Abstract
B cells play a central role in the pathogenesis of systemic sclerosis (SSc). Most B-cell studies have focused on their pathological role as antibody producers. However, in addition to immunoglobulin secretion, these cells have a wide range of functions in the immune response, including antigen presentation to T cells and cytokine production. Importantly, not all B-cell subsets promote the immune response. Regulatory B cells (Bregs) attenuate inflammation and contribute to the maintenance of immune tolerance. However, effector B cells (Beffs) positively modulate the immune response through the production of various cytokines. In SSc, Bregs are insufficient and/or dysfunctional. B-cell-targeting biologics have been trialled with promising results in the treatment of SSc. These therapies can affect Bregs or Beffs, which can potentially limit their long-term efficacy. Future strategies might involve the modulation of effector B cells in combination with the stimulation of regulatory subsets. Additionally, the monitoring of individual B-cell subsets in patients may lead to the discovery of novel biomarkers that could help predict disease relapse or progression. The purpose of this review is to summarize the relevant literatures and explain how Bregs and Beffs jointly participate in the pathogenesis of SSc.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Systemic sclerosis (SSc, also known as scleroderma) is a connective tissue disease that is characterized by excessive fibrosis of the skin and various internal organs and has an autoimmune aetiology [1]. The pathogenesis of SSc is unclear. More than 90% of SSc patients are positive for autoantibodies, such as anti–DNA topoisomerase I, anticentromere, and anti–RNA polymerase antibody (Ab) [2]. Increasing evidence shows that B cells are involved in SSc. One study demonstrated that SSc patients displayed distinct abnormalities in blood B-cell compartments; these abnormalities were characterized by expanded naïve B cells and activated memory B cells [3]. The sensitive balance between effector B cells (Beffs) and regulatory B cells (Bregs) is disrupted, the number of Bregs is reduced, and Bregs are functionally impaired [4]. Furthermore, studies have shown that interleukin-6 (IL-6) derived from effector B cells can promote fibrosis. However, regulatory B-cell-derived interleukin-10 (IL-10) has anti-inflammatory effects [5].
B cells promote the pathogenesis of SSc through diverse mechanisms, most notably via the production of antibodies, but also through antigen presentation, which leads to T cell activation and the secretion of various cytokines [6]. The crucial role of B cells in SSc is highlighted by the success of therapeutic B-cell depletion strategies [7, 8]. Research in preclinical models, together with insights from clinical studies of B-cell depletion therapy, has improved our understanding of the contribution of B cells to the aetiology and pathogenesis of SSc. B cells are important for Ab production, antigen presentation, and cytokine production [9]. In particular, cytokine-producing B cells play critical roles in multiple aspects of immunity. There are two opposing B-cell subsets: Bregs and Beffs [10]. Bregs are capable of modulating immune responses and contributing to immune tolerance [11]. B-cell depletion therapies target Beffs as well as Bregs; however, to date, no specific method has been developed to regulate Beffs without affecting Bregs. In addition, studies of B-cell subsets and their plasticity in patients with scleroderma are lacking. Understanding B-cell development and differentiation, as well as phenotypic and functional heterogeneity, will make the treatment of SSc by targeting B cells a possibility.
B-cell differentiation and development
The development of B cells begins in the foetal liver and continues in the bone marrow, where multiple checkpoints ensure that B cells released to the periphery express functional B-cell receptors (BCRs) and are not specific for their own antigens [12]. The autoreactivity of BCR can be detected in the immature B-cell stage [13]. Early bone marrow transitional B lymphocytes are also called transitional B cells. These immature B cells express immunoglobulin M (IgM) and immunoglobulin D (IgD) on their surface, which are usually characterized by a CD24hi CD38hi phenotype and a lack of the memory B-cell marker CD27 [14]. Immature B cells develop in the periphery and mature into antigen-naïve CD24int CD38int CD27− B cells [15]. Under antigen-specific stimulation, B cells can differentiate into plasmablasts, plasma cells and memory B cells [16]. Plasma cells are long-lived, terminally differentiated and antibody-producing cells, and plasmablasts are their short-lived proliferative counterparts [17]. These two cell types lack the expression of the pan-B-cell marker CD20 and are collectively referred to as antibody-secreting cells [18]. In the case of reinfection, plasma cells constantly produce antibodies in the bone marrow to provide immediate protection to the host. Memory B cells can be rapidly induced to differentiate into antibody-producing plasma cells through interactions with homologous T cells. This memory response is usually stronger than that of primary B cells to antigens, and these cells produce immunoglobulins with high affinity [19].
The definition of Beffs and Bregs: the lack of a specific phenotype
There are no phenotypic markers or transcription factors that can be used to specifically identify either Beffs or Bregs. However, Beffs and Bregs are defined by the expression of proinflammatory or anti-inflammatory cytokines. Beffs can positively modulate the immune response through the production of various cytokines [20]. For example, lymphotoxin-producing Beffs are essential for the ontogenesis, homeostasis, and the activation of secondary lymphoid organs, as well as the development of tertiary lymphoid tissues at ectopic sites. Other Beffs have been shown to modulate the development of effector and memory CD4+ T-cell responses via the production of cytokines such as IL-6, interferon-γ (IFN-γ), tumour necrosis factor-α (TNF-α) and granulocyte macrophage colony stimulating factor (GM-CSF) 20]. In contrast, IL-10-producing Bregs are now recognized as negative regulators of the immune system, inflammation, and autoimmunity based on studies with human subjects and mouse models of autoimmune diseases such as rheumatoid arthritis, systemic lupus erythematosus (SLE), and multiple sclerosis (MS) [21,22,23] . The phenotype of mouse splenic Bregs is derived from two different B-cell subsets: the marginal zone (MZ) and B1 B cells [24]. Furthermore, the CD9+ B-cell subset is enriched in IL-10-producing Bregs since this subset includes both the MZ and B1 B-cell subsets [25]. Therefore, a protocol that selectively depletes Beffs while sparing Bregs would be a potent therapy for autoimmune diseases.
Effector B cells in SSc
Beffs promote immune responses through the production of various proinflammatory cytokines [26]. In particular, IL-6- or GM-CSF-producing Beffs have emerged as important in the treatment of SSc [27, 28].
B cells are the main source of IL-6 [29]. IL-6 can induce naïve CD4+ T cells to differentiate into T follicular fluid (Tfh) and Th17 cells and induce B cells to differentiate into plasma cells [30, 31]. In addition, IL-6 also plays an important role in SSc. In a bleomycin-induced scleroderma model, IL-6 deficiency in B cells can reduce skin fibrosis, while IL-10 deficiency in B cells can increase skin fibrosis [32, 33]. IL-6 can also promote the secretion of collagen by fibroblasts [34]. Importantly, B-cell activating factor (BAFF) has been shown to increase IL-6+ Beffs [35]. In addition, BAFF antagonists reduce skin and pulmonary fibrosis in scleroderma models by reducing IL-6+ Beffs rather than IL-10+ Bregs [33]. In humans, B cells have been shown to secrete high levels of IL-6 in SSc patients [36]. Therefore, IL-6+ Beffs promote the pathogenesis of SSc. In an SSc mouse model, IL-6 played a key role in tissue fibrosis and autoimmunity [37]. Administration of an anti-IL-6 receptor Ab to mice reduced skin fibrosis in a mouse model of scleroderma. Studies have shown that skin and lung fibrosis in a scleroderma mouse model was attenuated in B-cell-specific IL-6-deficient mice.
GM-CSF was originally considered a haematopoietic growth factor that can induce the proliferation and differentiation of granulocytes and macrophages. However, GM-CSF also has a proinflammatory effect and plays a key role in autoimmune diseases [38, 39]. For example, GM-CSF is involved in the differentiation of fibroblasts into myofibroblasts, which may promote fibrosis in the pathogenesis of SSc [40]. A recent study showed that the Th2 cytokine IL-4 induces effector B cell production of GM-CSF (GM-Beffs) in humans [41]. GM-Beffs were enriched within CD20+CD30+CD38−/low cells, a population distinct from plasmablasts, suggesting that GM-Beffs exert antibody-independent functions [28]. The level of CD30+GM-Beffs were significantly greater in SSc patients than in healthy controls. A subpopulation of SSc patients with diffuse-type and concomitant interstitial lung disease exhibited high numbers of GM-Beffs. In summary, these findings suggest that human GM-Beffs are enriched in the CD30+ B-cell subset and play a role in the pathogenesis of SSc [28].
Regulatory B cells in SSc
Negative regulation of the immune response by B-cell subsets, called Bregs, is considered an important part of the immune system. Bregs play a crucial role in SSc. The production of IL-10 by Bregs is mainly related to the immune response [42]. IL-10 is an anti-inflammatory cytokine that negatively regulates the production of proinflammatory cytokines in T cells and the function of antigen-presenting cells (APCs), as well as dendritic cells and macrophages [43]. IL-10 is produced by a variety of immune cells, such as T cells, macrophages and B cells. IL-10-producing Bregs (IL-10+ Bregs) have been considered the main subgroup of cells that regulate the immune response because of their important functions. Mizoguchi et al. used the term “regulatory B cells” for the first time [44].
In SSc, IL-10+ Bregs have an inhibitory effect on pathogenesis [45]. Previous studies have shown that donor-derived IL-10+ Bregs are important for the suppression of SSc in the sclerodermatous chronic graft-versus-host disease (Scl-cGVHD) model [46]. The absence of IL-10+ Bregs induced severe Scl-cGVHD. In humans, the level of IL-10+ Bregs in patients with SSc is reduced, and a reduced level of IL-10+ Bregs is associated with interstitial lung disease [47, 48]. Moreover, the number of IL-10+ Bregs was negatively correlated with the extent of SSc in patients. In a follow-up study, compared with those before treatment, IL-10+ Breg levels in patients with SSc were found to be significantly increased after treatment, accompanied by decreased disease activity [49]. These results suggest that a decrease in the number of IL-10+ Bregs contributes to the development of SSc.
Potential relevance of Beffs and Bregs in SSc
Overall, different approaches have been tested to address the hyperactivation of B cells in SSc. Currently, some success has been achieved in targeting B cells for SSc therapy. B-cell depletion therapy with rituximab, a CD20 Ab that depletes human pan-B cells, has shown beneficial effects on skin fibrosis, pulmonary function and pulmonary hypertension in patients with SSc [7, 50, 51]. Furthermore, in a phase 2 study and a phase 3 study, the IL-6-receptor-α inhibitor tocilizumab failed to reduce skin thickening, but tended to improve the modified Rodnan Skin Score and pulmonary function [52, 53]. The clinical improvement in both trials might have been limited by the lack of specificity, as depletion of all B-cell subsets eliminated the protective effects of Bregs.
IL-6 is a pleiotropic cytokine whose activity stimulates the proliferation and differentiation of B and T lymphocytes, increased antibody production, activates T cells, stimulates the differentiation of haematopoietic precursor cells, affects the proliferation of nonlymphoid cells, and activates the acute phase protein response [54]. IL-6 plays a special role in the development of SSc, both in vascular damage and in the development of fibrosis. In the early stages, IL-6 participates in vascular endothelial activation and apoptosis [55]. Moreover, IL-6 plays an important role in the development of fibrotic changes by mediating the transformation of fibroblasts into myofibroblasts. All of these conditions are associated with disabling clinical manifestations, such as skin thickening, pulmonary fibrosis, pulmonary arterial hypertension (PAH), heart failure, and dysphagia. IL-6 induced a concentration-dependent increase in collagen and glycosaminoglycan production by human dermal fibroblasts in vitro [56]. Many reports have shown that the production of IL-6 in fibroblasts from SSc-affected skin is increased compared with that in normal fibroblasts [57]. In addition, blocking the IL-6 response with an anti-IL-6 antibody resulted in a significant reduction in type I procollagen in cultured SSc fibroblasts [58]. A previous study also revealed that the level of serum IL-6 was closely related to skin thickening, which confirmed the important role of IL-6 in the development of SSc skin sclerosis [59, 60].
IL-10 is an anti-inflammatory cytokine that can inhibit the synthesis of proinflammatory cytokines in monocytes or macrophages [61]. IL-10 can stimulate B cells to produce IgG, IgA and IgM. In vitro experiments showed that IL-10 could downregulate the expression of the type I collagen gene and increase the expression of the collagenase gene [62]. Another study revealed a negative correlation between IL-10+ B cells and IL-17+ T cells in SSc patients who were positive for the anti-topo I antibody and the anticentromere Ab [49]. The number of IL-10+B cells in SSc patients is very low, especially in those with SSc-related interstitial lung disease (ILD); furthermore, the number of IL-10+B cells is negatively correlated with the number of IL-17A-producing T cells, suggesting that IL-10 plays an important negative regulatory role in the pathogenesis of SSc. IL-10 was found to inhibit helper T-cell 1 and 17 (Th1 and Th17) immune responses [63]. However, Th17 cells play an important role in the pathogenesis of SSc. In a mouse fibroblast line, IL-17A increased the expression of TGF-β, connective tissue growth factor (CTGF) and collagen but increased skin and lung inflammation and fibrosis in a mouse bleomycin (BLM) model of SSc [64, 65]. The role of Th17 cells in human SSc may not involve direct fibrosis but indirect promotion of inflammation and vasculopathy [66, 67]. Elevated levels of IL-17 were detected in the peripheral blood, skin and lung lymphocytes of patients with SSc [68, 69]. In human SSc, IL-17 increases fibroblast proliferation and collagen production and induces fibroblasts to produce IL-6, IL-8 and adhesion molecules [70, 71]. IL-17A also induces human endothelial cells to produce IL-6 and adhesions [72, 73]. Furthermore, IL-17A promotes the proliferation, migration, collagen synthesis and secretion of SSc patient-derived dermal vascular smooth muscle cells, resulting in intimal thickening and aggravated vasculopathy and fibrosis [74,75,76].
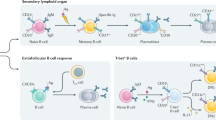
Consequently, we speculate that there may be an imbalance between IL-6+ Beffs and IL-10+ Bregs in the pathogenesis of systemic sclerosis and that directly acting on fibroblasts and vascular endothelial cells, or indirectly inducing Th17 cells to play a pathogenic role, could be involved in inflammation, abnormal immunity, vasculopathy and fibrosis (Fig. 1).
Disequilibrium between Beffs and Bregs in the pathogenesis of systemic sclerosis. There may be an imbalance between IL-6+ Beffs and IL-10+ Bregs in the pathogenesis of systemic sclerosis, as directly acting on fibroblasts and vascular endothelial cells or indirectly inducing Th17 cells to play a pathogenic role could be involved in inflammation, abnormal immunity, vasculopathy and fibrosis
Conclusion
B cells both positively and negatively regulate the pathogenesis of SSc via cytokine production. However, identifying the definitive markers and specific transcription factors of Bregs and Beffs is necessary for the development of new B-cell-targeted therapeutic strategies. Nevertheless, Bregs and Beffs are critically important components of the immune system. B cells are the key factor in the pathogenesis of SSc. Therefore, in the future, increasing the number of Bregs or reducing the number of Beffs may be one of the best strategies to treat refractory autoimmune diseases such as SSc.
Data availability
Data availability is not applicable to this article as no new data were created or analyzed in this study.
Abbreviations
- Ab:
-
Antibody
- APC:
-
Antigen presenting cells
- BAFF:
-
B-cell activating factor
- BCRs:
-
B-cell receptors
- Beffs:
-
Effector B cells
- BLM:
-
Bleomycin
- Bregs:
-
Regulatory B cells
- cGVHD:
-
Chronic graftversus-host disease
- CTGF:
-
Connective tissue growth factor
- DC:
-
Dendritic cells
- GM-CSF:
-
Granulocyte macrophage colony stimulating factor
- IFN-γ:
-
Interferon-γ
- IgD:
-
Immunoglobulin D
- IgM:
-
Immunoglobulin M
- IL-6:
-
qinterleukin-6
- IL-10:
-
Interleukin-10
- ILD:
-
Interstitial lung disease
- MS:
-
Multiple sclerosis
- MZ:
-
Marginal zone
- SLE:
-
Systemic lupus erythematosus
- SSc:
-
Systemic sclerosis
- Tfh:
-
T follicular fluid
- Th1:
-
Helper T-cell 1
- Th17:
-
Helper T-cell 17
- TNF-α:
-
Tumour necrosis factor-α
References
Denton CP, Khanna D (2017) Systemic sclerosis. Lancet 390:1685–1699
Nihtyanova SI, Denton CP (2010) Autoantibodies as predictive tools in systemic sclerosis. Nat Rev Rheumatol 6:112–116
Ly N, Ueda-Hayakawa I, Nguyen C, Huynh T, Kishimoto I, Fujimoto M, Okamoto H (2021) Imbalance toward TFH 1 cells playing a role in aberrant B cell differentiation in systemic sclerosis. Rheumatology (Oxford) 60:1553–1562
Sanges S, Jendoubi M, Kavian N, Hauspie C, Speca S, Crave JC, Guerrier T, Lefevre G, Sobanski V, Savina A, Hachulla E, Hatron PY, Labalette M, Batteux F, Dubucquoi S, Launay D (2017) B Cell Homeostasis and functional properties are altered in an hypochlorous acid-induced murine model of systemic sclerosis. Front Immunol 8:53
Einhaus J, Pecher AC, Asteriti E, Schmid H, Secker KA, Duerr-Stoerzer S, Keppeler H, Klein R, Schneidawind C, Henes J, Schneidawind D (2020) Inhibition of effector B cells by ibrutinib in systemic sclerosis. Arthritis Res Ther 22:66
Melissaropoulos K, Daoussis D (2021) B cells in systemic sclerosis: from pathophysiology to treatment. Clin Rheumatol 40:2621–2631
Bosello SL, De Luca G, Rucco M, Berardi G, Falcione M, Danza FM, Pirronti T, Ferraccioli G (2015) Long-term efficacy of B cell depletion therapy on lung and skin involvement in diffuse systemic sclerosis. Semin Arthritis Rheum 44:428–436
Daoussis D, Tsamandas A, Antonopoulos I, Filippopoulou A, Papachristou DJ, Papachristou NI, Andonopoulos AP, Liossis SN (2016) B cell depletion therapy upregulates Dkk-1 skin expression in patients with systemic sclerosis: association with enhanced resolution of skin fibrosis. Arthritis Res Ther 18:118
Romero-Ramirez S, Navarro-Hernandez IC, Cervantes-Diaz R, Sosa-Hernandez VA, Acevedo-Ochoa E, Kleinberg-Bild A, Valle-Rios R, Meza-Sanchez DE, Hernandez-Hernandez JM, Maravillas-Montero JL (2019) Innate-like B cell subsets during immune responses: Beyond antibody production. J Leukoc Biol 105:843–856
Ma X, Dai Y, Witzke O, Xu S, Lindemann M, Kribben A, Dolff S, Wilde B (2022) Chloroquine suppresses effector B-cell functions and has differential impact on regulatory B-cell subsets. Front Immunol 13:818704
Catalan D, Mansilla MA, Ferrier A, Soto L, Oleinika K, Aguillon JC, Aravena O (2021) Immunosuppressive mechanisms of regulatory B cells. Front Immunol 12:611795
Raza I, Clarke AJ (2021) B cell Metabolism and autophagy in autoimmunity. Front Immunol 12:681105
Kohler F, Hug E, Eschbach C, Meixlsperger S, Hobeika E, Kofer J, Wardemann H, Jumaa H (2008) Autoreactive B cell receptors mimic autonomous pre-B cell receptor signaling and induce proliferation of early B cells. Immunity 29:912–921
Martin VG, Wu YB, Townsend CL, Lu GH, O’Hare JS, Mozeika A, Coolen AC, Kipling D, Fraternali F, Dunn-Walters DK (2016) Transitional B cells in early human B cell development - time to revisit the paradigm? Front Immunol 7:546
Giltiay NV, Giordano D, Clark EA (2019) The plasticity of newly formed B cells. J Immunol 203:3095–3104
Shinnakasu R, Kurosaki T (2017) Regulation of memory B and plasma cell differentiation. Curr Opin Immunol 45:126–131
Nutt SL, Hodgkin PD, Tarlinton DM, Corcoran LM (2015) The generation of antibody-secreting plasma cells. Nat Rev Immunol 15:160–171
Tellier J, Nutt SL (2019) Plasma cells: The programming of an antibody-secreting machine. Eur J Immunol 49:30–37
Cancro MP, Tomayko MM (2021) Memory B cells and plasma cells: The differentiative continuum of humoral immunity. Immunol Rev 303:72–82
Shen P, Fillatreau S (2015) Antibody-independent functions of B cells: a focus on cytokines. Nat Rev Immunol 15:441–451
Banko Z, Pozsgay J, Szili D, Toth M, Gati T, Nagy G, Rojkovich B, Sarmay G (2017) Induction and differentiation of IL-10-producing regulatory B cells from healthy blood donors and rheumatoid arthritis patients. J Immunol 198:1512–1520
Matsushita T (2014) Regulatory B cells in mouse models of systemic lupus erythematosus (SLE). Methods Mol Biol 1190:195–205
Cencioni MT, Mattoscio M, Magliozzi R, Bar-Or A, Muraro PA (2021) B cells in multiple sclerosis - from targeted depletion to immune reconstitution therapies. Nat Rev Neurol 17:399–414
Wang Y, Liu J, Burrows PD, Wang JY (2020) B cell development and maturation. Adv Exp Med Biol 1254:1–22
Matsushita T, Le Huu D, Kobayashi T, Hamaguchi Y, Hasegawa M, Naka K, Hirao A, Muramatsu M, Takehara K, Fujimoto M (2016) A novel splenic B1 regulatory cell subset suppresses allergic disease through phosphatidylinositol 3-kinase-Akt pathway activation. J Allergy Clin Immunol 138:1170-1182.e9
Niiro H (2015) Effector B cells in autoimmune diseases. Nihon Rinsho Meneki Gakkai Kaishi 38:412–420
Horii M, Fushida N, Ikeda T, Oishi K, Hamaguchi Y, Ikawa Y, Komuro A, Matsushita T (2022) Cytokine-producing B-cell balance associates with skin fibrosis in patients with systemic sclerosis. J Dermatol 49:1012–1019
Higashioka K, Kikushige Y, Ayano M, Kimoto Y, Mitoma H, Kikukawa M, Akahoshi M, Arinobu Y, Horiuchi T, Akashi K, Niiro H (2020) Generation of a novel CD30(+) B cell subset producing GM-CSF and its possible link to the pathogenesis of systemic sclerosis. Clin Exp Immunol 201:233–243
Hobbs MV, McEvilly RJ, Koch RJ, Cardenas GJ, Noonan DJ (1991) Interleukin-6 production by murine B cells and B cell lines. Cell Immunol 132:442–450
Korn T, Hiltensperger M (2021) Role of IL-6 in the commitment of T cell subsets. Cytokine 146:155654
Sakkas LI (2016) Spotlight on tocilizumab and its potential in the treatment of systemic sclerosis. Drug Des Devel Ther 10:2723–2728
Desallais L, Avouac J, Frechet M, Elhai M, Ratsimandresy R, Montes M, Mouhsine H, Do H, Zagury JF, Allanore Y (2014) Targeting IL-6 by both passive or active immunization strategies prevents bleomycin-induced skin fibrosis. Arthritis Res Ther 16:R157
Matsushita T, Kobayashi T, Mizumaki K, Kano M, Sawada T, Tennichi M, Okamura A, Hamaguchi Y, Iwakura Y, Hasegawa M, Fujimoto M, Takehara K (2018) BAFF inhibition attenuates fibrosis in scleroderma by modulating the regulatory and effector B cell balance. Sci Adv 4:eaas9944
Juhl P, Bondesen S, Hawkins CL, Karsdal MA, Bay-Jensen AC, Davies MJ, Siebuhr AS (2020) Dermal fibroblasts have different extracellular matrix profiles induced by TGF-beta, PDGF and IL-6 in a model for skin fibrosis. Sci Rep 10:17300
Matsushita T, Fujimoto M, Hasegawa M, Matsushita Y, Komura K, Ogawa F, Watanabe R, Takehara K, Sato S (2007) BAFF antagonist attenuates the development of skin fibrosis in tight-skin mice. J Invest Dermatol 127:2772–2780
Rojas M, Rodriguez Y, Monsalve DM, Pacheco Y, Acosta-Ampudia Y, Rodriguez-Jimenez M, Molano-Gonzalez N, Mantilla RD, Ramirez-Santana C, Anaya JM (2019) Cytokine imbalance in patients with systemic sclerosis and resilience: the key role of interleukin-6. Clin Exp Rheumatol 37(Suppl 119):15–22
Kitaba S, Murota H, Terao M, Azukizawa H, Terabe F, Shima Y, Fujimoto M, Tanaka T, Naka T, Kishimoto T, Katayama I (2012) Blockade of interleukin-6 receptor alleviates disease in mouse model of scleroderma. Am J Pathol 180:165–176
Wicks IP, Roberts AW (2016) Targeting GM-CSF in inflammatory diseases. Nat Rev Rheumatol 12:37–48
Achuthan AA, Lee K, Hamilton JA (2021) Targeting GM-CSF in inflammatory and autoimmune disorders. Semin Immunol 54:101523
Binai N, O’Reilly S, Griffiths B, van Laar JM, Hugle T (2012) Differentiation potential of CD14+ monocytes into myofibroblasts in patients with systemic sclerosis. PLoS ONE 7:e33508
Li R, Rezk A, Miyazaki Y, Hilgenberg E, Touil H, Shen P, Moore CS, Michel L, Althekair F, Rajasekharan S, Gommerman JL, Prat A, Fillatreau S, Bar-Or A (2015) Proinflammatory GM-CSF-producing B cells in multiple sclerosis and B cell depletion therapy. Sci Transl Med 7:310ra166
Dasgupta S, Dasgupta S, Bandyopadhyay M (2020) Regulatory B cells in infection, inflammation, and autoimmunity. Cell Immunol 352:104076
Lykken JM, Candando KM, Tedder TF (2015) Regulatory B10 cell development and function. Int Immunol 27:471–477
Mizoguchi E, Mizoguchi A, Preffer FI, Bhan AK (2000) Regulatory role of mature B cells in a murine model of inflammatory bowel disease. Int Immunol 12:597–605
Sakkas LI, Daoussis D, Mavropoulos A, Liossis SN, Bogdanos DP (2019) Regulatory B cells: New players in inflammatory and autoimmune rheumatic diseases. Semin Arthritis Rheum 48:1133–1141
Le Huu D, Matsushita T, Jin G, Hamaguchi Y, Hasegawa M, Takehara K, Tedder TF, Fujimoto M (2013) Donor-derived regulatory B cells are important for suppression of murine sclerodermatous chronic graft-versus-host disease. Blood 121:3274–3283
Mavropoulos A, Simopoulou T, Varna A, Liaskos C, Katsiari CG, Bogdanos DP, Sakkas LI (2016) Breg cells are numerically decreased and functionally impaired in patients with systemic sclerosis. Arthritis Rheumatol 68:494–504
Mavropoulos A, Liaskos C, Simopoulou T, Bogdanos DP, Sakkas LI (2017) IL-10-producing regulatory B cells (B10 cells), IL-17+ T cells and autoantibodies in systemic sclerosis. Clin Immunol 184:26–32
Matsushita T, Hamaguchi Y, Hasegawa M, Takehara K, Fujimoto M (2016) Decreased levels of regulatory B cells in patients with systemic sclerosis: association with autoantibody production and disease activity. Rheumatology (Oxford) 55:263–267
Daoussis D, Melissaropoulos K, Sakellaropoulos G, Antonopoulos I, Markatseli TE, Simopoulou T, Georgiou P, Andonopoulos AP, Drosos AA, Sakkas L, Liossis SN (2017) A multicenter, open-label, comparative study of B-cell depletion therapy with Rituximab for systemic sclerosis-associated interstitial lung disease. Semin Arthritis Rheum 46:625–631
Zamanian RT, Badesch D, Chung L, Domsic RT, Medsger T, Pinckney A, Keyes-Elstein L, D’Aveta C, Spychala M, White RJ, Hassoun PM, Torres F, Sweatt AJ, Molitor JA, Khanna D, Maecker H, Welch B, Goldmuntz E, Nicolls MR (2021) Safety and efficacy of B-Cell depletion with rituximab for the treatment of systemic sclerosis-associated pulmonary arterial hypertension: A multicenter, double-blind, randomized, placebo-controlled trial. Am J Respir Crit Care Med 204:209–221
Khanna D, Denton CP, Jahreis A, van Laar JM, Frech TM, Anderson ME, Baron M, Chung L, Fierlbeck G, Lakshminarayanan S, Allanore Y, Pope JE, Riemekasten G, Steen V, Muller-Ladner U, Lafyatis R, Stifano G, Spotswood H, Chen-Harris H, Dziadek S, Morimoto A, Sornasse T, Siegel J, Furst DE (2016) Safety and efficacy of subcutaneous tocilizumab in adults with systemic sclerosis (faSScinate): a phase 2, randomised, controlled trial. Lancet 387:2630–2640
Khanna D, Lin C, Furst DE, Goldin J, Kim G, Kuwana M, Allanore Y, Matucci-Cerinic M, Distler O, Shima Y, van Laar JM, Spotswood H, Wagner B, Siegel J, Jahreis A, Denton CP (2020) Tocilizumab in systemic sclerosis: a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Respir Med 8:963–974
Uciechowski P, Dempke W (2020) Interleukin-6: A Masterplayer in the Cytokine Network. Oncology 98:131–137
Barnes TC, Spiller DG, Anderson ME, Edwards SW, Moots RJ (2011) Endothelial activation and apoptosis mediated by neutrophil-dependent interleukin 6 trans-signalling: a novel target for systemic sclerosis? Ann Rheum Dis 70:366–372
Duncan MR, Berman B (1991) Stimulation of collagen and glycosaminoglycan production in cultured human adult dermal fibroblasts by recombinant human interleukin 6. J Invest Dermatol 97:686–692
Francois A, Chatelus E, Wachsmann D, Sibilia J, Bahram S, Alsaleh G, Gottenberg JE (2013) B lymphocytes and B-cell activating factor promote collagen and profibrotic markers expression by dermal fibroblasts in systemic sclerosis. Arthritis Res Ther 15:R168
Kawaguchi Y, Hara M, Wright TM (1999) Endogenous IL-1alpha from systemic sclerosis fibroblasts induces IL-6 and PDGF-A. J Clin Invest 103:1253–1260
Sato S, Hasegawa M, Takehara K (2001) Serum levels of interleukin-6 and interleukin-10 correlate with total skin thickness score in patients with systemic sclerosis. J Dermatol Sci 27:140–146
Zhu HL, Q DU, Chen WL, Zuo XX, Li QZ, Liu SJ (2019) Altered serum cytokine expression profile in systemic sclerosis and its regulatory mechanisms. Beijing Da Xue Xue Bao Yi Xue Ban 51:716–722
Saraiva M, Vieira P, O’Garra A (2020) Biology and therapeutic potential of interleukin-10. J Exp Med 217:e20190418
Reitamo S, Remitz A, Tamai K, Uitto J (1994) Interleukin-10 modulates type I collagen and matrix metalloprotease gene expression in cultured human skin fibroblasts. J Clin Invest 94:2489–2492
Stadhouders R, Lubberts E, Hendriks RW (2018) A cellular and molecular view of T helper 17 cell plasticity in autoimmunity. J Autoimmun 87:1–15
Okamoto Y, Hasegawa M, Matsushita T, Hamaguchi Y, Huu DL, Iwakura Y, Fujimoto M, Takehara K (2012) Potential roles of interleukin-17A in the development of skin fibrosis in mice. Arthritis Rheum 64:3726–3735
Lei L, Zhao C, Qin F, He ZY, Wang X, Zhong XN (2016) Th17 cells and IL-17 promote the skin and lung inflammation and fibrosis process in a bleomycin-induced murine model of systemic sclerosis. Clin Exp Rheumatol 34(Suppl 100):14–22
Dufour AM, Borowczyk-Michalowska J, Alvarez M, Truchetet ME, Modarressi A, Brembilla NC, Chizzolini C (2020) IL-17A dissociates inflammation from fibrogenesis in systemic sclerosis. J Invest Dermatol 140:103-112.e8
Fukayama M, Yoshizaki A, Fukasawa T, Ebata S, Kuzumi A, Yoshizaki-Ogawa A, Asano Y, Oba K, Sato S (2020) Interleukin (IL)-17F and IL-17E are related to fibrosis and vasculopathy in systemic sclerosis. J Dermatol 47:1287–1292
Yang X, Yang J, Xing X, Wan L, Li M (2014) Increased frequency of Th17 cells in systemic sclerosis is related to disease activity and collagen overproduction. Arthritis Res Ther 16:R4
Rolla G, Fusaro E, Nicola S, Bucca C, Peroni C, Parisi S, Cassinis MC, Ferraris A, Angelino F, Heffler E, Boita M, Brussino L (2016) Th-17 cytokines and interstitial lung involvement in systemic sclerosis. J Breath Res 10:046013
Brembilla NC, Montanari E, Truchetet ME, Raschi E, Meroni P, Chizzolini C (2013) Th17 cells favor inflammatory responses while inhibiting type I collagen deposition by dermal fibroblasts: differential effects in healthy and systemic sclerosis fibroblasts. Arthritis Res Ther 15:R151
Dufour AM, Alvarez M, Russo B, Chizzolini C (2018) Interleukin-6 and type-I collagen production by systemic sclerosis fibroblasts are differentially regulated by interleukin-17A in the presence of transforming growth factor-beta 1. Front Immunol 9:1865
Xing X, Yang J, Yang X, Wei Y, Zhu L, Gao D, Li M (2013) IL-17A induces endothelial inflammation in systemic sclerosis via the ERK signaling pathway. PLoS ONE 8:e85032
Chizzolini C, Dufour AM, Brembilla NC (2018) Is there a role for IL-17 in the pathogenesis of systemic sclerosis? Immunol Lett 195:61–67
Liu M, Yang J, Xing X, Cui X, Li M (2014) Interleukin-17A promotes functional activation of systemic sclerosis patient-derived dermal vascular smooth muscle cells by extracellular-regulated protein kinases signalling pathway. Arthritis Res Ther 16:4223
Liu M, Yang J, Li M (2015) Tanshinone IIA attenuates interleukin-17A-induced systemic sclerosis patient-derived dermal vascular smooth muscle cell activation via inhibition of the extracellular signal-regulated kinase signaling pathway. Clinics (Sao Paulo) 70:250–256
Liu M, Wu W, Sun X, Yang J, Xu J, Fu W, Li M (2016) New insights into CD4(+) T cell abnormalities in systemic sclerosis. Cytokine Growth Factor Rev 28:31–36
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (No. 81602747).
Author information
Authors and Affiliations
Contributions
Mengguo Liu conceived of the idea for the review and wrote, proofread and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
I declare that the author have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, M. Effector and regulatory B-cell imbalance in systemic sclerosis: cooperation or competition?. Clin Rheumatol 43, 2783–2789 (2024). https://doi.org/10.1007/s10067-024-07086-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-024-07086-0