Abstract
The release of ornamental pets and associated pathogens outside their native range might directly or indirectly impact the recipient community. In temperate regions, e.g., central Europe, feral freshwater species of tropical and sub-tropical origins are mainly constrained to thermally polluted waters and thermal springs. However, species with high environmental plasticity and reproduction rates, such as the shrimp Neocaridina davidi, may adapt to colder water regimes over time. A widening thermal niche may eventually overcome thermal barriers, further expanding the range and enhancing transmission opportunities for host generalist parasites. This study assesses the observed (field observations) and theoretical (species distribution models) range expansion of N. davidi and associated parasites in Europe. We report three newly established N. davidi populations from thermally polluted waters in central Europe (Germany, Hungary, and Slovakia) and provide further evidence of its range expansion into colder environments. Species distribution models predict thermally suitable habitats in the Mediterranean and a foreseeable expansion into Western Europe and the Balkans by 2050. We confirm the presence of the microsporidian parasite Ecytonucleospora hepatopenaei in feral N. davidi populations across Europe and expand the list of microsporidians found in this host from two to four. Furthermore, we provide the first evidence of parasite spillover from/to the invasive crayfish Procambarus clarkii, suggesting that parasite exchange with native biota might be possible. Such possibility, coupled with an ongoing range expansion of N. davidi bolstered by human-mediated introductions and climate change, will likely exacerbate the impact on native biota.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Freshwater habitats are particularly prone to biological invasion (Casatti et al. 2006; Gherardi 2007). Invasive species can directly and indirectly impact entire ecosystems and their native communities. Generally, invaders lead to substantial declines in the abundance and diversity of resident species and alter ecosystem functioning (Ricciardi and MacIsaac 2011; Cucherousset and Olden 2011; Britton et al. 2023). Direct impacts typically involve biotic interactions such as predation, competition, and pathogen transmission, while indirect impacts often imply changes in habitat structure, water clarity, nutrients, and organic matter concentrations (Cucherousset and Olden 2011; Gallardo et al. 2016; Britton et al. 2023). Direct and indirect impacts might not follow the same trajectories; thus, the outcome of biological invasions depends on the complex interplay of various processes, some of which, including the role of invasive species as vectors for pathogens, urgently require further investigation. The interplay of all these processes makes the ecological impact of invasive species hard to predict. Nevertheless, the impact of biological invasion, bolstered by the worldwide introduction of non-native species, has likely led to the present-day global biota homogenization in freshwater ecosystems (Olden et al. 2018).
With increased global connectivity and trade, the number of introduction pathways for non-native freshwater species is growing at an unprecedented pace (Saul et al. 2017; Turbelin et al. 2022). The introduction of a broad spectrum of non-native organisms in freshwater habitats is commonly associated with aquaculture practice, recreational angling, pet trade, cultural activities, governmental stocking, mosquito biocontrol, or shipping and boating activities (Cambray 2003; Savini et al. 2010; Capinha et al. 2013; Patoka et al. 2018; Bernery et al. 2022). In Europe, the pet trade is increasingly recognized as an emerging pathway for the introduction of non-native species and their associated symbionts (Padilla and Williams 2004; Duggan 2010; Lipták and Vitázková 2015; Patoka et al. 2016).
The increased availability and popularity of aquarium animals, coupled with the inability of the owners to care for them due to high fecundity, aggressive behavior, large adult size, misinformation, and non-return policies by vendors, may result in deliberate introductions into natural ecosystems (Patoka et al. 2018; Banha et al. 2019; Gippet and Bertelsmeier 2021; Bláha et al. 2022). Consequently, introductions are tendentially biased toward the cheapest and most successful pets, which are also generally the most prolific and environmentally adaptable species (Gippet and Bertelsmeier 2021). Such traits make them more likely to adapt to local conditions and become invasive (Blackburn et al. 2011).
Although introduced non-native species might not necessarily become successful invaders in recipient ecosystems, they might negatively affect them, e.g., via pathogen transmission, which could persist in the system even after the original host disappearance (Simberloff et al. 2013). Enemy release theory suggests that invaders partially lose their associated parasites when released into new environments (Torchin et al. 2003). However, the remaining parasites can spillover to susceptible native species, conferring a competitive advantage to the invading hosts (Strauss et al. 2012; Telfer and Bown 2012). For instance, the oomycete Aphanomyces astaci (Schikora 1906), the causative agent of the crayfish plague, co-introduced in Europe alongside North American crayfish, induced mass mortality among the native crayfish species (Svoboda et al. 2017). On the other hand, invasive species might also acquire local parasites, amplifying (spillback) or diluting (dilution) their prevalence in native species (Strauss et al. 2012; Telfer and Bown 2012).
Parasite spillover is particularly relevant, considering that commercially successful freshwater pets are typically mass-produced in intensive aquaculture farms. Captive pets often have higher pathogen burdens than their wild counterpart due to potential immunosuppression from inbreeding, high densities, and lack of stringent biosecurity measures (Landaeta-Aqueveque et al. 2014; Maceda-Veiga and Cable 2019; Wood et al. 2022; Maciaszek et al. 2023). This is the case for pet fish like the guppy (Poecilia reticulata Peters, 1859), the convict cichlid (Amatitlania nigrofasciata Günther, 1867), and crustaceans like crayfish and atyid shrimps which have established feral populations across the globe including Europe (Mrugała et al. 2015; Emde et al. 2016; Maceda-Veiga and Cable 2019; Maciaszek et al. 2023).
Atyid shrimps, especially the cherry shrimp (Neocaridina davidi Bouvier, 1904), syn. N. denticulata sinensis, and N. heteropoda), are among the most successful organisms involved in the pet trade due to vibrant colors and ease of keeping (Maciaszek et al. 2018; Bláha et al. 2022). Neocardina davidi possesses high fecundity and environmental plasticity, with captive individuals successfully breeding in a wide range of conditions (14–30 °C, pH 6–8.2, gH 0–27 °C) (Namaei Kohal et al. 2018; Maciaszek et al. 2023). However, low winter temperatures in temperate regions commonly prevent their survival and range expansion once released, constraining them to thermally-polluted waterbodies (Veselý et al. 2015; Weiperth et al. 2019). Water temperatures in the native habitat of N. davidi in Southeast Asia range from 6 °C in winter to 30 °C in summer (Klotz et al. 2013). Those temperatures are higher than winter temperatures in central Europe; however, a warming climate might facilitate the establishment of this thermophilic species in the foreseeable future (Klotz et al. 2013; Jabłońska et al. 2018).
Permanent, self-sustaining feral populations of N. davidi are already known from thermal waters in Canada, Germany, Hungary, and Poland and even unheated waters in Israel, Japan, the French overseas territory of la Reunion, and the USA (Klotz et al. 2013; PM 2017; Jabłońska et al. 2018; deBruyn 2019; Levitt-Barmats et al. 2019; Weiperth et al. 2019). Moreover, further observations of likely N. davidi have been reported worldwide. The most recent was from southern France via the iNaturalist app (tomjamonneau 2023). However, these are single observations, often lack genetic data, and until proven otherwise, do not represent self-sustaining populations. Nevertheless, they highlight the ongoing releases of these non-native shrimps in natural environments.
The effects of N. davidi introduction are known to be multi-dimensional. Reported ecological impacts of N. davidi are, for instance, the replacement of native shrimps with similar ecological niches (Onuki and Fuke 2022), alteration of meiofaunal assemblages (Weber and Traunspurger 2016), and changes in leaf-litter breakdown in invaded areas (Schoolmann and Arndt 2017). The high feeding rate of the omnivorous N. davidi, estimated to be over half of its body weight per day, implies that other less efficient native crustaceans relying on the same resources may be outcompeted (Schoolmann and Arndt 2017). However, neither habitat suitability nor the ecological impact of N. davidi on European ecosystems has been thoroughly investigated. Likewise, the risk posed by co-introducing symbionts associated with N. davidi remains unclear.
Neocaridina davidi hosts a wide range of commensals and parasites (Ohtaka et al. 2012; Liao et al. 2018; Bauer et al. 2021; Maciaszek et al. 2023), some of which have been co-introduced with N. davidi outside their native range (Niwa and Ohtaka 2006; Patoka et al. 2016; Maciaszek et al. 2021b; Kakui and Komai 2022). Among them, Ecytonucleospora (= Enterocytozoon) hepatopenaei (EHP) (Wang et al. 2023) has been detected using molecular tools in a German population of feral N. davidi (Schneider et al. 2022). EHP is a microsporidian parasite known to infect the hepatopancreas of shrimps, resulting in stunted growth and impaired immunity. Its transmission occurs directly via the oral-fecal route, cannibalism, or exposure to contaminated water (Tangprasittipap et al. 2013; Subash et al. 2022). EHP is known to infect cultured penaeid shrimps (Chaijarasphong et al. 2021) but can also infect other freshwater, brackish, and marine invertebrates (Karthikeyan and Sudhakaran 2020; Krishnan et al. 2021; Munkongwongsiri et al. 2022; Wan Sajiri et al. 2023). Most recently, EHP has been reported in dragonflies with persisting infections in their terrestrial imago stage and, more worryingly, from the red swamp crayfish (Procambarus clarkii Girard, 1852), a prominent invader in freshwater ecosystems worldwide (Dewangan et al. 2023; Ling et al. 2024). Therefore, confirming the presence of EHP and other microsporidian infections in feral N. davidi populations is imperative. Likewise, assessing N. davidi-related parasite infections in co-occurring crustaceans is paramount, as they might spread to native and invasive biota.
This study aims to assess the range expansion of N. davidi and its associated parasites in Europe, confirm the presence of EHP in the feral populations, identify possible parasite spillover/spillback from and to co-occurring crustaceans (amphipods, crayfish, and isopods), and experimentally test the possibility of pathogen transmission between alien and native biota. Furthermore, in light of ongoing climatic changes, we infer current and near-future habitat suitability for N. davidi in Europe and the Mediterranean using species distribution models.
Materials and methods
Sampling sites
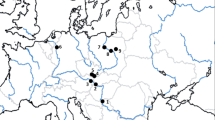
The sampling of N. davidi and co-occuring crustaceans (amphipods, crayfish, and isopods) took place between September 2021 and May 2023 in multiple locations across Germany, Hungary, Slovakia, and Poland (Table 1 and Fig. 1). Two catchments have been investigated in Germany: the Erft in North Rhine-Westphalia and the Blies in Saarland. The Erft is a tributary of the River Rhine with a long history of thermal pollution (Schoolmann and Arndt 2018). Neocaridina davidi was first reported from the Gillbach in 2013, a thermally polluted tributary of the River Erft, and later from the lower and upper Erft and the Rhine (Klotz et al. 2013; Schoolmann and Arndt 2018; Bierbach et al. 2022; Schneider et al. 2022). Two citizen scientists also reported sightings of N. davidi in the upper part of the Erft via the iNaturalist app (aymac 2021; drmichaelbraun 2021). The heated waters of the Gillbach, which are fed entirely by the cooling water of the nearby lignite-fired powerplant of Niederaußem, hosted a community of organisms commonly found in the pet trade, including, among others, P. reticulata and A. nigrofasciata (Klotz et al. 2013; Lukas et al. 2017b).
Map of sampling areas where Neocaridina davidi individuals were collected. Black dots indicate sites visited in the present study. White dots indicate additional areas in which N. davidi has been observed in previous studies or reported by citizen scientists via the iNaturalist app. The maps were generated using ArcGIS with data extracted from OpenStreetMap
The second German catchment, the Blies, is a tributary of the River Saar, subject to post-mining water discharges (Franzaring 2022). Warm and saline waters from a former coal mine now used as a geothermal plant are released in one of its tributaries, the Sinnerbach. The heated water is discharged into a so-called ‘water garden’ for cooling, which flows into a nearby creek, the Klinkenbach, before converging into the Sinnerbach. The water garden is an artificial recreational area characterized by cloudy, saline waters (pH 7.5–7.9, DO 3.6–9 mg/l, 1850–1900 µs) and a temperature constantly above 26 °C (Table 1). The garden is home to a thriving community of freshwater pets, which includes cichlids, cyprinids, poecilids, and crayfish such as P. clarkii (Lukas et al. 2017a). Neocaridina davidi was first filmed on-site in 2020 by a YouTuber (Krabbelkeller 2020). From the outflow of the water garden into the Klinkenbach, the water temperature still remains above 20 °C until the upper part to the Sinnerbach.
Two localities in Hungary have been investigated: the thermal water of Miskolctapolca and those of the Városliget Park in Budapest. In Miskolctapolca, the thermal waters of the Békás pond and the outflowing Hejő creek, a tributary of the River Tisza, host a wide variety of ornamental fish, shrimps, crayfish, amphibians, and turtles which have been extensively described in recent publications (Weiperth et al. 2019; Maciaszek et al. 2021a; Bláha et al. 2022). Here, N. davidi was first recorded in the autumn of 2017 and has maintained a stable population since then. Occasionally, shrimps have been observed to venture into the colder downstream water of Hejő creek (< 6 °C) between late autumn and early spring (Weiperth et al. 2019). The second location, the thermal water of Városliget Park, comprises large and small thermal spring ponds. The warmest of the large ponds hosts pet-traded crayfish such as P. clarkii, marbled crayfish (Procambarus virginalis Lyko, 2017), and five Cherax species (Weiperth et al. 2020). All ponds are interconnected and drain via a subsurface channel into the main arm of the Danube River. Among these is also Szent István spring pond, a small artificial pond at the base of a fountain fed by thermal water (28.9 °C, pH 8.37, DO 3.1 mg/l, 796 µs, Table 1).
In Slovakia, N. davidi was first observed in 2021 by Martin Dobrota, a citizen scientist in the upper part of the Čepčínsky creek, a tributary of the River Turiec (Váh River Basin). The creek originates from a thermal spring and has a length of 7 km. Its upper section hosts a population of P. clarkii, which likely escaped from one of Europe’s largest aquaculture facilities focused on breeding exotic fish species, mollusks, and crustaceans for the pet trade (Lipták et al. 2023).
Neocaridina davidi has been present in Poland at least since 2003 in the Ciepły canal, a thermally polluted artificial channel in the lower section of the River Oder, near Gryfino (Jabłońska et al. 2018). The 3.5 km long channel receives heated water from the Dolna Odra Power Station cooling system. In its proximity, the annual mean temperature is 20.9 °C (Maciaszek et al. 2021b). Besides N. davidi, several alien species, including pet trade-related ones like the Asian clam (Corbicula fluminea Müller, 1774), the pirapitinga (Piaractus brachypomus Cuvier, 1818), and P. clarkii are well-established on-site (Jabłońska et al. 2018; Maciaszek et al. 2021b; Ondračková et al. 2023). However, in the summer of 2022, fish and mollusks living in the River Oder, including the artificial canal where N. davidi was previously collected, experienced unprecedented mass mortality caused by the toxic golden alga Prymnesium parvum (Carter 1937), an alga associated with saline waters (Marchowski and Ławicki 2023).
Sampling collection and processing
Neocaridina davidi and co-occuring amphipods, decapods, and isopods were collected using hand nets and immediately preserved in 96% ethanol for morphological and molecular analyses of hosts and parasites. Moreover, the carapax, including a section of the hepatopancreas, of 50 additional freshly dissected individuals collected from the Kleine Erft (Germany) on November 2022 were fixed in Davidson’s freshwater solution for 24 h and later transferred to 70% ethanol for histological examination. The rest of the body was preserved in 96% ethanol and processed for morphological and molecular analyses of hosts and parasites. The Kleine Erft population was chosen after the first batch of individuals collected during this study (February 2022) were confirmed to be infected with EHP via molecular screening. A total of 50 pet-traded N. davidi were later bought from different retailers and private keepers. These were euthanized by ice submersion until the absence of eye stalk responses. After death, they were cut in half lengthwise, one side preserved in 96% ethanol for molecular identification of host and parasites and the other homogenized for a subsequent infection experiment with isopods and amphipods.
All hosts (amphipods, crayfish, isopods, and N. davidi) were morphologically and molecularly identified, measured, and dissected in the laboratory. The hosts were screened for internal parasites, and the guts were removed to avoid microsporidian contamination in downstream molecular analyses. Additionally, small portions of preserved muscles from five P.clarkii individuals collected during another study (Lipták et al. 2023) were also molecularly screened for microsporidians. Epibionts were not analyzed as their conservation in ethanol-preserved specimens is generally poor.
Molecular analyses
DNA was isolated from hosts using either a modified salt precipitation protocol described by Grabner et al. (2015) or using a 10% solution of Chelex 100 resin (Bio-Rad Laboratories, Hercules, CA, USA) following the protocol described in Hupało et al. (2023). Host molecular identification was performed using the universal eukaryotic primers LCO1490 (5′-GGTCAACAAATCATAAAGATATTGG-3′) and HCO2198 (5′-TAAACTTCAGGGTGACCAAAAAATCA-3′) (Folmer et al. 1994) or LCO1490-JJ (5′-CHACWAAYCATAAAGATATYGG-3′) and HCO2198-JJ (5′-AWACTTCVGGRTGVCCAAARAATCA-3′) (Astrin and Stüben 2008). When possible, ten N. davidi individuals for each sampling point and each retailer were randomly chosen for molecular identification. Other crustaceans were identified following the same procedure.
Microsporidians presence in the examined hosts was initially screened with the universal microsporidian primers V1F (5′-CACCAGGTTGATTCTGCCTGAC-3′) (Zhu et al. 1993) and Micuni3R (5′-ATTACCGCGGMTGCTGGCAC-3′) (Weigand et al. 2016) targeting the small subunit ribosomal RNA (SSU rRNA) gene. Subsequently, infected individuals (i.e., bands clearly visible in the gel) were screened for EHP using a single PCR with the species-specific primers ENF779 (5′-CAGCAGGCGCGAAAATTGTCCA-3′) and ENR779 (5′-AAGAGATATTGTATTGCGCTTGCTG-3′) targeting the SSU rRNA gene (Tangprasittipap et al. 2013) and a nested PCR with the SWP1F (5′-TTGCAGAGTGTTGTTAAGGGTTT-3′), SWP1R (5′-CACGATGTGTCTTTGCAATTTTC-3′), SWP2F (5′-TTGGCGGCACAATTCTCAAACA-3′), and SWP2R (5′-GCTGTTTGTCTCCAACTGTATTTGA-3′) primers targeting the spore wall protein of EHP (Jaroenlak et al. 2016). All PCR reactions excluding those used for the LCO1490-JJ and HCO2198-JJ primer pair consisted of 20 μL composed of 10 μL of 2 × AccuStart II PCR ToughMix (Quantabio), 1 μL of each primer (0.5 μM), 0.35 μL of 50 × GelTrack Loading Dye (Quantabio), 6.65 μL MilliQ water and 1 μL of DNA template. For the LCO1490-JJ and HCO2198-JJ primer pair, the PCR reaction consisted of 20 μL assay with 10 μL of Dream-TaqTM Hot Start Green PCR Master Mix (Thermo Fisher Scientific, Waltham, MA, USA), 1.6 μL (5 uM) of each primer, 4.8 μL of nuclease-free water and 2 uL of DNA template per reaction. PCR settings for DNA fragment amplification varied between different primer sets and are described in detail in the Supplementary file (S1). PCR products were sent either unpurified to Microsynth Seqlab (Germany) or purified (LCO1490-JJ and HCO2198-JJ) to Eurofins Genomics (Cologne, Germany) for Sanger sequencing. Enzymatical purification was obtained with 10 U of Exonuclease I (ExoI) and 1 U of thermosensitive alkaline phosphatase (FastAP) (both Thermo Fisher Scientific) for 5 μL of PCR product with an incubation step at 37 °C for 25 min followed by an inactivation step at 85 °C for 15 min.
Raw sequences were quality-checked and edited using Geneious v2023.0.1 (Biomatters, Ltd., New Zealand). Only sequences with a minimum length of 200 bp were retained for the analyses. Obtained sequences were compared against GenBank records using BLASTN (blast.ncbi.nlm.nih.gov). Host and parasite sequences were separately aligned using the MAFFT v7.490 algorithm with standard settings (Katoh et al. 2019). Maximum likelihood phylogenetic trees with bootstrap support values (1000 replicates) were produced in IQ-Tree 2.2.0 (Minh et al. 2020) using the TIM3 + F + G4 substitution model for microsporidians and the TPM2u + F + I + G4 substitution model for N. davidi based on Bayesian information criterion. A haplotype network was inferred using the Minimum Spanning Network method in PopArt v1.7 (Leigh and Bryant 2015), whereas the visualization of N. davidi haplotypes for each area investigated was obtained with the R package mapmixture (Jenkins 2023). All the sequences generated in this study were submitted to NCBI GenBank (accession numbers OR610860-71 and OR613110-12 for hosts, and OR616363-71 for microsporidians).
Histological analyses
Whole animal preparations were processed for histological analyses via initial fixation in Davidson’s freshwater fixative (~ 48 h) before being transferred into 70% ethanol. Only infected individuals (confirmed via molecular analyses) were used. These were infiltrated with paraffin wax in an automated tissue processor (ethanol-xylene substitute wax). Wax-infiltrated tissues were then embedded into wax blocks and sectioned at 4 µm to attain a single section per specimen through the center of the animal. Each section was then mounted upon a glass slide and stained with hematoxylin and eosin, following a standard de-wax and rehydration protocol. The slides were screened for pathogens using a Leica DM500 light microscope and photographed with the integrated camera system.
Infection experiment
Potential horizontal transmission of EHP from N. davidi to the native macroinvertebrates Asellus aquaticus (Linnaeus, 1758) and Gammarus pulex (Linnaeus, 1758) (co-habitants at the invasion site) was experimentally assessed. Three groups of ten A. aquaticus and ten G. pulex were transferred from the respective rearing tanks into the experimental tanks (acclimatization: 24 h). These were lab-reared, microsporidian-free individuals (F2 of individuals collected in the Boye catchment, NRW, Germany). After that, homogenized tissues of five EHP-infected N. davidi (EHP positive using nested PCR and preserved at 4 °C < 24 h) were provided as feed (inoculant) over three days to the treatment groups (two groups of ten individuals per species). The use of EHP-infected homogenized tissue provides simultaneous exposure to spore-infected water and direct oral infections via the ingestion of infected tissue remains. EHP-infected homogenized tissue was preferred over exposure to EHP-infected shrimp feces to maximize the possibility of transmission by exploiting the scavenger habits of A. aquaticus and G. pulex. In parallel, homogenized tissues of five EHP-free N. davidi individuals were fed to the control groups (one group of ten individuals per species). Each homogenized shrimp was diluted in 4 ml of water and evenly distributed among tanks on days one and three to minimize ammonia production.
Both G. pulex and A. aquaticus were observed to feed on the material provided. After 20 days, all G. pulex and A. aquaticus were collected and molecularly screened for EHP following the procedure described in “Molecular analyses” Section.
All G. pulex and A. aquaticus were held in 3-L containers illuminated daily for eight hours with 6500 K LED bars. The container setup consisted of a thin layer of gravel barely covering the bottom, Java moss (Taxiphyllum barbieri Iwatsuki, 1982), three alder leaves, and water from the respective rearing tank (17.1 °C, pH 7.92, 426 µs). No filtering, aeration, or heating was used. The experimental tanks were left for 4 weeks before the experiment to reduce potential ammonia spikes when feeding homogenized N. davidi and avoid water changes that could reduce the number of infective spores in the tank.
Influence of temperature on N.davidi and EHP infectivity
Temperature influences survival rate and sex ratios in N. davidi, resulting in a larger proportion of females born at higher temperatures (Serezli et al. 2017). This may boost propagule spreading from thermally altered to colder areas. Thus temperature-dependent sex ratio of sexed individuals was assessed using a pairwise Chi-square across different temperature categories (< 15, 15–19.99, 20–24.99, and > 25 °C). The infectivity of EHP may be affected by temperature, given its warm climate origins. Therefore, the influence of temperature on EHP prevalence in all feral N. davidi was investigated using Generalized Linear Model (GLM) logistic regression with infected/uninfected as the dependent variable and carapax size, sex (female, male, and unsexed), and temperature categories as independent variables. The results were reported as odd ratios. The model did not include water conductivity, a proxy for salinity, as this has not been consistently measured (Table 1), nor was a distinction made between sampling sites and sampling season. A direct comparison of prevalence across sampling sites and seasons was impossible due to a low number of samples and mismatched sampling periods. Therefore, we employed an exact binomial probability estimation with a 95% confidence interval to estimate the infection rate of background populations at each site using the exactici package (Fay 2023). Statistical and descriptive analyses were performed with the open-source software R (version 4.3.2, R Core Team 2023) via the RStudio GUI (version 2023.06.0, RStudio Inc.).
Species occurrence presence-only data
Species distribution models (SDMs) were employed to predict habitat suitability for N. davidi across central Europe and the Mediterranean region using the Maxent algorithm on presence-only data. The taxonomy of N. davidi remains somewhat unclear as N. davidi, and N. denticulata (De Haan 1844) are often used interchangeably in the literature, and natural hybridization between these species and N. palmata (Shen 1948) may occur (Onuki and Fuke 2022). Therefore, we constrained our data to N. davidi clade A and B, leaving out clade C as the latter, according to the most recent phylogenetic and morphological analyses of Onuki and Fuke (2022) are likely hybrids between N. davidi and N. palmata.
All available DNA sequences (COI gene) of Neocaridina with a minimum length of 500 bp were retrieved from NCBI GenBank on February 2024, totaling 307 entries (Supplementary file S2). Sequences not belonging to wild collected individuals, those from outside eastern Asia, those from individuals belonging to species or clades other than N. davidi clade A and B, and those where retrieving the sampling location was impossible even by screening the related literature were excluded. Geographical coordinates for samples with only the location name were retrieved from Google Maps. We eliminated duplicate entries and kept only those from sampling locations more than 10 km apart. The final dataset consisted of 51 entries, which were also used to construct the phylogenetic tree mentioned in “Molecular analyses” Section (Fig. 2). Native and invaded areas of N. davidi in eastern Asia span from southern China to northern Japan, encompassing a wide range of environmental variables. Different populations might have distinct thermal tolerances. Therefore, we split them into southern (Clade B1) and northern (Clade A and B2) populations (Fig. 2) to account for possible climatic adaptations and generated two separate SDMs. Note that the clades were heavily biased towards either Taiwan or northern China and northern Japan (Fig. 2).
Species distribution models
The software Maxent version 3.4.4 (Phillips et al. 2017) and R were employed for modeling habitat suitability of southern (n = 26) and northern (n = 25) N. davidi populations. Given the patchy distribution of available data and the sampling locations’ relative proximity, bias files for both models were generated using two-dimensional kernel density estimation via the MASS package (Ripley et al. 2023). Leave-one-out cross-validation was used to test the models, and the jackknife test was used to identify the most important variables. Maximum entropy modeling was done using 10.000 background sites, 500 iterations, a regularization multiplier of 1, auto features, and a logistic output.
Bioclimatic data with a spatial resolution of 2.5 arc minutes for 1979–2013 (Karger et al. 2017) provided by Paleoclim (http://www.paleoclim.org) and downscaled CMIP6 SSP245 (intermediate greenhouse gas emissions scenario) projections for 2050 (2041–2060 period) provided by WorldClim (https://worldclim.org) were used to model habitat suitably in the present day and near future. For near-future projections, we used the average values of four global climate model ensembles (ACCESS-CM2, HadGEM3-GC31-LL, MIROC6, and MPI-ESM1-2-HR) deemed suitable for continental Europe and the Mediterranean area (Raju and Kumar 2020; Seker and Gumus 2022).
With a lack of detailed knowledge of the focal species, simpler models are more accurate than those calibrated using a broader set of bioclimatic variables (Low et al. 2021). Therefore, as predictors, we prioritized the use of the minimum and maximum temperature variables. Neocaridina davidi is primarily constrained by its thermal tolerance, and artificial aquatic ecosystems exist even in relatively arid areas, as exemplified by invaded regions of Israel (Levitt-Barmats et al. 2019). To improve realism and transferability, we excluded precipitation variables that contributed more than the temperature and removed those that were highly correlated (Pearson’s r > 0.8) using the package ENMTools (Warren et al. 2021). The resulting bioclimatic variables used for the models were BIO5—maximum temperature of the warmest month, BIO6—minimum temperature of the coldest month, and BIO18—precipitation of the warmest quarter.
Present and near future habitat suitability were projected on Central Europe and the Mediterranean. The AUC value for the first model was 0.88 (good), while that of the second was 0.61 (poor). A value above 0.5 indicates that the Maxent model performs better than random. We kept the second model despite the poor score, as it was consistent with the known distribution of N. davidi and, therefore, deemed biologically relevant. The widely used AUC values are often misleading and unrealistic when it comes to the biological significance of SDMs (Lobo et al. 2008). N. davidi clade B1, exported from Taiwan, is currently present in the European pet trade and thermally altered waters. Still, they have been observed in the northern regions of China and Japan. Therefore, we created an average ensemble of habitat suitability projections derived from southern and northern regions for a more realistic representation of present and near-future predictions for this clade.
Results
A total of 658 shrimps and 353 co-occurring crustaceans (303 amphipods, 5 crayfish, and 45 isopods) were collected. All shrimps were N. davidi clade B1 (Fig. 2). Morphological identification of feral N. davidi was corroborated by molecular identification with five haplotypes detected, here named ‘Ndh1’, ‘Ndh2’, ‘Ndh3’, ‘Ndh4’, and ‘Ndh5’ (Figs. 2 and 3). Ndh1 showed 100% similarity to feral N. davidi previously collected in Germany (OM468123). ‘Ndh2’ was 100% similar to feral and pet-traded individuals previously found in Poland (MG816764 and MG816765), pet-traded individuals in Canada (MG319788), and wild individuals from Taiwan (MG816764 and MG734262), while ‘Ndh3’ and ‘Ndh5’ had 100% similarity to wild Taiwanese samples (MG734280 and MG734258). ‘Ndh4’ was 100% similar to feral individuals previously collected in Hungary (OM214466).
Neocaridina davidi was present at all sites, including the Gillbach, where other pet-traded species disappeared, and the Ciepły canal, which was affected by the mass mortality event in the Oder during the summer of 2022. In the water garden (Blies catchment), N. davidi co-occurred with pet-traded species, including previously unreported species such as Ancistrus sp., Hemichromis spp., molly (P. sphenops Valenciennes, 1846), platy (Xiphophorus maculatus Günther, 1866), and P. virginalis. In the Szent István spring pond N. davidi co-occurred with pet-traded fish (e.g., Poecilidae) and the invasive crayfish P. virginalis. Neocaridina davidi inhabited environments with a wide range of water parameters (9–29.3 °C, pH 6.9–8.6, 3.1–9.8 mg/l DO, and 551–2274 µs/cm, Table 1) and 30.2% of the collected individuals originated from water below 15 °C.
Among the N. davidi collected, 316 were females (119 were ovigerous), 237 were males, and 105 were individuals of undetermined gender. With an average carapax length of 5.09 ± 1.00 SD (range 3.06–7.99) mm, females were larger than males 4.38 ± 0.75 SD (3.01–6.38) mm. The proportion of females and males was relatively similar across temperature categories (Chi-square test, χ2(3, 553) = 6.88, p = 0.076). There was no clear evidence of a temperature-dependent sex ratio. Populations of N. davidi living at temperatures between 15 and 20 °C showed the highest proportion of ovigerous females (61.54%) and those living above 25 °C the lowest (14.63%).
Among the investigated feral N. davidi, 5.62% were infected with microsporidians, mostly EHP (5.02%, Table 2). However, neither carapax length, sex (female, male, unsexed), nor temperature predicted EHP infections (all OR 95% CI crossed 1, Table 3). Half of the investigated areas hosted feral populations of N. davidi infected with EHP. The highest prevalence was found in the Erft catchment (9.30%). However, binomial probability estimations calculated for each sampling site indicated a potential prevalence of up to 84.14% among the background populations (Table 4). EHP was the only microsporidian found in pet-traded N. davidi, reaching 14% prevalence. EHP was not found in other macroinvertebrates collected in the wild, and no EHP-infected individual was found in the infection experiment of G. pulex and A. aquaticus.
The detection of EHP relied on molecular analyses. SSU rRNA sequences showed 99.9–100% similarity with those obtained from the whiteleg shrimp (Penaeus vannamei Boone, 1931) from India, China, South Korea, and Vietnam, and polychaete worms Marphysa spp. collected in India. All infected N. davidi shared the same EHP haplotype (100% similarity). Likewise, EHP spore wall protein sequences belonged to a single haplotype, which showed 98.64% similarity to sequences obtained from P. vannamei collected in India, Thailand, and Malaysia. Histology of EHP-positive N. davidi revealed infected hepatopancreatic tubules, presenting eosinophilic cytoplasmic inclusions of early spore development as well as later mature spore stages (Fig. 4).
Histopathological images of the infected hepatopancreas of Neocaridina davidi, revealing cytoplasmic inclusions in the hepatopancreatic epithelial alpha and beta cells. A The relative morphology of the cytoplasmic inclusions reflects different development stages (reflecting the putative sporophorous vesicle, arrow 1; and infective spore stages, arrow 2). B Cytoplasmic inclusions of microsporidian spores (black arrows) within the hepatopancreatic epithelia. The white arrows indicate example host nuclei. HL hepatopancreatic tubule lumen, HS haemal sinus
Besides EHP, three other microsporidian isolates, two of which were new to science, were detected in N. davidi (Table 2, Fig. 5). Those in Čepčínsky creek were infected with an undescribed microsporidian isolate, here named Microsporidium sp. SCC01. The closest described species was Enterocytospora artemiae (Rode et al. 2013) (JX915760; 83.23% identity, 99% coverage, e-value = 6e−80). The same microsporidian was also detected in two P. clarkii collected on site. One N. davidi individual collected from the Békás pond was infected with an undescribed isolate, here named Microsporidium sp. HMI01. The closest described species was Jirovecia branchilis (Liu et al. 2023) (AF484694; 83.97% identity, 81% coverage, e-value = 1e−69). One individual from Finkelbach was infected with Microsporidium sp. EFB01 (OM467902; 99.50% identity, 69% coverage, e-value = 1e−94), a microsporidian isolate previously observed in N. davidi by Schneider et al. (2022).
Maximum likelihood phylogenetic tree of microsporidians identified in this study. Grey dots represent bootstrap support values above 90%. Sequences obtained in this study are indicated in bold, and the outgroup sequence in grey. The names and circumscriptions of microsporidian clades sensu Bojko et al. (2022) are indicated in red
Six other microsporidians were detected exclusively in amphipods or isopods co-occuring with N. davidi (Table 2, Fig. 5). Among these were Nosema sp. clade B sensu Bacela-Spychalska et al. (2023) (OR165989; 99.60% identity, 100% coverage, e-value = 9e−174), Dictyocoela roeselum (Haine et al. 2004) (MG773219; 100% identity, 100% coverage, e-value = 0.0), and Microsporidum sp.V-B (MK719363; 99.06% identity, 100% coverage, e-value = 5e−157) found in Gammarus roeselii (Gervais 1835), Microsporidium sp. 505 (FN434085; 99.14% identity, 100% coverage, e-value = 5e−171) found in G. pulex and two undescribed microsporidians. The first of which was found in G. roeselii and here named Microsporidium sp. HMI02. The closest described species was Helmichia lacustris (Voronin 1998) (GU130406; 90.46% identity, 84% coverage, e-value = 6e−86). The second, here named Microsporidium EFB02, was found in A. aquaticus. The closest described species was Euplotespora binucleata (Fokin et al. 2008) (DQ675604; 84.23% identity, 96% coverage, e-value = 2e−74). None of the other hosts were infected (Table 2).
In central Europe, N. davidi and their parasites occur in thermal and adjacent colder waters. Waters in Central Europe, if not thermally altered, have low predicted habitat suitability for southern and northern N. davidi populations (Fig. 6). More favorable conditions are present in the Mediterranean. Its coastline and islands offer moderately to highly suitable habitats to northern N. davidi populations, while the southeastern Mediterranean seems more favorable for the southern populations. Broader areas than those currently occupied by the species are deemed suitable. These are expected to expand under a climate change scenario with intermediate greenhouse gas emissions.
Maxent derived present-day and near-future habitat suitability projections for southern (Taiwan-biased) and northern (northern China-Japan-biased) populations of Neocaridina davidi in Central Europe and the Mediterranean. Near-future predictions (2050) based on CMIP6 SSP245 average values of four global climate model ensembles (ACCESS-CM2, HadGEM3-GC31-LL, MIROC6, and MPI-ESM1-2-HR) for the period 2041–2060. Unheated waters currently invaded by southern and northern population of Neocaridina davidi are indicated with a red dot on the respective map
Predictions for the northern populations of N.davidi indicate a notable increase in suitable habitats by 2050 in Western Europe and the Balkans but a worsening for the southern Mediterranean. Areas with suitable habitats might shrink for the southern N.davidi populations if no climatic adaptations or thermal plasticity is accounted for. The average projection ensemble of both populations for 2050 indicates limited habitat suitability in continental Europe, mainly across the Iberian peninsula. The most influential predictor of habitat suitability in all projections was BIO6—minimum temperature of the coldest month. This predictor contributed between 75.7 and 99.2%, thus retaining the most useful information by itself.
Discussion
The release of ornamental pets and associated parasites into novel environments might pose severe risks to native biota (Sures 2011; Svoboda et al. 2017; Patoka et al. 2018). Deepening the knowledge of invading hosts and parasites is paramount, especially for commercially successful ornamental pets. Commercially successful ornamental pets, like N. davidi, are widely available, easy to breed, and often tolerant to a wide range of environmental variables. This makes them more prone to releases and increases their chances of establishing in new locations (Lipták and Vitázková 2015; Bláha et al. 2022). A possible invasion risk is not covered by the EU regulation No. 1143/2014 on the prevention and management of the introduction and spread of invasive alien species, as shrimps are not currently listed as species of concern. We present the discovery of three newly established N. davidi populations across Europe and mounting evidence of an ongoing range expansion into colder water. Furthermore, we confirm the presence of EHP infections in feral N. davidi and other microsporidian parasites, including one shared with the highly invasive crayfish P. clarkii, highlighting a possible parasite spillover.
The presence of feral N. davidi in central Europe dates back to 2003, with the report of a population in Poland (Jabłońska et al. 2018). Since then, further populations have been discovered in Germany and Hungary (Klotz et al. 2013; Weiperth et al. 2019). The current study increased this list with three more thermally polluted sites in Germany, Hungary, and Slovakia. The latter is the first report of this species in the country. The geographic distribution of feral N. davidi, as evidenced by their haplotypes, primarily reflects what is available in the pet trade of the respective countries. The haplotype ‘Ndh2’ was dominant in Poland, Slovakia, and the Hungarian population of Városliget Park, Budapest. The dominance of this haplotype in the eastern part of central Europe is unsurprising, as the pet market in these countries often shares similar suppliers. Accordingly, this haplotype has previously been reported from the Polish pet trade (Jabłońska et al. 2018), and the Slovakian N. davidi population may have escaped from the nearby aquaculture farm that supplies the Slovak, Hungarian, and Polish markets. On the other hand, ‘Ndh1’ and ‘Ndh3’ were only found in Germany, where these haplotypes are traded.
Escapes from aquaculture farms are a known issue, particularly in Southwestern Europe, where biosecurity measures are often lacking (Maceda-Veiga et al. 2013). Colder water in temperate regions might prevent warm-adapted species from establishing. Nevertheless, numerous pets, including the topmouth gudgeon (Pseudorasbora parva Temminck and Schlegel, 1846), the oriental weather loach (Misgurnus anguillicaudatus Cantor, 1842), and apple snails (Pomacea spp.), have become invasive in Europe (Beyer 2004; Franch et al. 2008; Gilioli et al. 2017). Other introduced pets have failed to establish outside thermal waters due to a lack of physiological plasticity, but this might change in the future, thanks to a warming climate (Ribeiro et al. 2008; Weiperth et al. 2019).
High physiological plasticity may have favored the establishment of N. davidi even outside such boundaries. Accordingly, N. davidi seems to be relatively unaffected by perturbations in the thermal regime of cooling water systems. As seen for the Gillbach, where most ornamental species, except for N. davidi, perished following a temperature decrease due to the partial decommissioning of the powerplant in 2020 and 2021 (RWE 2021). Likewise, most ornamental species hosted in the water garden were also present in the lower warm part of the Klinkenbach until its confluence with the Sinnerbach, where cold and warm water mixed, with only N. davidi occurring in the colder downstream waters. Neocaridina davidi had higher thermal tolerance than other pets, with roughly one-third of the individuals collected in waters below 15 °C. Furthermore, N. davidi were collected from waters containing as little as 3.1 mg/l dissolved oxygen, indicating they can thrive even in oxygen-poor environments. Accordingly, the N. davidi population in the Ciepły Canal survived oxygen deprivation associated with the algal explosion event that infested the saline waters of the Oder River in the summer of 2022 (Marchowski and Ławicki 2023).
Temperature did not strongly influence N. davidi sex ratios. This contradicts previous experimental findings where the sex ratio appeared to be strongly influenced by temperature (Serezli et al. 2017). However, such experimental studies do not take predation into account. Females are generally larger and often more colorful than males. Accordingly, the coloration of females collected across different sampling sites was mixed, ranging from brown-grey to red, blue, green, and black, while that of males was mainly transparent-gray-brown. Consequently, increased visibility of females likely results in higher predation pressure and a more balanced sex ratio, as observed in the present study. Such a dynamic might lessen propagule pressure from thermal to colder waters, hindering colonization success. This might hold particularly true for water nearing 20 °C, in which 80% of the population is expected to be composed of females (Serezli et al. 2017). Ideal breeding conditions in feral N. davidi seem to be found between 15 and 20 °C, as shown by the large proportion of ovigerous females. However, ovigerous females were also observed at temperatures below 10 °C, suggesting that reproduction in these waters is not limited to the summer season only. This finding, coupled with the relatively high proportion of individuals collected in water below 15 °C, indicates that species’ range expansion to colder waters might be possible in the upcoming years.
A range expansion of N. davidi into colder waters is unsurprising; previous studies have postulated such a prediction (Klotz et al. 2013; Schneider et al. 2022). Although considered thermophilic, N. davidi is not strictly a tropical species. In truth, its native habitats includes waters ranging from 6 °C in winter to 30 °C in the summer (Klotz et al. 2013). Accordingly, even N. davidi clade B1, originating from the warm waters of Taiwan, has been observed in the colder waters of northern regions of China and Japan (Zhou et al. 2021; Onuki and Fuke 2022). However, waters in Central Europe, unlike those in the Mediterranean, have low predicted habitat suitability for southern and northern N. davidi populations, unless thermally altered. Winter temperatures are 2–4 °C lower than those registered in Asian water bodies but similar to those reported from thermally-polluted rivers such as the River Rhine, Europe’s seventh-longest and busiest waterway (Wetzel 2001; van Vliet et al. 2011), known for its enormous proportion of invasive species (Sures et al. 2019). Nevertheless, a rise in water temperatures, an increase in the frequency and duration of heat waves, and a decrease in recurring cold spells during winter facilitate the acclimatization of thermophilic species (van Vliet et al. 2013; Jabłońska et al. 2018; Woolway et al. 2021).
In a climate change scenario with intermediate greenhouse gas emissions, broader areas than those currently occupied are expected to become suitable by 2050 in Western Europe and the Balkans, largely sparing those of Central Europe. This is based on the assumption that southern N. davidi populations will show similar thermal tolerances to those observed in their northern counterpart. The critical thermal minima and the lower incipient lethal temperature in thermophilic freshwater decapods depend on the duration and temperature of the acclimation phase (Dı́az Herrera et al. 1998; Kır and Kumlu 2008). Since feral N. davidi have been present in Europe for approximately 2 decades, it is likely that, over time, a gradual insurgence of cold-adapted individuals among the established populations may result in a thermal niche expansion. However, thermal tolerance studies on N. davidi are urgently needed to confirm such assumptions. Furthermore, hybrids between N. denticulata, N. davidi, and N. palmata are found in nature (Onuki and Fuke 2022) and likely in the pet trade, as suggested by the relatively high genetic variability in feral N. davidi across the globe. Hybrids between populations with low genetic distance but high variability might have a higher thermal tolerance due to outbreeding overdominance, thus becoming more invasive than any parental population (Facon et al. 2005; Hupało et al. 2018).
An expansion of N. davidi into colder water might also result in the spread of associated pathogens, as seen for parasites that have been co-introduced outside their native range (Niwa and Ohtaka 2006; Maciaszek et al. 2021b; Kakui and Komai 2022; Schneider et al. 2022). Enemy release theory suggests that invaders partially lose their associated parasites when released into new environments (Torchin et al. 2003). Parasites may be lost due to a lack of suitable environmental conditions if their tolerance differs from that of their hosts or because they cannot complete their life cycle (Lymbery et al. 2014; Chalkowski et al. 2018). Therefore, the successful establishment of parasites in novel environments is more likely to occur in generalist species with simple life cycles, i.e., for species using vertical or horizontal transmission without the need for intermediate hosts (Lymbery et al. 2014; Prati et al. 2022).
These include the microsporidian EHP, which was previously detected in a German population of feral N. davidi (Schneider et al. 2022). This parasite possesses a simple life cycle, has been reported from a variety of invertebrate hosts, and can be considered a host generalist (Karthikeyan and Sudhakaran 2020; Krishnan et al. 2021; Munkongwongsiri et al. 2022; Wan Sajiri et al. 2023). The presence of EHP-infected feral N. davidi in sites with different water parameters indicates that this parasite is resilient to various environmental conditions. Accordingly, EHP is tolerant to salinity between 2 and 40 ppt, albeit its transmission at low salinity is reduced (Aranguren Caro et al. 2021), and its distribution range spans from tropical to temperate areas (Kim et al. 2022). Considering water salinization across Europe, bolstered by industrial discharges and droughts, EHP’s prevalence might increase in the forseable future. Moreover, the release of spores via waste water of home aquaria might constitute an additional source of propagules reaching aquatic environments.
The spread of EHP into European water can be attributed to pet trade dynamics as the vast majority of N. davidi sold on the global market are imported from Taiwan due to cheaper production costs (Maciaszek et al. 2018). Taiwanese farms are located in the southern part of the island in close proximity to the coast, an area also known for the intensive farming of non-native L. vannamei, the primary host for EHP (Aranguren Caro et al. 2021; Kim et al. 2022). This area relies heavily on groundwater for aquaculture, which is subject to salinization due to seawater intrusion (Dibaj et al. 2020). Water salinization coupled with intensive farming enhances the infectivity of EHP (Aranguren Caro et al. 2021); consequently, a higher load of spores is available in the environment. Thus, EHP spores might spread across aquaculture facilities via water movement and flying insects reaching N. davidi farms. Accordingly, dragonflies, often abundant near shrimp farms, serve as suitable hosts for EHP (Dewangan et al. 2023). Infected N. davidi may then end up unnoticed in the global market, favored by loose regulations and a lack of relevant information.
Spillover events, such as that of EHP from L. vannamei to susceptible native biota, are not rare events even in temperate regions, as exemplified by the outbreak of crayfish plague in Europe (Svoboda et al. 2017). Parasites that are able to switch from introduced to native host species can have greater pathogenic effects in native hosts because they lack a coevolutionary history (Mastitsky et al. 2010). However, non-native species might also acquire local parasites, amplifying (spillback) or diluting (dilution) their prevalence in native species (Strauss et al. 2012; Telfer and Bown 2012).
No parasite spillback from native species to N. davidi was detected. However, it is possible that other parasites like trematodes, e.g. Plagiorchidae, which are common in Central Europe, might find a suitable host in N. davidi, as has been seen for N. denticulata in its native range (Okabe and Shibue 1952). On the other hand, parasite spillover seems to have occurred between N. davidi and the invasive crayfish P. clarkii, as they shared a common microsporidian, Microsporidium sp. SCC01. Given the distant relationship between the two hosts, we can assume that this parasite is of a generalist nature in terms of host preference. The other two microsporidians found in N. davidi, namely Microsporidium sp. EFB01 and Microsporidium sp. HMI01 were not detected in other biota. Similarly, EHP was not detected in amphipods and isopods collected from the field or in G. pulex and A. aquaticus subjected to experimental infection. It is not excluded that a spillover might have occurred under experimental conditions with longer exposure to EHP infective spores. Nonetheless, it is still possible that EHP could spread to native biota, as has been observed for parasites in other invasive crustaceans (Bacela-Spychalska et al. 2012; Svoboda et al. 2017). Such spread may be facilitated by climate change and invasive species acting as vectors.
The ongoing rise in temperature and salinization of freshwater ecosystems is likely to bolster the spread of N. davidi and the transmissibility of EHP, enhancing the availability of propagules that might infect native biota. Procambarus clarkii, which often co-occurs with N. davidi in thermally altered waters, may act as a vector. Accordingly, EHP has recently been detected in Chinese populations of P. clarkii (Ling et al. 2024). This is particularly concerning as P. clarkii is widely distributed and is one of Europe’s most invasive crayfish species (Oficialdegui et al. 2019).
The discovery of two additional microsporidians in N. davidi compared to a previous study (Schneider et al. 2022) is a clear indication that screening for parasites invisible to the naked eye in pet-traded organisms is unfortunately not a common practice. Although microsporidians are more likely than other parasites to successfully establish in new areas due to their simple life cycle, generalist nature, and high environmental resilience, they are often neglected in biological invasion studies. The impact asserted by invading microsporidians on native crustaceans, with the exception of those found in prominent amphipod invaders such as the killer shrimp (Dikerogammarus villosus Sovinsky, 1894) and the demon shrimp (Dikerogammarus haemobaphes Eichwald, 1841), remains unclear and requires further studies.
To conclude, the range expansion of N. davidi in Europe is ongoing and is likely to continue in the future, supported by warming temperatures. The increasing presence of feral N. davidi and ongoing water salinization presents unique opportunities for the spread of EHP and potentially other host generalist parasites to native biota, leading to unpredictable outcomes. Therefore, it is imperative to closely monitor the expansion of feral N. davidi and undertake comprehensive experimental infection studies on a broader range of potential native hosts using various environmental conditions, spore concentrations, and exposures.
Data availability
The raw data supporting the conclusions of this article are available in the OSF repository, https://doi.org/https://doi.org/10.17605/OSF.IO/HQ5N2. The reported nucleotide sequence data are available in the GenBank database under the accession numbers OR610860-71 and OR613110-12 for hosts and OR616363-71 for microsporidians.
References
Aranguren Caro LF, Alghamdi F, De Belder K et al (2021) The effect of salinity on Enterocytozoon hepatopenaei infection in Penaeus vannamei under experimental conditions. BMC Vet Res 17:65. https://doi.org/10.1186/s12917-021-02778-0
Astrin JJ, Stüben PE (2008) Phylogeny in cryptic weevils: molecules, morphology and new genera of western Palaearctic Cryptorhynchinae (Coleoptera : Curculionidae). Invert Syst 22:503–522. https://doi.org/10.1071/IS07057
aymac (2021) iNaturalist observation. https://www.inaturalist.org/observations/80770934. Accessed 4 May 2023
Bacela-Spychalska K, Wattier RA, Genton C, Rigaud T (2012) Microsporidian disease of the invasive amphipod Dikerogammarus villosus and the potential for its transfer to local invertebrate fauna. Biol Invasions 14:1831–1842. https://doi.org/10.1007/s10530-012-0193-1
Bacela-Spychalska K, Wattier R, Teixeira M et al (2023) Widespread infection, diversification and old host associations of Nosema Microsporidia in European freshwater gammarids (Amphipoda). PLoS Pathog 19:e1011560. https://doi.org/10.1371/journal.ppat.1011560
Banha F, Diniz A, Anastácio PM (2019) Patterns and drivers of aquarium pet discharge in the wild. Ecol Ind 106:105513. https://doi.org/10.1016/j.ecolind.2019.105513
Bauer J, Jung-Schroers V, Teitge F et al (2021) Association of the alga Cladogonium sp. with a multifactorial disease outbreak in dwarf shrimp (Neocaridina davidi). Dis Aquat Org 146:107–115. https://doi.org/10.3354/dao03625
Bernery C, Bellard C, Courchamp F et al (2022) Freshwater fish invasions: a comprehensive review. Annu Rev Ecol Evol Syst 53:427–456. https://doi.org/10.1146/annurev-ecolsys-032522-015551
Beyer K (2004) Escapees of potentially invasive fishes from an ornamental aquaculture facility: the case of topmouth gudgeon Pseudorasbora parva. J Fish Biol 65:326–327. https://doi.org/10.1111/j.0022-1112.2004.00559ac.x
Bierbach D, Schutz C, Weimar N, et al (2022) Discovery of a new but established population of the guppy in Germany. https://doi.org/10.1101/2022.02.01.478389
Blackburn TM, Pyšek P, Bacher S et al (2011) A proposed unified framework for biological invasions. Trends Ecol Evol 26:333–339. https://doi.org/10.1016/j.tree.2011.03.023
Bláha M, Weiperth A, Patoka J et al (2022) The pet trade as a source of non-native decapods: the case of crayfish and shrimps in a thermal waterbody in Hungary. Environ Monit Assess 194:795. https://doi.org/10.1007/s10661-022-10361-9
Bojko J, Reinke AW, Stentiford GD et al (2022) Microsporidia: a new taxonomic, evolutionary, and ecological synthesis. Trends Parasitol. https://doi.org/10.1016/j.pt.2022.05.007
Britton JR, Lynch AJ, Bardal H et al (2023) Preventing and controlling nonnative species invasions to bend the curve of global freshwater biodiversity loss. Environ Rev 31:310–326. https://doi.org/10.1139/er-2022-0103
Cambray JA (2003) Impact on indigenous species biodiversity caused by the globalisation of alien recreational freshwater fisheries. Hydrobiologia 500:217–230. https://doi.org/10.1023/A:1024648719995
Capinha C, Larson ER, Tricarico E et al (2013) Effects of climate change, invasive species, and disease on the distribution of native European crayfishes. Biol Conserv 27:731–740. https://doi.org/10.1111/cobi.12043
Casatti L, Langeani F, Ferreira CP (2006) Effects of physical habitat degradation on the stream fish assemblage structure in a pasture region. Environ Manage 38:974. https://doi.org/10.1007/s00267-005-0212-4
Chaijarasphong T, Munkongwongsiri N, Stentiford GD et al (2021) The shrimp microsporidian Enterocytozoon hepatopenaei (EHP): biology, pathology, diagnostics and control. J Invertebr Pathol 186:107458. https://doi.org/10.1016/j.jip.2020.107458
Chalkowski K, Lepczyk CA, Zohdy S (2018) Parasite ecology of invasive species: conceptual framework and new hypotheses. Trends Parasitol 34:655–663. https://doi.org/10.1016/j.pt.2018.05.008
Cucherousset J, Olden JD (2011) Ecological impacts of nonnative freshwater fishes. Fisheries 36:215–230. https://doi.org/10.1080/03632415.2011.574578
deBruyn A (2019) Conservation ecology of a unique population of lake chub (Cyprinidae: Couesius plumbeus) : population size, movement ecology, habitat use and potential interactions with the exotic cherry shrimp (Neocaridina davidi var. red). University of British Columbia. https://open.library.ubc.ca/collections/ubctheses/24/items/1.0380902. Accessed 10 Feb 2024
Dewangan NK, Pang J, Zhao C et al (2023) Host and transmission route of Enterocytozoon hepatopenaei (EHP) from dragonfly to shrimp. Aquaculture 574:739642. https://doi.org/10.1016/j.aquaculture.2023.739642
Diaz Herrera F, Sierra Uribe E, Fernando Bückle Ramirez L, Garrido Mora A (1998) Critical thermal maxima and minima of Macrobrachium rosenbergii (Decapoda: Palaemonidae). J Therm Biol 23:381–385. https://doi.org/10.1016/S0306-4565(98)00029-1
Dibaj M, Javadi AA, Akrami M et al (2020) Modelling seawater intrusion in the Pingtung coastal aquifer in Taiwan, under the influence of sea-level rise and changing abstraction regime. Hydrogeol J 28:2085–2103. https://doi.org/10.1007/s10040-020-02172-4
drmichaelbraun (2021) iNaturalist observation. https://www.inaturalist.org/observations/80770934. Accessed 4 May 2023
Duggan IC (2010) The freshwater aquarium trade as a vector for incidental invertebrate fauna. Biol Invasions 12:3757–3770. https://doi.org/10.1007/s10530-010-9768-x
Emde S, Kochmann J, Kuhn T et al (2016) Cooling water of power plant creates “hot spots” for tropical fishes and parasites. Parasitol Res 115:85–98. https://doi.org/10.1007/s00436-015-4724-4
Facon B, Jarne P, Pointier JP, David P (2005) Hybridization and invasiveness in the freshwater snail Melanoides tuberculata: hybrid vigour is more important than increase in genetic variance. J Evol Biol 18:524–535. https://doi.org/10.1111/j.1420-9101.2005.00887.x
Fay MP (2023) exactci: exact P-values and matching confidence intervals for simple discrete parametric cases. https://github.com/cran/exactci
Folmer O, Black M, Hoeh W et al (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotechnol 3:294–299
Franch N, Clavero M, Garrido M et al (2008) On the establishment and range expansion of oriental weatherfish (Misgurnus anguillicaudatus) in NE Iberian Peninsula. Biol Invasions 10:1327–1331. https://doi.org/10.1007/s10530-007-9207-9
Franzaring J (2022) Discharge and water quality of the River Moselle from 1990 to 2020 as related to climatic changes and de-Industrialization. Water 14:3600. https://doi.org/10.3390/w14223600
Gallardo B, Clavero M, Sánchez MI, Vilà M (2016) Global ecological impacts of invasive species in aquatic ecosystems. Glob Change Biol 22:151–163. https://doi.org/10.1111/gcb.13004
Gherardi F (2007) Biological invaders in inland waters: profiles, distribution, and threats. Springer, Dordrecht
Gilioli G, Schrader G, Carlsson N et al (2017) Environmental risk assessment for invasive alien species: a case study of apple snails affecting ecosystem services in Europe. Environ Impact Assess Rev 65:1–11. https://doi.org/10.1016/j.eiar.2017.03.008
Gippet JMW, Bertelsmeier C (2021) Invasiveness is linked to greater commercial success in the global pet trade. PNAS. https://doi.org/10.1073/pnas.2016337118
Grabner DS, Weigand AM, Leese F et al (2015) Invaders, natives and their enemies: distribution patterns of amphipods and their microsporidian parasites in the Ruhr Metropolis, Germany. Parasit Vectors 8:419. https://doi.org/10.1186/s13071-015-1036-6
Hupało K, Riss HW, Grabowski M et al (2018) Climate change as a possible driver of invasion and differential in HSP70 expression in two genetically distinct populations of the invasive killer shrimp, Dikerogammarus villosus. Biol Invasions 20:2047–2059. https://doi.org/10.1007/s10530-018-1679-2
Hupało K, Copilaș-Ciocianu D, Leese F, Weiss M (2023) Morphology, nuclear SNPs and mate selection reveal that COI barcoding overestimates species diversity in a Mediterranean freshwater amphipod by an order of magnitude. Cladistics 39:129–143. https://doi.org/10.1111/cla.12520
Jabłońska A, Mamos T, Gruszka P et al (2018) First record and DNA barcodes of the aquarium shrimp, Neocaridina davidi, in Central Europe from thermally polluted River Oder canal. Poland Knowl Manag Aquat Ecosyst 419:14. https://doi.org/10.1051/kmae/2018004
Jaroenlak P, Sanguanrut P, Williams BAP et al (2016) A Nested PCR Assay to avoid false positive detection of the microsporidian Enterocytozoon hepatopenaei (EHP) in environmental samples in shrimp farms. PLoS ONE 11:e0166320. https://doi.org/10.1371/journal.pone.0166320
Jenkins T (2023) Mapmixture. https://github.com/Tom-Jenkins/Mapmixture
Kakui K, Komai T (2022) First record of Scutariella japonica (Platyhelminthes: Rhabdocoela) from Hokkaido, Japan, and notes on its host shrimp Neocaridina sp. Aff. davidi (Decapoda: Caridea: Atyidae). Aquat Anim. https://doi.org/10.34394/aquaticanimals.2022.0_AA2022-1
Karger DN, Conrad O, Böhner J et al (2017) Climatologies at high resolution for the earth’s land surface areas. Sci Data 4:170122. https://doi.org/10.1038/sdata.2017.122
Karthikeyan K, Sudhakaran R (2020) Exploring the potentiality of Artemia salina to act as a reservoir for microsporidian Enterocytozoon hepatopenaei of penaeid shrimp. Biocatal Agric Biotechnol 25:101607. https://doi.org/10.1016/j.bcab.2020.101607
Katoh K, Rozewicki J, Yamada KD (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20:1160–1166. https://doi.org/10.1093/bib/bbx108
Kim JH, Lee C, Jeon HJ et al (2022) First report on Enterocytozoon hepatopenaei (EHP) infection in Pacific white shrimp (Penaeus vannamei) cultured in Korea. Aquaculture 547:737525. https://doi.org/10.1016/j.aquaculture.2021.737525
Kır M, Kumlu M (2008) Critical thermal minima of Penaeus semisulcatus (Decapoda: Penaeidae) acclimated to four temperature levels. J World Aquac Soc 39:535–540. https://doi.org/10.1111/j.1749-7345.2008.00185.x
Klotz W, Miesen FW, Hüllen S, Herder F (2013) Two Asian fresh water shrimp species found in a thermally polluted stream system in North Rhine-Westphalia, Germany. Aquat Invasions 8:333–339. https://doi.org/10.3391/ai.2013.8.3.09
Krabbelkeller (2020) Garnelen im Guppybach. https://www.youtube.com/watch?v=szzkUtSSH4U. Accessed 12 Oct 2023
Krishnan AN, Kannappan S, Aneesh PT et al (2021) Polychaete worm: a passive carrier for Enterocytozoon hepatopenaei in shrimp. Aquaculture 545:737187. https://doi.org/10.1016/j.aquaculture.2021.737187
Landaeta-Aqueveque C, Henríquez A, Cattan PE (2014) Introduced species: domestic mammals are more significant transmitters of parasites to native mammals than are feral mammals. Int J Parasitol 44:243–249. https://doi.org/10.1016/j.ijpara.2013.12.002
Leigh JW, Bryant D (2015) POPART: full-feature software for haplotype network construction. Methods Ecol Evol 6:1110–1116. https://doi.org/10.1111/2041-210X.12410
Levitt-Barmats Y, Yanai Z, Cohen T, Shenkar N (2019) Life-history traits and ecological characteristics of the ornamental shrimp Neocaridina denticulata (De Haan, 1844), recently introduced into the freshwater systems of Israel. Aquat Invasions 14:684–702. https://doi.org/10.3391/ai.2019.14.4.08
Liao C-C, Shin J, Chen L-R et al (2018) First molecular identification of Vorticella sp. from freshwater shrimps in Tainan, Taiwan. Int J Parasitol Parasites Wildl 7:415–422. https://doi.org/10.1016/j.ijppaw.2018.10.002
Ling B, Wu Y, Yu Q et al (2024) Ecytonucleospora hepatopenaei proliferate in Procambarus clarkii: a warning for crayfish and shrimp aquaculture. Aquaculture 581:40457. https://doi.org/10.1016/j.aquaculture.2023.740457
Lipták B, Sebastian P, Francisco JO et al (2023) Springing up like mushrooms: established populations of invasive red swamp crayfish in Slovakia. https://www.researchsquare.com. Accessed 15 Feb 2024
Lipták B, Vitázková B (2015) Beautiful, but also potentially invasive. Ekol Bratisl 34:155–162. https://doi.org/10.1515/eko-2015-0016
Lobo JM, Jiménez-Valverde A, Real R (2008) AUC: a misleading measure of the performance of predictive distribution models. Glob Ecol Biogeogr 17:145–151. https://doi.org/10.1111/j.1466-8238.2007.00358.x
Low BW, Zeng Y, Tan HH, Yeo DCJ (2021) Predictor complexity and feature selection affect Maxent model transferability: evidence from global freshwater invasive species. Divers Distrib 27:497–511. https://doi.org/10.1111/ddi.13211
Lukas J, Kalinkat G, Kempkes M et al (2017a) Feral guppies in Germany: a critical evaluation of a citizen science approach as biomonitoring tool. Bull Fish Biol 17:13–27
Lukas J, Jourdan J, Kalinkat G et al (2017b) On the occurrence of three non-native cichlid species including the first record of a feral population of Pelmatolapia (Tilapia) mariae (Boulenger, 1899) in Europe. R Soc Open Sci 4:170160. https://doi.org/10.1098/rsos.170160
Lymbery AJ, Morine M, Kanani HG et al (2014) Co-invaders: the effects of alien parasites on native hosts. Int J Parasitol Parasites Wildl 3:171–177. https://doi.org/10.1016/j.ijppaw.2014.04.002
Maceda-Veiga A, Cable J (2019) Diseased fish in the freshwater trade: from retailers to private aquarists. Dis Aquat Org 132:157–162. https://doi.org/10.3354/dao03310
Maceda-Veiga A, Escribano-Alacid J, De Sostoa A, García-Berthou E (2013) The aquarium trade as a potential source of fish introductions in southwestern Europe. Biol Invasions 15:2707–2716. https://doi.org/10.1007/s10530-013-0485-0
Maciaszek R, Kamaszewski M, Struzynski W, Lapa P (2018) Epibionts of ornamental freshwater shrimps bred in Taiwan. Ann Wars Univ Life Sci-SGGW Anim Sci 57:2. https://doi.org/10.22630/AAS.2018.57.2.13
Maciaszek R, Jabłońska A, Hoitsy M et al (2021a) First record and DNA barcodes of non-native shrimp, Caridina babaulti (Bouvier, 1918) in Europe. Eur Zool J 88:816–823. https://doi.org/10.1080/24750263.2021.1944337
Maciaszek R, Świderek W, Kaliszewicz A et al (2021b) First report of Scutariella japonica (Matjašič, 1990), a temnocephalid epibiont from South-East Asia, found on introduced ornamental freshwater shrimp in European waters. Knowl Manag Aquat Ecosyst 422:19. https://doi.org/10.1051/kmae/2021018
Maciaszek R, Świderek W, Prati S et al (2023) Epibiont cohabitation in freshwater shrimp Neocaridina davidi with the description of two species new to science, Cladogonium kumaki sp. nov and Monodiscus kumaki sp. nov, and redescription of Scutariella japonica and Holtodrilus truncatus. Animals 13:1616. https://doi.org/10.3390/ani13101616
Marchowski D, Ławicki Ł (2023) Unprecedented mass mortality of aquatic organisms in the River Oder. Oryx 57:9. https://doi.org/10.1017/S0030605322001387
Mastitsky SE, Karatayev AY, Burlakova LE, Molloy DP (2010) Biodiversity research: Parasites of exotic species in invaded areas: does lower diversity mean lower epizootic impact? Divers Distrib 16:798–803. https://doi.org/10.1111/j.1472-4642.2010.00693.x
Minh BQ, Schmidt HA, Chernomor O et al (2020) IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol Biol Evol 37:1530–1534. https://doi.org/10.1093/molbev/msaa015
Mrugała A, Kozubíková-Balcarová E, Chucholl C et al (2015) Trade of ornamental crayfish in Europe as a possible introduction pathway for important crustacean diseases: crayfish plague and white spot syndrome. Biol Invasions 17:1313–1326. https://doi.org/10.1007/s10530-014-0795-x
Munkongwongsiri N, Thepmanee O, Lertsiri K et al (2022) False mussels (Mytilopsis leucophaeata) can be mechanical carriers of the shrimp microsporidian Enterocytozoon hepatopenaei (EHP). J Invertebr Pathol 187:107690. https://doi.org/10.1016/j.jip.2021.107690
Namaei Kohal M, Esmaeili Fereidouni A, Firouzbakhsh F, Hayati I (2018) Effects of dietary incorporation of Arthrospira (Spirulina) platensis meal on growth, survival, body composition, and reproductive performance of red cherry shrimp Neocaridina davidi (Crustacea, Atyidae) over successive spawnings. J Appl Phycol 30:431–443. https://doi.org/10.1007/s10811-017-1220-5
Niwa N, Ohtaka A (2006) Accidental introduction of symbionts with imported freshwater shrimps. Assess Control Biol Invasion Risks 1:182–186
Oficialdegui FJ, Clavero M, Sánchez MI et al (2019) Unravelling the global invasion routes of a worldwide invader, the red swamp crayfish (Procambarus clarkii). Freshw Biol 64:1382–1400. https://doi.org/10.1111/fwb.13312
Ohtaka A, Gelder SR, Nishino M et al (2012) Distributions of two ectosymbionts, branchiobdellidans (Annelida: Clitellata) and scutariellids (Platyhelminthes: “Turbellaria”: Temnocephalida), on atyid shrimp (Arthropoda: Crustacea) in southeast China. J Nat Hist 46:1547–1556. https://doi.org/10.1080/00222933.2012.692826
Okabe K, Shibue H (1952) A new second intermediate host, Neocaridina denticulata, for Plagiorchis muris (Tanabe): Plagiorchidae. Jpn J Med Sci Biol 5:257–258. https://doi.org/10.7883/yoken1952.5.257
Olden JD, Comte L, Giam X (2018) The Homogocene: a research prospectus for the study of biotic homogenisation. NeoBiota 37:23–36. https://doi.org/10.3897/neobiota.37.22552
Ondračková M, Tkachenko MY, Bartáková V et al (2023) Population genetic structure, parasite infection and somatic condition of pumpkinseed Lepomis gibbosus (Actinopterygii: Centrarchidae) in the Oder river basin. J Fish Biol 102:426–442. https://doi.org/10.1111/jfb.15273
Onuki K, Fuke Y (2022) Rediscovery of a native freshwater shrimp, Neocaridina denticulata, and expansion of an invasive species in and around Lake Biwa, Japan: genetic and morphological approach. Conserv Genet 23:967–980. https://doi.org/10.1007/s10592-022-01467-1
Padilla DK, Williams SL (2004) Beyond ballast water: aquarium and ornamental trades as sources of invasive species in aquatic ecosystems. Front Ecol Environ 2:131–138. https://doi.org/10.1890/1540-9295(2004)002[0131:BBWAAO]2.0.CO;2
Patoka J, Bláha M, Devetter M et al (2016) Aquarium hitchhikers: attached commensals imported with freshwater shrimps via the pet trade. Biol Invasions 18:457–461. https://doi.org/10.1007/s10530-015-1018-9
Patoka J, Magalhães ALB, Kouba A et al (2018) Invasive aquatic pets: failed policies increase risks of harmful invasions. Biodivers Conserv 27:3037–3046. https://doi.org/10.1007/s10531-018-1581-3
Phillips SJ, Anderson RP, Dudík M et al (2017) Opening the black box: an open-source release of Maxent. Ecography 40:887–893. https://doi.org/10.1111/ecog.03049
PM (2017) Une invasion de crevettes “d’aquarium” dans la riviere du Mât. In: Journal de Reunion Island. https://www.especesinvasives.re/presse/article/une-invasion-de-crevettes-d. Accessed 5 Jan 2023
Prati S, Grabner DS, Pfeifer SM et al (2022) Generalist parasites persist in degraded environments: a lesson learned from microsporidian diversity in amphipods. Parasitology 149:973–982. https://doi.org/10.1017/S0031182022000452
R Core Team (2023) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Raju KS, Kumar DN (2020) Review of approaches for selection and ensembling of GCMs. J Water Clim Chang 11:577–599. https://doi.org/10.2166/wcc.2020.128
Ribeiro F, Elvira B, Collares-Pereira MJ, Moyle PB (2008) Life-history traits of non-native fishes in Iberian watersheds across several invasion stages: a first approach. Biol Invasions 10:89–102. https://doi.org/10.1007/s10530-007-9112-2
Ricciardi A, MacIsaac HJ (2011) Impacts of biological invasions on freshwater ecosystems. Fifty years of invasion ecology: the legacy of Charles Elton. Wiley & Sons Ltd, Blackwell, Chichester, pp 211–224
Ripley B, Venables B, Bates DM, et al (2023) MASS: support functions and datasets for Venables and Ripley’s MASS. https://github.com/cran/MASS
RWE (2021) Decommissioning activities at year-end: RWE implements legally mandated exit from coal and nuclear power. https://www.rwe.com/en/press/rwe-power/2021-12-30-rwe-implements-legally-mandated-exit-from-coal-and-nuclear-power/. Accessed 10 Feb 2024
Saul W-C, Roy HE, Booy O et al (2017) Assessing patterns in introduction pathways of alien species by linking major invasion data bases. J Appl Ecol 54:657–669. https://doi.org/10.1111/1365-2664.12819
Savini D, Occhipinti-Ambrogi A, Marchini A et al (2010) The top 27 animal alien species introduced into Europe for aquaculture and related activities. J Appl Ichthyol 26:1–7. https://doi.org/10.1111/j.1439-0426.2010.01503.x
Schneider R, Prati S, Grabner D, Sures B (2022) First report of microsporidians in the non-native shrimp Neocaridina davidi from a temperate European stream. Dis Aquat Org 150:125–130. https://doi.org/10.3354/dao03681
Schoolmann G, Arndt H (2017) Leaf-litter preferences of the introduced freshwater shrimps Atyaephyra desmarestii and Neocaridina davidi. Crustaceana 90:1715–1730. https://doi.org/10.1163/15685403-00003736
Schoolmann G, Arndt H (2018) Population dynamics of the invasive freshwater shrimp Neocaridina davidi in the thermally polluted Gillbach stream (North Rhine-Westphalia, Germany). Limnologica 71:1–7. https://doi.org/10.1016/j.limno.2018.05.001
Seker M, Gumus V (2022) Projection of temperature and precipitation in the Mediterranean region through multi-model ensemble from CMIP6. Atmos Res 280:106440. https://doi.org/10.1016/j.atmosres.2022.106440
Serezli R, Atalar MS, Hamzacebi S et al (2017) To what extent does temperature affect sex ratio in red cherry shrimp, Neocaridina davidi? The scenario global warming to offspring sex ratio. Fresen Environ Bull 26:7575–7579
Simberloff D, Martin J-L, Genovesi P et al (2013) Impacts of biological invasions: what’s what and the way forward. Trends Ecol Evol 28:58–66. https://doi.org/10.1016/j.tree.2012.07.013
Strauss A, White A, Boots M (2012) Invading with biological weapons: the importance of disease-mediated invasions. Funct Ecol 26:1249–1261. https://doi.org/10.1111/1365-2435.12011
Subash P, Uma A, Ahilan B (2022) Early responses in Penaeus vannamei during experimental infection with Enterocytozoon hepatopenaei (EHP) spores by injection and oral routes. J Invertebr Pathol 190:107740. https://doi.org/10.1016/j.jip.2022.107740
Sures B (2011) Parasites of animals. In: Simberloff D, Rejmánek M (eds) Encyclopedia of biological invasions. Univ of California Press, Berkely and Los Angeles, pp 500–503
Sures B, Nachev M, Grabner D (2019) The Rhine river as hotspot of parasite invasions. In: Mehlhorn H, Klimpel S (eds) Parasitology research monographs 12: parasite and disease spread by major rivers on earth. Past and future perspectives. Springer Nature, Cham, Switzerland, pp 409–429
Svoboda J, Mrugała A, Kozubíková-Balcarová E, Petrusek A (2017) Hosts and transmission of the crayfish plague pathogen Aphanomyces astaci: a review. J Fish Dis 40:127–140. https://doi.org/10.1111/jfd.12472
Tangprasittipap A, Srisala J, Chouwdee S et al (2013) The microsporidian Enterocytozoon hepatopenaei is not the cause of white feces syndrome in whiteleg shrimp Penaeus (Litopenaeus) vannamei. BMC Vet Res 9:139. https://doi.org/10.1186/1746-6148-9-139
Telfer S, Bown K (2012) The effects of invasion on parasite dynamics and communities. Funct Ecol 26:1288–1299. https://doi.org/10.1111/j.1365-2435.2012.02049.x
Tomjamonneau (2023) iNaturalist observation. https://www.inaturalist.org/observations/194449320. Accessed 10 February 2024.
Torchin ME, Lafferty KD, Dobson AP et al (2003) Introduced species and their missing parasites. Nature 421:628–630. https://doi.org/10.1038/nature01346
Turbelin AJ, Diagne C, Hudgins EJ et al (2022) Introduction pathways of economically costly invasive alien species. Biol Invasions 24:2061–2079. https://doi.org/10.1007/s10530-022-02796-5
van Vliet MTH, Ludwig F, Zwolsman JJG et al (2011) Global river temperatures and sensitivity to atmospheric warming and changes in river flow. Water Resour Res 47:W02544. https://doi.org/10.1029/2010WR009198
van Vliet MTH, Franssen WHP, Yearsley JR et al (2013) Global river discharge and water temperature under climate change. Glob Environ Change 23:450–464. https://doi.org/10.1016/j.gloenvcha.2012.11.002
Veselý L, Buřič M, Kouba A (2015) Hardy exotics species in temperate zone: can “warm water” crayfish invaders establish regardless of low temperatures? Sci Rep 5:16340. https://doi.org/10.1038/srep16340
Wan Sajiri WMH, Kua BC, Borkhanuddin MH (2023) Detection of Enterocytozoon hepatopenaei (EHP) (microsporidia) in several species of potential macrofauna-carriers from shrimp (Penaeus vannamei) ponds in Malaysia. J Invertebr Pathol 198:107910. https://doi.org/10.1016/j.jip.2023.107910
Wang Y, Chen J, Na Y et al (2023) Ecytonucleospora hepatopenaei n. gen. et comb. (Microsporidia: Enterocytozoonidae): a redescription of the Enterocytozoon hepatopenaei (Tourtip et al., 2009), a microsporidian infecting the widely cultivated shrimp Penaeus vannamei. J Invertebr Pathol 201:107988. https://doi.org/10.1016/j.jip.2023.107988
Warren DL, Matzke NJ, Cardillo M et al (2021) ENMTools 1.0: an R package for comparative ecological biogeography. Ecography 44:504–511. https://doi.org/10.1111/ecog.05485
Weber S, Traunspurger W (2016) Influence of the ornamental red cherry shrimp Neocaridina davidi (Bouvier, 1904) on freshwater meiofaunal assemblages. Limnologica 59:155–161. https://doi.org/10.1016/j.limno.2016.06.001
Weigand AM, Kremers J, Grabner DS (2016) Shared microsporidian profiles between an obligate (Niphargus) and facultative subterranean amphipod population (Gammarus) at sympatry provide indications for underground transmission pathways. Limnologica 58:7–10. https://doi.org/10.1016/j.limno.2016.01.005
Weiperth A, Gábris V, Danyik T et al (2019) Occurrence of non-native red cherry shrimp in European temperate waterbodies: a case study from Hungary. Knowl Manag Aquat Ecosyst 420:9. https://doi.org/10.1051/kmae/2019002
Weiperth A, Bláha M, Szajbert B et al (2020) Hungary: a European hotspot of non-native crayfish biodiversity. Knowl Manag Aquat Ecosyst 421:43. https://doi.org/10.1051/kmae/2020035
Wetzel RG (2001) Limnology: lake and river ecosystems. Academic Press, San Diego
Wood LE, Guilder J, Brennan ML et al (2022) Biosecurity and the ornamental fish trade: a stakeholder perspective in England. J Fish Biol 100:352–365. https://doi.org/10.1111/jfb.14928
Woolway RI, Jennings E, Shatwell T et al (2021) Lake heatwaves under climate change. Nature 589:402–407. https://doi.org/10.1038/s41586-020-03119-1
Zhou C, Feng M, Tang Y et al (2021) Species diversity of freshwater shrimp in Henan Province, China, based on morphological characters and COI mitochondrial gene. Ecol Evol 11:10502–10514. https://doi.org/10.1002/ece3.7855
Zhu X, Wittner M, Tanowitz HB et al (1993) Small subunit rRNA sequence of Enterocytozoon bieneusi and its potential diagnostic role with use of the polymerase chain reaction. J Infect Dis 168:1570–1575. https://doi.org/10.1093/infdis/168.6.1570
Acknowledgements
This study was performed within the Collaborative Research Center (CRC) RESIST (A09) and funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)—CRC 1439/1—project number: 426547801. We are grateful for the support from the Open Access Publication Fund of the University of Duisburg-Essen. We thank Mandy Hajnsek for her assistance in the laboratory, Richard Schneider for assisting in the field, and Martin Dobrota for signaling the presence of N. davidi in Slovakia and for helping in the field. Lastly, we are very grateful to the anonymous reviewers for their extremely valuable and thorough comments and suggestions, which have helped substantially improve the quality of our manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was performed within the Collaborative Research Center (CRC) RESIST (A09) and funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)—CRC 1439/1—project number: 426547801. We acknowledge support from the Open Access Publication Fund of the University of Duisburg-Essen.
Author information
Authors and Affiliations
Contributions
SP and DG conceived the study, and BS supervised the project. SP, DG, KH, AW, RM, BL, and FB carried out the sampling. SP, KH, and JB carried out laboratory analyses. SP performed the data analyses. SP wrote the first draft of the manuscript, and all authors contributed critically to the final draft. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Prati, S., Grabner, D.S., Hupało, K. et al. Invisible invaders: range expansion of feral Neocaridina davidi offers new opportunities for generalist intracellular parasites. Biol Invasions 26, 2499–2523 (2024). https://doi.org/10.1007/s10530-024-03324-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-024-03324-3