Abstract
Alien species introduction is a global phenomenon involving different invasion patterns and is characterized by niche conservatism or shift. We describe the spatial distribution of Cacyreus marshalli Butler, [1898] (Lepidoptera: Lycaenidae) in its native (southern Africa) and invaded (Europe) ranges. C. marshalli is the only alien butterfly in Europe, introduced by the trade of ornamental Pelargonium plants, and might threaten native lycaenids because of the chance of its naturalization on indigenous Geranium spp. In Europe, C. marshalli is widespread in the Mediterranean basin, but absent in northern countries. We investigate invasion patterns and their temporal dynamics in Italy, the most extensively invaded country, identifying three phases and different rates of spread resulting from multiple introductions and human-mediated movements. We also characterize and compare the native and invasive ecological niches of C. marshalli with a multivariate approach based on bioclimatic, ecological and human demographic variables. The little overlap between the native and invaded niches (12.6%) indicates a shift in the realized niche of C. marshalli. While the expansion potential of C. marshalli in Europe remains constrained by the distribution of suitable host plants, our niche comparison analysis suggests the species has already invaded new ecological and climatic spaces. This includes colder areas than would be suggested by its native distribution in Africa.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The introduction of non-native species is a global phenomenon and one of the most important threats to biodiversity, second only to habitat destruction (Gurevitch and Padilla 2004; Roy et al. 2014). In recent years, the management of invasive species has been extensively debated. In Europe, for instance, regulations prioritise the eradication of harmful invasive organisms such as those in the Invasive Alien Species list (IAS—European Commission 2016). Yet, such programmes are rarely executed due to a lack of economic and human resources, complex logistic organisation, or ethical debates (Bertolino and Genovesi 2003; Jardine and Sanchirico 2018; Tiberti et al. 2019). Planning both preventive and longer-term management strategies is crucial to mitigate undesirable consequences, such as negative impacts on native ecosystems (Thuiller et al. 2005; Wittenberg and Cock 2001), but requires prior familiarity with alien species ecology and new knowledge of its behavior in the new invaded region, including spread ad habitat requirements. This knowledge can be gained by firstly monitoring the species with dedicated lab or field research, and then it can be used to refine predictive modelling tools that are useful for guiding preventive and management actions (Sharp et al. 2011).
One first step to predict how invasive species will colonise new environments is identifying which environmental variables (climatic, geographic, or ecological) affect the species distribution in its native range. Indeed, the success of a species in expanding its range depends on which environmental conditions it finds outside its native range. Areas outside the native range that have similar environmental conditions will be more likely to be colonized (the “climate match hypothesis”; Peterson and Robins 2003; Broennimann et al. 2007; Jiménez-Valverde et al. 2011).
A common assumption (often supported) in invasion ecology is that most invasive species conserve their native ecological niche even in the invaded area (“niche conservatism”; Wiens and Graham 2005; Pearman et al. 2008; Petitpierre et al. 2012; Strubbe et al. 2013). Recent studies suggest that at least some species are able to adapt to new conditions by shifting their native niche (Broennimann et al 2012; Strubbe et al. 2013; Guisan et al. 2014; Ørsted and Ørsted 2019). These “niche shifts” can occur in the invaded range, i.e., when a species expands its distribution in environmental conditions that were accessible but not occupied in its native range, perhaps due to the absence of competitors or predators (Keane and Crawley 2002; Oliveira et al. 2018). In other cases, shifts may be a consequence of rapid evolutionary processes acting on species under new selective pressures (Broennimann et al. 2007). Therefore, niche dynamics usually depend on case-specific ecological characteristics of a species and on the invasions stage that it is experiencing (Oliveira et al. 2018). Niche shifts can be quick, and have been described at early stages of invasion for several species (Early and Sax 2014; González‐Moreno et al. 2015).
Insects are one of the largest groups of invaders: about 9835 established alien insect species have been globally recorded (Turner et al. 2021), with an overall rate of invasions increasing at globalization events (Bonnamour et al. 2021) as they are often introduced accidentally by human-mediated movements (Roques et al. 2016). Only a small proportion of invasive insect species affects human goods and services, but some with serious economic, environmental, or human and animal health impacts (Venette and Hutchison 2021; Roques et al. 2016; Kenis et al. 2009). The global impacts and associated costs of invasive insects are grossly underestimated overall (Bradshaw et al. 2016). This situation partly results from an incomplete understanding of the ecology and distribution of invasive insect species (Diniz-Filho et al. 2010). Studies on niche dynamics in invaded ranges, notably, are often incomplete and will require more consideration in the future (Hill et al. 2017).
Some insect groups of species such as butterflies are biologically and ecologically better known and, because of their quick responses, provide ideal case studies to evaluate the effects of environmental or climatic changes (Thomas 2005).
Here, we first characterized and compared the realized niches of the butterfly Cacyreus marshalli Butler, [1898] (Lepidoptera: Lycaenidae) in its native and invaded range to assess the potential for further expansion across Europe. Cacyreus marshalli, native to southern Africa, is the only non-native butterfly in Europe (van Swaay et al. 2010) and it is potentially a threat to native biodiversity, due to the potential shift in larval foodplant and resulting competition with local butterfly species (Quacchia et al. 2008; Paradiso et al. 2019). Then, using a time series of occurrence data of the species in Italy, we examined the spatial dynamics of the expansion of its range. This work allowed us to identify key environmental factors of the ecological niches of C. marshalli in both its native (southern Africa) and non-native range (Europe). Our results provide tools to discuss the potential for range expansion of this species and management actions.
Materials and methods
Study species
The native distribution of C. marshalli includes a wide area of southern Africa: Zambia, Mozambique, Zimbabwe, Botswana, South Africa, Swaziland (Clark and Dickson 1971; Duke et al. 1999). Despite a first sighting in the United Kingdom in 1978 (Sarto i Monteys 1992), the species was first officially considered established in Europe from the record of a permanent population in Mallorca in 1989 (Eitschberger and Stamer 1990; Sarto i Monteys and Maso 1991). Cacyreus marshalli has since invaded many other European countries (Spain, Italy, Netherland, Switzerland, France, Portugal, Andorra, Malta, Slovenia, Albania, Austria, Denmark, Bulgaria, Croatia, Czech Republic, Greece, Bosnia, Germany, Montenegro, ordered by year of first record) (Maes et al. 2019). The species has been recorded in all these countries, even though its permanent establishment is not always certain. This is especially the case in northern Europe where the species is unlikely to overwinter (Paradiso et al. 2019).
In its native range, larvae of C. marshalli feed on several species of Pelargonium and Geranium (Clark and Dickson 1971). In Europe, Geraniaceae such as Geranium and Erodium are abundant but the genus Pelargonium is absent in nature. Only ornamental Pelargonium species are used as host plants out of its native range, especially P. zonale and P. peltatum. Thus, the invasion of C. marshalli in Europe probably results from Pelargonium trades. Eggs have been repeatedly reported on imports of ornamental plants, making control with insecticides difficult (Sarto i Monteys 1992). Since its first introduction, C. marshalli has never been observed feeding on wild European Geranium spp., but experimental trials in controlled conditions have shown it can oviposit and develop on some native European Geranium species (Quacchia et al. 2008). The species is polyvoltine, with up to six overlapping generations. Adults fly from May to October in the Mediterranean regions (Favilli and Manganelli 2006), while in southern Africa adults fly the whole year in warmer localities, or the summer months in cooler regions (Pringle et al. 1994).
Occurrence data on C. MARSHALLI
Occurrences in the native range
By selecting the Southern African countries where the presence of C. marshalli is confirmed by literature, i.e., South Africa, Lesotho, Swaziland and Zimbabwe (Cabral 2000; Heath et al. 2002; Gardiner 2004; Congdon et al. 2010), we obtained a total of 1744 georeferenced records of C. marshalli over the period 1867–2018.
Occurrence data have been provided by the Lepidopterists’ Society of Africa and the South African Lepidoptera Conservation Assessment project (Animal Demography Unit and Cape Town University). We did not consider GBIF records for southern Africa because of the lower number and reliability of records of the open-source database compared to the dataset provided, and the redundancies between them.
Occurrences in the invaded range
We obtained a total of 1070 georeferenced records of C. marshalli in Europe from GBIF (GBIF.org (16 October 2019) GBIF Occurrence Download https://doi.org/10.15468/dl.tjqfky). After filtering presence for reliable records in European countries in which the presence of the species is attested, i.e., Portugal, Spain, France, Italy, Switzerland, Austria, Slovenia, Croatia, Bosnia, Albania, Greece, Bulgaria, Czech Republic, Germany, Netherlands, Denmark, United Kingdom (Maes et al. 2019; Butterfly Conservation Europe personal communication), we reduced our data set to 484 georeferenced occurrence points.
The data were further integrated with 135 occurrences for nine countries obtained from the literature (Franeta 2018; Hermansen 2011; Kolev and Tsvetanov 2018; Koren and Kulijer 2016; Langourov and Simov 2014; Sachanowicz et al. 2016; Sammut 2007; Verovnik et al. 2011), and another set of 451 records for Italy from the Papilionoidea database (Balletto et al. 2007).
Spatial aggregation of records data
Since the original Italian database consisted of records mapped on a 10 × 10 km Universal Transverse Mercator (UTM) grid, and all the other records from both native and invaded ranges were available at a higher resolution, analyses were conducted at the coarser resolution (10 × 10 km). Given the low natural dispersal ability of the species (Paradiso et al. 2019), records at this resolution were considered indicative of locally established populations. C. marshalli was recorded in about 29% of the native range grid (420 out of 14,312 cells), and in about 14% of the invaded range grid (486 out of 34,567 cells, Supplementary Material 1).
Environmental data
Based on the ecology of C. marshalli, we selected fourteen environmental predictors to model the native and invaded realized niches of the species (Table 1). Climatic factors exert a strong influence on the survival and growth of ectothermic organisms and therefore are usually key predictors of invasive species’ long-term establishment probability (i.e., climate match hypothesis; Peterson 2003; Broennimann et al. 2007; Jiménez-Valverde et al. 2011).
We selected bioclimatic variables from Worldclim world databases (http://www.worldclim.org/) at a resolution of 1 km, and fitted different models from the native and invaded ranges. We considered annual mean temperature (bio1), isothermality (bio3 = (mean diurnal temperature / temperature annual range)*100), temperature seasonality (bio4 = standard deviation of the mean monthly temperature* 100), mean temperature of the warmest quarter (bio10), mean temperature of the coldest quarter (bio11), annual precipitation (bio12), precipitation seasonality (bio15 = coefficient of variation of monthly precipitations), precipitation of wettest quarter (bio16) and precipitation of driest quarter (bio17).
We also included elevation data, extracted from the Global Multi-resolution Terrain Elevation Data (GMTED2010, https://topotools.cr.usgs.gov/GMTED_viewer/) at a 1 km resolution, to capture variation in other abiotic parameters such as wind speed, partial pressure of atmospheric gases, radiation input, and a general decrease in habitat structural complexity (Hodkinson 2005).
Besides climate, habitat constraints also limit species establishment and are considered significant drivers of population dynamics in invasive species. Due to substantial differences between European and African landscapes, we categorized habitats according to their environmental permeability (Stamps et al. 1987; Wiens 1997). Indeed, insects respond to differences in vegetation structure (Stasek et al. 2008), and previous studies have shown butterflies can perceive a continuous wooded area as a barrier to dispersal and colonization (Roland et al. 2000; Casacci et al. 2015; Riva et al. 2018). We based environmental permeability on the identification of ecoregions on a global scale as established by Olson and colleagues (Olson et al. 2001), namely forests (ECO_1), ecotones (ECO_2), and grasslands (ECO_3).
In the invaded range, C. marshalli was recorded mainly from urban environments—mostly associated with ornamental Pelargonium spp. (not naturalized in Europe). Because data on the distribution of ornamental pelargoniums is not available, we considered the human population density (total number of people inhabiting per grid cell) as a proxy for the abundance of Pelargonium spp. in that cell. The human population density was retrieved from the GHS (Global Human Settlement) population grid (Joint Research Center of the European Commission 2015).
All predictors were rescaled to a common resolution (10 × 10 km). For climatic, elevation and human density data, we used the median value of each predictor. For environmental permeability, we computed the proportion of each habitat in each 10 × 10 km grid cell (ranging from 0/100 to 100/100).
Invasion patterns
Cacyreus marshalli spatial distribution in Europe
We quantified the European spatial distribution of C. marshalli. Calculating the invaded area of each country, we estimated the percentage of each European country invaded by C. marshalli.
Spatio-temporal trends of invasion in Italy
We regressed the annual distances from the first detection of C. marshalli in Italy (1996) as a function of time (Gilbert and Liebhold 2010; Liebhold et al. 1992) to evaluate its spreading process at the national level. We run a piecewise linear regression analysis (Aikio et al. 2010) using the segmented package in R (Muggeo and Muggeo 2017), to identify significant breaking points and to distinguish the different invasion phases. The significance of the breaking points was tested using a Davies test (Davies 1987). The slope of each segment provided an estimated rate of spread (V, measured in km/year) (Liebhold et al. 1992).
Relative to the assumed first area of establishment in Italy (near Rome), the spread of C. marshalli was characterized by a mixture of medium and long-distance first records. Therefore, we also investigated for specific expansion dynamics at range borders using linear quantile regressions (Groenen and Meurisse, 2012; Scharf et al. 1998). This was performed by separately regressing the longitude and latitude coordinates of the first record locations as a function of time, and repeating the procedure for the quantiles 0.05, 0.25, 0.5, 0.75 and 0.95 of the coordinates. The slope and significance of each regression model informed about possible temporal changes in the core distribution of the new records (using the median quantile = 0.5), as well as for southward/northward and westward/eastward expansions (using the more extreme quantiles, e.g., 0.05 and 0.95)”.
Moreover, we run a second piecewise linear regression analysis using the cumulative number of cells occupied by C. marshalli in Italy over time (from the first sighting in the country in 1996 until 2018) to also describe the evolution of the number of cells with confirmed records.
Niche comparison
Prior to niche comparison analysis, we mapped the environmental background of both native and invaded ranges, and overlapped it with C. marshalli distribution to visually highlight differences (Supplementary Material 1).
We then used the ecospat 3.0 package in R (Broennimann et al. 2017), whose functions provide tools to quantify and compare the niches of one or more species with an ordination approach (Di Cola et al. 2017).
After normalizing and scaling the explanatory variables, we applied a PCA (Principal Component Analysis) to reduce our 14 variables into two unrelated linear components. We calibrated the PCA on the environmental background values of both ranges; the PCA axes therefore maximized the ecological variance present in the study area. The PCA scores of the two C. marshalli distributions were projected onto a grid of cells defined by the first two axes of the PCA and delimited by minimum and maximum scores present in the study areas. The smoothed density of occurrence (oij) was then estimated in each PCA grid cell (with R = 100), using Kernel density function; this index goes from 0 (environmental circumstances in which the species has not been observed) to 1 (environmental circumstances in which the species was most commonly observed) (Broennimann et al. 2012). We calculated the niche overlap between the native and invasive range using the comparative D metric (Schoener 1968), which ranges from 0 (no overlap between the niches) to 1 (identical niches) (Broennimann et al. 2012). Moreover, we conducted two hypothesis tests (Warren et al. 2008) to evaluate the niche difference statistical significance, measured through a random relocation of the species presence points in the two ranges. We compared the actual value of D with 100 simulated values of D obtained through the overlap between two simulated niches (test of niche equivalency, evaluating niche conservatism in the strictest sense, i.e. the effective equivalency of the environmental niche; Warren et al. 2008) and 100 simulated values of D obtained through the overlap between the niche observed in a range and a simulated niche in the other range (test of niche similarity, whereby the rejection of the null hypothesis indicates that niche differentiation depends on habitat selection/suitability and it is not an artefact due to different habitat availability in the two ranges; Warren et al. 2008). Finally, to compare realized niches in native and invaded ranges, we evaluated four synthetic descriptors of the invasion process: (i) niche expansion, referred to conditions inside the invaded niche but outside the native one, taking into account the environments common to both ranges, i.e., analog environment; (ii) niche unfilling, referred to conditions inside the native niche but outside the invaded one in analog environment; (iii) niche stability, referred to conditions common to both native and invaded niches, large values of which may lead to niche conservatism; and (iv) centroid shift, the movement of the mean niche position within a multivariate environmental niche space (Petitpierre et al. 2012; Guisan et al. 2014).
All statistical and spatial analyses were performed in R version 3.3.3 (R Core Team 2017) and in QGIS “Essen” version 2.14.2 (2016).
Results
Invasion patterns
Cacyreus marshalli spatial distribution in Europe
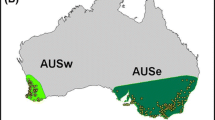
To date, C. marshalli was found in 20 European countries and across 1.5% of the European territory (52,300 km2, based on 10 × 10 km cells; Table 2). Italy (31,700 km2 invaded) and Spain (11,800 km2) are the two countries with the wider area occupied and the highest number of occurrences recorded since the 1990s (Fig. 1a). Switzerland (2200 km2) and Slovenia (1600 km2) later experienced substantial invasions. The species was recently (2016) recorded in Montenegro, Germany, and Bosnia Herzegovina, with less than three occupied cells each (Table 2).
Spatio-temporal trends of invasion in Italy
In Italy, records of the species date from 1996 near Rome (Trematerra et al. 1997; Bonelli et al. 2018). The piecewise linear regression of C. marshalli’s spread patterns in Italy (Fig. 2a) showed the presence of three relatively distinct invasion phases (a fast onset of expansion, a slower second phase of expansion, followed by a plateau). An initial lag phase, associated with the first population establishment of C. marshalli in Italy but without or with limited spatial expansion, is not clearly visible from our data. It may have taken place either earlier 1996, before the first species record, or to have been very short or unclearly defined because of multiple entries from other countries.
Piecewise linear regression of the annual distances from the Cacyreus marshalli’s first detection in Italy (a) (P < 0.05, adj.R2 = 0.171) provides rates of spread (V), corresponding to the slope of each segment (Davies test, P > 0.05), showing three possible phases of the spread process with two breakpoints in 2000 and 2006. Slopes ± SE values reveal two possible stages of expansion and a potential saturation phase for C. marshalli. Piecewise linear regression analysis of C. marshalli’s evolution of cell number with confirmed records in Italy (b) (P < 0.05, adj.R2 = 0.994), showing three phases with two significant breakpoints in 2004 and 2007 (Davies test, P > 0.05). Small horizontal bars represent confidence intervals
The trend of the distance from the first invaded cell abruptly rose during the first years of species observation (onset of the expansion phase, 1996–2000, V = 66.8 ± 30.73 km/year), probably due to a combination of short and long-distance dispersal, overall reaching more than 400 km in 8 years. The spread rates decelerated from 2000 to 2006 (second stage of the expansion phase, V = 13.7 ± 8.26 km/year), followed by a long period of very slow growth, with a slope not significantly different from zero (2006–2018, V = 0.7 ± 2.46 km/year), possibly representing a plateau (or saturation) phase (Fig. 2a).
The species does not show a geographical longitudinal and latitudinal gradient, proved by the absence of a significant range expansion from our quantile regression, another indicator of a rapid initial expansion (Supplementary Material 2).
The piecewise regression analysis of the C. marshalli’s evolution of cell number with confirmed records in Italy (Fig. 2b) revealed that the species' recorded range experienced a first slight growth (1996–2004), followed by a sharp increase (2004–2007) and then by another modest raise (2007–2018). An average number of 9.6 and 10.7 new occupied cells per year, with an average yearly percentage of 0.26% of the country invaded, characterize the first and third periods. The central period counts on average 31.5 new invaded cells per year, with on average 0.62% of the country yearly occupied. Accordingly, the Davies test revealed a significant change in slopes between these three phases (P ≤ 0.05).
Niche comparison
C. marshalli is currently more widespread in Central and South Europe, and missing from colder regions. This pattern is consistent with the bioclimatic condition typically observed in its native range, i.e., occurrences in warmer and dryer areas (Supplementary Material 1).
The first two PCA axes succeeded in capturing respectively 49.2% and 17.1% of the variance in “habitat” (i.e., the wider environmental space redefined by PCA accounting for climate, elevation, population density and environmental permeability variables) among the grid cells (Fig. 3a). The variables which contributed the most are annual mean temperature, isothermality, mean temperature of coldest quarter (axis 1), annual precipitation and precipitation of the wettest quarter (axis 2) (Supplementary Material 3, Figs. 3a and 4).
Correlation circle (a) resulting from the Principal Component Analysis, representing the single variables used and their contribution towards the two axes, and representation of Cacyreus marshalli niche overlap (b, c) between native (Africa, in blue) and invaded range (Europe, in red). The solid lines and dotted lines represent 100% and 50% of the available background, respectively. The grey shading shows the density of species occurrences per cell, in Africa (b) and Europe (c). The red arrows (c) represent how the niche centre has changed from Africa to Europe, along each of the PCA axes, linking the centroid of the native and non-native distribution (continuous line) and between native and invaded extent (dashed line). PC1 corresponds to bio_1 (Annual Mean Temperature, -0.941), bio_3 (Isothermality, -0.909), bio_11 (Mean Temperature of the Coldest Quarter,—0.947); PC2 corresponds to bio_12 (Annual Precipitation, − 0.818) and bio_16 (Precipitation of the Wettest Quarter, -0.907). The niche overlap is visible in purple and represents stability (proportion of the niche in the invaded area shared with the native area); the area of unfilling (proportion of the niche occupied in the native range but not in the secondary range) is shown in blue, and the zone of expansion (proportion of the niche occupied in the invaded area but not in the native area) is shown in red. The analog environment is defined by the black bold contour
Scatterplot of predictors; the density of presence of the species in relation to the values of variables in the native (in blue) and invaded ranges (in red) are represented; the niche overlap for each variable is shown in purple. The solid contour lines delimit the 100th quantiles of the density at the available variable, blue in the native area and red in the invaded
The niches occupied by C. marshalli in the native and invaded areas overlap by about 13% (D = 0.126). The equivalency test indicates the two niches are distinguishable (P = 0.049), but the observed differences could be a result of significant environmental differentiation between the two ranges. The similarity test indicates that niches are not more similar than would be expected by chance (P = 0.412), and thus that the realized niche in the native range cannot be used to predict the current distribution of C. marshalli in the invaded range.
We observed a centroid shift for the realized niches and an expansion of niche breadth into analog environmental space (Fig. 3b, c). Indeed, most of the analog environments have been occupied by C. marshalli (84%), as shown by the high stability index (0.839). However, expansion (0.160) and unfilling (0.055) index values indicate a movement of the species beyond the environmental and ecological borders of its native range conditions into analog environments and that, if records are found in those environments in the native range, they’re also likely to be found in the invaded range. A clear shift towards conditions present only in the invaded range is represented (Fig. 3c).
Discussion
European colonization and distribution
Often, new colonies of invasive organisms produce the ‘bridgehead effect’, whereby they expand rapidly outside their native range after the establishment of key source populations that represent a first important stepping stone to invade other regions (Garnas et al. 2016). Cacyreus marshalli appears to have followed a similar pattern. As suggested by the high rate of spread observed between 1996 and 2000 following the initial discovery (Fig. 2a), the Italian invasion was most likely originally determined by human-mediated long-range movements. Most likely, the Pelargonium trade caused multiple introductions starting from plant nurseries supplying local markets, followed by active dispersal over short distances starting from focal points (Paradiso et al. 2019). Our analysis of C. marshalli range expansion in Italy (Fig. 2) supports the hypothesis that the spread of C. marshalli marked an undefined initial rapid lag-phase (establishment) prior to 1996, followed by the onset of the expansion phase (1996–2000) defined by slow growth in occupied cell number but high spread rates (about 67 km/year) driven by long-distance human-mediated dissemination through transportation of infested plants. A slower invasion speed characterized the second stage of the expansion phase (2000–2006), dominated by short-distance local dispersal and build-up of local populations and during which, however, the occupied cell number quadrupled from 2004. Then, the expansion phase flowed into a saturation phase (2006–2018) with extremely low spread rates and lower (but still increasing) occupancy range, either because of biological constraints (Arim et al. 2006) and/or because of the filling of suitable niche environments (Williamson et al. 2005).
An extended saturation phase, with a continuous significant increase of cell number occupied by the species but low spread rates, is likely to be the consequence of a rapid initial establishment and onset of the expansion phase mediated by the broad Pelargonium trade that allowed the species to immediately reach most long-distance sites.
The observed changes in spread rate and its divergence with the invasion patterns underline the presence of different factors promoting the species spread, such as human-mediated jump dispersal (Levin 1989; Horvitz et al. 2017; Suarez et al. 2001). Other reasons explaining this variation could lie in the different levels of control and local eradication attempts throughout the invasion phases. The spatial expansion of incipient gypsy moth (Lymantria dispar Linnaeus, [1758] (Lepidoptera: Lymantriidae)) populations in North America (Liebhold et al. 1992) for instance, is affected by environmental factors (such as host availability), but also by changes in pest management practices (such as local eradication efforts, or restrictions limiting human-aided transportation of early life stages).
Potential distribution of C. marshalli
Our analysis demonstrates that a shift in the realized niche of C. marshalli took place along with the expansion from its native (African) to its invaded (European) range. This species, in fact, is characterized by a fundamental niche broader than expected (Fig. 3b, c). Temperature-related variables show a restricted niche overlap between the two ranges, mainly due to the colonization of no-analog climates in Europe (Fig. 4). In Europe, C. marshalli withstands a wide span of temperatures, which are lower on average than those represented in its native African range (Supplementary Material 1). The realized niche of the species in Europe is further constrained by the distribution of Pelargonium, its host plant, which is highly diffused as a popular balcony plant not only in the Mediterranean areas, where average temperatures are almost partially comparable with the southern-African ones, but also in the colder continental Europe and in low-mountain areas (Paradiso et al. 2019). Consequently, this has provided C. marshalli with the ability to persist on potted plants in colder areas. In any case, the species records in Europe indicate C. marshalli thrive under climatic conditions similar to the ones encountered in its native range, but remains sporadic in colder environments.
Consistently, in the invaded range, C. marshalli is mainly present at lower elevations, often associated with more favorable temperatures (Fig. 4, Supplementary Material 1). Another nonexclusive explanation is the negative correlation of elevation with population density, indicative of minor human presence at higher altitudes and therefore of a lower ornamental Pelargonium plant spread (Fig. 3a).
Furthermore, shifts in habitat choices suggest remarkable ecological plasticity of C. marshalli, a trait typical of many invasive species. In its native range, this species is primarily recorded in ecotones, while in Europe it is closely linked to prevailing woodland habitats, connected with the presence of towns or small villages (Fig. 4, Supplementary Material 1).
In southern Africa, where pelargoniums are rarely used for ornamental purposes, spontaneous Geraniaceae occur mainly in transition areas. Conversely, in Europe, despite some rare and occasional naturalization events recorded in central Italy (Guglielmone et al. 2009), pelargoniums are only grown as ornamental plants, because of their attractive inflorescences, variety of colors (Esser 1997), and resistance to adverse weather conditions such as drought.
The fact that many populated areas in Europe fall within woodland habitats is likely an artifact. Most of the continent indeed falls in the “forest” category, which consequently contains most European cities, due to a lack of definition for Europe ecoregions. Because of the bias with the used land covers, due to the differences in ecoregion definition between the two ranges, we suggest the use of better resolution land cover maps (e.g., the CORINE landcover) in future additional analyses. Niche modelling based on occurrences of a given species should always also consider biases in distribution data, due to the fact that available data are usually not random, due to different monitoring efforts (Diniz-Filho et al. 2010; Cardoso et al. 2011).
In our comparison analysis, the overlap between the native and invaded niches corresponds to approximately 13% of our defined environmental space (Fig. 3b, c). The similarity test did not yield significant similarity between the two niches, as often happens when comparing heterogeneous spatial scenarios that differ for many ecological features (Supplementary Material 1; Brown and Carnaval 2019). The native niche is partially contained in the invaded niche, but the latter has expanded into different ecological and climatic spaces and has mainly shifted away from its native niche into no-analog environments (Fig. 3b, c). The expansion of niche breadth from the native to the invaded range is likely the result of greater environmental variability in Europe.
Research on niche comparison of various plants (Atwater et al. 2018), birds (Strubbe et al. 2013) and vertebrates (Strubbe et al. 2015) has demonstrated that niche shifts are in most cases the result of unfilling, likely because of previous dispersal barriers and colonization time lag (Liu et al. 2014; Strubbe et al. 2015). The exact nature of C. marshalli shift can be interpreted by expansion and unfilling indexes, which can reveal the components of the change in the niche (Guisan et al. 2014).
The stability index has the greatest importance (> 80%), while the unfilling index has the lowest values (ca. 5%), showing how analog climate and environment have been almost fully occupied. Nevertheless, an actual realized niche shift is clearly suggested, probably driven by the distribution of Pelargonium, as shown both by the expansion index (> 10%) but even more by the rapid colonization of new no-analog environments in the invaded range (Fig. 3b, c).
Hill et al. (2017) compared niche overlaps in 22 globally invasive insect species. The niche overlap was low in all cases, while the phenomenon of expansion was evident for 12 species, and unfilling in 15 species (Hill et al. 2017). These invasive insects showed higher expansion values than other taxonomic groups, with the only exception of reptiles (Liu et al. 2014), whose climatic niche is generally more conserved along with the maximum temperatures (Liu et al. 2017). The prevalence of the unfilling phenomenon suggests that, in invasive insects, often there is a climate imbalance such that they have a potential for future niche expansions in the invaded area.
Another example of niche shift in insects is the Asian tiger mosquito Aedes albopictus Skuse, [1894] (Diptera: Culicidae). The area of invasion of this mosquito, resulting in a niche overlap of 29% and expansion and unfilling values of 28% and 7%, respectively, is strongly linked to urban areas, similarly to our results on C. marshalli (Hill et al. 2017). This species has been able to occupy new environments in the invaded range (Africa, the Middle East, Europe and America), and the invaded niche portion still potentially usable compared to the native area is very small (Gratz 2004). Despite great adaptability to climate and drought, A. albopictus has not yet been observed reproducing outside of anthropic environments.
The invasion of C. marshalli in Europe might impact populations of native lycaenids such as Aricia spp. and Eumedonia eumedon Esper, [1780] (Lepidoptera: Lycaenidae), especially if C. marshalli naturalizes on wild geraniums (Geranium spp., see Quacchia et al. 2008). The European Pollinator Initiative (IUCN 2019) recently reported potential threats for wild pollinators, mentioning C. marshalli as an alien species that might threaten native butterfly species. This risk is particularly serious in the Mediterranean basin, and in particular in Italy, where a tradition of growing pelargoniums as ornamental plants is widespread (Quacchia et al. 2008). Invasive species tend to have more competitive characteristics than native species: when sharing the same host plant, native species would probably be disadvantaged, with the risk of local extinctions or distributional changes (Quacchia et al. 2008). Audusseau et al. (2017), for example, investigated the effect of the recent Northward expansion of the butterfly Araschnia levana Linnaeus, [1758] (Lepidoptera: Nymphalidae) in Sweden on the distribution of the native Urtica-consuming species Aglais urticae and Aglais io Linnaeus, [1758] (Lepidoptera: Nymphalidae). Results showed a shift in the distribution of the two native species resulting in a reduced overlap with the invasive A. levana”.
These examples provide a unique opportunity to study how species colonize new environments, in the absence of native predators, competitors or dispersion barriers (Pearman et al. 2008). Changes in the climatic and ecological niches between different geographical areas or temporal periods are, nowadays, also mediated by climate change (Guisan et al. 2014). Climate warming, such as the over 2 °C-maximum temperature increase experienced in the Alps from the mid-1980s (Beniston 2006;), could have promoted the spread in Europe of C. marshalli and facilitated its survival during the colder months (Clark and Dickson 1971), in combination with the continuous new introductions of the species by international Pelargonium trade. Whereas, considering the species distribution, the fundamental niche of C. marshalli remains uninvestigated, the potential adaptation of this species in the invaded range cannot yet be evaluated. Demonstrating such changes would require comparing the genetic and physiological characteristics of the European and native populations (Vrba et al. 2014; Fischer and Karl 2010), and we propose it as a future research question.
General management recommendations
This study also demonstrates how important anthropic activities are in the introduction and maintenance of C. marshalli in Europe. Literature sources confirm that ornamental horticulture is among the most important sources of invasions of plants (Dehnen‐Schmutz 2007) and herbivorous insects (Roques et al. 2016). Imports and exports of pelargoniums follow various routes that involve southern Africa, Europe and the USA, as well as India and South America, particularly for essential oil extraction (https://www.seair.co.in/us-import/product-pelargonium.aspx). According to the International Register of Pelargonium Cultivars (http://thepelargoniumregister.com/), many nurseries are specialized in Pelargonium cultivar production, also in Europe, although new varieties of Pelargonium are also continuously imported from their native range. Pelargonium plants are commercially widespread because they are easy to grow, little affected by insects, resistant and carry showy and lasting blooms.
The European Commission has recently funded a project to improve the sale of ornamental Pelargonium spp. in Europe (PfE-Europe in Bloom: https://ec.europa.eu/chafea/agri/en/campaigns/pfe-europe-bloom), without outlining any measure taking into account the spread of C. marshalli in beneficiary states such as Austria, France, Germany, Italy, the Netherlands and Poland. The lack of regulation in the Pelargonium trade is at the root of multiple and continuous introductions of C. marshalli, together with the dispersal abilities shown by its females (Paradiso et al. 2019).
We call for a rise in attention towards the management of this species. Even the Alps, whose climatic conditions are extreme and usually limit the flight and dispersal of butterflies (Junker et al. 2010), have been invaded by this species. This has been encouraged by the widespread introduction of ornamental pelargoniums, particularly the cultivars most appreciated by C. marshalli (Costanzi et al. 2007), which consequently must be renewed every year in both public and private spaces. The potential threat that this species, still not listed as an IAS, represents to native biodiversity in the Alpine area should not be underestimated, because the proximity of small urban aggregates to natural ecotone habitats with abundant native Geranium spp. could prompt the host plant shift and the naturalization of this non-native butterfly species. According to Paradiso and colleagues (2019), gravid females of C. marshalli (a sedentary species in its native habitat: Clark and Dickson 1971) are able to fly over the tree canopy and other barriers, while looking for a Pelargonium plant to oviposit. These unexpected flying skills, together with the broad availability of hostplants, have allowed the rapid and widespread expansion of this species in Europe, confirmed by the small proportion of not yet occupied appropriate available niche described by the unfilling value.
Studies published on this species so far (Sarto i Monteys and Maso 1991; Quacchia et al. 2008; Paradiso et al. 2019) show that only a decrease in the ornamental Pelargonium use, combined with the choice of cultivars less appreciated by the butterfly and with meticulous control of the plant trade, could reduce the potential expansion of the species in Europe, prevent its naturalization and avoid the introduction in other continents.
In conclusion, our investigation of invasion patterns and their temporal dynamics in Italy revealed changes in spread rates and their divergence with the invasion patterns, implying a complexity of factors promoting the species spread, such as human-mediated movement or different levels of control throughout time and space.
Our results have also illustrated the existence of a realized niche shift of C. marshalli, which has expanded into analog environments from its native (African) to its invaded (European) range, and probably driven by the distribution of Pelargonium. Therefore, due to the importance of human activities in C. marshalli spread and the hypothetical future threat to native butterfly species, we encourage the application of monitoring and eradication plans where possible, to prevent its naturalization and avoid further introductions.
Data availability
Bioclimatic and ecological data used as predictors for the analysis are available online, such as most Cacyreus marshalli occurrences are open source (GBIF). Italian records can be sent under request by prof. Emilio Balletto (University of Turin), while requests for southern African records should be addressed to the Lepidopterists’ Society of Africa.
References
Aikio S, Duncan RP, Hulme PE (2010) Lag-phases in alien plant invasions: separating the facts from the artefacts. Oikos 119(2):370–378
Arim M, Abades S, Neill PE, Lima M, Marquet PA (2006) Spread dynamics of invasive species. P Natl A Sci 103(2):374–378
Atwater DZ, Ervine C, Barney JN (2018) Climatic niche shifts are common in introduced plants. Nat Ecol Evol 2(1):34–43
Audusseau H, Le Vaillant M, Janz N, Nylin S, Karlsson B, Schmucki R (2017) Species range expansion constrains the ecological niches of resident butterflies. J Biogeogr 44(1):28–38
Balletto E, Bonelli S, Cassulo LA (2007) Insecta Lepidoptera Papilionoidea. In: Ruffo S, Stoch F (eds) Checklist and Distribution of the Italian Fauna. 10,000 Terrestrial and Inland Water Species, vol 17. Memorie del Museo Civico di Storia Naturale di Verona, Italy, Verona, pp 257–261
Beniston M (2006) Mountain weather and climate: a general overview and a focus on climatic change in the Alps. Hydrobiologia 562(1):3–16
Bertolino S, Genovesi P (2003) Spread and attempted eradication of the grey squirrel (Sciurus carolinensis) in Italy, and consequences for the red squirrel (Sciurus vulgaris) in Eurasia. Biol Conserv 109(3):351–358
Bonelli S, Casacci LP, Barbero F, Cerrato C, Dapporto L, Sbordoni V, Rondinini C (2018) The first red list of Italian butterflies. Insect Conserv Diver 11(5):506–521
Bonnamour A, Gippet JM, Bertelsmeier C (2021) Insect and plant invasions follow two waves of globalisation. Ecol Lett 24(11):2418–2426
Bradshaw CJ, Leroy B, Bellard C, Roiz D, Albert C, Fournier A, Barbet-Massin M, Salles J, Simard F, Courchamp F (2016) Massive yet grossly underestimated global costs of invasive insects. Nat Commun 7:12986
Broennimann O, Treier UA, Müller-Schärer H, Thuiller W, Peterson A, Guisan A (2007) Evidence of climatic niche shift during biological invasion. Ecol Lett 10(8):701–709
Broennimann O, Fitzpatrick MC, Pearman PB, Petitpierre B, Pellissier L, Yoccoz NG et al (2012) Measuring ecological niche overlap from occurrence and spatial environmental data. Global Ecol Biogeogr 21(4):481–497
Broennimann O, Petitpierre B, Randin C, Engler R, Di Cola V, Breiner F, Guisan A et al (2017) ecospat: Spatial ecology miscellaneous methods. R Package version 3.0. https://CRAN.R-project.org/package=ecospat
Brown JL, and Carnaval AC (2019) A tale of two niches: methods, concepts, and evolution. Front Biogeogr 11(4)
Cabral A (2000) Borboletas de Moçambique. Grupo IPG, Maputo and Lisbon, p 69
Cardoso P, Erwin TL, Borges PA, New TR (2011) The seven impediments in invertebrate conservation and how to overcome them. Biol Conserv 144(11):2647–2655
Casacci LP, Cerrato C, Barbero F, Bosso L, Ghidotti S, Paveto M, Viterbi R (2015) Dispersal and connectivity effects at different altitudes in the Euphydryas aurinia complex. J Insect Conserv 19(2):265–277
Clark GC, Dickson CGC (1971) Life histories of the South African lycaenid butterflies. Purnell, Switzerland
Congdon C, Collins S, Bayliss J (2010) Butterflies of south east Africa’s mountains (Mozambique and Malawi). Metamorphosis 21(2):45–107
Costanzi M, Malausa LC, Groussier G, Tabone E, Ris N, Alma A, Ferracini C, Quacchia A (2007) Strategie di lotta biologica per contenere fitofagi esotici di recente introduzione nella zona transfrontaliera italo-francese, a salvaguardia delle diverse realtà ambientali. Progetto Interreg III Alcotra (UE) Italia-Francia pp 8–39
Davies RB (1987) Hypothesis testing when an invasive parameter is present only under the alternative. Biometrika 74:33–43
Dehnen-Schmutz K, Touza J, Perrings C, Williamson M (2007) A century of the ornamental plant trade and its impact on invasion success. Divers Distrib 13(5):527–534
Di Cola V, Broennimann O, Petitpierre B, Breiner FT, D’amen M, Randin C, Engler R, Pottier J, Pio D, Dubuis A, Pellissier L, Mateo RJ, Hordijk W, Salamin N, Guisan A (2017) Ecospat: an R package to support spatial analyses and modeling of species niches and distributions. Ecography 40(6):774–787
Diniz-Filho JAF, De Marco JRP, Hawkins BA (2010) Defying the curse of ignorance: perspectives in insect macroecology and conservation biogeography. Insect Conserv Diver 3(3):172–179
Duke N, Saunders J, Saunders C (1999) A checklist of insects of Swaziland. The Conservation Trust of Swaziland and The Natural History Society of Swaziland, Swaziland
Early R, Sax DF (2014) Climatic niche shifts between species’ native and naturalized ranges raise concern for ecological forecasts during invasions and climate change. Global Ecol Biogeogr 23(12):1356–1365
Eitschberger U, Stamer P (1990) Cacyreus marshalli Butler, 1898, eine neue Tagfalterart für sie europäische Fauna? (Lepidoptera, Lycaenidae). Atalanta 21(1/2):101–108
Esser M (1997) Pelargoniums. Rebo International, Olanda
European Commission (2016) Commission Implementing Regulation (EU) 2016/1141 of 13 July 2016 adopting a list of invasive alien species of Union concern pursuant to Regulation (EU) No 1143/2014 of the European Parliament and of the Council. Off J Eur Un L 189(4): 4–8
Favilli L, Manganelli G (2006) Life history of Cacyreus marshalli, a South African species recently introduced into Italy (Lepidoptera Lycaenidae). Boll Della Soc Entomol Italiana 138(1):51–61
Fischer K, Karl I (2010) Exploring plastic and genetic responses to temperature variation using copper butterflies. Clim Res 43(1–2):17–30
Franeta F (2018) Checklist of the butterflies (Lepidoptera: Papilionoidea) of Montenegro. Zootaxa 4392(1):128–148
Gardiner A (2004) Chapter 10. Butterflies of the four corners area. In: Timberlake JR, Childers SL (eds) Biodiversity of the Four Corners Area: Technical Review, vol 15. Occasional Publications on Biodiversity, Bulawayo, pp 381–397
Garnas JR, Auger-Rozenberg MA, Roques A, Bertelsmeier C, Wingfield MJ, Saccaggi DL, Slippers B (2016) Complex patterns of global spread in invasive insects: eco-evolutionary and management consequences. Biol Invasions 18(4):935–952
Gilbert M, Liebhold A (2010) Comparing methods for measuring the rate of spread of invading populations. Ecography 33(5):809–817
González-Moreno P, Diez JM, Richardson DM, Vilà M (2015) Beyond climate: disturbance niche shifts in invasive species. Global Ecol Biogeogr 24(3):360–370
Gratz NG (2004) Critical review of the vector status of Aedes albopictus. Med Vet Entomol 18(3):215–227
Groenen F, Meurisse N (2012) Historical distribution of the oak processionary moth Thaumetopoea processionea in Europe suggests recolonization instead of expansion. Agric for Entomol 14(2):147–155
Guglielmone L, Gallo L, Guiggi A (2009) The Italian “Succulent Flora”: a taxonomical, chorological and historical approach. Bocconea 23:345
Guisan A, Petitpierre B, Broennimann O, Daehler C, Kueffer C (2014) Unifying niche shift studies: insights from biological invasions. Trends Ecol Evol 29(5):260–269
Gurevitch J, Padilla DK (2004) Are invasive species a major cause of extinctions? Trends Ecol Evol 19(9):470–474
Heath A, Newport MA, and Hancock D (2002) Butterflies of Zambia. Afr Butterfly Res Inst.
Hermansen K (2011) Fund af dagsommerfuglen, Cacyreus marshalli (Butler, 1898) i Danmark. Lepidoptera bind X, 2
Hill MP, Gallardo B, Terblanche JS (2017) A global assessment of climatic niche shifts and human influence in insect invasions. Global Ecol Biogeogr 26(6):679–689
Hodkinson ID (2005) Terrestrial insects along elevation gradients: species and community responses to altitude. Biol Rev 80(3):489–513
Horvitz N, Wang R, Wan FH, Nathan R (2017) Pervasive human-mediated large-scale invasion: analysis of spread patterns and their underlying mechanisms in 17 of China’s worst invasive plants. J Ecol 105(1):85–94
IUCN (2019) Managing invasive alien species to protect wild pollinators. Technical guidance prepared for the European Commission under contract No 07.0202/2018/795538/SER/ENV.D.2 “Technical support related to the implementation of the EU Pollinators Initiative”
Jardine SL, Sanchirico JN (2018) Estimating the cost of invasive species control. J Environ Econ Manag 87:242–257
Jiménez-Valverde A, Peterson AT, Soberón J, Overton JM, Aragón P, Lobo JM (2011) Use of niche models in invasive species risk assessments. Biol Invasions 13(12):2785–2797
Junker M, Wagner S, Gros P, Schmitt T (2010) Changing demography and dispersal behaviour: ecological adaptations in an alpine butterfly. Oecologia 164(4):971–980
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17(4):164–170
Kenis M, Auger-Rozenberg MA, Roques A, Timms L, Péré C, Cock MJ, Settele J, Augustin S, Lopez-Vaamonde C (2009) Ecological effects of invasive alien insects. Biol Invasions 11(1):21–45
Kolev Z, Tsvetanov T (2018) Clarifications and new data on the distribution of Cacyreus marshalli Butler, 1898 in Bulgaria (Insecta, Lepidoptera, Lycaenidae). ZooNotes 122:1–4
Koren T, Kulijer D (2016) New or interesting records of three butterfly (Lepidoptera: Papilionoidea and Hesperioidea) species from Bosnia and Herzegovina and Croatia. Natura Croatica 25(2):321–326
Langourov M, Simov N (2014) Cacyreus marshalli Butler, 1898 (Lep.: Lycaenidae), a new species for Bulgaria. Entomol Record J Var 126:190–192
Levin SA (1989) Analysis of risk for invasions and control program. Biol Invasion Glob Perspect SCOPE 37:425–435
Liebhold AM, Halverson JA, Elmes GA (1992) Gypsy moth invasion in North America: a quantitative analysis. J Biogeogr 513–520
Liu X, Li X, Liu Z, Tingley R, Kraus F, Guo Z, Li Y (2014) Congener diversity, topographic heterogeneity and human-assisted dispersal predict spread rates of alien herpetofauna at a global scale. Ecol Lett 17(7):821–829
Liu X, Petitpierre B, Broennimann O, Li X, Guisan A, Li Y (2017) Realized climatic niches are conserved along maximum temperatures among herpetofaunal invaders. J Biogeogr 44(1):111–121
Maes D, Verovnik R, Wiemers M, Brosens D, Beshkov S, Bonelli S, Buszko J, Cantú-Salazar L, Cassar LF, Collins S, Dincă V, Djuric M, Dušej G, Elven H, Franeta F, Garcia-Pereira P, Geryak Y, Goffart P, Gór Á, Hiermann U, Höttinger H, Huemer P, Jakšić P, John E, Kalivoda H, Kati V, Kirkland P, Komac B, Kőrösi Á, Kulak A, Kuussaari M, L’Hoste L, Lelo S, Mestdagh X, Micevski N, Mihoci I, Mihut S, Monasterio-León Y, Morgun DV, Munguira ML, Murray T, Nielsen PS, Ólafsson E, Õunap E, Pamperis LN, Pavlíčko A, Pettersson LB, Popov S, Popović M, Pöyry J, Prentice M, Reyserhove L, Ryrholm N, Šašić M, Savenkov N, Settele J, Sielezniew M, Sinev S, Stefanescu C, Švitra G, Tammaru T, Tiitsaar A, Tzirkalli E, Tzortzakaki O, van Swaay CAM, Viborg AL, Wynhoff I, Zografou K, Warren MS (2019) Integrating national red lists for prioritising conservation actions for European butterflies. J Insect Conserv 23(2):1–30
Muggeo VM, Muggeo MVM (2017) Package ‘segmented.’ Biometrika 58:525–534
Oliveira BF, Costa GC, Fonseca CR (2018) Niche dynamics of two cryptic prosopis invading South American drylands. Biol Invasions 20(1):181–194
Olson DM, Dinerstein E, Wikramanayake ED, Burgess ND, Powell GV, Underwood EC, D’amico JA, Itoua I, Strand HE, Morrison JC, Allnutt TF, Ricketts TH, Kura Y, Lamoreux JF, Wettengel WW, Hedao P, Kassem KR, Loucks CJ (2001) Terrestrial ecoregions of the world: a new map of life on earth: a new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. Bioscience 51(11):933–938
Orsted IV, Orsted M (2019) Species distribution models of the spotted wing Drosophila (Drosophila suzukii, Diptera: Drosophilidae) in its native and invasive range reveal an ecological niche shift. J Appl Ec 56(2):423–435
Paradiso F, Martelli F, Cerrato C, Ghidotti S, Viterbi R, Canterino S, Ferracini C, Bonelli S (2019) From Africa to the Alps; risk assessment on an invasion by Cacyreus marshalli (Butler, 1898). J Insect Conserv 23(2):279–288
Pearman PB, Guisan A, Broennimann O, Randin CF (2008) Niche dynamics in space and time. Trends Ecol Evol 23(3):149–158
Peterson AT, Robins CR (2003) Using ecological-niche modeling to predict barred owl invasions with implications for spotted owl conservation. Conserv Biol 17(4):1161–1165
Petitpierre B, Kueffer C, Broennimann O, Randin C, Daehler C, Guisan A (2012) Climatic niche shifts are rare among terrestrial plant invaders. Science 335(6074):1344–1348
Pringle EL, Henning GA, Ball JB (eds) (1994) Pennington’s Butterflies of southern Africa, edition 2. Struik Winchester, Cape Town
Quacchia A, Ferracini C, Bonelli S, Balletto E, Alma A (2008) Can the Geranium Bronze, Cacyreus marshalli, become a threat for European biodiversity? Biodivers Conserv 17(6):1429–1437
Quantum GIS Development Team - Version 2.14.2 (2016) Quantum GIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.osgeo.org
R Core Team (2017) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Riva F, Acorn JH, Nielsen SE (2018) Narrow anthropogenic corridors direct the movement of a generalist boreal butterfly. Biol Let 14(2):20170770
Roland J, Keyghobadi N, Fownes S (2000) Alpine Parnassius butterfly dispersal: effects of landscape and population size. Ecology 81(6):1642–1653
Roques A, Auger-Rozenberg A, Blackburn TM, Garnas J, Pyšek P, Rabitsch W, Duncan RP (2016) Temporal and interspecific variation in rates of spread for insect species invading Europe during the last 200 years. Biol Invasions 18(4):907–920
Roy HE, Peyton J, Aldridge DC, Bantock T, Blackburn TM, Britton R, Dobson M et al (2014) Horizon scanning for invasive alien species with the potential to threaten biodiversity in Great Britain. Glob Change Biol 20(12):3859–3871
Sachanowicz K, Łuczkowski S, Larysz A (2016) State of knowledge of butterfly fauna of Albania, with three new species for the country. Acta Zool Bulgar 68(4):511–518
Sammut P (2007) Cacyreus marshalli Butler, [1898] reaches the Maltese Islands (Lepidoptera: Lycaenidae). SHILAP Revista De Lepidopterología 35(139):317–319
Sarto i Monteys V (1992) Spread of the Southern African Lycaenid butterfly, Cacyreus marshalli Butler, 1898, (LEP: Lycaenidae) in the Balearic Archipelago (Spain) and considerations on its likely introduction to continental Europe. J Res Lepidoptera 31(1–2):24–34
Sarto i Monteys V, Maso A (1991) Confirmation of Cacyreus marshalli Butler, 1898 (Lycaenidae: Polyommatinae) as a new species for the European fauna. Boletín De Sanidad Vegetal, Plagas 17(1):173–183
Scharf FS, Juanes F, Sutherland M (1998) Inferring ecological relationships from the edges of scatter diagrams: comparison of regression techniques. Ecology 79(2):448–460
Schoener TW (1968) Anolis lizards of Bimini: resource partitioning in a complex fauna. Ecology 49:704–726
Sharp RL, Larson LR, Green GT (2011) Factors influencing public preferences for invasive alien species management. Biol Conserv 144(8):2097–2104
Stamps JA, Buechner M, Krishnan VV (1987) The effects of edge permeability and habitat geometry on emigration from patches of habitat. Am Nat 129(4):533–552
Stasek DJ, Bean C, Crist TO (2008) Butterfly abundance and movements among prairie patches: the roles of habitat quality, edge, and forest matrix permeability. Environ Entomol 37(4):897–906
Strubbe D, Broennimann O, Chiron F, Matthysen E (2013) Niche conservatism in non-native birds in Europe: niche unfilling rather than niche expansion. Global Ecol Biogeogr 22(8):962–970
Strubbe D, Beauchard O, Matthysen E (2015) Niche conservatism among non-native vertebrates in Europe and North America. Ecography 38(3):321–329
Suarez AV, Holway DA, Case TJ (2001) Patterns of spread in biological invasions dominated by long-distance jump dispersal: insights from Argentine ants. P Natl A Sci 98(3):1095–1100
Thomas JA (2005) Monitoring change in the abundance and distribution of insects using butterflies and other indicator groups. Phil T R Soc B 360:339–357
Thuiller W, Richardson DM, Pyšek P, Midgley GF, Hughes GO, Rouget M (2005) Niche-based modelling as a tool for predicting the risk of alien plant invasions at a global scale. Glob Change Biol 11(12):2234–2250
Tiberti R, Bogliani G, Brighenti S, Iacobuzio R, Liautaud K, Rolla M, von Hardenberg A, Bassano B (2019) Recovery of high mountain Alpine lakes after the eradication of introduced brook trout Salvelinus fontinalis using non-chemical methods. Biol Invasions 21(3):875–894
Trematerra P, Zilli A, Valentini V, Mazzei P (1997) Cacyreus marshalli, a new pest of geranium in Italy. Informatore Fitopatologico 47(7/8):2–6
Turner R, Blake R, and Liebhold AM (2021) International Non-native Insect Establishment Data (0.1), Zenodo
Van Swaay C, Cuttelod A, Collins S, Maes D, Munguira ML, Šašić M, Settele J, Verovnik R, Verstrael T, Warren Ma, Wynhoff I, and Wiemers M (2010) European red list of butterflies. Luxembourg: Publications Office of the European Union (Crvena knjiga)
Venette RC, Hutchison WD (2021) Invasive insect species: global challenges, strategies and opportunities. Fron Insect Sci 1:1
Verovnik R, Polak S, Seljak G (2011) Pojav in øirjenje tujerodne vrste dnevnega metulja-pelargonijevega bakrenœka (Cacyreus marshalli (Butler 1898)) v sloveniji. Acta Entomologica Slovenica 19:1
Vrba P, Dolek M, Nedvěd O, Zahradníčková H, Cerrato C, Konvička M (2014) Overwintering of the boreal butterfly Colias palaeno in Central Europe. CryoLetters 35(3):247–254
Warren DL, Glor RE, Turelli M (2008) Environmental niche equivalency versus conservatism: quantitative approaches to niche evolution. Evolution 62(11):2868–2883
Wiens JA (1997) Metapopulation dynamics and landscape ecology. Metapopulation biology. Academic Press, Elsevier, pp 43–62
Wiens JJ, Graham CH (2005) Niche conservatism: integrating evolution, ecology, and conservation biology. Annu Rev Ecol Evol S 36:519–539
Williamson M, Pyšek P, Jarošík V, Prach K (2005) On the rates and patterns of spread of alien plants in the Czech Republic, Britain, and Ireland. Ecoscience 12(3):424–433
Wittenberg R, Cock MJW (2001) Invasive Alien species: a toolkit of best prevention and management practices. CAB International, Wallingford, Oxon, UK, p xvii:228
Acknowledgements
We are grateful to all the scientists from different European countries who shared the Cacyreus marshalli distribution data with us, namely Per Stadel Nielsen, Miloš Popović, Benjamin Komac, Suvad Lelo, Rudi Verovnik, Filip Franeta, Stoyan Beshkov, Zdravko Kolev, and Mario Langourov. Special thanks are due to the Lepidopterists’ Society of Africa, the South African Lepidoptera Conservation Assessment (SALCA), the Animal Demography Unit (ADU) and the University of Cape Town, for their kindness in sending us the C. marshalli presence database of Africa. We are also grateful to Dr. Massimo Meregalli, for the help and valuable knowledge on territories and the biodiversity of southern Africa, and to Dr. Laura Guglielmone, for providing us with valuable information and botanical material about the Geraniaceae family. Special thanks to Prof. Emilio Balletto, Dr. Martha Gibson, Dr. Federico Riva, Emi Husband and Emma White for the precious contribution in the revision phase.
Funding
Open access funding provided by Università degli Studi di Torino within the CRUI-CARE Agreement. The project is a collaboration between the Gran Paradiso National Park and the Department of Life Sciences and Systems Biology of Turin University, and was funded by the Gran Paradiso National Park (Italian Ministry for Ecological Transition).
Author information
Authors and Affiliations
Contributions
S.B., C.C., F.M. designed the study; F.M., C.C. collected occurrences data and performed the statistical analyses; F.P. realized species distribution maps; F.M., F.P., S.B., C.C. discussed the results; F.M. wrote the first draft of the manuscript, which was substantially improved through the contribution of F.P., S.B., S.G., C.C. All authors commented on the manuscript. S.B. and C.C. contributed equally to this study.
Corresponding author
Ethics declarations
Conflicts of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martelli, F., Paradiso, F., Ghidotti, S. et al. Invasion patterns and niche comparison of the butterfly Cacyreus marshalli among native and non-native range. Biol Invasions 24, 3935–3952 (2022). https://doi.org/10.1007/s10530-022-02895-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-022-02895-3