Abstract
Objectives
To prospectively assess the reliability and accuracy of high-resolution, dental MRI (dMRI) for endodontic working length (WL) measurements of premolars and molars under clinical conditions.
Materials and methods
Three-Tesla dMRI was performed in 9 subjects who also had undergone cone-beam computed tomography (CBCT) (mean age: 47 ± 13.5 years). A total of 34 root canals from 12 molars (4/8, upper/lower jaw; 22 root canals) and 11 premolars (2/9 upper/lower jaw; 12 root canals) were included. CBCT and dMRI datasets were reconstructed to visualize the root canal in one single slice. Subsequently, two radiologists measured the root canal lengths in both modalities twice in blinded fashion. Reliability and accuracy for both modalities were assessed using intraclass correlation coefficients (ICCs) and Bland–Altman analysis, respectively.
Results
Reliability (intra-rater I/II; inter-rater) of dental MRI measurements was excellent and comparable to CBCT for premolars (0.993/0.900; 0.958 vs. 0.993/0.956; 0.951) and for molars (0.978/0.995; 0.986 vs. 0.992/0.996; 0.989). Bland–Altman analysis revealed a mean underestimation/bias (95% confidence interval) of dMRI measurements of 0.8 (− 1.44/3.05) mm for premolars and 0.4 (− 1.55/2.39) mm for molars. In up to 59% of the cases, the accuracy of dMRI for WL measurements was within the underestimation margin of 0 to 2 mm short of the apical foramen AF.
Conclusions
In vivo demonstration and measurement of WL are feasible using dMRI. The reliability of measurements is high and equivalent to CBCT. Nonetheless, due to lower spatial resolution and longer acquisition time, the accuracy of dMRI is inferior to CBCT, impeding its current use for clinical treatment planning.
Clinical relevance
dMRI is a promising radiation-free imaging technique. Its reliability for endodontic working length measurements is high, but its accuracy is not satisfactory enough yet.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Accurate assessment of root canal morphology and preexisting pathology is crucial in treatment planning in endodontic therapy [1, 2]. Intraoral periapical radiography (PR) represents the first and easiest method of choice for visualizing periapical pathology and appropriate treatment planning [3]. However, this means three-dimensional (3D) structures are acquired in two-dimensional (2D) images and superimposed with the surrounding anatomy without any information about the spatial relationships. These limitations were recognized as early as in the 1960s [4, 5], acknowledging the restrictions of PR, in particular for the buccolingual dimension [6]. Consequently, periapical lesions are often missed on PR when compared to cone-beam computed tomography (CBCT) [7]. A new era of dento-maxillo-facial imaging emerged with the introduction of (CBCT) at the end of the last millennium [8]. Since then, CBCT has a well-established role in endodontics [9, 10].
Even though CBCT is associated with a significantly lower radiation dose compared to conventional CT-imaging [8, 11], this is substantially higher compared to conventional radiography [12]. In conjunction with the increasing application and relevance of CBCT in the management of endodontic disorders [13], this leads to an increased radiation exposure in the general population [14]. This in turn has severe effects on the young population, especially children who are more sensitive to radiation [15, 16] and therefore have a higher risk of developing cancer later on [17, 18]. Thus, the application of CBCT should be balanced out against radiation exposure. For these reasons, a 3D, non-ionizing imaging modality would be beneficial, most importantly for young patients. High-resolution three-dimensional dental MRI (dMRI) represents a new imaging modality with the potential to serve as a non-ionizing alternative to CBCT. dMRI has recently delivered promising results in endodontics [19], e.g., in detecting root fractures [20] and in detecting and differentiating periapical lesions [21, 22] as well in guided endodontics [23]. In addition, dMRI has the potential to assess pulp regeneration [24] and reperfusion [25]. These aspects have largely remained unamenable for ionizing radiation imaging modalities. Recent developments of dedicated coil and sequence techniques [26,27,28] have provided higher resolution and improved metal artifact–suppressed dMRI images. These improvements could allow the use of dMRI for further endodontic indications as well.
Several previous ex vivo studies have also concluded that CBCT can be used for accurate working length (WL) measurements, compared to the electronic apex locator (EAL) [29, 30]. Defining an accurate WL is an important factor for the success of root canal therapy [2, 31]. Although EAL remains the gold standard to determine accurate length measurements in most cases, using CBCT for WL measurements in patients in whom the EAL precision may be limited (e.g., due to a root perforation) provides an additional and reliable option [32]. This could therefore also be transferable to dMRI as a radiation-free diagnostic tool.
However, it remains unclear whether the new imaging capabilities of dMRI allow for a reliable and accurate analysis of the working length in clinical practice which should be comparable to CBCT. In our study, we, therefore, prospectively evaluated these parameters in vivo using dMRI and directly compared the results with the CBCT.
Materials and methods
The study was approved by the ethics committee of the University of Heidelberg (approval number S-404/2014), and written informed consent was obtained from all participants. Overall, 9 subjects (3 males, 6 females) were prospectively enrolled in the study. Inclusion criteria were age of 18 and availability of a CBCT scan, which was performed prior to dMRI for a clinical indication (e.g., implant planning). Exclusion criteria included pregnancy, claustrophobia, and implants considered not to be safe at a field strength of 3 Tesla (e.g., cochlear implants, pacemakers, implantable defibrillators, event recorder). Exclusion criteria on tooth level were caries and/or restorations affecting or compromising the pulp, root canal, or cuspal edge (e.g., crowns, bridges, and restorations made of non-precious alloys affecting the visibility of adjacent pulps).
Imaging
All MRI scans were obtained on a 3 Tesla MRI system (Magnetom Trio; Siemens Healthineers GmbH, Erlangen, Germany) using a dedicated 15-channel dental surface coil (Mandibula, Noras MRI products GmbH). A PD-weighted 3D MSVAT-SPACE (multiple slab acquisition with view angle tilting gradient based on a sampling perfection with application-optimized contrasts using different flip angle evolution) sequence was applied, optimized for dental MRI, as described elsewhere [27]. Sequence parameters were repetition time 1170 ms; echo time, 6.4 ms; FOV, 168 mm × 131 mm; voxel size, 0.44 mm × 0.44 mm × 0.44 mm; acquisition matrix, 384 × 300; slice oversampling 220%; slices 80; and time of acquisition, 7:45 min. All CBCT images were acquired using 3D Accuitomo 170 (J Morita; Kyoto, Japan) with the following acquisition parameters: an isotropic voxel size: 160 µm, field of view: 8 × 8 cm2, tube voltage: 90 kV, tube current: 7 mA, 14-bit, 360° rotation in 17 s, and 560 frames.
Image analysis
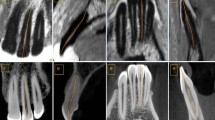
Osirix (v. 8.5.1., Geneva, Switzerland) was used to reformat the CBCT/MRI datasets and reconstruct the slices; first, we identified the deepest intercuspal point in three planes. Then, a plane visualizing the whole length of the pulp chamber and complete root canal till the major foramen was defined. Next, images were randomized and evaluated by two radiologists (with 3 and 8 years of experience in dental imaging) twice. To avoid any influence of the reference measurements in CBCT on MRI evaluation, MRI was evaluated first. A time interval of 2 weeks separated both evaluation rounds within each modality as well as MRI and CBCT image assessment to minimize learning bias (6 weeks between first MRI read and last CBCT read). In addition, CBCT and MRI datasets were presented in different order and names in each read. Conditions between both examiners were identical. Both examiners used the same computer (MacBook Pro 15.4 inches, Model A1707, 2880-by-1800 native resolution at 220 pixels per inch) and under ACR (American College of Radiology) recommended reading room conditions [33] (diffuse ambient light of 20 lx). The radiologists could adjust the contrast/saturation of the images. The root canal length of molars/premolars was defined as the distance between the deepest intercuspal point in the projected midline of the pulp cavity and the major foramen. The measurement line followed the curvatures of the pulp cavity and was positioned in the center of the root canal until the major foramen as described before [34] (Fig. 1).
Statistical analysis
Statistical analysis was done using SPSS 23 (SPSS Inc., Chicago, USA). To assess the reliability of both modalities, intra- and inter-rater agreement was determined by calculating intraclass correlation coefficients (ICCs) with a 95% confidence interval (IC). ICC values were interpreted as described before [30]. The accuracy of dMRI was analyzed with Bland–Altman plots using CBCT measurements as reference values.
Results
The study cohort comprised 9 subjects (3 males and 6 females) who met the inclusion criteria. Mean age ± SD was 47 years ± 13.5 (median, 45; range, 29–68). A total of 34 root canals from 12 molars (4 upper jaw, 8 lower jaw; 22 root canals) and 11 premolars (2 upper jaw, 9 lower jaw; 12 root canals) were included in this study of the correlation between CBCT and dMRI measurements. Molars and premolars with apparent movement or metal artifacts during image acquisition were excluded. All root canals identified in the reference modality CBCT were included and were visible in dMRI images.
Reliability
Intra-/inter-rater reliability of root canal length measurements for premolars was excellent for both modalities: 0.956–0.993/0.951 for CBCT and 0.900–0.993/0.958 for dMRI (Tables 1 and 2). An excellent intra-/inter-rater reliability was also noted for molars in both modalities as well with ICCs of 0.992–0.996/0.989 for CBCT and 0.978–0.995/0.986 for dMRI.
Accuracy
dMRI showed a tendency to underestimate the WL in 67.6% of all measurements compared to CBCT (underestimation in 75% in the premolars and 63.6% in the molars). Bland–Altman analysis uncovered a mean underestimation/bias (lower/upper limits of agreement) of 0.55 (− 1.51/2.62) mm. This translates to mean underestimation values of 0.8 (− 1.44/3.05) mm for premolars and 0.4 (− 1.55/2.39) mm for molars (Fig. 2).
Bland–Altman plots of the mean differences between CBCT and dMRI for working length measurements for premolars (A) and molars (B) illustrating an underestimation of dMRI-derived measurements for both premolars and molars. The solid line represents the mean of all differences (bias) and the dotted lines represent the upper and lower 95% limits of agreement (LOA)
The proportion of dMRI measurements within 0 to 2 mm short difference range from CBCT was 58.3% for premolars and 59% for molars. An overestimation beyond 0.5 mm of the AF was reported in 16.6/22.7% of measurements for premolars/molars. The maximum difference between dMRI and CBCT measurements was 2.83 mm in underestimations and 0.7 mm in overestimations for premolars as well as 2.63 mm in underestimations and 1.38 in overestimations for molars.
Discussion
CBCT has become the gold standard in dental imaging, particularly in the field of endodontics, e.g., preoperative planning, to assess the extent of periapical lesions and their proximity to adjacent anatomical structures, as well as to determine the accurate number of root canals and accompanying pathologies such a periapical periodontitis and root fractures [9, 10]. However, its application is still limited due to the higher radiation dose compared to PR [35]. The higher radiation dose is associated with an increased risk of cancer later on, especially in young, radiosensitive patients [14, 18]. Consequently, a comparable, non-ionizing, 3D imaging modality like dMRI would be of high clinical value. In this study, we assessed the reliability and accuracy of dMRI for measuring the WL of premolars/molars in vivo. In terms of reliability, dMRI performed excellently and comparable to the reference imaging modality CBCT. In terms of accuracy, however, dMRI systematically underestimated WL, with a mean underestimation of 0.55 mm.
A notable benefit of our investigation is the evaluation of dMRI reliability and accuracy under in vivo conditions, thereby reproducing real-time clinical settings, where metal and motion artifacts are incorporated in the evaluation. Furthermore, all root canals identified in the reference modality CBCT were included in the study. In contrast, available information on the accuracy and reliability of CBCT in endodontic working length measurements for premolar/molars is restricted predominantly to ex vivo studies [29, 30, 36, 37]. This is problematic because ex vivo studies exclude motion artifacts, which are typical under clinical conditions with possible impact on diagnostic accuracy [38]. Liang et al. and Connert et al. included 162 and 42 extracted teeth, respectively, of which 46/42 and 12/12 were premolars/molars, respectively. Previous in vivo studies evaluating the accuracy of CBCT WL measurements only evaluated small patient cohorts and included anterior teeth only: Janner et al. (9 teeth; 6 incisors, 2 canines, 1 premolar) and Jeger et al. (40 teeth; 32 incisors, 8 canines) [34, 39]. In opposition to the previously mentioned in vivo studies, our investigation offered a comparable large sample size of 11 premolars and 12 molars. Altogether, 46 datasets and 184 root canal measurements were evaluated.
Liang et al. observed a high reliability of CBCT measurements in WL in their ex vivo study with an excellent intra-rater ICC of 0.982 [30]. Correspondingly, the reliability assessment of our CBCT measurements delivered high intra-rater ICCs as well (0.956–0.996). In a similar fashion, our analysis delivered comparably high intra-rater ICCs for dMRI in the range of 0.900–0.995. Connert et al. analyzed the repeatability in their study and reported a mean of absolute differences in CBCT measurements of 0.14 mm with a range of 0.12–0.16 mm, for all teeth included in the study. Unfortunately, the authors of that study did not report separate repeatability results for premolars and molars. In contrast, our study offered a separate repeatability assessment of the premolar/molars in both modalities, an additional strength of our investigation. We delivered a mean of absolute differences in CBCT measurements of 0.14 (0.02–0.26) mm/0.17 (0.09–0.25) mm and in dMRI measurements of 0.08 (0.02–0.15) mm/0.12 (0.1–0.13) mm for premolars/molars, respectively, illustrating an excellent and comparable repeatability in our study.
The preceding investigation of Liang et al. observed a high accuracy for CBCT-based root canal length measurements compared to EAL. Pearson correlation coefficients of 0.958 for premolars and 0.936 for molars were reported [30]; our study demonstrated a lower correlation between CBCT and dMRI measurements of 0.602 for premolars and 0.880 for molars. For assessment of absolute mean discrepancies between CBCT and EAL, Connert et al. and Liang et al. reported mean differences (range) of 0.48 mm (0.30–0.68 mm)/0.49 mm (0.30–0.66 mm) and 0.42 mm (0.03–1.12 mm)/0.51 mm (0–1.33 mm) for premolars/molars, under ex vivo conditions, respectively [29, 30]. Again, our in vivo study revealed a larger/comparable error for premolars/molars (0.8 mm /0.41 mm). The range, however, was larger for both, premolars (− 1.44 to 3.05) and molars (− 1.55 to 2.39). In another ex vivo study, Metska et al. measured 40 root canals of 33 maxillary teeth, of which 7 were molars and 11 were premolars and compared it to direct measurements [36]. The authors reported a mean underestimation of CBCT-based measurements of 0.51 ± 0.73 mm for posterior teeth (premolars and molars). In direct comparison with our dMRI results, the combined mean underestimation of 0.55 ± 1.05 mm seems comparable for both tooth types. Finally, Segato et al. compared the CBCT measurements of 30 mandibular premolars to direct measurements in another ex vivo study and reported that 73% of all measurements were within ± 0.5 mm [37]. In contrast, in our study, only 16.6% of our premolar dMRI measurement errors were within ± 0.5 mm.
In the present study, dMRI WL measurements underestimated the length in 75/63.6% of the cases for premolars/molars, respectively. Previous systematic reviews reported an improved outcome of primary root canal treatment if the root canal filling was within 2 mm short to 0 mm to the apical foramen [2]. In the present study, 58.3/59% dMRI-based measurements were within that range for premolars/molars, respectively. Moreover, in 16.6/22.7% of our dMRI measurements, there was a WL overestimation of more than 0.5 mm for premolars/molars, respectively, highlighting the lower accuracy of current dMRI technique. Nonetheless, it is essential to note that the dMRI-derived error was within the same scale as previously reported CBCT errors even though the spatial resolution as well as the acquisition time was substantially lower/longer (factor of voxel volume 21; isotropic voxel size of 0.44 mm vs. 0.16 mm from CBCT; factor of acquisition time 26; 7.45 min vs. 17.5 s).
This study has several limitations. First, CBCT and not EAL was chosen as the reference modality; even though previous studies revealed a high accuracy of CBCT-based measurements, a direct comparison with EAL or even histological results might have increased accuracy evaluation. Second, the spatial resolution of dMRI was significantly lower than CBCT, despite the application of a high-resolution dMRI setup, resulting in a lower accuracy. Third, in vivo images are prone to motion artifacts due to the longer acquisition time of dMRI compared to CBCT. That is why further developments are needed to enhance MRI image acquisition speed as well as resolution. Finally, higher costs and restricted access to MRI machines may be additional limitations for the clinical application of dMRI.
Conclusion
This in vivo study demonstrated the feasibility of root canal length measurements for premolars and molars using high-resolution dMRI. Compared to CBCT, the intra- and inter-rater reliability of dMRI measurements was comparably high and overall excellent. In terms of accuracy of dMRI, however, root canal lengths were systematically underestimated compared to CBCT, restricting its clinical application in its current form. Further dMRI improvements resulting in higher spatial resolution and shorter acquisition times are necessary for application of dMRI in clinical routine.
References
Schloss T, Sonntag D, Kohli MR, Setzer FC (2017) A comparison of 2- and 3-dimensional healing assessment after endodontic surgery using cone-beam computed tomographic volumes or periapical radiographs. J Endod 43(7):1072–1079. https://doi.org/10.1016/j.joen.2017.02.007
Ng YL, Mann V, Rahbaran S, Lewsey J, Gulabivala K (2008) Outcome of primary root canal treatment: systematic review of the literature – part 2 influence of clinical factors. Int Endod J 41(1):6–31. https://doi.org/10.1111/j.1365-2591.2007.01323.x
E. European Society of (2006) Quality guidelines for endodontic treatment: consensus report of the European Society of Endodontology. Int Endod J 39(12):921–930. https://doi.org/10.1111/j.1365-2591.2006.01180.x
Bender IB, Seltzer S (2003) Roentgenographic and direct observation of experimental lesions in bone: I 1961. J Endod 29(11):702–6. https://doi.org/10.1097/00004770-200311000-00005 (discussion 701)
Bender IB (1961) S Seltzer (2003) Roentgenographic and direct observation of experimental lesions in bone: II. J Endod 29(11):707–12. https://doi.org/10.1097/00004770-200311000-00006 (discussion 701)
Schwartz SF, Foster JK Jr (1971) Roentgenographic interpretation of experimentally produced bony lesions. I Oral Surg Oral Med Oral Pathol 32(4):606–612. https://doi.org/10.1016/0030-4220(71)90326-4
Tsai P, Torabinejad M, Rice D, Azevedo B (2012) Accuracy of cone-beam computed tomography and periapical radiography in detecting small periapical lesions. J Endod 38(7):965–970. https://doi.org/10.1016/j.joen.2012.03.001
Mozzo P, Procacci C, Tacconi A, Martini PT, Andreis IA (1998) A new volumetric CT machine for dental imaging based on the cone-beam technique: preliminary results. Eur Radiol 8(9):1558–1564. https://doi.org/10.1007/s003300050586
Fayad MI, Nair M, Levin MD, Benavides E, Rubinstein RA, Barghan S, Hirschberg CS, Ruprecht A (2015) AAE and AAOMR joint position statement: use of cone beam computed tomography in endodontics 2015 update. Oral Surg Oral Med Oral Pathol Oral Radiol 120(4):508–512. https://doi.org/10.1016/j.oooo.2015.07.033
Chogle S, Zuaitar M, Sarkis R, Saadoun M, Mecham A, Zhao Y (2020) The recommendation of cone-beam computed tomography and its effect on endodontic diagnosis and treatment planning. J Endod 46(2):162–168. https://doi.org/10.1016/j.joen.2019.10.034
Okano T, Harata Y, Sugihara Y, Sakaino R, Tsuchida R, Iwai K, Seki K, Araki K (2009) Absorbed and effective doses from cone beam volumetric imaging for implant planning. Dentomaxillofac Radiol 38(2):79–85. https://doi.org/10.1259/dmfr/14769929
Pauwels R (2015) Cone beam CT for dental and maxillofacial imaging: dose matters. Radiat Prot Dosimetry 165(1–4):156–161. https://doi.org/10.1093/rpd/ncv057
Venskutonis T, Plotino G, Juodzbalys G, Mickeviciene L (2014) The importance of cone-beam computed tomography in the management of endodontic problems: a review of the literature. J Endod 40(12):1895–1901. https://doi.org/10.1016/j.joen.2014.05.009
R Pauwels, L Cockmartin, D Ivanauskaite, A Urboniene, S Gavala, C Donta, K Tsiklakis, R Jacobs, H Bosmans, R Bogaerts, K Horner, SP Consortium (2014) Estimating cancer risk from dental cone-beam CT exposures based on skin dosimetry. Phys Med Biol 59(14):3877–91. https://doi.org/10.1088/0031-9155/59/14/3877
Kleinerman RA (2006) Cancer risks following diagnostic and therapeutic radiation exposure in children. Pediatr Radiol 36(Suppl):2121–2125. https://doi.org/10.1007/s00247-006-0191-5
Applegate KE, Thomas K (2011) Pediatric CT–the challenge of dose records. Pediatr Radiol 41(Suppl):2523–2527. https://doi.org/10.1007/s00247-011-2161-9
Meulepas JM, Ronckers CM, Smets A, Nievelstein RAJ, Gradowska P, Lee C, Jahnen A, van Straten M, de Wit MY, Zonnenberg B, Klein WM, Merks JH, Visser O, van Leeuwen FE, Hauptmann M (2019) Radiation exposure from pediatric ct scans and subsequent cancer risk in the Netherlands. J Natl Cancer Inst 111(3):256–263. https://doi.org/10.1093/jnci/djy104
Yeh JK, Chen CH (2018) Estimated radiation risk of cancer from dental cone-beam computed tomography imaging in orthodontics patients. BMC Oral Health 18(1):131. https://doi.org/10.1186/s12903-018-0592-5
Ariji Y, Ariji E, Nakashima M, Iohara K (2018) Magnetic resonance imaging in endodontics: a literature review. Oral Radiol 34(1):10–16. https://doi.org/10.1007/s11282-017-0301-0
Schuurmans TJ, Nixdorf DR, Idiyatullin DS, Law AS, Barsness BD, Roach SH, Gaalaas L (2019) Accuracy and reliability of root crack and fracture detection in teeth using magnetic resonance imaging. J Endod 45(6):750-755 e2. https://doi.org/10.1016/j.joen.2019.03.008
Juerchott A, Pfefferle T, Flechtenmacher C, Mente J, Bendszus M, Heiland S, Hilgenfeld T (2018) Differentiation of periapical granulomas and cysts by using dental MRI: a pilot study. Int J Oral Sci 10(2):17. https://doi.org/10.1038/s41368-018-0017-y
Lizio G, Salizzoni E, Coe M, Gatto MR, Asioli S, Balbi T, Pelliccioni GA (2018) Differential diagnosis between a granuloma and radicular cyst: effectiveness of magnetic resonance imaging. Int Endod J 51(10):1077–1087. https://doi.org/10.1111/iej.12933
W Leontiev O Bieri P Madorin D Dagassan-Berndt S Kuhl G Krastl R Krug R Weiger T Connert (2021) Suitability of magnetic resonance imaging for guided endodontics: proof of principle. J Endod https://doi.org/10.1016/j.joen.2021.03.011
Iohara K, Fujita M, Ariji Y, Yoshikawa M, Watanabe H, Takashima A, Nakashima M (2016) Assessment of pulp regeneration induced by stem cell therapy by magnetic resonance imaging. J Endod 42(3):397–401. https://doi.org/10.1016/j.joen.2015.11.021
Ploder O, Partik B, Rand T, Fock N, Voracek M, Undt G, Baumann A (2001) Reperfusion of autotransplanted teeth–comparison of clinical measurements by means of dental magnetic resonance imaging. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 92(3):335–340. https://doi.org/10.1067/moe.2001.116505
Prager M, Heiland S, Gareis D, Hilgenfeld T, Bendszus M, Gaudino C (2015) Dental MRI using a dedicated RF-coil at 3 Tesla. J Craniomaxillofac Surg 43(10):2175–2182. https://doi.org/10.1016/j.jcms.2015.10.011
Hilgenfeld T, Prager M, Heil A, Schwindling FS, Nittka M, Grodzki D, Rammelsberg P, Bendszus M, Heiland S (2017) PETRA, MSVAT-SPACE and SEMAC sequences for metal artefact reduction in dental MR imaging. Eur Radiol 27(12):5104–5112. https://doi.org/10.1007/s00330-017-4901-1
Flugge T, Hovener JB, Ludwig U, Eisenbeiss AK, Spittau B, Hennig J, Schmelzeisen R, Nelson K (2016) Magnetic resonance imaging of intraoral hard and soft tissues using an intraoral coil and FLASH sequences. Eur Radiol 26(12):4616–4623. https://doi.org/10.1007/s00330-016-4254-1
Connert T, Hulber JM, Godt A, Lost C, ElAyouti A (2014) Accuracy of endodontic working length determination using cone beam computed tomography. Int Endod J 47(7):698–703. https://doi.org/10.1111/iej.12206
Liang YH, Jiang L, Chen C, Gao XJ, Wesselink PR, Wu MK, Shemesh H (2013) The validity of cone-beam computed tomography in measuring root canal length using a gold standard. J Endod 39(12):1607–1610. https://doi.org/10.1016/j.joen.2013.08.001
Sjogren U, Hagglund B, Sundqvist G, Wing K (1990) Factors affecting the long-term results of endodontic treatment. J Endod 16(10):498–504. https://doi.org/10.1016/S0099-2399(07)80180-4
Ugur Aydin Z, Goller Bulut D (2021) Determination of root canal length up to perforation area using different electronic apex locators and CBCT images obtained at different voxel sizes: a comparative ex vivo study. Chin J Dent Res 24(1):49–54. https://doi.org/10.3290/j.cjdr.b1105877
Norweck JT, Seibert JA, Andriole KP, Clunie DA, Curran BH, Flynn MJ, Krupinski E, Lieto RP, Peck DJ, Mian TA (2013) ACR-AAPM-SIIM technical standard for electronic practice of medical imaging. J Digit Imaging 26(1):38–52. https://doi.org/10.1007/s10278-012-9522-2
Janner SF, Jeger FB, Lussi A, Bornstein MM (2011) Precision of endodontic working length measurements: a pilot investigation comparing cone-beam computed tomography scanning with standard measurement techniques. J Endod 37(8):1046–1051. https://doi.org/10.1016/j.joen.2011.05.005
Kadesjo N, Lynds R, Nilsson M, Shi XQ (2018) Radiation dose from X-ray examinations of impacted canines: cone beam CT vs two-dimensional imaging. Dentomaxillofac Radiol 47(3):20170305. https://doi.org/10.1259/dmfr.20170305
Metska ME, Liem VM, Parsa A, Koolstra JH, Wesselink PR, Ozok AR (2014) Cone-beam computed tomographic scans in comparison with periapical radiographs for root canal length measurement: an in situ study. J Endod 40(8):1206–1209. https://doi.org/10.1016/j.joen.2013.12.036
Segato AVK, Piasecki L, M. Felipe Iparraguirre Nunovero, U.X. da Silva Neto, V.P.D. Westphalen, G. Gambarini, E. Carneiro, (2018) The accuracy of a new cone-beam computed tomographic software in the preoperative working length determination ex vivo. J Endod 44(6):1024–1029. https://doi.org/10.1016/j.joen.2018.02.027
Spin-Neto R, Kruse C, Hermann L, Kirkevang LL, Wenzel A (2020) Impact of motion artefacts and motion-artefact correction on diagnostic accuracy of apical periodontitis in CBCT images: an ex vivo study in human cadavers. Int Endod J 53(9):1275–1288. https://doi.org/10.1111/iej.13326
Jeger FB, Janner SF, Bornstein MM, Lussi A (2012) Endodontic working length measurement with preexisting cone-beam computed tomography scanning: a prospective, controlled clinical study. J Endod 38(7):884–888. https://doi.org/10.1016/j.joen.2012.03.024
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported by a grant from the Dietmar Hopp Foundation (grant number: 1DH2011152).
Author information
Authors and Affiliations
Contributions
M.Z. and T.H. designed the study, collected the data, and analyzed and led the writing; FSS designed the study and collected the data; A.J. collected and analyzed the data; M.N. and Z.H. contributed to methodology and sequence development; JM, HG, and JMEJ contributed to conception and interpretation; S.H. and M.B. contributed to conception, study design, and interpretation.
Corresponding author
Ethics declarations
Ethics approval
This study was performed in accordance with the ethical standards of the University of Heidelberg ethics committee and with the 1964 Helsinki Declaration and its later amendments. This study was approved by the ethics committee of the University of Heidelberg (approval number S-404/2014).
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Conflict of interest
Tim Hilgenfeld, Alexander Juerchott, and Johannes Mente received a grant from the Dietmar Hopp Foundation. Mathias Nittka and Zahra Hosseini are both employees at Siemens Healthcare GmbH. The rest of the authors stated explicitly that there are no conflicts of interest in connection with this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zidan, M., Schwindling, F.S., Juerchott, A. et al. Endodontic working length measurements of premolars and molars in high-resolution dental MRI: a clinical pilot study for assessment of reliability and accuracy. Clin Oral Invest 26, 6765–6772 (2022). https://doi.org/10.1007/s00784-022-04636-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04636-1