Abstract
Accurate prediction of the outcome of molecular target-based treatment in advanced renal cell carcinoma (RCC) is an important clinical problem. Positron emission tomography/computed tomography using [18F]-2-fluoro-2-deoxyglucose (FDG PET/CT) is a noninvasive tool for the assessment of glucose accumulation which can be a marker of the biological characteristics of the tumor. In this paper, we assess FDG PET/CT as a survival prognostic marker in patients with advanced RCC. The study included 121 patients treated in the years 2011–2016 with a diagnosis of advanced renal cell carcinoma (stage IV, multifocal metastases in all patients). Assessment using FDG PET/CT was conducted by measuring the maximum standard uptake value (SUVmax) for the marker used (the highest SUV measurement result for each patient in a single examination). SUVmax measurements were compared with various clinical risk factors used as prognostic markers. The median follow-up period was 19 months (ranging from 3 to 61 months). SUVmax measurements in all patients ranged from 1.3 to 30.0 (median 6.9). Higher SUVmax was correlated with poorer prognosis. Multi-way analysis with standard risk factors revealed that SUVmax was an independent factor for overall survival (OS; p < 0.003, hazard ratio 1.312, 95% CI 1.147–1.346). For SUVmax < 7.0, median OS was 32 months. For 7.0 ≤ SUVmax < 12.0, median OS was 12.5 months. For SUVmax ≥ 12.0, median OS was 10 months. The differences were statistically significant. A preliminary SUVmax assessment conducted using FDG PET/CT can provide information useful in the prediction of survival of patients with advanced RCC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Renal cell carcinoma (RCC) is not a common cancer, constituting 3% of all tumors in adults [1]. Metastases are detected in approximately 30% of RCC patients, with further 30–40% developing metastases after radical nephrectomy with curative intent [2, 3]. Cytokine treatments have been available for advanced RCC patients for a long time and have been associated with an uncertain prognosis [4, 5]. Molecular targets, such as vascular endothelial growth factor (VEGF) or mTOR kinase, have been selected for newer therapeutics to improve the therapeutic index [6,7,8,9] and are recommended as the main treatments for advanced RCC [10, 11]. It is commonly known that prognosis in RCC patients can vary, and the guidelines recommend treatments employing prognostic classifications based on a combination of clinical information and laboratory data [8, 10, 11].
Positron emission tomography/computed tomography using [18F]-2-fluoro-2-deoxyglucose (FDG PET/CT) is a useful noninvasive tool for the assessment of glucose metabolism which can be a marker of the biological activity of the tumor. We focused on the standard uptake value (SUV). SUVmax was described by other authors as a simplified quantitative measure of FDG accumulation (i.e., the highest SUV of all RCC lesions in each patient) that predicted overall survival (OS) of patients with advanced RCC [12].
In this article, we present our analyses of OS of patients with SUVmax higher and lower than the threshold value of 7.8. Kayani reported that high SUVmax was correlated with a shorter OS in patients treated with the tyrosine kinase inhibitor (TKI) sunitinib [13]. In another study, Chen reported that the baseline SUVmax was correlated with the OS of RCC patients treated with everolimus, an oral mTOR inhibitor (mTORi) [14]. Other authors also supported the usefulness of FDG PET/CT as a prognostic tool for patients with RCC [15, 16].
In this study, we present our results from a longer follow-up period.
Methods
Patients
The analysis was conducted in 121 patients diagnosed with clear-cell RCC, treated at the Oncology Center in Bydgoszcz, Poland, between April 2011 and April 2016.
Imaging
The patients reported for examination at least 6 h after the last meal and drank 0.5–0.75 L of still unflavored mineral water.
After administration of the radiopharmaceutical FDG, the patients waited for the examination for approximately 1 h in a recumbent or seated position and were advised to reduce physical activity. During that time, physiological distribution of the marker in the body occurred. After intravenous administration, most of the FDG is quickly cleared from the vascular space (T1/2 under 1 min) and undergoes biodistribution in tissues (T1/2 up to 1.5 h). Approximately 4% of the administered activity accumulates in the heart, 8% in the brain, 5% in the liver and 3% in the lungs. The remaining 80% of the administered activity is distributed in all other tissues. Approximately 30% of the fraction distributed in all other tissues (24% of the total administered activity) is excreted with urine (T1/2 12 min—25% and T1/2 1.5 h—75%). Physiological uptake of FDG by brown adipose tissue has also been observed.
Isotope Fluorine-18—positron-emitting isotope with a half-life of 109.771 min.
To conduct the examination, FDG with an activity of 5–7 MBq/kg b.w. was administered. Directly after the administration of FDG, 20 mg of furosemide was (optionally) administered to increase renal excretion.
-
Acquisition start time 60 minutes after FDG administration.
-
Patient body position Lying in a supine position, with hands behind the head.
-
Imaging range from the cranial base to mid-thigh. In cases requiring additional acquisitions: areas of the head and limbs.
-
The examinations were conducted using a PET/CT scanner Biograph mCT 128 (SIEMENS).
-
Acquisition parameters CT WB: Topogram—Standard, Eff.mAs—Care Dose 4d, kV—120, Slice—5.0 mm, Acq—32×1.2 mm, Pitch—0.7, Direction—Craniocaudal, Kernel—B30f, FoV—780 mm, Increment—3.0 mm.
-
PET WB Isotope—F-18, Pharm—FDG, Scan Range—Match CT Range, Scan Duration/Bed—1.8 min.
-
PET recon 1 Output Image—Corrected, Recon Meth—TRUeX+TOF(UltraHD-PET), Iteration—2, Subset—21, Image Size—200, Filter—Gaussian, Zoom—1.0, FWHM—5.0.
-
PET recon 2 Output Image—Uncorrected, Recon Meth—Iterative+TOF, Iteration—3, Subset—21, Image Size—200, Filter—Gaussian, Zoom—1.0, FWHM—5.0.
-
CT was performed as per protocol following diagnostic criteria, without or with intravenous administration of contrast depending on the clinical requirements.
-
Image reconstruction Iterative reconstruction algorithm taking into account the correction of absorption and dispersion phenomena.
-
Processing of quantitative and image data Assessment of the intensity of distribution of the marker in pathological lesions (SUV, semiquantitative assessment).
Statistical analysis
The arithmetic mean and median OS were calculated using the Microsoft® Excel 2011 spreadsheet. Statistical significance of differences between the obtained results was checked using the logrank test (p < 0.05) in the Statistica software v. 13.0 by StatSoft.
OS was calculated between the date of the FDG PET/CT examination and the date of death. OS curves were estimated using the Kaplan–Meier method and compared using the logrank test. The impact of SUVmax and other standard clinicopathological factors (fitness, gap between diagnosis and start of treatment, LDH and calcium levels, age, sex and histopathological assessment) on OS were analyzed using monodimensional Cox regression model at p < 0.05 and using a multidimensional Cox model.
Sample FDG PET/CT images as a function of identified SUVmax cutoff points (Fig. 1).
Results
Patient characteristics
The clinical characteristics of the 121 patients are presented in Table 1. Among 79 patients with stage IV RCC, 42 had not previously undergone nephrectomy. FDG PET/CT assessment in 12 patients who had received prior treatment was conducted more than 3 weeks after the end of the previous treatment.
Treatment methods
The median follow-up period was 19 months (ranging from 6 to 61 months). During the follow-up, 60 patients were treated with a single intervention (20 with sorafenib, 31 with sunitinib), 29 were treated with two interventions (20 with TKI and mTORi, 9 with two TKIs), while the remaining patients were treated with three or more interventions (Table 2). Sixty-three tumor-related deaths were observed; the remaining 58 patients were confirmed to be alive at the time of writing of this paper. There were deaths due to other causes.
FDG PET/CT assessment
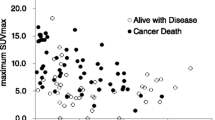
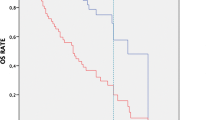
SUVmax measurements in all patients ranged from 1.3 to 30.0 (median 6.9). With SUVmax analyzed as a continuous variable, it was found that high SUVmax was associated with a shorter OS (Fig. 2; p < 0.003, hazard ratio 1.311, 95% CI 1.198–1.361). The impact of SUVmax on OS was compared with that of a range of standard risk factors. Multidimensional analysis of SUVmax in association with fitness status, calcium level and the gap between diagnosis and start of treatment (p < 0.016 in monodimensional analysis) revealed that SUVmax was a significant independent predictor of OS (Table 3).
We first checked the correctness of selecting SUVmax = 7.0 as the cutoff value for establishing prognosis in a report [17] regarding a group of 101 patients. We categorized 121 patients from the current study into three subgroups according to their SUVmax, as suggested by that report [17]. In the current study, median OS for 60 RCC patients with SUVmax < 7.0 was 32 months, while in 28 RCC patients with 7.0 ≤ SUVmax < 12.0, median OS was 12.5 months (95% CI 4.97–19.45; p < 0.003).
For 33 patients (27%) with SUVmax ≥ 12.0, median OS was 10 months (95% CI 1.3–9.7). Differences in OS for these subgroups of patients were statistically significant (< 7.0 vs. ≥ 7.0 and < 12.0: p = 0.003; ≥ 7.0 and < 12.0 vs. ≥ 12.0: p = 0.04; Fig. 1). Regardless of the tumor size and the organs in which metastases were found, patients with a lower SUVmax had a longer OS than patients with a higher SUVmax.
Discussion
We demonstrated that SUVmax in a FDG PET/CT examination is a useful prognostic marker of overall survival of patients with advanced RCC. Our results are consistent with those reported by Nakaigawa et al. (BMC Cancer (2016) 16:67) who showed that SUVmax was an independent prognostic factor for OS [17].
It is rational to claim that RCC with a high SUVmax would be associated with a poorer prognosis, because it is suggested that RCC with a fast metabolism requires more glucose as a source of energy.
Many researchers attempt to determine methods of establishing prognosis for RCC. The MSKCC classification recommended by Motzer et al. is the most common method [18] which divides patients into three subgroups: favorable-, intermediate- and high-risk patients with a median OS of 30, 14 and 5 months, respectively.
In our opinion, SUVmax could be a better prognostic marker compared to the risk factors used in the MSKCC if the results are confirmed and validated in future larger series of patients.
The main treatments in our study included those aimed at molecular targets that inhibited the biological activity of the tumor. As a result, the unique biological properties of RCC were significantly involved in obtaining clinical remission. Although the size of our study was relatively small, the results were statistically significant.
Some researchers suggest that FDG PET/CT is generally not appropriate for assessing RCC because of the urinary excretion of the radiopharmaceutical, which can mask the presence of primary lesions [19, 20]. Recently, several research groups have shown high usefulness of FDG PET/CT in assessing RCC response to molecular target-based treatments [21,22,23].
The prognostic assessment used in our study is clinically beneficial, since molecular target-based treatments, as opposed to the conventional cytotoxic antineoplastic agents, do not always cause an obvious response of the tumor. FDG PET/CT demonstrates that the uptake of FDG can be used not only as a prognostic marker before treatment, but also as a tool for a real-time assessment of the biological activity status of RCC.
Conclusions
A preliminary SUVmax assessment conducted using FDG PET/CT can provide information useful in the prediction of survival of patients with advanced RCC.
References
Chow WH, Devesa SS, Warren JL, Fraumeni JF Jr. Rising incidence of renal cell carcinoma in the United States. JAMA. 1999;281:1628–31.
Linehan WM, Walther MM, Alexander RB, Rosenberg SA. Adoptive immunotherapy of renal cell carcinoma: studies from the Surgery Branch, National Cancer Institute. Semin Urol. 1993;11:41–3.
Janzen NK, Kim HL, Figlin RA, Belldegrun AS. Surveillance after radical or partial nephrectomy for localized renal cell carcinoma and management of recurrent disease. Urol Clin North Am. 2003;30:843–52.
Kaehler KC, Sondak VK, Schadendorf D, Hauschild A. Pegylated interferons: prospects for the use in the adjuvant and palliative therapy of metastatic melanoma. Eur J Cancer. 2010;46(1):41–6.
Negrier S, Escudier B, Lasset C, et al. Recombinant human interleukin-2, recombinant human interferon alfa-2a, or both in metastatic renal cell carcinoma: Groupe Francais D’Immunotherapie. N Engl J Med. 1998;338:1272–8.
Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal cell carcinoma. N Engl J Med. 2007;356:115–24.
Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced clear cell renal cell carcinoma. N Engl J Med. 2007;356:125–34.
Song M. Recent developments in small molecule therapies for renal cell carcinoma. Eur J Med Chem. 2017;142:383–92.
Kumar R, Kapoor A. Current management of metastatic renal cell carcinoma: evolving new therapies. Curr Opin Support Palliat Care. 2017;11(3):231–7.
European Association of Urology: The compilation of the complete Guidelines should be referenced as: EAU Guidelines. Edn. presented at the EAU Annual Congress Copenhagen 2018. ISBN 978-94-92671-01-1. http://uroweb.org/guideline/renal-cell-carcinoma/.
National Comprehensive Cancer Network: NCCN Clinical Practice Guideline in Oncology: Kidney Cancer Version 2.2019—September 17, 2018. https://www.nccn.org/professionals/physician_gls/pdf/kidney.
Namura K, Minamimoto R, Yao M, et al. Impact of maximum standardized uptake value (SUVmax) evaluated by 18-fluoro-2-deoxy-d-glucose positron emission tomography/computed tomography (18 F-FDG-PET/CT) on survival for patients with advanced renal cell carcinoma: a preliminary report. BMC Cancer. 2010;10:667.
Kayani I, Avril N, Bomanji J, et al. Sequential FDG-PET/CT as a biomarker of response to Sunitinib in metastatic clear cell renal cancer. Clin Cancer Res. 2011;17:6021–8.
Chen JL, Appelbaum DE, Kocherginsky M, et al. FDG-PET as a predictive biomarker for therapy with everolimus in metastatic renal cell cancer. Cancer Med. 2013;2:545–52.
Ferda J, Ferdova E, Hora M, et al. 18 F-FDG-PET/CT in potentially advanced renal cell carcinoma: a role in treatment decisions and prognosis estimation. Anticancer Res. 2013;33:2665–72.
Mizuno T, Kamai T, Abe H, et al. Clinically significant association between the maximum standardized uptake value on 18 F-FDG PET and expression of phosphorylated Akt and S6 kinase for prediction of the biological characteristics of renal cell cancer. BMC Cancer. 2015;15:1097.
Nakaigawa N, Kondo K, Tateishi U, et al. FDG PET/CT as a prognostic biomarker in the era of molecular-targeting therapies: max SUVmax predicts survival of patients with advanced renal cell carcinoma. BMC Cancer. 2016;16:67.
Motzer RJ, Bacik J, Murphy BA, Russo P, Mazumdar M. Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol. 2002;20:289–96.
Ma H, Shen G, Liu B, Yang Y, Ren P, Kuang A. Diagnostic performance of 18F-FDG PET or PET/CT in restaging renal cell carcinoma: a systematic review and meta-analysis. Nucl Med Commun. 2017;38(2):156–63.
Kitajima K, Yamamoto S, Fukushima K, Minamimoto R, Kamai T, Jadvar H. Update on advances in molecular PET in urological oncology. Jpn J Radiol. 2016;34(7):470–85.
Ranieri G, Marech I, Niccoli Asabella A, et al. Tyrosine-Kinase inhibitors therapies with mainly anti-angiogenic activity in advanced renal cell carcinoma: value of PET/CT in response evaluation. Int J Mol Sci. 2017;18(9):pii: E1937.
Ueno D, Yao M, Tateishi U, et al. Early assessment by FDG-PET/CT of patients with advanced renal cell carcinoma treated with tyrosine kinase inhibitors is predictive of disease course. BMC Cancer. 2012;12:162.
Kayani I, Avril N, Bomanji J, et al. Sequential FDG-PET/CT as a biomarker of response to Sunitinib in metastatic clear cell renal cancer. Clin Cancer Res. 2011;17(18):6021–8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Pankowska, V., Malkowski, B., Wedrowski, M. et al. FDG PET/CT as a survival prognostic factor in patients with advanced renal cell carcinoma. Clin Exp Med 19, 143–148 (2019). https://doi.org/10.1007/s10238-018-0539-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-018-0539-9