Abstract
Alternative raw materials unsuitable for human consumption might represent a great interest in animal nutrition. Freshwater bivalves represent an unexploited potential feed resource. The effects of freshwater mussel Sinanodonta woodiana meal on growth performance, histomorphology, and morphometric characteristics of the digestive system of rainbow trout (Oncorhynchus mykiss) were evaluated. A total of 540 rainbow trout juveniles were fed with three experimental diets consisting of fish meal (FM), fish by-product meal (FPM), and freshwater mussel meal (MM). After 90 days, the fish tripled their initial weight, despite fish fed with MM showed lower weight gain, specific growth rates, and higher feed conversion ratio than those fed with control diets. It has been estimated that the total production period of fish fed with the MM diet was 12–14 days longer than fish fed with FPM and FM, respectively, due to lower growth rates. Histological and morphometrical analyses did not reveal any diet influence on the severity of the observed lesions both in the gut and liver. No negative effects were observed with the inclusion of the MM; therefore, the use of this ingredient could be considered for rainbow trout feeding.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The growing world population will lead to an increase in the demand for fish products; thus, aquaculture will play a key role in supporting this necessity (Duarte et al. 2009; Ytrestøyl et al. 2015; Fry et al. 2016). Increasing the diversity of protein sources in the aquaculture industry is thus a key factor for meeting this demand. Even though the aquafeed sector has recently improved feed quality, reducing fish meal (FM) and fish oil (FO) inclusion, by using alternative plant materials (soybeans, wheat, canola, corn, and nuts), animal by-products (poultry by-product meal, blood meal, and bone meal), or unicellular proteins such as bacteria, yeasts, and microalgae (Olsen and Hasan 2012; Chauton et al. 2015; Teves and Ragaza 2016; Hua et al. 2019; Jones et al. 2020), novel protein sources are still required to correspond such an increasing demand.
Aquatic organisms are also of particular interest as FM substitutes, since many of the farmed species require long-chain unsaturated fatty acids, which are found mainly in marine and freshwater food webs (Weiß and Buck 2017). Molluscs, as sources of marine and freshwater origin nutrients, such as Cristaria plicata (Luo et al. 2019), Mytilus edulis (Weiß and Buck 2017), Mytilus chilensis (Mongile et al. 2015), Holothuria polii (Piccinno et al. 2013), and Rapana venosa (Sahin et al. 2021; Sahin and Ergün 2021), have been investigated to replace fish meal in different fish species in various studies within this context. In particular, in the rainbow trout (Oncorhynchus mykiss), the partial replacement of FM has been tested using M. edulis (Berge and Austreng 1989) and Mytilus galloprovincialis (Aral et al. 1999). Mussels are of considerable interest for this purpose as they can be used as a protein source in fish feed (Langeland et al. 2016). Mussel meal has a high protein content with an adequate composition of amino acids. Moreover, it has low fat content and a favourable fatty acid profile, being rich in eicosapentaenoic (EPA) and docosahexaenoic (DHA) acids, which are particularly important in fish nutrition (Berge and Austreng 1989; Carlberg et al. 2018; Wagner et al. 2019). However, only in rare cases, freshwater mussels, such as Unionid (Anodonta anatina and Unio mancus), were used, suggesting the possibility of a partial replacement of FM with freshwater mussel meal (MM) (Sicuro et al. 2010). Moreover, considering freshwater bivalves are not only used for human consumption but also their production in the future could also increase as they can be reared in inland integrated aquaculture to reduce the environmental impact of freshwater fish farms (Sicuro et al. 2020).
Within molluscs, Sinanodonta woodiana is an Asian freshwater unionid, accidentally introduced to Europe in 1960 and is currently considered an invasive alien species (Douda et al. 2012; Poznańska-Kakareko et al. 2021; Urbańska et al. 2021). Its utilization in fish nutrition could mitigate and limit the negative effects of its expansion because of being a serious threat to autochthonous freshwater mussels and a vector of an exotic fish species, the European bitterling (Rhodeus amarus) (Urbańska et al. 2021).
In the present study, fish meal was partially replaced with the freshwater mussel S. woodiana meal (MM) to evaluate its potential use as a new alternative ingredient for the rainbow trout (O. mykiss). MM was compared with two of the most common commercial sources of protein in fish feeds: fish meal (FM) and fish by-product meal (FPM), the latter derived from fish trimmings and offal. FPM has strongly reduced the fish farming dependency on marine resources, thus reducing also the environmental impact of aquaculture (Ytrestøyl et al. 2015; Hua et al. 2019). For this reason, and to provide a broader comparison with MM, FPM has been used as a further diet. The use of MM and their effects was evaluated on the basis of zootechnical performance and histopathological alteration on the liver and gut.
Materials and methods
The trial was conducted at the Experimental Facility of the Department of Agricultural, Forest, and Food Sciences (DISAFA) of the University of Turin (Italy). All trial procedures involving fish were conducted according to the guidelines of the European Union Council (2010/63/EU) for the use and care of experimental animals. The Ethical Committee of the University of Turin approved the experimental protocol (protocol no. 314714).
Sinanodonta woodiana collection and preparation of the experimental feed
The bivalves S. woodiana (Fig. 1) were collected at Cavagnolo Lake, Piedmont, Italy (45°10′07.9″N, 8°02′21.2″E). The bivalves were kept under quarantine in a continuous flow-through system for 1 month to reduce the potential biological contaminants (Sicuro et al. 2020). Afterwards, the soft tissue of the mussels was separated from their shells for chemical analyses.
A total of 150 adult individuals (300–1000 g each) of S. woodiana were selected to obtain the MM. The soft tissue of the mussels was separated from their shells, dried in an oven at 70 °C for 72 h, and ground using a knife mill to obtain a homogeneous powder (Fig. 2).
MM was frozen at − 20 °C until proximate analyses were performed (Table 1). Each fish diet was prepared by cold pelleting through a meat grinder Labor 32 (Rheninghaus, San Mauro Torinese, Italy), and the obtained pellet was dried at 50 °C for 48 h (Fig. 2). Diets were stored at 4 °C for the entire duration of the growth trial. Proximate composition (crude protein, crude fat, ash, moisture, and estimated carbohydrate) of each diet (MM, FPM, and FM) was analysed following AOAC (AOAC 2000). Based on proximate composition (Table 1), three isoproteic and isolipidic diets were formulated including 20% of either MM or FPM in substitution of FM. This level of inclusion was selected as the maximum possible level of the MM inclusion compatible with the rainbow trout (O. mykiss) nutritional requirements and for evaluating the presence of antinutritional effects.
Fish and feeding trials
A total of 540 rainbow trout juveniles, with a mean individual weight of 19.11 ± 2.70 g, were randomly distributed into 12 experimental tanks (45 fish per tank) with a density of around 2 kg per m3. In the first part of the trial, fish were acclimated to the experimental conditions for 2 weeks and fed with a commercial feed. The 90-day feeding trial was conducted in a mono-factorial and balanced design with 3 levels of experimental treatment (diets) and 4 replicates. Tanks (400 L each) were supplied with artesian well water in a flow-through open system of 8 L min−1 water inflow. The fish were manually fed twice a day, 6 days a week, at 1.8% of their biomass, and weighed in bulk every 15 days to adjust the amount of feed. During the feeding trial, fish were observed daily for behavioural responses to test diets and calculate the mortality. Water temperature and dissolved oxygen were monitored daily in each tank using an oximeter YSI model 55 (YSI Incorporated, Yellow Springs, OH, USA), and concentration of ammonium (N–NH4+), nitrite (N–NO2), nitrate (N–NO3), and phosphate (P–PO4) was measured once a week, using a photometer NOVA 60 Spectroquant® (Sigma-Aldrich Canada Co., Winston Park, Ontario, Canada) (Table 2).
At the end of the trial, three fish per tank were euthanised by administering an overdose of anaesthetic (MS-222, PHARMAQ Ltd., Fordingbridge, UK; 500 mg/L) for histomorphological analyses.
Growth performance
The growth performance of rainbow trout fed with the experimental diets was evaluated through the final weight and the indexes of survival, weight gain (WG), specific growth rate (SGR), and feed conversion ratio (FCR). Hereafter are the equations of the used indexes:
A growth model was estimated for the three treatments using an exponential equation.
Histomorphological analysis
At the end of the trial, anterior and posterior gut segments, as well as the liver of twelve fish per treatment (three fish per tank), were collected and fixed in a 10% buffered formalin solution to perform histomorphological investigations. Gut segments and liver tissue were routinely embedded in paraffin wax blocks following standard histological techniques (Verdile et al. 2020) and then sectioned at 5-μm thickness through the automated rotary microtome Leica RM2155 (Leica Microsystems, Nussloch, Germany) and coloured with haematoxylin–eosin (H&E).
The observed histopathological findings were evaluated in all the organs using a semi-quantitative scoring system as follows: absent (score = 0), mild (score = 1), moderate (score = 2), and severe (score = 3). Gut histopathological findings were separately assessed for mucosa (inflammatory infiltrates) and submucosa (inflammatory infiltrates and gut-associated lymphoid tissue activation) for each segment. The total score of each gut segment was obtained by adding up the mucosa and submucosa scores. All the slides were blind assessed by two independent observers and the discordant cases were reviewed, using a multi-head microscope, until a unanimous consensus was reached.
For the morphometric evaluation, gut sections were examined by light microscopy and captured with a Nikon DS-Fi1 digital camera (Nikon Corporation, Minato, Tokyo, Japan) coupled to a Zeiss Axiophot microscope (Carl Zeiss, Oberkochen, Germania) using × 2.5 objective lens. NIS-Elements F software was used for image capturing. The analyses were made using the Image Pro-Plus software (6.0 version, Media Cybernetics, MD, USA). The evaluated morphometric indices were the villus height (Vh) and the villus width (Vw) of 10 well-oriented and intact villi. Moreover, the mucosal (MT) and muscular (MuT) thicknesses were also measured on three standardised points of the gut mucosal and muscular layers, respectively (Fig. 3).
Trace elements and rare earth elements
Due to the potential metal contamination and bioaccumulation in the tissues of filter-feeding organisms as bivalves (Storelli et al. 2000; McEneff et al. 2017), trace elements and rare earth element concentrations were analysed. Samples of mussels meal (N = 3) were homogenised and subjected to mineralisation with acids and oxidants (HNO3 and H2O2) using a microwave digestion lab station (Ethos 1, Milestone, Shelton, CT, USA), equipped with a 10-position rotor for high-pressure PTFE (polytetrafluoroethylene) digestion tubes. Trace elements (N = 23) and rare earth elements (N = 16) were analysed by inductively coupled plasma–mass spectrometry (ICP-MS Xseries II, Thermo Scientific, Bremen, Germany) following the protocols already described (Squadrone et al. 2016). The recovery of the analytical methods was verified using certified reference materials (Oyster Tissue-SRM 1566b and REE-1 from the National Institute of Standard and Technology). Recovery rates were found to be between 94 and 105%. A direct mercury analyser (DMA80, Milestone, Shelton, CT, USA), which performs thermal decomposition, catalytic reduction, amalgamation, desorption, and atomic absorption spectroscopy without sample pre-treatment, was instead used for total mercury determination. The limit of quantification (LOQ) was 0.010 mg kg−1 for trace elements and 0.001 mg kg−1 for REE. All values were expressed on a wet weight basis (Table 3). The heavy metal content in the mussel meal was compliant with the maximum limits set by European Regulations for feed (Dir 32/2002 UE and amendments).
Statistical analyses
Statistical analyses were conducted with the software R (R Core Team 2022), while the fitting of exponential growth models was performed through the software Excel.
The water parameters (temperature, oxygen, ammonium, nitrite, nitrate, and phosphate) and the growth performance indices (survival, WG, SGR, and FCR) were analysed to assess the presence of differences among diets (MM, FPM, and FM). For this purpose, an ANOVA test was adopted after the verification of normality (Shapiro–Wilk) and homogeneity of the variance (Levene’s test of the package car). The Tukey test (package multcomp) was adopted as a post-hoc test.
The presence of differences in the histomorphological alterations among diets was evaluated with a Kruskal–Wallis test (Field et al. 2012).
Results
Growth performance
The three experimental diets were accepted by all fish and consumed without waste. The growth performances are reported in Table 4.
At the end of the trial, the final fish weight increased more than triple for all treatments, varying between 67.6 and 77.3 g. The survival rate was equally high among the diets (97.59 ± 3.5%).
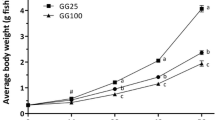
For all the indexes and the final weight, a difference was present among the diets. The WG and SGR were lower in trout fed with MM compared with FPM and FM, resulting in fish slightly smaller in MM group (Table 4 and Fig. 4). With respect to FCR, all values were below 1, despite higher in MM than in both FM and FPM. Although slight differences in the production performances have been reported, antinutritional effects were not observed.
The impact of the weight differences in a real farm production could be estimated by the growth curve models (Fig. 4). In fact, all the treatments could reach the European commercial size (around 400 g) between 190 and 204 days of rearing. It means that those fish fed with MM, in a commercial farm, should be reared for 12 or 14 days more than those fed with FPM and FM, respectively.
Histomorphological analyses
The results of the histopathological evaluation are shown in Table 5. Diet did not influence the severity of the observed histopathological lesions both in gut and liver (p > 0.05). Independently from diet, from absent to mild and multifocal lymphoplasmacytic inflammation was recorded in the gut, while liver showed from mild to severe and diffuse vacuolar degeneration (Fig. 5a) and mild and multifocal lymphoplasmacytic inflammation (Fig. 5b).
Data regarding morphometric indices are reported in Table 6. Non-significant differences were reported among dietary treatments for all the evaluated morphometric indices. However, Vh and mucosal thickness were numerically lower in the proximal gut compared to the distal gut.
Discussion
In this study, the use of the freshwater bivalve S. woodiana as an alternative protein source to replace FM or FPM in the feeding of rainbow trout (O. mykiss) was investigated, by evaluating the growth performance and the histopathological lesions on the gut and liver of the fish. Regardless of the diet, at the end of the trial all the fish have tripled their initial weight and no differences in the survival rates were recorded. However, the fish fed with MM had lower WG and SGR than fish fed with FM and FPM. Furthermore, the FCR was higher in fish fed with MM.
In the preparation of the MM experimental diet, an intense smell originating from the mussel meal was noted; this factor may have negatively affected the food intake in comparison with the other diets. During the drying process of mussel tissues in the oven, there was an evident change in the appearance of tissues, in particular, for the presence of mucus secreted by mussels during the separation of the shells. In the future, other methods to obtain mussel meal should be tested for separating the mucilage to improve palatability. The MM composition was lower in crude protein and higher in non-protein and non-fat compounds than FM and FPM, even though MM proximate composition was comparable with alternative protein sources tested before on rainbow trout diets, next to 40% (Hua et al. 2019). Considering the proximate composition of MM, the highest possible level of inclusion (20%) in fish feed was adopted to highlight any possible presence of antinutritional effects.
It has been estimated that the commercial size in fish fed with MM would be delayed by only 12–14 days. The increase of trout productive cycle duration causes an increase of production costs that is an obviously negative aspect for trout farmers (Carlberg et al. 2018). However, the use of S. woodiana in fish nutrition could be considered an added value that is appreciated by modern consumer, which pays more attention to food sustainability, as in the case of organic salmon (Carlberg et al. 2018).
Mussel meal derived from S. woodiana is a good source of proteins, carbohydrates, ashes, and amino acids and suitable for developing new pet’s feed (Konieczny et al. 2021). The MM proximate analyses have been compared with mussel meal derived by other species, which is the most similar feedstuff already tested in similar studies. Mussel meal derived from M. edulis and C. plicata contains a high percentage of proteins and lipids with a beneficial fatty acid profile; therefore, it is considered a valid substitute for FM and suitable for feeding fish (Berge and Austreng 1989; Luo et al. 2019). In Ussuri catfish (P. ussuriensis) it has been estimated that C. plicata meal can replace up to 50% of FM proteins without causing a negative effect on growth (Luo et al. 2019). Indeed, M. edulis and C. plicata meal can be used as a substitute for FM, but excessive content might affect growth performance (Weiß and Buck 2017; Luo et al. 2019). However, in the diet of Arctic charr (S. alpinus), the metabolism was less influenced by the M. edulis meal diet (Wagner et al. 2019). Poorer growth has been reported with the increase in the inclusion of M. edulis meal in the rainbow trout diet due to the low digestibility caused by the high ash content derived from mussel shells (Berge and Austreng 1989). However, in a study were shelled M. galloprovincialis were used, no negative effects were noted on the growth performance (Aral et al. 1999). In our study, MM was obtained using shelled mussels; consequentially, the ash content has not affected the diet. In this study, S. woodiana meal was tested, for the first time, as a novel ingredient of rainbow trout. Our productive results are consistent with those obtained in marine aquaculture for common sole (S. solea) (Mongile et al. 2015) and turbot (S. maximus) (Weiß and Buck 2017).
To understand the effect of the new fish diets on the digestive system, histological analyses were performed. In fact, the digestive system, including liver, is the main site of feed digestion and nutrient absorption, and its health status is capable of greatly affecting the utilisation of dietary nutrients (Caimi et al. 2020). Therefore, the absence of significant alterations in the gut and liver of the treated groups seems to suggest that MM can be safely used as alternative feed ingredients in aquaculture, maintaining gastrointestinal health. These results are in accordance with Mongile et al. (2015) and Nagel et al. (2014), who did not record any significant histopathological alteration in liver and gut after the administration of diets containing up to 75% of M. edulis and M. chilensis meal in common sole and turbot. Similarly to what has been observed in the present study, also Mongile et al. (2015) and Nagel et al. (2014) recorded a uniformly abundant vacuolar degeneration in the liver of all the groups, including control groups. This hepatic alteration has been previously detected in trout and it can be considered paraphysiological, depending on the intensive feeding and farming of rainbow trout (Chemello et al. 2021). Furthermore, at the end of the trial, all the fish have triplicated their initial weight, suggestive of unaltered liver functionality.
The feasibility of MM utilisation is also confirmed by the unaffected morphometric indices both in the proximal and distal gut. Moreover, the maintenance of a proximo-distal increasing gradient recorded for Vh and mucosal thickness is in accordance with the physiological intensity of the nutrient absorption processes that take place in the gut (Verdile et al. 2020). In fact, short and long villi are simultaneously present in trout proximal and distal gut, with the long villi length in the proximal intestine increasing when the fish reached their highest weight, and the distal intestine showing similar villi length along with the fish development (Verdile et al. 2020). Therefore, the identification of differences between the two gut compartments may be related to either the simultaneous presence of short and long villi or the villi length changes throughout the fish cycle (Chemello et al. 2021). The efficient nutrient absorption thanks to the maintenance of an adequate gut morphological asset is also confirmed by the unaltered growth performances herein recorded.
The presence of non-essential and toxic elements such as mercury, lead, cadmium, and arsenic may affect the use of MM. However, bioaccumulation is not an intrinsic feature of bivalves, but it is a consequence of the ecological conditions where they live, and this could nullify the successive utilisation of MM in fish feeds and in the aquafeed industry. In this study, the concentration of many elements was analysed also in bivalves with an estimated age up to about 20 years. Although the metal concentrations were below the legal limits set by European regulations for Hg, Pb, Cd, and As, the potential risk of bioaccumulation and the potential synergic effects of inorganic contaminants in fish tissues should not be underestimated (Storelli et al. 2000; McEneff et al. 2017). For this purpose, the use in the diet of substances capable of chelating heavy metals, such as humic and fulvic acids, could be evaluated (Rasidi et al. 2019; Jusadi et al. 2020). Additionally, it was found that the addition of humic and fulvic acids in the diet formulation can improve feed use and growth rate (Rasidi et al. 2019; Jusadi et al. 2020).
Conclusions
Our results indicate that MM can be utilised in rainbow trout nutrition even if fish fed with MM need some more day to reach commercial size with respect to those fed with fish meals. The survival rate during growth trial and the absence of histological lesions on the digestive system of the fish confirm the feasibility of using these new feed ingredients. The decrease in the productive parameters can be compensated by a general consideration of the reduction of ecological footprint of fish farming, and this is probably the most relevant aspect recorded in this research. Moreover, the use of a feedstuff obtained by an exotic species of bivalve that threatens the native species can be considered a valuable use of resources derived from the control of an alien population. In conclusion, we believe that this research not only suggest a novel ingredient in the panorama of alternative ingredients for fish nutrition in aquaculture but also represents an example of the modern concept of integration applied to aquaculture.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- MM:
-
Freshwater mussel meal
- FM:
-
Fish meal
- FPM:
-
Fish by-product meal
- FO:
-
Fish oil
- EPA:
-
Eicosapentaenoic acids
- DHA:
-
Docosahexaenoic acids
- WG:
-
Weight gain
- SGR:
-
Specific growth rates
- FCR:
-
Feed conversation ratio
- Vh:
-
Villus height
- Vw:
-
Villus width
- MT:
-
Mucosal thicknesses
- MuT:
-
Muscular thicknesses
References
AOAC (2000) Official methods of analysis, 17th edn. The Association of Official Analytical Chemist, Gaithersburg
Aral O, Ağirağaç C, Yi̇ği̇t MA (1999) Research on the use of Mediterranean mussel in feed for rainbow trout (Oncorhynchus mykiss W. 1782). Turk J Vet Anim Sci 23:23–28
Berge GM, Austreng E (1989) Blue mussel in feed for rainbow trout. Aquaculture 81:79–90. https://doi.org/10.1016/0044-8486(89)90232-9
Caimi C, Gasco L, Biasato I et al (2020) Could dietary black soldier fly meal inclusion affect the liver and intestinal histological traits and the oxidative stress biomarkers of Siberian Sturgeon (Acipenser baerii) Juveniles? Animals 10:155. https://doi.org/10.3390/ani10010155
Carlberg H, Lundh T, Cheng K, Pickova J, Langton M, Gutiérrez JLV, Kiessling A, Brännäs E (2018) In search for protein sources: evaluating an alternative to the traditional fish feed for Arctic charr (Salvelinus alpinus L.). Aquaculture 486:253–260. https://doi.org/10.1016/j.aquaculture.2017.12.027
Chauton MS, Reitan KI, Norsker NH, Tveterås R, Kleivdal HT (2015) A techno-economic analysis of industrial production of marine microalgae as a source of EPA and DHA-rich raw material for aquafeed: Research challenges and possibilities. Aquaculture 436:95–103. https://doi.org/10.1016/j.aquaculture.2014.10.038
Chemello G, Biasato I, Gai F, Capucchio MT, Colombino E, Schiavone A, Gasco L, Pauciullo A (2021) Effects of Tenebrio molitor larvae meal inclusion in rainbow trout feed: myogenesis-related gene expression and histomorphological features. Ital J Anim Sci 20:1211–1221. https://doi.org/10.1080/1828051X.2021.1945959
Douda K, Vrtílek M, Slavík O, Reichard M (2012) The role of host specificity in explaining the invasion success of the freshwater mussel Anodonta woodiana in Europe. Biol Invasions 14:127–137. https://doi.org/10.1007/s10530-011-9989-7
Duarte CM, Holmer M, Olsen Y, Soto D, Marbà N, Guiu J, Black K, Karakassis I (2009) Will the oceans help feed humanity? Bioscience 59:967–976. https://doi.org/10.1525/bio.2009.59.11.8
Field A, Miles J, Field Z (2012) Discovering statistics using R, 1st edn. Sage publications, London, UK
Fry JP, Love DC, MacDonald GK, West PC, Engstrom PM, Nachman KE, Lawrence RS (2016) Environmental health impacts of feeding crops to farmed fish. Environ Int 91:201–214. https://doi.org/10.1016/j.envint.2016.02.022
Hua K, Cobcroft JM, Cole A, Condon K, Jerry DR, Mangott A, Praeger C, Vucko MJ, Zeng C, Zenger K, Strugnell JM (2019) The future of aquatic protein: implications for protein sources in aquaculture diets. One Earth 1:316–329. https://doi.org/10.1016/j.oneear.2019.10.018
Jones SW, Karpol A, Friedman S, Maru BT, Tracy BP (2020) Recent advances in single cell protein use as a feed ingredient in aquaculture. Curr Opin Biotechnol 61:189–197. https://doi.org/10.1016/j.copbio.2019.12.026
Jusadi D, Aprilia T, Setiawati M, Agus Suprayudi M, Ekasari J (2020) Dietary supplementation of fulvic acid for growth improvement and prevention of heavy metal accumulation in Nile tilapia fed with green mussel. Egypt J Aquat Res 46:295–301. https://doi.org/10.1016/j.ejar.2020.04.002
Konieczny P, Andrzejewski W, Yang T, Urbańska M, Stangierski J, Tomczyk Ł, Mikołajczak B (2021) Selected quality attributes of freshwater mussel powder as a promising ingredient for pet food. Animals 12:90. https://doi.org/10.3390/ani12010090
Langeland M, Vidakovic A, Vielma J, Lindberg JE, Kiessling A, Lundh T (2016) Digestibility of microbial and mussel meal for Arctic charr (Salvelinus alpinus) and Eurasian perch (Perca fluviatilis). Aquac Nutr 22:485–495. https://doi.org/10.1111/anu.12268
Luo C, Wang Y, Tao S, Liao Y, Yang C, Cui C, Yang J, Yang Y (2019) Effects of replacing fish meal with mussel (Cristaria plicata) meat on growth, digestive ability, antioxidant capacity and hepatic IGF-I gene expression in juvenile Ussuri catfish (Pseudobagrus ussuriensis). Aquac Res 50:826–835. https://doi.org/10.1111/are.13953
McEneff G, Quinn B, Bennion M, Dolan S, O’Rourke K, Morrison L (2017) Bioaccumulation of metals in juvenile rainbow trout (Oncorhynchus mykiss) via dietary exposure to blue mussels. Chemosphere 188:548–556. https://doi.org/10.1016/j.chemosphere.2017.08.141
Mongile F, Mandrioli L, Mazzoni M, Pirini M, Zaccaroni A, Sirri R, Parma L, Gatta PP, Sarli G, Bonaldo A (2015) Dietary inclusion of mussel meal enhances performance and improves feed and protein utilization in common sole (Solea solea, Linnaeus, 1758) juveniles. J Appl Ichthyol 31:1077–1085. https://doi.org/10.1111/jai.12895
Nagel F, von Danwitz A, Schlachter M, Kroeckel S, Wagner C, Schulz C (2014) Blue mussel meal as feed attractant in rapeseed protein-based diets for turbot (Psetta maxima L.). Aquac Res 45:1964–1978. https://doi.org/10.1111/are.12140
Olsen RL, Hasan MR (2012) A limited supply of fishmeal: impact on future increases in global aquaculture production. Trends Food Sci Technol 27:120–128. https://doi.org/10.1016/j.tifs.2012.06.003
Piccinno M, Schiavone R, Zilli L, Sicuro B, Storelli C, Vilella S (2013) Sea cucumber meal as alternative protein source to fishmeal in gilthead sea bream (Sparus aurata) nutrition: effects on growth and welfare. Turk J Fish Aquat Sci 13:305–313. https://doi.org/10.4194/1303-2712-v13_2_12
Poznańska-Kakareko M, Wiśniewski K, Szarmach D, Witkowska A, Kakareko T, Jermacz Ł, Kobak J (2021) Importance of substratum quality for potential competitive niche overlap between native and invasive unionid mussels in Europe. Sci Total Environ 799:149345. https://doi.org/10.1016/j.scitotenv.2021.149345
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rasidi R, Jusadi D, Setiawati M, Yuhana M, Zairin MJR, Sugama K (2019) Effects of humic acid addition to feeds with heavy metal cadmium contamination from green mussels on the growth performance of Asian seabass. BIOTROPIA- Southeast Asian J Trop Biol 26:214–221
Sahin T, Ergün S (2021) Incorporation of Rapa Whelk (Rapana venosa) meal in diets for rainbow trout (Oncorhynchus mykiss) fry. Aquac Res 52:678–692. https://doi.org/10.1111/are.14925
Sahin T, Yılmaz S, Gürkan M, Ergün S (2021) Effects of Rapana venosa meal-supplemented diets on reproduction, histopathology and some blood parameters of rainbow trout (Oncorhynchus mykiss) broodstock. Aquac Res 52:4897–4910. https://doi.org/10.1111/are.15324
Sicuro B, Mioletti S, Abete C, Amedeo S, Panini E, Forneris G (2010) Potential utilisation of farmed freshwater mussels (Anodonta anatina and Unio mancus) in Italy. Cuba J Agric Sci 44:6
Sicuro B, Castelar B, Mugetti D, Pastorino P, Chiarandon A, Menconi V, Galloni M, Prearo M (2020) Bioremediation with freshwater bivalves: a sustainable approach to reducing the environmental impact of inland trout farms. J Environ Manag 276:111327. https://doi.org/10.1016/j.jenvman.2020.111327
Squadrone S, Brizio P, Stella C, Prearo M, Pastorino P, Serracca L, Ercolini C, Abete MC (2016) Presence of trace metals in aquaculture marine ecosystems of the northwestern Mediterranean Sea (Italy). Environ Pollut 215:77–83. https://doi.org/10.1016/j.envpol.2016.04.096
Storelli MM, Storelli A, Marcotregiano GO (2000) Heavy metals in mussels (Mytilus galloprovincialis) from the Ionian Sea, Italy. J Food Prot 63:273–276. https://doi.org/10.4315/0362-028X-63.2.273
Teves JFC, Ragaza JA (2016) The quest for indigenous aquafeed ingredients: a review. Rev Aquac 8:154–171. https://doi.org/10.1111/raq.12089
Urbańska M, Kamocki A, Kirschenstein M, Ożgo M (2021) The Chinese pond mussel Sinanodonta woodiana demographically outperforms European native mussels. Sci Rep 11:17058. https://doi.org/10.1038/s41598-021-96568-1
Verdile N, Pasquariello R, Scolari M, Scirè G, Brevini TAL, Gandolfi F (2020) A detailed study of rainbow trout (Onchorhynchus mykiss) intestine revealed that digestive and absorptive functions are not linearly distributed along its length. Animals 10:745. https://doi.org/10.3390/ani10040745
Wagner L, Gómez-Requeni P, Moazzami AA, Lundh T, Vidakovic A, Langeland M, Kiessling A, Pickova J (2019) 1H NMR-based metabolomics and lipid analyses revealed the effect of dietary replacement of microbial extracts or mussel meal with fish meal to Arctic Charr (Salvelinus alpinus). Fishes 4:46. https://doi.org/10.3390/fishes4030046
Weiß M, Buck BH (2017) Partial replacement of fishmeal in diets for turbot (Scophthalmus maximus, Linnaeus, 1758) culture using blue mussel (Mytilus edulis, Linneus, 1758) meat. J Appl Ichthyol 33:354–360. https://doi.org/10.1111/jai.13323
Ytrestøyl T, Aas TS, Åsgård T (2015) Utilisation of feed resources in production of Atlantic salmon (Salmo salar) in Norway. Aquaculture 448:365–374. https://doi.org/10.1016/j.aquaculture.2015.06.023
Acknowledgements
The authors are grateful to the personnel of the Department of Agricultural, Forest and Food Sciences of the University of Turin, in particular, Prof. Laura Gasco, and to the research group on Animal Science that hosted the fish for conducting the growth trial.
Funding
Open access funding provided by Università degli Studi di Torino within the CRUI-CARE Agreement. This research was funded by the European project Aquaval (ERA-NET WaterWorks 2015).
Author information
Authors and Affiliations
Contributions
B. Sicuro: conceptualisation, methodology, writing—original draft, writing—review and editing, supervision, and funding acquisition; B. Castelar: methodology, investigation, data curation, formal analysis, and writing—review and editing; C. Bergamino: methodology, investigation, data curation, formal analysis, writing—original draft, and writing—review and editing; S. Mioletti: methodology, formal analysis, and writing—review and editing; S. Squadrone: methodology, formal analysis, and writing—review and editing; A. Griglione: methodology, formal analysis, and writing—review and editing, M. Falzone: methodology, formal analysis, and writing—review and editing; E. Colombino: methodology, data curation, formal analysis, and writing—review and editing; M.T. Capucchio: methodology, formal analysis, writing—review and editing, and supervision. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval
The animal study protocol was approved by Ethics Committee of Torino University (Comitato bioetico di Ateneo, University of Turin) protocol code: 314714, July 27th, 2019.
Competing interests
The authors declare no competing interests.
Additional information
Handling Editor: Brian Austin
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sicuro, B., Castelar, B., Bergamino, C. et al. Freshwater mussel meal as new alternative ingredient for rainbow trout (Oncorhynchus mykiss) feeds: growth performance and histomorphological analyses. Aquacult Int 32, 431–445 (2024). https://doi.org/10.1007/s10499-023-01168-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-023-01168-7