Abstract
Seasonal pollen allergy is a major public health concern, with many different pollen aeroallergens being present in the atmosphere at varying levels during the season. In Norway, information about spatiotemporal variation of pollen aeroallergens is currently lacking, leading to reduced ability to manage and treat seasonal allergies. Seven pollen aeroallergens (alder, hazel, willow, birch, pine, grass and mugwort) were monitored daily for 16 years from 12 regions and coalesced to create regional pollen calendars. Seasonal statistics, such as seasonal pollen integral (SPIn), onset, duration and periods of high and very high concentrations, were calculated for all pollen types and regions. High days were further modelled with SPIn in a linear regression framework to investigate the connection between the strength of the season and number of days above high pollen thresholds. The tree pollen season occurred between January and mid-July, with the pollen aeroallergens birch and pine being the most prominent in all regions. The herb pollen season was observed to occur between June and mid-August, although mugwort was almost completely absent. The grass pollen season was mostly mild on average but more severe in some regions, primarily Kristiansand. South-east regions of Oslo, Kristiansand and Lillehammer had the overall highest pollen load, while northern regions of Bodø, Tromsø and Kirkenes had the overall lowest pollen loads. SPIn and days above high pollen thresholds had positive highly significant relationships (R2 > 0.85) for all pollen types, bar mugwort. Regional pollen calendars and seasonal statistics contribute to reliable information that can be used by medical professionals to effectively and timely manage and treat seasonal pollen allergies in Norway. Further research is needed to determine sensitization profiles of pine and willow.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Seasonal allergies have long been a substantial contributing factor of decline in global public health (Licari et al., 2014), which is expected to worsen with climate change (Pacheco et al., 2021). An estimated 10—20% of the global population is suffering from some type of seasonal pollen allergy (Akdis & Agache, 2014), with the number being considerably higher in Europe, exceeding 40% in many countries (Burbach et al., 2009; D’Amato et al., 2007). On an individual level, physical symptoms range from mild irritation and discomfort to severe allergic rhinitis and reduced lung function (Bousquet et al., 2001; Idrose et al., 2021). Unfortunately, symptoms are not only physical, but mental as well, including headaches, difficulty sleeping and reduced ability to concentrate all being frequently reported (Marshall et al., 2000; Papapostolou et al., 2021). The combined effect is the reduction in quality of life in general (Meltzer, 2001; Meltzer et al., 2017; Pitt et al., 2004). Additionally, the effects on the individual contribute to an impact on a societal level due to the number of sufferers affected, causing measurable decline on societal work performance and economic productivity (Lamb et al., 2006; Zuberbier et al., 2014).
While the umbrella term seasonal pollen allergy is frequently used, individuals are likely to be sensitized to multiple specific pollen types (polysensitization) rather than one or all combined (Ciprandi & Cirillo, 2011; Ciprandi et al., 2011; Moreno et al., 2014). Grass (Poaceae), birch (Betula) and ragweed (Ambrosia) are considered the three main pollen aeroallergens globally due to their atmospheric abundance, high allergenicity and large number of sufferers (Akdis & Agache, 2014). The high public health relevance of these three allergens have contributed to extensive research into their allergenic effects and the emitting plants ecological circumstances (Frisk et al., 2023; Raith & Swoboda, 2023; Smith et al., 2013). However, many other pollen types are also present in the atmosphere, such as the tree pollen alder (Alnus), ash (Fraxinus), aspen (Populus), elm (Ulmus), hazel (Corylus), oak (Quercus), olive (Olea), plane (Platanus), pine (Pinus), spruce (Picea) and willow (Salix) and also some herbs, such as mugwort (Artemisia), nettles (Urticaceae) and plantain (Plantago), but the list is not exhaustive (Hoebeke et al., 2018; Pashley et al., 2009; Perez-Badia et al., 2010; Ruiz-Valenzuela & Aguilera, 2018; Piotrowska-Weryszko and Weryszko-Chmielewska 2014). The pollen from these plants are frequent components of the atmospheric biodiversity and combined represent a substantial proportion of the natural flora in many European ecosystems (Heinken et al., 2022; Seregin, 2010). Furthermore, many of these pollen types can provoke allergic reactions in sensitive sufferers, commonly seen for alder, oak and olive (Bernstein et al., 2021; Biedermann et al., 2019; Feo Brito et al., 2011). While for other types, there are strong suspicions of allergenicity, but the degree has not been fully explored and research is lacking, as is the case with e.g. pine and willow (Costache et al., 2021; Gastaminza et al., 2009; Mansouritorghabeh et al., 2019).
Pollen aeroallergens do not only vary temporally through the season, but spatially as well, contributing to regional phenomena based on a combined ensemble of local weather, species flowering, atmospheric conditions and climate (Antépara et al., 1995; Grundström et al., 2019; Kurganskiy et al., 2021; Skjøth et al., 2007). To account for this variation, medical practitioners, healthcare providers, local governments and individual sufferers require reliable and intuitive information on which pollen aeroallergens are likely to be presented in their surroundings and their respective seasonality. This allows them to timely and accurately warn patients and the general public for upcoming and current pollen seasons (e.g. via online platforms Lee et al., 2015; Schober et al., 2022)), to facilitate optimal treatment by prescribing required medication ahead of time (Greiner et al., 2011; Wallace et al., 2008) and to plan public policy, such as the planning of urban parks (Cariñanos et al., 2014; Suanno et al., 2021). Pollen monitoring networks and pollen forecasters alike provide this expertise by creating pollen calendars and disseminating associated information regarding seasonal onset, strength and duration (Camacho et al., 2020; Pecero-Casimiro et al., 2020; Ravindra et al., 2021). They also provide relevant information of periods of high pollen levels, during which severe symptoms are more likely to occur (Adams-Groom et al., 2020; Verstraeten et al., 2019). While many countries have highly detailed pollen calendars and seasonal statistics, this is lacking in Norway, leading to reduced ability for the public health community to provide accurate information to sufferers. The lack of research into prevalent pollen types and their spatiotemporal seasonality further enhances the need for comprehensive investigation. The aim of our study is to provide detailed and informative regional pollen calendars of allergenic pollen types and associated seasonal statistics for the public health community to assist in the management and prevention of seasonal pollen allergies.
2 Material and methods
2.1 Regional locations and pollen sampling methods
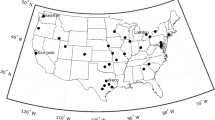
The Norwegian pollen monitoring network has developed continually since 1980, with the current modern network consisting of 12 stations (Table 1). The network has been expanded to encompass all major geographical regions (bar Svalbard in the Arctic Ocean) and most major population centres in Norway, being approximate and representative of nine of the ten largest cities (bar Fredrikstad to the south-east of Oslo) (Fig. 1). This has allowed for accurate pollen monitoring and forecasting on a regional and national level. The pollen monitoring stations representing Innlandet (Lillehammer) and Rogaland (Stavanger) counties have been relocated once and twice, respectively, due to local policies but remain representative as singular and continual datasets due to their proximity, sharing the same surrounding vegetation and climatological conditions as their previous sites.
Map over northern Europe with the 12 Norwegian pollen monitoring regions and the 15 individual sites marked (see Table 1)
Pollen was monitored daily using Hirst volumetric spore traps (Hirst, 1952) of Burkard design, was processed using standard pollen monitoring methods (Galán et al., 2014) and was counted using the latitudinal method at 400×magnification. The pollen monitoring network routinely monitors all recognizable airborne pollen types, with special interest to the main seven types with allergenic potential: alder (Alnus), hazel (Corylus), willow (Salix), birch (Betula), pine (Pinus), grass (Poaceae) and mugwort (Artemisia). All the allergenic types (bar grasses) were taxonomically identified using pollen morphology to genus level, while grasses can only be determined to family level due to lacking distinguishing pollen morphology below family level. All pollen types were monitored from the start of the growth season to the 30th of September, generally being considered the end of the growth period in Norway (Ruosteenoja et al., 2020; Skaugen & Tveito, 2004).
2.2 Seasonal pollen statistics, pollen threshold levels and statistical analyses
All available pollen data for the seven allergenic types during the years 2007—2022 (16 years) were used for the creation of the pollen calendars. Although additional data were available for many pollen stations, the analysis of recent and overlapping years between all stations allowed for representative comparisons of current data. The year 2017 was missing in the Stavanger dataset. The daily data from all years were averaged per county and used to calculate four pollen season metrics: first to last occurrence (onset and duration), the duration of the main season, high concentrations and very high concentrations. The duration of the main season was calculated using the 95% seasonal pollen integral (SPIn) method, removing the first and last 2.5% to avoid long tails of low and spurious pollen concentrations (Galán et al., 2017; Goldberg et al., 1988). High and very high pollen concentrations are threshold levels at which increasingly adverse and severe clinical health symptoms are likely to occur in sufferers.
Threshold levels vary between pollen types, with varied thresholds having been reported in the literature (de Weger et al., 2013; Galán et al., 2007; Pfaar et al., 2017; Rapiejko et al., 2007; Steckling-Muschack et al., 2021; Šukienė et al., 2021; Toro et al. 2015) and by medical organizations such as the American Academy of Allergy, Asthma and Immunology (AAAAI). Tree pollen are normally reported as requiring higher pollen concentrations to reach high and very high thresholds, while grass and other herb pollen tend to require lower pollen concentrations for the same thresholds. After reviewing the literature, we set the high and very high threshold for the tree pollen (alder, hazel, willow, birch and pine) at 80 and 150 pollen grains/m3, respectively, while we set the same for herb pollen (grass and mugwort) at 50 and 100 pollen grains/m3, respectively.
To investigate how the strength of each pollen season (SPIn) influenced the number of days above high thresholds, we constructed seven linear regression models, one of each pollen type in a combined location framework. Each linear regression model contained a polynomial function as to identify any nonlinear relationships between the variables. The strength of the linear association was assessed using adjusted R2-values. All statistical analyses were performed in the statistical software R (ver. 4.1.3.) (R Core Team 2022).
3 Results
3.1 Tree pollen seasons
3.1.1 Tree pollen general seasonality
Nationally, the general order of tree pollen appearance in the air from first to last is alder, hazel, willow, birch and pine, occurring from the end of January to the middle of July. On average, the most abundant tree pollen from highest to lowest are birch, pine, willow, alder and hazel.
3.1.2 Alder pollen statistics
The alder pollen main season onset occurs the 1st week of February on the west coast (Ørsta, Førde and Bergen), while starting later further east towards Lillehammer and Oslo around the end of February to the beginning of March, lasting until mid-April (Fig. 2). The season varies in strength over the country, being moderate in Trondheim, Førde, Lillehammer and Oslo while mostly absent in the northern regions (Table 2). The moderate regions tend to average 1 to 3 days of high pollen thresholds per year, with up to 7 to 8 days having been observed for Trondheim, Førde and Oslo (Table 3). A strong linear relationship between SPIn and days above high pollen thresholds was observed for the alder pollen type (p < 0.001, R2 = 0.909) (Fig. 3). Each model term can be viewed in the supplementary material (Supplementary Table 1).
Allergenic pollen calendars from 12 Norwegian regions. Not all seven pollen types are represented in all regions due to very low average seasonal pollen integral (SPIn) (see Table 2). Regions are arranged by latitude from north to south, and pollen types on average start in the season. Pollen concentrations for high (H) and very high (VH) thresholds for alder, hazel, willow, birch and pine are 80/150 (H/VH) and for grass and mugwort 50/100 (H/VH). For average and maximum days of high thresholds see Table 3
3.1.3 Hazel pollen statistics
Overlapping with alder is the hazel pollen season, with onset around mid-February and continuing to mid-April (Fig. 2). The season is mild on average in Norway being completely absent in northern regions and moderate only in Oslo (Table 2). Oslo is also the only region with observed high days, having on average 1 per year but reaching up to a maximum of 5 high days (Table 3). A strong linear relationship between SPIn and days above high pollen thresholds was observed for the hazel pollen type (p < 0.001, R2 = 0.903) (Fig. 3).
3.1.4 Willow pollen statistics
The willow pollen main season onset occurs as the hazel season ends in mid-April, starting about a week earlier in the far south (Stavanger and Kristiansand) and around mid-May in the far north (Kirkenes). The season lasts until the mid to end of June in most regions, extending further into August in the far north (Fig. 2). Furthermore, willow pollen can be observed in the air for a substantial amount of time, being found on average for 3 to 4 months in many regions. The season is mild in most regions, becoming moderate in Lillehammer, Oslo and Trondheim (Table 2). As with alder, the moderate regions tend to average 1 to 3 days of high pollen thresholds per year, with up to 9 and 13 having been observed in Lillehammer and Oslo, respectively (Table 3). A strong linear relationship between SPIn and days above high pollen thresholds was observed for the willow pollen type (p < 0.001, R2 = 0.851) (Fig. 3).
3.1.5 Birch pollen statistics
The birch pollen main season onset occurs during mid-April in the south, with onset as late as early to mid-May north of Trondheim and lasting until the mid- to late-May in the south while continuing to mid-June in northern regions (Fig. 2). The season tends to be moderate to severe, with the northern regions (Kirkenes, Tromsø and Bodø) having moderate seasons, Kristiansand and Ørsta having moderate-to-severe seasons and Oslo and Lillehammer on average severe seasons (Table 2). All regions have on average high threshold days, ranging from 1 day in the northern three regions to 11 in Olso and Lillehammer. The maximum number of seasonal high threshold days can exceed 20 days, with 26 and 27 having been observed in Ørsta and Lillehammer, respectively (Table 3). Days above high and very high pollen thresholds are observed frequently and continuously between late-April and mid-May in many regions, especially around Ørsta, Bergen, Oslo and Lillehammer. A strong linear relationship between SPIn and days above high pollen thresholds was observed for the birch pollen type (p < 0.001, R2 = 0.867) (Fig. 3), with a plateauing of the relationship in years of extreme SPIn.
3.1.6 Pine pollen statistics
The pine pollen main season onset occurs during mid- to late-May, with slight delays in the northern three regions. The average end date tends to vary between regions, ending in mid-June in Oslo while continuing until the beginning of July in the north (Trondheim and above) (Fig. 2). The season is usually mild in Tromsø, Bodø and Stavanger and severe in Lillehammer, Oslo, Geilo and Kristiansand, while being moderate elsewhere (Table 2). The same pattern is reflected in the number of high threshold days, with on average no high days for the mild regions, around 1 to 5 in the moderate regions and 10 to 11 in the severe regions (Table 3). The seasonal maximum number of days above high thresholds have been observed in Oslo and Geilo, of 20 and 23 days, respectively. Days above high and very high pollen thresholds occur in Oslo and Kristiansand between late-May and early-June while occurring during the entire July in Geilo. A strong linear relationship between SPIn and days above high pollen thresholds was observed for the pine pollen type (p < 0.001, R2 = 0.880) (Fig. 3), with a strong plateauing of the relationship even at lower SPIn.
3.2 Herb pollen seasons
3.2.1 Grass pollen statistics
The grass pollen season onset occurs during the beginning of June, with some delays until the end-June to early-July in the three northern regions, continuing until early to mid-August (Fig. 2). While the season duration averages around 2 months, being slightly shorter in the northern regions, it is also mild in most of Norway (Table 2). The exception is Kristiansand to the far south, where days exceeding very high thresholds are a common occurrence between the early-June and early-July. Most regions average between 0 and 2 days of high pollen thresholds per year, while Kristiansand averages just below 2 weeks of high days, with the maximum number of 26 high days having previously been observed (Table 3). Ørsta, Stavanger and Bodø have also been observed to have a noticeable number of high threshold days in some years. A strong linear relationship between SPIn and days above high pollen thresholds was observed for the grass pollen type (p < 0.001, R2 = 0.915) (Fig. 3).
3.2.2 Mugwort pollen statistics
The mugwort pollen season occurs during between mid-July and mid-August, with only four regions (Trondheim, Lillehammer, Oslo and Kristiansand) having levels high enough to establish a reliable calendar (Fig. 2). The season has been observed to be mostly absent in Norway, with pollen being observed only infrequently in regions other than the four mentioned above. In these four regions, the season is generally mild, with the highest SPIn identified in the south (Oslo and Kristiansand) (Table 2). No days exceeding the high pollen threshold have been observed during the study period in any regions (Table 3).
4 Discussion
We found that birch and pine were the pollen aeroallergens in the Norwegian atmosphere with the highest abundance, although there was substantial variation between regions. Grass, willow and alder pollen were abundant in some regions but moderate nationally, while hazel and mugwort pollen were on average low, although hazel was a more prominent aeroallergen in Oslo. Previous research on Norwegian pollen seasons is sparse (Havnen, 1973a), but two older studies have identified birch and grass as the primary pollen aeroallergens, followed by lesser abundances of alder and mugwort (Havnen, 1973b; Ramfjord, 1991). Havnen (1973b) also found significant abundances of pine pollen. This agrees with our research findings, as birch and pine were overwhelmingly the most abundant pollen but there were relevant abundances of grass pollen as well. This also corroborates the few studies available on local seasonal allergies, as birch and grass allergies have been shown to be prevalent among Norwegian children (Oftedal et al., 2007; Selnes et al., 2005). Studies have also shown a connection between seasonal allergies and asthma for the same demographic (Njå et al., 2000), and that the prevalence has increased since 1980s (Hansen et al., 2013; Hovland et al., 2014). While statistics on seasonal allergies for adults in Norway are sparse, it is likely to overlap with the general sensitization profiles found in Western Europe (Burbach et al., 2009), in which grass is the main pollen allergen, followed by birch, hazel and alder.
The relevance of birch pollen as a prominent aeroallergen in Norway is further emphasized by the allergenic prevalence in the surrounding Nordic countries (Jäger et al., 1996), such as Sweden (Lind et al., 2016), Finland (Ranta et al., 2008), Iceland (Przedpelska-Wasowicz et al., 2021) and Denmark (Ørby et al., 2015). Although the levels of birch pollen were magnitudes higher than the grass pollen in most regions, this does not mean that grass pollen is any less allergenically prevalent. Previous studies have found grass pollen to elicit strong symptoms in sensitive sufferers even when present in very low levels (Feo Brito et al., 2010; Kiotseridis et al., 2013; de Weger et al., 2011), implying that localized grass flowering can be just as harmful as regional phenomena of birch pollen. This suggests that even the low levels of grass pollen observed in most of Norway are likely still relevant for public health. Our findings further showed that Kristiansand, Lillehammer and Oslo, all regions in the south-east of the country, had the highest overall load of pollen. This likely makes them the worst regions for pollen sufferers, but longitudinal studies on allergenic symptoms would be needed to confirm this (Caruana et al., 2015; Kuehr et al., 1992; Schreurs et al., 2022). The opposite applies to Tromsø, Kirkenes and Bodø, being the northern-most regions with the lowest overall pollen load.
Pine, while not as commonly regarded as an allergenic pollen, has been identified as a potential allergen due to previous studies on pine pollen allergies (Freeman, 1993; Harris & German, 1985; Hosen, 1990) and the regular frequency of substantial pollen levels in the atmosphere (De Linares et al., 2017; López-Orozco et al., 2023; Ranta et al., 2008; Rodríguez-Rajo et al., 2003; Ruiz-Valenzuela & Aguilera, 2018; Toth et al., 2011). This is likely a combined outcome of large plant populations, high pollen production and optimal pollen release conditions of tall trees (Väli et al., 2022). Due to this, some authors have recommended further detailed investigations into pine pollen allergy (Gastaminza et al., 2009), with a possible outcome of reclassifying it as a relevant pollen allergen (Marcos et al., 2001), although overall allergenicity is likely to be low (Charpin et al., 2022). In Norway, where Scots pine (Pinus sylvestris) represents 30% of total tree volume (Statistics-Norway, 2022), it potentially contributes an allergenic effect to sufferers in the surrounding areas, even if only locally. While Monterey pine (Pinus radiata) has previously been linked as an allergenic species (Gastaminza et al., 2009; Harris & German, 1985), the allergenic status of Scots pine requires further research. As with pine, willow is not commonly regarded as a prevalent pollen aeroallergen (Weber, 2015), our results show that it can occur in relevant concentrations in certain regions, primarily Lillehammer and Oslo. A recent review has suggested that the pollen can contribute to moderate sensitization (Costache et al., 2021), showing its potential as an important aeroallergen in Norwegian regions in need of caution and further investigation.
5 Conclusion
We provide important seasonal statistics of relevant pollen aeroallergens present in the Norwegian atmosphere acquired from reliable long-term observations. We further show that SPIn is a directly contributing factor to the number of days above high pollen thresholds and periods of increased frequency of severe symptoms in sufferers. Birch and grass are likely the most prevalent pollen aeroallergens in Norway, but further research is needed to determine the extent of pine and willow sensitization. Our findings contribute practical guidelines and reference frames for medical professionals needed to mitigate and reduce the burden of seasonal pollen allergies.
References
Adams-Groom, B., Skjøth, C. A., Selby, K., Pashley, C., Satchwell, J., Head, K., & Ramsay, G. (2020). Regional calendars and seasonal statistics for the United Kingdom’s main pollen allergens. Allergy, 75(6), 1492–1494. https://doi.org/10.1111/all.14168
Akdis, C. A., & Agache, I. (2014). EAACI Global Atlas of Allergy. Zürich, Switzerland: European Academy of Allergy and Clinical Immunology.
Antépara, I., Fernández, J. C., Gamboa, P., Jauregui, I., & Miguel, F. (1995). Pollen allergy in the Bilbao area (European Atlantic seaboard climate): Pollination forecasting methods. Clinical and Experimental Allergy, 25(2), 133–140. https://doi.org/10.1111/j.1365-2222.1995.tb01018.x
Bernstein, D. I., Würtzen, P. A., DuBuske, L., Blaiss, M. S., Ellis, A. K., Weber, R. W., & Nolte, H. (2021). Allergy to oak pollen in North America. Allergy and Asthma Proceedings, 42(1), 43–54. https://doi.org/10.2500/aap.2021.42.200089
Biedermann, T., Winther, L., Till, S. J., Panzner, P., Knulst, A., & Valovirta, E. (2019). Birch pollen allergy in Europe. Allergy: European Journal of Allergy and Clinical Immunology, 74(7), 1237–1248. https://doi.org/10.1111/all.13758
Bousquet, J., Van Cauwenberge, P., & Khaltaev, N. (2001). Allergic rhinitis and its impact on asthma. The Journal of Allergy and Clinical Immunology. https://doi.org/10.1067/mai.2001.118891
Burbach, G. J., Heinzerling, L. M., Edenharter, G., Bachert, C., Bindslev-Jensen, C., Bonini, S., et al. (2009). GA2LEN skin test study II: Clinical relevance of inhalant allergen sensitizations in Europe. Allergy, 64(10), 1507–1515. https://doi.org/10.1111/j.1398-9995.2009.02089.x
Camacho, I., Caeiro, E., Nunes, C., & Morais-Almeida, M. (2020). Airborne pollen calendar of Portugal: A 15-year survey (2002–2017). Allergologia Et Immunopathologia, 48(2), 194–201. https://doi.org/10.1016/j.aller.2019.06.012
Cariñanos, P., Casares-Porcel, M., & Quesada-Rubio, J. M. (2014). Estimating the allergenic potential of urban green spaces: A case-study in Granada, Spain. Landscape and Urban Planning, 123, 134–144. https://doi.org/10.1016/j.landurbplan.2013.12.009
Caruana, E. J., Roman, M., Hernández-Sánchez, J., & Solli, P. (2015). Longitudinal studies. Journal of Thoracic Disease, 7(11), E537–E540. https://doi.org/10.3978/j.issn.2072-1439.2015.10.63
Charpin, D., Sénéchal, H., & Poncet, P. (2022). Respiratory Allergy to Conifers. In Conifers—Recent Advances (Vol. 4). IntechOpen. https://doi.org/10.5772/intechopen.101217
Ciprandi, G., & Cirillo, I. (2011). Monosensitization and polysensitization in allergic rhinitis. European Journal of Internal Medicine, 22(6), e75–e79. https://doi.org/10.1016/j.ejim.2011.05.009
Ciprandi, G., Incorvaia, C., Puccinelli, P., Soffia, S., Scurati, S., & Frati, F. (2011). Polysensitization as a challenge for the allergist: The suggestions provided by the polysensitization Impact on Allergen Immunotherapy studies. Expert Opinion on Biological Therapy, 11(6), 715–722. https://doi.org/10.1517/14712598.2011.576246
Costache, A., Berghi, O., Cergan, R., Dumitru, M., Neagos, A., Popa, L., et al. (2021). Respiratory allergies: Salicaceae sensitization (Review). Experimental and Therapeutic Medicine, 21(6), 609. https://doi.org/10.3892/etm.2021.10041
D’Amato, G., Cecchi, L., Bonini, S., Nunes, C., Annesi-Maesano, I., Behrendt, H., et al. (2007). Allergenic pollen and pollen allergy in Europe. Allergy: European Journal of Allergy and Clinical Immunology, 62(9), 976–990. https://doi.org/10.1111/j.1398-9995.2007.01393x
de Weger, L. A., Beerthuizen, T., Gast-Strookman, J. M., Plas, D. T., Terreehorst, I., Hiemstra, P. S., & Sont, J. K. (2011). Difference in symptom severity between early and late grass pollen season in patients with seasonal allergic rhinitis. Clinical and Translational Allergy, 1(1), 18. https://doi.org/10.1186/2045-7022-1-18
de Weger, L. A., Bergmann, K. C., Rantio-Lehtimäki, A., Dahl, Å., Buters, J., Déchamp, C., et al. (2013). Impact of Pollen. In M. Sofiev & K.-C. Bergmann (Eds.), Allergenic: Pollen A review of the Production, Release, Distribution and Health Impacts (Vol. 9789400748, pp. 161–215). Dordrecht: Springer Netherlands. https://doi.org/10.1007/978-94-007-4881-1_6
Feo Brito, F., Gimeno, P. M., Carnés, J., Martín, R., Fernández-Caldas, E., Lara, P., et al. (2011). Olea europaea pollen counts and aeroallergen levels predict clinical symptoms in patients allergic to olive pollen. Annals of Allergy, Asthma and Immunology, 106(2), 146–152. https://doi.org/10.1016/j.anai.2010.11.003
Feo Brito, F., Murgimeno, P., Carnés, J., Fernández-Caldas, E., Lara, P., Alonso, A. M., et al. (2010). Grass pollen, aeroallergens, and clinical symptoms in Ciudad Real, Spain. Journal of Investigational Allergology & Clinical Immunology, 20(4), 295–302.
Freeman, G. L. (1993). Pine pollen allergy in northern Arizona. Annals of Allergy, 70(6), 491–494.
Frisk, C. A., Adams-Groom, B., & Smith, M. (2023). Isolating the species element in grass pollen allergy: A review. Science of the Total Environment, 883, 163661. https://doi.org/10.1016/j.scitotenv.2023.163661
Galán, C., Ariatti, A., Bonini, M., Clot, B., Crouzy, B., Dahl, A., et al. (2017). Recommended terminology for aerobiological studies. Aerobiologia, 33(3), 293–295. https://doi.org/10.1007/s10453-017-9496-0
Galán, C., Cariñanos, P., Alcázar, P., & Domínguez, E. (2007). Spanish aerobiology network (REA): Management and quality manual. Servicio De Publicaciones De La Universidad De Córdoba, 184, 1–36.
Galán, C., Smith, M., Thibaudon, M., Frenguelli, G., Oteros, J., Gehrig, R., et al. (2014). Pollen monitoring: Minimum requirements and reproducibility of analysis. Aerobiologia, 30(4), 385–395. https://doi.org/10.1007/s10453-014-9335-5
Gastaminza, G., Lombardero, M., Bernaola, G., Antepara, I., Muñoz, D., Gamboa, P. M., et al. (2009). Allergenicity and cross-reactivity of pine pollen. Clinical & Experimental Allergy, 39(9), 1438–1446. https://doi.org/10.1111/j.1365-2222.2009.03308.x
Goldberg, C., Buch, H., Moseholm, L., & Weeke, E. R. (1988). Airborne pollen records in Denmark, 1977–1986. Grana, 27(3), 209–217. https://doi.org/10.1080/00173138809428928
Greiner, A. N., Hellings, P. W., Rotiroti, G., & Scadding, G. K. (2011). Allergic rhinitis. The Lancet, 378(9809), 2112–2122. https://doi.org/10.1016/S0140-6736(11)60130-X
Grundström, M., Adams-Groom, B., Pashley, C. H., Dahl, Å., Rasmussen, K., de Weger, L. A., et al. (2019). Oak pollen seasonality and severity across Europe and modelling the season start using a generalized phenological model. Science of the Total Environment, 663, 527–536. https://doi.org/10.1016/j.scitotenv.2019.01.212
Hansen, T. E., Evjenth, B., & Holt, J. (2013). Increasing prevalence of asthma, allergic rhinoconjunctivitis and eczema among schoolchildren: Three surveys during the period 1985–2008. Acta Paediatrica, International Journal of Paediatrics, 102(1), 47–52. https://doi.org/10.1111/apa.12030
Harris, R. M., & German, D. F. (1985). The incidence of pine pollen reactivity in an allergic atopic population. Annals of Allergy, 55(5), 678–679.
Havnen, J. (1973a). Aerobiology in Norway. Bulletins from the Ecological Research Committee, 18, 25–29.
Havnen, J. (1973b). Allergy research and aerobiology in Norway. In Bulletins from the Ecological Research Committee (NFR)—Scandinavian Aerobiology (Vol. 18, pp. 171–179). Oikos Editorial Office.
Heinken, T., Diekmann, M., Liira, J., Orczewska, A., Schmidt, M., Brunet, J., et al. (2022). The European forest plant species list (EuForPlant): Concept and applications. Journal of Vegetation Science. https://doi.org/10.1111/jvs.13132
Hirst, J. M. (1952). An automatic volumetric spore trap. Annals of Applied Biology, 39(2), 257–265. https://doi.org/10.1111/j.1744-7348.1952.tb00904.x
Hoebeke, L., Bruffaerts, N., Verstraeten, C., Delcloo, A., De Smedt, T., Packeu, A., et al. (2018). Thirty-four years of pollen monitoring: An evaluation of the temporal variation of pollen seasons in Belgium. Aerobiologia, 34(2), 139–155. https://doi.org/10.1007/s10453-017-9503-5
Hosen, H. (1990). Allergy to pine pollen (letter). Annals of Allergy, 64, 480.
Hovland, V., Riiser, A., Mowinckel, P., Carlsen, K.-H., & Carlsen, K. C. L. (2014). Asthma with allergic comorbidities in adolescence is associated with bronchial responsiveness and airways inflammation. Pediatric Allergy and Immunology, 25(4), 351–359. https://doi.org/10.1111/pai.12241
Idrose, N. S., Walters, E. H., Zhang, J., Vicendese, D., Newbigin, E. J., Douglass, J. A., et al. (2021). Outdoor pollen-related changes in lung function and markers of airway inflammation: A systematic review and meta-analysis. Clinical & Experimental Allergy, 51(5), 636–653. https://doi.org/10.1111/cea.13842
Jäger, S., Nilsson, S., Berggren, B., Pessi, A.-M., Helander, M., & Ramfjord, H. (1996). Trends of some airborne tree pollen in the Nordic countries and Austria, 1980–1993. Grana, 35(3), 171–178. https://doi.org/10.1080/00173139609429078
Kiotseridis, H., Cilio, C. M., Bjermer, L., Tunsäter, A., Jacobsson, H., & Dahl, Å. (2013). Grass pollen allergy in children and adolescents-symptoms, health related quality of life and the value of pollen prognosis. Clinical and Translational Allergy, 3(1), 19. https://doi.org/10.1186/2045-7022-3-19
Kuehr, J., Karmaus, W., Frischer, T., Hendel-Kramer, A., Weiss, S., Moseler, M., et al. (1992). Longitudinal variability of skin prick test results. Clinical and Experimental Allergy, 22(9), 839–844. https://doi.org/10.1111/j.1365-2222.1992.tb02829.x
Kurganskiy, A., Creer, S., de Vere, N., Griffith, G. W., Osborne, N. J., Wheeler, B. W., et al. (2021). Predicting the severity of the grass pollen season and the effect of climate change in Northwest Europe. Science Advances, 7(13), 1–12. https://doi.org/10.1126/sciadv.abd7658
Lamb, C. E., Ratner, P. H., Johnson, C. E., Ambegaonkar, A. J., Joshi, A. V., Day, D., et al. (2006). Economic impact of workplace productivity losses due to allergic rhinitis compared with select medical conditions in the United States from an employer perspective. Current Medical Research and Opinion, 22(6), 1203–1210. https://doi.org/10.1185/030079906X112552
Lee, K., Agrawal, A., & Choudhary, A. (2015). Mining Social Media Streams to Improve Public Health Allergy Surveillance. In Proceedings of the 2015 IEEE/ACM International Conference on Advances in Social Networks Analysis and Mining (pp. 815–822). New York, NY, USA: ACM. https://doi.org/10.1145/2808797.2808896
Licari, A., Ciprandi, G., Marseglia, A., Castagnoli, R., Barberi, S., Caimmi, S., & Marseglia, G. L. (2014). Current recommendations and emerging options for the treatment of allergic rhinitis. Expert Review of Clinical Immunology, 10(10), 1337–1347. https://doi.org/10.1586/1744666X.2014.955476
De Linares, C., Delgado, R., Aira, M. J., Alcázar, P., Alonso-Pérez, S., Boi, M., et al. (2017). Changes in the Mediterranean pine forest: Pollination patterns and annual trends of airborne pollen. Aerobiologia, 33(3), 375–391. https://doi.org/10.1007/s10453-017-9476-4
Lind, T., Ekebom, A., Alm Kübler, K., Östensson, P., Bellander, T., & Lõhmus, M. (2016). Pollen season trends (1973–2013) in Stockholm Area Sweden. PLoS ONE, 11(11), e0166887. https://doi.org/10.1371/journal.pone.0166887
López-Orozco, R., García-Mozo, H., Oteros, J., & Galán, C. (2023). Long-term trends and influence of climate and land-use changes on pollen profiles of a Mediterranean oak forest. Science of the Total Environment, 897, 165400. https://doi.org/10.1016/j.scitotenv.2023.165400
Mansouritorghabeh, H., Jabbari-Azad, F., Sankian, M., Varasteh, A., & Farid-Hosseini, R. (2019). The most common allergenic tree pollen grains in the Middle East: A narrative review. Iranian Journal of Medical Sciences, 44(2), 87–98.
Marcos, C., Rodriguez, F. J., Luna, I., Jato, V., & González, R. (2001). Pinus pollen aerobiology and clinical sensitization in northwest Spain. Annals of Allergy, Asthma & Immunology, 87(1), 39–42. https://doi.org/10.1016/S1081-1206(10)62320-4
Marshall, P. S., O’Hara, C., & Steinberg, P. (2000). Effects of seasonal allergic rhinitis on selected cognitive abilities. Annals of Allergy, Asthma & Immunology, 84(4), 403–410. https://doi.org/10.1016/S1081-1206(10)62273-9
Meltzer, E. O. (2001). Quality of life in adults and children with allergic rhinitis. Journal of Allergy and Clinical Immunology, 108(1), S45–S53. https://doi.org/10.1067/mai.2001.115566
Meltzer, E. O., Farrar, J. R., & Sennett, C. (2017). Findings from an online survey assessing the burden and management of seasonal allergic rhinoconjunctivitis in US patients. The Journal of Allergy and Clinical Immunology in: Practice, 5(3), 779-789.e6. https://doi.org/10.1016/j.jaip.2016.10.010
Moreno, C., Justicia, J. L., Quiralte, J., Moreno-Ancillo, Á., Iglesias-Cadarso, A., Torrecillas, M., et al. (2014). Olive, grass or both? Molecular diagnosis for the allergen immunotherapy selection in polysensitized pollinic patients. Allergy, 69(10), 1357–1363. https://doi.org/10.1111/all.12474
Njå, F., Røksund, O. D., Svidal, B., & Carlsen, K. (2000). Asthma and allergy among schoolchildren in a mountainous, dry, nonpolluted area in Norway. Pediatric Allergy and Immunology, 11(1), 40–48. https://doi.org/10.1034/j.1399-3038.2000.00044.x
Oftedal, B., Brunekreef, B., Nystad, W., & Nafstad, P. (2007). Residential outdoor air pollution and allergen sensitization in schoolchildren in Oslo Norway. Clinical & Experimental Allergy, 37(11), 1632–1640. https://doi.org/10.1111/j.1365-2222.2007.02823.x
Ørby, P. V., Peel, R. G., Skjøth, C., Schlünssen, V., Bønløkke, J. H., Ellermann, T., et al. (2015). An assessment of the potential for co-exposure to allergenic pollen and air pollution in Copenhagen, Denmark. Urban Climate, 14, 457–474. https://doi.org/10.1016/j.uclim.2014.12.002
Pacheco, S. E., Guidos, G., Annesi-Maesano, I., Pawankar, R., Amato, G. D. ’, Latour-Staffeld, P., et al. (2021). Climate change and global issues in allergy and immunology. Journal of Allergy and Clinical Immunology, 148(6), 1366–1377. https://doi.org/10.1016/j.jaci.2021.10.011
Papapostolou, G., Kiotseridis, H., Romberg, K., Dahl, Å., Bjermer, L., Lindgren, M., et al. (2021). Cognitive dysfunction and quality of life during pollen season in children with seasonal allergic rhinitis. Pediatric Allergy and Immunology, 32(1), 67–76. https://doi.org/10.1111/pai.13328
Pashley, C. H., Fairs, A., Edwards, R. E., Bailey, J. P., Corden, J. M., & Wardlaw, A. J. (2009). Reproducibility between counts of airborne allergenic pollen from two cities in the East Midlands. Aerobiologia, 25(4), 249–263. https://doi.org/10.1007/s10453-009-9130-x
Pecero-Casimiro, R., Maya-Manzano, J. M., Fernández-Rodríguez, S., Tormo-Molina, R., Silva-Palacios, I., Monroy-Colín, A., & Gonzalo-Garijo, Á. (2020). Pollen calendars and regional gradients as information tools in the Extremadura pollen monitoring network (SW Spain). Aerobiologia, 36(4), 731–748. https://doi.org/10.1007/s10453-020-09667-3
Perez-Badia, R., Rapp, A., Morales, C., Sardinero, S., Galan, C., & Garcia-Mozo, H. (2010). Pollen spectrum and risk of pollen allergy in central Spain. Annals of Agricultural and Environmental Medicine : AAEM, 17(1), 139–151. https://doi.org/10.1097/00005392-199503001-00016
Pfaar, O., Bastl, K., Berger, U., Buters, J., Calderon, M. A., Clot, B., et al. (2017). Defining pollen exposure times for clinical trials of allergen immunotherapy for pollen-induced rhinoconjunctivitis—an EAACI position paper. Allergy, 72(5), 713–722. https://doi.org/10.1111/all.13092
Piotrowska-Weryszko, K., & Weryszko-Chmielewska, E. (2014). Plant pollen content in the air of Lublin (central-eastern Poland) and risk of pollen allergy. Annals of Agricultural and Environmental Medicine, 21(4), 693–696. https://doi.org/10.5604/12321966.1129916
Pitt, A. D., Smith, A. F., Lindsell, L., Voon, L. W., Rose, P. W., & Bron, A. J. (2004). Economic and quality-of-life impact of seasonal allergic conjunctivitis in Oxfordshire. Ophthalmic Epidemiology, 11(1), 17–33. https://doi.org/10.1076/opep.11.1.17.26437
Przedpelska-Wasowicz, E. M., Wasowicz, P., Áskelsdóttir, A. Ó., Guðjohnsen, E. R., & Hallsdóttir, M. (2021). Characterisation of pollen seasons in Iceland based on long-term observations: 1988–2018. Aerobiologia, 37(3), 507–524. https://doi.org/10.1007/s10453-021-09701-y
Raith, M., & Swoboda, I. (2023). Birch pollen—the unpleasant herald of spring. Frontiers in Allergy, 4, 1–13. https://doi.org/10.3389/falgy.2023.1181675
Ramfjord, H. (1991). Outdoor appearance of aeroallergens in Norway. Grana, 30(1), 91–97. https://doi.org/10.1080/00173139109427778
Ranta, H., Sokol, C., Hicks, S., Heino, S., & Kubin, E. (2008). How do airborne and deposition pollen samplers reflect the atmospheric dispersal of different pollen types? An Example from Northern Finland. Grana, 47(4), 285–296. https://doi.org/10.1080/00173130802457230
Rapiejko, P., Stankiewicz, W., Szczygielski, K., & Jurkiewicz, D. (2007). Progowe stężenie pyłku roślin niezbędne do wywołania objawów alergicznych—threshold pollen count necessary to evoke allergic symptoms. Otolaryngologia Polska, 61(4), 591–594. https://doi.org/10.1016/S0030-6657(07)70491-2
Ravindra, K., Goyal, A., Kumar, S., Aggarwal, A., & Mor, S. (2021). Pollen calendar to depict seasonal periodicities of airborne pollen species in a city situated in Indo-Gangetic plain India. Atmospheric Environment, 262, 118649. https://doi.org/10.1016/j.atmosenv.2021.118649
Rodríguez-Rajo, F. J., Jato, V., & Aira, M. J. (2003). Pollen content in the atmosphere of Lugo (NW Spain) with reference to meteorological factors (1999–2001). Aerobiologia, 19(3–4), 213–225. https://doi.org/10.1023/B:AERO.0000006527.12928.26
Ruiz-Valenzuela, L., & Aguilera, F. (2018). Trends in airborne pollen and pollen-season-related features of anemophilous species in Jaen (South Spain): A 23-year perspective. Atmospheric Environment, 180, 234–243. https://doi.org/10.1016/j.atmosenv.2018.03.012
Ruosteenoja, K., Markkanen, T., & Räisänen, J. (2020). Thermal seasons in northern Europe in projected future climate. International Journal of Climatology, 40(10), 4444–4462. https://doi.org/10.1002/joc.6466
Schober, A., Tizek, L., Johansson, E. K., Ekebom, A., Wallin, J.-E., Buters, J., et al. (2022). Monitoring disease activity of pollen allergies: What crowdsourced data are telling us. World Allergy Organization Journal, 15(12), 100718. https://doi.org/10.1016/j.waojou.2022.100718
Schreurs, W., Schermer, T. R. J., Akkermans, R. P., Bischoff, E. W. M. A., & Luijks, H. D. (2022). 25-year retrospective longitudinal study on seasonal allergic rhinitis associations with air temperature in general practice. NPJ Primary Care Respiratory Medicine, 32(1), 54. https://doi.org/10.1038/s41533-022-00319-2
Selnes, A., Nystad, W., Bolle, R., & Lund, E. (2005). Diverging prevalence trends of atopic disorders in Norwegian children. Results from three crosssectional studies. Allergy: European Journal of Allergy and Clinical Immunology, 60(7), 894–899. https://doi.org/10.1111/j.1398-9995.2005.00797.x
Seregin, A. P. (2010). The most common plant species in temperate Europe based on frequency of occurrences in the national grid mapping projects. Feddes Repertorium, 121(5–6), 194–208. https://doi.org/10.1002/fedr.201000013
Skaugen, T., & Tveito, O. (2004). Growing-season and degree-day scenario in Norway for 2021–2050. Climate Research, 26(3), 221–232. https://doi.org/10.3354/cr026221
Skjøth, C. A., Sommer, J., Stach, A., Smith, M., & Brandt, J. (2007). The long-range transport of birch (Betula) pollen from Poland and Germany causes significant pre-season concentrations in Denmark. Clinical and Experimental Allergy, 37(8), 1204–1212. https://doi.org/10.1111/j.1365-2222.2007.02771.x
Smith, M., Cecchi, L., Skjøth, C. A., Karrer, G., & Šikoparija, B. (2013). Common ragweed: A threat to environmental health in Europe. Environment International, 61, 115–126. https://doi.org/10.1016/j.envint.2013.08.005
Statistics-Norway. (2022). The National Forest Inventory, Table 06289, Growing stock under bark and annual increment under bark, by species of tree (1 000 m3) 1933—2022. https://www.ssb.no/en/statbank/table/06289
Steckling-Muschack, N., Mertes, H., Mittermeier, I., Schutzmeier, P., Becker, J., Bergmann, K. C., et al. (2021). A systematic review of threshold values of pollen concentrations for symptoms of allergy. Aerobiologia, 37(3), 395–424. https://doi.org/10.1007/s10453-021-09709-4
Suanno, C., Aloisi, I., Parrotta, L., Fernández-González, D., & Del Duca, S. (2021). Allergenic risk assessment of urban parks: Towards a standard index. Environmental Research. https://doi.org/10.1016/j.envres.2021.111436
Šukienė, L., Šaulienė, I., Dubakienė, R., Rudzevičienė, O., & Daunys, G. (2021). Analysis of allergenic pollen data, focusing on a pollen load threshold statement. Aerobiologia, 37(4), 843–860. https://doi.org/10.1007/s10453-021-09727-2
Team, R. C. (2022). A Language and Environment for Statistical Computing. Version 4.1.3. Vienna, Austria.: R Foundation for Statistical Computing. http://www.r-project.org/
Toro, A. R., Córdova, J. A., Canales, M., Morales, S. R. G. E., Mardones, P. P., & Leiva, G. M. A. (2015). Trends and threshold exceedances analysis of airborne pollen concentrations in Metropolitan Santiago Chile. PLoS ONE, 10(5), e0123077. https://doi.org/10.1371/journal.pone.0123077
Toth, I., Peternel, R., Srnec, L., & Vojniković, B. (2011). Diurnal variation in airborne pollen concentrations of the selected taxa in Zagreb Croatia. Collegium Antropologicum, 35(Suppl 2), 43–50.
Verstraeten, W. W., Dujardin, S., Hoebeke, L., Bruffaerts, N., Kouznetsov, R., Dendoncker, N., et al. (2019). Spatio-temporal monitoring and modelling of birch pollen levels in Belgium. Aerobiologia, 35(4), 703–717. https://doi.org/10.1007/s10453-019-09607-w
Väli, V., Odgaard, B. V., Väli, Ü., & Poska, A. (2022). Pollen richness: A reflection of vegetation diversity or pollen-specific parameters? Vegetation History and Archaeobotany, 31(6), 611–622. https://doi.org/10.1007/s00334-022-00879-w
Wallace, D. V., Dykewicz, M. S., Bernstein, D. I., Blessing-Moore, J., Cox, L., Khan, D. A., et al. (2008). The diagnosis and management of rhinitis: An updated practice parameter. Journal of Allergy and Clinical Immunology, 122(2 SUPPL), 1–84. https://doi.org/10.1016/j.jaci.2008.06.003
Weber, R. W. (2015). Allergen of the Month—Sandbar Willow. Annals of Allergy, Asthma & Immunology, 114(2), A21. https://doi.org/10.1016/j.anai.2014.12.002
Zuberbier, T., Lötvall, J., Simoens, S., Subramanian, S. V., & Church, M. K. (2014). Economic burden of inadequate management of allergic diseases in the European Union: A GA2LEN review. Allergy: European Journal of Allergy and Clinical Immunology, 69(10), 1275–1279. https://doi.org/10.1111/all.12470
Acknowledgements
The project was funded by Foundation Dam (grant number HEL-435545). The authors acknowledge administrative support from the Norwegian Asthma and Allergy Association (NAAF).
Funding
Open access funding provided by Norwegian Institute of Bioeconomy Research.
Author information
Authors and Affiliations
Contributions
CAF was contributed to conceptualization, methodology, investigation, data curation, formal analysis, funding acquisition, supervision, writing—original draft and writing—review and editing. TEB was contributed to methodology, investigation, data curation, resources and writing—review and editing. HR was contributed to methodology, investigation, data curation, resources and writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any inappropriate financial or personal relationships that would bias or influence the research in this paper.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Frisk, C.A., Brobakk, T.E. & Ramfjord, H. Allergenic pollen seasons and regional pollen calendars for Norway. Aerobiologia 40, 145–159 (2024). https://doi.org/10.1007/s10453-023-09806-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10453-023-09806-6