Abstract
Invasive mosquitoes, Aedes aegypti (African) and Aedes albopictus (Asian), serve as global vectors for diseases such as dengue, Zika, and chikungunya, and they are currently present in Colombia. Consequently, the aim of this study was to assess the potential geographic distribution of these A. aegypti and A. albopictus vectors under both present and future environmental conditions in Colombia. To achieve this, a dataset was compiled using global records for each species, with those from Colombia being used for model validation. These records, in conjunction with layers of bioclimatic variables, were incorporated into ecological niche models. Predictions of potential distribution were made with the Maxent algorithm, using the Wallace EcoMod application for current conditions and the Java MaxEnt software for future projections, which represented three climate change scenarios (SSP245, SSP370, and SSP585) for two time periods (2041–2060 and 2081–2100). Additionally, niche overlap was calculated, and the suitable habitat range and population at risk in Colombia were quantified for current predictions. It is suggested that both species share ideal areas in the country with an overlap of 0.85, covering over 70% of the country's territory and reaching altitudes higher than 2,500 m, potentially affecting at least 45% of the population. In the future for both species it is estimated that, with increasing temperatures, the potential distribution could decrease, but they could invade areas with altitudes above 3,500 m (colder). It is concluded that, currently, A. aegypti and A. albopictus could have similar geographic distribution in Colombia and that, in the following years, climate change could lead to latitudinal and altitudinal variations in the distribution areas of these vectors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Aedes aegypti (African) and Aedes albopictus (Asian) mosquitoes are significant invasive species involved in the transmission of arboviral diseases (Rey and Lounibos 2015). In their native regions, both species serve as primary vectors for diseases with global public health implications, including yellow fever, dengue, chikungunya, and Zika (Paupy et al. 2009; Souza-Neto et al. 2019; Lwande et al. 2020). In the Americas, both species are distributed throughout the continent, with records of A. aegypti dating back around 300 years (Powell et al. 2018) and currently acting as the primary vector for dengue, chikungunya, and Zika (Urdaneta-Marquez and Failloux 2011; Ferreira-de-Brito et al. 2016; Costa-da-Silva et al. 2017). Meanwhile, A. albopictus has been recorded for 40 years and has the potential to be a significant vector in the Americas (Garcia-Rejon et al. 2021). This is supported by the discovery of dengue and Zika infected populations of A. albopictus (Figueiredo et al. 2010; Sanchez-Rodriguez et al. 2014; Correa-Morales et al. 2019; Alencar et al. 2021). Moreover, A. albopictus has been reported as infected with these arboviruses during outbreaks in Mexico and Brazil (Ibáñez-Bernal et al. 1997; Ricas et al. 2020).
The spread of these species to new locations beyond their native range is closely linked to human activities, particularly trade, especially maritime trade, and tourism, among others (Lounibos 2002; Bonizzoni et al. 2013; Eritja et al. 2017; Eskildsen et al. 2018). Furthermore, the presence of these species is associated with temperature and rainfall, which influence the abundance and distribution of their populations. In warmer climates, their life cycle accelerates, facilitating disease transmission, while in colder climates, their development is negatively affected, consequently impacting their vectorial capacity (Marinho et al. 2016; Cruz Ferreira et al. 2017; Ezeakacha and Yee 2019). Consequently, in areas where these species are found, A. aegypti reproduces in urban settings within artificial containers containing stagnant water, such as tires, barrels, water tanks, and other receptacles (Getachew et al. 2015; Ferede et al. 2018). In contrast, A. albopictus prefers suburban or rural habitats, where it reproduces in natural reservoirs like tree holes or bromeliads, and even in artificial containers (Gratz 2004; Delatte et al. 2008; Rao 2010). However, it is worth noting that A. aegypti presence and reproduction have also been detected in rural and suburban areas (Pérez-Castro et al. 2016; Djiappi-Tchamen et al. 2021; Dalpadado et al. 2022), while A. albopictus has been observed in urban areas (Abílio et al. 2018; Camargo et al. 2021).
The eggs of these species possess a unique characteristic in their shell, allowing them to withstand desiccation (Ezeakacha and Yee 2019) and enter a state of diapause when environmental conditions are unfavorable for hatching and survival (Diniz et al. 2017; Lee and Duvall 2022). Consequently, in their adult form, invasive populations of A. albopictus can establish themselves in transitional zones between the tropical and temperate regions of the planet, where A. aegypti cannot thrive. This is why their presence has been documented in northern parts of the United States and even in northern European countries (Thomas et al. 2012; Vega-Rúa et al. 2014). On the other hand, A. aegypti is distributed primarily in tropical areas, with some presence in subtropical regions, where it can establish itself in temperate climates during the warm months of the year (Kamal et al. 2018).
Currently, in Colombia, A. aegypti is present in approximately 80% of the continental territory up to an elevation of 2,600 m (Ruiz-López et al. 2016). In contrast, A. albopictus has been identified in certain municipalities in Colombia (Vélez et al. 1998; Suárez 2001; Cuéllar-Jiménez et al. 2007; Gutiérrez et al. 2011; Rúa-Uribe et al. 2011; Zamora et al. 2015; Carvajal et al. 2016; Ortiz-Canamejoy and Villota 2018). However, recent estimates from our research group suggest that A. albopictus could potentially inhabit as much as 96.14% of the country's territory up to an elevation of 3,000 m (Echeverry-Cárdenas et al. 2021). In Colombia, A. aegypti is the primary focus of vector control programs (Ardila-Roldán et al. 2013; Caldera et al. 2013; Aguirre-Obando et al. 2015; Conde et al. 2015; Cabezas et al. 2017). On the other hand, due to limited research and the relatively low transmission of arboviruses reported by A. albopictus in Colombia, this mosquito currently holds a somewhat secondary role in vector control programs, despite sporadic detections of dengue-infected specimens (Gómez-Palacio et al. 2017).
Given the epidemiological significance of both A. aegypti and A. albopictus, understanding their potential geographic distribution in Colombia is crucial due to their significant role in the country's epidemiological landscape and the potential threats they pose to public health (Pérez-Castro et al. 2016; Gómez-Palacio et al. 2017). Ecological niche modeling (ENM) serves as a valuable tool for predicting their distribution and associated epidemiological risk identifying regions with optimal environmental conditions for their presence (Soberón and Peterson 2005; Peterson et al. 2011). Extensive research using ENM has explored the potential distribution of A. aegypti and A. albopictus across various geographic scales. For example, ENM has been globally applied to both species, providing insights into their distribution in the past (Kraemer et al. 2019), present (Kraemer et al. 2015), and future (Kamal et al. 2018; Laporta et al. 2023). Studies on A. aegypti have scrutinized its potential distribution at both local levels (city and country) (Mweya et al. 2013; Fatima et al. 2016; Estallo et al. 2018) and globally (Liu-Helmersson et al. 2019). Similarly, research on A. albopictus has investigated its potential distribution at local levels (city and country) (Neteler et al. 2011; Kalan et al. 2017) and continental levels (Fischer et al. 2014; Oliveira et al. 2021). However, there is currently no study estimating the potential distribution of both mosquitoes specifically for Colombia. These comprehensive studies contribute valuable insights into the potential distribution of these mosquito species, aiding in the development of informed vector control strategies and our understanding of the dynamic interplay between these vectors and their environment. This information is essential for the strategic planning and execution of vector prevention and control measures, enabling the identification of high-risk areas.
This approach allows us to identify potential mosquito habitats, guiding the targeting of vector control programs. Additionally, it helps pinpoint areas where potential distributions of both species overlap, assess the human population's potential exposure to disease transmission, estimate their latitudinal and altitudinal distribution range, and predict future changes to their ranges given climate change (Kamal et al. 2018; Echeverry-Cárdenas et al. 2021). Furthermore, recognizing the potential impact of climate change on the distribution of these vectors is of utmost significance, as it provides a more precise understanding of how environmental conditions and, consequently, the distribution of these mosquitoes may evolve in the future. ENM has proven to be effective in estimating the possible distribution of diseases such as malaria and yellow fever, as well as the distribution of their respective vectors (Liria and Navarro 2010; Altamiranda-Saavedra et al. 2017; Uusitalo et al. 2019). Consequently, the objective of this study was to assess the potential geographic distribution of both A. aegypti and A. albopictus vectors under both current and future environmental conditions in Colombia.
Methods
Study area
Colombia is located in the northwestern corner of South America, sharing borders with Venezuela, Brazil, Peru, Ecuador, and Panama. Geographically, Colombia is divided into 32 departments and covers a total area of 2,070,408 km2, of which 1,141,748 km2 are continental territory and 928,660 km2 are maritime territory. On the other hand, geologically, the country is defined by a mountainous region that traverses it from north to south, known as the Colombian Andes. This mountainous region is divided into three ranges, or cordilleras: the Western Cordillera, the Central Cordillera, and the Eastern Cordillera. These cordilleras extend across various departments, including Antioquia, Boyacá, Caldas, Cauca, Cundinamarca, Huila, Nariño, Norte de Santander, Quindío, Risaralda, Santander, Tolima, and Valle del Cauca (Gómez et al. 2020) (Fig. 1). Additionally, Colombia can be divided into six regions with varying climatic conditions. The Andean region, being the most mountainous, exhibits a range of climates due to differences in altitude, encompassing tropical and subtropical climates that vary from temperate to cold, including high-mountain areas. In the Caribbean region, a tropical climate with high temperatures prevails throughout the year, while the Pacific region experiences a warm and humid climate with high levels of rainfall. The Orinoco region is characterized by a semi-arid climate with distinct rainy and dry seasons, while the Amazon region features a tropical and equatorial climate with high levels of precipitation throughout the year. The insular region comprises the Colombian Caribbean islands of San Andrés and Providencia, where a maritime tropical climate prevails, with warm and consistent temperatures year-round (Gómez et al. 2020) (Fig. 1).
To estimate the population at risk of arbovirus transmission, a search was conducted in the most recent population census by departments on the DANE website for the year 2018 (Departamento Administrativo Nacional de Estadística—https://www.dane.gov.co/). For this purpose, the total number of inhabitants in departments with a suitability probability > 0.5 was calculated under the current context (Table1). The information was delimited by region and department, and the population density was calculated using the area and population data for each department.
Records of occurrence
To determine the potential geographic distribution of A. aegypti (african) and A. albopictus (asian) in Colombia, global records for each species were collected from the GBIF database (GBIF 2023a, 2023b; https://www.gbif.org/). Additional records of their occurrence in Colombia were sourced from scientific articles (Supplementary Material 1). This dataset underwent a thorough cleaning process to remove duplicate occurrences, records with geospatial issues, and multiple occurrences within a single 5 km radius pixel. This cleaning was performed using R (CoreTeam 2017) in conjunction with the following packages: sp 1.4–1 (Pebesma et al. 2020), raster 3.4–5 (Hijmans et al. 2020a) and dismo 1.3–3 (Hijmans et al. 2020b). The resulting datasets for each species were employed for model calibration and validation. The calibration phase utilized global records for each species (A. aegypti n = 9,248; A. albopictus n = 10,884), while all records obtained for Colombia were used for validation (A. aegypti n = 482; A. albopictus n = 113).
Environmental variables
To estimate the current models for both species, we obtained the 19 bioclimatic variables available in Worldclim v2.1 (www.worldclim.org) at a spatial resolution of 2.5 min (approximately 5 km) (Fick and Hijmans 2017) due to the country-scale approach, as has been done in other studies (Lippi et al. 2019; Adeogun et al. 2023). To address multicollinearity issues among these variables, we applied a Variance Inflation Factor (VIF) using the occurrence records that were previously cleaned for each species (Pradhan 2016; Jiménez-García and Peterson 2019). This was performed within the R environment and involved the use of raster 3.4–5 (Hijmans et al. 2020a) and usdm 1.1–18 (Naimi 2017) packages. The environmental variables with low correlations were selected as the basis for current and future estimations, taking into account the variables that accounted for at least 90% of the contribution in the model (Table2).
Additionally, to estimate the potential future distribution, we used the selected bioclimatic variables under the Shared Socio-economic Pathways (SSP) 245, 370, and 585 scenarios for the periods 2041–2060 and 2081–2100. The Intergovernmental Panel on Climate Change (IPCC) has incorporated new scenarios called Shared Socioeconomic Pathways (SSP) in its 6th assessment report (CMIP6). These scenarios, including SSP245, SSP370, and SSP585, estimate different levels of greenhouse gas emissions and CO2 concentrations over time, based on the potential human actions taken to mitigate and adapt to climate change (O’Neill et al. 2017). Specifically, the SSP245 scenario anticipates moderate mitigation policies and technological advancements, leading to lower emissions and a moderate increase in global temperatures. In contrast, the SSP370 scenario predicts a world with less ambitious mitigation policies, higher emissions, and a significant rise in temperature. Finally, the SSP585 scenario envisions a future characterized by rapid economic growth, high energy demand, and continued reliance on fossil fuels, resulting in a pronounced increase in emissions and a notable temperature rise (Eyring et al. 2016; Riahi et al. 2017). These scenarios emphasize the importance of addressing climate change and implementing effective mitigation and adaptation measures to prevent the potential expansion of disease vectors like A. aegypti and A. albopictus, which can have significant public health implications (Jarvis et al. 2008; Caminade et al. 2019).
These variables were extracted from WorldClim v2.1 at a resolution of 2.5 min, and we employed various Global Circulation Models (GCM) to represent different climate sensitivities. The selection of climate models was carefully considered, focusing on equilibrium climate sensitivity (ECS). Our approach included models with ECS values ranging from 2.5 to 4 °C, such as MRI-ESM2-0, MPI-ESM1-2-HR, MIROC-ES2L, and GISS-E2-1-H, providing a nuanced exploration of climate scenarios within this range. To encompass a broader spectrum, models with ECS values exceeding 4 °C, specifically IPSL-CM6A-LR and UKESM1-0-LL. This diverse set of ECS values ensures a comprehensive coverage of potential climate conditions, enhancing the robustness and reliability of our modeling approach (Flynn and Mauritsen 2020; Hausfather et al. 2022).
Ecological niche modeling
All modelling were conducted using the maximum entropy algorithm, implemented in the MaxEnt software (Phillips et al. 2006). To achieve this, the rasters were masked to the extents of the accessible areas (M) of the mosquitoes using the global occurrence records. To perform the estimations of the current models for A. aegypti and A. albopictus, the Wallace EcoMod application run from R package wallace (Kass et al. 2018). In this platform, the accessible area (M) was adjusted by employing a polygon that covers global occurrences with a 0.2 buffer, excluding Colombia's polygon. We then sampled 10,000 background points randomly over the global polygon after removing the polygon representing Colombia. Spatial partitioning was employed for cross-validation, and the regularization multiplier was implemented within a range of 0–4, with the parameters Linear (L), Quadratic (Q), Hinge (H), and Product (P), using combinations of L, LQ, H, LQH, and LQHP (Jiménez-García and Peterson 2019; Valencia-Rodríguez et al. 2021). The models were selected based on the mean validation AUC average and were projected for Colombia, allowing us to determine the potential current geographic distribution of A. aegypti and A. albopictus.
Future estimations were conducted using the maxent.jar v3.4.3 software. Ten replicas were executed, each consisting of 5,000 iterations, using the complementary log–log output format and the "Do clamping" parameter. Extrapolation parameter was disabled to prevent extrapolations into non-analogous climates (Kamal et al. 2018). To obtain a final map that represented each emission scenario in both periods, the average between the predictions generated with each selected GCM was calculated (Outammassine et al. 2022). The results were presented in continuous format, and environmental suitability in mountainous areas was delineated in altitude range bins using a binary format, applying the threshold rule of the 10th percentile training presence. Additionally, the potential expansion area was quantified in all scenarios.
Given that A. aegypti and A. albopictus are important global vectors of arboviral diseases, understanding whether their niches overlap in Colombia is crucial due to their role in disease transmission and implications for public health. Based on available records of A. aegypti and A. albopictus in Colombia, it is hypothesized that both species may share niches and coexist. Therefore, niche overlap between both species in Colombia was calculated using 1,000 permutations to assess its statistical significance. This analysis was conducted in R software using the raster v3.4–5 (Hijmans et al. 2020a) and the PCA-env approach (Broennimann et al. 2012) inside ecospat v4.0.0 package (Di Cola et al. 2017), utilizing the estimates obtained under current environmental conditions.
Validation of the models
To validate the current model projections for Colombia, the NicheToolBox application run from R package ntbox (Osorio-Olvera et al. 2020) was utilized, using mosquito occurrences in this country. This approach involved calculating the area under the curve (AUC) and conducting a partial analysis of the Receiver Operating Characteristics (ROC-partial) for a more comprehensive assessment of model performance. In this analysis, the parameter E was set to 0.05 per 1000 iterations. The significance values were interpreted as follows: p > 0.05 suggests that the estimates are not superior to those generated by a random model, while AUC with p < 0.05 indicates that the estimated predictions are statistically superior to those obtained from a random model (Peterson et al. 2008; Echeverry-Cárdenas et al. 2021).
Results
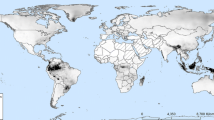
Globally, we obtained 9,248 curated occurrences for A. aegypti (Fig. 2a) and 10,884 for A. albopictus (Fig. 2b). In Colombia, the occurrences recorded for A. aegypti identify the species in 24 departments, primarily in the western part of the country, with records in the Caribbean, Andean, Pacific, Orinoco, and Amazon regions, ranging from sea level up to 3,540 m. In contrast, A. albopictus is recorded in 10 departments, with records in the Pacific, Andean, Orinoco, and Amazon regions, ranging from sea level up to 1,671 m (Fig. 2c).
On the other hand, following the selection and reduction tests, a total of 5 bioclimatic variables were obtained (Table2), which were used in the execution of the different proposed models.
Estimates in the current context suggest that A. aegypti (AUC = 0.75 and ROC partial p = 0.00) and A. albopictus (AUC = 0.74 and ROC partial p = 0.00) are distributed widely across the country with probabilities between 0.40 and 0.60 (Table 3). The calculation of niche overlap between both species resulted in an overlap value of 0.85 and p = 0.00, signifying that both species extensively share their ecological niche in Colombia, implying a substantial overlap in habitat and potential distribution range. Additionally, Table1 shows the population potentially at risk of arbovirus transmission, where A. aegypti would affect around 30 million people, A. albopictus around 26 million, and both approximately 20 million, with the Caribbean region potentially being the most affected. In Fig. 3, environmental suitability areas are presented on a probability scale, along with an altitude-delimited map, suggesting that A. aegypti (Fig. 3 Ib) and A. albopictus (Fig. 3 IId) have a potential distribution at altitudes ranging from 1,500 to 2,999 m around the Andes and the Sierra Nevada de Santa Marta (the highest altitude zone in the Caribbean region). Additionally, it is suggested that A. aegypti has a high likelihood of presenting suitable zones in all regions, with the insular region and the Orinoco being the most suitable, covering their entire extent. In the Caribbean region, the highest suitability is found throughout the department of La Guajira, with smaller areas in the departments of Magdalena, Cesar, Atlántico, Bolívar, Sucre, and Córdoba, which are densely populated (Table1). In the Andean region, the most populated in the country (Table1), the departments with the highest suitability areas are Norte de Santander, Antioquia, Huila, and Tolima. In the Amazon region, the Guaviare department shows high suitability, with some areas in Guainía, Vaupés, Caquetá, and Amazonas. Conversely, lower suitability is found in the Pacific region, as well as in the Andes and the Sierra Nevada de Santa Marta (Fig. 3I). Nevertheless, the latter has the potential to reach altitudes of up to 2,500 m (Fig. 3 Ib). Both species exhibit high environmental suitability on the islands of San Andrés and Providencia (Fig. 3).
Furthermore, the current estimation for A. albopictus suggests high suitability across areas in all regions, among which the Insular, Orinoco, and Amazon exhibit the highest probability throughout their geographical extent, regions with the lowest population (Table1). In the other regions, higher suitability areas are observed in different parts of all departments, covering a large portion of La Guajira, Norte de Santander, and Tolima. Similarly, lower environmental suitability is found in the high-altitude areas of the Andes Mountain range and in the Sierra Nevada de Santa Marta (Fig. 3II). Nevertheless, it has the potential distribution at altitudes of up to 2,500 m in the Sierra Nevada de Santa Marta (Fig. 3 IId). In general, A. aegypti exhibits higher suitability in all Colombian regions (suitability > 0.60) than A. albopictus (suitability ranging from 0.40 to 0.60) (Table 3).
Estimations in the context of climate change suggest that both A. aegypti and A. albopictus are expected to experience a reduction in the most suitable areas of their ranges across all regions. However, on average, over a period of 58 years, they have the potential to expand their altitudinal distribution range, reaching elevations between 3,500 and 3,999 m. In general, for the period 2041–2060, both species are projected to have potential distribution areas across much of Colombia, with a small percentage (up to 6.4%) having suitability values exceeding 0.60 (Table 3). During this period, both A. aegypti and A. albopictus exhibit decreased suitability probabilities in the Caribbean, Andean, and Pacific regions. However, high suitability probabilities are maintained in some areas of La Guajira department and around the Sierra Nevada de Santa Marta. Additionally, suitability is suggested in departments surrounding the mountain ranges and, in the Orinoco, and Amazon regions (Fig. 4). In the insular region, A. aegypti maintains a high probability of suitability under all three scenarios (Fig. 4 Ia, b, and c), whereas A. albopictus shows a decreased probability compared to the current context (Fig. 4 IId, e, f). During this period, both species could reach altitudes between 3,000 and 3,499 m under the SSP370 and SSP585 scenarios (Fig. 4 Ib, c, IIe, f). Specifically, A. aegypti has the potential to occupy areas between 2,500 and 2,999 m under the SSP585 scenario (Fig. 4 Ic).
For the period 2081–2100, a decrease in the area with suitability > 0.60 and between 0.40 and 0.60 is anticipated. Despite this, there is an increase in the area occupied in zones with suitability between 0.20 and 0.40 (Table 3). This suggests that high temperatures over extended periods could potentially influence the expansion of mosquitoes into areas that previously had a cooler climate. During this period, the probability of suitability is maintained in the insular region, La Guajira, around the mountain ranges, the Sierra Nevada de Santa Marta, and in the Orinoco and Amazon regions. For both species, a low probability is estimated in the high-altitude zones of the mountain ranges, the Pacific and Caribbean regions, as well as in the southern Amazon for A. aegypti under the SSP245 and SSP370 scenarios (Figs. 5 Ia, b). On the other hand, for both mosquitoes, it is suggested that under the SSP370 and SSP585 scenarios, there is increased suitability around the mountain ranges, covering a large extent of the Andean region, which is currently the most populated region in the country (Figs. 5 Ib, c, IIe, f). Additionally, A. aegypti exhibits environmental suitability for distribution at altitudes higher than 2,500 m under all three scenarios, reaching altitudes exceeding 3,500 m under the SSP370 and SSP585 scenarios, and even expanding its range to altitudes between 2,500 and 2,999 m (Figs. 5 Ib, c). For A. albopictus, altitudes exceeding 3,500 m are estimated under the SSP585 scenario (Fig. 5 IIf). Regarding the insular region, potential areas are estimated for both species, with A. aegypti being the mosquito with higher environmental suitability in both time periods and under all three climate change scenarios (Fig. 4 and 5).
Discussion
The primary vector for diseases like dengue, Zika, and chikungunya in Colombia is A. aegypti (Vega-Rúa et al. 2014; Pérez-Castro et al. 2016; Musso et al. 2018), while the epidemiological role of A. albopictus remains less understood in the country. Niche modeling for both species in this study suggests a wide area of potential distribution across the country with high suitability in various localities within each region. Furthermore, based on the records obtained, A. aegypti and A. albopictus are found in 75% and 31% of Colombia's departments, respectively. Our estimates also indicate distribution over a large portion of the country, with a potential range occupied of 83% for A. aegypti and 80% for A. albopictus (with suitability > 0.40). Currently, there are no studies relating to the total extent occupied by A. aegypti in Colombia, however, it is suggested that this mosquito occupies 80% of the national territory (Ruiz-López et al. 2016). Meanwhile, A. albopictus has been estimated in the 80% of Colombian territory for the present day (Echeverry-Cárdenas et al. 2021).
The current distribution of A. aegypti and A. albopictus in Colombia indicates altitudes ranging from 0 to 2,600 m and 0 to 1,800 m, respectively. Niche modeling estimates project suitable areas for both species at altitudes between 0 to 2,999 m in mountainous regions and the Sierra Nevada de Santa Marta. While A. aegypti exhibits a broader potential range, covering over 80% of Colombia with low suitability in the Pacific and Amazon regions, A. albopictus shows high suitability over a significant portion of the Amazon. Discrepancies among studies regarding their distribution in Colombia may arise from differences in model calibration and projection methods. For Colombia, there are three niche modeling studies which have contributed valuable insights into the potential distribution of Aedes aegypti and Aedes albopictus. The first study focused on A. aegypti, revealing high suitability in areas around the Andes Mountains and the Caribbean region, while suitability was low in the rest of the country (Portilla Cabrera and Selvaraj 2020). The second study focused on A. albopictus, showing suitability across a significant portion of the country and at altitudes exceeding 1,500 m (Echeverry-Cárdenas et al. 2021). The third study, which is a Master's thesis that has not been published yet, involved estimating the potential distribution for both species based on records spanning from 1979 to 2013. In this study, suitability was observed around the Andes Mountains and in the Caribbean region, with lower suitability noted in the rest of the country. Our estimations under the current context suggest that both species would exhibit environmental suitability across a significant portion of the country, but would not present environmental suitability in a part of the coastal region of the Pacific Ocean. Particularly, A. aegypti would not be present in much of the Amazon and A. albopictus in a part of La Guajira.
The present estimations considered all available occurrences, including their native areas in Africa and Asia, as well as occurrences in Colombia. Global data, excluding that from Colombia, was modeled and projected onto the study region. The estimations in this study were conducted using the new CMIP6 climate models and climate change scenarios (SSP245, 370, and 585) (Eyring et al. 2016; Riahi et al. 2017). It is evident that these updated models and climate change scenarios, such as CMIP6 and SSP245, SSP370, and SSP585, will indeed provide a more current and comprehensive understanding of the potential distribution of A. aegypti and A. albopictus in Colombia (Eyring et al. 2016; Riahi et al. 2017). Considering the inclusion of occurrences from both their native areas in Africa and Asia, as well as data from Colombia, these estimations provided valuable insights into how these mosquito species may respond to changing environmental conditions, particularly in the context of climate change. Although previous studies found increases or decreases in the suitability of areas nationwide for both species (Portilla Cabrera and Selvaraj 2020; Echeverry-Cárdenas et al. 2021), our study provides important regional context showing increases in suitability in some regions (Pacific and Andean), but decreases in others (Caribbean, Orinoco, and Amazon), demonstrating that future range shifts may exhibit spatial heterogeneity. This latter aspect indicates the persistent potential for both species to establish in much of the country, emphasizing the need for adaptive vector control and public health strategies.
The biological cycle of mosquitoes is influenced by environmental variables such as temperature, precipitation, and humidity (Tabachnick 2016; Reinhold et al. 2018). Specifically, the optimal temperature for the development of A. aegypti and A. albopictus falls between 20 and 32 °C, and they respond negatively to temperatures exceeding 39 °C and dropping below 16 °C (Marinho et al. 2016; Reinhold et al. 2018; Ezeakacha and Yee 2019). In the current and future estimations, unsuitable zones are suggested in the departments located in the Pacific region (Chocó, Valle, Cauca, Nariño). This region of Colombia is known to be one of the wettest in the neotropics, with annual precipitation ranging from 8,000 to 10,000 mm and maximum annual temperatures of 32.2 °C (Poveda-M et al. 2004; Rangel-ch and Arellano-P 2017). Therefore, being a region with high rainfall, the biological cycle of mosquitoes can be affected as excess moisture and precipitation impact the development of pupae and larvae (Costa et al. 2010; Dhimal et al. 2015).
Additionally, all estimations suggest decreases in the Caribbean region (La Guajira, Atlántico, Magdalena, Cesar, Sucre, Bolívar, and Córdoba) and the Orinoco region (Arauca, Casanare, Meta, Vichada, Guaviare, Guainía, Vaupés, Caquetá, and Amazonas). These areas are characterized by an average temperature ranging from 25 to 28 °C with relative humidity below 75% in some areas (La Guajira and Cesar) (IDEAM 2010). Consequently, in these regions in the future under the influence of climate change, it would become a region with high temperatures that could affect mosquito development, as at least a 0.6 °C (± 0.5) increase is estimated for the period 2041 – 2070 nationwide (IDEAM 2010).
According to climate change scenarios estimated in Colombia by the Institute of Hydrology, Meteorology, and Environmental Studies (IDEAM), for the period 2011–2070, the central-western region (around the Andean region) and the northern region (Caribbean region) are expected to experience a decrease in precipitation between 5 and 10%. In contrast, the eastern and southeastern regions (Orinoquia and Amazon regions, respectively) are expected to see increases between 5 and 25% (IDEAM 2010). Consequently, in the northern part of the country, the mosquito's biological cycle may be affected due to increased temperatures and decreased precipitation, while in the eastern part of the country, it may be affected by increased temperatures and excessive precipitation.
On the other hand, the distribution of these species in the future remains consistent over time and under different scenarios in a region of the western part. It is anticipated that in the future, the departments located in the Colombian Andes (Tolima, Santander, Risaralda, Quindío, Norte de Santander, Huila, Cundinamarca, Caldas, Boyacá, Antioquia, Cauca, and Nariño) will experience increased temperature and precipitation (López-Latorre and Neira 2016), potentially creating a suitable environment for the establishment of these species in these areas in the future. For example, in the Pacific region and the central-southern part of the western region (around the Andean region), an increase in precipitation between 5 and 15% is estimated (IDEAM 2010).
A study that assessed the ecophysiological plasticity of A. aegypti and A. albopictus revealed that the main barrier for A. aegypti egg hatching is low temperatures. However, populations found at lower altitudes can adapt to lower temperatures (Kramer et al. 2021). Additionally, A. albopictus exhibits greater success in its distribution due to its characteristics in its place of origin, ecological plasticity, and the ability for egg diapause (Diniz et al. 2017; Kramer et al. 2021). These findings differ from what was obtained in the current study, as our estimations suggest that A. aegypti has greater plasticity than A. albopictus to adapt to higher altitudes. Nevertheless, there are no studies on interspecific competition between these species in Colombia, so our observations would be subject to further research addressing this topic.
Passive transport plays a significant role in the dispersion of mosquitoes at very large geographic scales where these species couldn't naturally reach (Lounibos 2002; Lwande et al. 2020; Wilke et al. 2022). Colombia has several major maritime ports that handle a wide variety of cargo going to and from the country (Cantillo et al. 2018). These ports, along with the primary land transportation routes, connect different geographic areas throughout the country and can provide a pathway for the entry and exit of vectors to new locations. This can complicate the epidemiological landscape of public health (Eritja et al. 2017; Bennett et al. 2019). For example, a study documented the presence of A. aegypti and A. albopictus in a major Colombian seaport, which led to the first record of A. albopictus in western Colombia (Suárez 2001). Furthermore, a mathematical modeling study conducted by our research group suggests that passive transport mediated by cargo trucks plays an important role in the dispersion of A. aegypti and its potential capacity to transmit the Mayaro virus on a Colombian scale (Valencia-Marín et al. 2020).
The calculation of overlap between A. aegypti and A. albopictus suggests the potential coexistence of both species in Colombia. This coexistence between the two mosquitoes may arise through habitat segregation, as previous studies have shown the predominance of A. aegypti in urban areas and A. albopictus in rural areas, allowing for their coexistence in suburban areas (Braks et al. 2003; Hopperstad et al. 2021). Until now, studies on the coexistence of these species in Colombia have not been conducted, even though the presence of both A. aegypti and A. albopictus in the same locality has been reported (Ortiz-Canamejoy and Villota 2018; Camacho-Gómez and Zuleta 2019; Gómez-Vargas and Zapata-Úsuga 2019). However, the fact that these species share the same habitat may lead to competitive displacement between them, which influences their distribution and establishment (Rey and Lounibos 2015). For instance, studies have demonstrated that A. albopictus has a competitive advantage in its larval stage, as its population growth remains unaffected in environments with low nutrient levels, leading to the displacement of A. aegypti (Braks et al. 2004; Deerman and Yee 2023). In this context, the displacement of one of the mosquitoes can play a significant role in their geographic distribution, as previous studies have shown the disappearance or reduction of A. aegypti populations following the appearance of A. albopictus (O’Meara et al. 1995; Kaplan et al. 2010; Bagny et al. 2012). However, conducting competition studies is recommended to either confirm or refute our hypothesis regarding the coexistence of both species in Colombia.
In conclusion, at present, A. aegypti and A. albopictus are potentially widely distributed in Colombia, inhabiting altitudes from 0 to 2,999 m, with an occupancy area exceeding 80%. In the future, rising temperatures could enable these mosquito species to spread to higher altitudes than they currently inhabit, allowing them to expand into previously uncolonized areas. Currently, it is expected that the distribution of these mosquitoes will be influenced by climate change in Colombia. This information helps identify potential habitats for these vectors, allowing for the targeted implementation of vector control programs. Furthermore, it is suggested that due to the extensive potential distribution of A. albopictus, it has the potential to play an important role as a secondary vector for the same arboviruses transmitted by A. aegypti. However, future studies are recommended to confirm this hypothesis.
References
Abílio AP, Abudasse G, Kampango A et al (2018) Distribution and breeding sites of Aedes aegypti and Aedes albopictus in 32 urban/peri-urban districts of Mozambique: implication for assessing the risk of arbovirus outbreaks. PLoS Negl Trop Dis 12:1–15. https://doi.org/10.1371/journal.pntd.0006692
Adeogun A, Babalola AS, Okoko OO et al (2023) Spatial distribution and ecological niche modeling of geographical spread of Anopheles gambiae complex in Nigeria using real time data. Sci Rep 13:1–18. https://doi.org/10.1038/s41598-023-40929-5
Aguirre-Obando OA, Dalla Bona AC, Duque LJE, Navarro-Silva MA (2015) Insecticide resistance and genetic variability in natural populations of Aedes (Stegomyia) aegypti (Diptera: Culicidae) from Colombia. Zoologia 32:14–22. https://doi.org/10.1590/S1984-46702015000100003
Alencar J, De MCF, Marcondes CB et al (2021) Natural infection and vertical transmission of zika virus in sylvatic mosquitoes Aedes albopictus and Haemagogus leucocelaenus from Rio de Janeiro, Brazil. Trop Med Infect Dis 6:1–10
Altamiranda-Saavedra M, Arboleda S, Parra JL et al (2017) Potential distribution of mosquito vector species in a primary malaria endemic region of Colombia. PLoS ONE 12:1–14. https://doi.org/10.1371/journal.pone.0179093
Ardila-Roldán S, Santacoloma L, Brochero H (2013) Estado de la sensibilidad a los insecticidas de uso en salud pública en poblaciones naturales de Aedes aegypti (Diptera: Culicidae) del departamento de Casanare, Colombia. Biomedica 33:446–458. https://doi.org/10.7705/biomedica.v33i3.1534
Bagny L, Arnoux S, Delatte H et al (2012) Spread of invasive Aedes albopictus and decline of resident Aedes aegypti in urban areas of Mayotte 2007–2010. Biol Invasions 14:1623–1633. https://doi.org/10.1007/s10530-012-0177-1
Bennett KL, Gómez Martínez C, Almanza A et al (2019) High infestation of invasive Aedes mosquitoes in used tires along the local transport network of Panama. Parasit Vectors 12:264. https://doi.org/10.1186/s13071-019-3522-8
Bonizzoni M, Gasperi G, Chen X, James AA (2013) The invasive mosquito species Aedes albopictus: Current knowledge and future perspectives. Trends Parasitol 29:460–468. https://doi.org/10.1016/j.pt.2013.07.003
Braks MAH, Honório NA, Lourenço-De-Oliveira R et al (2003) Convergent habitat segregation of Aedes aegypti and Aedes albopictus (Diptera: Culicidae) in Southeastern Brazil and Florida. J Med Entomol 40:785–794. https://doi.org/10.1603/0022-2585-40.6.785
Braks MAH, Honório NA, Lounibos LP et al (2004) Interspecific competition between two invasive species of container mosquitoes, Aedes aegypti and Aedes albopictus (Diptera: Culicidae), in Brazil. Ann Entomol Soc Am 97:130–139. https://doi.org/10.1603/0013-8746(2004)097[0130:ICBTIS]2.0.CO;2
Broennimann O, Fitzpatrick MC, Pearman PB et al (2012) Measuring ecological niche overlap from occurrence and spatial environmental data. Glob Ecol Biogeogr 21:481–497. https://doi.org/10.1111/j.1466-8238.2011.00698.x
Cabezas L, Cabanzo W, Santa F et al (2017) Distribución espacial del mosquito Aedes aegypti (Diptera: Culicidae) en el área rural de dos municipios de Cundinamarca, Colombia. Biomedica 37:1–30
Caldera SM, Jaramillo MC, Cochero S et al (2013) Diferencias genéticas entre poblaciones de Aedes aegypti de municipios del norte de Colombia, con baja y alta incidencia de dengue. Biomedica 33:89–98. https://doi.org/10.7705/biomedica.v33i0.1573
Camacho-Gómez M, Zuleta LP (2019) Primer reporte de Aedes (Stegomyia) albopictus (Skuse) en la Orinoquía Colombiana. Biomedica 39:785–797
Camargo C, Alfonso-Parra C, Díaz S et al (2021) Spatial and temporal population dynamics of male and female Aedes albopictus at a local scale in Medellín, Colombia. Parasit Vectors 14:1–15. https://doi.org/10.1186/s13071-021-04806-2
Caminade C, McIntyre KM, Jones AE (2019) Impact of recent and future climate change on vector-borne diseases. Ann N Y Acad Sci 1436:157–173. https://doi.org/10.1111/nyas.13950
Cantillo J, Cantillo V, Arellana J (2018) Modelling with joint choice of ports and countries of origin and destination: application to Colombian ports. Marit Policy Manag 45:720–738. https://doi.org/10.1080/03088839.2018.1440090
Carvajal JJ, Honorio NA, Díaz SP et al (2016) Detección de Aedes albopictus (Skuse) (Diptera: Culicidae) en el municipio de Istmina, Chocó, Colombia. Biomedica 36:438–446
Conde M, Orjuela LI, Castellanos CA et al (2015) Evaluación de la sensibilidad a insecticidas en poblaciones de Aedes aegypti (Diptera: Culicidae) del departamento de Caldas, Colombia, en 2007 y 2011. Biomedica 35:43–52. https://doi.org/10.7705/biomedica.v35i1.2367
CoreTeam R (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, R Cran, Vienna, Austria
Correa-Morales F, González-Acosta C, Mejía-Zúñiga D et al (2019) Surveillance for zika in Mexico: naturally infected mosquitoes in urban and semi-urban areas. Pathog Glob Health 113:309–314. https://doi.org/10.1080/20477724.2019.1706291
Costa-da-Silva AL, Ioshino RS, Petersen V et al (2017) First report of naturally infected Aedes aegypti with chikungunya virus genotype ECSA in the Americas. PLoS Negl Trop Dis 11:1–11. https://doi.org/10.1371/journal.pntd.0005630
Cuéllar-Jiménez ME, Velásquez-Escobar OL, González-Obando R, Morales-Reichmann CA (2007) Detección de Aedes albopictus (Skuse) (Diptera: Culicidae) en la ciudad de Cali, Valle del Cauca, Colombia. Biomedica 27:273–279. https://doi.org/10.7705/biomedica.v27i2.224
da Cruz Ferreira DA, Degener CM, De Almeida M-T et al (2017) Meteorological variables and mosquito monitoring are good predictors for infestation trends of Aedes aegypti, the vector of dengue, chikungunya and Zika. Parasit Vectors 10:1–11. https://doi.org/10.1186/s13071-017-2025-8
Dalpadado R, Amarasinghe D, Gunathilaka N, Ariyarathna N (2022) Bionomic aspects of dengue vectors Aedes aegypti and Aedes albopictus at domestic settings in urban, suburban and rural areas in Gampaha district, western province of Sri Lanka. Parasit Vectors 15:1–14. https://doi.org/10.1186/s13071-022-05261-3
de Costa EAP, de Santos EM, Correia JC, de Albuquerque CMR (2010) Impact of small variations in temperature and humidity on the reproductive activity and survival of Aedes aegypti (Diptera, Culicidae). Rev Bras Entomol 54:488–493. https://doi.org/10.1590/S0085-56262010000300021
de Figueiredo ML, de Gomes A, Amarilla AA et al (2010) Mosquitoes infected with dengue viruses in Brazil. Virol J 7:1–5. https://doi.org/10.1186/1743-422X-7-152
Delatte H, Dehecq JS, Thiria J et al (2008) Geographic distribution and developmental sites of Aedes albopictus (Diptera: Culicidae) during a chikungunya epidemic event. Vector-Borne Zoonotic Dis 8:25–34. https://doi.org/10.1089/vbz.2007.0649
Deerman H, Yee DA (2023) Competitive interactions with Aedes albopictus alter the nutrient content of Aedes aegypti. Med Vet Entomol 37:715–722. https://doi.org/10.1111/mve.12675
Dhimal M, Gautam I, Joshi HD et al (2015) Risk factors for the presence of chikungunya and dengue vectors (Aedes aegypti and Aedes albopictus), their altitudinal distribution and climatic determinants of their abundance in central Nepal. PLoS Negl Trop Dis 9:e0003545. https://doi.org/10.1371/journal.pntd.0003545
Di Cola V, Broennimann O, Petitpierre B et al (2017) ecospat: an R package to support spatial analyses and modeling of species niches and distributions. Ecography (cop) 40:774–787
Diniz DFA, De Albuquerque CMR, Oliva LO et al (2017) Diapause and quiescence: dormancy mechanisms that contribute to the geographical expansion of mosquitoes and their evolutionary success. Parasit Vectors 10:1–13. https://doi.org/10.1186/s13071-017-2235-0
Djiappi-Tchamen B, Nana-Ndjangwo MS, Tchuinkam T et al (2021) Aedes mosquito distribution along a transect from rural to urban settings in Yaoundé, Cameroon. InSects. https://doi.org/10.3390/insects12090819
Echeverry-Cárdenas E, López-Castañeda C, Carvajal-Castro JD, Aguirre-Obando OA (2021) Potential geographic distribution of the tiger mosquito Aedes albopictus (Skuse, 1894) (Diptera: Culicidae) in current and future conditions for Colombia. PLoS Negl Trop Dis 15:e0008212. https://doi.org/10.1101/2020.03.13.990440
Eritja R, Palmer JRB, Roiz D et al (2017) Direct evidence of adult Aedes albopictus dispersal by car. Sci Rep 7:1–15. https://doi.org/10.1038/s41598-017-12652-5
Eskildsen GA, Rovira JR, Smith O et al (2018) Maternal invasion history of Aedes aegypti and Aedes albopictus into the Isthmus of Panama: implications for the control of emergent viral disease agents. PLoS ONE 13:1–19. https://doi.org/10.1371/journal.pone.0194874
Estallo EL, Sangermano F, Grech M et al (2018) Modelling the distribution of the vector Aedes aegypti in a central Argentine city. Med Vet Entomol 32:451–461. https://doi.org/10.1111/mve.12323
Eyring V, Bony S, Meehl GA et al (2016) Overview of the coupled model intercomparison project phase 6 (CMIP6) experimental design and organization. Geosci Model Dev 9:1937–1958. https://doi.org/10.5194/gmd-9-1937-2016
Ezeakacha NF, Yee DA (2019) The role of temperature in affecting carry-over effects and larval competition in the globally invasive mosquito Aedes albopictus. Parasit Vectors 12:1–11. https://doi.org/10.1186/s13071-019-3391-1
Fatima HS, Atif S, Rasheed BS et al (2016) Species distribution modelling of Aedes aegypti in two dengue-endemic regions of Pakistan. Trop Med Int Heal 21:427–436. https://doi.org/10.1111/tmi.12664
Ferede G, Tiruneh M, Abate E et al (2018) Distribution and larval breeding habitats of Aedes mosquito species in residential areas of northwest Ethiopia. Epidemiol Health 40:e2018015. https://doi.org/10.4178/epih.e2018015
Ferreira-de-Brito A, Ribeiro IP, de Miranda RM et al (2016) First detection of natural infection of Aedes aegypti with Zika virus in Brazil and throughout South America. Mem Inst Oswaldo Cruz 111:655–658. https://doi.org/10.1590/0074-02760160332
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Fischer D, Thomas SM, Neteler M et al (2014) Climatic suitability of Aedes albopictus in europe referring to climate change projections: comparison of mechanistic and correlative niche modelling approaches. Eurosurveillance 19:1–13. https://doi.org/10.2807/1560-7917.ES2014.19.6.20696
Flynn CM, Mauritsen T (2020) On the climate sensitivity and historical warming evolution in recent coupled model ensembles. Atmos Chem Phys 20:7829–7842. https://doi.org/10.5194/acp-20-7829-2020
Garcia-Rejon JE, Navarro JC, Cigarroa-Toledo N, Baak-Baak CM (2021) An updated review of the invasive Aedes albopictus in the americas; geographical distribution, host feeding patterns, arbovirus infection, and the potential for vertical transmission of dengue virus. InSects. https://doi.org/10.3390/insects12110967
GBIF.org (2023a) GBIF Occurrence Download. https://doi.org/10.15468/dl.fs2kdu
GBIF.org (2023b) GBIF Occurrence Download. https://doi.org/10.15468/dl.4w22ym
Getachew D, Tekie H, Gebre-Michael T et al (2015) Breeding sites of Aedes aegypti: potential dengue vectors in Dire Dawa, east Ethiopia. Interdiscip Perspect Infect Dis 2015:1–8. https://doi.org/10.1155/2015/706276
Gómez J, Núñez-Tello A, Mateus-Zabala D et al (2020) Physiographic and geological setting of the Colombian territory. In: Gómez J, Mateus-Zabala D (eds) The geology of Colombia proterozoic – paleozoic. Servicio geológico Colombiano. Publicaciones Geológicas Especiales, Bogotá, pp 1–16
Gómez-Palacio A, Suaza-Vasco J, Castaño S et al (2017) Infección de Aedes albopictus (Skuse, 1894) con el genotipo asiático-americano del virus del dengue serotipo 2 en Medellín y su posible papel como vector del dengue en Colombia. Biomedica 37:135–142. https://doi.org/10.7705/biomedica.v34i2.3474
Gómez-Vargas W, Zapata-Úsuga G (2019) Presence of Aedes aegypti and Aedes albopictus (Diptera: Culicidae) in the rural area of the department of santander, Colombia. Biosalud 18:7–21. https://doi.org/10.17151/biosa.2019.18.1.1
Gratz NG (2004) Critical review of the vector status of Aedes albopictus. Med Vet Entomol 18:215–227. https://doi.org/10.1111/j.0269-283X.2004.00513.x
Gutiérrez M, Almeida O, Barrios H et al (2011) Hallazgo de Aedes albopictus (Diptera: Culicidae) en el municipio de Barrancabermeja, Colombia. Biomedica 31:26–26
Hausfather Z, Marvel K, Schmidt GA et al (2022) Climate simulations: recognize the ‘hot model’ problem. Nature 605:26–29. https://doi.org/10.1038/d41586-022-01192-2
Hijmans R, Etten J, Sumner M et al (2020a) Package “raster” Geographic data analysis and modeling
Hijmans R, Phillips S, Leathwick J, Elith J (2020b) Package “dismo”. Species distribution modeling
Hopperstad KA, Sallam MF, Reiskind MH (2021) Estimations of fine-scale species distributions of Aedes aegypti and Aedes albopictus (Diptera: Culicidae) in Eastern Florida. J Med Entomol 58:699–707. https://doi.org/10.1093/jme/tjaa216
Ibáñez-Bernal S, Briseño B, Mutebi JP et al (1997) First record in America of Aedes albopictus naturally infected with dengue virus during the 1995 outbreak at Reynosa, Mexico. Med Vet Entomol 11:305–309
IDEAM (2010) Atlas climatológico de Colombia. http://atlas.ideam.gov.co/cclimatologicas/index.html. Acessed 25 March 2023
Jarvis A, Lane A, Hijmans RJ (2008) The effect of climate change on crop wild relatives. Agric Ecosyst Environ 126:13–23. https://doi.org/10.1016/j.agee.2008.01.013
Jiménez-García D, Peterson AT (2019) Climate change impact on endangered cloud forest tree species in Mexico. Rev Mex Biodivers. https://doi.org/10.22201/IB.20078706E.2019.90.2781
Kalan K, Ivović V, Glasnović P, Buzan E (2017) Presence and potential distribution of Aedes albopictus and Aedes japonicus japonicus (Diptera: Culicidae) in Slovenia. J Med Entomol 54:1510–1518. https://doi.org/10.1093/JME/TJX150
Kamal M, Kenawy MA, Rady MH et al (2018) Mapping the global potential distributions of two arboviral vectors Aedes aegypti and Aedes albopictus under changing climate. PLoS ONE 13:1–21. https://doi.org/10.1371/journal.pone.0210122
Kaplan L, Kendell D, Robertson D et al (2010) Aedes aegypti and Aedes albopictus in Bermuda: extinction, invasion, invasion and extinction. Biol Invasions 12:3277–3288. https://doi.org/10.1007/s10530-010-9721-z
Kass JM, Vilela B, Aiello-Lammens ME et al (2018) Wallace: a flexible platform for reproducible modeling of species niches and distributions built for community expansion. Methods Ecol Evol 9:1151–1156. https://doi.org/10.1111/2041-210X.12945
Kraemer MUG, Sinka ME, Duda KA et al (2015) The global distribution of the arbovirus vectors Aedes aegypti and Ae. albopictus Elife 4:e08347. https://doi.org/10.7554/eLife.08347
Kraemer MUG, Reiner RC, Brady OJ et al (2019) Past and future spread of the arbovirus vectors Aedes aegypti and Aedes albopictus. Nat Microbiol 4:854–863. https://doi.org/10.1038/s41564-019-0376-y
Kramer IM, Pfeiffer M, Steffens O et al (2021) The ecophysiological plasticity of Aedes aegypti and Aedes albopictus concerning overwintering in cooler ecoregions is driven by local climate and acclimation capacity. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2021.146128
Laporta GZ, Potter AM, Oliveira JFA et al (2023) Global distribution of Aedes aegypti and Aedes albopictus in a Climate change scenario of regional rivalry. InSects 14:49. https://doi.org/10.3390/insects14010049
Lee IH, Duvall LB (2022) Maternally Instigated diapause in Aedes albopictus: coordinating experience and internal state for survival in variable environments. Front Behav Neurosci 16:1–16. https://doi.org/10.3389/fnbeh.2022.778264
Lippi CA, Stewart-Ibarra AM, Franklin Bajaña Loor ME et al (2019) Geographic shifts in Aedes aegypti habitat suitability in Ecuador using larval surveillance data and ecological niche modeling: implications of climate change for public health vector control. PLoS Negl Trop Dis 13:e0007322. https://doi.org/10.1371/journal.pntd.0007322
Liria J, Navarro J (2010) Modelo de nicho ecológico en Haemagogus williston (Diptera: Culicidae), vectores del virus de la fiebre amarilla. Rev Biomed 21:149–161
Liu-Helmersson J, Brännström Å, Sewe MO et al (2019) Estimating past, present, and future trends in the global distribution and abundance of the arbovirus vector Aedes aegypti under climate change scenarios. Front Public Heal 7:1–10. https://doi.org/10.3389/fpubh.2019.00148
López-Latorre MA, Neira M (2016) Influencia del cambio climático en la biología de Aedes aegypti (Diptera: Culicidae) mosquito transmisor de arbovirosis humanas. Rev Ecuat Med Cienc Biol 37:11–21
Lounibos LP (2002) Invasive insect vectors of human disease. Annu Rev Entomol 47:233–266
Lwande OW, Obanda V, Lindström A et al (2020) Globe-trotting Aedes aegypti and Aedes albopictus: risk factors for arbovirus pandemics. Vector-Borne Zoonotic Dis 20:71–81. https://doi.org/10.1089/vbz.2019.2486
Marinho RA, Beserra EB, Bezerra-Gusmão MA et al (2016) Effects of temperature on the life cycle, expansion, and dispersion of Aedes aegypti (Diptera: Culicidae) in three cities in Paraiba, Brazil. J Vector Ecol 41:1–10. https://doi.org/10.1111/jvec.12187
Musso D, Rodriguez-Morales AJ, Levi JE et al (2018) Unexpected outbreaks of arbovirus infections: lessons learned from the Pacific and tropical America. Lancet Infect Dis 18:e355–e361. https://doi.org/10.1016/S1473-3099(18)30269-X
Mweya CN, Kimera SI, Kija JB, Mboera LEG (2013) Predicting distribution of Aedes aegypti and Culex pipiens complex, potential vectors of Rift Valley fever virus in relation to disease epidemics in East Africa. Infect Ecol Epidemiol 3:21748. https://doi.org/10.3402/iee.v3i0.21748
Naimi B (2017) Package “usdm”. Uncertainty analysis for species distribution models. R- Cran
Neteler M, Roiz D, Rocchini D et al (2011) Terra and aqua satellites track tiger mosquito invasion: modelling the potential distribution of Aedes albopictus in north-eastern Italy. Int J Health Geogr 10:49. https://doi.org/10.1186/1476-072X-10-49
O’Meara GF, Evans Leonard FJ, Gettman AD, Cuda JP (1995) Spread of Aedes albopictus and decline of Aedes aegypti (Diptera: Culicidae) in Florida. J Med Entomol 32:554–562
O’Neill BC, Kriegler E, Ebi KL et al (2017) The roads ahead: narratives for shared socioeconomic pathways describing world futures in the 21st century. Glob Environ Chang 42:169–180. https://doi.org/10.1016/j.gloenvcha.2015.01.004
Oliveira S, Rocha J, Sousa CA, Capinha C (2021) Wide and increasing suitability for Aedes albopictus in Europe is congruent across distribution models. Sci Rep 11:1–9. https://doi.org/10.1038/s41598-021-89096-5
Ortiz-Canamejoy K, Villota AC (2018) Primera evidencia de Aedes albopictus en el departamento del Putumayo, Colombia. MedUNAB 21:10–15. https://doi.org/10.29375/01237047.3416
Osorio-Olvera L, Lira-Noriega A, Soberón J et al (2020) ntbox: An r package with graphical user interface for modelling and evaluating multidimensional ecological niches. Methods Ecol Evol 11:1199–1206. https://doi.org/10.1111/2041-210X.13452
Outammassine A, Zouhair S, Loqman S (2022) Global potential distribution of three underappreciated arboviruses vectors (Aedes japonicus, Aedes vexans and Aedes vittatus) under current and future climate conditions. Transbound Emerg Dis 69:e1160–e1171. https://doi.org/10.1111/tbed.14404
Paupy C, Delatte H, Bagny L et al (2009) Aedes albopictus, an arbovirus vector: From the darkness to the light. Microbes Infect 11:1177–1185. https://doi.org/10.1016/j.micinf.2009.05.005
Pebesma E, Bivand R, Rowlingson B et al (2020) Package “sp”. Classes and methods for spatial data
Pérez-Castro R, Castellanos JE, Olano VA et al (2016) Detection of all four dengue serotypes in Aedes aegypti female mosquitoes collected in a rural area in Colombia. Mem Inst Oswaldo Cruz 111:233–240. https://doi.org/10.1590/0074-02760150363
Peterson AT, Papeş M, Soberón J (2008) Rethinking receiver operating characteristic analysis applications in ecological niche modeling. Ecol Modell 213:63–72. https://doi.org/10.1016/j.ecolmodel.2007.11.008
Peterson AT, Soberón J, Pearson RG et al (2011) Ecological niches and geographic distributions. Princeton University Press, Princeton
Phillips SB, Aneja VP, Kang D, Arya SP (2006) Maximum entropy modeling of species geographic distributions. Ecol Modell 190:231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Portilla Cabrera CV, Selvaraj JJ (2020) Geographic shifts in the bioclimatic suitability for Aedes aegypti under climate change scenarios in Colombia. Heliyon 6:e03101. https://doi.org/10.1016/j.heliyon.2019.e03101
Poveda-M IC, Rojas-P CA, Rudas-ll A, Rangel-ch JO (2004) El Chocó biogeográfico: ambiente físico
Powell JR, Gloria-Soria A, Kotsakiozi P (2018) Recent history of Aedes aegypti: vector genomics and epidemiology records. Bioscience 68:854–860. https://doi.org/10.1093/biosci/biy119
Pradhan P (2016) Strengthening MaxEnt modelling through screening of redundant explanatory bioclimatic variables with variance inflation factor analysis. Researcher 8:29–34. https://doi.org/10.7537/marsrsj080516.05
Rangel-ch JO, Arellano-P H (2017) Clima del chocó biogeográfico/costa Pacífica de Colombia
Rao B (2010) Larval habitats of Aedes albopictus (Skuse) in rural areas of Calicut, Kerala, India. J Vector Borne Dis 47:175–177
Reinhold JM, Lazzari CR, Lahondère C (2018) Effects of the environmental temperature on Aedes aegypti and Aedes albopictus mosquitoes: a review. InSects. https://doi.org/10.3390/insects9040158
Rey JR, Lounibos P (2015) Ecología de Aedes aegypti y Aedes albopictus en América y transmisión enfermedades. Biomedica 35:177–185. https://doi.org/10.7705/biomedica.v35i2.2514
Riahi K, van Vuuren DP, Kriegler E et al (2017) The shared socioeconomic pathways and their energy, land use, and greenhouse gas emissions implications: an overview. Glob Environ Chang 42:153–168. https://doi.org/10.1016/j.gloenvcha.2016.05.009
Ricas HR, Malta CR, Morales I et al (2020) First report of Aedes albopictus infected by dengue and zika virus in a rural outbreak in Brazil. PLoS ONE 15:1–11. https://doi.org/10.1371/journal.pone.0229847
Rúa-Uribe G, Suárez Acosta C, Londoño V et al (2011) Primera evidencia de Aedes albopictus (Skuse) (Diptera: Culicidae) en la ciudad de Medellín, Antioquia - Colombia. Rev Salud Pública Medellín 5:89–98
Ruiz-López F, González-Mazo A, Vélez-Mira A et al (2016) Presencia de Aedes (Stegomyia) aegypti (Linnaeus, 1762) y su infección natural con el virus del dengue en alturas no registradas para Colombia. Biomedica 36:303–308. https://doi.org/10.7705/biomedica.v36i2.3301
Sanchez-Rodriguez OS, Sanchez-Casas RM, Laguna-Aguilar M et al (2014) Natural transmission of dengue virus by Aedes albopictus at Monterrey, northeastern Mexico. Southwest Entomol 39:459–468. https://doi.org/10.3958/059.039.0307
Soberón J, Peterson AT (2005) Interpretation of models of fundamental ecological niches and species’ distributional areas. Biodivers Inform 2:1–10
Souza-Neto JA, Powell JR, Bonizzoni M (2019) Aedes aegypti vector competence studies: a review. Infect Genet Evol 67:191–209. https://doi.org/10.1016/j.meegid.2018.11.009
Suárez M (2001) Aedes albopictus (Skuse) (Diptera, Culicidae) en Buenaventura, Colombia. Inf Quinc Epidemiológico Nac 6:221–236
Tabachnick WJ (2016) Ecological effects on arbovirus-mosquito cycles of transmission. Curr Opin Virol 21:124–131. https://doi.org/10.1016/j.coviro.2016.09.008
Thomas SM, Obermayr U, Fischer D et al (2012) Low-temperature threshold for egg survival of a post-diapause and non-diapause European aedine strain, Aedes albopictus (Diptera: Culicidae). Parasit Vectors 5:1–7. https://doi.org/10.1186/1756-3305-5-100
Urdaneta-Marquez L, Failloux AB (2011) Population genetic structure of Aedes aegypti, the principal vector of dengue viruses. Infect Genet Evol 11:253–261. https://doi.org/10.1016/j.meegid.2010.11.020
Uusitalo R, Siljander M, Culverwell CL et al (2019) Predictive mapping of mosquito distribution based on environmental and anthropogenic factors in Taita Hills, Kenya. Int J Appl Earth Obs Geoinf 76:84–92. https://doi.org/10.1016/j.jag.2018.11.004
Valencia-Marín BS, Gandica ID, Aguirre-Obando OA (2020) The Mayaro virus and its potential epidemiological consequences in Colombia: an exploratory biomathematics analysis. Parasit Vectors 13:1–17. https://doi.org/10.1186/s13071-020-04354-1
Valencia-Rodríguez D, Jiménez-Segura L, Rogéliz CA, Parra JL (2021) Ecological niche modeling as an effective tool to predict the distribution of freshwater organisms: the case of the Sabaleta Brycon henni (Eigenmann, 1913). PLoS ONE 16:e0247876. https://doi.org/10.1371/journal.pone.0247876
Vega-Rúa A, Zouache K, Girod R et al (2014) High Level of vector competence of Aedes aegypti and Aedes albopictus from ten American countries as a crucial factor in the spread of chikungunya virus. J Virol 88:6294–6306. https://doi.org/10.1128/jvi.00370-14
Vélez ID, Quiñones ML, Suárez M et al (1998) Presencia de Aedes albopictus en Leticia, Amazonas, Colombia. Biomedica 18:192. https://doi.org/10.7705/biomedica.v18i3.990
Wilke ABB, Vasquez C, Carvajal A et al (2022) Mosquito surveillance in maritime entry ports in Miami-Dade county, florida to increase preparedness and allow the early detection of invasive mosquito species. PLoS ONE 17:e0267224. https://doi.org/10.1371/journal.pone.0267224
Zamora J, Castaño JC, Hoyos R (2015) Primer registro de Aedes albopictus, vector de los virus del dengue y chigunguña en La Tebaida, Quindío. Biomedica 35:114–115
Acknowledgements
The authors thank Universidad del Quindío for funding Project 1045 and covering the cost of the manuscript’s translation into English.
Funding
Open Access funding provided by Colombia Consortium. Universidad del Quindío, 1045, Oscar Alexander Aguirre Obando
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. EMJ and EEC prepared the material, collected the data, and contributed with analyses. The manuscript was written by EMJ, EEC, and OAAO. All the authors read and approved the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare having no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mejía-Jurado, E., Echeverry-Cárdenas, E. & Aguirre-Obando, O.A. Potential current and future distribution for Aedes aegypti and Aedes albopictus in Colombia: important disease vectors. Biol Invasions 26, 2119–2137 (2024). https://doi.org/10.1007/s10530-024-03298-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-024-03298-2