Abstract
The most important cause of the declining numbers of wading birds is a combination of habitat loss and degradation, as well as low productivity due to predation. Knowledge of the habitat requirements of this endangered group of species is critical for maintaining and restoring suitable breeding habitats. The aim of this paper is to characterize those habitat components of the Eurasian Curlew (Numenius arquata) which determine its choice of nesting site. This study was undertaken in three sites in Poland, the most important breeding areas of this species in this country. Our results showed that macrohabitat factors affect curlew nest-site selection in different ways. The most important ones were distance to a forest edge, the percentage cover of managed farmland in the vicinity and the number of farmland plots close to the nest. Other habitat components, such as distance to scattered trees, distance to roads and soil category, were of lesser importance and could be treated as localized. This study is a contribution to the knowledge of the Eurasian Curlew’s nest-site selection in farmland grasslands, which may have implications for its conservation.
Zusammenfassung
Wahl des Neststandorts beim Brachvogel (Numenius arquata) mitten in seinem europäischen Verbreitungsgebiet: Anpassung an örtliche Makrohabitat-Faktoren
Die wichtigste Ursache für den Rückgang der Watvögel ist eine Kombination aus Verlust oder Verschlechterung ihres Lebensraums und einer geringeren Produktivität durch Raubtiere. Ihre Anforderungen an den Lebensraum zu kennen ist entscheidend für die Erhaltung und Wiederherstellung geeigneter Bruthabitate für diese gefährdete Vogelgruppe. Ziel dieser Arbeit ist es, diejenigen Elemente des Lebensraums zu ermitteln, die für den Brachvogel (Numenius arquata) für die Wahl seines Nistplatzes ausschlaggebend sind. Die Untersuchung wurde in Polen an drei Standorten, den wichtigsten Brutgebieten dieser Art in diesem Land, durchgeführt. Unsere Ergebnisse zeigen, dass Makrohabitat-Faktoren die Standortwahl des Brachvogels auf unterschiedliche Weise beeinflussen. Die wichtigsten Faktoren waren die Entfernung zum nächsten Waldrand, der Anteil der bewirtschafteten landwirtschaftlichen Flächen in der Umgebung und die Anzahl von Feldern in der Nähe des Nests. Andere Faktoren wie die Entfernung zu einzeln stehenden Bäumen oder Straßen sowie die Bodenbeschaffenheit waren von geringerer und wohl nur örtlich begrenzter Bedeutung. Diese Studie trägt zu unserem Wissen darüber bei, wie der Brachvogel seinen Nistplatz auf landwirtschaftlich genutztem Weideland auswählt, was möglicherweise Konsequenzen für seinen Schutz haben kann.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The intensification of agriculture has led to the drainage and conversion to intensively managed, “improved” grasslands of a large proportion of wet, natural and semi-natural, extensively managed grasslands, which are key breeding habitats for waders (Silva-Monteiro et al. 2021). A denser sward and the rapid drying out of topsoil reduce the abundance of invertebrate prey dwelling there (Smart et al. 2006; Kleijn et al. 2010). Frequent mowing simplifies the sward structure, reduces the numbers of suitable nesting sites (Verhulst et al. 2011; Wilson et al. 2014) and heightens the risk of nest destruction and chick mortality (Kruk et al. 1997; Schekkerman et al. 2009). Land abandonment can also cause wader habitats to deteriorate and be lost. Fallow grassland transforms into tall herb and rush communities, subsequently overgrown with bushes and trees, thereby becoming unsuitable habitat for waders (Ławicki and Wylegała 2011; Ławicki et al. 2011; Leito et al. 2014). Another factor causing habitat loss is afforestation of low-value farmland and as mitigation of climate change. All these changes also lead to fragmentation of farmland and give rise to edge effects (Douglas et al. 2014; Wilson et al. 2014; Franks et al. 2017; Kaasiku et al. 2022; Pálsdóttir et al. 2022). Existing and newly growing trees in open agricultural landscapes have similar effects (Tamis and Heemskerk 2020).
Predation of wader nests and chicks reduces the productivity of shorebirds, precluding the maintenance of stable populations (Macdonald and Bolton 2008; Roodbergen et al. 2012). The principal predators are meso-mammals, such as Red Fox (Vulpes vulpes; Macdonald and Bolton 2008; Zielonka et al. 2020), and also birds, although the latter are more likely to prey on chicks than on eggs (Teunissen et al. 2008). In some areas, nests are plundered by non-native and invasive mammals like American Mink (Neovison vison) and Raccoon Dog (Nyctereutes procyonoides; Grant et al. 1999; Niemczynowicz et al. 2017). In response to these threats, preventive measures have been taken in important wader breeding areas, such as improving habitat condition (Eglington et al. 2008), lethal predator control (Fletcher et al. 2010), protection of nests and chicks by means of fencing (Malpas et al. 2013; Verhoeven et al. 2022) and headstarting (e.g. Laidlaw et al. 2021).
Habitat management, one of the main objectives of conservation projects, should be undertaken when sufficient knowledge of the relevant habitat selection has been gained (Korniluk et al. 2021). A variety of habitat factors may influence nest-site selection by grassland waders. At the fine scale, the height of the vegetation in which nests are concealed seems to play the most important role (Smart et al. 2006; Laidlaw et al. 2020; Kämpfer and Fartmann 2022). Nest-site selection by grassland waders may also be influenced by the landscape structure and macro-scale habitat features, e.g. patches of woodland (Bertholdt et al. 2017), man-made structures such as stone walls on pastures (Wallander et al. 2006), telephone poles (Johansson 2001) and ditches (Valkama et al. 1998). Moreover, the choice of nesting site may be determined by the availability of suitable feeding grounds in its vicinity (Berg 1992a).
For one bird species associated with grasslands, the Eurasian Curlew, subspecies Numenius arquata (hereafter referred to as the curlew), knowledge of its nesting site preferences appears to be important for conservation efforts aimed at maintaining stable populations. This charismatic bird breeds in Europe from the Atlantic in the west, Scandinavia in the north as far as the Ural Mountains in the east (Keller et al. 2020). While still common and abundant in some parts of its range (Brown 2015; Keller et al. 2020), it has been declining markedly in recent decades and has therefore been classified as globally near threatened (Birdlife International 2023). In Europe, curlews inhabit a variety of open lowlands (Zielonka et al. 2020; Leprince et al. 2022), uplands (Douglas et al. 2014) and coastal areas (Leito et al. 2014; Kämpfer and Fartmann 2022), whereas in the centre of its continental range, it nests mainly on grasslands and arable fields (Brown Brown 2015; Silva-Monteiro et al. 2021). The Polish curlew population breeds principally in extensively managed, wet grasslands in lowland river valleys, including floodplains (Ławicki and Wylegała 2011; Ławicki et al. 2011; Lewtak et al. 2016; Wilk et al. 2020).
The aim of this paper is thus to characterize the habitat components which mediate the curlews’ nest-site selection in human-modified river valleys in Poland. We expected that certain specific macro-scale habitat components would affect nest-site selection by curlews in managed grasslands, but that some might be universal, while others would be only of local importance. We hypothesized that these birds would avoid nesting close to environmental features that could increase the risk of nest predation: forest edges, scattered trees, ditches and roads (see Berg 1992a; Valkama et al. 1998). On the other hand, we expected curlews to prefer nesting near areas with a high cover of farmland, in patchy farmland and on the most humid soil type. These results may improve our understanding of nest-site selection by the curlew breeding in the centre of its European breeding range, and extend our knowledge of the habitat preferences of this endangered species.
Materials and methods
Study area
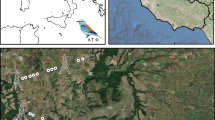
This work was undertaken in three open-landscape lowland river valleys (Fig. 1, see also electronic supplementary materials: Fig. S1–S3), which are among the most important curlew breeding areas in Poland (Chodkiewicz et al. 2022). The study sites partially or fully overlapped (39–100%) Natura 2000 sites for which the curlew was the subject of protection. In the years preceding this survey, nationwide monitoring of the breeding populations of four declining wader species (including the curlew) was carried out in key lowland grasslands and inland wetlands (Żmihorski et al. 2018). Therefore, precise knowledge regarding the occurrence of this rare species was used in the delimitation of the study sites.
The first site was located in the valley of the small, naturally flowing and meandering River Brzozówka (36.6 km2; 53°34′N, 23°4′E; 108–126 m a.s.l.), a left tributary of the River Biebrza (hereafter B). The valley was dominated by peat soils and the grassland was well drained. Land-use patches were small, invariable in shape, mainly long and narrow. Access to them was provided by a relatively dense network of unpaved roads (Table 1). The second site (55.4 km2; 53°14′N 21°13′E; 102–119 m a.s.l.) lay in the valleys of the Rivers Omulew and Płodownica, situated among forested inland dunes (hereafter O). Site O consisted of three areas separated by valley sections where curlews did not occur. Agricultural land was moderately drained. Mineral-muck and peat soils predominated, and alluvial soils were present locally. The road density was lower than at site B (Table 1). The third site (33.7 km2; 52°53′N, 16°20′E; 33–44 m a.s.l.) was situated in Western Poland, on the floodplain of the Noteć, the largest of the rivers described here (hereafter N). The soils were represented in the following order by alluvial soil, peat and mineral-muck, their areas decreasing in this order. The density of roads and drainage ditches was the lowest of all the sites (Table 1).
The main habitat type in the study areas was open grassland, managed mainly by mowing (1–3 times per year) and to a lesser extent by cattle grazing. There were few arable fields in all the locations. The vegetation was subject to natural processes and human activities, notably agricultural management. The grasslands were mainly semi-natural, but some were ploughed and undersown with varieties of grass for intensive hay production. Although grassland communities were dominant, there were also patches of sedges, rushes and herbs. Plant succession had taken over some abandoned meadows, which were now overgrown by tall herbs, sedges and shrubs. Singly growing trees were most common along roads, rivers, ditches and the boundaries of land plots. The few water bodies consisted mainly of small ponds, and in site N there were also oxbows. The landscapes surrounding the study sites were composed predominantly of arable land and forests.
Data collection
This study was carried out during six breeding seasons from 2017 to 2022. Each year, breeding pairs were located before the search for nests began. Observers walked or travelled by car, stopping at points with the best visibility to survey all potential curlew breeding habitats using binoculars and spotting scopes. Nests were found mostly between mid-April and the end of May by searching for birds sitting on or returning to their nests. In all the sites, observers looked for breeding pairs and searched for nests at least twice a week. The positions of the located nests were recorded with a handheld GPS device. A total of 158 first-clutch nests were found in the three study areas (nB = 62; nO = 65; nN = 31). Replacement nests (nB = 17; nO = 23; nN = 25) were not included in this analysis.
Data processing
Pseudo-absence points (n = 790) were randomly selected for each study site and each study year independently in a 5:1 ratio to the nests found (see electronic supplementary materials: Figs. S1–S3). Despite the fact that the use of a single draw is a frequently method used for analysis of habitat selection (Bertholdt et al. 2017; Kaasiku et al. 2019; Holmes et al. 2020; Kämpfer and Fartmann 2022), the ratio of random points to nest locations was increased. This approach was aimed at obtaining greater background stability from the random sample. Random points were generated only on managed grasslands, as curlews did not nest in other types of habitat such as arable fields. Seven habitat- and predation risk-related macro-scale variables, potentially explaining the curlews’ nest-site selection, were applied (Table 2). The ecological significance of these variables was based on the methodology applied in other studies of curlews and other grassland-breeding waders (see Valkama et al. 1998; Żmihorski et al. 2018). Soil type (Soil) was defined using soil and agriculture maps (scaled 1:5,000 or 1:25,000) from the “web map services” (WMS) of the Polish provincial geoportals. The most up-to-date aerial photographs (0.25 m pixel) from the “Central Office of Geodesy and Cartography” (GUGIK Poland) and satellite images (Google Earth) were used to measure the distance to the nearest tree (DistTree) for the year of the study. The number of farmland plots within 500 m of a nest’s location and random points (FarmPlot) was calculated using the vector database of the “Land and Building Register”, previously available in the “Land-parcel Identification System of the Agency for Restructuring and Modernization of Agriculture” (see Krupiński et al. 2021). Distances of the nearest landscape elements, i.e. roads (DistRoad), ditches and rivers (DistDitch), forest edges (DistForest) and managed farmland cover (FarmCov) within 500 m of a nest’s location and random points, were calculated based on the vector database of the “Topographic Objects Database” (BDOT10k) of GUGIK Poland. The original spatial dataset (BDOT10k) did not include certain small landscape features such as reed beds, shrubs and permanently abandoned farmland plots. Therefore, the vector land cover was compared with the aerial photographs, and these features were hand-removed to correctly calculate managed farmland cover.
The locations of curlews during the incubating period were distributed mostly up to a few hundred metres from the nest, but at night they could have been present more than 1 km away from the nest (authors’ unpubl. GPS-logger data, Ewing et al. 2017). Therefore, 500 m was assumed to be the distance at which curlews mainly use their feeding resources during the nesting period, so managed farmland cover and the number of farmland plots were calculated for this distance. QGIS 3.28 (QGIS Development Team 2023) with optional plugins was used for all the geospatial analyses.
Statistical analyses
In the first step, the multicollinearity of a quantitative variables was tested for the entire dataset and independently for a subset of each study site. No high values of the variance inflation factor were identified (VIF < 1.7). Generalized linear mixed models (GLMM) with a binomial structure and logit link function were employed to analyse nest-site selection by the curlews. Nest location (1) and random point (0) were treated as dependent variables, seven macro-scale continuous parameters and one categorical (Soil) were the main fixed predictors (Table 2), while year was introduced as a random factor. Three models were developed with the same variable structure, one for each site, to examine which macro-scale habitat factors affected nest-site selection. Models were constructed in the glmmTMB package (Brooks et al. 2017) and the final structure was thus as follows: Nest ~ Soil+DistRoad+DistDitch+DistForest+DistTree+AgriCov+AgriPatch+(1|Year). For reference, one summary model was also created with site as a random factor and an interaction between site and year: Nest ~ Soil+DistRoad+DistDitch+DistForest+DistTree+AgriCov+AgriPatch+(1|Year*Site). The results of this model confirmed the influence of general factors, but without considering the importance of local factors (see electronic supplementary materials: Tables S2–S3). Therefore, three separate models for each site were considered in the analyses and discussion of the results. An information-theoretic approach (AIC) and Akaike weights (ACIwt) were used in the model selection (Burnham and Anderson 2002). Sets of all possible combinations of the global models were computed using the MuMln package (Bartoń 2023). Using model averaging based on an information criterion, we calculated sets of models that fell within the 95% confidence limits. Only models with ΔAIC ≤ 2 were considered equally supported (Burnham and Anderson 2002). R-square for the final model of each site was calculated as R-square Nakagawa, as implemented in the MuMIn package (Nakagawa et al. 2017). All statistical analyses were run in R Studio (R Core Team 2023).
Results
The best models of the study sites had different numbers of variables. The model of site B contained five variables—distance to a road, distance to a forest edge, distance to a tree, managed farmland cover and soil type (Table 3)—and explained 43% of the variation in the probability of nest occurrence, as estimated by pseudo-r2 statistics for GLMM. The best model of site O contained four variables—distance to a forest edge, distance to a tree, managed farmland cover and soil type (Table 3)—and explained 28% of the variation. There were three variables—distance to a forest edge, managed farmland cover and number of farmland plots—in the top model of site N (Table 3). This model explained 32% of the variation in the probability of nest occurrence, as estimated by pseudo-r2.
The factors present in all the best models were distance to a forest edge and managed farmland cover (Table 4). Only distance to a forest edge had a significant effect on the curlews’ nest-site selection in all three study sites (Table 4). In all three models, the probability of a nest being present increased with distance to a forest edge (Fig. 2). The presence of a nest was positively related to the managed farmland cover in the vicinity of the nest (Fig. 2), but this variable was close to significant in the model of site N (Table 4). The top models of sites B and O displayed a higher probability of nests occurring in locations some distance away from scattered trees (Table 4, Fig. 3). Most nests in sites B and O were located in vegetation growing on peat soils (Fig. 4). This variable was significant for site B and close to significant for site O (Table 4). In site N, curlews built their nests more frequently on alluvial soils (Fig. 4), but this variable was not included in the final model (Table 3, Fig. 4). The best model of site B showed that there was a higher probability of a nest occurring in areas close to roads. In site N, the presence of a nest was positively related to the number of farmland plots within a radius of 500 m (Table 4, Fig. 3). Distance to a ditch was the only variable that did not fit into any top model (Table 4).
Discussion
As expected, curlews preferred placing their nests farther away from forest edges in all the study sites. The importance of this factor for nest-site selection is confirmed by studies in the northern European range of this species (Berg 1992a; Valkama 1998). In addition, the proximity of forested areas and afforestation has a negative impact on the occurrence, densities and population changes of curlews (Żmihorski et al. 2016; Franks et al. 2017). Forest avoidance is also common in other species of waders (Stroud 1990; Wilson et al. 2014; Żmihorski et al. 2016; Bertholdt et al. 2017; Kaasiku et al. 2019; Pálsdóttir et al. 2022). This is explained by the behavioural response to the risk of nest predation (Wilson et al. 2014). Meso-mammals such as Red Foxes, which are a threat to ground-nesting birds, are more numerous in and near forests than in open landscapes (Kaasiku et al. 2022). Recent research using temperature loggers clearly demonstrates that mammals are the main cause of curlew nest failures in the UK (Zielonka et al. 2020; Ewing et al. 2023). Moreover, crows, which likewise plunder birds’ nests (Macdonald and Bolton 2008; Teunissen et al. 2008), are very active along forest margins (Smedshaug et al. 2002). Curlews may also avoid breeding close to forests because trees obstruct the view from the nest, thus reducing the earlier detection of predators (Bertholdt et al. 2017; Kaasiku et al. 2019).
Nest predation by corvids was found in all study sites. Single trees and groups of trees are perches and nesting sites for corvids and other avian predators in farmland (Ottvall et al. 2005). In two sites (B and O), the probability of a curlew nest being present increased with distance from the nearest tree. A similar result was obtained by Johansson (2001), who demonstrated the avoidance of perches (trees and telephone poles) by Black-tailed Godwits (Limosa limosa). On the other hand, the cryptic colouration of their plumage helps incubating curlews remain undetected by avian predators scanning their surroundings from perches. In addition, they exhibit aggressive anti-predator behaviour and are well capable of defending their clutches against corvids (Berg 1992a; Zielonka et al. 2020). However, these strategies may fail if the nest is close to a tree and frequently visited by corvids, as was the case in the study sites. Studies of Northern Lapwings (Vanellus vanellus; Berg et al. 1992), Black-tailed Godwits (Johansson 2001) and Long-billed Curlews (Numenius americanus; Coates et al. 2019) showed that nesting farther from potential corvid perches (including trees) offered a higher probability of nesting success. In one study site (N), however, the curlews did not avoid the proximity of scattered trees. This may have been due to the markedly lower density of corvids there than at other nesting sites (Kuczyński and Chylarecki 2012) and therefore the lower frequency of corvids surveying their surroundings from trees.
Ditches and rivers are also a predator-related habitat component because, being linear, they provide corridors for medium-sized mammalian predators such as Red Fox (Bischof et al. 2019), American Mink and Polecat (Mustela putorius; Harrington et al. 2008). In river valleys in Poland, American Mink are common and may have a negative impact on the breeding success of waders (Niemczynowicz et al. 2017). Our results do not indicate any effect of ditches on curlew nest-site selection, which is in line with studies of curlews in Sweden (Berg 1992a) and Northern Lapwings in the UK (Bertholdt 2017). In Finland, by contrast, curlews nested farther away from ditches (Valkama et al. 1998), the authors explaining that this might be an adaptation to reduce the risk of nest predation. It is possible that in our study plots the risk of nest predation was not related to the proximity of ditches, and therefore, the curlews had no need to avoid them, although we did not investigate or demonstrate this. Other studies have shown that ditches may be attractive foraging sites for waders (Kaasiku et al. 2019). Therefore, the no-ditch effect may also be a trade-off between avoiding the predation risk and benefitting from the proximity to an attractive foraging area.
Another threat to curlew broods that we considered was that posed by roads, which mammalian predators use as corridors (Bischof et al. 2019). It has been experimentally shown that nest predation is highest along unpaved roads (Pescador and Peris 2007), and this was the prevailing type of road in the study sites. Roads can also be a source of human disturbance, as a lot of agricultural activities on grasslands (harrowing, seeding, rolling and fertilization) take place in the second half of April, when waders are laying and then incubating their eggs. A previous study has shown that curlews avoid nesting close to areas that are frequented by humans (Kämpfer and Fartmann 2022). Avoidance of nesting near roads has been reported in a study of a related species, the Long-billed Curlew (Coates et al. 2019). In our study, however, we found no effects of road proximity at two sites (O and N), but the results from the third site (B) were surprising, as the birds located their nests closer to roads. This phenomenon is interesting and requires more detailed study. However, based on the field observation, we speculated that it is associated with grassland management rather than with nesting preferences. The grassland closer to roads may have been managed in a way that matched the preferences of curlews, i.e. maintaining a moderate moisture content, keeping a small proportion of plots unmown or cut once in the previous year and ensuring the dominance of short to medium-height vegetation (Żmihorski et al. 2016). A study in lowland grasslands in the UK indicated that curlews preferred to place their nests in grassland with lower vegetation. This was explained by the better availability of prey near the nest and the better visibility and detection of predators from the nest (Zielonka et al. 2020).
The curlews showed a preference for a higher proportion of managed farmland cover in the vicinity of their nests in all the sites. The wide availability of potential foraging grounds during the incubation period is an important factor, as adults forage relatively close to the nest (Berg 1992b). The vegetation on managed farmland, mainly grassland in our study sites, was shorter than the vegetation of abandoned land, reed beds and scrub. Consequently, the field of view of the surrounding area from the nest is greater in sites with a higher proportion of managed agricultural land and may facilitate earlier predator detection. The selection of sites with a wide availability of potential foraging grounds may also be important for the chicks, which are particularly vulnerable to predation during the rearing period and usually move up to 300 m from the nest for the first 20 days after hatching (Ewing et al. 2017).
At one site (N), the curlews preferred more patchy grassland, selecting nest sites in locations surrounded by a greater number of farmland plots. However, in the other two sites (B and O), where no such preference was noted, the patchy fragmentation of grasslands was high and relatively even in both. As a result, curlews had a limited choice and always nested in patchy grassland. The farmland plots in the study sites are managed by a great many different landowners. Moreover, the plots are variably managed and differ in the number of cuts, mowing dates, fertilization and the grazing of animals, resulting in heterogeneous grasslands. Pearce-Higgins and Grant (2006) reported similar results, showing that curlews were the most abundant where the vegetation structure was relatively heterogeneous. The heterogeneity of vegetation resulting from different management practices in a mosaic enhances invertebrate richness (Bonari et al. 2017) and, as reported in previous studies, resource-abundant feeding habitats are crucial for breeding curlews (Berg 1993; Valkama 1998; Leprince et al. 2022).
In two sites (B and O), curlews preferred nesting in vegetation growing on peat soils, but in the third plot (N), dominated by alluvial soils, no selectivity of nesting sites was found. Curlews were more likely to nest in grasslands on peat soils, because the foraging conditions there were probably better than on the mineral-muck. The peat soils in the study sites are still more moist than other available soil types, despite having been drained decades ago. Soil moisture is related to soil penetrability and the availability of earthworms in the top soil layer, which is crucial for waders (Smart et al. 2006; Leito et al. 2014). In drier soils, earthworms may remain at greater depths, making them inaccessible to birds (Rhymer et al. 2010).
Our research has shown that nest-site selection in the curlew is determined not by one but by several macro-scale habitat factors. In all three study sites, the curlews preferred locations farther from forest edges, with a high cover of managed farmland (permanent grasslands) near the nests and high numbers of farmland plots; these factors are considered universal. Other factors had a local influence, however: distance to the nearest tree, distance to a road and soil type. The results of this research can inform the drawing up of specific recommendations for implementation in curlew conservation programmes. Management of the human-modified grassland agricultural landscape should take into account the habitat preferences of curlews in order to provide suitable nesting sites for this endangered species. This is important both for the preservation of existing breeding habitats and for their restoration.
Data availability
The dataset analysed during the current study is available from the corresponding author upon reasonable request.
References
Bartoń K (2023) MuMIn: multi-model inference. R package version 1.47.5. https://cran.r-project.org/web/packages/MuMIn/. Accessed 23 Mar 2023
Berg Å (1992a) Factors affecting nest-site choice and reproductive success of Curlews Numenius arquata on farmland. Ibis 134:44–51. https://doi.org/10.1111/j.1474-919X.1992.tb07228.x
Berg Å (1992b) Habitat selection by breeding Curlews Numenius arquata on mosaic farmland. Ibis 134:355–360. https://doi.org/10.1111/j.1474-919X.1992.tb07228.x
Berg Å (1993) Food resources and foraging success of Curlews Numenius arquata in different farmland habitats. Ornis Fen 70:22–31
Berg Å, Lindberg T, Källebrink K (1992) Hatching success of lapwings on farmland: differences between habitats and colonies of different sizes. J Anim Ecol 61:469–476. https://doi.org/10.2307/5337
Bertholdt NP, Gill JA, Laidlaw RA, Smart J (2017) Landscape effects on nest site selection and nest success of Northern Lapwing Vanellus vanellus in lowland wet grasslands. Bird Study 64:30–36. https://doi.org/10.1080/00063657.2016.1262816
Birdlife International (2023) Species factsheet: Numenius arquata. http://datazone.birdlife.org/species/factsheet/eurasian-curlew-numenius-arquata. Accessed 20 Sep 2023
Bischof R, Gjevestad JGO, Ordiz A, Eldegard K, Milleret C (2019) High frequency GPS bursts and path-level analysis reveal linear feature tracking by Red Foxes. Sci Rep 9:8849. https://doi.org/10.1038/s41598-019-45150-x
Bonari G, Fajmon K, Malenovský I, Zelený D, Holuša J, Jongepierová I, Kočárek P, Konvička O, Uřičář J, Chytrý M (2017) Management of semi-natural grasslands benefiting both plant and insect diversity: the importance of heterogeneity and tradition. Agric Ecosyst Environ 246:243–252. https://doi.org/10.1016/j.agee.2017.06.010
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Maechler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modelling. R J 9:378–400
Brown DJ (2015) International single species action plan for the conservation of the Eurasian Curlew Numenius arquata arquata, N. a. orientalis and N. a. suschkini. AEWA Technical Series No. 58, Bonn
Burnham KP, Anderson DR (2002) Model selection and multimodel inference, 2nd edn. Springer, New York
Chodkiewicz T, Beuch S, Ławicki Ł, Wylegała P et al (2022) Rare birds panel in Poland—report of 2020 for breeding species. Ornis Pol 63:347–379
Coates SE, Wright BW, Carlisle JD (2019) Long-billed Curlew nest site selection and success in the Intermountain West. Jour Wild Mgmt 83:1197–1213. https://doi.org/10.1002/jwmg.21661
Douglas DJT, Bellamy P, Stephen LS, Pearce-Higgins JW, Wilson JD, Grant MC (2014) Upland land use predicts population decline in a globally near-threatened wader. J Appl Ecol 51:194–203. https://doi.org/10.1111/1365-2664.12167
Eglington SM, Gill JA, Bolton M, Smart MA, Sutherland WJ, Watkinson AR (2008) Restoration of wet features for breeding waders on lowland grassland. J Appl Ecol 45:305–314. https://doi.org/10.1111/j.1365-2664.2007.01405.x
Ewing SR, Scragg ES, Butcher N, Douglas DJT (2017) GPS tracking reveals temporal patterns in breeding season habitat use and activity of a globally near threatened wader, the Eurasian Curlew. Wader Study 124:206–214
Ewing H, Franks S, Smart J, Burton N, Gill JA (2023) Nest survival of threatened Eurasian Curlew (Numenius arquata) breeding at low densities across a human-modified landscape. Ibis 165:753–766. https://doi.org/10.1111/ibi.13180
Fletcher K, Aebischer NJ, Baines D, Foster R, Hoodless AN (2010) Changes in breeding success and abundance of ground-nesting moorland birds in relation to the experimental deployment of legal predator control. J Appl Ecol 47:263–272. https://doi.org/10.1111/j.1365-2664.2010.01793.x
Franks SE, Douglas DJT, Gillings S, Pearce-Higgins JW (2017) Environmental correlates of breeding abundance and population change of Eurasian Curlew Numenius arquata in Britain. Bird Study 64:393–409. https://doi.org/10.1080/00063657.2017.1359233
Grant MC, Orsman C, Easton J, Lodge C, Smith M, Thompson G, Rodwell S, Moore N (1999) Breeding success and causes of breeding failure of Curlew Numenius arquata in Northern Ireland. J Appl Ecol 36:59–74. https://doi.org/10.1046/j.1365-2664.1999.00379.x
Harrington LA, Macdonald DW (2008) Spatial and temporal relationships between invasive American Mink and native European Polecats in the Southern United Kingdom. J Mammal 89:991–1000. https://doi.org/10.1644/07-MAMM-A-292.1
Holmes GI, Koloski L, Nol E (2020) Nest-site selection of a subarctic-breeding shorebird: evidence for tree avoidance without fitness consequences. Can J Zool 98:573–580. https://doi.org/10.1139/cjz-2019-0264
Johansson T (2001) Habitat selection, nest predation and conservation biology in a Black-tailed Godwit (Limosa limosa) population. Dissertation, Uppsala University
Kaasiku T, Rannap R, Kaart T (2019) Managing coastal grasslands for an endangered wader species can give positive results only when expanding the area of open landscape. J Nat Conserv 48:12–19. https://doi.org/10.1016/j.jnc.2018.12.004
Kaasiku T, Rannap R, Männil P (2022) Predation-mediated edge effects reduce survival of wader nests at a wet grassland-forest edge. Anim Conserv 25:692–703. https://doi.org/10.1111/acv.12774
Kämpfer S, Fartmann T (2022) Natural coastal dunes on Wadden Sea islands as a refuge for an endangered wader species. J Coast Conserv 26:53. https://doi.org/10.1007/s11852-022-00897-w
Keller V, Herrando S, Vorisek P, Franch M (2020) European breeding bird atlas 2: distribution, abundance and change. European Bird Census Council & Lynx Edicions, Barcelona
Kleijn D, Schekkerman H, Dimmers WJ, Van Kats RJ, Melman D, Teunissen WA (2010) Adverse effects of agricultural intensification and climate change on breeding habitat quality of Black-tailed Godwits Limosa l. limosa in The Netherlands. Ibis 152:475–486. https://doi.org/10.1111/j.1474-919X.2010.01025.x
Korniluk M, Białomyzy P, Grygoru G, Kozub Ł, Sielezniew M, Świętochowski P, Tumiel T, Wereszczuk M, Chylarecki P (2021) Habitat selection of foraging male Great Snipes on floodplain meadows: importance of proximity to the lek, vegetation cover and bare ground. Ibis 163:486–506. https://doi.org/10.1111/ibi.12898
Kruk M, Noordervliet MAW, ter Keurs WJ (1997) Survival of Black-tailed Godwit chicks Limosa limosa in intensively exploited grassland areas in The Netherlands. Biol Conserv 8:127–133. https://doi.org/10.1016/S0006-3207(96)00131-0
Krupiński D, Kotowska D, Recio MR, Żmihorski M, Obłoza P, Mirski P (2021) Ranging behaviour and habitat use in Montagu’s Harrier Circus pygargus in extensive farmland of Eastern Poland. J Ornithol 162:325–337. https://doi.org/10.1007/s10336-020-01837-x
Kuczyński L, Chylarecki P (2012) Atlas of common breeding birds in Poland: distribution, habitat preferences and population trends. GIOŚ, Warsaw
Laidlaw RA, Gunnarsson TG, Méndez V, Carneiro C, Þórisson B, Wentworth A, Gill JA, Alves JA (2020) Vegetation structure influences predation rates of early nests in subarctic breeding waders. Ibis 162:1225–1236. https://doi.org/10.1111/ibi.12827
Laidlaw R, Smart J, Ewing H, Franks S et al (2021) Predator management for breeding waders: a review of current evidence and priority knowledge gaps. Wader Study 128:44–55
Ławicki Ł, Wylegała P (2011) Recent data on the declining breeding population of Eurasian Curlew Numenius arquata in western Poland. Wader Study Group Bull 118:14–17
Ławicki Ł, Wylegała P, Batycki A, Kajzer Z et al (2011) Long-term decline of grassland waders in western Poland. Vogelwelt 132:101–108
Leito A, Sepp K, Pehlak H, Kuresoo A et al (2014) Coastal grassland wader abundance in relation to breeding habitat characteristics in Matsalu Bay, Estonia. Ornis Fen 91:149–165
Leprince R, Debenest E, Lartigau C, Turpaud-Fizzala V, Poirel C, Lachaussée N, Donnez M, Bocher P (2022) Influence of habitat quality and diversity on two populations of Eurasian Curlew (Numenius arquata) with contrasting dynamics in Western France. Ornis Fenn 99:132–149. https://doi.org/10.51812/of.124713
Lewtak J, Trzciński K, Krupiński D (2016) The national action plan for conservation of the Eurasian Curlew. Wildlife Society „Stork”, Warsaw (abstract in English)
MacDonald MA, Bolton M (2008) Predation on wader nests in Europe. Ibis 150:54–73. https://doi.org/10.1111/j.1474-919X.2008.00869.x
Malpas LR, Kennerley RJ, Hirons GJ, Sheldon RD, Ausden M, Gilbert JC, Smart J (2013) The use of predator-exclusion fencing as a management tool improves the breeding success of waders on lowland wet grassland. J Nat Conserv 21:37–47. https://doi.org/10.1016/j.jnc.2012.09.002
Nakagawa S, Johnson PCD, Schielzeth H (2017) The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J R Soc Interface 14:20170213. https://doi.org/10.1098/rsif.2017.0213
Niemczynowicz A, Świętochowski P, Brzeziński M, Zalewski A (2017) Non-native predator control increases the nesting success of birds: American Mink preying on wader nests. Biol Conserv 212:86–95. https://doi.org/10.1016/j.biocon.2017.05.032
Ottvall R, Larsson K, Smith HG (2005) Nesting success in Redshank Tringa totanus breeding on coastal meadows and the importance of habitat features used as perches by avian predators. Bird Study 52:289–296. https://doi.org/10.1080/00063650509461402
Pálsdóttir AE, Gill JA, Alves JA, Pálsson S, Méndez V, Ewing H, Gunnarsson TG (2022) Subarctic afforestation: effects of forest plantations on ground-nesting birds in lowland Iceland. J Appl Ecol 59:2456–2467. https://doi.org/10.1111/1365-2664.14238
Pearce-Higgins JW, Grant MC (2006) Relationships between bird abundance and the composition and structure of moorland vegetation. Bird Study 53:112–125. https://doi.org/10.1080/00063650609461424
Pescador M, Peris S (2007) Influence of roads on bird nest predation: an experimental study in the Iberian Peninsula. Land Urb Plan 82:66–71. https://doi.org/10.1016/j.landurbplan.2007.01.017
QGIS Development Team (2023) QGIS geographic information system. Open Source Geospatial Foundation Project. http://qgis.osgeo.org. Accessed 14 Sep 2023
R Core Team (2023) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna
Rhymer CM, Robinson RA, Smart J, Whittingham MJ (2010) Can ecosystem services be integrated with conservation? A case study of breeding waders on grassland. Ibis 152:698–712. https://doi.org/10.1111/j.1474-919X.2010.01049.x
Roodbergen M, van der Werf B, Hötker H (2012) Revealing the contributions of reproduction and survival to the Europe-wide decline in meadow birds: review and meta-analysis. J Ornithol 153:53–74. https://doi.org/10.1007/s10336-011-0733-y
Schekkerman H, Teunissen W, Oosterveld E (2009) Mortality of Black-tailed Godwit Limosa limosa and Northern Lapwing Vanellus vanellus chicks in wet grasslands: influence of predation and agriculture. J Ornithol 150:133–145. https://doi.org/10.1007/s10336-008-0328-4
Silva-Monteiro M, Pehlak H, Fokker C, Kingma D, Kleijn D (2021) Habitats supporting wader communities in Europe and relations between agricultural land use and breeding densities: a review. Glob Ecol Conserv 28:e01657. https://doi.org/10.1016/j.gecco.2021.e01657
Smart J, Gill JA, Sutherland WJ, Watkinson AR (2006) Grassland-breeding waders: identifying key habitat requirements for management. J Appl Ecol 43:454–463. https://doi.org/10.1111/j.1365-2664.2006.01166.x
Smedshaug CA, Lund SE, Brekke A, Sonerud GA, Rafoss T (2002) The importance of the farmland-forest edge for area use of breeding Hooded Crows as revealed by radio telemetry. Ornis Fenn 79:1–13
Stroud DA, Reed TM, Harding NJ (1990) Do moorland breeding waders avoid plantation edges? Bird Study 37:177–186. https://doi.org/10.1080/00063659009477055
Tamis W, Heemskerk P (2020) A longitudinal study of the effects of trees, geese and avian predators on breeding wader meadow birds: the case of the Demmerik polder, the Netherlands. Eur J Wildl Res 66:78. https://doi.org/10.1007/s10344-020-01415-9
Teunissen W, Schekkerman H, Willems F, Majoor F (2008) Identifying predators of eggs and chicks of Lapwing Vanellus vanellus and Black-tailed Godwit Limosa limosa in the Netherlands and the importance of predation on wader reproductive output. Ibis 150:74–85. https://doi.org/10.1111/j.1474-919X.2008.00861.x
Valkama J, Robertson P, Currie D (1998) Habitat selection by breeding Curlews (Numenius arquata) on farmland: the importance of grassland. Ann Zool Fennici 35:141–148
Verhoeven MA, Jelle Loonstra AH, Pringle T, Kaspersma W, Whiffin M, McBride AD, Sjoerdsma P, Roodhart C, Burgess MD, Piersma T, Smart J (2022) Do ditch-side electric fences improve the breeding productivity of ground-nesting waders? Ecol Solut Evid 3:e12143. https://doi.org/10.1002/2688-8319.12143
Verhulst J, Kleijn D, Loonen W, Berendse F, Smit C (2011) Seasonal distribution of meadow birds in relations to in-field heterogeneity and management. Agric Ecosyst Environ 142:161–168. https://doi.org/10.1016/j.agee.2011.04.016
Wallander J, Isaksson D, Lenberg T (2006) Wader nest distribution and predation in relation to man-made structures on coastal pastures. Biol Conserv 132:343–350. https://doi.org/10.1016/j.biocon.2006.04.026
Wilk T, Chodkiewicz T, Sikora A, Chylarecki P, Kuczyński L (2020) The red list of birds in Poland. OTOP, Marki (abstract in English)
Wilson JD, Anderson R, Bailey S, Chetcuti J et al (2014) Modelling edge effects of mature forest plantations on peatland waders informs landscape-scale conservation. J Appl Ecol 51:204–213. https://doi.org/10.1111/1365-2664.12173
Zielonka NB, Hawkes RW, Jones H, Burnside RJ, Dolman PM (2020) Placement, survival and predator identity of Eurasian Curlew Numenius arquata nests on lowland grass-heath. Bird Study 66:471–483. https://doi.org/10.1080/00063657.2020.1725421
Żmihorski M, Pärt T, Gustafson T, Berg Å (2016) Effects of water level and grassland management on alpha and beta diversity of birds in restored wetlands. J Appl Ecol 53:587–595. https://doi.org/10.1111/1365-2664.12588
Żmihorski M, Krupiński D, Kotowska D, Knape J, Pärt T, Obłoza P, Berg Å (2018) Habitat characteristics associated with occupancy of declining waders in Polish wet grasslands. Agric Ecosyst Environ 251:236–243. https://doi.org/10.1016/j.agee.2017.09.033
Acknowledgements
The authors thank Grzegorz Grygoruk, Marek Maluśkiewicz, Karol Trzciński and Tomasz Tumiel for their nest searches and for sharing their valuable observations. They are also grateful to Aneta Gajko, Edyta Kapowicz, Anna Suchowolec (Polish Society for Bird Protection) and Maciej Szajda (Regional Directorate for Environmental Protection in Poznań) for obtaining funding and the organizational support of this project.
Funding
The search for curlew nests was part of a scheme for protecting these against predators and farmwork, run in accordance with The National Action Plan for the conservation of the Eurasian Curlew (Lewtak et al. 2016) and coordinated by the “Stork” Wildlife Society, the Polish Society for Bird Protection (PTOP) and the Regional Directorate for Environmental Protection in Poznań. The project was co-financed by the EU Cohesion Fund (project POIS.05.01.00–00-380/12–00 in 2017–2020) and the Provincial Funds for Environmental Protection and Water Management in Białystok, Poznań and Warsaw (in 2021–2022). This study was also supported by the University of Siedlce.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
The field work was conducted in accordance with the guidelines set out in the National Action Plan for the conservation of the Eurasian curlew. The permit for the field work around the nests was issued by the General Directorate for Environmental Protection in Poland (DZP-WG.6401.03.6.2017.dł, DZP-WG.6401.95.2021.WW.2).
Additional information
Communicated by T. Gottschalk.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Obłoza, P., Krupiński, D. & Kasprzykowski, Z. Nest-site selection of Eurasian Curlew (Numenius arquata) in the centre of its European range: adaptation to local macrohabitat factors. J Ornithol (2024). https://doi.org/10.1007/s10336-024-02203-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10336-024-02203-x