Abstract
The Swedish wild boar (Sus scrofa) population has increased rapidly over the last decades, resulting in conflicts with human activities. Particularly, the increase has been challenging for agriculture as wild boar cause damage on crops and grasslands. To predict under what conditions to expect damage and where to prioritize management actions, basic knowledge about wild boar habitat and space use is needed. In this study, we used data from 99 wild boar equipped with GPS-collars, collected over a large temporal scale and throughout their distributional range in southern Sweden. We investigated wild boar home range size and habitat use across gradients of habitat availability and population density. Functional response in habitat use was assessed by estimating the use and availability of agricultural land on individual level and then, on population-level evaluating how use changed with changing availability. Finally, a potential response in habitat use was evaluated in relation to population density, i.e., the interaction between availability and population density. Home range size was negatively related to population density for both male and female wild boar. Wild boar used agricultural land more intensively with increasing population density and when other habitat types were less available. Our findings show that wild boar spatial behavior is highly context dependent and may vary considerably due to landscape characteristics and local conditions. Wild boars tend to overuse agricultural land at high densities which has strong implications for wildlife management. It is therefore important to consider local conditions when predicting space and habitat use by wild boar. Overall, this study provided a better understanding of the drivers of wild boar distribution and space use in agro-forested mosaic landscapes and how this knowledge can improve management practices.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
From being extirpated in the eighteenth century, the Swedish wild boar (Sus scrofa) population has increased rapidly since the 1970s, when some individuals escaped from enclosures in which they were held for recreational hunting and meat production (Truvé and Lemel 2003). Today, the population has re-established in the main parts of southern and central Sweden. The current population size is estimated to be over 300,000 animals and the reported annual hunting bag was more than 112,000 animals in 2023 (Swedish Association for Hunting and Wildlife Management). Wild boar can demonstrate high reproductive rates, adaptability, and opportunistic feeding habits (Massei et al. 1996; Schley and Roper 2003; Fonseca et al. 2011; Malmsten 2017). In Sweden, and other parts of Europe, this has led to a rapid range expansion and population increase as well as conflicts with human activities due to crop damage, vehicle collisions and disease transmission (Thurfjell et al. 2015; Gren et al. 2020; Stenberg et al. 2021). Agricultural fields are known to be a preferred habitat for wild boar (Thurfjell et al. 2009; Muthoka et al. 2022) and are used at a higher extent during summer than during the rest of the year (Sweden: Lemel et al. 2003, Thurfjell et al. 2009; Germany: Keuling et al. 2009). The challenge to agriculture has been of particular interest due to crop damage and severe economic losses for farmers. The modern agricultural landscape, providing a high abundance of nutritious feed over large areas, represents an interface for conflict between humans and wildlife. Farmers’ economic losses are expected to grow as both intensification of agricultural activities and wild boar abundance increase (Gren et al. 2020). Moreover, the risk of disease transmission between wild boar and pig farms have been accentuated due to the first case of Salmonella enterica subsp. enterica, serovar Choleraesuis in more than 40 years (Ernholm et al. 2022), and the recent outbreak of African swine fever (ASF) in the Swedish wild boar population (SVA 2023). Due to such conflicts, the management of wild boar has, as in other parts of the world, become an issue of national concern in Sweden, and an increased understanding of the wild boar distribution in agro-forested mosaic landscapes is crucial.
A common contemporary approach to monitor and map animal movements is the tagging of animals with GPS telemetry collars. By spatially locating individuals with high precision at a given time, this technique allows to investigate distributional variation in relation to landscape characteristics and local conditions (Cagnacci et al. 2010). A well-established and frequently used method of describing animal distribution is to estimate home range sizes. Animal spatial behavior is shaped by both social and environmental factors; forage availability and competition level are well known to effect space use (e.g., Tufto et al. 1996; van Beest et al. 2016). Home range size is by theory predicted to decrease with increasing food abundance (Ford 1983), and generally, higher-quality habitats are associated with smaller home ranges. Adjustment of home range size to resource levels has been demonstrated in a wide range of mammalian species (Ims 1987; Boitani et al. 1994; Lucherini and Lovari 1996; McLoughlin and Ferguson 2000; Schradin et al. 2010; Bjorneraas et al. 2012) and wild boar home range has shown to be negatively correlated to increasing resources as in years with tree seed mast (Bisi et al. 2018) and around baiting areas (Keuling et al. 2008b). The “density-dependent hypothesis” predicts that changes in home-range size are inversely related to population density (Massei et al. 1997). Increased local animal density increases the competition (Focardi et al. 2006), why competition is considered a main mechanism promoting density dependence. A reduction of home range size at high density has been confirmed in several species (roe deer: Kjellander et al. 2004; wild boar: Massei et al. 1997; moose: van Beest et al. 2016). In wild boar, inter-sexual differences in spatial behavior patterns are related to differences in reproductive strategies between males and females (Kurz and Marchinton 1972; Singer et al. 1981; Boitani et al. 1994; Cavazza et al. 2023; Miettinen et al. 2023). Females form family groups that can include several generations of adults and offspring, while adult males live isolated from the herd outside the rut period (Podgorski 2013). Although the territorial behavior in wild boar is poorly understood, male wild boar are reported to be less territorial than females, interacting more frequently with individuals of both sexes and with larger home ranges that overlap both sexes (Kay et al. 2017; Schlichting et al. 2022). Furthermore, due to their greater body size and advantageous physical characteristics (Spitz et al. 1998), males could be expected to be more resilient to interference competition.
Animals are known to adapt their spatial utilization according to perceived risk, often referred to as the “landscape of fear” (Gaynor et al. 2019). This suggests that animal movement patterns are influenced by predation risk. Although agricultural fields provide high energetic gain, it could also be a dangerous environment for wild boar considering that 20% of the annual hunting bag in Sweden is shot in crop fields (Swedish Association for Hunting and Wildlife Management 2017). However, the avoidance of risky areas (e.g., hunting areas) by wild boar is a debated issue, and is likely to vary at the local and individual scale (Tolon et al. 2009; Said et al. 2012; Brogi et al. 2020, 2022). Hence, context-dependency of animal spatial behavior is complex and behavioral decisions animals make often result in trade-offs between opposing needs, such as forage and safety (Brown et al. 1999).
While home range models may serve an important descriptive purpose, these models describe space use based solely on spatial location, and it is of ecological interest also to understand the causal processes of animal movement and distribution patterns. Analyzing a species’ distribution across habitats, i.e., habitat use, links individual animals to their environment by connecting behavior to resource availability (Johnson 1980) and habitat use is most commonly studied by comparing the use of a given habitat in relation to the availability of that habitat in the surrounding landscape (Boyce and McDonald 1999; Manly et al. 2002; Johnson et al. 2006). Although the availability of suitable habitats and resource abundance are proven central determinants for habitat use patterns (Mysterud and Ims 1998; Pellerin et al. 2010; Boyce et al. 2016; Holbrook et al. 2019), the use-available relationship is often more complex. Individuals may change their preference of a particular habitat as a function of its availability. Hence, the use of a given habitat may be conditional on the availability of that habitat (Holbrook et al. 2019). This phenomenon was first termed “functional response” by Mysterud and Ims (1998) and its importance has been demonstrated widely since (Godvik et al. 2009; Bjorneraas et al. 2012; Holbrook et al. 2017; Avgar et al. 2020; Oeser et al. 2023). In addition to adjustment in habitat use due to habitat availability, adaptive shifts in distribution may also be due to site-specific conditions in different populations (e.g., William et al. 2018). Avgar et al. (2020) showed that density dependence may provide a mechanistic explanation for the context-dependent outcomes often reported in habitat use analysis and empirical support for density-dependent habitat selection is growing (Mobæk et al. 2009; van Beest et al. 2014, 2016).
Functional responses have most commonly been studied by assessing how the use of a given habitat changes with its availability (Holbrook et al. 2019), but the number of studies linking variation in functional response to site-specific conditions is growing. Due to an expected increase in Swedish wild boar densities in areas recently recolonized, it is important to understand also how the species adjusts their space use under different population levels. Knowledge on the spatial behavior of wildlife is also crucial in order to predict, prevent, and manage diseases at the wild-domestic interface (Pascual‐rico et al. 2022, Podgorski and Smietanka 2018). Management practices aiming to mitigate human-wildlife conflicts in agricultural landscapes require science-oriented and ecologically reliable information to be effective. However, such essential information is still not very well examined, and the knowledge of context dependencies and plasticity in wild boar space use is very limited.
Aims
In this study, we used wild boar telemetry data collected between 2004 and 2021 and throughout the species’ distributional range in southern Sweden, allowing us to investigate two aspects of wild boar spatial behavior across gradients of habitat availability and population density. The aim was twofold: (1) investigate the influence of population density and availability of agricultural land on wild boar home range size, and (2) investigate the influence of population density and availability of agricultural land on wild boar use of agricultural land.
Based on the literature, we predict that wild boar home range size will (P1a) decrease at high population density (Massei et al. 1997), (P1b) the decrease in home range size due to population density will be more pronounced in females (Spitz et al. 1998; Kay et al. 2017; Schlichting et al. 2022), (P2a) home range size will decrease at high availability of agricultural land (Ford 1983), and (P2b) the decrease in home range size due to availability of agricultural land will be more pronounced in females (Podgorski 2013). In the context of landscape of fear, and the relatively high mortality risk associated with agricultural land, we predict that (P3) wild boar use of agricultural land (in relation to availability) will increase at high population density.
Material and methods
Study area
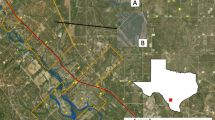
We conducted this study within the main distributional range of wild boar in Sweden (55°–60° N, 12°–18° E, Fig. 1). The landscape composition varies across the study area, with the boreal forest being the dominating habitat in the north and farmland in the south. The duration of the growing season (days with average temperature > 5 °C) varies from 190 to 220 days. Average annual precipitation varies from 500 to 1100 mm and average snow cover is between 25 and 100 days per year (SMHI 2022). There tends to be a longer period of snow-covered ground and a shorter duration of the growing season in the northern sites compared to the southern sites. Supplemental feeding is common throughout the study area, although of unknown quantities.
Hunting bag estimates for each hunting district in the study area for hunting year 2020/2021 (left), illustrating the large density variation across the study area. A hunting bag was used as a proxy for population density and assigned to each individual wild boar according to the location (hunting district) and time stamp (hunting year) of its first GPS location. The study area in southern Sweden with GPS-locations from 99 collared wild boar between 2004 and 2021 (right)
Capture, collars, and positional data
In this study, we used multiple telemetry datasets (e.g., Thurfjell et al. 2009; Muthoka et al. 2022) collected between 2004 and 2021, from 99 wild boar (16 males and 83 females) equipped with GPS-collars. The animals were immobilized with a tranquiliser gun (Dan-inject model JM, Dan-inject, Kolding, Denmark) from a vehicle on agricultural fields or close to feeding stations, or a blowpipe (Dan-inject model Blow 125) after being captured in coral traps. Wild boars were immobilized using one of the following anaesthetizing combinations and adjusted for their body size: 10 mg medetomidine + 20 mg butorphanol + 500 mg ketamine (Kreeger and Arnemo 2007; Thurfjell et al. 2009); 15–30 mg romifidine + 300 mg zolazepam-tiletamine; or 5–6 mg medetomidine + 300–400 mg zolazepam-tiletamine. After immobilization, wild boars were equipped with one of the following GPS/GSM collars: Vertex Plus 2D or 3D; Vertex Lite 2D; or GPS Pro Light 3D (Vectronic Aerospace GmbH). Telemetry data was collected or subsampled to one location every 1 h and for a maximum of 365 days to obtain homogeneity among individuals.
Population density and space use
We obtained hunting bag for each hunting district and hunting year between 2004 and 2021 from the Swedish Association for Hunting and Wildlife Management, game monitoring. Hunting bag statistics are commonly used as a proxy for assessing the relative abundance of animals in several species (Apollonio et al. 2010; Putman et al. 2011). However, since this approach has formerly been criticized for not being fully reliable (Focardi et al. 2020), we collected wild boar-vehicle collision data for each county and hunting year represented among wild boar location data and conducted a complementary calibration study. Collisions data was compiled to match hunting year (July 1 to June 30). Motivated by the strong correlation between wild boar collisions data (wild boar-vehicle collisions/km2) and hunting bag (shot wild boar/km2) (r = 0.89, df = 100, p < 0.0001; Fig. S1), also demonstrated by Massei et al. (2015) and Neumann et al. (2020), hunting bag (shot wild boar/km2) was assumed to be a reliable proxy for wild boar abundance and will hereon be referred to as “population density.” We assigned hunting bag to each individual animal according to the location (hunting district) and time stamp (hunting year) of its first GPS-location. For individuals with more than one monitoring period, we used the hunting bag for the first period.
To determine wild boar habitat use, we used CORINE land cover (CLC) data with 100 m spatial resolution, which has 44 categorized land cover and land use classes in total (European Union 2000, 2006, 2012, 2018). We reclassified the CLC classes into two habitat groups: agricultural land and others. Agricultural land was defined as all subclasses of the CLC category agricultural area (Kosztra et al. 2017): areas principally occupied by agriculture, arable land, fruit and berry plantations, pastures, meadows, and other permanent grasslands under agricultural use. All remaining land cover classes were grouped as others. We assigned land cover data to each individual according to the year for its first GPS-location and the closest corresponding year of the CLC data.
Both day and night location data from 83 female and 16 male collared wild boar (mean locations per individual = 4081, range = 304–8697) were used to compute an alpha-concave hull with a concave distance of 3 km in order to estimate home range used by each wild boar (Asaeedi et al. 2017). We estimated the use of agricultural land by calculating the proportion of true locations in agricultural land divided by the total number of true locations. We assessed available resources by generating an equal number of random locations to true locations, i.e., a ratio of 1:1, within the individual home range. This design ensured that availability was measured in the area known and in reach to each animal, and thus representing a type III analysis (Manly et al. 2002). Availability was calculated as the proportion of random locations in agricultural land divided by the total number of random locations. Seven individuals were omitted from the dataset before estimating habitat use due to a complete absence of agricultural land in their home range. The range of variables used in the statistical analysis is provided in Table 1. We used a two-stage approach (Fieberg et al. 2010): first, estimating the use of agricultural land and availability of agricultural land per individual and season; and second, on population-level assessing how the sample of use changed with the sample of availability (Holbrook et al. 2019).
As the hour of sunset is known to trigger the onset of activity (Boitani et al. 1994; Lemel et al. 2003; Thurfjell et al. 2014), each used location was assigned a daylight value (day = 1, night = 0) according to its sunrise and sunset (Schlyter 2021). We used night-time data from 76 female and 16 male collared wild boar (mean locations per individual = 1979, range = 110–4201) to analyze habitat use as wild boar are predominantly nocturnal (Boitani et al. 1994; Lemel et al. 2003; Keuling et al. 2008a; Podgorski et al. 2013; Thurfjell et al. 2014; Brivio et al. 2017).
We defined three different seasons related to the ecology of wild boar in Scandinavia: spring–early summer (reproductive season; March–June), summer–autumn (crop season; July–October), and winter (November–February; Mauget 1982; Thurfjell et al. 2014; Malmsten et al. 2017). To account for differences in habitat use across different seasons, all random locations were assigned a random season at an equal number to true locations for each season. This allowed us to test for the change in use while holding availability constant. Individuals with fewer than 10 days of data or 100 recorded locations per season were removed from the dataset before estimating habitat use.
Statistical analysis
We investigated variables influencing wild boar home range size (log10) by using a generalized linear mixed model with a Gaussian error term in the R package glmmTMB (Brooks et al. 2017). We investigated the effects of population density (shot wild boar/km2), availability (proportion of random locations in agricultural land), and sex (females coded as 0 and used as reference value, males coded as 1), including the interactions “population density * sex” and “availability * sex,” while controlling for monitoring days (log10).
We investigated variables influencing wild boar use of agricultural land by using a generalized linear mixed model with a Gaussian error term in the R package glmmTMB. We investigated the effects of population density and availability of agricultural land, including the interaction “population density * availability,” while controlling for season. Inter-sexual differences were excluded from the analysis due to the limited sample size of males. Animal ID was treated as a random factor to account for the dependency of repeated seasons within individuals (Zuur et al. 2010). For both analyses, we used Akaike information criterion, corrected for small sample sizes (AICC), to compare the relative strength of candidate models by calculating the ∆AICC (Akaike 1974), and performed AICC model selection on candidate models (Tables 2 and 4). To assess model quality and to further ensure that models fulfilled assumptions, models were screened using the package performance (Lüdecke et al. 2021). Figures were produced using the package ggplot2 (Wickham 2016). For data analysis, we used R version 4.2.2 (R Core Team 2022).
Results
Home range size
The mean estimated alpha-concave hull home range size for individuals monitored for a minimum of 14 days was for males 49.6 km2 (95% CI 24–75) and for females 17.3 km2 (95% CI 14–20). The best predictive model explaining home range size included the predictors availability of agricultural land, population density and sex, and the interaction between availability of agricultural land and sex, and the interaction between population density and sex (Table 2). We found a strong negative effect of population density but a weaker effect of availability of agricultural land on home range size (Fig. 2, Table 3). Male home ranges were in general larger than female home ranges. Both females and males showed an overall decrease in home range size with increasing population density. The effect of population density was more pronounced in male home range size than female home range size (Fig. 2). Males showed a decrease in home range size with increasing availability of agricultural land, while female home ranges increased with increasing availability of agricultural land, although the effect of availability of agricultural land was not as strong (Table 3).
Home range size for male and female wild boar in relation to population density index (shot wild boar/km2). Points represent individual observed values for males (dark gray) and females (light gray). Lines show predicted values with 95% confidence limits (shaded areas) for males (solid line) and females (dashed line). Data was collected from GPS-locations of collared wild boars (N = 99) in southern Sweden, 2004–2021
Use of agricultural land
The best predictive model explaining wild boar use of agricultural land included the predictors availability of agricultural land and population density as well as the interaction between these two variables (Table 4). We found that wild boar increased their use of agricultural land with increasing availability of agricultural land (Fig. 3, Table 5). Moreover, wild boar used agricultural land more intensively with increasing population density, and when the availability of agricultural land was high. We found an overuse of agricultural land at high wild boar densities, an underuse of agricultural land at low densities, and a proportional use of agricultural land (in relation to availability) at intermediate densities (Fig. 3). Additionally, wild boar showed a higher use of agricultural land during summer–autumn than in other seasons (Table 5).
Wild boar use of agricultural land in relation to availability of agricultural land and population density within the home range. Dashed line indicates proportional use as availability changes. Points represent observed values of each individual, repeated for several seasons (N = 211). Lines show predicted values for different population densities: min (0.0045/km2; light yellow), mean (1.42/km2; orange), and max (3.23/km2; dark brown) for season summer-autumn. Shading of points indicates the individual population density within the home range. Data was collected from GPS-locations of collared wild boars (N = 92) in southern Sweden, 2004–2021
Discussion
In this study, we show that high wild boar densities lead to an overuse of agricultural land. We found that wild boar home range size was negatively related to population density for both male and female wild boar, thus confirming our prediction that home range size will decrease at high population density (P1a). We found no support for that this negative relationship between home range size and population density should be more pronounced for females (P1b). On the contrary, we found the effect of population density to be more pronounced in male home range size. Males showed a decrease in home range size with increasing availability of agricultural land, thus partially confirming our prediction that home range size will decrease at high availability of agricultural land (P2a). However, we found no support for this in female home range size, nor for our prediction that the negative relationship between home range size and availability of agricultural land should be more pronounced for females (P2b). Wild boar adjusted their use of agricultural land in relation to availability of agricultural land and population density, supporting our prediction that wild boar will increase their use of agricultural land (in relation to availability) at high population density (P3).
Home range size
Wild boar exhibited smaller home ranges at higher densities (Fig. 2). This is in support of our prediction and suggests that the level of intraspecific competition may influence home range size. It is also in line with previous literature on wild boar spatial behavior (Massei et al. 1997), as well as in other ungulate species (roe deer: Kjellander et al. 2004; moose: van Beest et al. 2016). This knowledge can be used to inform management policies, particularly for disease control where adaptive strategies can be applied according to wildlife ecology (Smith et al. 2022). Realistic home range sizes are crucial when assessing contaminated areas or potential rate of disease transmission. Contrary to our prediction, population density was more important for male home range size than for female home range size. Keuling and Massei (2021) concluded that recreational hunting caused changes in wild boar home range size. In this study, we used hunting bag as a proxy for population density. As hunting bag also reflects hunting pressure, the more pronounced effect in male home range size could be due to the higher hunting pressure on males. The observed behavior in males may also been affected by age. Cederlund and Sand (1994) showed that size of male moose home ranges was strongly dependent on age, in contrast to that of females. Due to unknown animal age in our sample, besides being adults, we could not correct for this in the analysis.
Theory on food exploitation suggests that when food supply decreases, home range size should increase due to increased activity, a relationship that has been demonstrated in the wild boar literature (Singer et al. 1981; Boitani et al. 1994; Massei et al. 1997; Bisi et al. 2018). Although the availability of agricultural land alone does not provide a complete understanding of an area’s food supply, agricultural crops are highly nutritious and could therefore be considered a high-quality resource. We found that male home range size was negatively affected by the availability of agricultural land. For females, however, this relationship was more complex, and it is possible that the effect of population density may mask or override the effect of availability of agricultural land. Moreover, animal spatial behavior is often subjected to a food/cover trade-off (Mysterud and Ims 1998; Brown et al. 1999); thus, once the need for food is saturated, other factors, such as shelter, may become more important. In summary, our results suggest that home range size is context dependent and affected by a complex combination of multiple conditions, such as food availability, access to cover, and level of competition.
Our results support previous findings on that males of wild boar have larger home ranges than females (Kurz and Marchinton 1972; Singer et al. 1981; Boitani et al. 1994). This is a probable consequence of the wild boar social- and mating system through males trying to optimize mating opportunities by searching and visiting several spatially separated female groups for females in heat (Singer et al. 1981; Dardaillon 1988). In species where males compete for breeding females during the mating season and female movement is restricted by the limited physical capacity of their offspring during certain times of the year, this usually leads to larger home ranges in males (e.g., Kjellander et al. 2004).
Use of agricultural land
Wild boar use of agricultural land was influenced by the amount of agricultural land available (Fig. 3), confirming the importance of resource availability in habitat use patterns (Mysterud and Ims 1998; Pellerin et al. 2010; Boyce et al. 2016; Holbrook et al. 2019). Wild boar demonstrate an opportunistic feeding behavior, and its diet reflects local environmental conditions (Schley and Roper 2003), why we could expect the species to utilize resources in relation to availability and abundance. Population-density effects on spatial behavior are not well described in the wild boar literature, but have been demonstrated in several other ungulate species (e.g., moose: van Beest et al. 2014), and the importance of considering density-dependent changes in habitat selection in a theoretical framework was shown by Avgar et al. (2020). We found that the effect of population density on wild boar use of agricultural land was conditional on the availability of agricultural land. Wild boar adjusted their use of agricultural land in relation to population density, with an underuse of agricultural land at low densities and an overuse at high densities, and with a stronger effect of density at high availability of agricultural land (Fig. 3). Theory on density-dependent habitat selection predicts that animals exhibit a specialized behavior when densities are low and a more generalized behavior during high densities to reduce competition for resources. In this perspective, an increased use of a habitat type due to increased competition would suggest that it is not the most preferred habitat. Although previous studies suggest that agricultural crops are highly attractive and are selected for by wild boar (Keuling et al. 2009; Thurfjell et al. 2009; Muthoka et al. 2022), the observed response may be explained by that animal behavior is also influenced by human predation risk. Trade-offs between access to high-quality resources and risk-taking may result in adjustments in habitat use (Valeix et al. 2009; Bonnot et al. 2013). Agricultural land is due to its openness, a relatively unsafe environment. Under low competitive conditions, wild boar should prefer to forage in more concealed habitats, if available. The quality of an otherwise poor area can be artificially increased by providing supplementary food (Muthoka et al. 2022) and wild boar are known to modify their spatial behavior around feeding sites (Keuling et al. 2008b). In the Swedish context, artificial feeding sites are often placed in areas of high cover, and thus, when available, provides easy access to high energetic gain under safe conditions. Such local food sources, however, are likely to be monopolized quickly and therefore lack the capacity to sustain dense populations. Agricultural areas are harder to monopolize due to its larger size. This provides a potential explanation for the observed overuse of agricultural land at high densities: strong competitive conditions push wild boar towards high-risk forage areas, i.e., agricultural fields. A similar behavior was shown by van Beest et al. (2016) who demonstrated an increased selection for riskier habitat in elk with increasing animal density. Furthermore, we found a stronger effect of population density when availability of agricultural land was high. In this study, we focus on the use and availability of a single habitat type: agricultural land. As the selection for a given habitat is conditional on other habitat types being available to the individual, an increase in the proportional availability of agricultural land indicates a decrease in the proportional availability of other, “safer,” habitat types, and consequently, an increased competition in these areas. Competition may act on different limited resources, where food may be one and safety another. It is likely that the observed functional responses in wild boar habitat use reflect the competitive conditions for cover rather than the competitive conditions for food, or possibly a combination of both resources.
We confirmed a seasonal variation in wild boar use of agricultural land, previously demonstrated by several studies (Lemel et al. 2003; Keuling et al. 2009; Thurfjell et al. 2009). Wild boar overused agricultural land during late summer and early autumn while demonstrating a proportional use in relation to its availability during other seasons. This seasonal variation is likely due to that agricultural land offers different food items throughout the year and is most attractive during periods when crops are ripe.
Conclusions
Wild boar spatial behavior is highly context dependent and may vary considerably due to landscape characteristics and local conditions. In this study, we show that we can expect adjustments in habitat use due to both habitat availability and population density. High wild boar densities are expected to lead to disproportionally high damage levels on agricultural fields which has strong implications for management. Realistic and achievable goals increase the chances of farmers and hunters agreeing on management practices and our results imply that, in many cases, it may in fact be enough to reduce wild boar density to moderate levels to reduce crop damage. We demonstrate the importance of identifying the level of plasticity in wild boar spatial behavior due to population-level characteristics. Applying static space use models not considering density may thus lead to inaccurate inferences with ecological and management consequences. Still, this observed plasticity in wild boar use of agricultural land and its consequences for crop damages requires further research to better assess the consistency in our observed overuse at high densities. This study provided a better understanding of the drivers of wild boar distribution and space use in agro-forested mosaic landscapes and showed that improved knowledge in wildlife spatial behavior can enhance management practices by directing actions to where they operate most usefully.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Autom Control 19:716–723
Apollonio M, Andersen R, Putman R (2010) European ungulates and their management in the 21st century. Cambridge University Press, Cambridge, UK
Asaeedi S, Didehvar F, Mohades A (2017) alpha-Concave hull, a generalization of convex hull. Theoret Comput Sci 702:48–59
Avgar T, Betini GS, Fryxell JM (2020) Habitat selection patterns are density dependent under the ideal free distribution. J Anim Ecol 89:2777–2787
Bisi F, Chirichella R, Chianucci F, Von Hardenberg J, Cutini A, Martinoli A, Apollonio M (2018) Climate, tree masting and spatial behaviour in wild boar (Sus scrofa L.): insight from a long-term study. Ann For Sci 75:46
Bjorneraas K, Herfindal I, Solberg EJ, Sther BE, van Moorter B, Rolandsen CM (2012) Habitat quality influences population distribution, individual space use and functional responses in habitat selection by a large herbivore. Oecologia 168:231–243
Boitani L, Mattei L, Nonis D, Corsi F (1994) Spatial and activity patterns of wild boars in Tuscany, Italy. J Mammal 75:600–612
Bonnot N, Morellet N, Verheyden H, Cargnelutti B, Lourtet B, Klein F, Hewison AM (2013) Habitat use under predation risk: hunting, roads and human dwellings influence the spatial behaviour of roe deer. Eur J Wildl Res 59:185–193
Boyce MS, McDonald LL (1999) Relating populations to habitats using resource selection functions. Trends Ecol Evol 14:268–272
Boyce MS, Johnson CJ, Merrill EH, Nielsen SE, Solberg EJ, van Moorter B (2016) Can habitat selection predict abundance? J Anim Ecol 85:11–20
Brivio F, Grignolio S, Brogi R, Benazzi M, Bertolucci C, Apollonio M (2017) An analysis of intrinsic and extrinsic factors affecting the activity of a nocturnal species: the wild boar. Mamm Biol 84:73–81
Brogi R, Grignolio S, Brivio F, Apollonio M (2020) Protected areas as refuges for pest species? The case of wild boar. Glob Ecol Conserv 22:e00969
Brogi R, Apollonio M, Brivio F, Merli E, Grignolio S (2022) Behavioural syndromes going wild: individual risk-taking behaviours of free-ranging wild boar. Anim Behav 194:79–88
Brooks ME, Kristensen K, van Benthem K, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM (2017) GlmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R Journal 9:378–400
Brown JS, Laundré JW, Gurung M (1999) The ecology of fear: optimal foraging, game theory, and trophic interactions. J Mammal 80:385–399
Cagnacci F, Boitani L, Powell RA, Boyce MS (2010) Animal ecology meets GPS-based radiotelemetry: a perfect storm of opportunities and challenges. Philos Trans R Soc B Biol Sci 365:2157–2162
Cavazza S, Brogi R and Apollonio M (2023) Sex-specific seasonal variations of wild boar distance traveled and home range size. Curr Zool zoad021
Cederlund G, Sand H (1994) Home-range size in relation to age and sex in moose. J Mammal 75:1005–1012
European Union (2000) Copernicus Land Monitoring Service 2000. European Environment Agency (EEA). Corine Land Cover (CLC) 2000
European Union (2006) Copernicus Land Monitoring Service 2006. European Environment Agency (EEA). Corine Land Cover (CLC) 2006
European Union (2012) Copernicus Land Monitoring Service 2012. European Environment Agency (EEA). Corine Land Cover (CLC) 2012
European Union (2018) Copernicus Land Monitoring Service 2018. European Environment Agency (EEA). Corine Land Cover (CLC) 2018
Dardaillon M (1988) Wild boar social grouping and their seasonal changes in the Camargue. Zeitschrift fur Saugtierkunde 53:22–30
Ernholm L, Sternberg-Lewerin S, Ågren E, Ståhl K, Hultén C (2022) First Detection of Salmonella enterica Serovar Choleraesuis in free ranging European wild boar in Sweden. Pathogens 11:723
Fieberg J, Matthiopoulos J, Hebblewhite M, Boyce MS, Frair JL (2010) Correlation and studies of habitat selection: problem, red herring or opportunity? Philos Trans R Soc B 365:2233–2244
Focardi S, Aragno P, Montanaro P, Riga F (2006) Inter-specific competition from fallow deer Dama dama reduces habitat quality for the Italian roe deer Capreolus capreolus italicus. Ecography 29:407–417
Focardi S, La Morgia V, Montanaro P, Riga F, Calabrese A, Ronchi F, Aragno P, Scacco M, Calmanti R, Franzetti B (2020) Reliable estimates of wild boar populations by nocturnal distance sampling. Wildl Biol 4:1–15
Fonseca C, Da Silva A, Alves J, Vingada J, Soares A (2011) Reproductive performance of wild boar females in Portugal. Eur J Wildl Res 57:363–371
Ford RG (1983) Home range in a patchy environment: optimal foraging predictions. Am Zool 23:315–326
Gaynor KM, Brown JS, Middleton AD, Power ME, Brashares JS (2019) Landscapes of fear: spatial patterns of risk perception and response. Trends Ecol Evol 34:355–368
Godvik IMR, Loe LE, Vik JO, Veiberg V, Langvatn R, Mysterud A (2009) Temporal scales, trade-offs, and functional responses in red deer habitat selection. Ecology 90:699–710
Gren IM, Andersson H, Mensah J, Pettersson T (2020) Cost of wild boar to farmers in Sweden. Eur Rev Agric Econ 47:226–246
Holbrook JD, Squires JR, Olson LE, DeCesare NJ, Lawrence RL (2017) Understanding and predicting habitat for wildlife conservation: the case of Canada lynx at the range periphery. Ecosphere 8:e01939
Holbrook JD, Olson LE, DeCesare NJ, Hebblewhite M, Squires JR, Steenweg R (2019) Functional responses in habitat selection: clarifying hypotheses and interpretations. Ecol Appl 29:e01852
Ims RA (1987) Responses in spatial organization and behaviour to manipulations of the food resource in the vole Clethrionomys rufocanus. J Anim Ecol 56:585–596
Johnson DH (1980) The comparison of usage and availability measurements for evaluating resourse preference. Ecology 61:65–71
Johnson CJ, Nielsen SE, Merrill EH, McDonald TL, Boyce MS (2006) Resource selection functions based on use-availability data: theoretical motivation and evaluation methods. J Wildl Manag 70:347–357
Kay SL, Fischer JW, Monaghan AJ, Beasley JC, Boughton R, Campbell TA, Cooper SM, Ditchkoff SS, Hartley SB, Kilgo JC, Wisely SM, Wyckoff AC, VerCauteren KC, Pepin KM (2017) Quantifying drivers of wild pig movement across multiple spatial and temporal scales. Mov Ecol 5:1–15
Keuling O, Massei G (2021) Does hunting affect the behavior of wild pigs? Human-Wildlife Interactions 15:44–55
Keuling O, Stier N, Roth M (2008a) How does hunting influence activity and spatial usage in wild boar Sus scrofa L.? Eur J Wildl Res 54:729–737
Keuling O, Stier N, Roth M (2008b) Annual and seasonal space use of different age classes of female wild boar Sus scrofa L. Eur J Wildl Res 54:403–412
Keuling O, Stier N, Roth M (2009) Commuting, shifting or remaining? Different spatial utilisation patterns of wild boar Sus scrofa L. in forest and field crops during summer. Mamm Biol 74:145–152
Kjellander P, Hewison AJM, Liberg O, Angibault JM, Bideau E, Cargnelutti B (2004) Experimental evidence for density-dependence of home-range size in roe deer (Capreolus capreolus L.): a comparison of two long-term studies. Oecologia 139:478–485
Kosztra B, Büttner G, Hazeu G, Arnold S (2017) Updated CLC illustrated nomenclature guidelines. European Environment Agency, Wien, Austria 1–124
Kreeger TJ, Arnemo JM (2007) Handbook of wildlife chemical immobilization (3rd ed). Wildlife Pharmaceuticals, Fort Collins, CO, USA
Kurz JC, Marchinton RL (1972) Radiotelemetry studies of feral hogs in South Carolina. J Wildl Manag 36:1240–1248
Lemel J, Truvé J, Söderberg B (2003) Variation in ranging and activity behaviour of European wild boar Sus scrofa in Sweden. Wildl Biol 9:29–36
Lucherini M, Lovari S (1996) Habitat richness affects home range size in the red fox Vulpes vulpes. Behav Proc 36:103–105
Lüdecke D, Ben-Shachar MS, Patil I, Waggoner P, Makowski D (2021) Performance: an R package for assessment, comparison and testing of statistical models. Journal of Open Source Software 6:3139
Malmsten A, Jansson G, Lundeheim N, Dalin AM (2017) The reproductive pattern and potential of free ranging female wild boars (Sus scrofa) in Sweden. Acta Vet Scand 59:52
Malmsten A (2017) On the reproduction of female wild boar (Sus scrofa) in Sweden. Dissertation, Swedish University of Agricultural Sciences Uppsala, Sweden
Manly BF, McDonald LL, Thomas DL, McDonald TL, Erickson WP (2002) Resource selection by animals: statistical design and analysis for field studies, 2nd edn. Kluwer Academic Publishers, Dordrech, Netherlands
Massei G, Genov PV, Staines BW (1996) Diet, food availability and reproduction of wild boar in a Mediterranean coastal area. Acta Theriol 41:307–320
Massei G, Genov PV, Staines W, Gorman ML (1997) Factors influencing home range and activity of wild boar (Sus scrofa) in a Mediterranean coastal area. J Zool 242:411–423
Massei G, Kindberg J, Licoppe A, Gacic D, Sprem N, Kamler J, Baubet E, Hohmann U, Monaco A, Ozolins J, Cellina S, Podgorski T, Fonseca C, Markov N, Pokorny B, Rosell C, Nahlik A (2015) Wild boar populations up, numbers of hunters down? A review of trends and implications for Europe. Pest Manag Sci 71:492–500
Mauget R (1982) Seasonality of reproduction in the wild boar. In: Cole DJA, Foxcroft GR (eds) Control of Pig Reproduction. Butterworth Scientific, London, pp 509–526
McLoughlin PD, Ferguson SH (2000) A hierarchical pattern of limiting factors helps explain variation in home range size. Ecoscience 7:123–130
Miettinen E, Melin M, Holmala K, Meller A, Väänänen VM, Huitu O, Kunnasranta M (2023) Home ranges and movement patterns of wild boars (Sus scrofa) at the northern edge of the species’ distribution range. Mammal Res 68:611–623
Mobæk R, Mysterud A, Loe LE, Holand Ø, Austrheim G (2009) Density dependent and temporal variability in habitat selection by a large herbivore; an experimental approach. Oikos 118:209–218
Muthoka CM, Andrén H, Nyaga J, Augustsson E, Kjellander P (2022) Effect of supplemental feeding on habitat and crop selection by wild boar in Sweden. Ethol Ecol Evol 35:106–124
Mysterud A, Ims RA (1998) Functional responses in habitat use: availability influences relative use in trade-off situations. Ecology 79:1435–1441
Neumann W, Widemo F, Singh NJ, Seiler A, Cromsigt JP (2020) Strength of correlation between wildlife collision data and hunting bags varies among ungulate species and with management scale. Eur J Wildl Res 66:86
Oeser J, Heurich M, Kramer-Schadt S, Andrén H, Bagrade G, Belotti E, Bufka L, Breitenmoser-Würsten C, Černe R, Duľa M, Fuxjäger C, Gomerčić T, Jędrzejewski W, Kont R, Koubek P, Kowalczyk R, Krofel M, Krojerová-Prokešová J, Kubala J, Kusak J, Kutal M, Linnell JDC, Mattisson J, Molinari-Jobin A, Männil P, Odden J, Okarma H, Oliveira T, Pagon N, Persson J, Remm J, Schmidt K, Signer S, Tám B, Vogt K, Zimmermann F, Kuemmerle T (2023) Prerequisites for coexistence: human pressure and refuge habitat availability shape continental-scale habitat use patterns of a large carnivore. Landscape Ecol 38:1713–1728
Pascual-rico R, Acevedo P, Apollonio M, Blanco-aguiar J, Body G, Del Rio L, Ferroglio E, Gomez A, Keuling O, Plis K (2022) Wild boar ecology: a review of wild boar ecological and demographic parameters by bioregion all over Europe. EFSA J 19:1–27
Pellerin M, Calenge C, Said S, Gaillard JM, Fritz H, Duncan P, Van Laere G (2010) Habitat use by female western roe deer (Capreolus capreolus): influence of resource availability on habitat selection in two contrasting years. Can J Zool 88:1052–1062
Podgorski T, Smietanka K (2018) Do wild boar movements drive the spread of African Swine Fever? Transbound Emerg Dis 65:1588–1596
Podgorski T, Bas G, Jedrzejewska B, Sonnichsen L, Sniezko S, Jedrzejewski W, Okarma H (2013) Spatiotemporal behavioral plasticity of wild boar (Sus scrofa) under contrasting conditions of human pressure: primeval forest and metropolitan area. J Mammal 94:109–119
Podgorski T (2013) Effect of relatedness on spatial and social structure of the wild boar Sus scrofa population in Białowieża Primeval Forest. Dissertation, University of Warsaw, Faculty of Biology, Białowieża, Poland
Putman R, Watson P, Langbein J (2011) Assessing deer densities and impacts at the appropriate level for management: a review of methodologies for use beyond the site scale. Mammal Rev 41:197–219
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Said S, Tolon V, Brandt S, Baubet E (2012) Sex effect on habitat selection in response to hunting disturbance: the study of wild boar. Eur J Wildl Res 58:107–115
Schley L, Roper TJ (2003) Diet of wild boar Sus scrofa in Western Europe, with particular reference to consumption of agricultural crops. Mammal Rev 33:43–56
Schlichting PE, Boughton RK, Anderson W, Wight B, VerCauteren KC, Miller RS, Lewis JS (2022) Seasonal variation in space use and territoriality in a large mammal (Sus scrofa). Sci Rep 12:4023
Schlyter P (2021) Sunrise and sunset in different locations in Sweden. Available at: http://www.stjarnhimlen.se
Schradin C, Schmohl G, Rodel HG, Schoepf I, Treffler SM, Brenner J, Bleeker M, Schubert M, Konig B, Pillay N (2010) Female home range size is regulated by resource distribution and intraspecific competition: a long-term field study. Anim Behav 79:195–203
Singer FJ, Otto DK, Tipton AR, Hable CP (1981) Home ranges, movements, and habitat use of European wild boar in Tennessee. J Wildl Manag 45:343–353
SMHI (Swedish Meteorological and Hydrological Institute) (2022) Meteorology data. Available at: https://www.smhi.se
Smith GC, Brough T, Podgórski T, Ježek M, Šatrán P, Vaclavek P, Delahay R (2022) Defining and testing a wildlife intervention framework for exotic disease control. Ecol Solut Evid 3:e12192
Spitz F, Valet G, Lehr Brisbin Jr I (1998) Variation in body mass of wild boars from southern France. J Mammal 79:251–259
Stenberg H, Leveringhaus E, Malmsten A, Dalin AM, Postel A, Malmberg M (2021) Atypical porcine pestivirus - a widespread virus in the Swedish wild boar population. Transbound Emerg Dis 69:2349–2360
SVA (National Veterinary Institute) (2023) Wildlife. Available at: https://www.sva.se/aktuellt/pressmeddelanden/african-swine-fever-in-wild-boar-outside-fagersta-sweden/
Swedish Association for Hunting and Wildlife Management (2017) Game monitoring. Available at: https://jagareforbundet.se/vilt/viltovervakning/
Thurfjell H, Ball JP, Ahlen PA, Kornacher P, Dettki H, Sjoberg K (2009) Habitat use and spatial patterns of wild boar Sus scrofa (L.): agricultural fields and edges. Eur J Wildl Res 55:517–523
Thurfjell H, Spong G, Ericsson G (2014) Effects of weather, season, and daylight on female wild boar movement. Acta Theriol 59:467–472
Thurfjell H, Spong G, Olsson M, Ericsson G (2015) Avoidance of high traffic levels results in lower risk of wild boar-vehicle accidents. Landsc Urban Plan 133:98–104
Tolon V, Dray S, Loison A, Zeileis A, Fischer C, Baubet E (2009) Responding to spatial and temporal variations in predation risk: space use of a game species in a changing landscape of fear. Can J Zool 87:1129–1137
Truvé J, Lemel J (2003) Timing and distance of natal dispersal for wild boar Sus scrofa in Sweden. Wildl Biol 9:51–57
Tufto J, Andersen R, Linnell JDC (1996) Habitat use and ecological correlates of home range size in a small cervid: the roe deer. J Anim Ecol 65:715–724
Valeix M, Loveridge AJ, Chamaillé-Jammes S, Davidson Z, Murindagomo F, Fritz H, Macdonald DW (2009) Behavioral adjustments of African herbivores to predation risk by lions: spatiotemporal variations influence habitat use. Ecology 90:23–30
van Beest FM, McLoughlin PD, Vander Wal E, Brook RK (2014) Density-dependent habitat selection and partitioning between two sympatric ungulates. Oecologia 175:1155–1165
van Beest FM, McLoughlin PD, Mysterud A, Brook RK (2016) Functional responses in habitat selection are density dependent in a large herbivore. Ecography 39:515–523
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York. ISBN 978-3-319-24277-4
William G, Jean-Michel G, Sonia S, Christophe B, Atle M, Nicolas M, Maryline P, Clément C (2018) Same habitat types but different use: evidence of context-dependent habitat selection in roe deer across populations. Sci Rep 8:1–13
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14
Acknowledgements
To landowners and staff for letting us conduct the study on their land and helping with fieldwork and to all the fieldworkers involved in the capture and collaring of wild boar. We also thank the Swedish Association for Hunting and Wildlife Management, game monitoring, for providing the national hunting bag data. Lastly, we thank Gunnar Jansson for providing GPS-data and Manisha Bhardwaj for revised English.
Funding
Open access funding provided by Swedish University of Agricultural Sciences. P. Kjellander and E. Augustsson were supported by the Swedish Environmental Protection Agency (SEPA), Swedish University of Agricultural Sciences (SLU), Swedish Association for Hunting and Wildlife Management (Svenska Jägareförbundet), and the private foundations Marie-Claire Cronstedts Stiftelse and Karl Erik Önnesjös Stiftelse. H. Kim and S. Widgren were supported by the Swedish Research Council for Sustainable Development (FORMAS) - Grant number 2020-00733. S. Widgren and J. Malmsten were supported by the Swedish Research Council for Sustainable Development (FORMAS) - Grant number 2016-000823. J. Månsson and H. Thurfjell were supported by the Swedish Environmental Protection Agency (SEPA) and Swedish Association for Hunting and Wildlife Management (Svenska Jägareförbundet).
Author information
Authors and Affiliations
Contributions
P. Kjellander, H. Thurfjell, S. Widgren, H. Kim, and E. Augustsson developed the study design. P. Kjellander, H. Thurfjell, S. Widgren, H. Kim, J. Malmsten, and J. Månsson provided data. Data were analyzed by E. Augustsson, H. Thurfjell, H. Kim, H. Andrén, and L. Graf. E. Augustsson led the writing of the manuscript and drafted the initial version. All authors revised the manuscript, contributed with supportive information, and gave final approval for publication.
Corresponding author
Ethics declarations
Ethics approval
All wild boar captures and handling were approved by the Ethical Committee in Animal Research, Umeå, Sweden (permit A 18-04), and Uppsala, Sweden (permit C 80/9, C 77/10, C 5.2.18-2830/16, and 5.8.18-00845/2017), and in compliance with Swedish laws and regulations.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Augustsson, E., Kim, H., Andrén, H. et al. Density-dependent dinner: Wild boar overuse agricultural land at high densities. Eur J Wildl Res 70, 15 (2024). https://doi.org/10.1007/s10344-024-01766-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-024-01766-7