Abstract
Purpose
The use of electronic patient-reported outcome (ePRO) data in routine care has been tied to direct patient benefits such as improved quality of care and symptom control and even overall survival. The modes of action behind such benefits are seldom described in detail. Here, we describe the development of a model of care leveraging ePRO data to monitor and manage symptoms of patients treated with immune checkpoint inhibitors.
Methods
Development was split into four stages: (1) identification of an underlying theoretical framework, (2) the selection of an ePRO measure (ePROM), (3) the adaptation of an electronic application to collect ePRO data, and (4) the description of an ePRO-oriented workflow. The model of care is currently evaluated in a bicentric longitudinal randomized controlled phase II trial, the IePRO study.
Results
The IePRO model of care is grounded in the eHealth Enhanced Chronic Care Model. Patients are prompted to report symptoms using an electronic mobile application. Triage nurses are alerted, review the reported symptoms, and contact patients in case of a new or worsening symptom. Nurses use the UKONS 24-hour telephone triage tool to issue patient management recommendations to the oncology team. Adapted care coordinating procedures facilitate team collaboration and provide patients with timely feedback.
Conclusion
This report clarifies how components of care are created and modified to leverage ePRO to enhance care. The model describes a workflow that enables care teams to be proactive and provide patients with timely, multidisciplinary support to manage symptoms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Immune checkpoint inhibitors (ICI) have become part of the standard of treatment for an expanding range of cancer types [1]. Despite having shown a lower toxicity profile compared to other treatments, immune-related adverse events (IrAE) caused by ICI can nevertheless be severe and potentially fatal [2, 3]. The likelihood of experiencing an IrAE is influenced by treatment modality: between 40 and 75% of patients treated with a single ICI experience an IrAE (any grade), with 10 to 30% experiencing severe events (grade ≥ 3) [3, 4]. About 95% of patients experience at least one IrAE when treated with combined ICI, and nearly 60% of patients experience at least one severe IrAE [5].
These IrAE are notably heterogeneous, occasionally resembling disease progression and mimicking auto-immune conditions [4]. Severe IrAE can be persistent or occur several months into and beyond treatment [6,7,8], thus adding on to the already considerable acute and chronic symptom burden patients experience.
Patient education and symptom self-management, particularly self-monitoring, contribute to more timely detection of IrAE, better short-term outcomes for patients, and lower incidence of chronic symptoms [9, 10]. However, patients treated with ICI may not be sufficiently supported in that domain [11, 12]. Mild symptoms are often under-recognized and under-reported by patients and clinicians, though they may be indicative of more serious developing conditions impacting quality of life [13, 14]. Close and frequent communication between patients and healthcare providers is thus essential in preventing severe IrAE. Information flyers and telephone follow-up targeting symptoms related to ICI treatment have been used to support patients and anticipate the delivery of care [15]. However, evidence-based procedures to monitor and manage them in a real-world setting are still lacking [16, 17].
The use of patient-reported outcome measures (PROM) has been shown to improve symptom detection, monitoring, and management by empowering patients to convey their perception of symptoms to healthcare providers, while also providing valuable treatment safety and tolerability data [12, 13, 18, 19]. Electronic PROM (ePROM) can play a role in shared clinical decision support by influencing treatment decisions and improving the scope and efficiency of patient-provider communication [20,21,22]. Remote real-time symptom reporting and monitoring facilitated by the use of ePROM may lead to more accurate insights into patients’ health status than delayed self-reports [23].
Studies involving the use of electronic PRO (ePRO) data in oncology reported a decrease in hospitalization rates and emergency department visits, with favorable outcomes on quality of life, perceived self-efficacy, and overall survival [13, 24]. How these studies’ interventions mobilized and interacted with existing care structures and procedures to produce beneficial outcomes is seldom described in detail [25]. Some interventions used ePRO to assess symptoms remotely as complementary clinical decision support to modify treatment or to refer patients to emergency or acute care services, among others [13]. To our knowledge, no studies targeting the remote management of symptoms of patients treated with ICI have detailed the conception and integration of ePRO-based care models, within existing care delivery structures.
In this report, we describe the development of a model of care that leverages ePRO data to monitor and manage symptoms in patients treated with ICI, in an outpatient care setting. This model is currently being tested in a randomized controlled phase II trial, the IePRO trial, at two Swiss university hospitals (ClinicalTrials.gov Identifier: NCT05530187).
Toward the development of an ePRO-based model of care
Development of the IePRO model of care took place between November 2020 and November 2021. A team of four physicians and five nurses of the participating institutions’ oncology departments and one patient-representative collaborated in the creation of its core components and their integration in the existing workflows of each hospital. All members had previous experience in collecting and interpreting PRO data in clinical oncology trials. Two nurses have published research on PROM aimed toward patients treated with ICI [26]. The patient representative was identified by screening Swiss and French patient advocacy groups related to oncology. A brief in-person interview allowed to assess their knowledge of ICI and their side effects, expertise in using PROs, and experience in collaborating in clinical trials.
This ePRO-based model of care was developed in four stages: (1) identification of an underlying theoretical framework, (2) selection of an ePROM, (3) adaptation of an electronic mobile application to collect ePRO data, and (4) ePRO-oriented workflow and clinical roles.
Theoretical framework
As ICI-related symptoms may add to the symptom burden of patients, effective management of these symptoms requires a holistic approach. To reflect upon and address the complexity and resources required for symptom management, we grounded the development of this intervention in the eHealth Enhanced Chronic Care Model (eCCM) [27], which is itself an extension of the Chronic Care Model (CCM) [28, 29].
The major components of the CCM, community resources and health systems, are complemented by eCommunity and eHealth in the eCCM [27]. eHealth includes the digital tools and resources available to patients that complement those provided by the healthcare system. Online communities and health-related social networks constitute the eCommunity, which supports patient engagement and activation for self-management.
The major components of the eCCM encapsulate five smaller interdependent components: self-management support (SMS), clinical decision support (CDS), delivery system design (DSD), clinical information systems (CIS), and eHealth education (eHE). These are brought together to ensure informed and activated patients interact with prepared and proactive practice teams, leading to satisfying encounters and improved outcomes [27]. They are described in further detail in Fig. 1.
The eHealth Enhanced Chronic Care Model (eCCM), adapted from: Gee PM, Greenwood DA, Paterniti DA, Ward D, Miller LMS. The eHealth Enhanced Chronic Care Model: A Theory Derivation Approach. Journal of Medical Internet Research 2015;17:e86. https://doi.org/10.2196/jmir.4067. The original is licensed under a Creative Commons Attribution license (CC-BY)
We address each of these smaller components and clarify their role in achieving productive interactions between patients and care providers as we describe the following development phases of the IePRO model.
Selection of an ePROM
Active discussions between the model development team allowed to identify an ePROM of particular interest, to both clinicians and patients. The patient-representative mobilized her patient-advocacy network to collect and convey general perceptions on existing PROM, such as their perceived advantages and disadvantages to assess symptomatic ICI-related toxicity, via e-mail. The PRO version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE™) item library was considered comprehensive and suitably flexible, measuring a broad spectrum of symptoms [26, 30]. Using the results of a previous Delphi study, we identified a set of 37 priority PRO-CTCAE™ items for routine symptom monitoring in this patient population, which compose the IePRO trial’s weekly symptom questionnaire [31].
Patients participate in the IePRO trial for the first six months of their ICI treatment. Because the majority of IrAE occur within the first three to four months of treatment [7], active symptoms are re-assessed daily for the first three months, using a modified recall period of 24 hours, between weekly questionnaires. In addition, patients can add any of the 80 PRO-CTCAE™ items to the daily and weekly assessments.
Adaptation of an electronic mobile application
The main goal in using an ePRO application is to enhance self-management support (SMS). As an eCCM component, SMS includes the provision of tools and resources for patients to acquire the skills and confidence to manage and monitor their health condition [32]. We adapted an application developed by Kaiku Health Ltd., where the developed ePROM was integrated. Studies using similar iteration versions of the Kaiku Health app have reported high agreement across patients and providers on its ease of use and high levels of satisfaction and relevance for clinical practice [33, 34].
The application sends patient reminders to fill out the ePROM at the previously mentioned time points, to facilitate data collection [35]. It displays all previous replies to any questionnaire, facilitating self-care and self-monitoring tasks [35]. In addition, at the end of each symptom questionnaire, a summary portraying symptom evolution is displayed (Fig. 2).
Since these features may increase symptom awareness, guidance to perceive their detection as empowering to manage and prevent complications is required, as they can also be perceived as signs of deterioration or disease progression, decreasing perceived self-efficacy [36].
To enable patients to navigate the complete item bank of the PRO-CTCAE™, a symptom selection screen was developed in collaboration with patients from the oncology department and the patient-representative, through a card-sorting exercise. Results were used to adapt the screen presented to patients allowing adding symptoms to be monitored.
Integration of ePRO data into clinical information systems (CIS) like the patient’s electronic health record (EHR) is a desired outcome, as it can decrease the technological burden and enhance accessibility of data [13, 32, 37]. The IePRO trial is conducted in two university hospitals operating different EHR platforms. An initial assessment for readiness to implement PRO data concluded that the CIS could not be modified to directly integrate ePRO data in similar ways. Nurses are thus prompted to access the application directly via e-mail when patients report new symptoms.
Development of an ePRO-oriented workflow and clinical roles
In the eCCM, delivery system design (DSD) relates to how care is coordinated and delivered across the network of health resources. The participating oncology departments treat a similar range of tumor types and number of patients, with similar provider team compositions. Physicians and nurses involved in direct patient care revealed service-level and provider-level barriers such as the time required to navigate, collect, and process PRO data, the integration or lack thereof within the EHR, and internal communication pathways to ensure the continuity of care [38, 39]. These barriers were included in the development of the model of care.
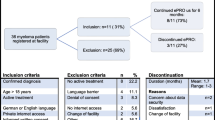
We hereafter describe how patients are engaged, the triage process and the triage nurse role, and the nurse-physician coordination to provide care. An overview of the model is featured in Fig. 3, and components of the eCCM represented in the IePRO model are summarized in Table 1.
Overview of the IePRO model of care: patients perform self-assessment (1) and declare potential symptoms using the symptom ePROM in the electronic application (2). Telephone triage nurses review PRO data and coordinate with the oncology team preemptively when necessary (3) and contact patients by telephone using a standardized triage process (4)
Patient engagement
As in the eCCM, informed and activated patients are key to create productive interactions with the healthcare providers [27]. Patients receive information on treatment side effects from clinical nurse specialists (CNS), physicians, and nurses. Triage nurses present the electronic application to the patient, provide a setup guide (online supplement A), and assist in its configuration.
Patients fill out the 37-item ePROM within the first week of ICI treatment by logging in to the online or mobile (smartphone) version of the application. They are prompted to complete subsequent daily and/or weekly questionnaires via an e-mail reminder or push notifications. Patients are made aware their answers in the ePROM will be reviewed by a team of triage nurses on weekdays between 8 and 12 pm. As part of the standard of care, patients are nevertheless encouraged to contact their oncology team directly in case any of any symptoms self-perceived as a cause of immediate concern.
Telephone triage nurses and triage process
Telephone triage nurses are the main vector of communication between the patient and the clinical oncology team in the IePRO model. This role was developed and reviewed with oncology physicians, nurses, and CNS. For some oncology subspecialties, the CNS provide sporadic telephone consultations for the most vulnerable patients; therefore, clarifying the role of triage nurses was essential to avoid confusion among providers and patients. While triage nurses work as gatekeepers, helping patients access and appropriate level of care, CNS are a resource to ensure evidence-based symptom management and provide highly specialized care.
Triage nurses were trained to use the United Kingdom Oncology Nursing Society (UKONS) 24-hour triage tool [40]. It was translated and validated in French, in collaboration with the UKONS, for use in the IePRO trial. Two members of the nursing team in one hospital received online training directly from UKONS, who trained the remaining three nurses.
The tool standardizes remote symptom assessment and provides clear guidance on remote symptom management. Triage procedures are triggered when triage nurses detect a new or worsening symptom in the ePRO application. The triage algorithm outputs three types of alerts according to symptom severity: (1) green alerts are issued for mild and stable symptoms where self-management support is recommended, (2) amber alerts represent symptoms that may increase or decrease in severity and thus require a new assessment within 24 hours, and (3) red alerts are issued when symptoms are moderate-to-severe, and in-person assessment is recommended. Nurses log triage procedures in the EHR using an electronic version of the tool’s triage log form. Since CIS integration was not possible, triage nurses alert physicians, CNS, and nurse practitioners of triaged symptoms and of their recommendations by sending a daily summary of all calls.
In the event of a green alert, triage nurses provide self-care guidance, and the oncology team is notified via the e-mail summary. When an amber alert is issued, the oncology team is immediately contacted via e-mail to validate the triage nurses’ assessment and determine if any additional care should be provided. More than one amber alert or at least one red alert triggers triage nurses to call the patient’s oncology physician to seek their specific recommendations and call the patient back to convey the latter. As this model of care is complementary to the standard of care, outside of the triage nurses’ operating schedule, standard procedures apply.
As part of the eHealth education (eHE) component in the IePRO model, triage nurses were trained extensively with the ePROM application between April and November 2021. It presents nurses with a visual and numerical representation of the reported symptoms (Fig. 4) that reflect a combination of PRO-CTCAE™ attributes (frequency, severity, interference, amount, and presence/absence). As outlined in the eCCM, access to this type of remote patient-reported symptom data enables the care team to be proactive and prepared for triage calls in advance [27].
Role of physicians and other healthcare professionals
Physicians are the primary collaborators with the triage nurses and are responsible for reviewing triage reports. When their assessment differs from the nurse’s, the physician is to contact them and the patient to provide their recommendation. Triage nurses and physicians may also forward requests to other professionals, such as psycho-oncologists and physiotherapists. An automated e-mail reminder to follow-up on and assess previously reported symptoms is sent in the morning of each in-person patient visit.
Assessing usability of the ePRO application and acceptability of the model of care
Assessment of the usability of the ePRO application and the acceptability of the model of care from the patient’s perspective takes place up to two weeks after study discontinuation. The mobile application’s usability and the model of care’s acceptability are assessed through semi-structured interviews with patients. Based on the mHealth app usability questionnaire (MAUQ) by Zhou et al. [41], interview guides have been developed. Items were grouped by their scope and nine open-ended questions were formulated by the research team, available as an online supplement (Supplement B).
A semi-structured patient interview guide to assess the acceptability of the model of care was created by the research team, using the definition of acceptability by Sekhon et al. [42]. Questions were derived from the seven constructs of acceptability: “affective attitude,” “burden,” “ethicality,” “intervention coherence,” “opportunity costs,” “perceived effectiveness,” and “self-efficacy” (Supplement C).
To assess the intervention’s acceptability from the healthcare provider’s perspective, an interview guide was developed based on the Consolidated Framework for Implementation Research (CFIR) [43]. It includes questions addressing the model of care’s characteristics, the outer and inner settings, the characteristics of the individuals using the model, and the process of implementation (Supplement D). Acceptability of the model of care will be assessed up to two weeks after the end of the trial.
Discussion
The IePRO model of care supports the detection and timely management of symptoms of patients treated with ICI. It represents a pragmatic research approach to the use of ePRO data in the context of two university hospitals that retain minor differences in resources and infrastructure, standard operating procedures, and care culture. It describes workflow changes that exist in parallel to usual care, complementing clinical activity and outlining a closed feedback loop between patients and care providers based on electronic monitoring of PRO data.
We consider this model of care to have notable strengths. Due to the potential of symptomatic IrAEs to become chronic conditions, there is a need for forward-looking transformations in care delivery that focus on both short and long-term care [8]. The model ensures that pre-existing and new symptoms are equally taken into account and that the full range of resources is mobilized to manage them. Contrasting with other trials using PRO-CTCAE™ items [44, 45], it accommodates the use of the full item library, lending itself to the heterogeneous toxicity of ICI. Alternating weekly fixed-length and adaptive daily questionnaires enables the detection of quick and sudden fluctuations in symptom severity, while potentially minimizing patient burden. Guided by the eCCM, the model aligns with recommendations from previous studies and with recent guidelines for implementing PRO in routine care, despite preceding them [13, 25, 46, 47]. As part of a clinical trial, some of the eCCM’s components were not developed in this iteration, namely, the community and eCommunity. Integration of these components in the future should be considered to broaden the support for patient self-management.
The model ensures patients receive tailored feedback every weekday they complete a questionnaire, without the requirement of a hospital visit. This closed feedback loop attempts to value the time patients invest in symptom reporting and encourage patients to continue self-monitoring.
Some challenges relating to future implementation, patient engagement, the triage nurse role, and the clinical and technological burden remain. There are no CNS and nurse practitioners available in one of the sites, and thus, the triage nurses are likely to more often strictly rely on physician collaboration to manage symptoms. In the same site, physician teams are less differentiated across tumor types, which may simplify the flow of information with nurses. E-mail reports for mild to moderate symptoms may not facilitate as timely of an intervention as direct telephone or face-to-face contact. However, the care teams agreed it would be the most effective way to request multidisciplinary support and update all relevant parties on patient status. This may increase the burden on triage nurses to obtain a timely reply. An integrated system in the EHR could potentially save time and provide a clearer transfer of responsibility across the oncology team.
The allocation of dedicated resources is recommended for successful implementation of PRO data in routine care, as there is the possibility of increased clinical burden [21, 24, 48]. Training in interpreting PRO data was focused on triage nurses, as time and technical constraints prevented deeper integration with the broader oncology team. Universal access to ePRO data would decrease friction, despite being more resource-intensive in its initial deployment [48]. Currently, triage nurses require more time to process data and create an accessible output for the oncology team. There is a clear risk of incomplete or inaccurate information between the triage reports and the self-reported patient data, which constitutes the most significant limitations of this model. Our preliminary experiences in the IePRO trial suggest clear benefits in training all providers to use PRO data and in integrating it directly in the EHR to minimize the technological burden. Weekly meetings between the nursing triage staff and the PIs of the IePRO trial, who are involved in direct patient care, facilitate discussions on matters related to the workflow and patient and provider burden. These include optimizing how pending issues can be handled more efficiently and derive consensus on how to manage unanticipated situations.
Features of electronic applications clearly play a role in patient engagement and compliance, with integrated communication with care providers and other patients being among the most desirable functionalities, which is included in the Kaiku Health app [35]. The development team considered patients could feel compelled to use the messaging service instead of contacting the medical team via telephone. Given the limited activity period of the nursing triage team, there was considerable risk that some messages would not be addressed in a timely manner, prompting the decision to deactivate this functionality. To accommodate those features in the IePRO model, the flow of communication between patient and providers would need to be revised. The impact on the burden of clinical teams would also need to be considered, as it may result in more frequent prompts to intervene than a system where the decision to initiate contact lies with the provider. Other eHealth interventions have used automated written feedback [35], which could be integrated in this model as well. Ongoing data collection from patient interviews may highlight the strengths and limitations of the application in its current version. Patient feedback will be addressed in future publications.
Data collection concerning the acceptability of the model of care from the provider’s perspective will take place after the trial and will be analyzed and disseminated in a later stage of the project. It is unclear how patients will perceive the novel role of the triage nurse and how it may interfere in their relationship with other providers like the CNS. International guidelines for managing IrAE often require skills, such as prescribing medication and diagnostic tests that most nurses in Switzerland cannot autonomously enact. While close collaboration with physicians in symptom management is essential, the lack of autonomy increases the complexity of the workflow and introduces additional points of failure. Further standardization of practice and continued investment in advanced nursing practice roles may further optimize care delivery and improve the model. Because IrAE management guidelines do not primarily focus on self-management support, some variability in what interventions are put in place by triage nurses is likely. More comprehensive self-management support coverage in those guidelines would empower nurses and patients and further clarify how beneficial outcomes can be achieved [17].
The development of this model benefited from the collaboration with a patient-representative to assess the tools and PROM used in its different components. This triggered deeper discussions with the care team, relating to symptom management and administrative challenges. As patients’ acceptability of the model of care is assessed, we believe future iterations also stand to gain significantly from deeper patient and public involvement.
Conclusion
The described based model of care provides insight into the complexity of using ePRO data to facilitate potential benefits for both patients and care providers. It attempts to draw a closed feedback loop between patients and providers, to ensure symptoms related to ICI treatments and beyond are monitored and managed by a proactive, prepared provider team.
The IePRO model is not intended as a blueprint for other institutions with that goal. Rather, it is an example of the complexity of such an endeavor, by reworking several components of care in the attempt to generate beneficial outcomes to patients. Under that light, we believe it furthers the discussion around PRO implementation by exposing some of the pragmatic difficulties and compromises that researcher and clinicians may have to manage.
Data availability
Not applicable.
Abbreviations
- CCM :
-
chronic care model
- CDS :
-
clinical decision support
- CIS :
-
clinical information systems
- DSD :
-
delivery system design
- eCCM :
-
eHealth Enhanced Chronic Care Model
- eHE :
-
eHealth education
- ePRO :
-
electronic patient-reported outcomes
- ePROM :
-
electronic patient-reported outcome measures
- ICI :
-
immune checkpoint inhibitors
- IrAE :
-
immune-related adverse events
- MAUQ :
-
mHealth app usability questionnaire
- PRO :
-
patient-reported outcomes
- PRO-CTCAE™ :
-
Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events
- PROM :
-
patient-reported outcome measures
- SMS :
-
self-management support
References
Vaddepally RK, Kharel P, Pandey R, Garje R, Chandra AB (2020) Review of indications of FDA-approved immune checkpoint inhibitors per NCCN guidelines with the level of evidence. Cancers 12:738. https://doi.org/10.3390/cancers12030738
Schneider BJ, Naidoo J, Santomasso BD, Lacchetti C, Adkins S, Anadkat M et al (2021) Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO guideline update. J Clin Oncol 39:4073–4126. https://doi.org/10.1200/JCO.21.01440
Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chávez A, Keegan N, Khamashta MA et al (2020) Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Primer 6:38. https://doi.org/10.1038/s41572-020-0160-6
Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J, et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology 2017;28:iv119–42. https://doi.org/10.1093/annonc/mdx225
Wolchok JD, Chiarion-Sileni V, Gonzalez R, Rutkowski P, Grob J-J, Cowey CL et al (2017) Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med 377:1345–1356. https://doi.org/10.1056/NEJMoa1709684
Owen CN, Bai X, Quah T, Lo SN, Allayous C, Callaghan S et al (2021) Delayed immune-related adverse events with anti-PD-1-based immunotherapy in melanoma. Ann Oncol 32:917–925. https://doi.org/10.1016/j.annonc.2021.03.204
Tang S-Q, Tang L-L, Mao Y-P, Li W-F, Chen L, Zhang Y et al (2021) The pattern of time to onset and resolution of immune-related adverse events caused by immune checkpoint inhibitors in cancer: a pooled analysis of 23 clinical trials and 8,436 patients. Cancer Res Treat 53:339–354. https://doi.org/10.4143/crt.2020.790
Johnson DB, Nebhan CA, Moslehi JJ, Balko JM (2022) Immune-checkpoint inhibitors: long-term implications of toxicity. Nat Rev Clin Oncol 19:254–267. https://doi.org/10.1038/s41571-022-00600-w
Zhang L, Zlotoff DA, Awadalla M, Mahmood SS, Nohria A, Hassan MZO et al (2020) Major adverse cardiovascular events and the timing and dose of corticosteroids in immune checkpoint inhibitor–associated myocarditis. Circulation 141:2031–2034. https://doi.org/10.1161/CIRCULATIONAHA.119.044703
Plaçais L, Michot J-M, Champiat S, Romano-Martin P, Baldini C, Joao MS et al (2021) Neurological complications induced by immune checkpoint inhibitors: a comprehensive descriptive case-series unravelling high risk of long-term sequelae. Brain Commun 3:fcab220. https://doi.org/10.1093/braincomms/fcab220
Champiat S, Lambotte O, Barreau E, Belkhir R, Berdelou A, Carbonnel F et al (2016) Management of immune checkpoint blockade dysimmune toxicities: a collaborative position paper. Ann Oncol 27:559–574. https://doi.org/10.1093/annonc/mdv623
Naing A, Hajjar J, Gulley JL, Atkins MB, Ciliberto G, Meric-Bernstam F et al (2020) Strategies for improving the management of immune-related adverse events. J Immunother Cancer 8:e001754. https://doi.org/10.1136/jitc-2020-001754
Di Maio M, Basch E, Denis F, Fallowfield LJ, Ganz PA, Howell D et al (2022) The role of patient-reported outcome measures in the continuum of cancer clinical care: ESMO clinical practice guideline. Ann Oncol:S0923753422006913. https://doi.org/10.1016/j.annonc.2022.04.007
Brahmer JR, Lacchetti C, Schneider BJ, Atkins MB, Brassil KJ, Caterino JM et al (2018) Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 36:1714–1768. https://doi.org/10.1200/JCO.2017.77.6385
Hoffner MSN, ANP-BC, AOCNP B, M. Rubin, MS, FNP-BC K. (2019) Meeting the challenge of immune-related adverse events with optimized telephone triage and dedicated oncology acute care. J Adv Pract Oncol:10. https://doi.org/10.6004/jadpro.2019.10.2.10
Martins F, Sofiya L, Sykiotis GP, Lamine F, Maillard M, Fraga M et al (2019) Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat Rev Clin Oncol. https://doi.org/10.1038/s41571-019-0218-0
Cheema PK, Iafolla MAJ, Nematollahi M, Berco F, Kaushik D, Matthews P et al (2022) A guide to implementing immune checkpoint inhibitors within a cancer program: experience from a large Canadian community centre. Curr Oncol 29:869–880. https://doi.org/10.3390/curroncol29020074
Mendoza T (2018) Understanding the toxicity of cancer immunotherapies: use of patient-reported outcomes. J Immunother Precis Oncol 1:38. https://doi.org/10.4103/JIPO.JIPO_10_18
Zhang L, Zhang X, Shen L, Zhu D, Ma S, Cong L (2022) Efficiency of electronic health record assessment of patient-reported outcomes after cancer immunotherapy: a randomized clinical trial. JAMA Netw Open 5:e224427. https://doi.org/10.1001/jamanetworkopen.2022.4427
Field J, Holmes MM, Newell D (2019) PROMs data: can it be used to make decisions for individual patients? A narrative review. Patient Relat Outcome Meas 10:233–241. https://doi.org/10.2147/PROM.S156291
Basch E, Stover AM, Schrag D, Chung A, Jansen J, Henson S et al (2020) Clinical utility and user perceptions of a digital system for electronic patient-reported symptom monitoring during routine cancer care: findings from the PRO-TECT trial. JCO Clin Cancer Inform:947–957. https://doi.org/10.1200/CCI.20.00081
Absolom K, Gibson A, Velikova G (2019) Engaging patients and clinicians in online reporting of adverse effects during chemotherapy for cancer: the eRAPID system electronic patient self-reporting of adverse events. Med Care 57:S59–S65. https://doi.org/10.1097/MLR.0000000000001085
Coolbrandt A, Van den Heede K, Vanhove E, De Bom A, Milisen K, Wildiers H (2011) Immediate versus delayed self-reporting of symptoms and side effects during chemotherapy: does timing matter? Eur J Oncol Nurs 15:130–136. https://doi.org/10.1016/j.ejon.2010.06.010
Kotronoulas G, Kearney N, Maguire R, Harrow A, Di Domenico D, Croy S et al (2014) What is the value of the routine use of patient-reported outcome measures toward improvement of patient outcomes, processes of care, and health service outcomes in cancer care? A systematic review of controlled trials. J Clin Oncol 32:1480–1501. https://doi.org/10.1200/JCO.2013.53.5948
Leppla L, Schmid A, Valenta S, Mielke J, Beckmann S, Ribaut J et al (2021) Development of an integrated model of care for allogeneic stem cell transplantation facilitated by eHealth—the SMILe study. Support Care Cancer 29:8045–8057. https://doi.org/10.1007/s00520-021-06328-0
Colomer-Lahiguera S, Bryant-Lukosius D, Rietkoetter S, Martelli L, Ribi K, Fitzpatrick-Lewis D et al (2020) Patient-reported outcome instruments used in immune-checkpoint inhibitor clinical trials in oncology: a systematic review. J Patient-Rep Outcomes:4. https://doi.org/10.1186/s41687-020-00210-z
Gee PM, Greenwood DA, Paterniti DA, Ward D, Miller LMS (2015) The eHealth Enhanced Chronic Care Model: a theory derivation approach. J Med Internet Res 17:e86. https://doi.org/10.2196/jmir.4067
Bodenheimer T, Wagner EH, Grumbach K. Improving primary care for patients with chronic illness. JAMA 2002;288:1775–9. https://doi.org/10.1001/jama.288.14.1775.
Bodenheimer T, Wagner EH, Grumbach K (2002) Improving primary care for patients with chronic illness - the chronic care model, part 2. JAMA 288:1909–1914. https://doi.org/10.1001/jama.288.15.1909
Basch E, Reeve BB, Mitchell SA, Clauser SB, Minasian LM, Dueck AC et al (2014) Development of the National Cancer Institute’s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). JNCI. J Natl Cancer Inst 106:dju244–dju244. https://doi.org/10.1093/jnci/dju244
Da Silva Lopes AM, Colomer-Lahiguera S, Mederos Alfonso N, Aedo-Lopez V, Spurrier-Bernard G, Tolstrup LK et al (2021) Patient-reported outcomes for monitoring symptomatic toxicities in cancer patients treated with immune-checkpoint inhibitors: a Delphi study. Eur J Cancer 157:225–237. https://doi.org/10.1016/j.ejca.2021.08.026
Wagner EH, Davis C, Homer CJ, Hagedorn SD, Austin B (2002) Abbi Kaplan. Curing the system - stories of change in chronic illness care, National Coalition on Health Care and the Institute for Healthcare Improvement
Iivanainen S, Alanko T, Peltola K, Konkola T, Ekström J, Virtanen H et al (2019) ePROs in the follow-up of cancer patients treated with immune checkpoint inhibitors: a retrospective study. J Cancer Res Clin Oncol 145:765–774. https://doi.org/10.1007/s00432-018-02835-6
Schmalz O, Jacob C, Ammann J, Liss B, Iivanainen S, Kammermann M et al (2020) Digital monitoring and management of patients with advanced or metastatic non-small cell lung cancer treated with cancer immunotherapy and its impact on quality of clinical care: interview and survey study among health care professionals and patients. J Med Internet Res 22:e18655. https://doi.org/10.2196/18655
Warrington L, Absolom K, Conner M, Kellar I, Clayton B, Ayres M et al (2019) Electronic systems for patients to report and manage side effects of cancer treatment: systematic review. J Med Internet Res 21:e10875. https://doi.org/10.2196/10875
Hoffman AJ (2013) Enhancing self-efficacy for optimized patient outcomes through the theory of symptom self-management. Cancer Nurs 36:E16–E26. https://doi.org/10.1097/NCC.0b013e31824a730a
Wendy H (2021) Oldenmenger, Corina J. G. van der Hurk, Doris Howell. Utilizing technology to manage symptoms. In: Charalambous A (ed) Dev. Util. Digit. Technol. Healthc. Assess. Monit. Springer, Cham, pp 55–72
Girgis A, Durcinoska I, Levesque JV, Gerges M, Sandell T, Arnold A et al (2017) eHealth system for collecting and utilizing patient reported outcome measures for personalized treatment and care (PROMPT-Care) among cancer patients: mixed methods approach to evaluate feasibility and acceptability. J Med Internet Res 19:e330. https://doi.org/10.2196/jmir.8360
Baeksted C, Pappot H, Nissen A, Hjollund NH, Mitchell SA, Basch E et al (2017) Feasibility and acceptability of electronic symptom surveillance with clinician feedback using the Patient-Reported Outcomes version of Common Terminology Criteria for Adverse Events (PRO-CTCAE) in Danish prostate cancer patients. J Patient-Rep Outcomes 1:1. https://doi.org/10.1186/s41687-017-0005-6
UKONS 24 hour triage rapid assessment and access toolkit. 2nd United Kingdom Oncology Nursing Society; 2016.
Zhou L, Bao J, Setiawan IMA, Saptono A, Parmanto B (2019) The mHealth app usability questionnaire (MAUQ): development and validation study. JMIR MHealth UHealth 7:e11500. https://doi.org/10.2196/11500
Sekhon M, Cartwright M, Francis JJ (2017) Acceptability of healthcare interventions: an overview of reviews and development of a theoretical framework. BMC Health Serv Res:17. https://doi.org/10.1186/s12913-017-2031-8
CFIR research team - center for clinical management research (2020) CFIR Interview Guide Tool. Consol Framew Implement Res. https://cfirguide.org/guide/app/#/. Accessed 8 Feb 2021
Bryant AL, Coffman E, Phillips B, Tan X, Bullard E, Hirschey R et al (2020) Pilot randomized trial of an electronic symptom monitoring and reporting intervention for hospitalized adults undergoing hematopoietic stem cell transplantation. Support Care Cancer 28:1223–1231. https://doi.org/10.1007/s00520-019-04932-9
Basch E, Deal AM, Kris MG, Scher HI, Hudis CA, Sabbatini P et al (2016) Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol 34:557–565. https://doi.org/10.1200/JCO.2015.63.0830
McCann L, Ream E, Armes J, Harris J, Kotronoulas G, Miaskowski C et al (2018) Remote monitoring systems in the cancer setting: eSMART: electronic Symptom Management using the Advanced Symptom Management System (ASyMS) Remote Technology for patients with cancer. The Breast 41:S4–S5. https://doi.org/10.1016/j.breast.2018.08.017
Mir O, Ferrua M, Fourcade A, Mathivon D, Duflot-Boukobza A, Dumont S et al (2022) Digital remote monitoring plus usual care versus usual care in patients treated with oral anticancer agents: the randomized phase 3 CAPRI trial. Nat Med 28:1224–1231. https://doi.org/10.1038/s41591-022-01788-1
Basch E, Barbera L, Kerrigan CL, Velikova G (2018) Implementation of patient-reported outcomes in routine medical care. Am Soc Clin Oncol Educ Book:122–134. https://doi.org/10.1200/EDBK_200383
Funding
Open access funding provided by University of Lausanne The present study was funded by the ISREC Foundation and Kaiku Health Ltd.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by André Manuel da Silva Lopes, Sara Colomer-Lahiguera, and Manuela Eicher. The first draft of the manuscript was written by André Manuel da Silva Lopes, Sara Colomer-Lahiguera and Manuela Eicher, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
A.D.S.L., C.D., S.G., S.B., G.G., V.A.L., N.M.A., S.L., and A. A. have no relevant financial or non-financial interests to disclose. S.C.L. reports grants from the ISREC Foundation. G.S-B. reports grants from MSD France, grants from Novartis, and personal fees from Bayer, MPNE, and WECAN, outside the submitted work. O.M. reports grants and personal fees from BMS, MSD, Pierre-Fabre, and Amgem; personal fees from Roche, Novartis, and GSL; and grants from Merck, outside the submitted work. M.E. received institutional research grants from Kaiku Health and reports grants from Bristol Myers Squibb, Roche and institutional fees as a Scientific Advisory Board Member/Consultant from Roche, outside the submitted work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
da Silva Lopes, A.M., Colomer-Lahiguera, S., Darnac, C. et al. Development of an eHealth-enhanced model of care for the monitoring and management of immune-related adverse events in patients treated with immune checkpoint inhibitors. Support Care Cancer 31, 484 (2023). https://doi.org/10.1007/s00520-023-07934-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-023-07934-w