Abstract
The Lesser Grey Shrike is a typical Palaearctic songbird for which we have limited knowledge of its migration ecology. All that is known about its non-breeding movements is inferred from observational data of birds on passage. The few available ring recovery data do not link breeding and African non-breeding grounds. By deploying two types of loggers, light-level geolocators and multi-sensor loggers, on birds from a declining Slovak breeding population, we present the first direct evidence for non-breeding grounds, loop migration, stopover sites and the timing of annual cycle events. With barometric data, we provide details on flight altitudes during migration. The two tracked birds migrated in a clear anti-clockwise loop to S Africa. Autumn migration tracks went through the Balkan Peninsula, Mediterranean Sea towards Libya with unusually long stays around N Chad and Niger. The next stopovers were in Angola, and the main non-breeding sites were in Botswana. Spring migration commenced on March 29 and April 7 and the birds took routes along East African countries, with stopovers later in Somalia and Saudi Arabia, before crossing the eastern Mediterranean Sea. Nocturnal migration dominated, but for three days in August, while crossing the Sahara Desert, the bird extended flights into the day with a sudden increase in flight altitudes at dawn. Flight altitudes were higher during barrier crossing and during the last phase of spring migration compared to the remaining periods, with the most extreme event recorded at 4530 m asl.
Zusammenfassung
Nichtbrutgebiete, Schleifenzug und Aktivitätsmuster im Jahreszyklus bei Schwarzstirnwürgern Lanius minor vom nordwestlichen Rand seines Verbreitungsgebietes
Der Schwarzstirnwürger ist eine der paläarktischer Singvogelarten, für die nur begrenzte Kenntnisse der Zugökologie vorliegen. Alle Informationen zu Bewegungen außerhalb der Brutzeit stammen von Beobachtungsdaten durchziehender Vögel. Die wenigen verfügbaren Ringwiederfunde erlauben keine Verbindungen zwischen Brut- und Überwinterungsplätzen. Wir präsentieren hier den ersten direkten Nachweis des Schleifenzuges, die zeitliche Abfolge der Ereignisse im Jahreszyklus, sowie die Rast- und Überwinterungsgebiete von Schwarzstirnwürgern einer abnehmenden, slowakischen Brutpopulation basierend auf Licht- und Multisensor-Loggerdaten. Barometrische Daten liefern zudem detaillierte Angaben zu individuellen Flughöhen während des Zuges. Die beiden getrackten Vögel zogen in einer klaren Schleife gegen den Uhrzeigersinn nach Südafrika. Der Herbstzug führte über die Balkanhalbinsel und das Mittelmeer nach Libyen, gefolgt von unerwartet langen Zwischenstopps im nördlichen Tschad und Niger. Die nächsten Rastgebiete lagen in Angola, die Überwinterungsgebiete in Botswana. Der Frühlingszug begann am 29. März und 7. April. Die Vögel flogen über Ostafrika mit Zwischenstopps in Somalia und in Saudi-Arabien, ehe sie das östliche Mittelmeer überquerten. Nächtliche Flüge dominierten während des Zuges. Bei der Durchquerung der Sahara im August waren jedoch drei der Nachtflüge um mehrere Stunden in den Tag hinein verlängert und wiesen einen plötzlichen Anstieg der Flughöhen während der Morgendämmerung auf. Die Flughöhen während der Barrieren-Überquerung und während der letzten Phase im Frühjahrszug waren zudem höher als in der restlichen Zugzeit, wobei eine maximale Flughöhe mit 4530 m ü. M. erreicht wurde.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Lesser Grey Shrike has a considerable breeding range spanning from NE Spain in the West, the Pannonian basin and the eastern Europe to Altai in the East, but unusually, its non-breeding range funnels in Southern Africa to a relatively small area (Lefranc and Worfolk 1997). Its populations at the western edge of its distributional range underwent substantial declines and area shrinkages, and decreasing numbers are also reported at the African non-breeding grounds (Herremans 1998; Bronskov and Keller 2020; PECMBS 2022). At the same time, we have very limited knowledge on the migration and overwintering ecology of the species. All available information on migration is inferred from bird observations. Earlier authors proposed an anticlockwise loop migration pattern, where in spring most birds use the Arabian Peninsula and Anatolia when heading towards the breeding sites (Stresemann 1943; Moreau 1961; Dowsett 1971; Lefranc and Worfolk 1997). This migration pattern is clearly visible in seasonal observation maps generated by bird observations submitted to the eBird database (Fink et al. 2022; Fig. 1b). To date, there is only a single observation linking breeding and non-breeding grounds (Krištín 2008). To fill parts of this gap, we analysed available ring recovery data and deployed two types of loggers (light-level geolocators and multi-sensor loggers) on shrikes in a breeding population at the North-western edge of its distributional range. We aimed at identifying for the first time individual-based migration tracks, locations of stopover sites and non-breeding sites of Lesser Grey Shrikes.
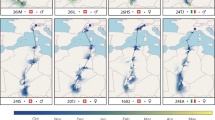
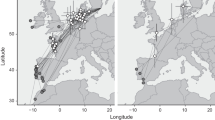
Comparison of available Lesser Grey Shrike distribution data relative to the data collected in this study. a Ringing and recovery sites of Lesser Grey Shrikes connected by great circle distance lines relative to eBird observer data. b Modelled relative abundance of Lesser Grey Shrikes per season from eBird (Fink et al. 2022). Abundance is presented as a gradient in red (breeding season), yellow (post-breeding/pre-breeding season) or blue (non-breeding season), and darker colours indicate higher relative abundance and lighter colours indicate lower relative abundance within the season. c Estimated migration tracks and stationary sites used by individuals 16KN (autumn = light blue, spring = light red) and 22UL (autumn = dark blue, spring = dark red) in this study. The track of 16KN was estimated using GeoPressureR, and shows the shortest most likely path of the bird. The dashed portion of the track connecting the last pre-breeding stopover site to the breeding site is the great circle distance between the two sites. The track of 22UL was estimated using SGAT and shows the median most likely path of the bird. The circles indicate the stationary sites used during each season (red = breeding, dark yellow = post-breeding, blue = non-breeding, light yellow = pre-breeding). Stationary sites were defined as stops ≥ 48 h
Material and methods
We studied Lesser Grey Shrikes at the foothills of Poľana Mts., Central Slovakia (48.5909N, 19.5161E as a centre of study area, 460–750 m asl). Here, this declining and endangered species breeds in a traditionally cultivated rural landscape; c.f. Krištín et al. (2000, 2007), still with a local population size of 30–35 nests/20 km2 in 2017–2022 (own unpublished data). Adult shrikes were trapped within their breeding territories with clap nets (baited with crickets or larvae of Zophobas) during the spring of 2017 and 2019. All trapped birds were marked with aluminium rings and an individual combination of two colour rings. In 2017, we equipped 10 birds (8 males and 2 females) with GDL3-PAM multi-sensor loggers (Swiss Ornithological Institute, Sempach). In 2019, we equipped another 10 birds (6 males and 4 females) with uTags (Swiss Ornithological Institute, Sempach). Loggers were attached using a leg-loop backpack harness (1 mm thick braided nylon string). The mean weight of the GDL3-PAM logger was 1.35 g and for the uTags 1.90 g. The mean male body mass was 44.6 ± 2.4 g (range 39.6–48.6 g), and the female body mass was 49.0 ± 5.74 (44.1–52.6 g). In the following years, we detected four birds (males only) with loggers. We retrieved three loggers and one bird we failed to catch. Return rates of logger-tracked birds were 28.6% (4/14) for males and 0% (0/6) for females. As the population is very small and declining, we did not have return rates on ringed-only birds during the period of this study. However, return rates were 40% for males and 25.2% for females during 1996–2000 (Krištín et al. 2007). The two loggers contained data for the full year, while the third logger stopped working after two months. We do not present data on this bird. Raw data on all three birds are available at the Zenodo data repository (Adamík et al. 2023).
To supplement the geolocator data with other available data sources on bird migration, we requested data on ringing recoveries from the EURING and SAFRING. We requested recoveries which fulfilled the condition of > 100 km between the ringing and recovery site. Only EURING had relevant data. This yielded nine encounters but we used only seven as two were of short distance and uninformative about migration (138 and 182 km within France and the UK, respectively, see Online Supplement 1, Table S1). For estimating distances, we used the ‘orthodrom.dist’ function (great circle distance) in the R-package birdring (Korner-Nievergelt and Robinson 2014). In addition, we used the eBird relative abundance maps to depict the predicted seasonal distribution of shrikes across the annual cycle (Sullivan et al. 2009; Fink et al. 2022). We used the eBird classification of seasons: breeding Jun 21 –Jul 27, non-breeding Dec 14 –Mar 1, post-breeding Aug 3–Dec 7, pre-breeding Mar 8 –Jun 14. Note that these map layers are modelled data and as such inevitably contain inaccuracies, e.g. they model presence of the species in Madagascar where it does not occur.
Light-level data collected from the uTag (bird 22UL) were analyzed using R packages TwGeos (Lisovski et al. 2016) and SGAT (Wotherspoon et al. 2013) to obtain the most likely geographic positions of the shrike. We followed the approach outlined in Lisovski et al. (2020) and the parameters used in Wong et al. (2022), first annotating twilight events using a light-level threshold of 1 in TwGeos. Stationary periods were defined as stops lasting ≥ 2 days. Briefly, we derived median reference solar zenith angles based on the recorded light readings at a non-breeding site (i.e., Hill-Ekstrom calibration). We discerned stationary and movement periods based on abrupt changes in sunrise, sunset, midday and midnight times, and used the ‘mergeGroups’ function in SGAT to group together similar twilight times to a single site. Using SGAT’s Group model, we specified the parameters for the twilight error distribution (gamma), the flight speed distribution (shape: 2.2, rate: 0.08), and a land mask constraining positions to land. A modified gamma model was first run for 1000 iterations to initiate the model, before the model was tuned for five runs with 300 iterations. The final run of the model for 2000 iterations produced the median migration tracks and associated 95% confidence intervals. Calibration parameters used in SGAT are available in Online Supplement 1, Table S2.
The addition of atmospheric pressure information to light-based geolocation has been shown to greatly enhance the accuracy of location estimates (Nussbaumer et al. 2023a). Thus, pressure, activity, and light data recorded from the single GDL3-PAM multi-sensor logger (16KN) were used to predict positions with the R package GeoPressureR (Nussbaumer et al. 2023b). We followed the steps detailed in Nussbaumer (2022). Essentially, GeoPressureR creates a likelihood map of positions based on matching geolocator-measured ambient air pressure data and global reference weather data, and a second likelihood map based on ambient light intensity data to further refine location estimates (Nussbaumer et al. 2023b). We distinguished between stationary and migratory periods based on simultaneous drops in pressure and sustained high activity. We handled outliers using a visual inspection as suggested in Nussbaumer (2022) using TRAINSET (https://www.trainset.geocene.com). This allowed us to compare pressure and activity data simultaneously, and we removed outlying single pressure points that differed significantly from preceding or following points (within 30 min) and did not match the activity level. We used a two-day threshold for stationary sites (i.e. ≥ 48 h), and light data was analyzed based on the threshold method, using breeding-site calibration. The combination of pressure and light likelihood maps is then used to model the trajectory of the bird, based on a gamma-distributed flight speed distribution (min. groundspeed: 20 km/h, average groundspeed: < 120 km/h). The resulting simulated most-likely path is the shortest path between the estimated stationary sites, and a marginal probability of each stationary site regardless of the bird’s trajectory is also produced. Parameters used in GeoPressureR are available in Online Supplement 1, Table S3.
To gain insight on the migratory behaviour of the shrikes throughout the annual cycle, we further classified the activity data recorded by the multi-sensor logger using the R package PAMLr (Dhanjal-Adams 2022). Following the guidelines in Dhanjal-Adams et al. (2022), we used the ‘classify_flap’ function to identify periods of low to high activity (flapping flight) using k-means clustering. Sustained high activity of 60 min or more was considered migration. The function ‘classify_summary_statistics’ was then used to summarize pressure changes and respective flight altitudes per classified flight. All analyses were conducted in R version 4.1.3 (R Core Team 2022). All times are given as UTC.
Results
Ring recoveries
All seven recoveries were related to autumn migration, linking breeding and southern passage sites (Fig. 1a), and none linked breeding and African non-breeding sites. Four birds were ringed as nestlings. Six birds were recovered in Greece in late August and early September (range 23 Aug–20 Sept, mean 8 Sept, mean distance 1200 km). One bird ringed as a nestling in Germany was recovered after 478 km and 60 days on 28 Aug in N Italy. Four birds were recovered in Greece within the same season. Two birds ringed at Hungarian breeding sites were recovered in Greece in the following autumn. Details on recoveries are provided in Online Supplement 1, Table S1.
Geolocation
The first bird (22UL, Fig. 1c) left the breeding site on July 21 (Table 1) and the first stopover was detected for 23 days in Bulgaria (July 25–Aug 18) and a second stay in northern Chad on the border with Libya for 57 days (Aug 24–Oct 20). After that, the bird made a quick 3700 km long movement and on Oct 23 it arrived at Angola where it stayed for 19 days. On Nov 13, the bird arrived at its first main non-breeding site in W Botswana, where it remained until Jan 10 (58 days). After that, it moved about 2° further South within Botswana to spend another 73 days (Jan 15–Mar 29) at the second main non-breeding site. The bird initiated the spring migration on Mar 29 and between Apr 7–9 it took a short stopover in the area of W Mozambique. After that, it took another 10-day stopover from April 15 in NW Kenya, west of Lake Turkana. The last detected stopover was estimated for three days (Apr 29–May 2) in Saudi Arabia. The bird flew towards the breeding site via Cyprus, Anatolia and Balkan with final arrival to the breeding site on May 15 (Fig. 1c).
The second individual (16KN, Table 1; Fig. 1c) departed the breeding site on July 23. It quickly moved to the border region of W Romania and SE Hungary around the Mureș River, where it stayed for 25 days (July 25–Aug 18). On Aug 18, the bird took off for migration. Changes in pressure data show flight for six consecutive nights (Online Supplement 1, Fig. S1). On Aug 24, following the Mediterranean Sea and Sahara crossing, it took a three-day stopover (Aug 24–27) at an estimated location in the border area of Niger and Chad. After that, on Aug 27 it made a short movement further south, where after two nocturnal flights arrived on Aug 29 in the Kanem region, Chad. There, the bird stopped for 52 days (Aug 29–Oct 19), which resembles the pattern in the previously tracked bird (22UL, with departure on Oct 20). On Oct 19, the bird took off and over a series of 9 nights, it moved further south to the next stopover in Angola, where it stayed for 4 days (Oct 27–31). After that, the bird moved southeast, staying 11 days (Nov 3–13) in Western Zambia, in the border region between Angola and Namibia, near the Zambezi River. After this stop, the bird continued further and on Nov 14 it arrived at its final non-breeding site in Central Kalahari Game Reserve, Ghanzi District, Botswana. On Apr 7, after 144 days of residency, the bird initiated spring migration. For eight nights in a row it migrated, took a break from Apr 14, and continued with flight on the night of Apr 15. After that, it took a stopover around Lake Turkana, Kenya for 3.5 days (Apr 16–19). Over the next four nights, the bird moved northeast and it took another 7-day stopover (Apr 23–30) in Somaliland, Somalia, in the area between Karkaar Mts and the Nugaal Valley (Fig. 1c). On Apr 30, the bird departed from the site. Changes in pressure show that the bird continued to make a series of nocturnal flights until May 15, with only one 1-day short stopover on May 7. However, these stopovers are below the set 2-day threshold, and therefore not shown. For comparison, a track modelled with a 12 h threshold, can be seen in Online Supplement 2.
Flight behaviour on migration
Nocturnal flights were the norm in both autumn and spring (Fig. 2). However, three prolonged flights into the day (22–24 Aug) occurred during the barrier crossing of the Mediterranean Sea and the Sahara Desert. Cumulatively, autumn migration lasted 176.8 h for this individual (with events classified as flights ≥ 1 h). On July 23, the bird initiated the first migration flights (4.7 and 1.3 h) across two consecutive nights to its first stopover site in Romania (Fig. 3). Flight durations while barrier crossing ranged from 12.9 to 14.9 h per night. Between the stopover sites in Romania and Chad, the bird made nine flights in total for 69.5 h, mean 7.7 (range 1.8–14.9). From Chad towards the main non-breeding site in Botswana, the bird flew in several bouts, totalling 101.4 h (mean 6.8, range 2.7–11.0).
Actogram of the Lesser Grey Shrike (16KN) over its annual cycle. The left figure shows unclassified activity, the right one classified into four behavioural states (resting, low activity, high activity and migration) using PAMLr. The two white vertical lines indicate the local sunrise and sunset times as recorder by the logger
Spring migration was initiated with a series of nocturnal flights across eight consecutive nights (cumulatively 69.8 flight hours, mean 8.7). After a one-day break, the bird continued with a 9.3 h flight on April 15. Another set of nocturnal flights was initiated on Apr 19 for four consecutive nights (cumulatively 29.5 flight hours). On Apr 30, the bird initiated another set of nocturnal flights (mean 8.5 h, range 5–10.3) but the logger stopped working while the bird was on the move (May 15). By May 11, the spring part of migration took cumulatively 201.8 flight hours. Interestingly, on several occasions while the bird was residing in the same area, it took short nocturnal flights (Fig. 3). For example, while residing in Chad, on Sept 17 it took 2.1 h climb flight up 1096 m. Similarly, on Dec 18 it took a short flight up 1085 m (Online Supplement 1, Fig. S2).
Altitudes of migratory flights
Flight altitudes in autumn were the lowest during the first two flights between the breeding site in Slovakia and the first stopover site, when the single bird reached the first night max 1329 m. Altitude range and heights reached were higher during the barrier crossing phase than during the period when on the move between Chad and Botswana (Table 2). Maximum flight altitude during barrier crossing was 3274 m asl, mean 1198 ± 949 SD m asl. Interestingly, we detected an increase in altitudes at dawn, when the bird started its ascent. This happened on all three mornings when the bird extended nocturnal flight into the day (Fig. 4). At the non-breeding residency site in Botswana, the bird stayed at a relatively constant altitude of 1000 m asl (Online Supplement 1, Fig. S2). Climbing height, record flight altitude (4530 m asl) and altitude range were the highest during the last stretch of migration, after Apr 30 (Table 2).
Discussion
The few available ringing recoveries of the Lesser Grey Shrike show that birds from European breeding sites migrate via Greece in autumn. Funnelling via Greece was likely common for birds from the extinct western European breeding sites (as indicated by the two recoveries of birds from German breeding sites). The only recovery in Africa is a female colour-ringed at our Slovak breeding site on June 10, 1998 by AK and resighted on April 12, 2000 in the area of Awash, Ethiopia (9.00N, 40.17E; Krištín 2008). Data from our two-tracked individuals confirm the earlier suggestions of Stresemann (1943), and Moreau (1961) of an anticlockwise loop migration pattern in this species. This is comparable to the loop migration pattern in the Red-backed Shrike (Tøttrup et al. 2012). Furthermore, both birds in our study made long stops either in the Sahel zone or in an oasis in the Sahara Desert. There, both birds spent nearly 2 months before moving further south to stopover sites in Angola. Again, this resembles the behaviour of the Red-backed Shrike for which Tøttrup et al. (2012, 2017) and Pedersen et al. (2020) found that the birds spent 1 to 2 months in S Sudan after Sahara crossing, before moving further south to the final non-breeding sites. Such a long residency indicates that this region might be of critical importance for shrikes after barrier crossing. Tøttrup et al. (2012) suggested that the shrikes likely encounter favourable foraging conditions in this area and store fuel for rapid southward movements.
Both tracked individuals resided during boreal winter in the area of the Kalahari Desert. This is the area where the entire global population of the species is seen to concentrate at that time of year (Herremans 1998). Earlier observational studies reported the first arriving birds from late October (Zimbabwe) to late November (Cape Province, S Africa) (Dowsett 1971; Lefranc & Worfolk 1997). Our tracked birds arrived at remarkably similar dates in mid-November. For the spring migration, we confirmed that the species migrates along the E African countries towards Ethiopia and Somalia, the Red Sea, the Middle East, Anatolia and the Balkan Peninsula. In line with the expected pattern of faster migration in spring (Nilsson et al. 2013; Briedis et al. 2020a), the migration speeds of shrikes were also much faster in spring than in autumn. However, travel speeds (i.e. speed excluding stopovers) were much faster in autumn. This resembles the pattern in the Great Reed Warbler when the total flight duration used for covering the full-migration distance was on average shorter in autumn than in spring (Emmenegger et al. 2021). However, all of these estimates should be taken with caution as no geolocator study can provide data on the first fuelling period prior to departure. The question of whether to consider the long autumn residency period in Chad as a stopover also arises. It is likely the birds undergo a partial moult there (Lefranc and Worfolk 1997), and if we assume residency in Chad, our migration speed estimates would be underestimated (but note that we also provide migration speed estimates without the Chad residencies). For example, Schmaljohann et al. (2022) do not consider an interruption of endurance flights due to moult as a stopover. Thus, if the shrikes undergo moult in this area, we should consider this residency period as a unique annual cycle event. This raises the question of how to define autumn migration, e.g. whether to view it as two events – prior and after the Chad residencies. Arrivals of the two birds to the breeding site in mid-May correspond with the long-term observations (mean arrival May 6; Krištín et al. 2007 and Krištín unpubl. data).
For the single individual with the multi-sensor logger data, we show that nocturnal migration was the prevailing pattern. This is in line with the typical pattern of prevailing nocturnal migration found for most trans-Saharan avian migrants (Dorka 1966; Schmaljohann et al. 2007b). The bird only extended its flights shortly into the day on three nights in autumn, during its crossing of the Mediterranean Sea and the Sahara Desert. This observation fits into the expected pattern that prolonged flights occur during barrier crossings (Adamík et al. 2016; Jiguet et al. 2019). Similarly, the extensions of nocturnal flights into the day during Sahara crossing were also found for two Red-backed Shrikes tracked with data loggers (Bäckman et al. 2017a, b). Schmaljohann et al. (2007a) showed that under favourable tail winds, the passerine nocturnal migration measured by radar continued into the day. In their study, landing tendency (sink rate) correlated negatively with tail winds. Thus, both radar and geolocation from this study provide solid evidence for the extension of flights into the day by trans-Saharan migrants. Activity data also significantly improved our understanding of migration behaviour. For example, we could see that migratory flights in spring are more compressed in time than in autumn, with the exception of a series of short nocturnal flights in the autumn. The single individual migrated on average for around 6 h per night in autumn and over 8 h per night in spring. In autumn, the migration was divided into three segments, with similar numbers of flights as was found for the Red-backed Shrike (Sjöberg et al. 2018). Flight altitudes were usually in the range of 1000–3000 m asl, with a record of 4530 m asl., and lowest during the first segment of autumn migration in Europe. These findings are in line with the flight altitudes recorded by the Red-backed Shrike (Tøttrup et al. 2017; Sjöberg et al. 2018). A similar pattern of low flight altitudes over Europe and a sudden increase in altitudes during barrier crossing was also found in European Nightjars (Norevik et al. 2021). Our tracked individual climbed the most metres during the diurnal part of the barrier crossing phase in autumn and in the last stretch of spring migration after leaving Somalia. We found that starting the ascent at dawn occurred over three barrier-crossing events. This was a similar pattern to the Great Reed Warbler described by Sjöberg et al. (2021) or to the sudden daytime ascents in migratory Great Snipes (Lindström et al. 2021). It is likely that the sudden increase in flight altitudes served the bird to find more favourable wind conditions or to reduce water loss (e.g., Schmaljohann et al. 2009). The highest flight altitude of 4530 m asl was recorded during the spring passage (May 2). It was higher than in the Red-backed Shrike or Tawny Pipit (Briedis et al. 2020b), similar to Eurasian Hoopoe, but lower than in the diurnal flights of the Great Reed Warbler (up to 6267 m asl; Sjöberg et al. 2021; Liechti et al. 2018). However, we should interpret this with caution as we had altitudinal data only from a single individual.
Although we provide details on migration behaviour of only two individuals, the gathered data greatly enhance our knowledge of this little-known and endangered long-distance migratory species. Our findings fit well into what was expected (loop migration, prevailing nocturnal migration) or what is already known about nocturnal migrants (extending nocturnal flights into the day, climbs at dawn). Thus we suggest that our findings likely show the general migration pattern in the Lesser Grey Shrike. Collecting data on these few individuals was a challenge in the field. The breeding population continues to decline in our study area and within few years there could be no individuals to track.
Data availability
Raw datalogger data from the tracked birds are available at Zenodo (Adamík et al. 2023).
References
Adamík P, Emmenegger T, Briedis M, Gustafsson L, Henshaw I, Krist M, Laaksonen T, Liechti F, Procházka P, Salewski V, Hahn S (2016) Barrier crossing in small avian migrants: individual tracking reveals prolonged nocturnal flights into the day as a common migratory strategy. Sci Rep 6:21560. https://doi.org/10.1038/srep21560
Adamík, P., Wong, JB., Hahn, S. & Krištín, A. Data from: Non-breeding sites, loop migration and activity patterns over the annual cycle in the Lesser Grey Shrike Lanius minor from a north-western edge of its range (1.0) [Data set], Zenodo, https://doi.org/10.5281/zenodo.7556866 (2023).
Bäckman J, Andersson A, Alerstam T, Pedersen L, Sjöberg S, Thorup K, Tøttrup AP (2017a) Activity and migratory flights of individual free-flying songbirds throughout the annual cycle: method and first case study. J Avian Biol 48:309–319. https://doi.org/10.1111/jav.01068
Bäckman J, Andersson A, Pedersen L, Sjöberg S, Tøttrup AP, Alerstam T (2017b) Actogram analysis of free-flying migratory birds: new perspectives based on acceleration logging. J Comp Physiol A 203:543–564. https://doi.org/10.1007/s00359-017-1165-9
Briedis M, Bauer S, Adamík P et al (2020a) Broad-scale patterns of the Afro-Palaearctic landbird migration. Global Ecol Biogeogr 29:722–735. https://doi.org/10.1111/geb.13063
Briedis M, Beran V, Adamík P, Hahn S (2020b) Integrating light-level geolocation with activity tracking reveals unexpected nocturnal migration patterns of the tawny pipit. J Avian Biol 51:e02546. https://doi.org/10.1111/jav.02546
Bronskov O, Keller V (2020) Lesser Grey Shrike. In: Keller V, Herrando S, Voříšek P, Franch M, Kipson M, Milanesi P, Martí D, Anton M, Klvaňová A, Kalyakin MV, Bauer HG, Foppen RPB (eds) European Breeding Bird Atlas 2: Distribution, Abundance and Change. EBCC & Lynx Edicions, Barcelona, pp 536–537
Dhanjal-Adams K (2022) pamlr: Suite of functions for manipulating pressure, activity, magnetism, temperature and light data in R. R Package Version 2:2
Dhanjal-Adams K, Willener AST, Liechti F (2022) pamlr: A toolbox for analysing animal behaviour using pressure, acceleration, temperature, magnetic or light data in R. J Anim Ecol 91:1345–1360. https://doi.org/10.1111/1365-2656.13695
Dorka V (1966) Das jahres- und tageszeitliche Zugmuster von Kurz- und Langstreckenziehern nach Beobachtungen auf den Alpenpässen Cou/Bretolet (Wallis). Ornithol Beobachter 63:165–223
Dowsett RJ (1971) The Lesser Grey Shrike in Africa. Ostrich 42:259–269. https://doi.org/10.1080/00306525.1971.9634416
Emmenegger T, Bensch S, Hahn S, Kishkinev D, Procházka P, Zehtindjiev P, Bauer S (2021) Effects of blood parasite infections on spatiotemporal migration patterns and activity budgets in a long-distance migratory passerine. Ecol Evol 11:753–762. https://doi.org/10.1002/ece3.7030
Fink D, Auer T, Johnston A, Strimas-Mackey M, Ligocki S, Robinson O, Hochachka W, Jaromczyk L, Rodewald A, Wood C, Davies I, Spencer A (2022) eBird Status and Trends, Data Version: 2021; Released: 2022. Cornell Lab of Ornithology, Ithaca, New York. https://doi.org/10.2173/ebirdst.2021
Herremans M (1998) Monitoring the world population of the Lesser Grey Shrike (Lanius minor) on the non-breeding grounds in southern Africa. J Ornithol 139:485–493. https://doi.org/10.1007/BF01653472
Jiguet F et al (2019) Desert crossing strategies of migrant songbirds vary between and within species. Sci Rep 9:20248. https://doi.org/10.1038/s41598-019-56677-4
Korner-Nievergelt F, Robinson RA (2014) Introducing the R-package ‘birdring.’ Ringing Migr 29:51–61. https://doi.org/10.1080/03078698.2014.933053
Krištín A, Hoi H, Valera F, Hoi C (2000) Breeding biology and breeding success of the Lesser Grey Shrike (Lanius minor) in a stable and dense population. Ibis 142:305–311. https://doi.org/10.1111/j.1474-919X.2000.tb04870.x
Krištín A, Hoi H, Valera F, Hoi C (2007) Philopatry, dispersal patterns and nest-site reuse in Lesser Grey Shrikes (Lanius minor). Biodivers Conserv 16:987–995. https://doi.org/10.1007/s10531-006-9019-8
Krištín A (2008) Ťuhýk menší Lanius minor. In: Cepák J, Klvaňa P, Škopek J, Schröpfer L, Jelínek M, Hořák D, Formánek J, Zárybnický J (eds) Atlas of migratory birds in the Czech and Slovak Republics. Aventinum, Prague, pp 491–492
Lefranc N, Worfolk T (1997) Shrikes. Pica Press, Sussex, A Guide to the Shrikes of the World
Liechti F, Bauer S, Dhanjal-Adams KL, Emmenegger T, Zehtindjiev P, Hahn S (2018) Miniaturized multi-sensor loggers provide new insight into year-round flight behaviour of small trans-Sahara avian migrants. Movement Ecol 6(1):19. https://doi.org/10.1186/s40462-018-0137-1
Lindström Å, Alerstam T, Andersson A, Bäckman J et al (2021) Extreme altitude changes between night and day during marathon flights of great snipes. Cur Biol 31:3433–3439. https://doi.org/10.1016/j.cub.2021.05.047
Lisovski S, Bauer S, Briedis M et al (2020) Light-level geolocator analyses: A user’s guide. J Anim Ecol 89:221–236. https://doi.org/10.1111/1365-2656.13036
Lisovski S, Wotherspoon S, Sumner M (2016) TwGeos: Basic data processing for light-level geolocation archival tags. R package version 0.1.2
Moreau RE (1961) Problems of Mediterranean-Saharan migration. Ibis 103:580–623. https://doi.org/10.1111/j.1474-919X.1961.tb02454.x
Nilsson C, Klaassen RH, Alerstam T (2013) Differences in speed and duration of bird migration between spring and autumn. Am Nat 181:837–845. https://doi.org/10.1086/670335
Norevik G, Åkesson S, Andersson A, Bäckman J, Hedenström A (2021) Flight altitude dynamics of migrating European nightjars across regions and seasons. J Exp Biol 224:jeb242836. https://doi.org/10.1242/jeb.242836
Nussbaumer R, Gravey M, Briedis M, Liechti F (2023a) Global positioning with animal-borne pressure sensors. Methods Ecol Evol 14:1104–1117. https://doi.org/10.1111/2041-210X.14043
Nussbaumer R, Gravey M, Briedis M, Liechti F, Sheldon D (2023b) Reconstructing bird trajectories from pressure and wind data using a highly optimised hidden Markov model. Methods Ecol Evol 14:1118–1129. https://doi.org/10.1111/2041-210X.14082
Nussbaumer R (2022) A user manual for GeoPressureR. https://raphaelnussbaumer.com/GeoPressureManual/
PECMBS (2022) Pan‐European common bird monitoring scheme. Trends of common birds in Europe, Accessed 20 Dec 2022 https://pecbms.info
Pedersen L, Onrubia A, Vardanis Y, Barboutis C, Waasdorp S, van Helvert M, Geertsma M, Ekberg P, Willemoes M, Strandberg R, Matsyna E, Matsyna A, Klaassen RHG, Alerstam T, Thorup K, Tøttrup AP (2020) Remarkably similar migration patterns between different red-backed shrike populations suggest that migration rather than breeding area phenology determines the annual cycle. J Avian Biol 51:e02475. https://doi.org/10.1111/jav.02475
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Schmaljohann H, Liechti F, Bruderer B (2007a) Daytime passerine migrants over the Sahara - are these diurnal migrants or prolonged flights of nocturnal migrants? Ostrich 78:357–362. https://doi.org/10.2989/OSTRICH.2007.78.2.38.118
Schmaljohann H, Liechti F, Bruderer B (2007) Songbird migration across the Sahara: the non-stop hypothesis rejected. Proc R Soc B 274:735–739. https://doi.org/10.1098/rspb.2006.0011
Schmaljohann H, Liechti F, Bruderer B (2009) Trans-Sahara migrants select flight altitudes to minimize energy costs rather than water loss. Beh Ecol Sociobiol 63:1609–1619. https://doi.org/10.1007/s00265-009-0758-x
Schmaljohann H, Eikenaar C, Sapir N (2022) Understanding the ecological and evolutionary function of stopover in migrating birds. Biol Rev 97:1231–1252. https://doi.org/10.1111/brv.12839
Sjöberg S, Malmiga G, Nord A, Andersson A, Bäckman J, Tarka M, Willemoes M, Thorup K, Hansson B, Alerstam T, Hasselquist D (2021) Extreme altitudes during diurnal flights in a nocturnal songbird migrant. Science 372:646–648. https://doi.org/10.1126/science.abe7291
Sjöberg S, Pedersen L, Malmiga G, Alerstam T, Hansson B, Hasselquist D, Thorup K, Tøttrup AP, Andersson A, Bäckman J (2018) Barometer logging reveals new dimensions of individual songbird migration. J Avian Biol 49:e01821. https://doi.org/10.1111/jav.01821
Stresemann E (1943) Überblick über die Vögel Kretas und den Vogelzug in der Aegäis. J Ornithol 91:448–514. https://doi.org/10.1007/BF02013136
Sullivan BL, Wood CL, Iliff MJ, Bonney RE, Fink D, Kelling S (2009) eBird: a citizen-based bird observation network in the biological sciences. Biol Conserv 142:2282–2292. https://doi.org/10.1016/j.biocon.2009.05.006
Tøttrup AP, Klaasen RHG, Strandberg R, Thorup K, Kristensen MW, Jørgensen PS, Fox J, Afanasyev V, Rahbeck C, Alerstam T (2012) The annual cycle of a trans-equatorial Eurasian-African passerine migrant: different spatio–temporal strategies for autumn and spring migration. Proc R Soc 279:1008–1016. https://doi.org/10.1098/rspb.2011.1323
Tøttrup AP, Pedersen L, Onrubia A, Klaasen RHG, Thorup K (2017) Migration of red-backed shrikes from the Iberian Peninsula: optimal or sub-optimal detour? J Avian Biol 48:149–154. https://doi.org/10.1111/jav.01352
Wong J, Turon F, Fernández-Tizón M, Hahn S (2022) First insights into migration routes and nonbreeding sites used by Red-rumped Swallows (Cecropis daurica rufula) breeding in the Iberian Peninsula. J Ornithol 163:1045–1049. https://doi.org/10.1007/s10336-022-02011-1
Wotherspoon SJ, Sumner MD, Lisovski S (2013) R Package SGAT: Solar/Satellite Geolocation for Animal Tracking. GitHub Repository. Retrieved from https://github.com/SWotherspoon/SGAT
Acknowledgements
We thank P. Tuček, M. Krist, T, Koutný, M. Briedis and J. Adamík for help with fieldwork, E. Bächler for providing the set of uTags in 2019 and the three reviewers for helpful comments. We are grateful to the European Union for Bird Ringing (EURING) which made the recovery data available through the EURING Data Bank and to the many ringers and ringing scheme staff who have gathered and prepared the data. This is publication #3 from the Tracking Least-Known Species Project at the Swiss Ornithological Institute.
Funding
Open access publishing supported by the National Technical Library in Prague. Funding was provided by grants from VEGA grant No. 2/0065/20 and Czech Science Foundation 20-00648S.
Author information
Authors and Affiliations
Contributions
PA and AK designed the study, collected data, and wrote the manuscript. PA requested and processed the EURING data. JBW analysed the geolocator and multi-sensor logger data, and wrote parts of methods with an input from SH. Maps and actogram: JBW; flight duration and altitude visualization: PA. All authors reviewed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
None.
Ethics statement
The permissions to handle the birds were issued by the Slovak Ministry of Environment, and the Slovak ringing scheme to AK.
Additional information
Communicated by F. Bairlein.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Adamík, P., Wong, J.B., Hahn, S. et al. Non-breeding sites, loop migration and flight activity patterns over the annual cycle in the Lesser Grey Shrike Lanius minor from a north-western edge of its range. J Ornithol 165, 247–256 (2024). https://doi.org/10.1007/s10336-023-02102-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-023-02102-7