Abstract
This paper investigates the challenges of implementing antibiotic stewardship – reducing and optimizing the use of antibiotics – in agricultural settings of Low- and Middle-Income Countries (LMICs) as a strategic part of addressing the global problem of antimicrobial resistance (AMR). It does so through analysis of the rapidly transforming yet diverse Kenyan poultry sector, characterized by growing commercial operations alongside traditional smallholder farming. Our research involves interviews with farmers, processors, policymakers, and agro-veterinary stores in these settings. We blend Chandler’s (2019, p. 8) notion of “antibiotics as infrastructure” with value chain frameworks to understand how the structural role of antibiotics in agriculture plays out through contrasting value chains, with different implications for stewardship. Weak regulation and intense market-based pressures are shown to drive widespread antibiotic use in poultry value chains involving small- and medium-sized farms supplying open markets. Antibiotic stewardship through adherence to agricultural and food safety standards is more evident, though unevenly observed, in value chains involving large commercial farms and processors supplying corporate buyers. Our findings reveal the complex structural roles of antibiotics in maintaining producer livelihoods in an intensely competitive and heterogeneous Kenyan poultry sector. This highlights challenges with applying global AMR policy to transforming food systems in LMICs without appropriate translation. We argue that attempts to reduce and optimize the use of antibiotics in agriculture must be informed by nuanced understandings of the roles of antibiotics in food systems in specific places including where very different scales and models of farming and value chain co-exist.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
This paper addresses the ways in which changing food systems in Low- and Middle-Income Countries (LMICs) shape antibiotic use in farming, with implications for tackling the global challenge of antimicrobial resistance (AMR). Confronting the growing problem of AMR (pathogens becoming resistant to antibiotics and other antimicrobials) is a key priority for the World Health Organization (WHO) involving agriculture as well as human healthcare.Footnote 1 Antibiotic stewardship has become central to policy designed to address the problem. In agriculture, this involves “a multifaceted approach to maintain the efficacy of antibiotics through reducing and optimizing drug use through disease prevention, biosecurity, good animal husbandry and herd health planning” (Helliwell et al. 2020, p. 447). Our paper contributes to critical research on the challenges of implementing such policy, particularly in LMICs. We focus on poultry supply chainsFootnote 2 in Kenya, where consumption of poultry meat is rising (Carron et al. 2017) and where antibiotics are used by both smallholders and larger commercial farms as part of the production process to help meet this demand (Kiambi et al. 2021; Mbugua et al. 2014). Conceptually, Clare Chandler’s (2019, p. 8) notion of “antibiotics as infrastructure”, appreciating the major role played by antibiotics in food and healthcare systems, is blended with value chain perspectives on the organizational structures and governance of supply chains to understand how the structural role of antibiotics in agriculture plays out differently through contrasting supply chains with varied implications for stewardship. This helps to foster a nuanced critique of antibiotic use in agricultural sectors of LMICs, which shed light on the problematic translation of global AMR policy in these settings.
Our paper investigates the practices and drivers of antibiotic use associated with changing markets and economic pressures across the Kenyan poultry sector, and we evaluate the challenges of reducing and optimizing antibiotic use in this context. In so doing, we advance research on how economic conditions in particular settings shape practices of antibiotic use as well as attempts to modify and reduce it (Begemann et al. 2018; Chandler 2019; Craddock and Hinchliffe 2015; Hinchliffe et al. 2018; Kirchhelle et al. 2020; Tompson et al. 2021ab). We contribute to this literature by focusing on the ways in which antibiotic use and the challenges of antibiotic stewardship are embedded in different kinds of poultry supply chains and scales and types of farming in Kenya.
In terms of defining the global health challenge the paper is addressing, “Antimicrobial resistance (AMR) is resistance of a microorganism to an antimicrobial drug to which it was previously sensitive”.Footnote 3 AMR in agriculture and food systems is a critical area of concern (Innes et al. 2021), with increasing cases reported of strains of bacteria such as Escherichia coli (E. coli), Campylobacter, Salmonella, and Enterococcus developing resistance to groups of antibiotics as one set of antimicrobials (Hudson et al. 2017; Mshana et al. 2021). There is evidence that pathogens resistant to treatment with antibiotics, and bacteria containing resistance genes, can be transmitted from animals to humans through the food animal supply chain (Food Standards Agency 2016; Graham et al. 2019), though the risk of such transmission is relatively small and uncertain (Cheng et al. 2019). Despite acknowledgement that antibiotic use is just one of many drivers of AMR (Hinchliffe et al. 2018), widespread use is nonetheless suggested to catalyse resistance and it also reduces the availability of antibiotics for humans (Buller et al. 2015; Capita and Alonso-Calleja 2013; Guetiya Wadoum 2016; O’Neill 2016; Van Boeckel et al. 2017). A wide variety of antibiotics are used in agriculture in most countries, including in poultry production, the majority of which are also needed for human healthcare (Craddock and Hincliffe 2015; Guetiya Wadoum 2016; Hudson et al. 2017; WHO 2016). Better stewardship of antibiotic use in agriculture, as defined above, is therefore viewed by global policymakers as one approach to tackling the AMR problem. This is alongside awareness raising (Alhaji et al. 2018; Manyi-Loh et al. 2018), surveillance of both AMR and antibiotic use (Agyare et al. 2018), and wider infection prevention and control (Cox et al. 2017). As such, antibiotic stewardship has formed a pivotal part of the WHO’s Global Action Plan for AMR since 2016, which cascades through National Action Plans (WHO 2016). The Kenyan National Action Plan on the Prevention and Containment of Antimicrobial Resistance (2017-22) is one example. A question raised by this is whether a one-size-fits-all approach to antibiotic stewardship across the globe is appropriate.
Pertinent to our paper is a burgeoning critical social science literature on the problems with implementing National Action Plans for AMR, including components on antibiotic stewardship. A policy “implementation challenge” (Chandler 2019. p. 2) is argued to be particularly significant for LMICs, where resources for surveillance and training are limited, and where agricultural production and antibiotic provision and use tend to be weakly regulated (Caudell et al. 2020; Hinchliffe 2022). Social scientists draw attention to the economic, political, and social structures and networks driving antibiotic use in agriculture and healthcare, including in LMICs, which can compromise stewardship initiatives (Chandler 2019; Chandler et al. 2019; Tompson 2021ab). Chandler (2019) argues that policymakers attempting to modify antibiotic use therefore need to better understand how antibiotics have become the fundamental infrastructure enabling healthcare and agricultural systems to operate and to appreciate how actors in these systems are thus compelled to rely on them in order to function. From this perspective, reducing and optimizing antibiotic use is not achieved simply by making individual actors, including those in food systems, more aware of AMR and by “responsibilizing” them (Helliwell et al. 2022, p. 14), irrespective of context. Rather, critical perspectives and improved policy on AMR need to recognize, and more sensitively respond to, the embeddedness of antibiotics in the everyday functioning and pressures of home and working lives associated with agriculture and healthcare.
Our paper shines a spotlight on antibiotic use and the challenges for antibiotic stewardship in agriculture and food systems in LMICs, focusing on the Kenyan poultry sector. Kenya represents an appropriate setting for assessing approaches to improving antibiotic stewardship in poultry value chains, as an LMIC with rapidly rising poultry meat consumption, significant commercialization of production alongside small-scale farming, and a National Action Plan for AMR at an embryonic stage in terms of its implementation. Moreover, there is evidence of AMR isolates obtained from both commercial poultry and smallholder farm poultry in Kenya (Carron et al. 2018; Kiambi et al. 2021).Footnote 4
We investigate the rapidly transforming, but also very diverse, poultry value chains in and around Nairobi, showing how antibiotics and attempts to modify and reduce their use are embedded in intensive modes of production and different kinds of supply chain. These supply chains include those involving smallholder and medium-scale production supplying informal urban markets often via intermediaries (brokers) and those involving larger commercial producers providing poultry meat for supermarket chains and the growing food service and hospitality sectors via large processors (Carron et al. 2017). While critical social science perspectives on AMR have begun to recognize how commercial pressures drive antibiotic use and compromise stewardship, including in agriculture and aquaculture (see, for example, Butcher et al. 2021 and Hinchliffe et al. 2018), the significance of different kinds of food supply chain structures tied into contrasting markets have received less attention. It is this important research gap that our paper intends to fill.
The paper proceeds, first, by outlining the research conducted in Kenya in 2021 with poultry supply chain actors and policymakers. We then summarize critical perspectives from the social sciences on both the AMR challenge and policy developed to confront it in agricultural settings, including those in LMICs. Elaborating the two conceptual framings we blend in the paper – the infrastructural role of antibiotics (Chandler 2019) and value chain perspectives (Reardon et al. 2014, 2019; Gereffi 2018; Gereffi et al. 2005) – we position our paper’s political-economic contribution. The paper then sets the scene for its analysis by providing an overview of the Kenyan National Action Plan on the Prevention and Containment of Antimicrobial Resistance (2017-22), including components on antibiotic stewardship, and transforming poultry value chains. Following this, we proceed to a three-part analysis of antibiotic use and challenges for antibiotic stewardship in diverse poultry supply chains in and around Nairobi. The first part examines antibiotic use embedded in weakly regulated antibiotic supply channels and shaped by existing norms of managing infection and product quality on a range of farm sizes, alongside different levels of awareness of AMR and antibiotic usage standards. The second part investigates the economic drivers of antibiotic use associated with competition between producers, increasing input costs, and market expectations, showing how these commercial pressures present in different ways for smallholder farms than for larger producers tied into different markets. The third part of the analysis considers questions of governance (understood as power and influence) in the different kinds of value chain in terms of opportunities for addressing the challenges of antibiotic stewardship. The analysis concurs with recent research on AMR in LMICs (Butcher et al. 2021; Hinchliffe et al. 2018) by revealing the problems of applying global policy on AMR to low resource settings in LMICs. It advances this research by considering how those problems are confronted through contrasting supply chain structures.
Methods
The paper’s analysis of antibiotic use and responses to the AMR challenge in poultry value chains in Kenya is underpinned by interview-based research conducted in and around Nairobi between March and July 2021. The project was funded by the UK’s Arts and Humanities Research Council and Global Challenges Research Fund. It was designed to explore antibiotic use and disease management on poultry (specifically chicken) farms and to make recommendations to modify these practices in light of the AMR challenge and the Kenyan National Action Plan. In line with the scale and scope of the research funding, we focused on the knowledges and practices of actors in the poultry food system rather than setting out to conduct a comprehensive survey of patterns of antibiotic use, awareness of AMR, and epidemiological analysis of AMR transmission. This is therefore a paper providing qualitative analysis of the challenges associated with implementing antibiotic stewardship policy in particular commercial settings. We thus targeted a range of actors across the spectrum of poultry value chains and in AMR policymaking.
We conducted 35 interviews overall, including 19 with chicken farmers, four with national-level AMR policymaking stakeholders, six with agro-veterinary stores (agro-vets), four with retailers, and two with large commercial processors. Participant observation on farms to engage directly with antibiotic dosing in practice was not possible given the mobility restrictions during the COVID-19 pandemic. Interviews were conducted, some on the farms studied, and were typically socially distanced in line with institutional rules and risk-assessed research procedures at the time. Barnett et al. (2011, p. 119) explain that using interviews as a research method creates “evaluative accounts” and “ordinary reasoning” on the part of participants. Whilst interviewing is therefore more directly engaged with respondents’ narratives than their day-to-day practices, we concur with Barnett et al. (2011) in recognizing the possibility of connecting them to practices. As Barnett et al. (2011, p. 120) argue, “[T]alk and action need to be understood as reflexively related in practices” (Barnett et al. 2011: 120).
Table 1 lists all anonymized interviewees by category, including interview dates. Interview codes are used to preserve anonymity and are referenced in the paper’s analysis to link findings to respondents where appropriate. Of the farms, the research team classified them into small, medium, and large depending on the number of chickens farmed and the markets supplied. Ten were classed as small commercial farms with 50 to 1,000 chickens and mainly supplying individual consumers, small retailers, and some open markets. Five were medium-sized commercial farms with between 1,000 and 10,000 chickens and also supplying individual consumers, small retailers, and open markets; and four were large commercial farms with more than 10,000 birds (15,000 to 30,000 per farm) and supplying processors, larger retailers, public institutions, and, in the case of one of these farms, open markets (i.e. open-air auctions) too.Footnote 5 Table 2 provides more detail on the farms interviewed, including their size and main markets. We identified these farms and other value chain and policymaking stakeholders through purposive sampling, and we were supported by the Kenyan Poultry Farmers Association (KEPOFA) and the Kenyan Government to gain access to them.
Interviews covered the following themes: the food system context and the position of the participants in production, the supply chain, and/or policymaking; infection prevention, disease management, and animal welfare; practices of antibiotic prescription, sale, and use, and their drivers; knowledge of AMR including policies, guidelines, and regulations designed to address it; challenges regarding antibiotic stewardship and infection prevention and management; and suggestions for improvements to policy and practice. Interviews were semi-structured and included a mix of closed and open-ended questions on these themes. Some questions were simple ones to establish the scale of production and the types of markets served, antibiotics used, and particular standards for their use (where they existed). Other questions were more open-ended in the style of “tell me about” (Valentine 2005, p. 110) to address, for example, how and why antibiotics are used in farming, the nature of challenges for reducing and optimizing their use, and awareness of AMR. By way of example, specific questions on the theme of infection prevention and animal welfare were: “What do you do to treat a sick bird?” And “Who decides the type and amount of medicine to give to each bird?”
The data underwent an initial round of open coding by two researchers using NVivo qualitative data analysis software. Data was coded according to initial broad themes (e.g. treatment of disease; experiences with AMR). Researchers then compared and discussed initial results to identify and refine possible concepts and categories. A single researcher subsequently conducted another round of open coding. This identified multiple sub-themes through a focus on instances of actions, processes, and practices reported in the data (e.g. lack of regulation and standard operating procedures; agro-vet dispensing practices). AMR risk sub-themes were mainly “etic” themes identified from literature reviews (Crang and Cook 2007, p. 139). A round of focused coding refined sub-themes. Separate codes were denoted for AMR risks applying to small/medium producers and large farms and processors in order to identify differences connected to the size of producer. Sub-themes were then categorized into four overarching themes (changes in food systems and supply chains; AMR risks; knowledgeable practices; Government interventions), which were derived from (a) extant theoretical concepts (supply chain; AMR risk) and (b) abstraction from empirically related practices and actions into higher level organizing concepts (e.g. knowledge; Government interventions). Table 3 shows the higher-level themes and sub-themes derived through the coding processes.
The approach taken thus contained elements of inductive grounded theory (Thornberg and Charmaz 2014) but used an “abductive” approach (Timmermans and Tavoy 2012, p. 167) to frame empirical data in relation to pre-existing theoretical frameworks, especially on value chain structures and AMR risk, to extend this theory. Analysis then identified connected sub-themes and used process tracing to infer relationships between and within the overarching themes. Two other researchers then reviewed the coding framework and relationships in the data and integrated them into the paper’s conceptual framework.
Critical perspectives on AMR policy and food systems in low- and middle-income countries
In this section, we first summarize the critical literature on global AMR policy and its implications for agriculture in LMICs. We then explain the two perspectives we bring together to understand the specific challenges for implementing antibiotic stewardship in diverse poultry value chains. The first is Chandler’s (2019) understanding of the structural role played by antibiotics in food and healthcare systems, and the second is frameworks for understanding value chain structures and governance (Gereffi 2018; Gereffi et al. 2005; Reardon et al. 2014, 2019).
Within critical social science literature there is growing criticism of global health policy, including for AMR, with its general focus on awareness, behavioural change, and choice shaped largely by institutions based in the global North. Hinchliffe (2022, p. 149) argues that this often ignores contextual “social, cultural and material drivers of health and illness, the social and cultural meanings and practices that relate to medicines, and the economic margins and pressures that relate to health care provision and food production”. Critical voices therefore argue for examining the structural nature of the health challenges and responses to them, including how these play out in LMICs. Clare Chandler’s anthropological research has been at the forefront of these interventions on AMR policy, and our paper works specifically with her notion of “antibiotics as infrastructure” (Chandler 2019, p. 8).
The framing of “antibiotics as infrastructure” (Chandler 2019, p. 8), informed by the work of Bowker and Star (2000), is where antibiotics, as a subset of antimicrobials, are explicitly recognized for the fundamental roles they play in enabling healthcare and food systems to function. Where AMR is flagged as a crisis in need of attention, revealing those roles constitutes “infrastructural inversion” (page 1). This makes knowable the normally hidden and assumed roles played by antibiotics in healthcare and food systems, including their use as ‘quick fixes’ in agricultural production to ensure product quality and to manage disease (Butcher et al. 2021; Denyer-Willis and Chandler 2019; Kirchhelle et al. 2020). Moreover, it helps to appreciate how livelihoods and food security can potentially be jeopardized by efforts to reduce antibiotic usage despite recognition of the potential dangers of over-use in the spheres of animal and human health. Taking this view, we thus recognize that antibiotics are central to modernizing agricultural systems in a way that can compromise attempts to modify their use as part of antibiotic stewardship initiatives (Helliwell et al. 2022). This engenders a more sympathetic critique of their use as a so-called “quick fix” to ensure product quality and manage infections where few other competitive options exist, for example affordable vaccines (Butcher et al. 2021; Denyer-Willis and Chandler 2019, p. 1; Kirchhelle et al. 2020; Tompson et al 2021a).
In the context of intensive farming, antibiotics have therefore become a part of modern agriculture and food systems, and the work of “infrastructural inversion” explicitly recognizes that. Following Chandler (2019, p. 1), Kirchhelle et al. (2020, p. 2) argue that “antibiotic policymaking should take into account antibiotics’ infrastructural relevance in medicine and food production”. Our paper contributes to these critical interventions from the social sciences by focusing in particular on the economic dimensions of the AMR challenge presented in food systems of Low- and Middle- Income Countries. We agree with Tompson et al. (2021a, p. 27), who argue that:
“Antibiotics have become a lynchpin in our political-economic systems, simply removing them is not a realistic solution. Instead, interventions are needed that address the economic and political imperatives that require resort to antibiotics as quick fixes”.
We argue for greater attention to these economic imperatives and the conditions, including drivers of product quality and value, shaping them in LMICs. This requires a firmer grasp of transforming food systems, both as broader political-economic context and by taking a closer look at supply chain dynamics and governance within these systems. To do this, we bring in our second conceptual dimension - political-economic perspectives on supply chains incorporating transforming food systems (Reardon et al. 2014; 2019) and Global Value Chains (GVCs) (Gereffi 2018; Gereffi et al. 2005). Reardon et al. (2014, p. 108) recognize how regional agri-food economies in Africa, as well as those across Asia and Latin America, comprise “an integrated system” developed from the “five interlinked transformations” of urbanization, changing diets, agri-food system transitions, rural market shifts and intensifying and more technologically driven food production. These transformations are shifting food systems in many LMICs away from traditional structures of subsistence agriculture and short supply chains to “transitional” and, they argue, ultimately “modern” stages marked by increasing commercialization, with domestic downstream buyers more prominent in the transitional phase and international market actors and standards becoming more dominant in the modern stage (Reardon et al. 2019, p. 50). However, importantly, Reardon et al. (2019) point to the current co-existence of traditional, transitional, and modern structures and food supply channels in many LMICs (see also Lee et al. 2012; Ortenblad 2021), suggesting the need to explore a diverse set of food supply chains and markets in any given setting. We thus argue for evaluation and conceptualization of these diverse food supply channels in the Kenyan poultry sector and what they mean for antibiotic use and attempts to foster better stewardship.
Our paper uses Reardon et al.’s (2019) categorization of modern, transitional, and traditional supply chain structures and recognition of their co-existence in transforming food systems to understand Kenyan poultry supply chains. Complementing this typology and enabling its analytical extension to the realm of governance and standards are insights from Gereffi et al’s (2005) GVC framework. This describes and explains key organizational forms, inter-firm linkages, and modes of coordination through which power and influence operate in the supply chain (primarily focused on a global scale, but the framework can also be applied at national and local levels) (Gereffi et al. 2005). It identifies five forms of inter-firm coordination in value chains – hierarchy, captive, relational, modular, and market. Hierarchy refers to a form of control through the vertical integration of activities on the part of a single firm. Captive value chains are those marked by high levels of power and coordination on the part of lead firm buyers. Although early GVC theories tended to see captive value chains involving low supplier capabilities, this is not always the case. Relational value chains tend to operate where complex products are traded, and they involve close buyer-supplier relations often embodying trust. Modular value chains are governed through various codes and standards and typically involve arm’s length relationships between buyers and suppliers. Market-based value chains involve governance through the market and arms-length relations between buyers and suppliers.
Bringing this categorization of governance to bear on modern, transitional, and traditional supply chains, modern structures incorporating global retailers and fast-food chains can be seen to involve more captive forms of governance where corporate buyers, and in some cases large processors, tend to manage and coordinate a consolidated supply base. There are also dimensions of modular governance at play where various quality-based and production standards are involved (Carron et al. 2017; Lee et al. 2012). Traditional structures, still dominant in many LMIC contexts (Vetter et al. 2019), are more likely to involve smallholders and small- and medium-sized commercial farms and informal markets connected through arms-length, market-based, and looser forms of coordination (Carron et al. 2017; Lee et al. 2012; Ortenblad 2021). Transitional structures in between can involve a combination of domestic retailers and open markets linked to small- and medium-sized producers through a mix of modular and market-based forms of coordination involving minimal standards and protocols.
Antibiotic use and any attempts to respond to AMR challenges, including through antibiotic stewardship, are likely to present differently in these contrasting configurations of value chain, with their different sets of actors, commercial drivers, production methods, and modes of governance. We thus argue for evaluation and conceptualization of these diverse value chains to understand how economic pressures (linked to wider drivers of value production) drive antibiotic dosing and to appreciate the challenges for reducing and optimizing antibiotic use in these different commercial contexts. Blending notions of “antibiotics as infrastructure” (Chandler 2019, p. 8) with perspectives on value chains offers a framework to grapple with antibiotics’ infrastructural roles in these economically diverse food supply chains and their implications for developing antibiotic stewardship.
Antimicrobial resistance policy and transforming poultry value chains in Kenya
The national action plan on the prevention and containment of antimicrobial resistance (2017-22)
The Kenyan National Action Plan on the Prevention and Containment of Antimicrobial Resistance was launched in 2017, aligning with the One Health approach and the UN’s Global Action Plan for AMR. It focuses on the following five strategic areas: (i) improving awareness and understanding of AMR; (ii) surveillance and research; (iii) reducing infection; (iv) optimization of antimicrobial use in human and animal health; and (v) investment in new medicines, interventions, diagnostics, and vaccinations. The Kenyan Government is responsible for executing the strategy, incorporating central roles for the Ministry of Health (MoH) and the Ministry of Agriculture, Livestock and Fisheries (MALF), as well as national and county ministries. Lead ministries have formed a National Antimicrobial Stewardship Interagency Committee (NASIC) with steering and technical committees. The first section of the plan highlights that, “… Reducing the emergence and spread of antimicrobial resistance (AMR) requires antimicrobial stewardship … and infection prevention and control … among professionals” (p. 17).
Implementation began in 2017 (ReAct 2017). However, several challenges have been reported, including limited funding and resources, limited infrastructure, information technology challenges, and what is referred to as disengagement of health workers, animal farmers, and other stakeholders (Kariuki et al. 2018; ReAct 2017). We acknowledge these challenges, but in our study, we aim to present a sympathetic critique that recognizes economic pressures in the sector and the structural role of antibiotics in addressing them. Moreover, we acknowledge that antibiotic dosing practices now deemed by global and Kenyan AMR policies to be unnecessary have only recently been curtailed or banned in the global North, for example the banning of antibiotic use for growth promotion in 2017 in the United States (Branswell 2017) and by the early 2000s in the European Union (Millet and Maertens 2011). This invites critique of global AMR policy when some of its standards and initiatives have only relatively recently been applied in higher income countries, and when it is sometimes imposed uncritically on agricultural and healthcare systems in LMICs.
Transforming poultry value chains in Kenya
Rapid urbanization, increasing economic growth, growing middle classes, and transformations in food retail landscapes are contributing to the rise in chicken meat consumption (Robinson and Pozzi 2011). The most significant increase in Kenyan chicken meat consumption is in urban areas (Interviews, Policymakers B and D, Retailer B, Processors A and B, Agro-vets B and F, Farms K, M and Q), a pattern strongly linked to the tastes and purchasing ability of the growing numbers of middle-class consumers (Bosire et al. 2016; Interviews, Policymaker B and Processor A). Regarding changing retail landscapes, supermarket chains accounted for 10% of Kenyan grocery sales, rising to 20% in cities in the mid-2010s (van Berkum et al. 2017), reflecting broader patterns of supermarket growth across LMICs (Humphrey 2007). Historically, there has been a preference for beef rather than chicken meat, but chicken meat has been growing in popularity (Carron et al. 2017), in part for nutritional reasons (Joel 2018). Chicken and other white meats are valued as being healthier than red meats, particularly by urban consumers (Ngula 2014; Interviews, Processors A and B, Farm Q), with indigenous chicken meat tending to be viewed more as a necessity than as a luxury food product (Bett et al. 2012). Alongside this, fast food in urban areas is dominated by chicken meat, spurred by aggressive marketing and affordable pricing.
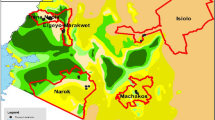
Illustrating the influence of these changing poultry consumption patterns on production systems, Nairobi and its environs (as well as other major cities) is a centre with a high concentration of commercial poultry producers because they are supplying these proximate urban markets (Carron et al. 2017; Nyaga 2007; van Berkum et al. 2017). Overall, in 2010, Kenya’s annual poultry meat production was about 20,000 metric tonnes, with around 90% consumed domestically (Ngula 2014). This figure is predicted to grow to approximately 48,000 metric tonnes by 2030.Footnote 6 Refrigeration and distribution systems from farms, via slaughterhouses, to markets have improved, easing transportation of large volumes of product to supermarkets and food service outlets. Such a significant poultry industry, with a diverse range of value chains, has grown in Kenya to both provision and shape the aforementioned markets in cities (Vosevwa 2014). Figure 1 depicts this diversity found in and around Nairobi through our research.
Carron et al. (2017) note a shift to intensive broiler production systems and increasingly industrialized systems of slaughter and processing. Such transforming systems mirror, and are influenced by, transformations of the industry in advanced capitalist economies through the twentieth century (Watts 2014). It is presumed that since antibiotics have been a key part of that globalizing agricultural industrialization (Kirchhelle 2018), it would also be the case in Kenya. Modernized, medium-sized commercial producers farming around 1,000 to 10,000 chickens are more directly linked into formal urban markets. At the larger scale, modern commercial producers can be vertically integrated into hierarchical production and distribution systems, or they can be part of captive value chains mainly to supply the rapidly expanding supermarket and fast-food sectors (Shaw et al. 2019). Larger commercial operations, with 10,000 to hundreds of thousands of birds live in purpose-built sheds and partially automated and climate-controlled environments (Shaw et al. 2019). Commercial and hybrid breeds of chicken are produced on these large farms, the latter by two dominant corporations, ‘Kenchic’ and ‘Kukuchick’ who have introduced parent breed stock from the Cobb 500, the fast-growing, low-input bird, dominant globally across large-scale broiler systems (Carron et al. 2017; Shaw et al. 2019).
Alongside these commercial transformations, however, traditional small-scale production continues, supplying informal markets via intermediaries, as well as farming on a subsistence basis (Abu Hatab et al. 2019; Carron et al. 2017; McCarron et al. 2015). These small commercial farms operating with fewer than 1,000 birds are generally not well integrated into value chains and are shaped largely by market-based forms of governance. Indigenous chickens are widely kept in Kenya by rural and urban households farming on a small scale (Magothe et al. 2012). Some farmers report that they keep indigenous kiyenyeji, but that these are “improved” kienyeji (improved through breeding programmes) (Interviews, Farms A, C, D, E, K and L). Domestic consumption and continued tastes for indigenous chicken meat influence this (Bett et al. 2012; Kagira and Kanyari 2010), and significant production still also comes from households deploying low-technology farming methods on plots of land smaller than two hectares (Njisane et al. 2020; Shaw et al. 2019). The risk of disease is argued to be high for these smallholders (Gillespie and van den Bold 2017; Njisane et al. 2020), who are reported to purchase drugs from agro-veterinary shops themselves as well as looking to vets and government for support (Kagira and Kanyari, 2010; Kiambi et al. 2021).
The challenges of antibiotic stewardship in diverse poultry value chains
Antibiotic use on farms, standards for stewardship, and awareness of AMR
In this section, we conduct some “infrastructural inversion” work advocated by Chandler (2019, p. 1) in terms of rendering knowable the structural role of antibiotics in Kenyan poultry production in order to grasp the challenges posed for antibiotic stewardship. This first entails showing when and where antibiotic use with farmed poultry is routine. Alongside this, we capture awareness of the need to address the AMR challenge through antibiotic stewardship. The picture in traditional value chains is contrasted with that for transitional and modern value chains.
The case of traditional value chains and small and medium-sized farms
Only two of the medium-scale farm respondents evidenced knowledge of AMR but they did not consider AMR an on-farm risk (Interviews, Farms K and O); this was from a total of fifteen small and medium sized farmers interviewed. This suggests that AMR, when partially known, is often disconnected from the day-to-day biosecurity practices, antibiotic dosing of poultry, and the pressures, disease or otherwise, of making a livelihood from poultry farming; and it certainly is not an identifiable challenge within poultry farming currently. This concurs with the observations of Helliwell et al. (2019) and Rees et al. (2021) in the case of UK dairy farming, where farmers tend not to recognize AMR as a problem directly concerning them. Rather, they contend with the more immediate issues of managing animal disease and maintaining product quality in order to ensure their farms’ economic viability. We return to this issue of what it is to know about AMR in the later sections of the paper on governance and opportunities for developing more effective antibiotic stewardship.
Our interviewees reported preventative or prophylactic antibiotic dosing at specific times in the chicken’s life, especially when they first arrive at the farm, and preventative treating of the whole flock when sickness is detected in just one or a few birds (Interviews, Farms D, E, F, O and N; Agro-Vets, A and B; Processor B; Policymakers B and C). Agro-vet expertise is critical for targeted antibiotic dosing of sick chicken with a known bacterial disease. However, the cost of agro-vet expertise can be prohibitive when budgeting to grow a flock in the context of livelihoods with slim margins that are crucial to the community’s food security. Consequently, farmers regularly engage in disease diagnosis and identifying suitable treatment (Interviews, Farms A, B, D, J, and K), often in consultation with a local peer farmer (Policymaker B; Hinchliffe et al. 2018). Where inexperience lies with various diseases, this can culminate in mis- or un-diagnosis and consequent antibiotic dosing might be unnecessary, for example for chickens with a viral disease. Indeed, agro-vets said that farmers usually approach them as a last resort or in emergencies after they have tried medicating the chickens themselves or after they have sought advice from other farmers and nothing seemed to work (Interviews, Agro-Vets A and D). Some small-scale farmers reported that they relied on agro-vets offering a verbal diagnosis, based solely on a farmer’s description of disease symptoms to the agro-vets (Interviews, Farms A, B, C, D, F, I, and K; see also Caudell et al. 2020 and Muloi et al. 2019). Farmers reported that they sometimes were sold multiple types of antibiotics by agro-vets, to counter one being ineffective (Interviews, Farmers A, C, I, J, K, N), a practice also recounted by agro-vets themselves (Interviews, Agro-Vets B, E and F) and Policymaker A.
With or without agro-vet expertise, chickens that are not getting better following antibiotic treatment can be dosed with either more of the same drug, or multiple others were tried. Equally, the practice of visiting different agro-veterinary retailers, known as polypharmacy, can entail lost veterinary records to trace previous antibiotic prescribing of a flock, with risk of repeating ineffective antibiotic treatment. One policymaker (Interview, Policymaker C) was aware of poultry given feed or growth promoting products labelled or mislabelled as containing, or not, antibiotics. With antibiotic withdrawal rules, farmers generally appeared to be aware of the need to observe withdrawal periods, but other stakeholders claimed non-observance was a common practice (Interviews, Agro-vets A, B, and E; Processor A; Policymaker A).
Respondents representing small and medium-scale farms as part of traditional value chains also discussed wider farming practices compromising effective infection prevention and disease management and therefore raising the risk of needing to dose birds with antibiotics. These practices included: on-farm slaughtering, including by brokers (Interviews, Farms E, F, I, J, K, M and Agro-Vet E; McCarron et al. 2015); challenges with animal husbandry (Interviews, Agro-Vets B, E and F; Policymakers A, B, and C); inappropriate housing structures for birds often living at high stocking density (Interviews, Farms B, C, E, G, K, Agro-Vet B, and Policymaker B); and procurement of birds from untrusted sources, potentially harbouring sickness, and so less suited to intensive, fast-growing meat production (Interviews, Farms A and B and Agro-Vet B).
The case of transitional and modern value chains involving processors and larger farms
Awareness of AMR as a health challenge involving agriculture was far greater amongst larger commercial farms and other stakeholders within modern and transitional value chains. The processors and their contract farmers interviewed all demonstrated awareness of AMR issues and risks. Returning to the insights of Helliwell et al. (2019) and Rees at al. (2021) and contrasting this position with smaller farmers being unaware of AMR, the ways in which larger contract farmers and those representing processors have come to know about AMR is largely through the antibiotic usage and stewardship protocols embodied in the food safety standards their buyers require them to follow. We acknowledge that our sample is small, but this also reflects a small number of large and highly influential processing companies in the area. Processors indicated a significant level of quality control and antibiotic use protocols at work in their supply chains (Interviews, Processors A and B), as this interviewee explains:
“Our policy and food safety is to prevent as much as possible antibiotic use, antimicrobial use. So trained people have to train well the farmers on other good manufacturing practices without using antibiotics.” (Interview, Processor A).
Contract farmers explicitly confirmed that quality control standards are higher in processor-led supply chains than for small-scale production for informal markets (Interviews, Farms P and S). The interviewee representing Farm P reflects explicitly on these regulatory contrasts between processor-led production and production for the open markets:
“When you have the contract, the major players regulate their [supply chains] by monitoring. They have their field officers, they give you the standard operating procedures, so you follow to the letter if you are dealing with the formal, I mean, like when you are doing the contract [for processors], they monitor closely. But otherwise, if you’re doing for the informal market, you produce and go to the market with your products and that’s it.“
This concurs with observations by Carron et al. (2017), who demonstrate some of the food safety standards adhered to by large poultry producers, including ISO 22,000 − 2005 and Hazard Analysis and Critical Control Points (HACCP), which importantly incorporate guidelines for reducing antibiotic use. Processors explain that they aim for their contract farmers to adhere to international standards and auditing in terms of antibiotic use, associated with Reardon et al.’s (2019) modern food systems and the combination of captive and modular forms of governance they incorporate. Corporate customers also aim to audit farms to demonstrate adherence to international standards, influenced by some of the final buyers being international brand name companies such as Kentucky Fried Chicken and Carrefour, both customers of one of our large participant farms.
Quality control and standards restricting and guiding antibiotic dosing of poultry along the lines of antibiotic stewardship protocol thus appear to be present in both integrated/own farms (hierarchical modes of governance, in GVC terms) and contract farms supplying processor-led (captive) value chains in a way that is absent from traditional value chains that are less coordinated and are governed more immediately by cost-based concerns than by codes and standards. Some processors had contract farmers who had measures in place to monitor their use of antibiotics (Interviews, Processors A and B and Farm Q). Despite these measures, however, a processor commented that non-observance of an antibiotic withdrawal period prior to the supply of chicken can still be evident: “Even our own contract farmers, despite putting [in place] all those measures, you will take a bird and when you test, you find tetracycline” (Interview, Processor A). There does not therefore appear to be comprehensive control of processors’ supply chains. Unregulated antibiotic purchase from agro-vets was also reported within parts of processor-led value chains that were supplying domestic retailers and public sector organizations, especially in less common cases where non-contract farmers are used (Interviews, Processor A and Farm P). Contract and processor-owned farms can therefore share similar challenges to small and medium-sized farms that lead to resort to additional antibiotic dosing, at times, in their poultry management system.
Our analysis of interviews with a range of supply chain actors demonstrates mixed awareness of AMR as a risk (disconnection from AMR in traditional value chains and connection largely through modular forms of governance (codes and standards) in the case of transitional and modern value chains). Respondents also reflected on significant unregulated dosing of poultry including unregulated agro-vet dispensing practices, and limited enforcement of policies and standards. This leads to widespread and weakly regulated dosing of poultry with antibiotics reported on both larger commercial as well as smallholder farms in order to manage infection, as well as for growth promotion (Kiambi et al. 2021; Mbugua et al. 2014; Odede 2016; Shaw et al. 2019).Footnote 7 More specifically, we show that small and medium-sized farms associated with traditional value chains demonstrate more weakly regulated (in some cases, unregulated) drug use and animal husbandry practices compared with larger farms supplying commercial processors, retailers, and the food service sector where various production standards are in evidence, if not wholly adhered to.
Economic pressures in value chains and their roles in driving antibiotic use
Having discussed contemporary practices of antibiotic dosing of poultry, including use beyond the targeted treatment of diagnosed bacterial infections, we focus on the effects of variable input costs and quality, market-pricing, and pressures on the funding for infrastructure. This is to demonstrate how the interactions between bird breeds, which have different growth rates and disease susceptibilities, and which are variously housed, create poultry products that are priced and valued differently for farmers trying to make a living in the context of intense competition (Interviews, Farms C, F, J, and O; Policymaker B). Competition is particularly acute among small farms as part of traditional value chains. We show how economic unpredictability relating to various contingencies around inputs leads to the antibiotic dosing of poultry by farmers as a hopeful aid to address business challenges. This appears to be more significant in the market-driven value chains involving small and medium sized farms supplying open markets via brokers than in processor-driven value chains where larger commercial farms are tied into more captive value chains and longer-term supply relationships. In so doing, we move to a second step in our “infrastructural inversion” work (Chandler 2019, p. 1), deepening the commercial explanation for why dosing birds with antibiotics is so common across Kenyan poultry farming despite the need to address the challenge of AMR through reducing the practice.
Dynamic input costs and intense competition between small-scale farmers, along with a disadvantageous position of farmers in relation to brokers, constrain farmers’ ability to engage in antibiotic stewardship. Small- and medium-sized farms using market intermediaries are under pressure to accept low prices from brokers (Interviews, Farms A, B, D, F, J, K, M, N, O and P), as this farmer articulates:
“The major challenge is actually the markets, the fluctuation of the market, because you can produce and find that there is oversupply at one point. Especially in the informal markets, we get oversupply, the prices go so low, and you can’t even break even.” (Interview, Farm P).
Interviewees discussed an increase in the number of poultry farms in and around Nairobi, triggered by rising urban poultry meat consumption, which has intensified small farm price pressures. In addition, inconsistent availability of some raw materials for feed results in fluctuating prices that often lead to the purchase of low quality or inadequate feeds resulting in deficiency in nutritional requirements for the growing chicken, which in turn raises the risk of disease that can then lead to treatment with antibiotics. Farmers indicated that they felt squeezed by rising feed costs (Interviews, Farms A, B, D, F, J, K, M, N, and O). Notably, small-scale farmers raised this issue most often, illustrated by this farmer’s reflection:
“When I started, we used to have quality products in the market. By quality products, I mean from the side of the feeds, which translates to the [chicken as] products that are going to the market. But now recently the quality of the feeds has really gone down, and it has affected the productivity“(Interview, Farm K).
Given the high cost of feed, getting a flock to market earlier by dosing with antibiotics can potentially represent a significant saving. Farmers also indicated that chicks were expensive and that farmers can therefore be forced to purchase slow growing and thus cheaper bird breeds (Interviews, Farms F, J, and O). The following small-scale farmer reflects on the rising costs and declining quality of inputs:
“The chick itself has become very expensive because for the last two years we have been buying the chick at 50 shillings (~$0.5), but now it has reached 80 shillings, then the food, for instance like the starter feeds, we used to buy a sack at 2,800 shillings and now it is 3,300 shillings, that is, 50 kilograms” (Interview, Farm F).
This agro-vet describes the market instability in terms of the price for which farmers can sell their broilers:
“With this free market when there is an abundance in terms of supply, the price is the driver but there are no guidelines in terms of … how the prices are supposed to be structured, so you realize farmers are actually left in a state of limbo or a state of mercy depending on the industry” (Interview, Agro-Vet E).
Our research suggested that larger farms tied into contracts with processors as part of captive value chains appear to have more secure and stable access to markets and pricing, demonstrating a contrast in commercial pressures and experiences, particularly between traditional and modern value chains and at a time of crisis through the COVID-19 pandemic and resulting economic impacts. Adhering to the food safety and agricultural standards incorporating antibiotic usage protocols has in fact become a part of maintaining access to those commercial markets.
Access to capital has also led to compromised animal husbandry practices, including inadequate poultry housing; this issue was raised by farmers of varying sizes from smallholders to large contractors (Interviews, Farms E, K, O, and Q), and was confirmed by processors (Interviews, Processors A and B). Whilst some farmers have invested in poultry housing with the aid of SACCOs (Savings and Credit Co-Operatives), financial institutions, perhaps for reasons associated with market instability described above, see poultry as a risky investment and are reluctant to support it (Interview, Farm L). Lack of capital results in converting existing farming structures to housing for poultry, as opposed to constructing purpose-built housing (interviews with Farms C and O). This can result in poorer biosecurity practices, which increase the spread of disease and lead farmers to resort to antibiotic use. Such a scenario is indicative of a traditional value chain influenced by intermediaries and shaped largely by market-based forms of coordination, to use GVC terms, driven by imperatives to minimize costs, where small and medium sized farms must continually compete to get their chickens to open markets. One medium-sized farmer explicitly linked difficulties with accessing open-air auctions to a dependence on brokers (Interview, Farm O). Broker activity can limit farmer profit, which can affect disease management investment and, by extension, antibiotic use.
Respondents suggested that smaller farmers, in particular, are tempted to dose birds with antibiotics to accelerate the growth of a bird with lower input costs (shorter production time with housing, feed, and labour saving), as short cuts to reach increasingly competitive and at times saturated markets (Interviews, Farms B, K, Agro-Vet E, Processor A, and Policymaker B). This suggests that antibiotics are known as cheap, available, and effective aids for smaller farmers striving for a profit and reflects literature evaluating antibiotic use as a “quick fix” (Butcher et al. 2021; Chandler 2019; Denyer-Willis and Chandler 2019, p. 1; Kirchhelle et al. 2020; Tompson et al. 2021). We support a sympathetic reading of such antibiotic use, given the role it plays in accessing markets and securing livelihoods, and the fact that it has only relatively recently been addressed in wealthier economies of the global North.
Antibiotic stewardship: addressing the policy “implementation challenge”
In its sections on optimizing and reducing the use of antimicrobials including antibiotics, the Kenyan National Action Plan mainly refers to implementing existing international standards and guidelines concerning product quality and food safety, which include infection prevention measures helping to reduce the risk of disease and AMR. These standards include ISO 22,000 − 2005 and HACCP, noted earlier. International guidelines covering the use of veterinary antimicrobials developed by OIE (World Organization for Animal Health) and Codex Alimentarius (the United Nations’ (UN’s) global food standards body)Footnote 8 are also mentioned. Against this backdrop, the plan suggests that, “Appropriate use of veterinary antimicrobials will be ensured through various regulatory systems based on applicable laws” (p. 33). Our research suggests, however, that the policy “implementation challenge” broadly identified by Chandler (2019, p. 2) is evident in the Kenyan poultry sector, particularly in the case of traditional value chains involving small- and medium-sized farms. Whilst standards are present in modern and some transitional value chains we studied, and appear to be partially applied, they are rarely finding their way into traditional value chains involving small- and medium-sized farms.
We argue that there are significant shortcomings in simply taking standards and guidelines derived from international policy and aiming to roll them out through the Kenyan poultry sector without sensitivity to the infrastructural role of antibiotics in the sector and the different governance structures of contrasting types of value chain. The Kenyan National Action Plan does not attend to the national and local specificities of how food supply chains operate, which our findings indicate challenge the implementation of international standards and guidelines. Whilst it refers to international product quality and food safety guidelines addressing the use of veterinary antimicrobials including antibiotics, nothing is said about how to address market price instability, low-quality feed, bird supply, or limited agro-vet access, which small- and medium-scale farmers in traditional and transitional value chains report as challenges and drivers of antibiotic use.Footnote 9 This is significant to note and reflects a tendency for policy to transfer responsibility to farmers and vets rather than addressing the systems leading to antibiotic use.
Policymakers point to institutional constraints in terms of a lack of budget allocation for addressing AMR and a lack of coordination between various agencies and organizations, including international organizations (Interviews, Policymakers B and C). Policymaker B explained that international and national government bodies are now developing manuals so that training and education are developed by government rather than commercial bodies. In terms of reaching farms and shaping farming practice, however, this was limited at the time of our study. Poultry farmers recognize that there is a lot more they could know about AMR and how to reduce and optimize antibiotic use (Interviews, Farms A, C, I, J, K, M, N, and O), with a more significant role to be played by extension services for smaller scale farmers who are not contracted by processors and who therefore cannot learn through that supply relationship (Interview, Farm P). A key policymaker (Interview, Policymaker B) and retailers (Interviews, Retailers A, B, and D) supported that view, but from experience, processors also identified the need to include incentivization for farms to participate (Interviews, Processors A and B). In parallel, agro-vets argued for government investment in both disease diagnostic services (Interview, Agrovet F) and improvements in their own training (Interviews, Agro-vets B, E, and F).
We suggest that improvements to antibiotic stewardship focused on education and training concerning AMR and its complex connection to antibiotic use are likely to be limited on their own in reducing antibiotic dosing of poultry across the whole sector. They are most likely to be effective in modern, and some transitional, value chains where a canvas of codes and protocols already exists and on which further training and regulatory efforts can be built. As we have noted, it is really through the codes and standards themselves and guidance on their implementation that the risks of AMR are addressed rather than through detailed knowledge of AMR. It is more challenging to see how manuals and standards would be operationalized through traditional value chains where both regulatory apparatus and educational opportunities are minimal at present, though this could be the subject of further research and policy intervention.
Beyond education, training, investment, and tighter regulation of antibiotic dosing, there is critical need to address the economic pressures, linked as they are to wider drivers of product value creation, that underpin antibiotic use. One policymaker contrasted the largely deregulated poultry sector with the more tightly regulated dairy industry:
“We don’t have a structure in the poultry industry and by this I mean if you look at the dairy industry, you have a dairy board, you have something that regulates it. But the poultry industry has nobody that regulates it.“ (Interview, Policymaker B).
Farmers agreed with this observation and articulated a preference for the government to implement a controlled market in the poultry industry with a regulated market structure incorporating price controls along with quality checks of poultry products. This issue was especially pressing for small- and medium-scale farmers (Interviews, Farms A, B, C, D, E, I, J, K, M, O), but was also mentioned by a large producer (Interviews, Farm R). It links to the evidence presented above, suggesting that farmers find it difficult to market products in an environment of high competition, fluctuating prices, and the perceived disadvantages of working through brokers. Interviewees similarly called for more market regulation of input costs (Interviews, Farms D, E, F, I, J, K, M, O P, and Q; Agro-vets B and E; Retailers A and B) and access to credit (Interview, Farm Q), also discussed above. Importantly, these respondents voice a desire for regulation that goes beyond the enforcement of standards on access and use of antibiotics in poultry farming, as identified in Nigeria (Alhaji et al. 2018) and Ghana (Boamah et al. 2016), to a tighter regulation of market structures themselves. With this might come a reduction in at least some of the commercial pressures driving the antibiotic dosing of poultry, as well as potentially providing more regulatory apparatus through which to run AMR policy and guidance on antibiotic stewardship.
In a different vein, more informal ways to reduce and optimize antibiotic use in the case of small and medium-sized farms have been argued for in the critical social science literature, including working much more closely through inter-farm networks and local knowledge of farming and disease management. This is advocated by Hinchliffe et al. (2018) and illustrated in the case of aquaculture in Bangladesh. It might also be more appropriate in such contexts where assembling data-driven, resource-intensive, and institutionalized forms of AMR governance common in the higher-resource settings of Europe (Helliwell et al. 2022; Hughes et al. 2021) and North America is not achievable. In our research, the value of informal farmer-to-farmer knowledge exchange was discussed (Interviews, Farms C, I, K, L; Policymakers A and B; Agrovet E). Farmers reported being an information resource for other farmers, including through social media communication applications such as WhatsApp, as well as asking others for advice. Whilst at present this method of information exchange appears to influence farmers to seek out specific drugs, apparently without prescription (as suggested earlier in the paper), the same networks could potentially be used more progressively to share best practice in infection prevention and disease management. It was clear from the interviews that farmers of a range of sizes had developed significant experience in managing disease on farms through sensory human-animal relations, for example by observational methods concerning birds’ stools, movement and posture, physical condition and symptoms, and feeding behaviour (Interviews, A, C, D, E, F, I, and J). It is this knowledge and experience of localized farmer-animal relations that critical scholars such as Hinchliffe et al., (2018: 1) argue should be harnessed in terms of addressing AMR as an “adaptive rather than technical challenge”, with antibiotic stewardship programmes too often envisaged as a data-driven and technical approach. We suggest that such adaptive work through localized farming networks focused on moving away from the multiple contingencies that result in dosing poultry with antibiotics could work in tandem with tighter regulation of markets and antibiotic use.
Conclusion
This paper has brought perspectives on value chains (Gereffi et al. 2005; Reardon et al. 2019) into Chandler’s (2019, p. 1) project of “infrastructural inversion” to understand how antibiotics have become central to the workings of Kenyan poultry farms as part of a transforming food system in this setting. We have evaluated the commercial drivers of antibiotic use on poultry farms as part of the management of disease, infection prevention, and product quality enhancement in an increasingly competitive and transforming market. A key contribution of the paper is to foreground the contrasting antibiotic dosing practices driven in traditional, transitional, and modern value chains and to identify the different ways in which they are currently governed. Antibiotic use is weakly regulated and strongly driven by market-based pressures, particularly in traditional value chains involving small- and medium-sized farms supplying open markets via brokers: these operations have limited engagement with policies of antibiotic stewardship. Engagement with antibiotic stewardship is more evident in the case of transitional value chains supplying corporate buyers and modern poultry value chains, particularly those involving influential processors and brand name buyers that require and adhere to standards involving the reduction and optimization of antibiotic use. However, these standards are unevenly observed and monitored. These findings help to explain how and why AMR policy, including antibiotic stewardship, developed in the global North struggles to translate meaningfully in settings of LMICs. It also suggests a careful critique of current antibiotic use in LMICs is needed, not least when standards for reducing and optimizing use are also at a relatively early stage in wealthier economies.
We concur with Kirchhelle et al.’s (2020, p. 2) call for “antibiotic policymaking [to] take into account antibiotics’ infrastructural relevance … in food production”. From our findings, which show a wide range of commercial drivers of antibiotic use, we argue that engagement with the dynamics of diverse value chains is critical to grasping this “infrastructural relevance” (Kirchhelle et al. 2020, p. 2). Whilst international standards and guidelines covering infection prevention, disease management, and drug use, along with education and training manuals, are mentioned in Kenya’s AMR National Action Plan and partially shape practice in processor-led value chains, the transitional and traditional value chains involving small- and medium-sized farms and shaped by market conventions remain largely untouched by AMR policy. Traditional value chains in particular embody significant pressures leading to widespread antibiotic use that is weakly regulated. This concurs with a wider body of critical literature on problems with translating policies on antibiotic stewardship (Butcher at al. 2021). Developments in antibiotic stewardship cannot follow a one-size-fits all approach, neither globally, nor even within national economies. Moreover, responsibility must not be narrowly transferred to individual value chain actors at the expense of more systemic change.
Participants in our research suggested a set of possible solutions to support the reduction and optimization of antibiotic use, mainly focused on tightening the regulation of production standards but crucially also strengthening the governance of poultry markets themselves that drive commercial pressures to use antibiotics. We therefore argue for close engagement with the structures and operations of the diverse value chains in which agricultural production is embedded, in order to better align developments in antibiotic stewardship with everyday pressures and practices. Social science perspectives, including those associated with political economy, are critical to developing a better understanding of the challenges and to making more geographically sensitive interventions.
Notes
Antimicrobials refer to antibiotics, antivirals, antimalarials, and antifungals. Our paper and underpinning research focus on antibiotics, as they have received most attention in stewardship programmes and are the focus of the conceptual literature most directly informing our analysis (notably Chandler 2019 and Helliwell et al. 2020). Moreover, antibiotics were the subset of antimicrobials most discussed by our interviewees. However, we acknowledge that attention needs to be paid, including in future research in LMICs, to other antimicrobials and non-bacterial infections aggravated by AMR.
Kenyan National Action Plan on the Prevention and Containment of Antimicrobial Resistance (2017-22), p. 6.
Studies conducted in Sub-Saharan Africa, including in Kenya, have found concentrations of common bacteria in poultry isolates that also affect humans including E. coli, Enterococci, Salmonella, Staphylococcus, Streptococcus and Campylobacter (Carron et al. 2018; Gahamanyi et al. 2020; Govender et al. 2019; Osman et al. 2019; Sarba et al. 2019). Literature suggests that the common antimicrobials for which bacteria in poultry isolates have shown resistance include tetracycline, co-trimoxazole, ampicillin, amoxicillin, sulphonamides, ciprofloxacin, and chloramphenicol (Adelaide et al. 2008; Carron et al. 2018; Dione et al. 2009; Langata et al. 2019; Wesonga et al. 2011).
This is a categorization used by the research team, influenced by policy-facing research (Shaw et al. 2019), rather than an official government classification.
PoultryWorld - FAO Sustainable Livestock Africa 2050 report (accessed 21/02/21).
Antibiotics used across a range of farm sizes reported by our interviewees were: promycine; bactim (sulfamethoxazole and trimethoprim); doxycycline; clarithromycin; tetracycline; colistin; polymixin; and oxytetracycline.
See for the Codex Code of Practice to Minimize and Contain Foodborne Antimicrobial Resistance, last revised in 2021. https://www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253 A%252 F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCXC%2B61-2005%252FCXC_061e.pdf (accessed 06/08/23).
Addressing market price instability is challenging in the contemporary Kenyan food system. However, provision of standard inputs such as vaccines could potentially be enabled by policy and action at national level.
References
Abu Hatab, A., M. E. R. Cavinato, and C. J. Lagerkvist. 2019. Urbanization, livestock systems and food security in developing countries: a systematic review of the literature. Food Security 11(2): 279–299. https://doi.org/10.1007/s12571-019-00906-1.
Adelaide, O. A., C. Bii, and P. Okemo. 2008. Antibiotic resistance and virulence factors in Escherichia coli from broiler chicken slaughtered at Tigoni processing plant in Limuru, Kenya. East African Medical Journal 85(12): 597–606.
Agyare, Christian, Vivian Etsiapa Boamah, and Crystal Ngofi Zumbi and Frank Boateng Osei. 2018. Antibiotic use in Poultry Production and its effects on Bacterial Resistance. In Antimicrobial Resistance - A Global threat, no pages.
Alhaji, N. B., A. E. Haruna, B. Muhammad, and M. K. Lawan, M.K. T.O.Isola. 2018. Antimicrobials usage assessments in commercial poultry and local birds in North-central Nigeria: Associated pathways and factors for resistance emergence and spread. Preventative Veterinary Medicine 154: 139–147. https://doi.org/10.1016/j.prevetmed.2018.04.001.
Barnett, C., P. Cloke, N. Clarke, and A. Malpass, A. 2011. Globalizing responsibility: the political rationalities of ethical consumption. Malden, Oxford, and Chichester: Wiley-Blackwell.
Begemann, S., E. Perkins, I. Van Hoyweghen, R. Christley, and F. Watkins. 2018. How political cultures produce different antibiotic policies in agriculture: a historical comparative case study between the United Kingdom and Sweden. Sociologia Ruralis 58(4): 765–785. https://doi.org/10.1111/soru.12206.
Bett, J. K., M. P. Musyoka, K. J. Peters, and W. Bokelmann. 2012. Demand for meat in the rural and urban areas of Kenya: a focus on the indigenous chicken. Economics Research International 10 pages. https://doi.org/10.1155/2012/401472).
Boamah, V. E., C. Agyare, H. Odoi, and A. Dalsgaard. 2016. Antibiotic practices and factors influencing the use of antibiotics in selected poultry farms in Ghana. Journal of Antimicrobial Agents 2(2): 8. https://doi.org/10.4172/2472-1212.1000120.
Bosire, C. K., M. Lannerstad, J. de Leeuw, M. S. Krol, J. O. Ogutu, P. A. Ochungo, and A. Y. Hoekstra. 2016. Urban consumption of meat and milk and its green and blue water footprints—patterns in the 1980s and 2000s for Nairobi, Kenya. https://doi.org/10.1016/j.scitotenv.2016.11.027.
Bowker, G. C., and S. L. Star. 2000. Sorting things out. Classification and its consequences. Cambridge, MA: MIT Press.
Branswell, H. 2017. Tightened rules for antibiotics for food livestock go into effect. StatNews com https://www.statnews.com/2017/01/03/fda-livestock-antibiotics/#:~:text=Under%20the%20Food%20and%20Drug%20Administration%20policy%2 C%20antibiotics,given%20to%20healthy%20animals%20to%20speed%20their%20 (accessed 22/07/23).
Buller, H., S. Hinchliffe, J. Hockenhull, D. Barrett, K. Reyher, A. Butterworth, and C. Heath. 2015. Systematic review and social research to further understanding of current practice in the context of using antimicrobials in livestock farming and to inform appropriate interventions to reduce antimicrobial resistance within the livestock sector. Report for DEFRA, August 2015.
Butcher, A., J. A. Cañada, and S. Sariola. 2021. How to make noncoherent problems more productive: towards an AMR management plan for low resource livestock sectors. Humanities and Social Science Communications 8: 287. https://doi.org/10.1057/s41599-021-00965-w.
Capita, R., and C. Alonso-Calleja. 2013. Antibiotic-resistant bacteria: a challenge for the food industry. Critical Reviews in Food Science and Nutrition 53(1): 11–48. https://doi.org/10.1080/10408398.2010.519837.
Carron, M., P. Alarcon, M. Karani, P. Muinde, J. Akoko, J. Onono, E. M. Fevre, B. Hasler, and J. Rushton. 2017. The broiler meat system in Nairobi, Kenya: using a value chain framework to understand animal and product flows, governance and sanitary risk. Preventative Veterinary Medicine 147: 90–99. https://doi.org/10.1016/j.prevetmed.2017.08.013.
Carron, M., Y-M. Chang, K. Momanyi, J. Akoko, J. Kiiru, J. Bettridge, G. Chaloner, J. Rushton, S. O’Brien, N. Williams, E. M. Fèvre, and B. Hasler. 2018. Campylobacter, a zoonotic pathogen of global importance: prevalence and risk factors in the fast evolving chicken meat system of Nairobi, Kenya. Plos Neglected Tropical Diseases 12(8): e0006658. https://doi.org/10.1371/journal.pntd.0006658.
Caudell, M. A., A. Dorado-Garcia, S. Eckford, C. Creese, D. K. Byarugaba, K. Afakye, T. Chansa-Kabali, F. O. Fasina, E. Kabali, S. Kiambi, T. Kimani, G. Mainda, P. E. Mangesho, F. Chimpangu, D. Kululeko, B. B. Kikimoto, E. Koka, T. Mugara, B. Rubegwa, and S. Swiswa. 2020. Towards a bottom-up understanding of antimicrobial use and resistance on the farm: a knowledge, attitudes, and practices survey across livestock systems in five African countries. Plos One 15(1): e0220274. https://doi.org/10.1371/journal.pone.0220274.
Chandler, C. I. R. 2019. Current accounts of antimicrobial resistance: stabilisation, individualisation, and antibiotics as infrastructure. Palgrave Communications 5(53) https://doi.org/10.1057/s41599-019-0263-4.
Cheng, G., J. Ning, S. Ahmed, J. Huang, R. Ullah, B. An, H. Hao, M. Dai, L. Huang, X. Wang, and Z. Yuan. 2019. Selection and dissemination of antimicrobial resistance in agri-food production. Antimicrobial Resistance and Infection Control 8(158): 1–3. https://doi.org/10.1186/s13756-019-0623-2.
Cox, J. A., E. R. Vlieghe, M. Mendelson, H. F. Wertheim, L. K. Ndegwa, M. V. Villegas, I. Gould, and G. Levy. 2017. Hara. Antibiotic stewardship in low- and middle-income countries: the same but different? Clinical Microbiology and Infection: The Official Publication of the European Society of Clinical Microbiology and Infectious Diseases 23(11): 812–818.
Craddock, S., and S. Hinchliffe. 2015. One world, one health? Social science engagements with the one health agenda. Social Science and Medicine 129: 1–4. https://doi.org/10.1016/j.socscimed.2014.11.016.
Crang, M., and I. Cook. 2007. Doing ethnographies. London: Sage.
Denyer-Willis, L., and C. I. R. Chandler. 2019. Quick fix for care, productivity, hygiene and inequality: reframing the entrenched problem of antibiotic overuse. British Medical Journal Global Health 4(4) https://doi.org/10.1136/bmjgh-2019-001590.
Dione, M. M., M. Leven, T. Marcotty, and S. Geertz. 2009. Prevalence and antimicrobial resistance of Salmonella isolated from broiler farms, chicken carcasses, and street-vended restaurants in Casamance, Senegal. Journal of Food Protection 72 (11): 2423-7. https://doi.org/10.4315/0362-028x-72.11.2423. PMID: 19903412.
Food Standards Agency. 2016. Chief Scientific Adviser’s Science Report. Issue four: Antimicrobial Resistance in the Food Supply Chain. London: Food Standards Agency.
Gahamanyi, N., L. E. G. Mboera, M. I. Matee, D. Mutangana, and E. V. G. Komba. 2020. Prevalence, risk factors, and antimicrobial resistance profiles of thermophilic Campylobacter species in humans and animals in SSA: a systematic review. International Journal of Microbiology. https://doi.org/10.1155/2020/2092478. PMID: 32025233; PMCID: PMC6983289.
Gereffi, G. 2018. Global value chains and development: redefining the contours of 21st century Capitalism. Cambridge: Cambridge University Press.
Gereffi, G., J. Humphrey, and T. Sturgeon. 2005. The governance of global value chains. Review of International Political Economy 12(1): 78–104. https://doi.org/10.1080/09692290500049805.
Gillespie, S., and M. van den Bold. 2017. Agriculture, food systems, and nutrition: meeting the challenge. Global Challenges 1(3): 1600002. https://doi.org/10.1002/gch2.201600002. PMID: 31565265; PMCID: PMC6607350.
Govender, V., E. Madoroba, K. Magwedere, G. Fosgate, and L. Kuonza. 2019. Prevalence and risk factors contributing to antibiotic-resistant Staphylococcus aureus isolates from poultry meat products in South Africa, 2015–2016. Journal of South African Veterinary Association 90(0): e1–e8. https://doi.org/10.4102/jsava.v90i0.1738. PMID: 31478731; PMCID: PMC6739513.
Graham, D. W., G. Bergeron, and M. W. Bourassa. 2019. Complexities in understanding antimicrobial resistance across domesticated animal, human, and environmental systems. Annals of the New York Academy of Sciences 1441(1): 17–30.
Guetiya Wadoum, R. E., N. F. Zambou, F. F. Anyangwe, J. R. Njimou, M. M. Coman, M. C. Verdenelli, C. Cecchini, S. Silvi, C. Orpianesi, A. Cresci, and V. Colizzi. 2016. Abusive use of antibiotics in poultry farming in Cameroon and the public health implications. British Poultry Science 57(4): 483–493. https://doi.org/10.1080/00071668.2016.1180668.
Helliwell, R., C. Morris, and S. Rama. 2019. Can resistant Infections be perceptible in UK dairy farming? Palgrave Communications 5: 12. https://doi.org/10.1057/s41599-019-0220-2.
Helliwell, R., C. Morris, and S. Raman. 2020. Antibiotic stewardship and its implications for agricultural animal-human relationships: insights from an intensive dairy farm in England. Journal of Rural Studies 78: 447–456. https://doi.org/10.1016/j.jrurstud.2020.07.008.
Helliwell, R., C. Morris, and S. Jones. 2022. Assembling antimicrobial resistance governance in UK Animal agriculture. Sociologia Ruralis. https://doi.org/10.1111/soru.12377.
Hinchliffe, S. J. 2022. Postcolonial global health, post-colony microbes and antimicrobial resistance. Theory Culture & Society 39(3): 145–168. https://doi.org/10.1177/0263276420981606.
Hinchliffe, S. J., and A. Butcher, M.M.Rahman. 2018. The AMR problem: demanding economies, biological margins, and co-producing alternative strategies. Palgrave Communications 4. https://doi.org/10.1057/s41599-018-0195-4.
Hudson, J. A., L. J. Frewer, G. Jones, M. Brereton, M. J. Whittingham, and G. Stewart. 2017. The agri-food chain and antimicrobial resistance: a review. Trends in Food Science & Technology 69: 131–147. https://doi.org/10.1016/j.tifs.2017.09.007.
Hughes, A., E. Roe, and S. Hocknell. 2021. Food supply chains and the antimicrobial resistance challenge: on the framing, accomplishments and limitations of corporate responsibility. Environment and Planning A: Economy & Space 53(6): 1373–1390. https://doi.org/10.1177/0308518X2110152.
Humphrey, J. 2007. The supermarket revolution in developing countries: tidal wave or tough competitive struggle? Journal of Economic Geography 7(4): 433–450. https://doi.org/10.1093/jeg/lbm008.
Innes, G. K., A. Markos, K. R. Dalton, C. A. Gould, K. E. Nachman, J. Fanzo, A. Barnhill, S. Frattaroli, and M. F. Davis. 2021. How animal agriculture stakeholders define, perceive, and are impacted by antimicrobial resistance: challenging the Wellcome Trust’s Reframing Resistance principles. Agriculture and Human Values 38(4): 893–909. https://doi.org/10.1007/s10460-021-10197-y.
Joel, H. M. 2018. Consumer willingness to pay for chicken meat derived from chicken fed on insect-based feed in Kenya http://erepository.uonbi.ac.ke/bitstream/handle/11295/105845/Harriet%20Mawia%20Joel.pdf?sequence=1&isAllowed=y.
Kagira, J. M., and P. W. N. Kanyari. 2010. Questionnaire survey on urban and peri-urban livestock farming practices and disease control in Kisumu Municipality. Kenya https://doi.org/10.4102/jsava.v81i2.110.
Kariuki, S., K. H. Keddy, M. Antonio, and I. N. Okeke. 2018. Antimicrobial resistance surveillance in Africa: successes, gaps and a roadmap for the future. African Journal of Laboratory Medicine 7(2): 1–2. https://doi.org/10.4102/ajlm.v7i2.924.
Kenyan National Action Plan on the Prevention and Containment of Antimicrobial Resistance 2017. -22).
Kiambi, S. R., A. Mwanza, C. Sirma, T. Czerniak, E. Kimani, A. Kabali, S. Dorado-Garcia, C. Eckford, S. Price, and S. Gikonyo et al. 2021. Understanding Antimicrobial Use contexts in the Poultry Sector: challenges for small-scale layer farms in Kenya. Antibiotics 10: 106. https://doi.org/10.3390/antibiotics10020106.
Kirchhelle, C. 2018. Pharming animals: a global history of antibiotics in food production (1935–2017). Palgrave Communications 4. https://doi.org/10.1057/s41599-018-0152-2.
Kirchhelle, C., P. Atkinson, A. Broom, K. Chuengsatiansup, J. P. Ferreira, N. Fortané, I. Frost, C. Gradmann, S. Hinchliffe, S. J. Hoffman, J. Lezaun, S. Nayiga, K. Outterson, S. H. Podolsky, S. Raymond, A. P. Roberts, A. C. Singer, A. D. So, L. Sringernyuang, E. Tayler, S. Rogers, Van Katwyk, and C. I. R. Chandler. 2020. Setting the standard: multidisciplinary hallmarks for structural, equitable and tracked antibiotic policy. British Medical Journal of Global Health Sep 5(9): e003091. https://doi.org/10.1136/bmjgh-2020-003091. PMID: 32967980; PMCID: PMC7513567.
Langata, L. M., J. M. Maingi, H. A. Musonye, J. Kiiru, and A. K. Nyamache. 2019. Antimicrobial resistance genes in Salmonella and Escherichia coli isolates from chicken droppings in Nairobi, Kenya. BMC Research Notes 2019;12(1):22. https://doi.org/10.1186/s13104-019-4068-8. PMID: 30642404; PMCID: PMC6332563.
Lee, J., G. Gereffi, and J. Beauvais. 2012. Global value chains and agrifood standards: challenges and possibilities for smallholders in developing countries. Proceedings of the National Academy of Sciences 109 (31): 12326–12331. https://doi.org/10.1073/pnas.0913714108.
Manyi-Loh, C., S. Mamphweli, E. Meyer, and A. Okoh. 2018. Antibiotic use in agriculture and its consequential resistance in environmental sources: potential public health implications. Molecules 23(4): 795.
Magothe, T.M., T.O. Okeno, W.B. Muhuyi and A.K. Kahi. 2012. Indigenous chicken production in Kenya: I. Current status. World’s Poultry Science Journal 68(1): 119–132.
Mbugua, M. W., P. Irungu, and J. Nzuma. 2014. Analysis of Demand for Antibiotics in Poultry Production in Kiambu County, Kenya.
McCarron, M., P. Munyua, P.-Y. Cheng, M. T. Po-Yung, C. Wanjohi, A. Moen, and M. A. Katz. 2015. Understanding the Poultry Trade Network in Kenya: Implications for Regional Disease Prevention and Control. https://reader.elsevier.com/reader/sd/pii/S0167587715001166?token=7BCF154F1A18011CD00E2470F9D9CE3A8BE7780CBD6C0BC35BA0ECCE459B92987C6672DFEDF2B9ADDE01F68CB9742175.
Millet, S., and L. Maertens. 2011. The European ban on antibiotic growth promoters in animal feed: from challenges to opportunities. The Veterinary Journal 187: 143–144. https://doi.org/10.1016/j.tvjl.2010.05.001.
Mshana, S. E., C. Sindato, M. I. Matee, and L. E. G. Mboera. 2021. Antimicrobial use and resistance in agriculture and food production systems in Africa: a systematic review. Antibiotics 10(8): 976. https://doi.org/10.3390/antibiotics10080976. PMID: 34439026; PMCID: PMC8389036.
Muloi, D., E. M. Fèvre, J. Bettridge, R. Rono, D. Ong’are, J. M. Hassell, M. K. Karani, P. Muinde, B. van Bunnik, A. Street, M. Chase-Topping, A. B. Pedersen, M. J. Ward, and M. Woolhouse. 2019. A cross-sectional survey of practices and knowledge among antibiotic retailers in Nairobi, Kenya. Journal of Global Health Dec 9(2): 010412. https://doi.org/10.7189/jogh.09.020412. PMID: 31489183; PMCID: PMC6708591.
Njisane, Y. Z., F. E. Mukumbo, and V. Muchenje. 2020. An outlook on livestock welfare conditions in African communities: a review. Asian-Australasian Journal of Animal Science Jun 33(6): 867–878. https://doi.org/10.5713/ajas.19.0282. Epub 2019 Jul 1. PMID: 31480203; PMCID: PMC7206380.
Nyaga, P. 2007. Poultry sector country review http://www.fao.org/3/a-ai379e.pdf.
O’Neill, J. 2016. Tackling drug-resistant infections globally.
Odede, R. 2016. Use of non-antibiotic growth promoters in chicken broiler production in Kenya. University of Nairobi.
Ortenblad, S. B. 2021. Despite the ‘supermarket revolution’: an exploration of dynamics in agri-food value chain in Thailand and implications for smallholders, traders and flows of produce. 233. PhD Theses, Department of geosciences and Natural Resource management, University of Copenhagen.
Osman, K. M., J. M. Badr, A. Orabi, A. Elbehiry, A. S. Saad, M. D. Ibrahim, and M. H. Hanafy. 2019. Poultry as a vector for emerging multidrug resistant Enterococcus spp.: first report of Vancomycin (Van) and the chloramphenicol-florfenicol (cat-fex-cfr) resistance genes from pigeon and duck faeces. Microbial Pathogenesis 128: 195–205. https://doi.org/10.1016/j.micpath.2019.01.006.
ReAct. 2017. Moving Beyond Antimicrobial Resistance (AMR) National Action Plans Development to Implementation. Action on antibiotic resistance (react) africa annual conference 2017. Retrieved from https://www.reactgroup.org/wp-content/uploads/2017/10/RAN_Conference-2017-Report.pdf.
Reardon, T., and C. P. Timmer. 2014. Five inter-linked transformations in the Asian agrifood economy: food security implications. Global Food Security 3(2): 108–117. https://doi.org/10.1016/j.gfs.2014.02.001.
Reardon, T., R. Echeverria, J. Berdegue, B. Minten, S. Liverpool-Tasie, D. Tschirley, and D. Zilberman. 2019. Rapid transformation of food systems in developing regions: highlighting the role of agricultural research and innovations. Agricultural Systems 172: 47–59. https://doi.org/10.1016/j.agsy.2018.01.022.
Rees, G. M., K. K. Reyher, D. C. Barrett, and H. Buller. 2021. It’s cheaper than a dead cow: understanding veterinary medicine use on dairy farms. Journal of Rural Studies 86: 587–598. https://doi.org/10.1016/j.jrurstud.2021.07.020.
Robinson, T., and F. Pozzi. 2011. Working paper-mapping supply and demand for animal-source foods to 2030. Rome. http://www.fao.org/docrep/014/i2425e/i2425e00.pdf.
Sarba, E. J., K. A. Kelbesa, M. D. Bayu, E. Z. Gebremedhin, B. M. Borena, and A. Teshale. 2019. Identification and antimicrobial susceptibility profile of Escherichia coli isolated from backyard chicken in and around ambo, Central Ethiopia. BMC Veterinary Research 15(1): 85. https://doi.org/10.1186/s12917-019-1830-z.
Shaw, M., H. Nielson, and M. Rose. 2019. Poultry Sector Study (DfID UKAid Report) http://www.bdsknowledge.org/dyn/bds/docs/960/DFID%20Poultry%20Sector%20Study%20180419.pdf.
Thornberg, R., and K. Charmaz. 2014. Grounded theory and theoretical coding. In The sage handbook of qualitative data analysis, ed. U. Flick. 153–169. London: Sage.
Timmermans, S., and I. Tavory. 2012. Theory construction in qualitative research: from grounded theory to abductive analysis. Sociological Theory 30(3): 167–186. https://doi.org/10.1177/0735275112457914.
Tompson, A. C., and C. I. R. Chandler. 2021a. Addressing antibiotic use: insights from social science around the world. Project Report. London School of Hygiene & Tropical Medicine. https://doi.org/10.17037/PUBS.04659562.
Tompson, A. C., L. Manderson, and C. I. R. Chandler. 2021b. Understanding antibiotic use: Practices, structures and networks. JAC-antimicrobial resistance 3 (4) dlab150. ISSN 2632 – 1823 https://doi.org/10.1093/jacamr/dlab150.
Valentine, G. 2005. Tell me about … using interviews as a research methodology. In Methods in human geography: a guide for students doing a Research Project, eds. R. Flowerdew, and D. Martin. 110–127. Harlow, UK: Prentice Hall.
van Berkum, S., T. Achterbosch, and V. Linderhof. 2017. Dynamics of food systems in Sub-saharan Africa. Waganingen. https://doi.org/10.18174/417176.
Van Boeckel, T. P., E. E. Glennon, D. Chen, M. Gilbert, T. P. Robinson, B. T. Grenfell, S. A. Levin, S. Bonhoeffer, and R. Laxminarayan. 2017. Reducing antimicrobial use in food animals. Science 357(6358): 1350–1352. https://doi.org/10.1126/science.aao1495.
Vetter, T., M. N. Larsen, and T. Bech Bruun. 2019. Supermarket-led development and the neglect of traditional food value chains: reflections on Indonesia’s agri-food system transformation. Sustainability 11(2): 498. https://doi.org/10.3390/su11020498.
Vosevwa, E. 2014. Factors Affecting Uptake of Processed Chicken Meat in Nairobi County: A case of Kenchic Ltd. http://docplayer.net/36064444-Factors-affecting-uptake-of-processed-chicken-meat-in-nairobi-county-a-case-of-kenchic-ltd.html 2014.
Watts, M. J. 2014 Commodities. In Introducing human geographies, ed. P. Cloke P., P. Crang, and M. Goodwin (3rd edition), 391–412. London: Taylor & Francis.
Wesonga, S. M., G. M. Muluvi, G.M., P.O. Okemo, and S. M. Kariuki. 2011. Antibiotic resistant salmonella and Escherichia coli isolated from indigenous Gallus domesticus in Nairobi, Kenya. East African Medical Journal 87(5): 205–210. https://doi.org/10.4314/EAMJ.V8715.63075.
World Health Organization. 2016. AMR: A manual for developing national action plans.
Acknowledgements
The authors acknowledge funding from the Arts and Humanities Research Council Global Challenges Research Fund (Award Number: AH/T004207/1). We are also grateful to Newcastle University for supporting the project during the Covid-19 pandemic. We thank all those interviewed as part of the field research, Caroline Wangari Njoroge for support with field materials, and Dadirai Mkombe for support with related research on the same project in Malawi. We appreciated feedback from a multi-stakeholder, including academic, audience at the project’s online inception workshop in November 2020. We are very grateful to the Editor and three anonymous reviewers for constructive feedback on an earlier version of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hughes, A., Roe, E., Wambiya, E. et al. The challenges of implementing antibiotic stewardship in diverse poultry value chains in Kenya. Agric Hum Values 41, 749–767 (2024). https://doi.org/10.1007/s10460-023-10518-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10460-023-10518-3