Abstract
The impact of climate change on ecosystems can be assessed through pollen dispersion data, which acts as a proxy for the plant flowering stage. The aim of this study is to verify if changes occurred in the season and concentration of airborne pollen in Trentino (Northern Italy), and to evaluate if temperature (T), precipitation (P), and/or land use influenced such changes in the period 1989–2018. Airborne pollen, sampled by a Hirst-type trap, was analyzed by light microscopy, and pollen concentrations were obtained. Twenty-four taxa, covering 95% of the local pollen spectrum, were considered for this study. A significant upward trend in annual pollen integral (APIn) is the main outcome over the study period. The increase in APIn is more evident when analyzing the data in three decadic blocks, both for all the considered taxa (+ 58%) and for Arboreal Pollen (AP; + 155%). Considering both the annual data and the decadic blocks, APIn shows a significant positive trend for Cupressaceae/Taxaceae, Ulmus, Populus, Salix, Ostrya, Quercus, Olea (AP), and Plantago, Cannabaceae, Ambrosia (non-arboreal pollen, NAP); a significantly earlier start of the Main Pollen Season (MPS) is proved for Rumex and Poaceae. APIn for 24 taxa shows a significant positive correlation with annual T min (< 0.005) and T mean (< 0.001), both of which showing a significant increase, and a negative correlation (< 0.025) with the number of frost days. From a human health perspective, higher temperatures, driven by climate changes, lead to higher concentrations of allergenic airborne pollen, turning into a higher risk for allergy sufferers.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ongoing climate change (IPCC Report 2022) has wide-ranging effects on ecosystems, human health, and economic sectors, with numerous repercussions for global well-being (EAA Report 2017).
Phenology, the science of natural recurring events, is one of the indicators for observing the impacts of climate change on ecosystems and biological processes (Parmesan 2006). The shift in phenology as a high-temporal resolution signal of this impact (Menzel et al. 2006) is examined in different geographical contexts, taking both animal and plant organisms into consideration (Parmesan 2006; Walther et al. 2002).
In the plant world, different phenological phases such as bud burst, leaf unfolding, flowering, and senescence are investigated through direct observations, but an increasing number of studies consider pollen dispersal data as a proxy for flowering in anemophilous plants (Bock et al. 2014; Hidalgo-Galvez et al. 2018; Morin et al. 2009). Even though plant flowering and pollen dispersal are not exactly overlapping phases, and airborne pollen dispersion is also influenced by transport phenomena (Damialis et al. 2020), airborne pollen data can be useful for its multiple advantages such as standardized data collection and analysis (e.g., UNI EN 16868:2019 at European level), global level sampling (https://www.zaum-online.de/pollen/pollen-monitoring-map-of-the-world.html, accessed on 19 December 2023), and the possibility to rely on long time series. The detection of trends, in fact, is extremely dependent on extended data series, in order to overcome the signal of the natural interannual variability in pollen production, particularly robust in the case of arboreal plants (Pesendorfer et al. 2016).
To highlight trends and evaluate changes in airborne pollen data, a set of pollen season descriptors has been elaborated and recommendations for a coherent terminology were published (Bastl et al. 2018). An increase in the total pollen sampled by aerobiological monitoring was reported over the northern hemisphere (Ziska et al. 2019), reflecting the effects of an increase in the annual pollen index (APIn) for arboreal pollen (AP) (Anderegg et al. 2021; Bruffaerts et al. 2018; Gehrig and Clot 2021; Glick et al. 2021; Ziello et al. 2012), often detailed in single species trends, such as Corylus (Bruffaerts et al. 2018; Hoebeke et al. 2018; Jochner-Oette et al. 2019), Betula and Quercus (Adams-Groom et al. 2022), and Cupressaceae/Taxaceae (Gehrig and Clot 2021).
Similarly, time trends of other pollen season descriptors were reported by several authors; an earlier MPS start, in particular, was highlighted for many taxa, such as Corylus, Quercus, Poaceae, and Urticaceae (Cristofolini et al. 2020; Glick et al. 2021; Hoebeke et al. 2018; Rojo et al. 2021), Betula (Frei and Gassner 2008; Lind et al. 2016; Rojo et al. 2021), Cupressaceae/Taxaceae (Gehrig and Clot 2021), and Olea (Garcia-Mozo et al. 2014). Results, though, can be very variable depending on the site (Ziello et al. 2012) and taxon (Damialis et al. 2007; Galan et al. 2016) considered and do not lead to general conclusions.
The possible drivers of change for pollen season descriptors, and thus for changes in plant phenology and biodiversity, can be several, but the most relevant include global change, mainly described through meteorological variables and land use. Pollen season is more often analyzed in relation to climate change, with temperature as the most investigated variable. Overall, air temperature evidences a marked effect on seasonality (Frei and Gassner 2008; Gehrig 2006; Hoebeke et al. 2018; Makra et al. 2014), determining an earlier and longer pollen season (Aguilera and Ruiz-Valenzuela 2014; Gehrig and Clot 2021; Majeed et al. 2018; Schramm et al. 2021; Zhang et al. 2015; Zhang and Steiner 2022), and higher pollen concentrations (Bogawski et al. 2014; Damialis et al. 2007; Wan et al. 2002; Ziska et al. 2019).
Precipitation is also often considered among meteorological parameters influencing pollen season descriptors (Schramm et al. 2021). Precipitation contributes to water availability, ensuring plant viability, development, and reproduction, with the concurrent pollen release, especially in Mediterranean areas, where water shortage may occur (Penuelas et al. 2004). On the short term, though, precipitation may reduce the airborne pollen concentration by removing it from the atmosphere.
In addition to meteorological variables, land use can play a role in plant species composition and spatial distribution, which in turn is reflected in the airborne pollen spectrum (Maya-Manzano et al. 2017; Rojo et al. 2015).
The study of pollen and its spatio-temporal changes, other than its ecological significance for interpreting plant phenology and biodiversity, is of paramount importance due to the allergenicity of many airborne pollen taxa. Current estimates place the incidence of pollen allergy to approximately 25–40% of the population, globally (D’Amato et al. 2007; Husna et al. 2022; Gani et al. 2018), following an increasing trend (Barnes 2018; D’Amato et al. 2015). Such increase is partly related to a higher exposure to airborne allergenic pollen, which is in turn connected to climate change. Milder weather, air pollution, and elevated CO2 levels, in fact, influence pollen production and allergenicity, as well as the spread of neophytes producing allergenic pollen (Beck et al. 2013; D’Amato et al. 2020; Luschkova et al. 2022; Rauer et al. 2021).
The present study, based on a 30-year aerobiological dataset collected in San Michele all’Adige, Northern Italy, aims to (i) verify if significant changes occurred in the timing and quantity of airborne pollen from 1989 to 2018 and (ii) evaluate the relationship between pollen season descriptors and air temperature, precipitation, and land use.
Materials and methods
Study area
The aerobiological monitoring site is located at San Michele all’Adige, Trentino, North Italy. The region is orographically complex, within the Alpine biogeographical region, characterized by a predominantly oceanic climate (Eccel et al. 2016) and a high diversity of plant associations. The sampling site is at the bottom of the Adige Valley (46.19, 11.13; 206 m a.s.l.), in a rural area mainly covered by vineyards and apple orchards, where also herbaceous species as Poaceae, Parietaria, Artemisia, and Plantago are present. Broadleaved and mixed forests (Ostrya carpinifolia Scop., Fraxinus ornus L., Quercus pubescens Willd., Corylus avellana L., Alnus sp., and Salix sp.) dominate the lateral slopes, while beech (Fagus sylvatica L.) and conifers (Pinus sylvestris L., Pinus nigra Arnold., and Picea abies (L.) H.Karst.) expand as the altitude increases.
Data collection
Data on airborne pollen concentration were collected over 30 years, from 1989 to 2018, by a Hirst-type volumetric sampler (VPPS 2000, Lanzoni, Bologna, Italy), installed at the top of a 10-m tilt-down pole. Identification and counting of pollen were performed by light microscopy (Leitz Diaplan, Wild Leitz GmbH, Wetzlar, Germany), and the daily concentration of airborne pollen (P*m−3) was calculated for each taxon. The sampling and analysis of airborne pollen were conducted in accordance with the UNI EN 16868:2019 European standard procedure.
Air temperature (°C) and precipitation (mm) were recorded within 1.5 km of the aerobiological sampling site, in the Adige valley floor at the same elevation as the aerobiological sampler. The weather station consists of a TMF500 datalogger (NESA s.r.l, Treviso, Italy), connected to several sensors including a Vaisala HMP155 thermo-hygrometer and a SIAP+MICROS rain gauge with a 400 cmq opening. The datalogger reads the sensors every 10 s and stores the meteorological data every 15 min.
Land cover was analyzed within a 1-km, 5-km, and 30-km radius area centered on the aerobiological sampler, considering the Corine Land Cover (CLC) level 1 classes (1. Artificial surfaces, 2. Agricultural areas, and 3. Forests and semi-natural areas; https://land.copernicus.eu/pan-european/corine-land-cover, accessed on 12 December 2023) for the years 1990, 2000, 2006, 2012, and 2018.
Data analysis
The San Michele all’Adige pollen spectrum consists of more than 60 taxa. For this study, the main 24 pollen taxa, covering 95% of the total pollen, were considered. Pollen taxa were assigned to the taxonomical level of plant species, genus, or family based on morphological features, referring to the updated nomenclature (https://www.worldfloraonline.org/, accessed on 12 December 2023).
The following pollen season descriptors were calculated for each taxon: (i) annual pollen integral (APIn; pollen*m−3); (ii) main pollen season (MPS) start and end, as the day of the year when 2.5% and 97.5% of the APIn was reached, respectively (i.e., MPS start, MPS end); (iii) MPS length, as the difference between MPS end and MPS start, plus 1; (iv) MPS peak value, as the maximum concentration registered during the MPS; (v) MPS peak, as the day when the MPS peak value occurs. Pollen descriptors were calculated for each calendar year.
In order to verify data completeness, the mean MPS over the 30-year study period was calculated for each taxon; the mean MPS start corresponds to the average of the annual MPS start dates minus a standard deviation (SD), and the mean MPS end corresponds to the average of the annual MPS end dates plus a SD. The analyses were performed only on years with at least 80% of data available within the mean MPS. The annual APIn for arboreal (AP) and non-arboreal (NAP) taxa and the number of high-concentration days (i.e., the number of days in a year when pollen concentration of at least one taxon exceeds a threshold of 100 P*m−3 for AP (AP > 100), and 50 P*m-3 for NAP (NAP > 50)) were also calculated (Pfaar et al. 2017).
Air temperature data (Tmean, Tmin, Tmax) were averaged on a yearly and monthly basis. Daily precipitation values (P) were accumulated on annual (P tot) basis. The number of days with precipitation (P days) was calculated for each year. The number of frost days was calculated for each year as the number of days when the minimum temperature was < 0 °C.
The presence of a monotonic upward or downward temporal trend in pollen season descriptors and meteorological variables was verified by means of the non-parametric Mann-Kendall test; the slope of the linear trend was calculated using Sen’s estimate (MAKESENS 1.0 version freeware, Finnish Meteorological Institute).
Correlations between pollen season descriptors and meteorological variables were analyzed using the non-parametric Spearman’s rank correlation (TIBCO Statistica® 14.1.0). Temperature and precipitation data considered for the analysis covered the period from the month including the MPS descriptor (i.e., MPS start, peak, end) backwards to October of the previous year.
In addition, pollen season descriptors and meteorological variables were averaged within each of three decadic blocks (i.e., 1989–1998, 1999–2008, and 2009–2018), to minimize the effect of interannual fluctuations in pollen production and to highlight relevant changes during the study period. Parametric one-way ANOVA (for meteorological variables) and non-parametric Kruskal-Wallis ANOVA (for pollen season descriptors) were performed to identify significant differences between the three 10-year data blocks, followed by the post-hoc LSD Fisher (parametric) and Mann-Whitney U (non-parametric) test to highlight the significantly different pairs.
Results
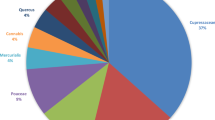
Figure 1 shows the pollen spectrum of San Michele all’Adige, based on the 30-year dataset. In terms of plant growth form, 16 out of 24 pollen taxa considered belong to arboreal plants (i.e., AP), and 8 out of 24 taxa belong to non-arboreal plants (i.e., NAP). Urticaceae and Ostrya are responsible for nearly half of the total airborne pollen (47.6%), followed by Poaceae (11.2%) and Cupressaceae/Taxaceae (6.1%).
Pollen spectrum of San Michele all’Adige, calculated for the 1989–2018 period. Arboreal taxa are indicated with _AP; non-arboreal with _NAP. Other pollen (9%) groups minor taxa, including the AP taxa Ulmus, Salix, Fagus, Picea, and Olea, and the NAP taxa Rumex, Plantago, Chenopodiaceae, and Ambrosia
Trend analysis
The most evident signal deriving from the analyses of the local time series of pollen data concerns the monotonic significant trend for the increase in total pollen, expressed by the APIn index, even more pronounced for arboreal pollen (Fig. 2). AP displays also a significant (p = 0.08) upward trend in the number of high-concentration days (AP > 100), with an increase of about 9 days per 10 years.
Time trend (Sen’s slope) of the annual pollen integral (APIn) for the total pollen of the 24 taxa considered in the study (black line for the trend and black dots for annual values) and separated for arboreal pollen (gray line and dots), and non-arboreal pollen (dashed line, white dots). P values are indicated next to the line for the significant trends
When detailing the analysis at taxon level, more than half of the taxa shows a significantly increasing trend for APIn (13 on 24); a very high significance (p < 0.001) is reported for Cupressaceae/Taxaceae, Populus, Salix, Quercus, Plantago, and Cannabaceae; a high significance (p < 0.01) is evidenced for Ulmus, Ostrya, Olea, and Ambrosia; Alnus, Picea, and Rumex display a significant trend (p < 0.05) (Table 1).
Coherently, also MPS peak values show a significant upward trend for most of the taxa (11 on 24), except for Poaceae, which peak values decrease during the study period. The significant upward trend for MPS peak value is generally associated with an APIn upward trend, except for Amaranthaceae, which MPS peak value has an increasing trend despite APIn remains stable during the study period.
A significantly earlier MPS start is observed for 6 out of 24 taxa (two AP, Fagus and Pinus; four NAP, Rumex, Poaceae, Urticaceae, Plantago), with an advance of 5.4 (min 3.3–max 8.0) days every ten years, on average.
As for MPS peak, end, and length, no coherent change is observable across the different taxa.
As for meteorological variables, precipitation does not show a trend along the time series. Analyzing temperatures and number of frost days, on the contrary, significant trends are highlighted, as reported in Table 2.. There is a significant upward trend in the annual Tmin and Tmean. Monthly Tmin for November of the previous year (Tmin 11py), February (Tmin 2), April (Tmin 4), and June (Tmin 6) significantly increase. Monthly Tmean for November of the previous year (Tmean 11py), and April (Tmean 4) also increase significantly. Conversely, the annual Tmax and the monthly Tmax for January (Tmax 1) and February (Tmax 2) have a significant negative trend. A significant decrease is observed in the annual number of frost days.
Pollen season descriptors and meteorological variables
Total pollen amount (APIn for 24 taxa) shows a significant positive correlation with annual Tmin (< 0.005) and Tmean (< 0.001), and negative (< 0.025) with the number of frost days. The significant positive correlation with annual Tmin and Tmean is evidenced also for APIn of AP (p < 0.01 and p < 0.005, respectively) and NAP (p < 0.01 and p < 0.0025, respectively); APIn of NAP shows a significant positive correlation with the number of days with precipitation (p < 0.025) and negative correlation with the number of frost days (p < 0.025). The number of high-concentration days, both for arboreal (AP > 100) and non-arboreal (NAP > 50) taxa, positively correlates with annual mean temperature (p < 0.02); a significant positive correlation is also evidenced between NAP > 50 and the number of days with precipitation (P days; Table 3).
The significant (p < 0.05) correlations between pollen season descriptors (MPS start, peak, and end) and monthly air temperatures are reported in Online Resource 1 ((a) Tmin; (b) Tmean; (c) Tmax). MPS start shows the highest number of correlations—mostly negative—with the pre-seasonal air temperature, more frequent for AP taxa flowering from February to April.
MPS peak of AP taxa shows a negative significant correlation with air temperature, mostly Tmean and Tmax, occurring up to ca. 2–3 months prior to the event. NAP taxa show a less evident effect of temperature on MPS peak timing.
MPS end evidences very few negative correlations with temperatures, mainly for AP taxa ending the pollen season in April with March Tmean (Tmean 3) and Tmax (Tmax 3).
Land cover
Table 4 details the first level of classification of CLC in 1990, 2000, 2006, 2012, and 2018 within three buffer zones centered on the aerobiological sampler, and with a radius of 1 km, 5 km, and 30 km.
Within the 1-km buffer, a reduction in the agricultural area between 2006 and 2018 is evident, with the concurrent increase in artificial surfaces between 2000 and 2018, and in forest and seminatural areas between 2012 and 2018. The overall changes from 1990 to 2018 range from 10.6 to 12% of the area related to each level I CLC category, and only from − 0.6 to 0.3% when considering the change in relation to the buffer area.
Within the 5-km buffer zone, there is an overall increase of 51 ha in artificial surfaces, corresponding to 8.3% of the category area, but only to 0.01% of the buffer area; agricultural and forest seminatural areas decrease slightly between 1990 and 2018 (− 1.3% and 0.3% of each category area). The maximum percentual change when referring to the buffer area is 0.1%.
Within the 30-km buffer, a remarkable expansion of artificial surfaces is noted (14.6%), mainly referrable to the period 1990–2006; on the contrary, agricultural areas slightly decrease (− 5.3%). Forest and seminatural areas remain quite stable during the period 1990–2018. When referring to the buffer area, changes are between − 0.01% and 0.01%.
Decadic block data analysis
The meteorological and pollen season descriptor data (mean ± sd) for the three decadic blocks are reported in Table 5, only when a significant variation between the blocks is verified. As for meteorological variables (Table 5), temperature exhibits significant changes across the decades. A significant increase of 1.0 °C in annual minimum temperature occurs from the first to the third decadic block, mainly driven by the considerable increase in April minimum temperature (+ 2.0 °C). No significant change occurs in the annual mean temperature across the decadic blocks, while it increases significantly particularly in April (+ 1.7 °C) and June (+ 0.9 °C). In contrast, the annual maximum temperature decreases significantly, in January (− 1.1 °C) and February (− 2.4 °C).
Regarding pollen amount (Table 6), the most evident signal corresponds to the highly significant increase in APIn (+ 58%), which is even more marked when considering only AP (+ 155%). Concurrently, AP shows a significant increase (+ 20 days) in the number of days with high pollen concentrations (i.e., AP > 100).
When considering the AP individual taxa, both APIn and MPS peak value significantly increase across the decadic blocks for Cupressaceae/Taxaceae, Ulmus, Populus, Quercus, Salix, and Olea; Ostrya APIn has a remarkable and significant increase (+ 189% from the first to the third decade), while MPS peak value significantly increases for Picea. MPS peak values exhibit a significant downward trend only for Vitis.
As for NAP taxa, APIn and MPS peak values significantly increase for Plantago, Amaranthaceae, Cannabaceae, and Artemisia; APIn increase is limited to the first and second decadic blocks for Ambrosia.
When analyzing MPS timing (Table 7), Poaceae express a significantly earlier MPS start (− 11 days) and peak (− 6 days) and a significantly later MPS end (+ 20 days) across the three decadic blocks, resulting in a longer pollen season. Rumex shows a significantly earlier MPS start and peak, and a significantly later MPS peak is pointed out for Artemisia only. A shorter MPS length is demonstrated for the Betula and Castanea AP taxa, connected to an earlier MPS end. Cupressaceae/Taxaceae, on the contrary, undergo a lengthening of the MPS.
Urticaceae, the most represented taxon in the local pollen spectrum, shows no significant change in any pollen season descriptors.
Discussion and conclusion
Overall, the key findings of this 30-year aerobiological study in Trentino (Northern Italy), carried out on 24 pollen taxa, are (i) an impressive increase in annual pollen amount and (ii) an earlier pollen season start, both related to rising air temperatures.
The significant increase in annual pollen amount, consistent with previous studies at different spatial scales (Anderegg et al. 2021; Cristofori et al. 2010; Gehrig and Clot 2021; Rojo et al. 2021; Ziello et al. 2012; Ziska 2020), is even more evident when considering data aggregated in decadic blocks, thus minimizing the effects of interannual fluctuations in pollen production. The increase in annual pollen amount can be due to several—even concurring—factors, such as to changes in vegetation composition (García-Mozo et al. 2016), to a larger production of pollen by the individual plants (Katz et al. 2020), or to changes in atmospheric factors that may influence pollen release and airborne transport (Picornell et al. 2023). In this study, no relevant changes in land use are verified within the buffer areas centered on the aerobiological sampler, leading to the exclusion of this as a possible cause of the increase in pollen amount. Rather, the significant increase in annual minimum and mean air temperatures registered in the study area, as well as the decrease in frost days, which positively correlate with the annual pollen amount, supports the evidence about the important role of meteorological variables on pollen production (Zhang and Steiner 2022). Precipitation does not show any change and there is no significant correlation with pollen season descriptors, likely because of the absence of hydric stress in the study area, in contrast to Mediterranean sites (Penuelas et al. 2004). The number of rainy days is the only precipitation-related variable that significantly correlates with the annual pollen integral of non-arboreal taxa. Pollen production can also depend on plant nutritional state (Jochner et al. 2013) and on carbon dioxide (CO2) levels in the atmosphere (Ziska 2020). It has been proven, mostly under controlled conditions, that elevated CO2 levels induce pollen production (Albertine et al. 2014; Rogers et al. 2006). Moreover, some authors (Zhang and Steiner 2022; Ziello et al. 2012) report that climate-driven changes alone have a smaller impact on pollen production when compared to the additional effect of CO2, even if temperature is a stronger driver in real conditions (Anderegg et al. 2021). In this study, the effect of CO2 on pollen amount was not directly investigated, but since CO2 and air temperature are two interconnected factors, it may have contributed to its radiative forcing (Koutsoyiannis and Kundzewicz 2020).
Basically, several climate and non-climate drivers affect pollen production, to varying degrees depending on the pollen taxa considered. In this study, Ostrya (hop hornbeam) and Cupressaceae/Taxaceae are the most represented arboreal taxa, accounting for 24% of the pollen spectrum. Hop hornbeam, which pollen amount tripled from the first to the third decadic block, is a native plant species that characterizes the mixed deciduous woodlands in the surroundings of the sampling area. The distribution of this species in the study area can be deemed to have remained constant during the last decades, due to the low management on this species, and to the scarce use of this plant in public and private greenery. Thus, the observed increase in pollen amount could be attributable to a larger production by the native plants, likely driven by climate change; we cannot exclude a concurrent age effect of the tree stands, which entails an expansion of the crown and consequently of the number of flowers. Hop hornbeam pollen is seldom taken into due consideration in aerobiological studies (Puljak et al. 2016), despite its high allergenicity (Voltolini et al. 2011) and the high potential levels of pollen exposure due to the large distribution area of this species (https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:295664-1, accessed on 13 December 2023).
Unlike hop hornbeam, the Cupressaceae/Taxaceae taxon is widely considered in the literature; the increase in pollen amount observed in this study is in line with previous results obtained in Switzerland (Gehrig and Clot 2021), Germany (Rojo et al. 2021), Iberian Peninsula (Galan et al. 2016), and Greece (Damialis et al. 2007). The marked increase (+ 142%) of this pollen in our study area can be explained with the more and more frequent use of these plants in urban parks and gardens for ornamental purposes, which is reflected in the aerobiological spectrum (Ciani et al. 2021). Such an increase in pollen amount, coupled by a concurrent lengthening of the pollen season, aggravates the exposure to this allergenic pollen, responsible for winter pollinosis, which is rising especially in Mediterranean countries (Charpin et al. 2019). Moreover, due to global warming, the distribution of these species is likely to expand in the Alps (Zorer et al. 2014).
Although Olea is not one of the most representative taxa in the study area, it can be discussed as an example of the potential impact of agronomic practices. Olea pollen significantly increased in the last decade, 2009–2018, with threefold values compared to the previous 20 years, confirming the trend already observed by other authors (Galera et al. 2018; Garcia-Mozo et al. 2014). For Olea, as well as for Vitis, the agronomic practices (e.g., fertilization, pest control, irrigation) may have had an impact on the reproductive phase and thus on the pollen amount.
Among the non-arboreal taxa (NAP), Ambrosia pollen markedly increased from the first to the second decade; then, it remained almost unchanged. Among the factors influencing changes in pollen amount, it is important to consider the spatial dispersion dynamics of this invasive plant species. The appearance of ragweed pollen in the local aerobiological samples was first observed in 1992 and further confirmed by the finding of this plant in the surrounding area (Gottardini and Cristofolini 1996). As expected, the distribution of this invasive species rapidly spread in the region (Prosser et al. 2019), with a consequent increase in airborne pollen amount during the expansion phase, likely reaching a stabilization afterwards, as reflected by the steady pollen amount. A concurrent impact on Ambrosia pollen might have been played by the containment measures adopted to reduce the plant spread at larger scale during the third decade (Bonini and Ceriotti 2020). The dynamics of invasive species help to also justify the shift in MPS peak for Artemisia pollen, driven by the spread of the late flowering invasive species A. annua and A. verlotiorum across the region (Cristofori et al. 2020).
A general tendency toward an earlier pollen season start, particularly pronounced for herbaceous taxa, is the second key finding of this study. Six pollen taxa are starting the MPS at a significant earlier date, with an average advance of 5.4 (min 3.3–max 8.0) days per 10 years, much higher than reported by other authors (e.g., ca. 2 days/10 years (Gehrig and Clot 2021); 3 days/10 years (Zhang et al. 2015)). Several studies pointed out an earlier pollen season start trend that interests the norther hemisphere (Bruffaerts et al. 2018; Cristofolini et al. 2020; Lind et al. 2016; Zhang et al. 2015; Ziska et al. 2019). This phenomenon is often related to the increase of air temperature and, in a broader sense, to climate change. Pre-seasonal temperature impacts on flowering and pollen release, and the extent of responses may vary depending on the plant species (Menzel et al. 2020). In our study, the correlations are negative between MPS start and temperature of the pre-flowering period, meaning that an increase of temperature close to the event leads to an earlier pollen season start. This is especially evident for taxa which MPS starts from February to April, as Poaceae and Urticaceae, confirming that pre-flowering temperature is an important driver of pollen release for spring and early summer flowering plants (Gehrig and Clot 2021). The minimum temperature rising after the vernalization period may accelerate the heat accumulation phase, inducing the start of the pollen season (Dahl et al. 2013). It should also be considered that Poaceae taxon includes a large variety of species; thus, shifts in pollen seasonality could also be due to changes in species composition, with species flowering in slightly different periods (Ghitarrini et al. 2017).
The present study, whose results are largely consistent with other surveys, has some peculiarities: (i) the high number of pollen taxa analyzed, reflecting the complexity of the local pollen spectrum, and (ii) the rural context, less frequently considered in aerobiological studies. Since the study area is scarcely influenced by typically urban phenomena such as the heat island (Chapman et al. 2017), the observed changes in MPS timing may likely be the result of a background environmental situation, without additional urban forcing factors on phenological phases (Ziello et al. 2012).
In conclusion, from a human health perspective, the increasing amount of pollen, linked to the air temperature rise, as verified in the study area over a 30-year period, can negatively affect the well-being of people suffering from pollen allergies. This finding assumes even greater importance in the perspective of climate change, where global temperatures are expected to warm significantly. In addition to the well-recognized value of airborne pollen data for allergy prophylaxis, this study underlines how long-term aerobiological monitoring may represent a valid tool to detect the consequences of the ongoing environmental changes.
Data Availability
Original data are available under request.
References
Adams-Groom B, Selby K, Derrett S, Frisk CA, Pashley CH et al (2022) Pollen season trends as markers of climate change impact: Betula Quercus and Poaceae. Sci Total Environ 831:154882. https://doi.org/10.1016/j.scitotenv.2022.154882
Aguilera F, Ruiz-Valenzuela L (2014) Forecasting olive crop yields based on long-term aerobiological data series and bioclimatic conditions for the southern Iberian Peninsula. Span J Agric Res 12(1):215–224. https://doi.org/10.5424/sjar/2014121-4532
Albertine JM, Manning WJ, DaCosta M, Stinson KA, Muilenberg ML, Rogers CA (2014) Projected carbon dioxide to increase grass pollen and allergen exposure despite higher ozone levels. Plos One 9(11). https://doi.org/10.1371/journalpone0111712
Anderegg WRL, Abatzoglou JT, Anderegg LDL, Bielory L, Kinney PL, Ziska L (2021) Anthropogenic climate change is worsening North American pollen seasons. PNAS USA 118(7). https://doi.org/10.1073/pnas2013284118
Barnes CS (2018) Impact of climate change on pollen and respiratory disease. Curr Allergy Asthma Rep, 18(11). https://doi.org/10.1007/s11882-018-0813-7
Bastl K, Kmenta M, Berger UE (2018) Defining pollen seasons: background and recommendations. Curr Allergy Asthma Rep 18(12):73. https://doi.org/10.1007/s11882-018-0829-z
Beck I, Jochner S, Gilles S, McIntyre M, Buters JTM, et al. (2013) High environmental ozone levels lead to enhanced allergenicity of birch pollen. Plos One, 8(11). https://doi.org/10.1371/journal.pone.0080147
Bock A, Sparks TH, Estrella N, Jee N, Casebow A, et al. (2014) Changes in first flowering dates and flowering duration of 232 plant species on the island of Guernsey. Glob Change Biol 20(11): 3508-3519. https://doi.org/10.1111/gcb12579
Bogawski P, Grewling L, Nowak M, Smith M, Jackowiak B (2014) Trends in atmospheric concentrations of weed pollen in the context of recent climate warming in Poznan (Western Poland). Int J Biometeorol 58(8):1759–1768. https://doi.org/10.1007/s00484-013-0781-5
Bonini M, Ceriotti V (2020) Ragweed story: from the plant to the patient. Aerobiologia 36(1):45–48. https://doi.org/10.1007/s10453-019-09571-5
Bruffaerts N, De Smedt T, Delcloo A, Simons K, Hoebeke L, et al. (2018) Comparative long-term trend analysis of daily weather conditions with daily pollen concentrations in Brussels, Belgium. Int J Biometeorol 62(3): 483-491. https://doi.org/10.1007/s00484-017-1457-3
Chapman S, Watson JEM, Salazar A, Thatcher M, McAlpine CA (2017) The impact of urbanization and climate change on urban temperatures: a systematic review. Landsc Ecol 32(10):1921–1935. https://doi.org/10.1007/s10980-017-0561-4
Charpin D, Pichot C, Belmonte J, Sutra JP, Zidkova J et al (2019) Cypress pollinosis: from tree to clinic. Clin Rev Allergy Immunol 56(2):174–195. https://doi.org/10.1007/s12016-017-8602-y
Ciani F, Dell’Olmo L, Foggi B, Lippi MM (2021) The effect of urban green areas on pollen concentrations at ground level: a study in the city of Florence (Italy). Urban For Urban Gree, 60. https://doi.org/10.1016/j.ufug.2021.127045
Cristofolini F, Anelli P, Billi BM, Bocchi C, Borney MF et al (2020) Temporal trends in airborne pollen seasonality: evidence from the Italian POLLnet network data. Aerobiologia 36(1):63–70. https://doi.org/10.1007/s10453-019-09609-8
Cristofori A, Bucher E, Rossi M, Cristofolini F, Kofler V, et al. (2020) The late flowering of invasive species contributes to the increase of Artemisia allergenic pollen in autumn: an analysis of 25 years of aerobiological data (1995–2019) in Trentino-Alto Adige (Northern Italy). Aerobiologia 36(4):669–682. https://doi.org/10.1007/s10453-020-09663-7
Cristofori A, Cristofolini F, Gottardini E (2010) Twenty years of aerobiological monitoring in Trentino (Italy): assessment and evaluation of airborne pollen variability. Aerobiologia 26(3): 253-261. https://doi.org/10.1007/s10453-010-9161-3
D’Amato G, Chong-Neto HJ, Monge Ortega OP, Vitale C, Ansotegui I et al (2020) The effects of climate change on respiratory allergy and asthma induced by pollen and mold allergens. Allergy 75(9):2219–2228. https://doi.org/10.1111/all.14476
D’Amato G, Cecchi L, Bonini S, Nunes C, Annesi-Maesano I, et al. (2007) Allergenic pollen and pollen allergy in Europe. Allergy 62(9): 976-990. https://doi.org/10.1111/j1398-9995200701393x
D’Amato, G, Holgate ST, Pawankar R, Ledford DK, Cecchi L, et al. (2015) Meteorological conditions, climate change, new emerging factors, and asthma and related allergic disorders A statement of the World Allergy Organization. WAOJournal 8: 52. https://doi.org/10.1186/s40413-015-0073-0
Dahl Å, Galán C, Hajkova L, Pauling A, Sikoparija B, et al. (2013) The onset, course and intensity of the pollen season In: Sofiev M, Bergmann K-C, editors Allergenic pollen: a review of the production, release, distribution and health impacts Dordrecht: Springer Netherlands (2013) p 29–70. https://doi.org/10.1007/978-94-007-4881-1_3
Damialis A, Halley JM, Gioulekas D, Vokou D (2007) Long-term trends in atmospheric pollen levels in the city of Thessaloniki, Greece. Atmos Environ 41(33): 7011-7021. https://doi.org/10.1016/jatmosenv200705009
Damialis A, Charalampopoulos A, Lazarina M, Diamanti E, Almpanidou V, et al. (2020) Plant flowering mirrored in airborne pollen seasons? Evidence from phenological observations in 14 woody taxa. Atmos Environ 240: 14. https://doi.org/10.1016/jatmosenv2020117708
EAA: European Environmental Agency (2017) Climate change, impacts and vulnerability in Europe 2016. An indicator-based report ISBN 978-92-9213-835-6 ISSN 1977-8449. https://doi.org/10.2800/534806
Eccel E, Zollo AL, Mercogliano P, Zorer R (2016) Simulations of quantitative shift in bio-climatic indices in the viticultural areas of Trentino (Italian Alps) by an open source R package. Comput Electron Agric 127:92–100. https://doi.org/10.1016/j.compag.2016.05.019
Frei T, Gassner E (2008) Climate change and its impact on birch pollen quantities and the start of the pollen season an example from Switzerland for the period 1969–2006. Int J Biometeorol 52(7):667–674. https://doi.org/10.1007/s00484-008-0159-2
Galan C, Alcazar P, Oteros J, Garcia-Mozo H, Aira MJ, et al. (2016) Airborne pollen trends in the Iberian Peninsula. Sci Total Environ 550: 53-59. https://doi.org/10.1016/jscitotenv201601069
Galera MD, Elvira-Rendueles B, Moreno JM, Negral L, Ruiz-Abellón MC et al (2018) Analysis of airborne Olea pollen in Cartagena (Spain). Sci Total Environ 622:436–445. https://doi.org/10.1016/j.scitotenv.2017.11.349
Gani F, Lombardi C, Barrocu L, Landi M, Ridolo E, Bugiani M, Rolla G, Senna G, Passalacqua G (2018) The control of allergic rhinitis in real life: a multicenter cross-sectional Italian study. Clin Mol Allergy 16: https://doi.org/10.1186/s12948-018-0082-y
Garcia-Mozo H, Yaezel L, Oteros J, Galan C (2014) Statistical approach to the analysis of olive long-term pollen season trends in southern Spain. Sci Total Environ 473:103–109. https://doi.org/10.1016/j.scitotenv.2013.11.142
Garcia-Mozo H, Oteros JA, Galan C (2016) Impact of land cover changes and climate on the main airborne pollen types in Southern Spain. Sci Total Environ 548: 221-228. https://doi.org/10.1016/jscitotenv201601005
Gehrig R (2006) The influence of the hot and dry summer 2003 on the pollen season in Switzerland. Aerobiologia 22(1):27–34. https://doi.org/10.1007/s10453-005-9013-8
Gehrig R, Clot B (2021) 50 years of pollen monitoring in Basel (Switzerland) demonstrate the influence of climate change on airborne pollen. Front Allergy, 2. https://doi.org/10.3389/falgy.2021.677159
Ghitarrini S, Galan C, Frenguelli G, Tedeschini E (2017) Phenological analysis of grasses (Poaceae) as a support for the dissection of their pollen season in Perugia (Central Italy). Aerobiologia 33(3):339–349. https://doi.org/10.1007/s10453-017-9473-7
Glick S, Gehrig R, Eeftens M (2021) Multi-decade changes in pollen season onset, duration, and intensity: a concern for public health? Sci Total Environ 781. https://doi.org/10.1016/jscitotenv2021146382
Gottardini E, Cristofolini F (1996) Survey of Ambrosia sp. presence by aerobiological monitoring. Colloquium Phytosociology, XXIV, 697–700.
Hidalgo-Galvez MD, Garcia-Mozo H, Oteros J, Mestre A, Botey R, Galan C (2018) Phenological behaviour of early spring flowering trees in Spain in response to recent climate changes. Theor Appl Climatol 132(1-2): 263-273. https://doi.org/10.1007/s00704-017-2089-6
Hoebeke L, Bruffaerts N, Verstraeten C, Delcloo A, De Smedt T, et al. (2018) Thirty-four years of pollen monitoring: an evaluation of the temporal variation of pollen seasons in Belgium. Aerobiologia 34(2): 139-155. https://doi.org/10.1007/s10453-017-9503-5
Husna SMN, Tan HTT, Shukri NM, Ashari NSM, Wong KK (2022) Allergic rhinitis: a clinical and pathophysiological overview. Front Med, 9. https://doi.org/10.3389/fmed.2022.874114
IPCC (2022) Climate Change 2022: Impacts, adaptation and vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [H.-O. Pörtner, D.C. Roberts, M. Tignor, E.S. Poloczanska, K. Mintenbeck, A. Alegría, M. Craig, S. Langsdorf, S. Löschke, V. Möller, A. Okem, B. Rama (eds.)]. Cambridge University Press. Cambridge University Press, Cambridge, UK and New York, NY, USA, 3056 pp., https://doi.org/10.1017/9781009325844.
Jochner S, Hofler J, Beck I, Gottlein A, Ankerst DP, et al. (2013) Nutrient status: a missing factor in phenological and pollen research? J Exp Bot 64(7): 2081-2092. https://doi.org/10.1093/jxb/ert061
Jochner-Oette S, Menzel A, Gehrig R, Clot B (2019) Decrease or increase? Temporal changes in pollen concentrations assessed by Bayesian statistics. Aerobiologia 35:153-163. https://doi.org/10.1007/s10453-018-9547-1
Katz DSW, Morris JR, Batterman SA (2020) Pollen production for 13 urban North American tree species: allometric equations for tree trunk diameter and crown area. Aerobiologia 36(3):401–415. https://doi.org/10.1007/s10453-020-09638-8
Koutsoyiannis D, Kundzewicz Z (2020) Atmospheric temperature and CO2: Hen-Or-Egg Causality? Sci, 2(4), 83. https://doi.org/10.3390/sci2040083
Lind T, Ekebom A, Kubler KA, Ostensson P, Bellander T, Lohmus M (2016) Pollen season trends (1973-2013) in Stockholm Area, Sweden. Plos One 11(11). https://doi.org/10.1371/journalpone0166887
Luschkova D, Traidl-Hoffmann C, Ludwig A (2022) Climate change and allergies. Allergo J Int 31:114–120. https://doi.org/10.1007/s40629-022-00212-x
Majeed HT, Periago C, Alarcon M, Belmonte J (2018) Airborne pollen parameters and their relationship with meteorological variables in NE Iberian Peninsula. Aerobiologia 34(3):375–388. https://doi.org/10.1007/s10453-018-9520-z
Makra L, Csepe Z, Matyasovszky I, Deak AJ, Sumeghy Z, Tusnady G (2014) The effects of the current and past meteorological elements influencing the current pollen concentrations for different taxa. Bot Stud, 55. https://doi.org/10.1186/s40529-014-0043-9
Maya-Manzano JM, Sadys M, Tormo-Molina R, Fernandez-Rodriguez S, Oteros J, et al. (2017) Relationships between airborne pollen grains, wind direction and land cover using GIS and circular statistics. Sci Total Environ 584: 603-613. https://doi.org/10.1016/jscitotenv201701085
Menzel A, Sparks TH, Estrella N, Koch E, Aasa A, et al. (2006) European phenological response to climate change matches the warming pattern. Glob Change Biol, 12(10), 1969–1976. https://doi.org/10.1111/j1365-2486200601193x
Menzel A, Yuan Y, Matiu M, Sparks T, Scheifinger H, et al. (2020) Climate change fingerprints in recent European plant phenology. Glob Change Biol 26:2599–612. https://doi.org/10.1111/gcb15000
Morin X, Lechowicz MJ, Augspurger C, O’ Keefe J, Viner D, Chuine I (2009) Leaf phenology in 22 North American tree species during the 21st century. Glob Change Biol 15-4: 961-975. https://doi.org/10.1111/j1365-2486200801735x
Parmesan C (2006) Ecological and evolutionary responses to recent climate change. Annu Rev Ecol Evol S 37: 637-669. https://doi.org/10.1146/annurevecolsys37091305110100
Penuelas J, Filella I, Zhang XY, Llorens L, Ogaya R, et al. (2004) Complex spatiotemporal phenological shifts as a response to rainfall changes. New Phytol 161(3): 837-846. https://doi.org/10.1111/j1469-8137200401003x
Pesendorfer MB, Koenig WD, Pearse IS, Knops JMH, Funk KA (2016) Individual resource limitation combined with population-wide pollen availability drives masting in the valley oak (Quercus lobata). J Ecol 104(3):637–645. https://doi.org/10.1111/1365-2745.12554
Pfaar O, Bastl K, Berger U, Buters J, Calderon MA, Clot B, Darsow U, Demoly P, Durham SR, Galan C, Gehrig R, Gerth van Wijk R, Jacobsen L, Klimek L, Sofiev M, Thibaudon M, Bergmann KC (2017) Defining pollen exposure times for clinical trials of allergen immunotherapy for pollen induced rhinoconjunctivitis – an EAACI Position Paper. Allergy 72:713–722. https://doi.org/10.1111/all.13092
Picornell A, Ruiz-Mata R, Rojo J, Oteros J, Recio M, de Gálvez-Montañez E, Trigo MM (2023) Applying wind patterns and land use to estimate the concentrations of airborne pollen of herbaceous taxa in a statistical framework. Urban Clim, 49. https://doi.org/10.1016/j.uclim.2023.101496
Prosser F, Bertolli A, Festi F, Perazza G (2019) Flora del Trentino. Fondazione Museo Civico di Rovereto ISBN: 9788874982974
Puljak T, Mamic M, Mitic B, Hrga I, Hrusevar D (2016) First aerobiological study in Mediterranean part of Croatia (Dalmatia): pollen spectrum and seasonal dynamics in the air of Split. Aerobiologia 32(4):709–723. https://doi.org/10.1007/s10453-016-9444-4
Rauer D, Gilles S, Wimmer M, Frank U, Mueller C et al (2021) Ragweed plants grown under elevated CO2 levels produce pollen which elicit stronger allergic lung inflammation. Allergy 76(6):1718–1730. https://doi.org/10.1111/all.14618
Rogers CA, Wayne PM, Macklin EA, Muilenberg ML, Wagner CJ, et al. (2006) Interaction of the onset of spring and elevated atmospheric CO2 on ragweed (Ambrosia artemisiifolia L) pollen production. Environ Health Perspect 114(6): 865-869. https://doi.org/10.1289/ehp8549
Rojo J, Rapp A, Lara B, Fernandez-Gonzalez F, Perez-Badia R (2015) Effect of land uses and wind direction on the contribution of local sources to airborne pollen. Sci Total Environ 538: 672-682. https://doi.org/10.1016/jscitotenv201508074
Rojo J, Picornell A, Oteros J, Werchan M, Werchan B, et al. (2021) Consequences of climate change on airborne pollen in Bavaria, Central Europe. Reg Environ Change 21(1). https://doi.org/10.1007/s10113-020-01729-z
Schramm PJ, Brown CL, Saha S, Conlon KC, Manangan AP, et al. (2021) A systematic review of the effects of temperature and precipitation on pollen concentrations and season timing, and implications for human health. Int J Biometeorol 65(10): 1615-1628. https://doi.org/10.1007/s00484-021-02128-7
Voltolini S, Dignetti P, Rogkakou A, Altomonte F, Mezzetta S, et al. (2011) Importance of Ostrya carpinifolia tree pollen exposure in exacerbating seasonal asthma. Allergy 66:3–3
Walther GR, Post E, Convey P, Menzel A, Parmesan C, et al. (2002) Ecological responses to recent climate change. Nature 416(6879): 389-395. https://doi.org/10.1038/416389a
Wan SQ, Yuan T, Bowdish S, Wallace L, Russell SD, et al. (2002) Response of an allergenic species Ambrosia psilostachya (Asteraceae), to experimental warming and clipping: Implications for public health. Am J Bot 89(11):1843–1846. https://doi.org/10.3732/ajb.89.11.1843
Zhang Y, Seiner A (2022) Projected climate-driven changes in pollen emission season length and magnitude over the continental United States. Nat Commun 13:1234. https://doi.org/10.1038/s41467-022-28764-0
Zhang Y, Bielory L, Mi ZY, Cai T, Robock A, Georgopoulos P (2015) Allergenic pollen season variations in the past two decades under changing climate in the United States. Glob Change Biol 21(4): 1581-1589. https://doi.org/10.1111/gcb12755
Ziello C, Sparks TH, Estrella N, Belmonte J, Bergmann KC, et al. (2012) Changes to airborne pollen counts across Europe. Plos One 7(4): 8. https://doi.org/10.1371/journalpone0034076
Ziska LH (2020) An overview of rising CO2 and climatic change on aeroallergens and allergic diseases. Allergy Asthma Immunol Res 12(5):771–782. https://doi.org/10.4168/aair.2020.12.5.771
Ziska LH, Makra L, Harry SK, Bruffaerts N, Hendrickx M, et al. (2019) Temperature-related changes in airborne allergenic pollen abundance and seasonality across the northern hemisphere: a retrospective data analysis. Lancet Planet Health 3(3): E124-E131. https://doi.org/10.1016/s2542-5196(19)30015-4
Zorer R, Eccel E, Tognetti R, La Porta N (2014) A Mediterranean conifer under vegetation shift: seasonal changes of photochemical activity in Cupressus sempervirens (L.) and evidence of correlation with temperature models. Ital J Agrometeorol, 19(1), 29-40.
Acknowledgements
We are grateful to Maria Cristina Viola for her invaluable work in analyzing pollen samples, to Roberto Zorer for his support in analyzing the changes in land use, and to Josè Oteros for his input in the first phase of data analysis.
Funding
Open access funding provided by Fondazione Edmund Mach - Istituto Agrario di San Michele all'Adige within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by George Zittis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cristofolini, F., Cristofori, A., Corradini, S. et al. The impact of temperature on increased airborne pollen and earlier onset of the pollen season in Trentino, Northern Italy. Reg Environ Change 24, 60 (2024). https://doi.org/10.1007/s10113-024-02223-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10113-024-02223-6