Abstract
In nutrient poor and dry forest sites common to northern Scandinavia, Scots pine (Pinus sylvestris L.) is the most common species both in managed and natural forests. However, Scots pine is sensitive to competition during establishment. Harvesting of all trees within a given area, i.e., clear-cutting, liberates regenerating seedlings from competition with mature trees. However, recently, clear-cut-free or continuous cover forestry has been the subject of substantial debate. When choosing a management method, it is important to recognize how competitive interactions direct the success of Scots pine regeneration. We studied Scots pine regeneration at three environments: beneath the canopy of mature trees, at the canopy edge in full sunlight, and distant from the canopy with no influence of mature trees. We imposed three treatments in each of these environments: root isolation (i.e., trenching), nitrogen (N) fertilization, and control plots. Root isolation enhanced seedling performance under the canopy of mature trees. Nitrogen fertilization enhanced seedling performance to a greater extent in the clear-cut than at the forest edge. However, N fertilization had no effect under the canopy. In the N-fertilized plots, we measured higher N content in the soil under the canopy than in the open environments, indicating that not all excess N was obtained by the mature trees. N-uptake might have been limited by competition for water in the N-fertilized plots. Our results suggest that belowground competition limits the success of regeneration of Scots pine. However, N fertilization presents a tool to compensate for underground competition along canopy edges.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forest regeneration is at the core of sustainable forest management as the mature trees are harvested and new seedlings establish. A better understanding of the environmental controls on regeneration is needed to predict how it can be successful under the future climate or under new forest policies. In Scandinavian boreal forests, the main tree species used in forestry are the native conifers Scots pine (Pinus sylvestris L.) and Norway spruce [Picea abies (L.) H. Karst]. Norway spruce is shade tolerant and can reach full growth potential in canopy openings ~ 0.1 ha, while Scots pine is a shade-intolerant competition sensitive pioneer species that has been suggested to require canopy openings larger than 0.4 ha to achieve maximum growth (Malcolm et al. 2001). Shade-intolerant species in general grow better than shade-tolerant species in high-light conditions, while suffering from higher mortality in low-light conditions (Kobe et al. 1995).
Aboveground growth of both Scots pine and Norway spruce has been found to increase with increasing solar radiation, and the highest growth rates have been found north of gap centers (Gray and Spies 1996; Erefur et al. 2011). However, Scots pine has a more flexible morphology that allows it to allocate growth to increased needle length under high-light conditions, which give it a higher capacity to utilize incoming sunlight (de Chantal et al. 2003). Yet, through the history of forestry in Sweden, it has repeatedly been observed that pine regeneration is suppressed within 5–10 m from single retained mature trees and along clear-cut edges, despite having access to full sunlight (Aaltonen 1919; Björkman 1945; Hagner 1962; Valkonen et al. 2002; Elfving and Jakobsson 2006; Kuuluvainen and Yllasjarvi 2011). Contrary to suggestions that adult trees promote seedling growth via common mycorrhizal networks, i.e., the “mother-tree” or the “wood-wide-web” (Simard et al. 1997), these patterns suggest that belowground competition serves as an important control on seedling growth in this area since the zone of suppressed seedling growth is similar in size to the zone of mature tree root system activity (Göttlicher et al. 2008; Henriksson et al. 2021).
Soil conditions affect the importance of below versus aboveground competition; belowground resources are naturally more limiting on poor and dry soils than on nutrient-rich, moist soils (Coomes and Grubb 2000). Since Scots pine is relatively drought-hardy, and it is the dominant species on dry sites in northern boreal Fennoscandia where few other tree species thrive, dry and poor soil conditions are common site conditions for Scots pine. Here, light would be expected to be of lower relative importance than soil resources, and in this region, nitrogen (N) availability is generally limiting for forest growth (Tamm 1991; Grossnickle 2000; Bhatti et al. 2006). Also, N is available to plants mainly in organic form (amino acids) in boreal forest soil, but inorganic sources (ammonia and nitrate) are also utilized (Inselsbacher and Näsholm 2012; Högberg et al. 2017). Soil microorganisms further limit the N availability for plants by retaining most of the nitrogen available from decomposing organic material and carbon substrates exudated by tree roots (Nazir et al. 2009; Näsholm et al. 2013; Högberg et al. 2017). When trees die or are harvested, the supply of carbon to the microorganisms decrease, limiting their demand for N. Consequently, N is released which can be utilized by the next generation of trees (Högberg et al. 2017). The simultaneous harvesting of all trees within a given area also liberates regenerating seedlings from competition with mature trees, enabling high initial growth rates (Petritan et al. 2011). This facilitation of regeneration by clear-cutting has been one of the main arguments for proponents of the method (Lundmark 2020).
Rotational forest management (RFM), which includes clear-cutting of mature trees, is currently the most common modern forestry practice in Scandinavia and other boreal forest regions (Lundmark et al. 2013). It enables economically sustainable forest management by ensuring a continuous interannual supply of wood through rotation between different even-aged plots (D'Amato et al. 2020). However, clear-cutting has been the subject of intense debates within the past decades for its potential negative impacts on ecological aspects including biodiversity, habitat quality, and soils as well as aesthetics and climate (Simonsson et al. 2015; Lundmark 2020; Larsen et al. 2022). An alternative to RFM is continuous cover forestry (CCF), i.e., uneven-aged management, where forest cover is maintained throughout the sylvicultural cycle by performing selective cuttings at regular intervals within uneven-aged plots (Pommerening and Murphy 2004; Schütz et al. 2012; Ahlstrom and Lundqvist 2015; Mason et al. 2022). However, CCF requires seedlings to establish and grow within gaps of limited size, where seedlings are influenced by interaction with mature trees to a higher degree than in RFM (McCarthy 2001). The proportion of edge environments where seedlings are under competition from the adjacent mature trees increases as the gap size decreases, eventually leading to a decrease in volume growth of the trees (Walters et al. 2006; Ruuska et al. 2008). Understanding the competitive interactions between seedlings and mature trees is important in assessing how seedling recruitment and growth is affected by different forestry management practices.
Though many observational studies have shown that Scots pine regeneration can suffer from competition of mature trees, experimental data isolating the specific mechanisms for these interactions are scarce (Högberg and Högberg 2022). The aim of our study was to clarify the environmental factors that control survival and growth of Scots pine seedlings in relation to mature trees, which is highly relevant in the context of CCF. We compared seedling establishment and growth of sown and planted Scots pine seedlings in three adjacent environments: within a pine forest, near a clear-cut edge, and on a clear-cut. In each of these environments, we also applied three paired treatments: control, N fertilization to increase the availability of N, and root isolation plots to physically reduce belowground competition between seedlings and mature trees. We tested the following hypotheses: (1) in untreated control plots, seedling survival and growth would be highest in the clear-cut due to the combination of high-light availability and absence of belowground competition with mature trees, (2) root isolation would enhance survival and growth of seedlings, with the greatest effect at the forest edge, where the reduced belowground competition would positively interact with the high-light availability, and (3) N fertilization would enhance seedling survival and growth, with this effect expected to be greatest at the in the clear-cut due to the combination of high-light availability and absence of belowground competition with mature trees. Testing these hypotheses in combination will provide valuable insight into the mechanisms that control pine regeneration, which are highly relevant in a context of adapting management approaches toward new regulations, e.g., clear-cut free forestry (EC 2021).
Material and methods
Study site conditions and experimental treatments
The study site is located near Åheden (64°13′ N, 19°48′ E) within the Svartberget research forest in Västerbotten county, northern Sweden. The site is located approximately 60 km from the coast on an alluvial sand-silt plain. Forests in Västerbotten county are dominated by Scots pine (Pinus sylvestris) and Norway spruce (Picea abies) either in single species (46.1% and 23.7% of productive forest area, respectively) or mixed stands (12.9%) (Petersson 2021).
The normal mean annual precipitation in the area is between 600 and 800 mm, the mean annual temperature is between + 2 and + 3 °C, and the length of growing season 150–160 days (SMHI 2023). Snow cover typically lasts from early November to early may (Axelsson et al. 2014). Climate data during the experiment are presented in Table 1.
The soil at the study site is a weakly podzolized sediment of sandy silt, classified as a Cambic Podzol (FAO 1988). The organic mor layer has a depth of ca. 2 cm (Högberg and Högberg 2002). The understory is dominated by ericaceous shrubs; lingonberry (Vaccinium vitis-idea L.) and bilberry (Vaccinium myrtillus L.), as well as a dense moss layer [mostly Dicranum spp. Hedw. with some Pleurozium schreberi (Brid.) Mitt] (Forsmark et al. 2020). In the clear-cut area, lingonberry also dominates the community but with a much lower cover. Reindeer lichen [Cladonia rangiferina (L.) F. H. Wigg.] and common heather [Calluna vulgaris (L.) Hull] constitute most of the remaining vegetation cover in the clear-cut.
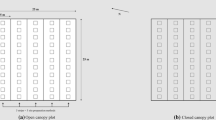
We set up an experiment at the site utilizing three alternative environments in relation to an intact mature forest (Fig. 1). First, we stablished plots directly beneath the intact canopy, where trees could influence both light and soil resource availability (hereafter “Forest”). Second, plots were established at the edge of the clear-cut (i.e., the south facing edge of the adjacent forest) where belowground competition with mature trees was present, but light competition was absent (hereafter “Edge”). The third environment was in the open clear-cut, where neither below nor above ground competition with the adjacent mature forest was present (hereafter “Clear-cut”). The clear-cut utilized for the study was done in early June 2013, forming a gap in the forest of approximately 100 m by 50 m. The clear-cut area had its long side adjacent to a road, which increased the total open area so that shadow from the trees on the south side would not affect the light availability on the clear-cut. No stump removal or other silvicultural soil preparation methods were used prior to setting up the experiment. Within each of these three environments, fifteen experimental plots (1.5 m × 1.5 m, 2.25 m2) were laid out in late June 2013 parallel to the forest edge. The plots inside the forest and out on the clear-cut were placed 15 m from the edge boundary, respectively, whereas the edge plots were placed 2 m into the clear-cut.
Schematic representation of the experimental design at the study site. The design incorporates three growing environments: “Forest” is located 15 m into the mature pine forest from the forest edge, “Edge” is located 2 m out from the forest edge, and “Clear-cut” is located 15 m from the forest edge out on the harvested area. For each growing environment, there are five replicates (blocks 1–5). For each replicate, there are three randomly distributed treatment plots: control (white), nitrogen (N) addition (green), and root isolation (yellow). Each treatment plot is split in one half where eight nursery grown seedlings were planted (P), and two quarters with sown seeds (S) and spontaneously regenerated seedlings (C) were measured, respectively
Within each of the three environments, fifteen experimental plots were arranged into five blocks where each of three nutrient competition treatments was represented: a control, root isolation, and N fertilization (Fig. 1). The control treatment plots were left untreated, whereas the root isolation treatment involved inserting a 1.5 m × 1.5 m steel frame 75-cm deep to physically prevent competition with mature tree roots, while maintaining water infiltration capacity. The N addition treatment aimed at evaluating whether fertilization with N would result in a similar seedling response pattern to a physical release from root competition. Nitrogen fertilizer was applied at a rate of 75 kg N ha−1 yr−1, which was applied in June annually in liquid form. This was accomplished by diluting 16.8 g of NH4NO3 in 10 l of tap water and applying evenly on designated experimental plots (2.25 m2). The unfertilized experimental plots (control and root isolation) were simultaneously watered with 10 l of H2O without any fertilizer. These three treatments were randomly placed within each of the fifteen blocks (Fig. 1).
Each experimental plot was separated into two 1.5 m × 0.75 m subplots. One was used to plant eight pine seedlings. The other was further divided into two 0.75 m × 0.75 m subplots. One was sown with 120 Scots pine seeds following manual soil preparation (organic layer removed and small depressions created in the soil), and the other left as is as a “spontaneous control” for measuring natural seedling regeneration. Planting and sowing were performed June 19–20, 2013. The planted pine seedlings were attacked in 2014 by large pine weevils (Hylobius abietis L.) leading to the death of 72% of planted seedlings (92% in the clear-cut, 80% at the forest edge, and 46% in the forest). This is consistent with what is frequently observed for seedlings planted after a clear-cut without any soil preparation, which provides ideal conditions (i.e., stumps, exposed roots, bark, slash, etc.) for the larval and pupal stage of the weevil (Örlander et al. 1997). The remaining seedlings that were planted in 2013 were, therefore, harvested in 2014 and replaced by new seedlings. The data on planted seedlings presented here are, therefore, from the second cohort of seedlings planted in May 2014. The planted seedlings were obtained from Gideå nursery and had an original mean height of 8.5 cm, a mean diameter of 2.3 mm, and a dry weight root:shoot ratio of 0.33.
PRS™ probes
We estimated soil nitrogen in the form of nitrate (NO3−) and ammonium (NH4+) availability using Plant Root SimulatorTM (PRSTM) probes (Western Ag Enterprises, Inc., Saskatoon, Canada). PRSTM probes consist of ion-exchange resin membranes (IEM) fitted on a plastic frame and are inserted vertically into the soil to sorb ions. Each measurement included two pairs of probes (four probes in total) with either an anion IEM (adsorbs NO3−) or cation IEM (adsorbs NH4+).
The nitrogen supply rate was measured in the planted subplots only. The probes were inserted into the soil on July 6, 2020, and removed after 6 weeks on August 17, 2020. A first cleaning of the probes was done on site using a light brush removing most of the attached soil. The probes were then taken to the laboratory and thoroughly cleaned using distilled water. The four probes per sample were packaged together in individual resealable plastic bags and sent to Western Ag for processing (elution of the adsorbed ions using HCl) and analysis. Results from both probes of each type were pooled prior to measurements by Western Ag. Inorganic N concentration (NH4+–N and NO3−–N) was measured colorimetrically using an automated flow injection analysis system (Skalar San + + Analyzer, Skalar Inc., NL), while the remaining elements were measured by inductively coupled plasma optical emission spectrometry (ICP-OES). Data are reported as the total quantity of each element adsorbed in µg per 10 cm2 of ion-exchange membrane surface area per incubation period.
Seedling inventories
We counted the number of surviving pine seedlings in each treatment plot the first 4 years in the field (2013–2016 for sown and 2014–2017 for planted seedlings) to follow the early development. A follow-up inventory was done in June 2020, when we in addition measured the individual height of each surviving seedling as well as stem base diameter for the planted seedlings. The 2020 inventory data were analyzed in this paper. For the sown seedlings, we did not measure the stem base diameter because high seedling density made this intractable. Naturally regenerated seedlings were absent in most of the spontaneous control plots that were neither sown nor planted, we, therefore, excluded these spontaneous control plots from our analysis.
Statistical analysis
RStudio was used for all analyses (R Core Team 2021). Seedling survival and nitrogen availability were analyzed using linear models, with the lm function in [stats] package (R Core Team 2021). The number of living seedlings per plot was used as response variable for survival analyses of planted and sown seedlings, respectively. Linear mixed models were made for height with plot nested within forest type as random factor to account for within-site variability, using the lmer function in the [lmerTest] package (Kuznetsova et al. 2017). Environment and treatment were used as fixed factors in all models. The natural logarithm of the response variable was used in all models except for planted seedling survival to fulfill the assumptions of homoscedasticity and normal distribution of residuals for the models. For sown seedlings, some subplots had 0 survival; therefore, 1 was added to each plot prior to analysis to be able to use the natural logarithm transformation.
Each model was then tested for the main effects of and interaction between forest type and treatment with ANOVA type III using the [car] package (Fox and Weisberg 2018). The [emmeans] package (Lenth 2021) was used to produce estimated marginal means from the models, and the [ggplot2] package (Wickham 2016) was used to present results graphicly.
Results
Seedling survival
For survival, there was significant interaction between environment and treatment for both sown and planted seedlings (Table 2). In the control plots, survival of sown seedlings was significantly higher in the clear-cut and forest edge than in the forest (Fig. 2A). The only significant effect of any treatment on sown seedling survival was inside the forest (Fig. 2A), where root isolation resulted in approximately 10 times higher sown seedling survival compared to the control plots (Fig. 2A). For the planted seedlings, there was no significant difference in survival between either control plots in the different environments or the three treatments within each environment (Fig. 2B). The only noticeable effect was that survival was significantly lower in the fertilized forest plots than on the fertilized clear-cut as well as fertilized and root isolated forest edge plots (Fig. 2B).
Estimated marginal means from linear models for survival of A germinated seedlings out of 120 sown per plot plus additional self-sown seedlings after 7 years and B planted seedlings after 6 years in field. Letters indicate significant differences including interaction. Values are back-transformed from natural logarithmic scale. Error bars indicate standard error
Seedling growth
For growth, there was significant interaction between environment and treatment (Table 3). In the control plots, height growth of sown seedlings was not significantly different between the three environments (Fig. 3A) while planted seedlings had grown significantly higher, and with thicker stems, in the clear-cut than in the forest edge and forest (Fig. 3B and C). The planted seedlings had significantly thicker stems in the forest edge than in the forest (Fig. 3C). Fertilization promoted the growth of seedlings most noticeably in the clear-cut, and this was the case for both the sown and the planted seedlings (Fig. 3). In the clear-cut, both sown and planted seedlings on fertilized plots were > 50% higher, and the planted seedlings had about 90% thicker stems than their respective counterparts on control plots (Fig. 3). In the forest edge, both the fertilization and the root isolation had a significant positive effect on seedling growth, for both sown and planted seedlings, the heights were > 50% higher (Fig. 3A, B), and the planted seedlings had c. 70% thicker stems in these plots than on control plots. In the forest, it was only the root isolation that had a positive effect on seedling growth, but only for the planted seedlings (Fig. 3B and C).
Estimated marginal means from linear mixed models for height of A sown seedlings after 7 years, B planted seedlings, and C stem base diameter of planted seedlings after 6 years in field. Letters indicate significant differences including interaction. Values are back-transformed from natural logarithmic scale. Error bars indicate standard error
Soil nitrogen concentration
In the fertilized plots, the nitrate supply rate was significantly higher than in control and root isolated plots in all environments, but with no significant difference between the three environments in any treatment. There was no significant difference between spontaneous control and planted subplots within any treatment in any of the three environments (Fig. 4A).
Nitrogen supply rate measured by PRS™ probes over 6 weeks in the summer of 2020 in the planted subplots in form of A nitrate and B ammonium. The results are estimated marginal means from linear models. Letters indicate significant differences including interaction. Values are back-transformed from natural logarithmic scale. Error bars indicate standard error
The ammonium supply rate was significantly higher in fertilized plots compared to both control and root isolated plots in the forest and forest edge, but not in the clear-cut. As with nitrate, there was neither any significant difference in ammonium supply rate between root isolated and control plots in any of the three environments, nor between environments in these treatments (Fig. 4B).
Discussion
In northern forest ecosystems, success of forest regeneration is known to be influenced by both above- and belowground competitions. A key aim of this study was to separate between these two controls, by applying three treatments along a forest to clear-cut gradient.
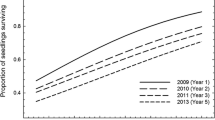
A general observation regarding both sown and planted seedlings in this experiment was a consistent visible trend of one or two seedlings being larger than the others in each plot, clearly displaying internal competition among the seedlings. Further, a larger share of sown seedlings died during the period of the study as compared to the planted seedlings, leaving a higher variability for sown (0–39 per plot) than for planted (3–8 per plot) seedlings. This was expected since survival of direct sown seedlings in general is lower than for planted seedlings (Grossnickle and Ivetić 2017). The higher variability allows for larger differences in survival between treatments and environments among the sown seedlings than the planted ones. The most severe decreases in sown seedling survival occurred in 2014, in both the clear-cut and in the N-fertilized plots in the forest edge (i.e., the environments where the initial germination numbers were highest). In 2020, mortality was high in all environments and treatments. These peaks in mortality followed years with dryer than average early summer conditions (2014, 2018, and 2019), which likely intensified inter-specific competition for water (Fig. 5). An increase in sown seedling numbers in 2016, that was highest in the forest, suggests that there were also newly germinated self-sown seeds included in the measurements. The internal competition and the addition of self-sown seedlings caused a large variability in growth in most plots that made patterns difficult to detect. Regardless, we found significant treatment responses which were large enough to be detected against this background noise caused by internal seedling competition.
An overview of the development of sown seedling germination and survival from 2013 to 2020. The summers 2014, 2018, and 2019 were unusually dry, marked by arrows. Large decreases in numbers of sown seedlings coincided with those dry summers, especially between 2016 and 2020. An increase in numbers of sown seedlings in 2016 indicates that addition of self-sown seedlings occurred
With our first hypothesis, we expected that seedling survival and growth in the control plots would be higher the further away from the mature forest canopy cover due to less belowground competition from mature trees. In support of our hypothesis, the number of surviving sown seedlings in the control plots was about ten times higher in the clear-cut environment than in the forest environment, while it occurred at an intermediate level at the forest edge. Regarding the lower germination and survival success of sown seedlings in the forest, direct competition for resources is the most likely explanation for these patterns. However, it is possible that several other processes contributed to this response, such as seed predation by carabid beetles and seedling predation by slugs, which have been shown to be higher under canopies than in clear-cut areas (Nystrand and Granstrom 2000). Further, the soil temperature increases following canopy removal, since more light energy reaches the ground (Grossnickle 2000). The optimal temperature for germination of Scots pine is 20–30 °C (Kamra and Simak 1968; Örlander et al. 1990), and these temperatures are less likely to occur under the shading canopy than on the clear-cut. It should in this context also be mentioned that the higher occurrence of ericaceous shrubs under the canopy than in the open areas could have inhibitory effect on regeneration of conifers (Mallik 2003). However, since the ground vegetation and organic layer was removed prior to sowing in the sown plots, this would affect the planted seedlings more than the sown. In addition to survival, the growth patterns we observed also supported our hypothesis, as growth of planted seedlings was highest in the clear-cut, intermediate in the forest edge, and lowest in the forest. However, the trend of increased height growth of sown seedlings away from the forest canopy (i.e., in the clear-cut) was not significant due to the large variability in heights.
For our second hypothesis, we expected that root isolation would enhance survival and growth of seedlings by suppressing competition from mature trees for water and nutrients. We also expected this effect to be strongest at the forest edge, where the seedlings still have full access to light, but where they are suppressed by belowground competition. In partial support of our hypothesis, we found that root isolation enhanced survival of sown seedlings in the edge environment, achieving similar survival as observed in clear-cut control plots. However, inconsistent with our hypothesis, we found that this response occurred also in the forest environment, suggesting that the higher light in the edge environment was not an important factor contributing to the root isolation response. This indicates that underground competition rather than light competition is the primary limitation for the establishment of sown seedlings in the forest. This brings up a question regarding the previous hypothesis: If light availability was not a main driver of survival in the forest, why was the survival of sown seedlings higher in the forest edge control plots than in the forest control plots when the main difference between the two environments was light availability? One explanation is that the belowground competition in the forest edge was reduced, since approximately half of the roots of mature trees in the forest edge environment are dead ones from the harvested side of the edge. An additional explanation might be the increase in temperature following canopy removal. The optimal soil temperature for root growth and water uptake capacity is 30–35 °C for Scots pine (Örlander et al. 1990), temperatures less likely to occur under the canopy than in full light. Further, the nutrient uptake capacity of seedlings has been found to decrease at soil temperatures below 15 °C (Örlander et al. 1990). Temperature increase can also enhance nutrient availability, through higher mineralization rates (Rustad et al. 2001; Strömgren and Linder 2002).
Regarding growth, both sown and planted seedling growths were enhanced by root isolation in the edge environment, in line with our hypothesis. Growth of planted seedlings was also enhanced by root isolation in the forest, achieving similar growth as observed in clear-cut control plots, despite the lower light conditions in the forest. This indicates that underground competition rather than light competition limits the growth of seedlings in the forest, similarly as for the survival of sown seedlings as described above. An early root isolation experiment conducted in the same area as the current study showed that pine seedlings growing in a root isolated area under closed canopy of mature pine grew better than the seedlings outside the isolated area, despite the lower light conditions; though the experiment was only made on a single plot and was not replicated (Björkman 1945). However, we found no effect of root isolation on sown seedling growth in the forest, indicating that mortality (described above) may be a more important mechanism by which belowground competition impacts sown seedlings. The absence of enhanced growth of sown seedlings in response to root isolation in the forest may also have been due to higher intra-specific competition among the sown seedlings themselves, as the significantly higher survival resulted in more seedlings to share the resources in the plot. Nevertheless, the positive responses of root isolation in our study are in line with experimental root trenching studies on other tree species resulting in positive responses from seedlings that are relieved from underground competition (Coomes and Grubb 2000; Devine and Harrington 2008; Wagner et al. 2010; Petritan et al. 2011). These findings are also relevant regarding recently popular discussions regarding the “mother-tree” or the “wood-wide-web,” where positive effects of adult trees on seedlings are proposed (Simard et al. 1997; Klein et al. 2016; Cahanovitc et al. 2022). Clearly, our data do not provide any support for these propositions. To the contrary, our results suggest that strong belowground competitive effects from adult trees suppress seedlings, in line with recent arguments questioning the validity of the mechanisms of common ectomycorrhiza networks as a resource transfer tool for adult trees to support seedlings (Karst et al. 2023; Henriksson et al. 2023).
Our third hypothesis was that N fertilization will enhance survival and growth, with the effect expected to be strongest in the clear-cut and weakest in the forest due to greater competition for other soil resources (e.g., water) from mature trees. Inconsistent with our third hypothesis, we found that N fertilization had no effect on sown or planted seedling survival relative to control plots within any of the environments; however, survival of N-fertilized planted seedlings was significantly lower in the forest than in the other environments. A potential explanation for this is that fertilized forest plots showed the highest supply rate of NH4+ we observed. The competition for water from mature trees may have increased the salt concentration, which potentially could have had a toxicity effect for small seedlings (Öhlund and Nasholm 2001). Since N fertilization increases nutritional value of plants, predation by herbivores could also have played a role (Ball et al. 2000). A potential increase in seedling predation by slugs in the forest environment (Nystrand and Granstrom 2000) might have been further enhanced by N fertilization since N fertilization has been found to increase the susceptibility of seedling predation by slugs (Albrectsen et al. 2004). Regarding growth, N fertilization significantly enhanced the growth of both sown and planted seedlings in the clear-cut, and to a smaller extent in the forest edge, while in the forest, there was not any significant effect of fertilization on growth at all. This pattern matched the pattern of the nitrate and ammonium supply analyses where less of the N-supply remained in the clear-cut, more in the forest edge and most in the forest, which is likely indicative of the biologically utilized N-supply in each environment. There was no indication from the PRS™ probe measurements that the supply of other major nutrients (P, K, S, Ca, and Mg) was lower in the forest than in the other environments, which otherwise could have helped explain the results if the added nitrogen caused deficiency of other nutrients (see supplementary material Fig. S1). The significantly higher remaining supply of ammonium in fertilized forest plots suggests that competition for other soil resources (e.g., water) between seedlings and mature trees likely also hindered seedling growth, which may have also limited seedling N fertilizer uptake. This would agree with the previous studies finding that water uptake affects uptake of N via mass flow in high N environments (McMurtrie and Nasholm 2018; Henriksson et al. 2021). Likewise, the aboveground growth of fertilized mature trees has been found to co-vary with precipitation (Lim et al. 2015). Soil resource availability may also interact with light availability since light conditions affect the capability of plants to utilize nutrients in the soil (Coomes and Grubb 2000). More light results in a higher potential for photosynthesis and, therefore, higher transpiration, which, in turn, leads to increased N uptake via mass flow (Oyewole et al. 2016; McMurtrie and Nasholm 2018; Henriksson et al. 2021). This would explain why the effect of N fertilization was higher in the clear-cut since the absence of competition for water allows for higher transpiration rates.
The release from competition for nutrients vs water has been studied in a previous experiment near our study site, where mature pine trees were girdled to release belowground competition for nutrients (Högberg et al. 2001; Ahlstrom and Lundqvist 2015). The needles of the mature trees remained the two 2 years after girdling, i.e., the trees still took up water for this period (Bhupinderpal‐Singh et al. 2003). In that experiment, the release of competition for nutrients caused by tree girdling resulted in a regeneration pulse already during these first 2 years, when the girdled trees maintained their needles (Axelsson et al. 2014). However, the number of seedlings established doubled the following 2 years after the needles were dropped, i.e., when competition for light and water decreased together with a release of nitrogen from decomposing needles (Axelsson et al. 2014). The seedlings established during the 3rd year after girdling showed the highest yearly increment and total height in a 12-year inventory period after girdling (Axelsson et al. 2014). Our data show similar patterns complementary to these findings by suggesting that simultaneous release from competition of water and nutrients has a better effect than N fertilization under low-light conditions (i.e., under the canopy), that both measures are effective under high-light conditions with underground competition (i.e., at the forest edge), while N fertilization is most effective where the seedlings have full light and no underground competition (i.e., in the clear-cut). This also agrees with the previous findings indicating that competition for water and nutrients is of relatively higher importance for Scots pine regeneration than competition for light, at least on nutrient poor soil with high permeability which is common in Scots pine forests of northern Sweden.
Conclusions
The results from this study support the previous findings that Scots pine seedlings suffer from belowground competition near mature trees. Despite internal competition among seedlings that increase as seedlings grow, we found an overall pattern that the seedlings grew best in clear-cut environments, followed by forest edge, and lastly under mature forest. We have here showed that the belowground competition along a forest edge may be alleviated by N fertilization and root isolation. Physical root isolation by steel frames and annual fertilization would be impractical to implement in practice; however, targeted microsite fertilization at planting locations in small gaps or clear-cut edges might be an option to improve regeneration of Scots pine in proximity to mature trees. Root isolation could potentially be achieved by deep soil scarification along forest edges that would interrupt root connection from mature trees. To avoid that the positive effect on seedling establishment is not counteracted by damage to the mature trees by root loss, the scarification should preferably be limited to cut off mature tree roots only in proximity to the planting position of the new seedling. Of course, this would have to be tested, preferably in a larger scale at more sites and in forest edges facing all directions. Overall, our results highlight that a transition to CCF forestry for northern European pine forests will necessitate substantial management innovation to alleviate these belowground resource constraints on seedlings in proximity to mature trees if aimed regeneration goals are to be maintained.
References
Aaltonen VT (1919) Über die natürliche Verjungung der Heidevälder im Finnischen Lappland I. Communicationes Ex Instituto Quaestionum Forestalium Finlandiae 1:319
Ahlstrom MA, Lundqvist L (2015) Stand development during 16–57 years in partially harvested sub-alpine uneven-aged Norway spruce stands reconstructed from increment cores. For Ecol Manag 350:81–86. https://doi.org/10.1016/j.foreco.2015.04.021
Albrectsen B, Gardfjell H, Orians C, Murray B, Fritz R (2004) Slugs, willow seedlings and nutrient fertilization: Intrinsic vigor inversely affects palatability. Oikos 105:268–278. https://doi.org/10.1111/j.0030-1299.2004.12892.x
Axelsson EP, Lundmark T, Hogberg P, Nordin A (2014) Belowground competition directs spatial patterns of seedling growth in boreal pine forests in Fennoscandia. Forests 5:2106–2121. https://doi.org/10.3390/f5092106
Ball JP, Danell K, Sunesson P (2000) Response of a herbivore community to increased food quality and quantity: an experiment with nitrogen fertilizer in a boreal forest. J Appl Ecol 37:247–255. https://doi.org/10.1046/j.1365-2664.2000.00487.x
Bhatti JS, Apps MJ, Lal R (2006) Anthropogenic changes and the global carbon cycle. In: Bhatti JS, Lal R, Apps MJ, Price MA (eds) Climate change and managed ecosystems. CRC Press, pp 71–91
Bhupinderpal-Singh NA, Ottosson Löfvenius M, Högberg M, Mellander PE, Högberg P (2003) Tree root and soil heterotrophic respiration as revealed by girdling of boreal Scots pine forest: extending observations beyond the first year. Plant Cell Environ 26:1287–1296
Björkman E (1945) Studier över ljusets betydelse för föryngringens höjdtillväxt på norrländska tallhedar. [Studies of the importance of light for regeneration height growth at Northern pine heaths.]
Cahanovitc R, Livne-Luzon S, Angel R, Klein T (2022) Ectomycorrhizal fungi mediate belowground carbon transfer between pines and oaks. ISME J 16:1420–1429. https://doi.org/10.1038/s41396-022-01193-z
Coomes DA, Grubb PJ (2000) Impacts of root competition in forests and woodlands: a theoretical framework and review of experiments. Ecol Monogr 70:171–207. https://doi.org/10.1890/0012-9615(2000)070[0171:Iorcif]2.0.Co;2
D’Amato D, Veijonaho S, Toppinen A (2020) Towards sustainability? Forest-based circular bioeconomy business models in Finnish SMEs. For Policy Econ 110:101848. https://doi.org/10.1016/j.forpol.2018.12.004
de Chantal M, Leinonen K, Kuuluvainen T, Cescatti A (2003) Early response of Pinus sylvestris and Picea abies seedlings to an experimental canopy gap in a boreal spruce forest. Forest Ecol Manag 176(1–3):321–336
Devine WD, Harrington TB (2008) Belowground competition influences growth of natural regeneration in thinned Douglas-fir stands. Can J for Res 38:3085–3097. https://doi.org/10.1139/X08-150
EC (2021) European commission, New EU forest strategy for 2030. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52021DC0572
Elfving B, Jakobsson R (2006) Effects of retained trees on tree growth and field vegetation in Pinus sylvestris stands in Sweden. Scand J for Res 21:29–36. https://doi.org/10.1080/14004080500487250
Erefur C, Bergsten U, Lundmark T, de Chantal M (2011) Establishment of planted Norway spruce and Scots pine seedlings: effects of light environment, fertilisation, and orientation and distance with respect to shelter trees. New for 41:263–276. https://doi.org/10.1007/s11056-010-9226-8
FAO (1988) FAO/Unesco soil map of the Wordl, revised legend, with corrections and updates. Technical Paper 20, ISRIC.
Forsmark B, Nordin A, Maaroufi NI, Lundmark T, Gundale MJ (2020) Low and high nitrogen deposition rates in northern coniferous forests have different impacts on aboveground litter production, soil respiration, and soil carbon stocks. Ecosystems 23:1423–1436. https://doi.org/10.1007/s10021-020-00478-8
Fox J, Weisberg S (2018) An R companion to applied regression. Sage publications
Göttlicher SG, Taylor AFS, Grip H, Betson NR, Valinger E, Högberg MN, Högberg P (2008) The lateral spread of tree root systems in boreal forests: estimates based on 15N uptake and distribution of sporocarps of ectomycorrhizal fungi. For Ecol Manag 255:75–81. https://doi.org/10.1016/j.foreco.2007.08.032
Gray AN, Spies TA (1996) Gap size, within-gap position and canopy structure effects on conifer seedling establishment. J Ecol 84:635–645. https://doi.org/10.2307/2261327
Grossnickle SC (2000) Ecophysiology of northern spruce species: the performance of planted seedlings. NRC Research Press
Grossnickle SC, Ivetić V (2017) Direct seeding in reforestation–a field performance review. Reforesta. https://doi.org/10.21750/REFOR.4.07.46
Hagner S (1962) Naturlig föryngring under skärm. [Natural regeneration under shelterwood.] https://pub.epsilon.slu.se/10058/1/medd_statens_skogsforskningsinst_052_04.pdf
Henriksson N, Lim H, Marshall J, Franklin O, McMurtrie RE, Lutter R, Magh R, Lundmark T, Näsholm T (2021) Tree water uptake enhances nitrogen acquisition in a fertilized boreal forest - but not under nitrogen-poor conditions. New Phytol 232:113–122. https://doi.org/10.1111/nph.17578
Henriksson N, Marshall J, Högberg MN, Högberg P, Polle A, Franklin O, Näsholm T (2023) Re-examining the evidence for the mother tree hypothesis – resource sharing among trees via ectomycorrhizal networks. New Phytol 239:19–28. https://doi.org/10.1111/nph.18935
Högberg MN, Högberg P (2002) Extramatrical ectomycorrhizal mycelium contributes one-third of microbial biomass and produces, together with associated roots, half the dissolved organic carbon in a forest soil. New Phytol 154:791–795. https://doi.org/10.1046/j.1469-8137.2002.00417.x
Högberg P, Högberg MN (2022) Does successful forest regeneration require the nursing of seedlings by nurse trees through mycorrhizal interconnections? For Ecol Manag. https://doi.org/10.1016/j.foreco.2022.120252
Högberg P, Nordgren A, Buchmann N, Taylor AFS, Ekblad A, Högberg MN, Nyberg G, Ottosson-Löfvenius M, Read DJ (2001) Large-scale forest girdling shows that current photosynthesis drives soil respiration. Nature 411:789–792. https://doi.org/10.1038/35081058
Högberg P, Näsholm T, Franklin O, Högberg MN (2017) Tamm Review: on the nature of the nitrogen limitation to plant growth in Fennoscandian boreal forests. For Ecol Manag 403:161–185. https://doi.org/10.1016/j.foreco.2017.04.045
Inselsbacher E, Näsholm T (2012) The below-ground perspective of forest plants: soil provides mainly organic nitrogen for plants and mycorrhizal fungi. New Phytol 195:329–334. https://doi.org/10.1111/j.1469-8137.2012.04169.x
Kamra SK, Simak M (1968) Germination studies on Scots pine (Pinus silvestris L.) seed of different provenances under alternating and constant temperatures.
Karst J, Jones MD, Hoeksema JD (2023) Positive citation bias and overinterpreted results lead to misinformation on common mycorrhizal networks in forests. Nat Ecol Evol 7:501–511. https://doi.org/10.1038/s41559-023-01986-1
Klein T, Siegwolf RTW, Körner C (2016) Belowground carbon trade among tall trees in a temperate forest. Science 352:342–344. https://doi.org/10.1126/science.aad6188
Kobe RK, Pacala SW, Silander JA, Canham CD (1995) Juvenile Tree Survivorship as a Component of Shade Tolerance. Ecol Appl 5:517–532. https://doi.org/10.2307/1942040
Kuuluvainen T, Yllasjarvi I (2011) On the natural regeneration of dry heath forests in Finnish Lapland: a review of V. T. Aaltonen (1919). Scand J for Res 26:34–44. https://doi.org/10.1080/02827581.2011.517951
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest Package: Tests in Linear Mixed Effects Models. J Stat Softw 82:1–26. https://doi.org/10.18637/jss.v082.i13
Larsen JB, Angelstam P, Bauhus J, Carvalho JF, Diaci J, Dobrowolska D, Gazda A, Gustafsson L, Krumm F, Knoke T (2022) Closer-to-Nature Forest Management. From Science to policy 12. European Forest Institute. https://doi.org/10.36333/fs12
Lenth R (2021) Emmeans: Estimated marginal means, aka least-squares means. R package version 1.7.1–1. https://CRAN.R-project.org/package=emmeans
Lim H, Oren R, Palmroth S, Tor-ngern P, Mörling T, Näsholm T, Lundmark T, Helmisaari H-S, Leppälammi-Kujansuu J, Linder S (2015) Inter-annual variability of precipitation constrains the production response of boreal Pinus sylvestris to nitrogen fertilization. For Ecol Manage 348:31–45. https://doi.org/10.1016/j.foreco.2015.03.029
Lundmark H, Josefsson T, Ostlund L (2013) The history of clear-cutting in northern Sweden - driving forces and myths in boreal silviculture. For Ecol Manag 307:112–122. https://doi.org/10.1016/j.foreco.2013.07.003
Lundmark H (2020) Clear-cutting - The most discussed logging method in Swedish forest history. Acta Univ agric Suec 2020:64, https://res.slu.se/id/publ/108511
Malcolm DC, Mason WL, Clarke GC (2001) The transformation of conifer forests in Britain - regeneration, gap size and silvicultural systems. For Ecol Manag 151:7–23. https://doi.org/10.1016/s0378-1127(00)00692-7
Mallik AU (2003) Conifer regeneration problems in boreal and temperate forests with ericaceous understory: role of disturbance, seedbed limitation, and keytsone species change. Crit Rev Plant Sci 22:341–366. https://doi.org/10.1080/713610860
Mason WL, Diaci J, Carvalho J, Valkonen S (2022) Continuous cover forestry in Europe: usage and the knowledge gaps and challenges to wider adoption. Forestry 95:1–12. https://doi.org/10.1093/forestry/cpac008
McCarthy J (2001) Gap dynamics of forest trees: a review with particular attention to boreal forests. Environ Rev 9:1–59. https://doi.org/10.1139/a00-012
McMurtrie RE, Näsholm T (2018) Quantifying the contribution of mass flow to nitrogen acquisition by an individual plant root. New Phytol 218:119–130. https://doi.org/10.1111/nph.14927
Näsholm T, Högberg P, Franklin O, Metcalfe D, Keel SG, Campbell C, Hurry V, Linder S, Högberg MN (2013) Are ectomycorrhizal fungi alleviating or aggravating nitrogen limitation of tree growth in boreal forests? New Phytol 198:214–221. https://doi.org/10.1111/nph.12139
Nazir R, Warmink JA, Boersma H, Van Elsas JD (2009) Mechanisms that promote bacterial fitness in fungal-affected soil microhabitats. FEMS Microbiol Ecol 71:169–185. https://doi.org/10.1111/j.1574-6941.2009.00807.x
Nystrand O, Granstrom A (2000) Predation on Pinus sylvestris seeds and juvenile seedlings in Swedish boreal forest in relation to stand disturbance by logging. J Appl Ecol 37:449–463. https://doi.org/10.1046/j.1365-2664.2000.00505.x
Öhlund J, Näsholm T (2001) Growth of conifer seedlings on organic and inorganic nitrogen sources. Tree Physiol 21:1319–1326. https://doi.org/10.1093/treephys/21.18.1319
Örlander G, Gemmel P, Hunt J (1990) Site preparation: a Swedish overview. BC Ministry of Forests
Örlander G, Nilsson U, Nordlander G (1997) Pine weevil abundance on clear-cuttings of different ages: a 6-year study using pitfall traps. Scand J for Res 12:225–240. https://doi.org/10.1080/02827589709355405
Oyewole OA, Jämtgard S, Gruffman L, Inselsbacher E, Näsholm T (2016) Soil diffusive fluxes constitute the bottleneck to tree nitrogen nutrition in a Scots pine forest. Plant Soil 399:109–120. https://doi.org/10.1007/s11104-015-2680-5
Petersson H (2021) Forest statistics 2021 - official statistics of Sweden. Swedish University of Agricultural Sciences, Department of Forest Resource Management, Umeå
Petritan IC, von Lupke B, Petritan AM (2011) Effects of root trenching of overstorey Norway spruce (Picea abies) on growth and biomass of underplanted beech (Fagus sylvatica) and Douglas fir (Pseudotsuga menziesii) saplings. Eur J Res 130:813–828. https://doi.org/10.1007/s10342-010-0473-1
Pommerening A, Murphy ST (2004) A review of the history, definitions and methods of continuous cover forestry with special attention to afforestation and restocking. Forestry 77:27–44. https://doi.org/10.1093/forestry/77.1.27
R Core Team (2021) R: A language and environment for statistical computing. https://www.R-project.org/
Rustad L, Campbell J, Marion G, Norby R, Mitchell M, Hartley A, Cornelissen J, Gurevitch J, Gcte N (2001) A meta-analysis of the response of soil respiration, net nitrogen mineralization, and aboveground plant growth to experimental ecosystem warming. Oecologia 126:543–562. https://doi.org/10.1007/s004420000544
Ruuska J, Siipilehto J, Valkonen S (2008) Effect of edge stands on the development of young Pinus sylvestris stands in southern Finland. Scand J for Res 23:214–226. https://doi.org/10.1080/02827580802098127
Schütz JP, Pukkala T, Donoso PJ, von Gadow K (2012) Historical emergence and current application of CCF. In: Pukkala T, von Gadow K (eds) Continuous cover forestry. Managing forest ecosystems. Springer, Dordrecht
Simard SW, Perry DA, Jones MD, Myrold DD, Durall DM, Molina R (1997) Net transfer of carbon between ectomycorrhizal tree species in the field. Nature 388:579–582. https://doi.org/10.1038/41557
Simonsson P, Gustafsson L, Ostlund L (2015) Retention forestry in Sweden: driving forces, debate and implementation 1968–2003. Scand J for Res 30:154–173. https://doi.org/10.1080/02827581.2014.968201
SMHI (2023). Monthly, season and annual maps - weather summary 1991–2020. Swedish Meteorological and Hydrological Institute. https://www.smhi.se/data/meteorologi/kartor/normal/arsmedeltemperatur-normal
Strömgren M, Linder S (2002) Effects of nutrition and soil warming on stemwood production in a boreal Norway spruce stand. Glob Chang Biol 8:1194–1204. https://doi.org/10.1046/j.1365-2486.2002.00546.x
Tamm CO (1991) Nitrogen-limited and nitrogen-depleted terrestrial ecosystems: ecological characteristics. In: Tamm CO (ed) Nitrogen in terrestrial ecosystems: questions of productivity, vegetational changes, and ecosystem stability. Springer, Berlin, pp 34–49
Valkonen S, Ruuska J, Siipilehto J (2002) Effect of retained trees on the development of young Scots pine stands in Southern Finland. For Ecol Manag 166:227–243. https://doi.org/10.1016/S0378-1127(01)00668-5
Wagner S, Collet C, Madsen P, Nakashizuka T, Nyland RD, Sagheb-Talebi K (2010) Beech regeneration research: from ecological to silvicultural aspects. For Ecol Manag 259:2172–2182. https://doi.org/10.1016/j.foreco.2010.02.029
Walters MB, Lajzerowicz CC, Coates KD (2006) Soil resources and the growth and nutrition of tree seedlings near harvest gap – forest edges in interior cedar–hemlock forests of British Columbia. Can J Res 36:62–76. https://doi.org/10.1139/x05-237
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Acknowledgements
We thank Tomas Lundmark for enabling the experimental set-up, Linda Gruffman for establishing the experiment, Corentin Abgrall for field measurements, and the field crew at Svartberget field station performing the yearly maintenance. We acknowledge the funding of BH’s PhD position from the Research School in Forest Genetics, Biotechnology, and Breeding at the Umeå Plant Science Centre, UPSC, at the Swedish University of Agricultural Sciences (SLU) being part of the Competence Centre program of the Swedish Governmental Agency for Innovation Systems (VINNOVA).
Funding
Open access funding provided by Swedish University of Agricultural Sciences. This work was supported by the Swedish Governmental Agency for Innovation Systems under Grant number 2016-00504, the Knut and Alice Wallenberg Foundation under Grant number KAW 2018.0259, and the Kempe Foundation.
Author information
Authors and Affiliations
Contributions
AN contributed to the conception of the research question and design of the work, BH contributed with analysis and interpretation of data and results as well as scientific writing of the work, AN and MJG contributed with scientific writing and revising it critically for sound and intellectual content.
Corresponding author
Ethics declarations
Conflict of interest
AN is besides her affiliation to the Swedish University of Agricultural Sciences employed at Stora Enso, a forest industry company. MJG and BH declare no conflict of interest and have no commercial affiliations.
Additional information
Communicated by Martin Ehbrecht.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Häggström, B., Gundale, M.J. & Nordin, A. Environmental controls on seedling establishment in a boreal forest: implications for Scots pine regeneration in continuous cover forestry. Eur J Forest Res 143, 95–106 (2024). https://doi.org/10.1007/s10342-023-01609-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-023-01609-1