Abstract
Objectives
To investigate the histomorphometric changes occurring in alveolar ridge preservation (ARP) based on the use of different plasma concentrates (PCs) in randomized clinical trials (RCT). There is controversy whether the placement of PCs in ARP is effective in the formation of new bone.
Materials and methods
A systematic review search was conducted in PubMed, Scopus, Web of Science, and Cochrane Database to answer the PICO question: In patients undergoing tooth extraction followed by ARP, do PCs alone in the post-extraction socket in comparison with spontaneous healing improve new vital bone formation percentage in histomorphometric analysis after more than 10 weeks? The risk of bias was assessed and a meta-analysis was conducted.
Results
Of 3809 results, 8 studies were considered suitable for inclusion. A total of 255 teeth were extracted in 250 patients. Regarding the PCs used, ARP was performed with platelet- and leukocyte-rich fibrin (L-PRF) in 120 sockets, and with pure platelet-rich plasma (P-PRP) in 31 sockets and 104 sockets were controlled. PCs improved new bone formation in ARP with respect to the spontaneous healing group (SMD = 1.77, 95%C.I. = 1.47–2.06, p-value < 000.1). There were no differences between the different PCs (L-PRF and P-PRP).
Conclusion
The results of this meta-analysis support the efficacy of the use of PCs in new bone formation in ARP. With respect to the different types of PCs studied, no differences were observed.
Clinical relevance
When planning implant surgery after tooth extraction, treatment with PCs should be considered for ARP. Any PC increases new bone formation compared to spontaneous healing.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Implantology is a rapid developing specialty of dentistry. Dental implant is the preferred treatment option for patients with missing teeth [1]. Nowadays, there are many studies assessing new techniques to improve treatment protocols, survival, and predictability of implant treatment [2,3,4].
When a tooth is removed, the alveolar bone undergoes several changes mainly during the first 3 months, that lead to resorption and loss of surrounding bone [5]. Adequate bone quantity and quality is a prerequisite for the success of dental implant treatment [6]. Nowadays, thanks to the studies carried out to improve bone biology, technique, and regenerative materials [7], different treatments have been proposed to reduce bone resorption and improve implant treatment [8,9,10,11,12]. Among these, alveolar ridge preservation (ARP) has shown promising results [13].
ARP includes the use of filling materials in the post-extraction alveolar socket. Many bone substitutes and other biomaterials have been tested for ARP. However, none of them has shown superior results [13,14,15]. In fact, there are studies highlighting that there is no filling material capable of preventing bone resorption completely [16, 17]. And, in a clinical setting, operators must opt for the one which is able to guarantee the best ARP [18].
In the last decade, autologous platelet concentrates (PCs) have been successfully used to this purpose. PCs are obtained by autologous blood centrifugation [19] and have broad applications in regenerative medicine [20] representing a biocompatible and low-cost option [18]. They were firstly introduced in oral and maxillofacial surgery by Whitman et al. in 1997 [21, 22]. Since then, many protocols have been proposed, resulting in various end products with different characteristics [22, 23], which could influence the amount and kinetics of growth factors release, fibrin architecture, and, therefore, clinical outcomes [24,25,26,27]. PC classification is still an important issue in the scientific community [28, 29]. To improve standardization, the classification by Ehrenfest et al. has been introduced [28]. PCs were classified into four groups based on fibrin architecture and leukocyte content. The structure of the fibrin matrix depends on whether or not an anticoagulant is used during preparation. This results in platelet-rich plasma (PRP) if anticoagulant is used and platelet-rich fibrin (PRF) if not. In turn, PRP and PRF may or may not contain leukocytes, giving pure-PRP (P-PRP), leukocyte-rich PRP (L-PRP), pure-PRF (P-PRF), and leukocyte-rich PRF (L-PRF) [24, 30, 31] (Table 1). L-PRF is considered a second generation of PCs [28] and was introduced by Choukroun et al. as a time-saving option compared to PRP [29]. However, PRF may also include leukocytes, which role in inflammation, wound healing, and regeneration is still unclear [26, 32, 33].
PCs have been shown to promote soft tissue healing [52,53,54], whereas the effects on bone tissue remain controversial [52, 55,56,57,58]. While some studies reported improved bone filling, increased bone density, and less ridge width reduction [52, 53, 59,60,61,62,63], others did not [56, 64, 65]. This scenario may be the result of the different protocols used and, therefore, of the different characteristics of each PC. Despite their wide application in clinical practice, there is heterogeneity among different preparation protocols and it is unclear which PC can lead to better results in vital new bone formation.
From this point of view, the aim of this systematic review and meta-analysis was to investigate the histomorphometric changes occurring in ARP based on the use of different PCs in a randomized clinical trial setting.
Materials and methods
Registration of this systematic review and meta-analysis was performed in the PROSPERO database (Registration No.: CRD42022340941). Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines were followed [66].
Search strategy and database screening
A literature search was conducted in the following databases: PubMed, Scopus, and Web of Science, and Cochrane Database. The first inspection was conducted on June 2, 2022. Retrieved results were updated during last search, performed on December 19, 2022. In each database, a combination of keywords and terms was input to generate an ad-hoc search strategy. The search strategies used for each database are shown in Supplementary Table 1. Resulting references were downloaded and uploaded in EndNote software (EndNote X9.3.2, Clarivate Analytics), which automatically removed the duplicates. Resulting list was furtherly manually screened for extra duplicates.
Eligibility criteria
The list of references and abstracts resulting from the search were examined. Studies meeting the following inclusion criteria were selected: (1) no restrictions on publication year; (2) English publication language; (3) only randomized clinical trials, also with a split-mouth design; (4) involving patients over 18 years of age; and (5) requiring non-traumatic tooth extraction. Specifically, the eligible study had to address the population (P), intervention (I), comparison (C), and outcome (O) [67] question described below:
-
(P): To include patients undergoing tooth extraction followed by ARP.
-
(I): ARP was performed by the addition of PCs, for example, PRP or PRF alone in the post-extraction socket.
-
(C): Post-extraction sockets were left without any ARP and spontaneous healing was observed.
-
(O): Suitable studies evaluated as outcome the effects of healing (ARP with PCs versus spontaneous healing) in terms of new vital bone formation percentage by histomorphometric analysis. The minimum follow-up required of 10 weeks was set to take into account the bone tissue physiology healing process, in which most dimensional alterations take place in the first 3 months following tooth extraction [68, 69], while greater new vital bone formation occurs later [70, 71].
The exclusion criteria were as follows: (1) Studies including only observations taken before 10 weeks of follow-up after the intervention; (2) Studies including third molars post-extraction sockets; (3)Studies realized in patients undergoing head and neck radiotherapy, patients with bone diseases, patients with immune-systemic diseases or uncontrolled diabetes; (4) Studies on cell-line models or animal models; (5) Studies investigating the combination of PCs with other materials or compared to other materials alone and—or without a spontaneous healing group as comparison; and (6) Case reports, case series, cohort, and case-control studies as study designs without a randomization process of patients.
Reference screening and inclusion
Two authors (VCAC and LBG) independently screened the resulting list for eligible references to be included in this systematic review, according to the inclusion/exclusion criteria listed above. In the first instance, only the title and abstract were assessed, and suitable studies were furtherly evaluated on full-text appraisal. The k-agreement calculation was evaluated to rank the reviewer’s agreement. A k-agreement of 0.77 showed excellent agreement between the two reviewers. A third author (JGS) participated in this phase to resolve discrepancies.
Data extraction
Independently, two reviewers (VCAC and LBG) performed data extraction based on items collected in ad-hoc extraction Excel sheets. The two reviewers, in a joint meeting with a third reviewer (JGS), merged the extraction Excel files to find for discrepancies, which were fixed in the same meeting after full-text evaluation.
The following information were recorded:
-
First author, year of publication, and country where the study was performed.
-
Study design.
-
Type of PCs: P-PRP, P-PRF, L-PRP, or L-PRF.
-
Characteristics of the patients: included number of patients, gender, mean age (Standard Deviation (S.D.) or range), smoking habit, and periodontal status.
-
Information about the tooth extraction: teeth extracted, the reason for extraction, information about the extraction procedure (with or without flap, type of suture), and the number of walls in the socket.
-
Information about the biopsy sampling, histomorphometric protocol, outcomes collected, and follow-up(s) in weeks.
-
Platelet concentrates protocols: use of anticoagulants, use of activators, and cycles of centrifugation, speed, and time.
-
New bone formation: number of tooth sockets treated for each group, new bone formation percentage expressed as mean and S.D.
Risk of bias assessment
The analysis of the risk of bias of the studies included was performed according to the Cochrane Risk of Bias in randomized interventional studies tool (RoB 2) in the last version, dated 22 August 2019 [72]. The assessment was specific to estimate the relative effect of two interventions on a target outcome. All participants underwent atraumatic tooth extraction and ARP using PCs (intervention) versus the physiological healing by a regular blood clot (control) in order to assess the percentage of new formed bone (outcome).
Concerning split-mouth design studies, RoB assessment was performed adopting an extension of the CONSORT guidelines for withing person trials [73].
RoB was performed independently by two authors (VCAC and LBG) and disagreements were solved in a joint meeting with a third reviewer (JGS).
Statistical analysis and data pooling
A meta-analysis was performed for pooled percentages of new vital bone formation for both RCTs and split-mouth RCT design studies. A meta-epidemiological study did not provide sufficient evidence for a difference in intervention effect estimates between parallel-arm RCTs and RCT-split mouth design studies, so a meta-analysis was performed including both study designs. However, subgroup analysis was also performed [74]. Data were input as mean values of percentages of new vital bone formation with respective S.Ds. and sample size for the control group versus the test group. In the study of Castro et al. [39] two different protocols of PCs were used, however, resulting in both in L-PRF. For this reason, the means and S.Ds. of both groups were combined in contrast to the control, employing the formula from the Cochrane Handbook for Systematic Reviews of Interventions version 6.3 [75].
Overall standardized mean difference (SMD) and relative 95% confidence interval (95% C.I.) were estimated by Hedges’ g weighted data and were graphically represented by forest plots in a fixed or random effect model, based on heterogeneity. Heterogeneity between studies was assessed by Cochran’s Q test and quantified by the I2 index. For I2 values higher than 50%, a random model was set, whether for lower values a fixed effect model was adopted [76]. Heterogeneity was furtherly evaluated by investigating differences among studies and was grouped as moderators, in particular, sensitivity analysis was run for (1) follow-up(s) in weeks; (2) type of PCs as L-PRF and P-PRP; (3) publication year; and (4) study design as RCT versus RCT split-mouth. ANOVA Q-test was used to assess statistically significant differences among subgroups [77].
To inspect the influence of individual studies on overall standardized mean difference, leaving one out method was employed [78]. In the last instance, a funnel plot was generated to graphically visualize the publication bias and was integrated by trim and fill analysis [79], Egger’s test [80], and the safe N test [81].
Trial Sequential Analysis [82] was employed to evaluate the strength of evidence and adjust for potential errors. The TSA software was used in its version 0.9 beta from the Copenhagen Trial Unit. The analysis set specific values for type 1 and 2 errors (5% and 10%) and used these values to calculate trial sequential monitoring boundaries, futility boundaries, and the required information size (RIS) [83, 84]. The mean difference to generate RIS was user-defined with the objective of detecting a mean difference of 7% of new vital bone formation between the test and control. The variance was based on an empirical model. The study also applied a model variance-based approach to correct for heterogeneity and used a graphical evaluation to determine if the cumulative Z-curve met defined thresholds [85].
Results
Search strategy and screening
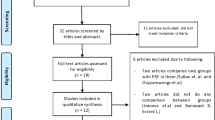
The last search in the mentioned databases yielded 3809 results (PubMed = 1788, Scopus = 744, Web of Science = 976, Cochrane Database = 301). These references were integrated into the EndNote reference software tool (Endnote X9.3.2, Clarivate Analytics). Once duplicates were removed, the titles and abstracts of a total of 2376 references were examined and 2316 were excluded. Sixty references were evaluated in the full text, and of these 52 studies were excluded (the list and rationale for exclusion are summarized in Supplementary Table 2). In the end, 8 studies were considered suitable for inclusion in the systematic review and meta-analysis. Figure 1 shows the flowchart.
General characteristics of included studies
The selection process resulted in 8 studies eligible for meta-analysis [37, 39, 40, 42, 57, 86,87,88]. The studies were published between 2015 and 2021. The studies were mostly performed in Europe [39, 42, 57, 86, 87], but two studies were performed in Brazil [40, 88], and one in South Africa [37].
Clinical characteristics are summarized in Table 2. A total of 6 studies adopted a RCT design, and two studies a split-mouth design [37, 39]. ARP was most frequently performed in the sockets of single-rooted teeth in the upper anterior maxilla. L-PRF was the most commonly investigated PC and only two studies employed PRP [57, 87]. All studies used sutures to stabilize clots, most without primary closure intention. Two studies completely covered the extraction site with a mucoperiosteal flap [42, 86]. One study sealed the sockets with L-PRF membranes placed in full-thickness buccal and lingual/palatal envelopes [39]. Only one study included heavy smokers (≥ 10 cigarettes/day), representing a total of 6 patients evenly distributed across the control and test groups [57]. This was the only study [57] that specifically included patients with periodontal disease, present in 52.7% and 45.8% of the patients assigned to the experimental and control groups, respectively. The percentage of new bone formation was evaluated after 10–24 weeks of follow-up.
Biopsy sampling was performed at the time of implant placement in all the studies. Before implant placement, a trephine bur was employed to collect the bone sample. The diameter ranged from 2 to 3 mm and the length from 4 to 7 mm, but Ivanova et al. studies and Castro et al. did not report biopsy sample length. Histomorphometric protocol differed among studies. The detailed histomorphometric protocol is reported in Table 3.
Concerning PCs preparation (Table 4), different centrifugation protocols emerged, both in time of centrifugation and revolutions per minute of the rotor (rpm) or relative centrifugal force (g). Even in similar reported rpm, g changed because of the different radius of rotors employed for the centrifugation.
In total 255 teeth were extracted and ARP was performed with L-PRF in 120 sockets and with P-PRP in 31 sockets. On the other hand, 104 sockets were left healing spontaneously. These procedures were performed in 225 patients from studies with an RCT design and 25 patients from studies with a split-mouth RCT design. Previous information and the mean and S.Ds of new bone formation percentages of each group in all included studies are reported in Table 5.
Risk of bias
Table 6 summarizes the risk of bias results for each study. Among RCTs, 3 studies showed some concerns in the randomization process. No information in this regard was reported in the Ivanova et al. study [86]. While in the other two studies, it was unclear whether the allocation sequence was concealed until participants were enrolled and assigned to interventions [87, 88]. When assessing “deviation from intervention”, no indications were provided in the study of Ivanova et al. [86], for this reason, this study was evaluated at high risk of bias. All the studies resulted in a low risk of bias in the “missing outcome data” item. The study by Ivanova et al. was the only one with a high risk of bias in the item “measurement of the outcome”, as it did not indicate whether the assessors collecting the results were aware of the intervention received by the study participants [86]. The “Selection of reported results” item had some concerns in three studies, since a pre-specified analysis plan for data was not indicated [42, 86, 88]. Overall, only one study resulted in a low risk of bias [40], three reported some concerns [42, 57, 87], while two studies were at high risk of bias [86, 88].
For Castro et al. [39] and Du Toit et al. [37] 46 items were evaluated from merging the standard CONSORT guideline checklist and the one for withing person trial [73]. Du Toit et al. and Castro et al. scored respectively 38 and 36 points. Du Toit et al. failed to report “other information” because the description of registration, protocol, and funding was missing. Castro et al. failed to provide information in different subfields of “Methods” (items and scores are collected in Supplementary Tables 3 and 4 for each study). Both studies were attributed to “some concerns” [37, 39].
Meta-analysis and trial sequential analysis
Heterogeneity results showed average heterogeneity among studies (I2 = 44.05%). Fixed effect model meta-analysis showed a positive SMD in patients undergoing ARP with PCs with respect to the spontaneous healing group (SMD = 1.77, 95% C.I. = 1.47–2.06, p-value < 000.1, Fig. 2). Heterogeneity among studies was furtherly investigated by leave-one-out method (Fig. 3), which showed different SMD after elimination of Castro et al. (SMD = 1.65, 95% C.I. = 1.32–1.98, p-value < 0.001) and Du Toit et al. (SMD = 1.87, 95% C.I. = 1.56–2.18, p-value < 000.1) studies, which were the only two studies with a split-mouth design.
Subgroup meta-analysis considering RCTs and split-mouth design RCTs separately showed that study design accounted partially for heterogeneity, since I2 statistics resulted in 0 only in the RCT subgroup. Similar overall fixed SMD was achieved from RCTs (SMD = 1.77, 95% C.I. = 1.42–2.11, p-value < 0.001; vs split-mouth SMD = 1.76, 95% C.I. = 1.18–2.35, p-value < 0.001; ANOVA Q-test p-value = 0.987—Supplemental Fig. 1). Indeed, differences emerged when investigating PC subtypes, in particular L-PRF and P-PRP. The absence of heterogeneity was found in the P-PRP subgroup analysis. However, the L-PRF subgroup included both split-mouth designs, raising I2 = 58.10%. However the difference in SMD between P-PRP and L-PRF was not statistically significant (L-PRF SMD = 1.75, 95% C.I. = 1.21–2.28, p-value < 0.001; vs P-PRP SMD = 1.58, 95% C.I. = 0.87–2.29, p-value < 0.001; ANOVA Q-test p-value = 0.71—Supplemental Fig. 2).
Surprisingly, meta-regression for publication year found a statistically significant difference in SMDs (fixed effect model p-value = 0.048). Meta-regression did not find a statistically significant difference between SMDs and follow-up, besides it seemed to increase for studies with higher follow-ups (fixed effect model p-value = 0.27—Fig. 4).
The absence of publication bias was highlighted by the trim and fill method which found 0 trimmed studies and graphically represented by funnel plot (Fig. 5). Also Egger’s linear regression test showed an absence of publication bias (p-value = 0.44). Moreover, the safe N test required the publication of 239 studies to revert the current overall SMD p-value.
TSA analysis approved results from meta-analysis after the addition of the third study and confirmed the efficacy of PCs versus spontaneous healing in determining a greater formation of vital bone percentage, since the cumulative z-curve crossed the monitoring boundaries. After the addition of the sixth study, RIS was reached, providing a conclusive statistically significant difference in the results for this meta-analysis (Fig. 6).
Discussion
This systematic review and meta-analysis with TSA showed conclusive results in the efficacy of PCs in new bone formation in ARP with respect to the spontaneous healing group (SMD = 1.77, 95% C.I. = 1.47–2.06, p-value < 000.1). Furthermore, the results of our study observed that there was no difference between the use of the different PCs included (P-PRP and L-PRF).
After tooth extraction, significant alveolar bone remodeling has been documented, leading to a decrease in alveolar height and width mainly at the expense of the vestibular plate [5]. This situation could influence the proper three-dimensional placement of implant-supported restorations as well as the esthetics mainly in the anterior sector. Therefore, one of the main goals of oral implantology is the preservation of the remining healthy bone after tooth extraction using highly predictable procedures [6]. After the first RCT about the success of P-PRP in APR compared to spontaneous healing by Anitua et al. in 2015 [57], new published evidence support the use of PCs in ARP. In the network meta-analysis published by Canellas et al. more than twenty materials were compared in ARP. L-PRF showed no statistically significant differences in ARP with the other best-performing graft materials (MP3®, Apatos®, Gen-Os® and Bond-apatite®).
The inconclusive results about the use of PCs in ARP may be due to the low number of studies using PCs without a xenograft, since most of the studies included combinations of materials which could modify the biological properties of PCs. According to previous studies, the use of PCs have certain advantages such as rapid reabsorption and formation of new trabecular bone while promoting healing due to abundant growth factors. Possibly, a good choice is the combination of a low resorbable material, as xenografts, that preserves the volume of the socket, together with another material that favors the formation of new bone to promote osseointegration and primary stability [89]. This approach is supported by other studies who have reported that ARP with any material is superior to spontaneous healing, and the use of different scaffold materials could favor the reduction of postextraction socket volume [13]. In addition, the application of PCs could improve the healing of the area increasing the formation of new bone [13, 18, 89]. Our study shows that the use of PCs in ARP, regardless of the type of PCs used, improves bone formation compared to spontaneous healing. This amount of neoformed bone must be taken into account in terms of its therapeutic significance. In any case, it should be noted that in our meta-analysis alveolar remodeling measures were not taken into consideration. This could be a limitation of this study, as current knowledge in ARP considers the formation of new vital bone and the preservation of ridge dimension together. This is because both processes can influence primary and secondary implant stability and osseointegration. To overcome this limitation, it is necessary to include studies that combine a xenograft with PCs that may increase the formation of new vital bone, compared to allograft alone. But, this does not allow us to know what effect PCs alone have on bone regeneration [90, 91].
To evaluate differences between the different PCs, it is necessary to unravel their biological behavior. Bone regeneration needs a complex coordination between cytokines, proteins, and grow factors (GFs), and the controlled release of these bioactive substances seems to play a major role in this process. Many studies analyze the release kinetics of GFs from PCs, but there is enormous variability among authors in reporting these results. It has been suggested that these observed differences in the controlled release of GFs from different PCs depend on the architecture of the fibrin matrix and its degree of cross-linking. Some studies consider that L-PRF produces a progressive release of growth factors, whereas PRP triggers a cascade release in the first hours [25, 31]. In contrast, other studies suggest the opposite based on a more rapid degradation of the fibrin matrix of L-PRF due to proinflammatory metalloproteinases produced by leukocytes [26]. In this meta-analysis, further considerations emerged. The scientific scenario offers a wide number of PC types and protocols, increasing heterogeneity. Changes in rotor diameter, number of spins, time and speed of centrifugation could contribute to different biologic characteristics of PCs, even though classified in the same group as P- L-PRP/PRF. In a rat model, different protocols for L-PRP preparation were employed, leading to differences in platelets and minerals concentrations, which impacted significally in reducing the bone defects [27]. This phenomenon, however, is still controversial and limited to short follow-up of bio-molecular events [26] since bone healing is a longer process [68,69,70,71]. Indeed, meta-regression showed an increased of new bone formation when measurements were done at longer follow-ups. In any case, no differences could be found in this study between the two PCs analyzed (P-PRP and L-PRF). Therefore, the PC with the simplest and cheapest technique should be used. Normally, the PRF technique is simpler but it has the disadvantage that it is not useful to vehicle other biomaterials. Therefore, depending on the ARP technique to be performed, the clinician will have to decide which one to use.
This study has certain limitations. Only two studies analyzed the use of P-PRP, so that in the future it would be convenient to perform more studies with this type of PC. Another limitation is that PCs protocols differed among studies and the outcome was observed at different follow-ups. Also the inclusion of two split-mouth RCTs can have an impact in the results. But, different analyses have been performed to minimize these issues. It is also worth considering that only new vital bone formation analyzed by histomorphometry was evaluated as an outcome in this study. Although it is expected that a higher percentage of vital bone will result in more bone tissue being available at the time of implant surgery, this is uncertain. And there were no other variables associated with clinical, function or treatment success analyzed in all the studies [92, 93]. It is worth considering that only Anitua et al. collected patient-reported outcomes among the included studies. The P-PRP group showed a statistically significant reduction in reported pain in the first week after extraction, supported by a lower inflammation score. It is necessary to analyze in future studies clinical variables such as changes in height and width, as well as outcomes associated with the patient's perspective such as pain or oral health-related quality of life.
Another limitation of this study is the great heterogeneity, since certain characteristics were different among the studies. These included data on smoking, periodontal status, included teeth (uni- or multiradicular), number of bony walls of the defects, and type of surgery (with or without flap). All these differences constitute potential confounding factors.
Conclusion
In conclusion, PCs are widely used in clinical practice, despite poor standardization and deep knowledge of molecular events happening in the healing process. Current evidence coming from this meta-analysis of RCTs supports the efficacy of PCs in the new bone formation process, compared to spontaneous healing. P-PRP has been tested in only two RCTs, while L-PRF has found wide application. However, subgroup analysis did not show a statistical difference between these two different PCs.
Data availability
Data to perform these studies are available after a justified request to the authors.
Materials availability
Not applicable
References
Hunt PR, Gartner JL, Norkin FJ (2005) Choice of a dental implant system. Compend Contin Educ Dent 26(4):239–40 (242, 245-8 passim; quiz 251, 271)
Saito H, Aichelmann-Reidy MB, Oates TW (2020) Advances in implant therapy in North America: improved outcomes and application in the compromised dentition. Periodontol 2000 82(1):225–237
Salgado-Peralvo AO, Garcia-Sanchez A, Kewalramani N, Barone A, Martínez-González JM, Velasco-Ortega E, López-López J, Kaiser-Cifuentes R, Guerra F, Matos-Garrido N, Moreno-Muñoz J, Núñez-Márquez E, Ortiz-García I, Jiménez-Guerra Á, Monsalve-Guil L (2022) Consensus report on preventive antibiotic therapy in dental implant procedures: summary of recommendations from the Spanish Society of Implants. Antibiotics (Basel) 11(5):655. https://doi.org/10.3390/antibiotics11050655
Sanz M, Heitz-Mayfield L (2022) Consensus report of DGI/SEPA/osteology workshop. Clin Oral Implants Res 33(Suppl 23):3–7
Schropp L et al (2003) Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent 23(4):313–323
Chrcanovic BR, Albrektsson T, Wennerberg A (2017) Bone quality and quantity and dental implant failure: a systematic review and meta-analysis. Int J Prosthodont 30(3):219–237
Ono T, Nakashima T (2022) Oral bone biology. J Oral Biosci 64(1):8–17
Baumer D et al (2017) Socket shield technique for immediate implant placement - clinical, radiographic and volumetric data after 5 years. Clin Oral Implants Res 28(11):1450–1458
Hurzeler MB et al (2010) The socket-shield technique: a proof-of-principle report. J Clin Periodontol 37(9):855–862
Joo JY, Son S, Lee JY (2016) Implant site development for enhancing esthetics of soft and hard tissue and simplification of implant surgery using a forced eruption. Int J Periodontics Restorative Dent 36(4):583–589
Salama H, Salama M (1993) The role of orthodontic extrusive remodeling in the enhancement of soft and hard tissue profiles prior to implant placement: a systematic approach to the management of extraction site defects. Int J Periodontics Restorative Dent 13(4):312–333
Saez-Alcaide LM et al (2021) Socket shield technique: a systematic review of human studies. Ann Anat 238:151779
Canullo L et al (2022) Dimensional and histomorphometric evaluation of biomaterials used for alveolar ridge preservation: a systematic review and network meta-analysis. Clin Oral Investig 26(1):141–158
Tan-Chu JH et al (2014) Analysis of buccolingual dimensional changes of the extraction socket using the “ice cream cone” flapless grafting technique. Int J Periodontics Restorative Dent 34(3):399–403
Atieh MA et al (2021) Interventions for replacing missing teeth: alveolar ridge preservation techniques for dental implant site development. Cochrane Database Syst Rev 4:CD010176
Avila-Ortiz G et al (2014) Effect of alveolar ridge preservation after tooth extraction: a systematic review and meta-analysis. J Dent Res 93(10):950–958
Morjaria KR, Wilson R, Palmer RM (2014) Bone healing after tooth extraction with or without an intervention: a systematic review of randomized controlled trials. Clin Implant Dent Relat Res 16(1):1–20
Canellas J et al (2021) What grafting materials produce greater alveolar ridge preservation after tooth extraction? A systematic review and network meta-analysis. J Craniomaxillofac Surg 49(11):1064–1071
Al-Hamed FS et al (2019) Regenerative effect of platelet concentrates in oral and craniofacial regeneration. Front Cardiovasc Med 6:126
Moscicka P, Przylipiak A (2021) History of autologous platelet-rich plasma: a short review. J Cosmet Dermatol 20(9):2712–2714
Whitman DH, Berry RL, Green DM (1997) Platelet gel: an autologous alternative to fibrin glue with applications in oral and maxillofacial surgery. J Oral Maxillofac Surg 55(11):1294–1299
Xu J et al (2020) Platelet-rich plasma and regenerative dentistry. Aust Dent J 65(2):131–142
Grambart ST (2015) Sports medicine and platelet-rich plasma: nonsurgical therapy. Clin Podiatr Med Surg 32(1):99–107
He L et al (2009) A comparative study of platelet-rich fibrin (PRF) and platelet-rich plasma (PRP) on the effect of proliferation and differentiation of rat osteoblasts in vitro. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 108(5):707–713
Kobayashi E et al (2016) Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin Oral Investig 20(9):2353–2360
Baca-Gonzalez L et al (2022) Plasma rich in growth factors (PRGF) and leukocyte-platelet rich fibrin (L-PRF): comparative release of growth factors and biological effect on osteoblasts. Int J Implant Dent 8(1):39
Al-Hamed FS et al (2021) Differences in platelet-rich plasma composition influence bone healing. J Clin Periodontol 48(12):1613–1623
Dohan Ehrenfest DM, Rasmusson L, Albrektsson T (2009) Classification of platelet concentrates: from pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol 27(3):158–67
Dohan Ehrenfest DM et al (2010) Shedding light in the controversial terminology for platelet-rich products: platelet-rich plasma (PRP), platelet-rich fibrin (PRF), platelet-leukocyte gel (PLG), preparation rich in growth factors (PRGF), classification and commercialism. J Biomed Mater Res A 95(4):1280–1282
Davis VL et al (2014) Platelet-rich preparations to improve healing. Part I: workable options for every size practice. J Oral Implantol 40(4):500–10
Dohan Ehrenfest DM et al (2012) Do the fibrin architecture and leukocyte content influence the growth factor release of platelet concentrates? An evidence-based answer comparing a pure platelet-rich plasma (P-PRP) gel and a leukocyte- and platelet-rich fibrin (L-PRF). Curr Pharm Biotechnol 13(7):1145–1152
Anitua E et al (2015) Leukocyte inclusion within a platelet rich plasma-derived fibrin scaffold stimulates a more pro-inflammatory environment and alters fibrin properties. PLoS ONE 10(3):e0121713
Filardo G et al (2012) Platelet-rich plasma intra-articular injections for cartilage degeneration and osteoarthritis: single- versus double-spinning approach. Knee Surg Sports Traumatol Arthrosc 20(10):2082–2091
Anitua E et al (2020) The influence of sodium citrate on the characteristics and biological activity of plasma rich in growth factors. Regen Med 15(10):2181–2192
Tamimi FM et al (2007) A comparative study of 2 methods for obtaining platelet-rich plasma. J Oral Maxillofac Surg 65(6):1084–1093
Marx RE et al (1998) Platelet-rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 85(6):638–646
Du Toit J et al (2016) Choukroun platelet-rich fibrin as an autogenous graft biomaterial in preimplant surgery: results of a preliminary randomized, human histomorphometric, split-mouth study. Int J Periodontics Restorative Dent 36(Suppl):s75-86
Dohan DM et al (2006) Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part III: leucocyte activation: a new feature for platelet concentrates? Oral Surg Oral Med Oral Pathol Oral Radiol Endod 101(3):e51-5
Castro AB et al (2021) Effect of different platelet-rich fibrin matrices for ridge preservation in multiple tooth extractions: A split-mouth randomized controlled clinical trial. J Clin Periodontol 48(7):984–995
Canellas J et al (2020) Tomographic and histomorphometric evaluation of socket healing after tooth extraction using leukocyte- and platelet-rich fibrin: A randomized, single-blind, controlled clinical trial. J Craniomaxillofac Surg 48(1):24–32
Weibrich G, Kleis WK (2002) Curasan PRP kit vs. PCCS PRP system. Collection efficiency and platelet counts of two different methods for the preparation of platelet-rich plasma. Clin Oral Implants Res 13(4):437–43
Ivanova V, Chenchev I, Zlatev S, Mijiritsky E (2021) Comparison study of the histomorphometric results after socket preservation with PRF and allograft used for socket preservation-randomized controlled trials. Int J Environ Res Public Health 18(14):7451. https://doi.org/10.3390/ijerph18147451
Weibrich G et al (2003) The harvest smart PRePTM system versus the Friadent-Schutze platelet-rich plasma kit. Clin Oral Implants Res 14(2):233–239
Lee C et al (2009) A quantitative radiological assessment of outcomes of autogenous bone graft combined with platelet-rich plasma in the alveolar cleft. Int J Oral Maxillofac Surg 38(2):117–125
Jovani-Sancho MD et al (2016) Platelet-rich plasma: a study of the variables that may influence its effect on bone regeneration. Clin Implant Dent Relat Res 18(5):1051–1064
Gnatek A, Janas-Naze A (2019) The influence of application of concentrated growth factors on post-operative complications – in vivo split-mouth study. J Stomatol 72(4):147–152
Ma F et al (2021) The impact of autologous concentrated growth factors on the alveolar ridge preservation after posterior tooth extraction: a prospective, randomized controlled clinical trial. Clin Implant Dent Relat Res 23(4):579–592
Rodella LF et al (2011) Growth factors, CD34 positive cells, and fibrin network analysis in concentrated growth factors fraction. Microsc Res Tech 74(8):772–777
Zhang Z et al (2019) Effect of autogenous growth factors released from platelet concentrates on the osteogenic differentiation of periodontal ligament fibroblasts: a comparative study. PeerJ 7:e7984
Mourao CF et al (2015) Obtention of injectable platelets rich-fibrin (i-PRF) and its polymerization with bone graft: technical note. Rev Col Bras Cir 42(6):421–423
Wang X et al (2018) Effects of an injectable platelet-rich fibrin on osteoblast behavior and bone tissue formation in comparison to platelet-rich plasma. Platelets 29(1):48–55
Suttapreyasri S, Leepong N (2013) Influence of platelet-rich fibrin on alveolar ridge preservation. J Craniofac Surg 24(4):1088–1094
Marenzi G et al (2015) Influence of Leukocyte- and Platelet-Rich Fibrin (L-PRF) in the Healing of Simple Postextraction Sockets: A Split-Mouth Study. Biomed Res Int 2015:369273
Anitua E, Pino A, Orive G (2016) Plasma rich in growth factors promotes dermal fibroblast proliferation, migration and biosynthetic activity. J Wound Care 25(11):680–687
Chakravarthi S (2017) Platelet rich fibrin in the management of established dry socket. J Korean Assoc Oral Maxillofac Surg 43(3):160–165
Pan J et al (2019) Effect of platelet-rich fibrin on alveolar ridge preservation: a systematic review. J Am Dent Assoc 150(9):766–778
Anitua E et al (2015) Clinical, radiographical, and histological outcomes of plasma rich in growth factors in extraction socket: a randomized controlled clinical trial. Clin Oral Investig 19(3):589–600
Badran Z et al (2018) Platelet concentrates for bone regeneration: current evidence and future challenges. Platelets 29(2):105–112
Alzahrani AA, Murriky A, Shafik S (2017) Influence of platelet rich fibrin on post-extraction socket healing: A clinical and radiographic study. Saudi Dent J 29(4):149–155
Hauser F et al (2013) Clinical and histological evaluation of postextraction platelet-rich fibrin socket filling: a prospective randomized controlled study. Implant Dent 22(3):295–303
Das S et al (2016) Socket preservation by beta-tri-calcium phosphate with collagen compared to platelet-rich fibrin: A clinico-radiographic study. Eur J Dent 10(2):264–276
Temmerman A et al (2016) The use of leucocyte and platelet-rich fibrin in socket management and ridge preservation: a split-mouth, randomized, controlled clinical trial. J Clin Periodontol 43(11):990–999
Thakkar DJ et al (2016) A comparative evaluation of extraction socket preservation with demineralized freeze-dried bone allograft alone and along with platelet-rich fibrin: a clinical and radiographic study. Contemp Clin Dent 7(3):371–376
Bennardo F et al (2021) Efficacy of platelet-rich fibrin compared with triamcinolone acetonide as injective therapy in the treatment of symptomatic oral lichen planus: a pilot study. Clin Oral Investig 25(6):3747–3755
Strauss FJ, Stahli A, Gruber R (2018) The use of platelet-rich fibrin to enhance the outcomes of implant therapy: A systematic review. Clin Oral Implants Res 29(Suppl 18):6–19
Page MJ et al (2021) PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ 372:n160
Guiglia R et al (2007) A combined treatment regimen for desquamative gingivitis in patients with oral lichen planus. J Oral Pathol Med 36(2):110–116
Araujo MG, Lindhe J (2005) Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol 32(2):212–8
Srinivas B et al (2018) Wound Healing and Bone Regeneration in Postextraction Sockets with and without Platelet-rich Fibrin. Ann Maxillofac Surg 8(1):28–34
Whetman J, Mealey BL (2016) Effect of healing time on new bone formation after tooth extraction and ridge preservation with demineralized freeze-dried bone allograft: a randomized controlled clinical trial. J Periodontol 87(9):1022–1029
Beck TM, Mealey BL (2010) Histologic analysis of healing after tooth extraction with ridge preservation using mineralized human bone allograft. J Periodontol 81(12):1765–1772
Sterne JAC et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898
Pandis N et al (2017) CONSORT 2010 statement: extension checklist for reporting within person randomised trials. BMJ 357:j2835
Smail-Faugeron V et al (2014) Comparison of intervention effects in split-mouth and parallel-arm randomized controlled trials: a meta-epidemiological study. BMC Med Res Methodol 14:64
Cumpston MS et al (2022) Strengthening systematic reviews in public health: guidance in the Cochrane Handbook for Systematic Reviews of Interventions, 2nd edition. J Public Health (Oxf) 44(4):e588–e592
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558
Altobelli E, Angeletti PM, Marziliano C, Mastrodomenico M, Giuliani AR, Petrocelli R (2021) Potential Therapeutic Effects of Curcumin on Glycemic and Lipid Profile in Uncomplicated Type 2 Diabetes-A Meta-Analysis of Randomized Controlled Trial. Nutrients 13(2):404. https://doi.org/10.3390/nu13020404
Willis BH, Riley RD (2017) Measuring the statistical validity of summary meta-analysis and meta-regression results for use in clinical practice. Stat Med 36(21):3283–3301
Duval S, Tweedie R (2000) Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56(2):455–463
Egger M et al (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315(7109):629–634
Persaud R (1996) Misleading meta-analysis. “Fail safe N” is a useful mathematical measure of the stability of results. BMJ 312(7023):125
Pisalsitsakul N et al (2022) Taking 200 mg vitamin C three times per day improved extraction socket wound healing parameters: a randomized clinical trial. Int J Dent 2022:6437200
Kang H (2021) Trial sequential analysis: novel approach for meta-analysis. Anesth Pain Med (Seoul) 16(2):138–150
Wetterslev J, Jakobsen JC, Gluud C (2017) Trial sequential analysis in systematic reviews with meta-analysis. BMC Med Res Methodol 17(1):39
Miladinovic B et al (2013) Optimal information size in trial sequential analysis of time-to-event outcomes reveals potentially inconclusive results because of the risk of random error. J Clin Epidemiol 66(6):654–659
Ivanova V, Chenchev I, Zlatev S, Kanazirski N (2019) Dimensional ridge alterations and histomorphometric analysis following socket preservation with prf or allograft. randomized controlled clinical study. J IMAB - Ann Proc (Scientific Papers) 25(4):2853–2861
Stumbras A et al (2020) Randomized and controlled clinical trial of bone healing after alveolar ridge preservation using xenografts and allografts versus plasma rich in growth factors. J Oral Implantol 46(5):515–525
Fontes Martins LC et al (2021) Bone marrow aspirate concentrate and platelet-rich fibrin in fresh extraction sockets: a histomorphometric and immunohistochemical study in humans. J Craniomaxillofac Surg 49(2):104–109
Canellas JVDS, Ritto FG, Figueredo CMDS, Fischer RG, de Oliveira GP, Thole AA, Medeiros PJD (2020) Histomorphometric evaluation of different grafting materials used for alveolar ridge preservation: a systematic review and network meta-analysis. Int J Oral Maxillofac Surg 49(6):797–810. https://doi.org/10.1016/j.ijom.2019.10.007
Clark D, Rajendran Y, Paydar S, Ho S, Cox D, Ryder M, Dollard J, Kao RT (2018) Advanced platelet-rich fibrin and freeze-dried bone allograft for ridge preservation: a randomized controlled clinical trial. J Periodontol 89(4):379–387. https://doi.org/10.1002/JPER.17-0466
Ntounis A et al (2015) Clinical assessment of bone quality of human extraction sockets after conversion with growth factors. Int J Oral Maxillofac Implants 30(1):196–201
Adams RJ (2022) Is there clinical evidence to support alveolar ridge preservation over extraction alone? A review of recent literature and case reports of late graft failure. Br Dent J 233(6):469–474
Mardas N et al (2015) Does ridge preservation following tooth extraction improve implant treatment outcomes: a systematic review: Group 4: Therapeutic concepts & methods. Clin Oral Implants Res 26(Suppl 11):180–201
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Contributions
Concept/Design: JGS, RMLP, JT; Data analysis/interpretation: VCAC, LBG, JGS, RMLP; Drafting article: VCAC, LBG, JGS, and RMLP; Critical revision of the article: JT; Approval of article: VCAC, LBG, JGS, JT, and RMLP; Statistics: VCAC, JGS; Funding secured by: N/A; Data collection: VCAC, LBG, JGS.
Corresponding author
Ethics declarations
Ethical approval
Not applicable
Patient consent
Not applicable
Conflict of interest
The authors declare no competing interest
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Caponio, V.C.A., Baca-González, L., González-Serrano, J. et al. Effect of the use of platelet concentrates on new bone formation in alveolar ridge preservation: a systematic review, meta-analysis, and trial sequential analysis. Clin Oral Invest 27, 4131–4146 (2023). https://doi.org/10.1007/s00784-023-05126-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-023-05126-8