Abstract
Background
The aim of this study was to investigate whether clinical outcomes in patients aged ≥ 70 undergoing decompressive surgery for degenerative cervical myelopathy (DCM) differ from those of younger patients (50–70 years) at 1 year.
Methods
Data were obtained from the Norwegian Registry for Spine Surgery (NORspine). Among 651 patients included, 177 (27.2%) were ≥ 70 years old. The primary outcome was change in the Neck Disability Index (NDI). Secondary outcomes were changes in the European Myelopathy Score (EMS), quality of life (EuroQoL EQ-5D), numeric rating scales (NRS) for headache, neck pain, and arm pain, and complications.
Results
Significant improvements in all patient-reported outcomes (PROMs) were detected for both age cohorts at 1 year. For the two age cohorts combined, there was a statistically significant improvement in the NDI score (mean 9.2, 95% CI 7.7 to 10.6, P < 0.001). There were no differences between age cohorts in mean change of NDI (− 8.9 vs. − 10.1, P = 0.48), EQ-5D (0.13 vs. 0.17, P = 0.37), or NRS pain scores, but elderly patients experienced a larger improvement in EMS (0.7 vs. 1.3, P = 0.02). A total of 74 patients (15.6%) in the younger cohort and 43 patients (24.3%) in the older cohort experienced complications or adverse effects within 3 months of surgery, mainly urinary and respiratory tract infections.
Conclusion
Surgery for DCM was associated with significant improvement across a wide range of PROMs for both younger and elderly patients. Surgery for DCM should not be denied based on age alone.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Degenerative cervical myelopathy (DCM), or cervical spondylotic myelopathy, is the most common cause of spinal cord impairment [19] and is associated with a wide range of symptoms and findings including gait disturbances, imbalance, loss of dexterity, impaired coordination, frequent falls, pain and stiffness in the neck, pain and numbness in limbs, and autonomic alterations that may cause bowel, urinary, and sexual problems [1, 28]. The initial symptoms are often subtle, and considerable delay in diagnosis is common. Clinicians need to be aware of the diagnosis and refer to MRI when DCM is suspected. There is growing evidence that decompressive surgery in selected patients can arrest progression of myelopathy and provide meaningful improvements in functional status, neurological outcomes, pain, and quality of life [10, 13]. However, complete resolution of symptoms is unlikely following surgery and risk associated with surgery is not negligible, as almost one in three patients reports adverse events within 3 months [12]. One suggested predictor for a less fortunate outcome after surgery is age [38]. As the elderly segment of the population continues to grow and MRI is readily available, the incidence of DCM is expected to rise. The prevalence and incidence of DCM is not well studied [3]. Studies on the general health of the Norwegian population show that at 65 years of age, life expectancy for men is 18.6 years, of which 14.4 will be healthy life years, and 21.5 years for women, of which 16.7 will be healthy life years. We do, however, not have good numbers for general health and quality of life for persons > 75 years of age [9].
High-quality data on surgical outcomes among elderly patients operated for degenerative cervical myelopathy are scarce [36], and there is concern that elderly patients experience less favorable outcome and more complications compared with younger patients [38]. However, the significance of age on the outcome after DCM surgery remains unclear. The purpose of this study was to compare the effectiveness and safety of surgery for DCM in patients aged ≥ 70 years vs. patients aged 50–70 years. We hypothesized that the elderly cohort would improve less than the younger cohort, and that they would have more complications.
Methods
The reporting in this paper is consistent with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines [35]. The study was approved by the Regional Committee for Medical Research and Health Research Ethics in Central Norway (2016/840), and all participants provided written informed consent.
Study population
The Norwegian Registry for Spine Surgery (NORspine) provides prospectively collected data on patients undergoing surgery for degenerative spinal disorders. NORspine is used for quality control and research, and provides data on demographics, lifestyle, and comorbidity, as well as patient-reported outcomes after spinal surgery [27]. All clinics that perform surgery for degenerative cervical disorders in Norway report to the registry [24, 26]. More than 80% of all surgeries on degenerative cervical spine in Norway are registered [13]. The study was planned after the data were collected, but before retrieval of data from the registry. Patients were included in the study if they had a primary diagnosis of DCM, were ≥ 50 years, and had undergone decompressive surgery between January 2012 and June 2018. Patients undergoing surgery for myelopathy for all other reasons, such as trauma, malignancies, infection, or deformity, are excluded from the registry. The patients were dichotomized into patients 50–70 years and patients aged ≥ 70 years.
Surgical procedures
The patients were referred to surgery based on clinical symptoms of myelopathy and corresponding radiographic findings. The surgical approach, number of operated levels, and the use and type of instrumentation were performed at the surgeons’ discretion.
Outcome measures
The primary outcome measure was change in Neck Disability Index (NDI) from baseline to 1-year follow-up. Secondary outcome measures were changes in the European Myelopathy Score (EMS), EQ-5D (EuroQoL’s instrument for measuring quality of life), and numeric rating scales (NRS) for headache, neck pain, and arm pain. The Global Perceived Effect (GPE) scale was used to measure the patients’ assessment of their condition 1 year after surgery. In addition, we reported surgeon and patient-reported complications that occurred within 3 months of surgery.
The NDI is a self-rated questionnaire developed for patients with neck disability. It has been translated into Norwegian and tested for psychometric properties [18]. The questionnaire is composed of ten items: seven related to activities of daily living (personal care, lifting, reading, work/daily activities, driving, sleep, and recreation), two related to pain (pain, headache), and one related to concentration. Each item is rated from 0 to 5. The NDI summary score ranges from 0 to 100, with lower scores indicating less disability.
The severity of cervical myelopathy was assessed by using the EMS [14, 34]. The EMS has five subscores: gait (1–5 points), bladder and bowel function (1–3 points), hand function (1–4 points), proprioception and coordination (1–3 points), and dysesthesia and paresthesia (1–3 points) [14, 34]. All subscores are functional, self-rated criteria that do not require formal testing. The total score ranges between 5 and 18. The EMS scores were dichotomized into mild DCM (scores of ≥ 13) and moderate-to-severe (scores between 5 and 12) [13].
Changes in health-related quality of life were measured with EQ-5D. The Norwegian version has shown good psychometric properties [30]. It evaluates five dimensions of the quality of life: mobility, self-care, activities of daily living, pain, and anxiety and/or depression. For each dimension, the patient describes three possible levels of problems (none, mild-to-moderate, and severe). An index value for health status is generated for each patient. Scores range from − 0.6 to 1, where 1 indicates perfect health.
Headache, neck and arm pain were measured with NRS [15]. NRS is a one-dimensional pain scale ranging from 0 (no pain) to 10 (worst imaginable pain).
The GPE scale [20] has seven categories: (1) complete recovery, (2) much better, (3) slightly better, (4) unchanged, (5) slightly worse, (6) much worse, and (7) worse than ever.
Surgeons provided data related to the following perioperative complications: unintentional durotomy, nerve root injury, wrong level of surgery, misplacement of implant, intraoperative hemorrhage requiring blood replacement, respiratory complications, anaphylactic reaction, spinal cord injury, esophageal injury, major vessel injury, cardiovascular complications, and other nerve injury. Patient-reported complications that occurred within 3 months of surgery were superficial wound infection, deep wound infection, urinary tract infection, pneumonia, pulmonary embolism, deep vein thrombosis, dysphagia, and dysphonia.
Data collection
Patients completed a self-administered questionnaire with baseline data on admission for surgery. The questionnaire included questions about demographics and personal characteristics (marital status, education, body mass index, and smoking). In addition, baseline data on patient-reported outcome measures (PROMs) were collected. Using a standard registration form, surgeons recorded data on diagnosis, comorbidity (including rheumatic diseases, hip or knee osteoarthritis, depression or anxiety, musculoskeletal pain, neurological disorder, cerebrovascular disease, cardiovascular disease, vascular claudication, lung disease, cancer, osteoporosis, hypertension, endocrine disorders), American Society of Anesthesiologists (ASA) score, image-related findings, hospital stay, and surgical procedure. NORspine distributed self-administered questionnaires to the patients by mail 3 and 12 months after surgery, without involving the treating hospitals. Non-responders received one reminder, together with a second copy of the questionnaire.
Statistical analysis
Statistical analyses were performed with SPSS version 26 (IBM) and Software R version 3.6.3. For statistical comparison tests, the significance level was defined as P ≤ 0.05. Frequencies were used for demographic variables at baseline, and changes in EMS, NDI, EQ-5D, and NRS were analyzed with paired sample T tests. Independent sample T tests were used to compare the changes between the two groups.
Missing data were handled with linear mixed model analyses. This strategy was in line with studies showing that imputations are not necessary before performing linear mixed model analysis of longitudinal data [23, 33]. In the mixed model, patients were not excluded from the analysis if a variable was missing at some, but not all, time points after baseline.
Results
In total, 651 patients were included in the study. There were 474 (72.8%) patients in the age group 50–70 years and 177 (27.2%) patients aged ≥ 70 years. A total of 525 participants (81%) provided PROMs at 3 and/or 12 months, and the response rate was similar for both age cohorts (80% vs. 84%, P = 0.24). Baseline characteristics are presented in Table 1.
Surgical outcomes are listed in Table 2. For both cohorts combined, there was a statistically significant improvement in NDI score (mean change 9.2, 95% CI, 7.7–10.6, P < 0.001). Complete case analyses showed no difference between age cohorts in change in NDI (mean difference 1.3, 95% CI − 2.2–4.7, P = 0.48), EQ-5D (mean difference − 0.04, 95% CI − 0.11–0.04, P = 0.37), NRS headache (mean difference 0.2, 95% CI − 0.6–0.9, P = 0.64), NRS neck pain (mean difference 0.5, 95% CI − 0.2–1.2, P = 0.18), or NRS arm pain (mean difference 0.5, 95% CI − 0.5–1.1, P = 0.44) from baseline to 1 year. EMS measured on the whole cohort changed from 14.0 to 14.9 (mean change 0.9 (95% CI 0.7–1.1, P < 0.001). Elderly patients had lower EMS scores at both baseline (14.5 vs. 12.7, P < 0.001) and 1 year (15.2 vs. 14.0, P < 0.001), and moderate-to-severe DCM was more common in the elderly group (41.6% vs. 18.2%, P < 0.001) compared to younger patients. The mean EMS change was slightly larger in the older age cohort (mean difference 0.6, 95% CI 0.1–1.1, P = 0.02).
The results of linear mixed model analyses were similar to those of the complete case analyses for all PROMs.
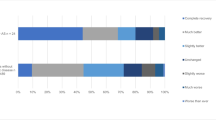
GPE for both age groups combined revealed that 167 out of 400 patients (41.8%) reported either complete recovery or much better at 1 year, and 64 out of 400 (16%) reported that they were slightly worse, much worse, or worse than ever at 1 year (Fig. 1). There was no difference in perceived benefit at 3 (P = 0.34) or 12 (P = 0.39) months.
Most patients were operated via either anterior or posterior approach, and very few underwent circumferential surgery and/or instrumentation. In the older age cohort, 132 patients (74.6%) were operated via posterior approach versus 207 (43.7%) in the younger age cohort.
Patients aged ≥ 70 years had 0.5 days longer hospital stay, mean 2.3 days (CI 95% 2.0 to 2.5) vs. 1.8 days (CI 95% 1.6 to 1.9) in the younger age cohort, P = 0.001.
Table 3 provides details of surgeon and patient-reported complications. There were no deaths within 30 days of surgery and no differences in surgeon-reported complications. In total, 117 patients (18.0%) reported complications within 3 months of surgery, 74 (15.6%) in the younger cohort vs. 43 (24.3%) in the older age cohort. Patients aged ≥ 70 years reported more urinary and respiratory tract infections within 3 months of surgery compared with patients in the younger age group.
Dysphagia was reported by 32/368 (8.7%) in the younger age cohort and 14/146 (9.6%) in the older age cohort.
Discussion
Patients aged ≥ 70 years experienced similar change in PROMs after decompressive surgery compared to younger patients. Moreover, elderly patients reported a larger improvement in EMS compared to younger patients. Elderly patients had longer hospital stays and experienced more minor complications compared with those aged 50–70 years.
Patients should be informed that complete resolution of symptoms and disability is unlikely, and that there is a risk of complications and neurological deterioration following surgery for DCM. Elderly patients had higher disability at baseline, and this may explain more residual symptoms at 1 year. Still, almost three out of four patients in both age cohorts reported at least some improvement according to GPE assessments at 1 year. Both groups experienced statistically significant improvement in all outcome measures. The clinical improvement is, however, modest, and the study population as a whole did not meet the recently suggested MCIC for NDI, NRS pain scores, and EQ-5D in a study from the Swedish Spine Registry [29]. One could argue that MCIC is of lesser importance given the often progressive nature of DCM [7].
The choice of cutoff age for “elderly” varies between studies. Some define elderly as > 65 years of age [25] and others as > 75 years of age [21]. A study trying to define a cutoff age for what is considered elderly by interviewing 300 individuals landed on 73.7 years [6]. In a recent AOSpine study on DCM surgery in the elderly, the cutoff age for elderly was set to 70 years [37]. In concordance with this, we chose 70 years of age or older as our definition of elderly.
Cervical spinal degeneration and comorbidity increase with age, and as expected, the two cohorts were not balanced for baseline factors. Patients in the older age cohort had more disability at baseline and at 1 year after surgery compared with patients in the younger age cohort. Still, the older age cohort had similar improvement in all PROMs at 1 year despite more comorbidity and higher ASA grade which possibly could impact the results in a negative manner. The proportion of patients operated for moderate-to-severe myelopathy was significantly higher in the older age cohort. Patients in both age groups who were operated for moderate-to-severe myelopathy experienced a larger improvement measured in EMS than those operated for mild myelopathy.
When comparing the study population to the general population, we find that the younger study population has somewhat higher prevalence of DMII and chronic obstructive lung disease and somewhat lower prevalence of stroke, hypertension, and cancer than the general population. The older age cohort has somewhat higher prevalence of chronic obstructive lung disease and somewhat lower prevalence of hypertension and cancer than the general population [16]. The numbers and different disease categories are not directly comparable, so statistical analysis is not sensible, but the numbers indicate that the populations studied are not very different from the general population.
In recent years, studies on frailty have been published, and one can argue that frail vs. non-frail patients are a better comparison than young vs. old patients. Frailty is defined as reduced physiological reserve which means that patients are susceptible to sudden, disproportionate functional decline following stressor events [8]. Age is incorporated in many of the frailty assessment scales, and frailty is strongly associated with age, but should be viewed as a separate entity [4]. Only patients that have been operated are included in NORspine. All these patients have been through preoperative screening, and those deemed unfit for surgery are already excluded. This would comprise many of the frail patients with cardiovascular challenges believed to be a risk factor for surgery. One must bear in mind that frailty is a dynamic entity which can both worsen and improve [17], that DCM patients score higher on frailty scales due to the nature of their diagnosis, and that improvement in DCM symptoms means improvement in frailty. Data from NORspine do not comprise frailty scales, but baseline data including comorbidity and ASA grade do not imply less frailty in the elderly group, so this does not weaken our results or conclusion.
Results from studies on elderly patients undergoing surgery for DCM vary (Table 4). A previous study has shown that age is a predictor of less favorable outcomes after surgery for DCM [38]. This contrasts to our study that demonstrated improvement, also in patients in the age group 70 years or older. They discuss whether this is due to age-related changes in the spinal cord, general degeneration associated with normal aging, and comorbidity. Results from the AOSpine CSM study show that elderly patients experience improvement, but not to the same extent as patients aged < 70, and that they have a similar complication rate as younger patients [37]. Similar improvement in PROMs were also observed in a recent study comparing different age groups [5].
In our study, the surgeons decided upon surgical approach based on patient factors, clinical symptoms and findings, and radiographic imaging. Elderly patients were more likely to undergo posterior decompression (Table 2). It would be interesting to compare the effectiveness of different surgical treatments, but this was beyond the scope of our study. The authors of a recent RCT comparing ventral vs. dorsal surgery for DCM concluded that there were no significant differences in outcomes, but more complications in the ventral surgery group [12] mainly due to dysphagia. We found similar incidence rates for dysphagia in both the younger and older age cohorts. The proportion of dysphagia was a bit higher among elderly patients operated via anterior approach, but this did not reach statistical significance. Interestingly, some patients that were operated via posterior approach also reported dysphagia at 3 months after surgery. This suggests that other factors than manipulation with the esophagus during surgery might play a role in the development of dysphagia.
Total complications were higher in the older age cohort mainly due to urinary and respiratory tract infections within 3 months. This is in line with previous reports on the outcomes of degenerative lumbar spine surgery [11, 22] and the recent AOSpine article on surgery for DCM in the elderly [37]. Life-threatening complications were fortunately rare for both age cohorts.
On average, patients aged ≥ 70 years had 0.5 days longer hospital stays than those in the 50–70 age group. The reasons are probably multifactorial and might include the possibility that fewer patients in the old age group had a life partner or spouse, differences in comorbidity, or different routines for postoperative mobilization, pain management, and hospital discharge. In addition, there were more posterior surgical approaches in the older cohort that probably reflect that degeneration of the spine increases with age and may imply more postoperative pain.
DCM is a frequently encountered diagnosis in spine surgery practice, but the time from symptom debut to surgical assessment is often long. One of the risk factors for poor surgical outcome is long duration of symptoms [38]. In one study, the mean time from symptom debut to diagnosis exceeded 2 years [2]. Approximately 20% of our patients had duration of symptoms more than 1 year. Patients should be identified and promptly referred for MRI and to a spine specialist for surgical assessment. Delayed diagnosis can lead to neurological deterioration and unnecessary residual symptoms because of delayed surgery [32].
Strengths and limitations
The pragmatic study design based on prospective registry data in an everyday clinical setting with a large study population ensures high external validity. Another strength is the wide range of patient-reported outcome measures. However, the two age cohorts were not balanced for all baseline and treatment factors, as cervical spine degeneration and comorbidity increase with age, and confounding factors were not explored. The main limitation was the loss to follow-up of 19%. However, a previous study from the NORspine registry found no differences in outcomes between responders and non-responders [31]. Another limitation is that patients in the older age cohort are carefully selected for surgery and might not be representative of the total population of elderly DCM patients. NORspine only includes patients that actually undergo surgery, and unfortunately, we do not have any information about patients ineligible for surgical treatment due to frailty and comorbidity. Patient characteristics, indications, and surgical strategies may vary between institutions and countries, and results from our study might differ from other countries and clinical settings.
Conclusions
Patient-reported outcomes following surgery for DCM showed that patients aged ≥ 70 years experienced similar improvements when compared to younger patients (50–70 years). Hospital stays were slightly longer for those in the elderly age group, but there were no relevant differences in perioperative complications between the two age groups. Patients who were ≥ 70 years of age were more likely to report minor complications within 3 months. Thus, age alone should not be a general contraindication for surgical treatment of DCM.
Data availability
No additional data are available.
Abbreviations
- DCM:
-
Degenerative cervical myelopathy
- NORspine:
-
Norwegian Registry for Spine Surgery
- NDI:
-
Neck Disability Index
- EMS:
-
European Myelopathy Score
- EQ-5D:
-
EuroQoL five dimensions
- NRS:
-
Numeric rating scale
- PROM:
-
Patient-reported outcome measure
- MRI:
-
Magnetic resonance imaging
- STROBE:
-
Strengthening the Reporting of Observational Studies in Epidemiology
- GPE:
-
Global Perceived Effect
- ASA:
-
American Society of Anesthesiologists
- SPSS:
-
Statistical Package for the Social Sciences
References
Badhiwala JH et al (2020) Degenerative cervical myelopathy - update and future directions. Nat Rev Neurol 16(2):108–124
Behrbalk E et al (2013) Delayed diagnosis of cervical spondylotic myelopathy by primary care physicians. Neurosurg Focus 35(1):E1
Boogaarts HD, Bartels RH (2015) Prevalence of cervical spondylotic myelopathy. Eur Spine J 24(Suppl 2):139–141
Clegg A et al (2013) Frailty in elderly people. Lancet 381(9868):752–762
Croci, D.M., et al., 2022 Differences in postoperative quality of life in young, early elderly, and late elderly patients undergoing surgical treatment for degenerative cervical myelopathy. J Neurosurg Spine. p. 1–11.
Daignault M, Wassef A, Nguyen QD (2021) How old is old? Identifying a chronological age and factors related with the perception of old age. J Am Geriatr Soc 69(11):3330–3333
Davies BM et al (2018) Degenerative cervical myelopathy. BMJ 360:k186
Dent E et al (2019) Management of frailty: opportunities, challenges, and future directions. Lancet 394(10206):1376–1386
Engdahl, B.L., 2016 Health among the elderly in Norway.
Fehlings MG et al (2017) A clinical practice guideline for the management of patients with degenerative cervical myelopathy: recommendations for patients with mild, moderate, and severe disease and nonmyelopathic patients with evidence of cord compression. Global Spine J 7(3 Suppl):70S-83S
Giannadakis C et al (2016) Surgery for lumbar spinal stenosis in individuals aged 80 and older: a multicenter observational study. J Am Geriatr Soc 64(10):2011–2018
Ghogawala Z et al (2021) Effect of ventral vs dorsal spinal surgery on patient-reported physical functioning in patients with cervical spondylotic myelopathy: a randomized clinical trial. JAMA 325(10):942–951
Gulati, S., et al., 2021 Surgery for degenerative cervical myelopathy: a nationwide registry-based observational study with patient-reported outcomes. Neurosurgery.
Herdmann J., L M., Krzan M., Dvorak J., Bock W.J., 1994 The European Myelopathy Score. Cerebellar Infarct. Midline Tumors. Minimally Invasive Endoscopic Neurosurgery (MIEN). Advances in Neurosurgery, vol 22.
Hjermstad MJ et al (2011) Studies comparing Numerical Rating Scales, Verbal Rating Scales, and Visual Analogue Scales for assessment of pain intensity in adults: a systematic literature review. J Pain Symptom Manage 41(6):1073–1093
Hopstock, L.A., et al., 2022 The seventh survey of the Tromso Study (Tromso7) 2015–2016: study design, data collection, attendance, and prevalence of risk factors and disease in a multipurpose population-based health survey. Scand J Public Health,14034948221092294.
Iqbal J, Denvir M, Gunn J (2013) Frailty assessment in elderly people. Lancet 381(9882):1985–1986
Johansen JB et al (2013) Measurement properties of the norwegian version of the neck disability index in chronic neck pain. Spine (Phila Pa 1976) 38(10):851–6
Kalsi-Ryan S, Karadimas SK, Fehlings MG (2013) Cervical spondylotic myelopathy: the clinical phenomenon and the current pathobiology of an increasingly prevalent and devastating disorder. Neuroscientist 19(4):409–421
Kamper SJ et al (2010) Global Perceived Effect scales provided reliable assessments of health transition in people with musculoskeletal disorders, but ratings are strongly influenced by current status. J Clin Epidemiol 63(7):760-766 e1
Madhavan K et al (2016) Surgical outcomes of elderly patients with cervical spondylotic myelopathy: a meta-analysis of studies reporting on 2868 patients. Neurosurg Focus 40(6):E13
Madsbu MA et al (2017) Surgery for herniated lumbar disk in individuals 65 years of age or older: a multicenter observational study. JAMA Surg 152(5):503–506
Madsbu, M.A., et al., 2017 Surgery for herniated lumbar disk in individuals 65 years of age or older: a multicenter observational study. JAMA Surg.
Madsbu MA et al (2020) Surgery for herniated lumbar disc in private vs public hospitals: a pragmatic comparative effectiveness study. Acta Neurochir (Wien) 162(3):703–711
Nakashima H et al (2016) Does age affect surgical outcomes in patients with degenerative cervical myelopathy? Results from the prospective multicenter AOSpine International study on 479 patients. J Neurol Neurosurg Psychiatry 87(7):734–740
Nerland US et al (2015) Minimally invasive decompression versus open laminectomy for central stenosis of the lumbar spine: pragmatic comparative effectiveness study. BMJ 350:h1603
NORSpine Annual Report. 2019.
Nouri A et al (2015) Degenerative cervical myelopathy: epidemiology, genetics, and pathogenesis. Spine (Phila Pa 1976) 40(12):E675-93
Revesz, D.F., 2022 A. Charalampidis, and P. Gerdhem, Effectiveness of laminectomy with fusion and laminectomy alone in degenerative cervical myelopathy. Eur Spine J.
Solberg TK et al (2005) Health-related quality of life assessment by the EuroQol-5D can provide cost-utility data in the field of low-back surgery. Eur Spine J 14(10):1000–1007
Solberg TK et al (2011) Would loss to follow-up bias the outcome evaluation of patients operated for degenerative disorders of the lumbar spine? Acta Orthop 82(1):56–63
Tetreault L et al (2019) Is preoperative duration of symptoms a significant predictor of functional outcomes in patients undergoing surgery for the treatment of degenerative cervical myelopathy? Neurosurgery 85(5):642–647
Twisk J et al (2013) Multiple imputation of missing values was not necessary before performing a longitudinal mixed-model analysis. J Clin Epidemiol 66(9):1022–1028
Vitzthum HE, Dalitz K (2007) Analysis of five specific scores for cervical spondylogenic myelopathy. Eur Spine J 16(12):2096–2103
von Elm E et al (2007) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet 370(9596):1453–1457
Wang, T.Y. and K.D. Than, 2021 Commentary: surgery for degenerative cervical myelopathy: a nationwide registry-based observational study with patient-reported outcomes. Neurosurgery.
Wilson JRF et al (2019) The impact of older age on functional recovery and quality of life outcomes after surgical decompression for degenerative cervical myelopathy: results from an ambispective, propensity-matched analysis from the CSM-NA and CSM-I International, multi-center studies. J Clin Med 8:10
Zhang JT et al (2016) Risk factors for poor outcome of surgery for cervical spondylotic myelopathy. Spinal Cord 54(12):1127–1131
Acknowledgements
The authors thank the Norwegian Registry for Spine Surgery (NORspine) for proving access to data used in the study. We are greatly indebted to all patients and spine surgeons who participated in NORspine registration.
Funding
Open access funding provided by NTNU Norwegian University of Science and Technology (incl St. Olavs Hospital - Trondheim University Hospital)
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript. TOJ, SG, ØPN, VVL, and ØOS took part in the study design, statistical analyses, interpretation of results, and writing. ØPN and TKS were involved in collection of the data, interpretation of results, and writing of the manuscript. AMG and STH were involved in the interpretation of results and writing.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Regional Committee for Medical Research and Health Research Ethics in Central Norway (2016/840), and all participants provided written informed consent.
Competing interests
The authors declare no competing interests.
Transparency
The lead author (TOJ) confirms that this paper is an honest, accurate, and transparent account of the reported study, that no important aspects of the study have been omitted, and that any discrepancies from the study as planned have been explained.
Additional information
Comments
Johansen et al. presented an interesting Norwegian registry-based observational study on surgery for degenerative cervical myelopathy in the elderly. Surprisingly, the results of the younger and older age group are comparable although with age degenerative changes are more advanced and frequent. This speaks even more for the fact that also and especially elderly patients can highly benefit from operative treatment.The authors have well discussed the limitations of their study. Although not all of them, most of the interventions for the treatment of degenerative diseases of the cervical spine were registered. However, possible complications after a period of half a year were not recorded. Here, for example, the frequency of adjacent segment degeneration, instability and reoperations would have been of interest. In addition, it would be interesting to know whether the results last in the long-term course and whether there would be differences between the age groups in this regard. The fact that complications such as urinary and respiratory tract infections are more common in the older age group seems to speak for a disposition of and higher risk for older patients. But also, many other factors such as comorbidities and duration as well as extent of symptoms and degree of myelopathy can play an important role. I completely agree with the authors of this article that old age per se should not be a contraindication to surgery for degenerative spine diseases. Since the life expectancy and number of elderly people are constantly increasing, close cooperation with colleagues from other disciplines such as gerontologists who care especially for elderly patients and their concomitant diseases is helpful and to be strived for. Due to the expected further demographic development with more and more elderly patients and since they present a distinct patient group with special requirements, it would even probably be suggested in the future to create a new kind of specialization in the sense of a geriatric neurosurgeon or spine specialist for patients with advanced age. I have read the article with great curiosity and congratulate the authors on their important contribution to spinal treatment of elderly patients.
Markus Florian Oertel
Zurich, Switzerland
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Spine degenerative.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Johansen, T.O., Vangen-Lønne, V., Holmberg, S.T. et al. Surgery for degenerative cervical myelopathy in the elderly: a nationwide registry-based observational study with patient-reported outcomes. Acta Neurochir 164, 2317–2326 (2022). https://doi.org/10.1007/s00701-022-05282-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-022-05282-y