgeons believed that a 2-stage approach should be offered to patients with BMI > 50 kg/m2, with SG being the first step. Postoperative thromboprophylaxis was recommended for 2 and 4 weeks by 37.8% and 37.7% of participants, respectively.
Conclusion
This survey demonstrated worldwide variations in bariatric surgery practice regarding patients with a BMI superior to 50 kg/m2. Careful analysis of these results is useful for identifying several areas for future research and consensus building.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
According to the World Health Organization, in 2016, 650 million of adults were obese [1]. Despite all efforts to support prevention campaigns and nonsurgical treatments (e.g., lifestyle modifications, medications, behavioral therapy and diets) [2, 3], the prevalence of obesity is steadily increasing worldwide becoming an important health and financial burden. Bariatric surgery represents a safe and effective treatment for patients with morbid obesity and its related diseases [4,5,6]. Over the past few decades, diffusion and standardization of principal bariatric procedures has led to a significant reduction of peri- and postoperative complications [7,8,9,10]. However, these interventions may be challenging in patients with BMI > 50 kg/m2 with higher risks [11,12,13,14,15]. Indeed, the large amount of visceral fat makes the surgical approach more technically demanding, while, on the other hand, the advanced stage of obesity often carries severe comorbidities that could worsen perioperative outcomes. Moreover, subjects with BMI over 50 kg/m2 are poor respondents in terms of weight loss and metabolic outcomes when compared to patients with lower values of BMI. Despite all these concerns, there is no consensus on the perioperative management of these patients.
The aim of this study was to address the issue of perioperative management of patients with BMI over 50 kg/m2 undergoing bariatric surgery using a specific well-designed survey.
To comply with new policy of the International Federation for the Surgery for Obesity and Related Metabolic Disorders (IFSO), the term “super-obese” was not used.
Methods
A confidential, voluntary, online questionnaire-based survey was sent to the bariatric community through IFSO mailing list and social media. The 48 questions were conceived (Online Appendix 1) by 15 bariatric surgeons from 9 different countries in an attempt to address existing controversies and challenges on perioperative management of patients with BMI over 50 kg/m2. The questionnaire was divided into 4 parts: (1) preoperative general evaluation and management, (2) preoperative weight loss management, (3) surgical technique details, and (4) postoperative management (see Online Appendix 1).
Each part included multiple-choice questions with a comment box at the end. The survey started on January 15th and closed for analysis on March 2nd, 2021. The survey was emailed twice to IFSO members and the link was also freely shared on social and scientific media (Facebook™, Researchgate™, Whatsapp™, LinkedIn™ and WeChat®) and through personal networks and societies of bariatric and metabolic surgeons of a few countries. Responses were collected and reported as percentages, while graphs were used for representation where applicable. Being a survey among surgeons, Institution Review Board approval was not needed for this study.
Surgical procedures included in this survey were: (i) sleeve gastrectomy (SG): removal of about 80% of the stomach leaving a tube-shaped stomach; (ii) one anastomosis gastric bypass (OAGB): division of the upper part of the stomach into a tube, similar to the top three quarters of a sleeve, and then joined to a loop of intestine; (iii) Roux-en-Y gastric bypass (RYGB): creation of a small pouch of the upper stomach by stapling and direct attachment to it to part of the small intestine called the Roux limb ("Y" shape); (iv) laparoscopic adjustable gastric banding (LAGB): placement of an adjustable band around the top part of the stomach; (v) single anastomosis duodeno-ileal bypass with sleeve gastrectomy (SADI-S): creation of a sleeve gastrectomy followed by an end-to-side duodeno-ileal diversion; (vi) biliopancreatic diversion with duodenal switch (BPD/DS): creation of a sleeve gastrectomy and connection of the end portion of the intestine to the duodenum near the stomach.
Results
A total of 820 participants responded to the survey. However, 31 were not bariatric surgeons and their responses were excluded. Therefore, a final number of 789 responses were analyzed.
Three hundred and twenty surgeons (40.6%) had performed more than 1000 metabolic and bariatric surgeries (MBS), while 138 (17.5%), 219 (27.8%), and 112 (14.2%) had done 501 to 1000, 100 to 500, and Less than 100 MBS, respectively.
Nationality of participants
Bariatric surgeons from 73 countries took part in the survey. Online Table 1 shows the number and percentages of participants from each nation in alphabetical order.
Preoperative evaluations for patients with BMIs over 50 kg/m2
A total of 709 (89.9%) surgeons believe that MBS on patients with BMI over 50 kg/m2, should only be performed by expert bariatric surgeons and 621 (78.7%) of them also stated that only experienced bariatric anesthesiologists should be involved. Most surgeons (79.2%) do not use any surgical risk scoring system for these patients. Almost 73% of participants had performed MBS on adolescent patients (under 18 years old) with BMI over 50 kg/m2, while72.2% of them reported to have carried out bariatric interventions on elderly patients (over 65 years old). Preoperative eating and psychological disorders are routinely assessed by 73.7% of surgeons. About 76.5% of participants recommend preoperative and postoperative CPAP only in selected cases with sleep apnea. Responses on preoperative evaluation are summarized in Online Appendix 2.
Preoperative weight loss management in patients with BMIs over 50 kg/m2
Weight loss must be encouraged before surgery for 55.34% of participants and 42.6% recommend an excess weight loss of at least 10%. Only 3.6% of surgeons recommended insertion of an Intragastric Balloon (IGB) for patients and most of them (62.2%) only in selected cases. Likewise, 66.6% recommended preoperative Liraglutide only for selected cases. Forty-two percent of participants ask patients to start a very low-calorie diet (VLCD) 4 weeks prior to surgery for liver shrinkage and 23.3% of them only for patients with hepatomegaly. On the other hand, 38.3% believed rate of weight loss is not important to proceed with surgery. Data on preoperative weight loss management are summarized in Online Appendix 3.
Choice of surgical procedure
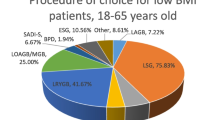
SG was considered the best choice for patients younger than 18 (62.1%) or older than 65 years old (54.7%). SG and OAGB were the most chosen procedures for individuals between 18 and 65 years (28% vs 20.6%, respectively) (Figs. 1, 2). There was no consensus regarding limb lengths and total small bowel measurement. Almost half of respondents (45.9%) recommended a 2-stage approach. SG should be the first step for 98% of participants, while LAGB only for 1.8%. As second stage, the OAGB with tailored limb, the SADI-S, the standard RYGB, and the RYGB with a long biliary limb over 150 cm were chosen by 24.5%, 18.4%, 16.5%, and 14.2%, respectively. Other options for second stage procedure were the OAGB with fixed limb (9.8%), the RYGB with long alimentary limb over 100 cm (5.5%), and the BPD/DS (3.8%). A small percentage (6.9%) of participants perform other surgical procedures.
Procedure of choice for three age categories of patients with BMIs over 50 kg/m2 as reported by the participants of the survey. A Under 18 years old, B 18 to 65 years old, and C older than 65 years old. LAGB laparoscopic adjustable gastric banding, LSG laparoscopic sleeve gastrectomy, LRYGB-S standard Roux-en-Y gastric bypass, LRYGB-LAL Roux-en-Y long alimentary limb > 100 cm gastric bypass, LRYGB-LBL Roux-en-Y long biliary limb > 150 cm gastric bypass, OAGB-F one anastomosis gastric bypass with fixed limb measures, OAGB-T one anastomosis Gastric Bypass with tailored limb measures, SADI-S single anastomosis duodeno–ileal bypass with sleeve gastrectomy, BPD-DS biliopancreatic diversion with duodenal switch
Metabolic and bariatric surgeries (MBS) algorithm in patients with BMIs over 50 based on respondent's answers. The percentage of the most responses of each items was mentioned. LSG laparoscopic sleeve gastrectomy, LRYGB-S standard Roux-en-Y Gastric Bypass, OAGB-T one anastomosis gastric bypass with tailored limb measures, SADI-S single anastomosis duodeno–ileal bypass with sleeve gastrectomy, BPD-DS biliopancreatic diversion with duodenal switch
Most participants (62.5%) stated that they had not performed robotic bariatric surgery on patients with BMIs over 50 kg/m2. However, 25.1% of participants who were experienced in robotic bariatric surgery believed that robotic surgery did not decrease postoperative morbidity in these patients. Only 3.3% of respondents considered single-incision laparoscopic surgery (SILS) to be safe for these patients, while 73% advised against it.
Thirty percent of participants recommended that revisional surgery or conversion for poor weight loss in patients should always be done, but most of them (68.6%) advised to perform an extensive preoperative evaluation.
Half of the participating surgeons (50.4%) recommended a time interval between the first and second bariatric procedures of 12 to 24 months. However, other surgeons would perform the second surgical procedure after less than 6 months (6.3%), 6–12 months (20.8%) and more than 24 months (22.3%).
The majority of participants (76.5%) believe that if there is resolution of comorbidities and adequate weight loss after the first procedure, performing the second step of the two-step procedure is not recommended.
When a micro/macro-nodular liver cirrhosis is found, 60.7% of surgeons perform liver biopsies, while 8.3% perform biopsies routinely and 14.5% only in case of fatty liver; 16.3% declared that they never perform biopsies during MBS.
Postoperative management (Online Appendix 4)
The majority of participants, 567 of the 789 surgeons (71.8%), did not recommend any postoperative imaging (upper gastrointestinal series, CT scan, etc.). 122 surgeons (15.5%) do believe that imaging should be performed routinely before hospital discharge and another 100 (12.6%) of participants believe that imaging should be done shortly after surgery.
Almost eighty percent (78.7%) of participants recommended postoperative intensive care unit admission for selected cases. Serum levels of creatine kinase to exclude rhabdomyolysis were routinely assessed by 43.6%, while 41.3% of participants measured it only when the intervention lasted longer than 2 h.
Postoperative thromboprophylaxis was recommended for 2 weeks by 37.8% of participants and for 4 weeks by 37.7%; 27.8% of surgeons prescribe postoperative anticoagulation for only one week. Postoperative vitamin supplementation was considered necessary by 95.6% of respondents.
According to 65.1%, there was no need to shorten the interval for postoperative follow-up appointments.
Definition of weight loss failure and weight loss outcome
The most frequent response (68.6%) for defining weight loss failure after MBS was weight loss lower than 50% of excess weight. Other responses included BMI over 40 kg/m2 (14.2%) and BMI over 35 kg/m2 (11.6%). Most surgeons (44.2%), believed that %EWL was a more accurate index for reporting weight loss outcomes compared to TWL (19.8%) or EBMIL (19.8%). Almost thirty-seven percent of respondents felt that the definition of ideal body weight for patients with BMIs over 50 kg/m2 was a BMI of 30 kg/m2. Data are summarized in online Table 6.
Interval to other types of surgery
Furthermore, the top three-time intervals surgeons considered appropriate for weight stabilization so that patients with BMIs over 50 kg/m2 could undergo other types of surgery was: 12 months, 18 months and 24 months for 39.8%, 32.3%, and 21.3% of respondents, respectively.
Discussion
Patients with BMI over 50 kg/m2 are unanimously considered high-risk candidates for MBS. Indeed, surgery on these patients should be preferably performed by expert bariatric surgeons with the support of experienced bariatric anesthesiologists [16]. However, in this survey, most surgeons responded that they did not use a different surgical risk score for these patients.
A recent study revealed that BMI over 50 kg/m2 was independently associated with female sex and left ventricle hypertrophy but not with hypertension, diabetes, or a higher rate of surgical complications [17]. This may explain why 66.1% of surgeons in this survey acknowledged that BMI over 50 kg/m2 is an indication for thorough preoperative cardiac evaluation.
Interestingly, most respondents believed that it is safe and effective to perform bariatric surgery in both elderly and adolescents with BMI over 50 kg/m2 [18,19,20].
Several studies have showed that after MBS, patients with preoperative eating disorders lost significantly less weight than patients without. Preoperative assessment and interventions targeting psychosocial dysfunction could decrease eating disorder symptoms [21, 22]. This was supported in the present survey as 73.7% of respondents routinely perform preoperative eating disorder evaluation and psychological assessment.
Despite that a clear majority of respondents support preoperative weight loss for patients with BMI over 50 kg/m2, there is no consensus on how to reach this goal. Very few surgeons advised to insert an IGB and VLCDs were not routinely recommended due to bad compliance from bariatric subjects. However, a systematic review showed that a low-calorie diet (LCD, 800–1200 kcal), instead of a very-low-calorie diet (VLCDs, 450–800 kcal per day), for 2 to 4 weeks should be preferred [23]. Indeed, individuals on VLCDs may experience symptoms like fatigue, headache and nausea compromising the compliance [23], but another systematic review recommended that VLCD should be used for 4–8 weeks prior to surgery [24].
Consistent with previous publications in the literature, the respondents’ opinion of the benefits of preoperative weight loss on outcomes was inconsistent. There is not enough high quality evidence showing benefits of preoperative weight loss [25]. Further validation may need to be carried out. The results of this survey were also controversial as 39% of surgeons believe that the amount of weight loss was not important for proceeding with surgery.
Moreover, it is universally accepted that bariatric surgery in patients with BMIs over 50 kg/m2 results in less weight loss and more complications when compared to patients with BMIs less than 50 kg/m2. Therefore, for patients with obesity and BMIs > 50 kg/m2 it is very important to offer these patients procedures that are safe and effective in the long term [26,27,28]. In this view, it is interesting that most surgeons in this survey preferred to perform SG on adolescents, as also suggested by current literature [29].
Procedure selection in patients between the ages of 18 to 65 years maybe more flexible as there are many published studies demonstrating safety and efficacy in this range of ages. Most surgeons participating in this study prefer SG and OAGB. Some studies have reported that SG is a safe and effective one-stage procedure for patients with BMIs over 50 kg/m2 [30, 31]. However, other studies have reported that SG results in insufficient weight loss in these patients and should only be used as first-stage operation of a 2-stage gastric bypass or BPD/DS [32,33,34,35,36]. Thereaux et al. concluded that SG can be an independent factor for weight loss failure in patients with BMIs over 50 kg/m2 [37].
The OAGB with a tailored limb length of about 200 cm has shown to achieve good weight loss for patients with BMIs over 50 kg/m2 [38] and has shown superiority when compared to the SG [39,40,41, 42]. A systematic review by Parmar et al. reported mean %EWL of 90.75%, three years after OAGB patients with BMIs over 50 kg/m2 [43]. Additionally, Miller et al. reported %EWL of 72.2% five years after a banded OAGB in these patients [44].
Some authors have recommended RYGB as a safe and effective procedure in patients with BMI over 50 kg/m2, since it achieves better weight loss than SG [31, 45,46,47,48,49,50]. Obeid et al. reported a 52.9%EWL at 10 years after RYGB [51]. Some studies have recommended creating the RYGB with longer biliopancreatic limbs (BPL) in RYGB for patients with BMIs over 50 kg/m2 to achieve better weight loss and lower rates of weight regain [52, 53]. Only 11.1% of surgeons create their RYGB with long BPL over 150 cm for these patients. There is no consensus concerning whether a longer alimentary limb (AL) results in better weight loss in patients with BMIs over 50 kg/m2. A systematic review by Orci et al. found that RYGB with a long AL over 100 cm had better efficacy for patients with severe obesity [54], but Sarhan et al. showed that longer ALs, had no significant effect on weight loss and weight regain was noted at 3 years follow-up [55].
The BPD/DS maybe the most effective bariatric procedure for patients with BMIs over 50 kg/m2 as primary procedure [56, 57] or as conversion after primary SG [58]. However, it may have higher rates of malnutrition, diarrhea and fecal incontinence [59]. In recent studies, the SADI-S procedure has also been shown to achieve good results in patients with BMIs over 50 kg/m2 as a one [56, 60] or two-stage procedure [61,62,63]. As expected, the LAGB is the least effective procedure for patients with BMIs over 50 kg/m2 and should not be recommended as primary procedure [46, 64, 65].
Advanced age and high BMIs are risk factors for weight loss failure and weight regain after bariatric surgery [65, 66]. Additionally, elderly patients may be high risk for greater postoperative morbidity and mortality [28, 67]. SG is the most commonly performed bariatric/metabolic procedure by participating surgeons (54.7%) for elderly patients with BMIs over 50 kg/m2, as also supported by current literature [68]. Other standard procedures such as RYGB, OAGB, BPD/DS and revisional procedures have been shown to be effective but may have greater morbidity compared to younger patients [38, 67, 69, 70].
Surprisingly, only 1 out of 3 surgeons routinely measure the length of the entire small intestine during any type of gastric bypass in patients with BMI over 50 kg/m2. Abellan et al. showed that small bowel length during RYGB had no effect on weight loss in this group of patients. However, when creating longer AL, measurement of small bowel length may be necessary to assure that the common limb will be greater than 50% of total bowel length to prevent nutritional deficiencies [71]. Ahuja et al. recommended total bowel length measurement during OAGB where the BPL limbs will be 250 cm or more [40]. It seems reasonable that total bowel length should be measured for any gastric bypass with long limbs.
This survey found that 54% of surgeons perform one-stage bariatric surgery in this patient population, while 45.9% prefer a two-stage approach. Some evidence suggests that a two-stage surgical approach may decrease morbidity and mortality in patients with BMIs over 50 kg/m2 [27, 35]. SG is recommended as the first-stage procedure due to its simplicity and shorter operative time [35]. About 98.2% of surgeons participating in this study supported this approach. For the second stage, most of participants performed OAGB with tailored limb lengths. SADI-S, standard RYGB and RYGB with a long biliary limb over 150 cm were less commonly performed. Only 3.9% of surgeons would perform BPD/DS as second stage procedure for patients BMI greater than 50 kg/m2. Safety and efficacy of conversional surgeries have been confirmed by several studies [26, 27, 32, 35, 58, 61,61,63]. Moreover, as reported in previous published studies [27, 32, 35, 61, 63], there was a wide time interval between the two stages.
Robotic surgery for severely obese patients provides ergonomic advantages for the surgeon, but without any documented clinical advantages when compared to laparoscopic approaches [15, 72]. However, vast majority of respondents did not support the use of the robot for subjects with BMI superior to 50 kg/m2.
Similarly, despite single-incision laparoscopic SG was reported by Pourcher et al. [12] with acceptable outcomes and low complication rates (4.8%), participants of this survey do not recommend SILS for individuals with severe obesity.
Intraoperative liver biopsy has been a matter of contextual debate among bariatric and metabolic surgeons. In this survey, 61% of participants prefer to biopsy the liver only if micro- or macro-nodular cirrhosis is observed intraoperatively rather than routine biopsy. A systematic review of 27 studies on liver biopsy during bariatric surgery (regardless of BMI) showed that 25% of patients had NASH with a large degree of heterogeneity [73]. Bedossa et al. reported that in a cohort of 798 patients with severe obesity, older age as well as visceral adiposity may play a more relevant role in NASH than high BMI [74].
In this survey, 79% surgeons believed that ICU is not mandatory for all patients with BMI more than 50 kg/m2 and it is only necessary for selected patients. However, two studies demonstrated higher unplanned ICU admission rate among these subjects. Both studies used a national database, the American College of Surgeons Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP) Data Registry. Both studies divided patients into BMI greater than 50 kg/m2 and BMI greater than 60 kg/m2. Wilkinson et al. [75] analyzed the outcomes of over 30,000 patients with BMI over 50 kg/m2 and Nasser et al. [76] analyzed the outcomes of over 85,000 patients BMI over 50 kg/m2. Both studies confirmed that the frequency of unplanned ICU care was higher than for patients with severe obesity.
This survey found that 72.2% of surgeons favored postoperative prophylactic anticoagulation for at least 2 weeks (up to 6 weeks). This practice is supported by published data by Wilkinson et al. [75] and Nasser et al. [76], who reported higher incidences of DVT/PTE among patients with BMI superior to 50 kg/m2. In addition, Stier et al. demonstrated that patients with morbid obesity (at least those with BMI greater than 50 kg/m2) treated with routine prophylactic enoxaparin did not achieve the defined target range for aFXa [77].
More than 95% of participants in this survey believed that patients with BMI more than 50 kg/m2 need postoperative vitamin supplementation. In addition, 65% of surgeons supported shorter time intervals between follow-up visits for patients with high BMIs. Apparently, they believe that a higher degree of postoperative care is necessary for patients with BMIs greater than 50 kg/m2 as compared to patients with morbid obesity.
Predictive factors for rhabdomyolysis after bariatric surgery are: patients having a BMI superior to 40 kg/m2, patients classified as ASA III or IV physical status, diabetics and those whose operation lasts longer than 4 h [78]. Similarly, a systematic review by Chakravartty et al. showed that patients with higher BMIs, male patients and those who had prolonged surgery are at greater risk of rhabdomyolysis [79]. The above-mentioned studies have proposed measuring serum CK levels in high-risk patients [78, 79]. In addition, another cohort of 485 patients reported an increased risk of rhabdomyolysis when the duration of surgery was greater than 230 min and suggested the serum CK should be checked for all bariatric patients within the first 24 h postoperatively [80]. Considering the lack of sufficient evidence regarding CK measurements, it is not surprising that in this survey there was no consensus. 43.6% of surgeons did not measure CK routinely, while 41.4% checked CK only when the operative time exceeded 2 h.
The ASMBS guidelines advocate the use of %EWL as an item for weight loss outcomes reporting [81]. Usually the Reinhold's criteria [82], with definition of excess weight loss less than 50% or BMI > 35 for insufficient weight loss is used as the definition of weight loss failure. However, previous studies supported the use of “initial weight” and “ideal weight” as mandatory items for %EWL calculation. However, they are not clearly defined and can cause outcome variability [81]. Although “ideal weight” is also considered as BMI 25 kg/m2 by the ASMBS, another study proposed that for patients with BMI more than 50 kg/m2, BMI 25 could not reflect “ideal weight” because of wide range between initial weight and ideal weight [83]. In this survey, despite 68.8% agreement on %EWL less than 50% for weight loss failure, once again significant diversity was observed regarding ideal weight. More studies clarifying definitions for ideal weight, and initial weight are needed for patients with BMI greater than 50 kg/m2.
Some papers revealed improved outcomes of lower extremity joint replacements after bariatric surgeries [84, 85], while in contrast other reports found no significant reduction in the incidence of complications of lower extremity joint replacements after bariatric operations [86]. The interval time between the initial bariatric operation and joint replacement (knee or hip) surgery was evaluated by Schwarzkopf et al. He recommended waiting at least 6 months after bariatric surgery before proceeding with joint replacement surgeries [87]. However, respondents of this survey believed that a time interval of 12 months was sufficient.
This first survey addressing MBS on patients with BMI over 50 kg/m2 provides common ground regarding different aspects of this hot topic. First, MBS in this subgroup of patients should only be performed by expert bariatric surgeons and only experienced bariatric anesthesiologists should be involved. Second, no surgical risk scoring system is used for the moment for these patients. Third, MBS seems to be safe for all age groups of patients with a BMI over 50 kg/m2, including adolescents (under 18 years old) and elderly (over 65 years old) patients. Fourth, patients should be routinely assessed for preoperative eating and psychological disorders. Fifth, preoperative and postoperative CPAP should be used in selected cases with sleep apnea. Sixth, SG is considered by far the best choice for patients younger than 18 or older than 65 years old with BMI over 50 kg/m2. Seventh, revisional surgery or conversion for poor weight loss in these patients should only be performed after extensive preoperative evaluation. Eighth, in a two-stage MBS approach in this subgroup of patients, if resolution of comorbidities and adequate weight loss after the first procedure is achieved, then performing the second step of the two-step procedure is not recommended. Ninth, when a micro/macro-nodular liver cirrhosis is found, performing a liver biopsy is recommended. Tenth, postoperative imaging is not mandatory. Eleventh, postoperative intensive care unit admission for selected cases is recommended. Finally,
weight loss failure definition after MBS on patients with BMI over 50 kg/m2 is weight loss lower than 50% of excess weight.
Limitations and Strength of Study
IFSO currently counts more than 6500 members, and while the vast majority of them are bariatric surgeons only 11.8% (769) participated in this survey. Subsequently, a minority of members have responded to our questionnaire. However, to date, this is the largest survey addressing perioperative management on subjects with BMI over 50 kg/m2.
Conclusion
This survey is the first survey addressing the issues concerning patients with BMI over 50 kg/m2 and showed significant variations in the management of bariatric surgery for this class of obesity. An international consensus on this topic should be carried out.
References:
https://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight
Kushner RF (2018) Weight loss strategies for treatment of obesity: lifestyle management and pharmacotherapy. Prog Cardiovasc Dis 61(2):246–252
Gadde KM, Martin CK, Berthoud HR, Heymsfield SB (2018) Obesity: pathophysiology and management. J Am Coll Cardiol 71(1):69–84
Peterli R, Wölnerhanssen BK, Peters T, Vetter D, Kröll D, Borbély Y et al (2018) Effect of laparoscopic sleeve gastrectomy vs laparoscopic Roux-en-Y gastric bypass on weight loss in patients with morbid obesity: the SM-BOSS randomized clinical trial. JAMA 319(3):255–265
Kang JH, Le QA (2017) Effectiveness of bariatric surgical procedures: a systematic review and network meta-analysis of randomized controlled trials. Medicine 96(46):e8632
Schauer PR, Kashyap SR, Wolski K, Brethauer SA, Kirwan JP, Pothier CE et al (2012) Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med 366(17):1567–1576
Gagner M (2014) Decreased incidence of leaks after sleeve gastrectomy and improved treatments. Surg Obes Relat Dis 10(4):611–612
Salminen P, Helmiö M, Ovaska J, Juuti A, Leivonen M, Peromaa-Haavisto P et al (2018) Effect of laparoscopic sleeve gastrectomy vs laparoscopic Roux-en-Y gastric bypass on weight loss at 5 years among patients with morbid obesity: the SLEEVEPASS Randomized Clinical Trial. JAMA 319(3):241–254
Robert M, Espalieu P, Pelascini E, Caiazzo R, Sterkers A, Khamphommala L et al (2019) Efficacy and safety of one anastomosis gastric bypass versus Roux-en-Y gastric bypass for obesity (YOMEGA): a multicentre, randomised, open-label, non-inferiority trial. Lancet (London, England) 393(10178):1299–1309
Duvoisin C, Favre L, Allemann P, Fournier P, Demartines N, Suter M (2018) Roux-en-Y gastric bypass: ten-year results in a cohort of 658 patients. Ann Surg 268(6):1019–1025
Kakarla VR, Nandipati K, Lalla M, Castro A, Merola S (2011) Are laparoscopic bariatric procedures safe in superobese (BMI ≥50 kg/m2) patients? An NSQIP data analysis. Surg Obes Relat Dis 7(4):452–458
Pourcher G, Ferretti S, Akakpo W, Lainas P, Tranchart H, Dagher I (2016) Single-port sleeve gastrectomy for super-obese patients. Surg Obes Relat Dis 12(3):522–527
Arapis K, Macrina N, Kadouch D, Ribeiro Parenti L, Marmuse JP, Hansel B (2019) Outcomes of Roux-en-Y gastric bypass versus sleeve gastrectomy in super-super-obese patients (BMI ≥60 kg/m(2)): 6-year follow-up at a single university. Surg Obes Relat Dis 15(1):23–33
Lee Y, Dang JT, Switzer N, Malhan R, Birch DW, Karmali S (2019) Bridging interventions before bariatric surgery in patients with BMI ≥ 50 kg/m(2): a systematic review and meta-analysis. Surg Endosc 33(11):3578–3588
Pastrana M, Stoltzfus J, Claros L, El Chaar M (2020) Outcomes of robotic bariatric surgery in super-obese patients: first report based on MBSAQIP database. Surg Obes Relat Dis 16(1):71–79
Wang C, Yang W, Yang J (2014) Surgical results of laparoscopic Roux-en-Y gastric bypass in super obese patients with BMI≥60 in China. Surg Laparosc Endosc Percutan Tech 24(6):e216–e220
Brownell NK, Rodriguez-Flores M, Garcia-Garcia E, Ordoñez-Ortega S, Oseguera-Moguel J, Aguilar-Salinas CA et al (2016) Impact of body mass index >50 on cardiac structural and functional characteristics and surgical outcomes after bariatric surgery. Obes Surg 26(11):2772–2778
Dewberry L, Khoury J, Schmiege S, Jenkins T, Boles R, Inge T (2020) Gastrointestinal symptoms in relation to quality of life after metabolic surgery in adolescents. Surg Obes Relat Dis 16:554
Prasad J, Vogels E, Dove JT, Wood C, Petrick AT, Parker DM (2019) Is age a real or perceived discriminator for bariatric surgery? A long-term analysis of bariatric surgery in the elderly. Surg Obes Relat Dis 15(5):725–731
Yang W, Zhu S, Cheng Z, Zhang N, Wu L, Chen Y et al (2021) Laparoscopic Roux-en-Y gastric bypass for excess weight and diabetes: a multicenter retrospective cohort study in China. Mini-Invas Surg 5:11
Kops NL, Vivan MA, Fülber ER, Fleuri M, Fagundes J, Friedman R (2021) Preoperative binge eating and weight loss after bariatric surgery: a systematic review and meta-analysis. Obes Surg 31(3):1239–1248
Martin-Fernandez KW, Martin-Fernandez J, Marek RJ, Ben-Porath YS, Heinberg LJ (2021) Associations among psychopathology and eating disorder symptoms and behaviors in post-bariatric surgery patients. Eat Weight Disord 26:2545
Romeijn MM, Kolen AM, Holthuijsen DDB, Janssen L, Schep G, Leclercq WKG et al (2021) Effectiveness of a low-calorie diet for liver volume reduction prior to bariatric surgery: a systematic review. Obes Surg 31(1):350–356
Martínez-Ortega AJ, Olveira G, Pereira-Cunill JL, Arraiza-Irigoyen C, García-Almeida JM, Irles Rocamora JA et al (2020) Recommendations based on evidence by the andalusian group for nutrition reflection and investigation (GARIN) for the pre- and postoperative management of patients undergoing obesity surgery. Nutrients 12(7):2002
Chinaka U, Fultang J, Ali A, Rankin J, Bakhshi A (2020) Pre-specified weight loss before bariatric surgery and postoperative outcomes. Cureus 12(12):e12406
Díaz-Tobarra M, Cassinello Fernández N, Jordá Gómez P, Nofal MN, Alfonso Ballester R, Ortega SJ (2017) One-stage vs two-stage laparoscopic roux-en-Y gastric bypass in obese patients with body mass index >55 Kg/m(2); 5-year follow up. Obes Surg 27(4):955–960
Dapri G, Cadière GB, Himpens J (2011) Superobese and super-superobese patients: 2-step laparoscopic duodenal switch. Surg Obes Relat Dis 7(6):703–708
Minhem MA, Safadi BY, Habib RH, Raad EPB, Alami RS (2018) Increased adverse outcomes after laparoscopic sleeve gastrectomy in older super-obese patients: analysis of American College of Surgeons National Surgical Quality Improvement Program Database. Surg Obes Relat Dis 14(10):1463–1470
Norain A, Arafat M, Burjonrappa S (2019) Trending weight loss patterns in obese and super obese adolescents: does laparoscopic sleeve gastrectomy provide equivalent outcomes in both groups? Obes Surg 29(8):2511–2516
Ece I, Yilmaz H, Alptekin H, Yormaz S, Colak B, Yilmaz F et al (2018) Comparative effectiveness of laparoscopic sleeve gastrectomy on morbidly obese, super-obese, and super-super obese patients for the treatment of morbid obesity. Obes Surg 28(6):1484–1491
Hong J, Park S, Menzo EL, Rosenthal R (2018) Midterm outcomes of laparoscopic sleeve gastrectomy as a stand-alone procedure in super-obese patients. Surg Obes Relat Dis 14(3):297–303
Catheline JM, Fysekidis M, Dbouk R, Boschetto A, Bihan H, Reach G et al (2012) Weight loss after sleeve gastrectomy in super superobesity. J Obes 2012:959260
Lemanu DP, Singh PP, Rahman H, Hill AG, Babor R, MacCormick AD (2015) Five-year results after laparoscopic sleeve gastrectomy: a prospective study. Surg Obes Relat Dis 11(3):518–524
Noel P, Nedelcu M, Eddbali I, Manos T, Gagner M (2017) What are the long-term results 8 years after sleeve gastrectomy? Surg Obes Relat Dis 13(7):1110–1115
Alexandrou A, Felekouras E, Giannopoulos A, Tsigris C, Diamantis T (2012) What is the actual fate of super-morbid-obese patients who undergo laparoscopic sleeve gastrectomy as the first step of a two-stage weight-reduction operative strategy? Obes Surg 22(10):1623–1628
Vitiello A, Berardi G, Velotti N, De Palma GD, Musella M (2020) Should sleeve gastrectomy be considered only as a first step in super obese patients? 5-year results from a single center. Surg Laparosc Endosc Percutaneous Techn 31(2):203–207
Thereaux J, Corigliano N, Poitou C, Oppert JM, Czernichow S, Bouillot JL (2015) Comparison of results after one year between sleeve gastrectomy and gastric bypass in patients with BMI ≥ 50 kg/m2. Surg Obes Relat Dis 11(4):785–790
Kermansaravi M, DavarpanahJazi AH, ShahabiShahmiri S, Carbajo M, Vitiello A, Parmar CD et al (2021) Areas of non-consensus around one anastomosis/mini gastric bypass (OAGB/MGB): a narrative review. Obes Surg 31:2453
Singla V, Aggarwal S, Singh B, Tharun G, Katiyar V, Bhambri A (2019) Outcomes in Super obese patients undergoing one anastomosis gastric bypass or laparoscopic sleeve gastrectomy. Obes Surg 29(4):1242–1247
Ahuja A, Tantia O, Goyal G, Chaudhuri T, Khanna S, Poddar A et al (2018) MGB-OAGB: effect of biliopancreatic limb length on nutritional deficiency, weight loss, and comorbidity resolution. Obes Surg 28(11):3439–3445
Madhok B, Mahawar KK, Boyle M, Carr WR, Jennings N, Schroeder N et al (2016) Management of super-super obese patients: comparison between mini (one anastomosis) gastric bypass and sleeve gastrectomy. Obes Surg 26(7):1646–1649
Musella M, Vitiello A, Berardi G, Velotti N, Pesce M, Sarnelli G (2021) Evaluation of reflux following sleeve gastrectomy and one anastomosis gastric bypass: 1-year results from a randomized open-label controlled trial. Surg Endosc 35(12):6777–6785. https://doi.org/10.1007/s00464-020-08182-3
Parmar CD, Bryant C, Luque-de-Leon E, Peraglie C, Prasad A, Rheinwalt K et al (2019) One Anastomosis gastric bypass in morbidly obese patients with BMI ≥ 50 kg/m(2): a systematic review comparing it with Roux-En-Y gastric bypass and sleeve gastrectomy. Obes Surg 29(9):3039–3046
Miller KA, Radauer M, Buchwald JN, McGlennon TW, Ardelt-Gattinger E (2020) 5-Year results of banded one-anastomosis gastric bypass: a pilot study in super-obese patients. Obes Surg 30(11):4307–4314
Aftab H, Risstad H, Søvik TT, Bernklev T, Hewitt S, Kristinsson JA et al (2014) Five-year outcome after gastric bypass for morbid obesity in a Norwegian cohort. Surg Obes Relat Dis 10(1):71–78
Bettencourt-Silva R, Neves JS, Pedro J, Guerreiro V, Ferreira MJ, Salazar D et al (2019) Comparative effectiveness of different bariatric procedures in super morbid obesity. Obes Surg 29(1):281–291
Hariri K, Guevara D, Dong M, Kini SU, Herron DM, Fernandez-Ranvier G (2018) Is bariatric surgery effective for co-morbidity resolution in the super-obese patients? Surg Obes Relat Dis 14(9):1261–1268
Jain D, Sill A, Averbach A (2018) Do patients with higher baseline BMI have improved weight loss with Roux-en-Y gastric bypass versus sleeve gastrectomy? Surg Obes Relat Dis 14(9):1304–1309
Uno K, Seki Y, Kasama K, Wakamatsu K, Umezawa A, Yanaga K et al (2017) A comparison of the bariatric procedures that are performed in the treatment of super morbid obesity. Obes Surg 27(10):2537–2545
Zerrweck C, Sepúlveda EM, Maydón HG, Campos F, Spaventa AG, Pratti V et al (2014) Laparoscopic gastric bypass vs sleeve gastrectomy in the super obese patient: early outcomes of an observational study. Obes Surg 24(5):712–717
Obeid NR, Malick W, Concors SJ, Fielding GA, Kurian MS, Ren-Fielding CJ (2016) Long-term outcomes after Roux-en-Y gastric bypass: 10- to 13-year data. Surg Obes Relat Dis 12(1):11–20
Shah K, Nergård BJ, Fagerland MW, Gislason H (2019) Limb length in gastric bypass in super-obese patients-importance of length of total alimentary small bowel tract. Obes Surg 29(7):2012–2021
Kalfarentzos F, Skroubis G, Karamanakos S, Argentou M, Mead N, Kehagias I et al (2011) Biliopancreatic diversion with Roux-en-Y gastric bypass and long limbs: advances in surgical treatment for super-obesity. Obes Surg 21(12):1849–1858
Orci L, Chilcott M, Huber O (2011) Short versus long Roux-limb length in Roux-en-Y gastric bypass surgery for the treatment of morbid and super obesity: a systematic review of the literature. Obes Surg 21(6):797–804
Sarhan M, Choi JJ, Al Sawwaf M, Murtaza G, Getty JL, Ahmed L (2011) Is weight loss better sustained with long-limb gastric bypass in the super-obese? Obes Surg 21(9):1337–1343
Pereira AM, Guimarães M, Pereira SS, Ferreira-de-Almeida R, Monteiro MP, Nora M (2021) Single and dual anastomosis duodenal switch for obesity treatment: a single-center experience. Surg Obes Relat Dis 17(1):12–19
Rezvani M, Sucandy I, Klar A, Bonanni F, Antanavicius G (2014) Is laparoscopic single-stage biliopancreatic diversion with duodenal switch safe in super morbidly obese patients? Surg Obes Relat Dis 10(3):427–430
Merz AE, Blackstone RB, Gagner M, Torres AJ, Himpens J, Higa KD et al (2019) Duodenal switch in revisional bariatric surgery: conclusions from an expert consensus panel. Surg Obes Relat Dis 15(6):894–899
Skogar ML, Sundbom M (2017) Duodenal switch is superior to gastric bypass in patients with super obesity when evaluated with the bariatric analysis and reporting outcome system (BAROS). Obes Surg 27(9):2308–2316
Moon RC, Gaskins L, Teixeira AF, Jawad MA (2018) Safety and effectiveness of single-anastomosis duodenal switch procedure: 2-year result from a single US institution. Obes Surg 28(6):1571–1577
Zaveri H, Surve A, Cottam D, Ng PC, Enochs P, Billy H et al (2019) A multi-institutional study on the mid-term outcomes of single anastomosis duodeno-ileal bypass as a surgical revision option after sleeve gastrectomy. Obes Surg 29(10):3165–3173
de la Cruz M, Büsing M, Dukovska R, Torres AJ, Reiser M (2020) Short- to medium-term results of single-anastomosis duodeno-ileal bypass compared with one-anastomosis gastric bypass for weight recidivism after laparoscopic sleeve gastrectomy. Surg Obes Relat Dis 16(8):1060–1066
Balibrea JM, Vilallonga R, Hidalgo M, Ciudin A, González Ó, Caubet E et al (2017) Mid-term results and responsiveness predictors after two-step single-anastomosis duodeno-ileal bypass with sleeve gastrectomy. Obes Surg 27(5):1302–1308
Giet L, Baker J, Favretti F, Segato G, Super P, Singhal R et al (2018) Medium and long-term results of gastric banding: outcomes from a large private clinic in UK. BMC Obes 5:12
Robert M, Poncet G, Boulez J, Mion F, Espalieu P (2011) Laparoscopic gastric bypass for failure of adjustable gastric banding: a review of 85 cases. Obes Surg 21(10):1513–1519
Lunel T, Iceta S, Pasquer A, Pelascini E, Perinel J, Poncet G et al (2021) Third bariatric procedure for insufficient weight loss or weight regain: how far should we go? Surg Obes Relat Dis 17(1):96–103
Daigle CR, Andalib A, Corcelles R, Cetin D, Schauer PR, Brethauer SA (2016) Bariatric and metabolic outcomes in the super-obese elderly. Surg obes Relat Dis 12(1):132–137
Gil-Rendo A, Muñoz-Rodríguez JR, Domper Bardají F, Menchén Trujillo B, Martínez-de Paz F, Caro González MDP et al (2019) Laparoscopic sleeve gastrectomy for high-risk patients in a monocentric series: long-term outcomes and predictors of success. Obes Surg 29(11):3629–3637
McGlone ER, Bond A, Reddy M, Khan OA, Wan AC (2015) Super-Obesity in the elderly: is bariatric surgery justified? Obes Surg 25(9):1750–1755
Elbahrawy A, Bougie A, Loiselle SE, Demyttenaere S, Court O, Andalib A (2018) Medium to long-term outcomes of bariatric surgery in older adults with super obesity. Surg Obes Relat Dis 14(4):470–476
Abellan I, Luján J, Frutos MD, Abrisqueta J, Hernández Q, López V et al (2014) The influence of the percentage of the common limb in weight loss and nutritional alterations after laparoscopic gastric bypass. Surg Obes Relat Dis 10(5):829–833
Buchs NC, Pugin F, Chassot G, Volonte F, Koutny-Fong P, Hagen ME et al (2013) Robot-assisted Roux-en-Y gastric bypass for super obese patients: a comparative study. Obes Surg 23(3):353–357
Barbois S, Arvieux C, Leroy V, Reche F, Sturm N, Borel AL (2017) Benefit-risk of intraoperative liver biopsy during bariatric surgery: review and perspectives. Surg Obes Relat Dis 13(10):1780–1786
Bedossa P, Tordjman J, Aron-Wisnewsky J, Poitou C, Oppert JM, Torcivia A et al (2017) Systematic review of bariatric surgery liver biopsies clarifies the natural history of liver disease in patients with severe obesity. Gut 66(9):1688–1696
Wilkinson KH, Helm M, Lak K, Higgins RM, Gould JC, Kindel TL (2019) The risk of post-operative complications in super-super obesity compared to super obesity in accredited bariatric surgery centers. Obes Surg 29(9):2964–2971
Nasser H, Ivanics T, Leonard-Murali S, Shakaroun D, Genaw J (2019) Perioperative outcomes of laparoscopic Roux-en-Y gastric bypass and sleeve gastrectomy in super-obese and super-super-obese patients: a national database analysis. Surg Obes Relat Dis 15(10):1696–1703
Stier C, Koschker AC, Stier R, Sosnierz A, Chiappetta S (2020) Are we missing treatment standards for thromboprophylaxis of the obese and super-obese patient population? A prospective systematic cohort study. Obes Surg 30(5):1704–1711
Lagandre S, Arnalsteen L, Vallet B, Robin E, Jany T, Onraed B et al (2006) Predictive factors for rhabdomyolysis after bariatric surgery. Obes Surg 16(10):1365–1370
Chakravartty S, Sarma DR, Patel AG (2013) Rhabdomyolysis in bariatric surgery: a systematic review. Obes Surg 23(8):1333–1340
Tolone S, Pilone V, Musella M, Rossetti G, Milone M, Fei L et al (2016) Rhabdomyolysis after bariatric surgery: a multicenter, prospective study on incidence, risk factors, and therapeutic strategy in a cohort from South Italy. Surg Obes Relat Dis 12(2):384–390
Brethauer SA, Kim J, el Chaar M, Papasavas P, Eisenberg D, Rogers A et al (2015) Standardized outcomes reporting in metabolic and bariatric surgery. Surg Obes Relat Dis 11(3):489–506
Reinhold RB (1982) Critical analysis of long term weight loss following gastric bypass. Surg Gynecol Obstet 155(3):385–394
Baltasar A, Perez N, Serra C, Bou R, Bengochea M, Borras F (2011) Weight loss reporting: predicted body mass index after bariatric surgery. Obes Surg 21(3):367–372
McLawhorn AS, Levack AE, Lee YY, Ge Y, Do H, Dodwell ER (2018) Bariatric surgery improves outcomes after lower extremity arthroplasty in the morbidly obese: a propensity score-matched analysis of a New York statewide database. J Arthroplast 33(7):2062–9.e4
Li S, Luo X, Sun H, Wang K, Zhang K, Sun X (2019) Does prior bariatric surgery improve outcomes following total joint arthroplasty in the morbidly obese? A meta-analysis. J Arthroplast 34(3):577–585
Smith TO, Aboelmagd T, Hing CB, MacGregor A (2016) Does bariatric surgery prior to total hip or knee arthroplasty reduce post-operative complications and improve clinical outcomes for obese patients? Systematic review and meta-analysis. Bone Jt J 98(9):1160–1166
Schwarzkopf R, Lavery JA, Hooper J, Parikh M, Gold HT (2018) Bariatric surgery and time to total joint arthroplasty: does it affect readmission and complication rates? Obes Surg 28(5):1395–1401
Acknowledgements
All authors disclose the absence of any financial and personal relationships with other people or organizations that could inappropriately influence (bias) their work except for Mr. Kamal Mahawar (see below). Kamal Mahawar has received honoraria from Ethicon, Medtronic, Olympus, Gore, and various NHS Trusts for educational activities related to bariatric surgery.
Funding
No funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Mohammad Kermansaravi, Panagiotis Lainas, Shahab Shahabi Shahmiri, Wah Yang, Amirhossein Davarpanah Jazi, Ramon Villalonga, Luciano Antozzi, Chetan Parmar, Radwan Kassir, Sonja Chiappetta, Lorea Zubiaga, Antonio Vitiello, Kamal Mahawar, Miguel Carbajo, Mario Musella, and Scott Shikora declare that they have no conflict of interest.
Informed consent
All surgeons participated to the survey voluntarily and gave informed consent.
Research involving human and animal rights
This is a survey among surgeons, and thus, no human or animal was included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kermansaravi, M., Lainas, P., Shahmiri, S.S. et al. The first survey addressing patients with BMI over 50: a survey of 789 bariatric surgeons. Surg Endosc 36, 6170–6180 (2022). https://doi.org/10.1007/s00464-021-08979-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-021-08979-w