Abstract
Traditional coffee cultivation in Cuba is the result of a complex interaction between different flora species creating agroforestry systems widely spread in mountainous area. The systems, product of local traditional knowledge, are mainly devoted to coffee production but, thanks to the interaction with other species, farmers provide different food products both for self-consumption and to be sold. Furthermore, the adoption of shade trees in order to reach a better quality of the coffee cultivated creates particular microclimate conditions favorable for microorganisms, fauna species and also for spontaneous flora species. According to this it is clear the relationships between traditional knowledge and biodiversity preservation which is fundamental also for improving the surrounding environment, avoiding floods or hydrogeological instability damages, concurring to climate change mitigation and carbon storage. Traditional agroforestry systems are one of the best example of coexistence and coevolution between man and nature, being an historical system adopted by local communities to satisfy their needs in total respect of the surrounding environment. Considering this, the promotion and maintenance of this kind of systems and knowledge related might constitute a valid example to actively preserve biodiversity while respecting human needs for food and livelihood security. These systems are also of particular importance considering the importance of coffee as a beverage served in many countries of the world, but often produced in intensive plantations. This paper shows the high sustainability of coffee production under the shade of trees and support a new concept of food quality contributing to preserve local cultures and environments.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Agrobiodiversity is the synergy and interaction between living things, land, technology, and social systems (Long et al. 2003) and conserving it in farming systems could provide direct and indirect benefits necessary for livelihoods and ecosystem functioning (Chirwa et al. 2008). Agroforestry systems are characterized by important elements that can play a significant role in the adaptation to climate change including changes in microclimate structure, protection through provision of permanent cover and opportunities for diversification of the agricultural systems, improving efficiency of use of soil, water and climatic resources, contribution to soil fertility improvement, reducing carbon emissions and increasing sequestration, and promoting gender equity (Rao et al. 2007) while being characterized by high socioeconomic and ecological complexity (Trosper et al. 2011). In addition, it has been exposed that agroforestry systems with greater structural complexity are capable of harboring high biodiversity (Santoro et al. 2020). The biodiversity associated to agroforestry is part of the wider concept of biocultural diversity (Agnoletti and Rotherham 2015) adopted by FAO in the criteria for the designation of agricultural heritage systems among the GIAHS sites (Globally Important Agricultural Heritage Systems).
Coffee cultivation is traditionally based on agroforestry systems considering that shade trees reduce the stress of coffee by ameliorating adverse climatic conditions and nutritional imbalances (Beer et al. 1997). Coffee is an extremely important agricultural commodity, produced in about 80 tropical countries, with an estimated 125 million people depending on it for their livelihoods in Latin America, Africa, and Asia (Krishnan 2017). Over the past 50 years, both production and consumption of coffee have risen considerably but, especially smallholders, who are the main producers, are facing growing challenges derived from climate change and more difficult natural growing conditions (FAO 2015). In Cuba, the culture of coffee has been important in many mountainous areas as a historically key element of the agricultural evolution of its landscapes (Ramírez and Paredes 2003). The first coffee plants were introduced in Cuba from 1748, having place in a farm of Wajay, a town located at the periphery of Havana city in the western geography of the country (Lapique and García 2014); but it is from the Revolution of Haiti that the coffee production in Cuba has had a particular increment (Fernandez 2012). Coffee plantations are predominant as coffee is a commercialized product in the majority of the island farms even if it is not the only species cultivated as it coexists with other cultivated species increasing the crops agricultural diversity being important repositories of biological richness for groups such as trees and epiphytes, mammals, birds, reptiles, amphibians, and arthropods (Moguel and Toledo 1999). Beside this, Cuba has the privilege of having a remarkable wealth of endemic or exclusive flora and fauna. Vales and Vilamajo (2001) highlight Cuba as the island of the Antilles with greater biological diversity, both in total species richness and in the degree of endemism, which considerably increases the value of Cuban biodiversity.
Considering this the present paper takes in consideration two traditional coffee production agroforestry systems located at the opposite extremes of Cuba with the aim of analyzing their structure and the agrobiodiversity related. The two systems considered are Sierra del Rosario and Sierra Maestra, both covering mountainous surfaces and still characterized by traditional practices related to coffee production. The paper aims to stress the importance of presence and maintenance of traditional agroforestry systems for local agrobiodiversity preservation. The agrobiodiversity linked to the presence of traditional practices, in fact, can be fundamental both from an economic and natural point of view providing multiple benefits. In addition, the maintenance of such systems is a valid example of a sustainable alternative in order to satisfy social-economical needs while preserving the local environment and cultural heritage. The spatial scale chosen for this paper is also aimed at understanding “gamma” diversity, the one at landscape level, considering larger geographic regions compared to small sampling units, including human activity.
Material and Methods
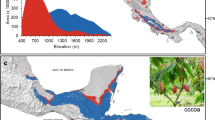
The present study focused on two areas located at the two opposite extremes of the island of Cuba (Fig. 1): Sierra Maestra and Sierra del Rosario. The choice has been made mainly considering the importance of coffee production in both areas history. Furthermore, in both areas the coffee production has been maintained according to the traditional practices creating complex and sustainable agroforestry systems.
The area of Sierra del Rosario, in the north-west of Cuba, covers 80.000 hectares and it is located in Artemisa province. It is 70 km away from the capital, Havana and it is part of a mountainous area with maximum high of 560 m a.s.l.. The eastern portion of the site analyzed is also part of the Sierra del Rosario Biosphere Reserve, included in the official UNESCO list in 1984 and recognized as a reserve showing a complex geological structure, with a great diversity of rocks that produce different and special soils, which in part, determine flora endemism in its landscape (García and Castiñeiras 2006). Sierra del Rosario, according to the Koppen classification (Fig. 3), belongs to the tropical area including both the monsoon (Am) and the savannah (Aw) climate subcategories (Kottek et al. 2006).
Sierra Maestra is set in the opposite side of the island, in the south-east of Cuba. The area considered covers a surface of 494.889 hectares of which 59.066 hectares are characterized by coffee and cocoa cultivation (Fig. 2). It extends across 14 municipalities in Granma, Santiago de Cuba and Guantanamo provinces. The area is set along the Sierra Maestra massif from 240 to 1200 m a.s.l.. According to the Koppen climate classification it belongs to Tropical, both monsoon (Am) and savannah (Aw), and Temperate, no dry season, warm summer (Cfb) classes (Fig. 3). Inside the study area boundaries there are also protected areas, part of the National System of Protected areas which are of as national or local significance (Fig. 2). Furthermore, as in the case of Sierra del Rosario even in this area there is a surface included in UNESCO Biosphere Reserve List from 1987: the Baconao Biosphere Reserve, located in the south of the provinces of Santiago de Cuba and Guantánamo, in the eastern region of Cuba.
The two areas choice has been made according to the recognized importance of traditional coffee cultivation in both of them and because they are, even in the present days, a good example of agroforestry systems sustainable management.
The study carried out is based on the comparison between the two sites. The analysis mainly focuses on the agroforestry systems structure characteristics, the ecological interactions created by the systems themselves and on the traditional practices and agrobiodiversity related. In particular: the first part of the study aims to provide a description of coffee agroforestry system structure, practices and traditional knowledge, focusing on their effects on the surrounding environment, while the second part provides a detailed assessment of coffee agroforestry system agrobiodiversity richness considering the different varieties of species both endemic and cultivated which are deeply related to the maintenance of the systems themselves.
The biodiversity inventories in Sierra del Rosario is the accumulative knowledge resulting of expeditions and field work jointly conducted over the last 35 years between Sierra del Rosario Ecological Station (EESR) and different institutions involved in systematics and taxonomy in Cuba, as is the case of the Cuban National Garden, the Institute of Ecology and Systematics and the National Museum of Natural History. Agricultural diversity has been compiled as the result of field works jointly conducted by INIFAT and EESR since 1999 and recently updated by COBARB project (2012–2019) due to expeditions and surveys conducted among 75 traditional farmers placed in agricultural landscapes.
Results
In Cuba, the introduction of agroforestry system and practices related dates back to the eighteenth century when French settlers introduced coffee cultivation (Coffea arabica) in the mountainous areas of the country, where by the first time trees were associated with permanent cultivation, with the goal of obtaining sustained yields in hillside conditions, not forgetting the “conucos” or family parcels, a traditional form of integrated production known as an agrosilvopastoral practice.
In traditional coffee plantations in the mountainous areas of the Sierra Maestra de Cuba, a multiple cultivation method is practiced, since they are established on the same surface, in addition to coffee trees and trees, Dioscore alata and Xathosoma sp., some varieties of banana are cultivated. Even if coffee is the main source of resources, fruit species are important supplements to the family's daily diet. Currently 57 673.36 ha are dedicated to coffee cultivation in Sierra Maestra.
Sierra del Rosario traditional system is similar to the one of Sierra Maestra: the farms characterized by coffee cultivation of important extension see also the presence of other species also important for farmers livelihood like Xanthosoma sagittifolium, Citrus, Pouteria sapota, Manhiot esculenta, Cucurbita moschata and Ipomoea batatas. Coffee cultivation is in fact part of the system diversity being particularly valorized in polycultural systems as when it is mixed with fruit trees as plátano (Musa spp.), mamey colorado (Pouteria sapota) and aguacate (Persea americana) used as shade trees. In addition, the traditional coffee system is characterized by a series of herbaceous species used as food.
The traditional structure of traditional agroforestry system for coffee production is the result of a complex combination of species strongly influencing the agrobiodiversity richness of the system itself (Fig. 4). The farmers use to create shade with species like fruit trees that can contribute to their sustenance but it is also common the utilization of forestry species like Ficus aurea and Trophis racemosa. Among the species adopted to create shade farmers prefer Gliricidia sepium and Samanea saman (Gonzalez Alvarez et al. 2016).
The traditional polyculture coffee garden is a complex combination of multiple species needed both from an economic and ecological point of view. It is important for farmers’ sustenance the presence of different species in the system that contributes to food security and good livelihood conditions but, at the same time, it results fundamental from an environmental point of view providing multiple ecological benefits while improving the agrobiodiversity richness (from Moguel and Toledo 1999)
From an ecological point of view, in agroforestry systems, nutrients enter through various sources such as rain and organic residues; these can be accumulated either in the shade trees cultivation, soil or litter. At the same time, interactions occur between crop layers such as residue deposition, infiltration, absorption and mineralization; likewise, outputs can occur by crops yields, leaching, runoff, and denitrification processes. The increase in litter shade trees promotes a diversity of decomposer organisms and other species can provide ecosystem services such as pest control (Petite Aldana et al. 2019) (Fig. 5) while protecting the soil from direct insolation, helping maintain organic matter, reducing evaporation and maintaining soil productivity (Siebert 2002).
Besides the ecological functions derived from the system complexity, agricultural diversification is promoted by local farmers as a way to increase options of keep economic inputs in face of extreme climatic impacts. In general, the traditional farms are featured in a high integration of crop production and animal breeding, which is highly desirable under the perspective of agroecology (Figs. 6 and 7).
Sierra del Rosario is recognized for its crop genetic resources with high levels of varietal diversity of coffee (Coffea sp), maize (Zea mays), lima bean (Phaseolus lunatus), common bean (Phaseolus vulgaris), chilli (Capsicum sp.), etc. (Castiñeiras et al. 2006). The multistate agroecosystems areas can include up to 500 plant species most of which are ornamental and medicinal, followed by fruit and timber species. There is also an amount of plants used and preserved by farmers because of its sacred conditions. Data base of the Ecological Station of RBSR reports a floristic diversity that reaches 889 upper and 281 lower plants, of which 11% are endemic. The richness of Plant Genetic resources of Sierra del Rosario has been object of research and in situ conservation by INIFAT, in collaboration with international partners like Bioversity International (García and Castiñeiras 2006). In this sense, the last project conducted between this organizations (COBARB), budged by 7 GEF and implemented by UNEP, had actualize the inventories of the agrobiodiversity in many traditional systems placed inside or around the RBSR. The actions of this project have been also aware of the landscape peculiarities of the regions (Sánchez et al. 2015).
In the following tables are expressed the species characterizing the systems: the Table 3 is only dedicated to the Sierra del Rosario flora species, Supplementary Material Table 1 to Sierra Maestra species and the last one (Supplementary Material Table 2) contains the species that the two systems have in common.
The fauna diversity is also very rich, both for wild species and livestock. In Sierra del Rosario, among livestock species (Table 1), the most important in terms of number of individuals are pigs. In traditional farms the traditional race of pigs is named “creole”. The term “creole” is used academically in reference to the animals considered genetically descendants from those brought to Cuba by Spaniard conquerors. However, the farmers make a broader use of the term; many times they use the word “creole” to name the colored animals or those born in their farms without racial control.
Wild fauna reported at Sierra del Rosario, in studies also conducted at the RBSR, shows a high rate of endemics, among them the best represented groups are Birds (131 species identified including 12 endemics), Reptiles (33 species identified including 27 endemics), Amphibia (16 species identified including 13 endemics), Mammals: (including a remarkable number of bats, with 11 species and among there is very important the presence of two species of genus Capromys, commonly known as hutias).
Some of the most charismatic species are reported in the best-preserved areas, but is also possible to observed them in the mountains traditional systems. Ramírez y Paredes and Pupo (2005) remarks that in Sierra del Rosario is not very difficult to see the high flight of the “Gavilán del Monte” (rapacios bird: Buteo jamaicensis) or the hummingbird (Chlorostilbon ricordii), even in the home gardens, but also other birds like: the national one “Tocororo” (Plioterus temnurus), the “Cartacuba” (Todus multicolor), the colorful of the “Bijirita” (Dendroica discolor) and the Common Bijirita and the familiar and endemic Tomeguín del Pinar (Tiaris canora), as well as the beautiful and laborious Carpenters: the “Scapular” (Colaptes auratus), the “Churroso” (Colaptes fernandinae), the “Jobado” (Centurus superciliaris) and the “Green” (Xiphidiopicus percussus). Population of butterflies such as: Calisto herophile, Eurema larae, the beautiful Helicornius charithonius, Appias drucilla, Utetheisa ornatrix and Marpesia chiron, among many others.
In the areas where calcareous rocks predominate, it is easy to find several species of mollusks, with showy shapes and colors, from the Zachrycia rangelina, the largest terrestrial mollusk in the region, but also the species Emoda sagraiana and Emoda marginata, Helicina adspersa, Plicathyrella assimilis, and Vianas regina Morelet, among other twenty-three species reported. This diversity is consistent with the character of Cuba as a world center of diversity of mollusks.
In the case of Sierra del Rosario, as the result of the field visits conducted by COBARB project, there were observed examples of the use of wild animal used for feeding, as is the case of shrimps and hutias, which are endemic species. In the case of the Cuban majá (Epicrates angulifer) the biggest snake of the island, which is also an endemic, it is reported the use of its fats for medical purposes. In the case of bees, there are either the species “abeja europea” (Apis mellifera) and “abeja de la tierra” (Melipona beecheii) that can be managed. Even when both bees are considered as introduced, there are wild populations of these species which are present in the forest or agroforestry areas of the landscapes.
Also Sierra Maestra has a remarkable endemic fauna (Tables 2, 3 and 4).
Among all the species in particular the mollusks could be highlighted considering their multiple uses in Sierra Maestra where they have been used as jewels and personal ornaments, for their color and beauty, including the species: Zachrysia bayamensis and Coryda lindoni. In addition, also lepidopterans perform various functions such as forming part of food chains and participating in the pollination of many plants. In the ecosystems of the Sierra Maestra can be see Greta cubana, Virbia heros (restricted to the Nipe-Sagua-Baracoa and Sierra Maestra mountain ranges), Calisto isnaeli. In Sierra Mastra there is also a great diversity of amphibians and reptiles, in particular the species Eleutherodactylus albipes, Eleutherodactylus cubanus, Eleutherodactylus jaumei are endemic of the Sierra Maestra.
Discussion
Coffee culture, as part of mixed systems, tends to be more important in the south of Sierra del Rosario range system while livestock, mainly cows and pigs, tend to be predominant in the more rural farms at the northern side of the range system. The production of coffee seems to be not very high in traditional farms in terms of gross production or productivity. This is in concordance with the arguments of Ospina (2008) pointing that what is remarkable in these ecological coffee systems is the kaleidoscope of food products and ecosystems service. However, in the case of the traditional systems of Sierra del Rosario, there is a surplus of coffee production that is placed in the national markets by different ways, but mainly because of State´s purchases. In the traditional farms placed in Sierra del Rosario, agricultural typologies vary according to differences in topography. For example, is very notable the nonuse of the slopes for practicing the agriculture when the availability of cropping areas in valleys is enough for food requirements. This conducts to differences in the cultures of managing these areas diversifying the traditional practices.
As has been pointed out, there is a diversity of sources of incomes for the traditional farming systems, condition that can be considered a guarantee of resilience. It is hypothesized that shade promotes slower and more balanced filling and uniform ripening of berries, thus yielding a better-quality product than unshaded coffee plants (Muschler 2001). In addition, the multiplicity of agricultural activities and the traditional knowledge is an opportunity for keeping the functioning in the system itself when a stressing situation occurs. The multifunctionality of many biological resources is also a distinctive attribute of the traditional systems still present in Sierra del Rosario. A very iconic example is the case of Cuban national tree (Roystonea regia), a forest resource with multiple uses and cultural signification. The species is a source of traditional materials for buildings, food for humans and animals, and also row materials for packaging. In this sense the petioles of the royal palm are very demanded in the elaboration of boxes for transportation of tobacco leaves.
Even when coffee and fruits trees are very important components of the traditional systems of Sierra del Rosario, other crops species like annual crops are important as complement of the systems. As Brown and Hodgkin pointed (2007), there are three categories of plant species make up plant biodiversity in the rural landscape:
-
1.
The plant species that are deliberately cropped or tended and harvested for food, fiber, fuel, fodder, timber, medicine, decoration, or other uses.
-
2.
At the other extreme, wild species that occur in natural communities and benefit the agricultural environment by providing protection, shade, and groundwater regulation
-
3.
Between these extremes, the wild related species of domesticates that can interbreed with and contribute to the genepool of their crop cousins, that survive autonomously, that share many of the pests and diseases of crops, and that sometimes are eaten to relieve famine
Regarding the Sierra Maestra, it represents one of the main nucleus of biodiversity in Cuba. The system traditional structure, today maintained, results to be a tool for the conservation and management of biodiversity, inside or outside protected areas or where the habitat is very disturbed. The use of forest trees as a shadow of coffee and cocoa cultivation has proven to be sustainable over three centuries creating also a favorable scenario to increase biodiversity. Forest trees are not the only one used to create shade in coffee cultivation, it is, in fact, easy and common to find fruit trees that apart from the function as shade and crop protection, also provide additional food products so that coffee and cocoa systems in Sierra Maestra not only generate coffee and cocoa as a product, but also high quality wood, fruits and agricultural products. Moreover, diversified coffee and cocoa plantations that look like a natural forest are ideal for protecting the soil, conserving water and maintaining high biodiversity constituting excellent wildlife habitat sites.
As seen from the result of the present study traditional agroforestry systems, as the ones in Sierra del Rosario and Sierra Maestra, have the potential to hold high species richness and constitute a valuable tool that could be used to complement conservation effort while being designed to improve farmers' livelihoods by generally increasing productivity, profitability and sustainability.
According to the World Bank (2008), improving these three aspects of small-scale agriculture is a key way out of poverty, emphasizing the potential of agroforestry practices to alleviate shortages. In addition, it has been exposed that agroforestry systems with greater structural complexity are capable of harboring high biodiversity and in particular the functional biodiversity, which can increase productivity and ecological resilience. For example, cross-pollination can increase coffee production by up to 50% compared to self-pollination (Tscharntke et al. 2011) and biological control can reduce outbreaks of plagues or herbivores (Kellerman et al. 2008; Perfecto et al. 2004). Interactions between woody and herbaceous plants in agroforestry systems usually improve the microclimate and nutrient availability in the soil. The belowground presence of trees affects moisture availability and soil temperature and these, in turn, affect transpiration and energy conversion of nearby plants (Rosenberg et al. 1983; Atangana et al. 2014). Biologically complex agroforestry systems often reveal greater ecosystem functioning and a reduced reliance on chemical inputs (Drinkwater and Snapp 2007; Malézieux et al. 2009; Martin and Isaac 2015). Such benefits have been observed from farm to landscape levels of integration, and across temperate and tropical agroecosystems. While these benefits are a key target in future food production landscapes, such success requires well developed diagnostics of the plant-soil continuum (Isaac and Borden 2019).
Conclusions
Agroforestry systems are recognized as an alternative for land use planning on farms and interfluvial areas being considered as a sustainable example for the management of tree, shrub, soil, crops and animals’ resources. The system must integrate the function and interaction between its components, otherwise it may be a good association, but hardly adequate to protect the soil-crop-tree and produce sustainably. Many of the alternatives improve soil conditions, others influence water production or relate to crop protection (World Vision 2005). The high diversity in terms of cultivated and spontaneous species characterizing the systems object of the present paper allows a strong resilience of the systems themselves, tested after the impact of devastating hurricanes, changes in rainfall patterns and droughts. In addition, at the genetic, species, and farming systems levels, biodiversity provides valuable ecosystems services and functions for agricultural production (Thrupp 2000).
“Agroforestry” is a relatively new term, the traditional knowledge and practices underpinning these land use systems is ancient, having originated in the ancestral "shifting cultivation" practices of African peoples and indigenous peoples of the Americas (and elsewhere). Landscape structure, field area and margins, and polycultures that are part of the indigenous agricultural strategy appear to increase the biodiversity of traditional agroecosystems (Altieri et al. 1987; Oldfield and Alcorn 1987; Parrotta et al. 2015). Thus, there is increasing evidence that the mosaic structure of landscapes under indigenous management maintains and even improves biodiversity (Gonzalez-Bernaldez 1991; Brown and Brown 1992; Reichhardt et al. 1994), as well as preserving the associated cultural values (Agnoletti 2014; Agnoletti et al. 2015).
Furthermore, the complex system created thanks to the introduction of shade trees and other cultivated species contribute to obtain multiple benefits from both an ecological and economic point of view. In fact, shade trees play an important role in erosion control and in maintaining soil productivity by stimulating the decomposition of residuals while generating additional products, such as wood, firewood and fruits, providing important contributions to farmers' livelihoods, especially in seasons where productivity it is low. It is increasingly clear that shade trees provide direct and indirect benefits, so it is difficult to fully quantify the total benefits. However, these benefits are expected to improve farmers' livelihoods by stabilizing their income and increasing their ability to recover in general. Considering this and the fact that being system with no chemical inputs and completely sustainable they can mitigate the effects of climate change by propitiating a favorable microclimate and increasing carbon storage it is clear that results important their preservation and protection in order to provide multiple benefits to the environment sustaining local communities. The present study, in this sense, has contributed to give a framework of the traditional structure of agroforestry systems for coffee production in Cuba providing at the same time the list of all the flora and fauna species involved in the systems themselves. This turns to be fundamental to understand the importance of maintaining and promoting traditional knowledge which, in this particular case, is the key to preserve the agrobiodiversity, sustain local communities and implement sustainable economic strategies. These systems are also an important alternative to intensive plantations of coffee, especially when we relate this production to the worldwide market of coffee consumed in many countries, for their sustainability and as an expression of a wider concept of food quality.
Data availability
Not applicable.
Code availability
Not applicable.
References
Agnoletti M (2014) Rural landscape, nature conservation and culture: Some notes on research trends and management approaches from a (southern) European perspective. Landsc Urban Plan 126:66–73
Agnoletti M, Rotherham ID (2015) Landscape and biocultural diversity. Biodivers Conserv 24:3155–3165. https://doi.org/10.1007/s10531-015-1003-8
Agnoletti M, Tredici M, Santoro A (2015) Biocultural diversity and landscape patterns in three historical rural areas of Morocco, Cuba and Italy. Biodivers Conserv 24:3387–3404. https://doi.org/10.1007/s10531-015-1013-6
Altieri M, Merrick L, Anderson MK (1987) Peasant agriculture and the conservation of crop and wild plant resources. Conserv Biol 1(1):49–58
Atangana A, Khasa D, Chang S, Degrande A (2014) Ecological interactions and productivity in agroforestry systems. In: Atangana A (ed) Tropical agroforestry. Springer, Dordrecht
Beer J, Muschler R, Kass D, Somarriba E (1997) Shade management in coffee and cacao plantations. Agrofor Syst 38(1):139–164
Brown KS, Brown GG (1992) Habitat alteration and species loss in Brazilian forests. In: Whitmore TC, Sayer JA (eds) Tropical deforestation and species extinction. Chapman & Hall, New York
Brown AH, Hodgkin T (2007) Measuring, Managing and Maintaining crop genetic diversity on farm. In: Jarvis DI, Padoch C, Cooper HD (eds) Managing biodiversity in agricultural ecosystems. Columbia University Press, New Yok, pp 13–33
Castiñeiras L, Barrios O, Fernández L, León N, Cristóbal R, Shagarodsky T, Fuentes V, Fundora Z, Moreno V (2006) Catálogo de cultivares tradicionales y nombres locales en fincas de las regiones occidental y oriental de Cuba. Agrinfor. La Habana, Cuba, 64 pp.
Drinkwater LE, Snapp SS (2007) Nutrients in agroecosystems: rethinking the management paradigm. Adv Agron 92:163–186
Fernández AH (2012) Sistema de asentamiento de las haciendas cafetaleras en la Sierra dl Rosario (1790 1850), Artemisa. Cuba Cuba Arqueológica Año V 1:12–19
Food and Agriculture Organization (2015) FAO statistical pocketbook coffee 2015. Food and Agriculture Organization, Rome
García M, Castiñeiras L (2006) La diversidad agrícola en reservas de la biosfera de Cuba. Editorial Academia, La Habana, pp 10–20
Gonzalez-Bernaldez F (1991) Diversidad biológica, gestión de ecosistemas y nuevas políticas agrarias. In: Pineda FF, Casado MA, de Miguel JM, Montalvo J (eds) Diversidad biológica. Fundación R Areces, Madrid
Gonzalez Alvarez A, Rodríguez YS, Delgado DA, Zamora JL (2016) Agrobiodiversidad en La Sierra Del Rosario, Cuba: El Café (Coffea Arabica L.) y otras claves de su configuración, Agrotecnia de Cuba 40(2):3–8
Kellermann JL, Johnson MD, Stercho AM, Hackett SC (2008) Ecological and economic services provided by birds on Jamaican Blue Mountain coffee farms. Conserv Biol 22(5):1177–1185
Kottek M, Grieser J, Beck C, Rudolf B, Rubel F (2006) Rubel F (2006) World Map of the Köppen-Geiger climate classification updated. Meteorol Z 15(3):259–263
Krishnan S (2017) Sustainable coffee production. Oxford research encyclopedia of environmental science. Oxford University Press, Oxford. https://doi.org/10.1093/acrefore/9780199389414.013.224
Isaac ME, Borden KA (2019) Nutrient acquisition strategies in agroforestry systems. Plant Soil 444:1–19. https://doi.org/10.1007/s11104-019-04232-5
Lapique Z, García L (2014) La Periferia Habanera. Rev Bimestre Cubana 41(2):89–102
Long CL, Li H, Ouyang Z, Yang X, Li Q, Trangmar B (2003) Strategies for agrobiodiversity conservation and promotion: a case from Yunnan, China. Biodivers Conserv 12:1145–1156. https://doi.org/10.1023/A:1023085922265
Malézieux E, Crozat Y, Dupraz C, Laurans M, Makowski D, Ozier-Lafontaine H, Rapidel B, Tourdonnet S, Valantin-Morison M (2009) Mixing plant species in cropping systems: concepts, tools and models: a review. Agron Sustain Dev 29:43–62
Martin AR, Isaac ME (2015) Functional traits in agroecology: a blueprint for research. J Appl Ecol 52:1425–1435
Muschler RG (2001) Shade improves coffee quality in a sub-optimal coffee-zone of Costa Rica. Agrofor Syst 51:131–139. https://doi.org/10.1023/A:1010603320653
Moguel P, Toledo VM (1999) Biodiversity conservation in traditional coffee systems of Mexico. Conserv Biol 13(1):11–21
Oldfield M, Alcorn JB (1987) Conservation of traditional agroecosystems. Bioscience 37(3):199–208
Ospina A (2008) Aproximación a la conservación de especies vegetales nativas con bosque ecológico con sombrío. Cali, Colombia. Consulted at www. agroforesteriaecologica.com. Accessed 26 Aug, 2019
Parrotta JA, Dey de Pryck J, Darko Obiri B, Padoch C, Powell B, Sandbrook C (2015) The Historical, Environmental and Socio-Economic Context of Forests and Tree-Based Systems for Food Security and Nutrition. Chapter 3 (pp. 71–134) In: Vira B, Mansourian S, Wildburger C (eds) forests and food: addressing hunger and nutrition across sustainable landscapes. Open Book Publishers, Cambridge, UK. http://dx.doi.org/https://doi.org/10.11647/OBP.0085
Chirwa PW, Akinnifesi FK, Sileshi G, Syampungani S, Kalaba FK, Ajayi OC (2008) Opportunity for conserving and utilizing agrobiodiversity through agroforestry in Southern Africa. Biodiversity 9(1–2):45–48. https://doi.org/10.1080/14888386.2008.9712881
Petit-Aldana J, Rahman MM, Parraguirre-Lezama C, Infante-Cruz A, Romero-Arenas O (2019) Litter decomposition process in coffee agroforestry systems. J For Environ Sci 35(2):121–139. https://doi.org/10.7747/JFES.2019.35.2.121
Perfecto I, Vandermeer J, Bautista G, Nunez G, Greenberg R, Bichier P, Langridge S (2004) Greater predation in shaded coffee farms: The role of resident neotropical birds. Ecology 85:2677–2681
Thrupp LA (2000) Linking agricultural biodiversity and food security: the valuable role of agrobiodiversity for sustainable agriculture. Int Aff 6(2):265–281. https://doi.org/10.1111/1468-2346.00133
Trosper L, Parrotta AJ, Agnoletti M et al (2011) The unique charachter of traditional forest-related knowldege: threats and challanges ahead. In: Parrotta JA, Trosper RL (eds) Traditional forest-related knowledge. Springer, Dordrecht
Ramírez Pérez JF, Paredes Pupo FA (2005) Cuba, paisaje a la naturaleza. Las Terrazas. Pinar del Río, 29 pp.
Rao KPC, Verchot LV, Laarman J (2007) Adaptation to climate change through sustainable management and development of agroforestry systems. J SAT Agric Res 4(1):1–30
Reichhardt L, Mellink E, Nahan GP, Rea A (1994) Habitat heterogeneity and biodiversity associated with indigenous agriculture in the Sonoran desert. Etnoecológica 3:21–36
Rosenberg NJ, Blad BL, Verma DB (1983) Microclimate: the biological environment. Wiley and Sons, New York
Sánchez Y, González A, Kauffman M, Zamora JL, Pérez HM (2015) Family farming and its landscape imbrication. An assessment in Sierra del Rosario and Cuchillas del Toa biosphere reserve. Agrotec De Cuba 5:73–85
Santoro A, Venturi M, Bertani R, Agnoletti M (2020) A review of the role of forests and agroforestry systems in the FAO Globally Important Agricultural Heritage Systems (GIAHS) programme. Forests 11:860. https://doi.org/10.3390/f11080860
Siebert SF (2002) From shade to sun-grown perennial crops in Sulawesi, Indonesia: implications for biodiversity conservation and soil fertility. Biodivers Conserv 11:1889–1902
Tscharntke T, Clough Y, Bhagwat SA, Buchori D, Faust H, Hertel D, Wanger TC (2011) Multifunctional ****shade-tree management in tropical agroforestry landscapes—a review. J Appl Ecol 48(3):619–629
Vales MA, Vilamajo D (2001) Biodiversidad biológica cubana, conservación y uso sostenile. Ciencia Innov y Desarro 6(1):39–44
World Vision International (2005). Annual report.
World Bank (2008) The World Bank annual report 2008: year in review. World Bank, Washington DC, USA. https://openknowledge.worldbank.org/handle/10986/7524.
Funding
This research is part of the “GIAHS Building Capacity” project, funded by the Italian Agency for Development Cooperation (AICS) and by the Department of Agriculture, Food, Environment and Forestry (DAGRI) of the University of Florence.
Author information
Authors and Affiliations
Contributions
MA: conceptualization; YMP, AGA: methodology; YMP, AGA: investigation; MA: writing; MA: supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Antonio Santoro.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Appendix 1: Complete list of the useful species at Sierra Del Rosario, with the botanical family of each species
Appendix 1: Complete list of the useful species at Sierra Del Rosario, with the botanical family of each species
List of species used for animal feeding.
Scientific name | Family | |
|---|---|---|
1 | Commelina diffusa Burm. f | Commelinaceae |
2 | Cynodon dactylon (L.) Pers | Poaceae |
3 | Dendropanax arboreus (L.) Decne. & Planch | Araliaceae |
4 | Echinochloa colona (L.) Link | Poaceae |
5 | Gerascanthus coloccocus (L.) Borhidi | Boraginaceae |
6 | Guazuma ulmifolia Lam | Malvaceae |
7 | Helianthus annuus L. | Asteraceae |
8 | Hypharrenia rufa Nees | Poaceae |
9 | Pennisetum purpureum Schumacher | Poaceae |
10 | Roystonea regia (Kunth) O.F. Cook | Arecace |
11 | Saccharum officinarum L. | Poaceae |
12 | Samanea saman (Jacq.) Merr | Leguminosae |
13 | Sorghum bicolor (L.) Moench | Poaceae |
14 | Sporobolus indicus (L.) R.Br | Poaceae |
15 | Trophis racemosa (L.) Urb | Moraceae |
List of species used for beverage elaboration.
Scientific name | Family | |
|---|---|---|
1 | Canavalia ensiformis L. | Leguminosae |
2 | Cassia grandis L. f | Leguminosae |
3 | Citrus x aurantifolia (Christm.) Swingle | Rutaceae |
4 | Coffea arabica L. | Rubiaceae |
5 | Coffea canephora L. | Rubiaceae |
6 | Passiflora edulis Sims | Passifloraceae |
7 | Smilax dominguensis Willd | Smilacaceae |
8 | Smilax havanensis Jacq | Smilacaceae |
9 | Smilax lanceolata L. | Smilacaceae |
10 | Smilax mollis Willd | Smilacaceae |
11 | Vitis tiliifolia Humb. & Bonpl. Ex Schult | Vitaceae |
12 | Vitis vinifera L. | Vitaceae |
List of the species used as condiments.
Scientific name | Family | |
|---|---|---|
1 | Allium cepa L. | Amaryllidaceae |
2 | Allium cepa var. aggregatum G. Don | Amaryllidaceae |
3 | Allium chinense G. Don | Amaryllidaceae |
4 | Allium fistulosum L. | Amaryllidaceae |
5 | Allium sativum L. | Amaryllidaceae |
6 | Allium tuberosum Rottler ex Spreng | Amaryllidaceae |
7 | Averrhoa bilimbi L. | Oxalidaceae |
8 | Bixa orellana L. | Bixaceae |
9 | Brassica juncea (L.) Coss | Brassicaceae |
10 | Capsicum annuum L. | Solanaceae |
11 | Capsicum chinense Jacq | Solanaceae |
12 | Capsicum frutescens L. | Solanaceae |
13 | Cinnamomum cassia (Nees & T. Nees) J. Presl | Lauraceae |
14 | Citrus x aurantium L. | Rutaceae |
15 | Citrus limon (L.) Burm | Rutaceae |
16 | Eryngium foetidum L. | Apiaceae |
17 | Foeniculum vulgare Mill | Apiaceae |
18 | Lippia micromera Schauer | Verbenaceae |
19 | Ocimum gratissimum L. | Lamiaceae |
20 | Plectranthus amboinicus (Lour.) Spreng | Lamiaceae |
21 | Sinapis alba L. | Brassicaceae |
List of the species used as fruits.
Scientific name | Family | |
|---|---|---|
1 | Anacardium occidentale L. | Anacardiaceae |
2 | Ananas comosus (L.) Merr | Bromeliaceae |
3 | Annona muricata L. | Annonaceae |
4 | Annona squamosa L. | Annonaceae |
5 | Carica papaya L. | Caricaceae |
6 | Chrysophyllum cainito L. | Sapotaceae |
7 | Chrysophyllum oliviforme L. | Sapotaceae |
8 | Citrullus lunatus (Thunb.) Matsum. Y Nakai | Cucurbitaceae |
9 | Citrofortunella microcarpa (Bunge) Wijnands | Rutaceae |
10 | Citrus reticulata Blanco | Rutaceae |
11 | Citrus x sinensis Osbeck | Rutaceae |
12 | Citrus x paradisi Macfad | Rutaceae |
13 | Cocos nucifera L. | Arecaceae |
14 | Cucumis melo L. | Cucurbitaceae |
15 | Dovyalis hebecarpa (Gardner) Warb | Salicaceae |
16 | Flacourtia indica (Burm. F.) Merr | Salicaceae |
17 | Malpighia emarginata DC | Malpighiaceae |
18 | Malus domestica L. | Rosaceae |
19 | Mammea americana L. | Callophyllaceae |
20 | Mangifera indica L. | Anacardiaceae |
21 | Manilkara zapota (L.) P. Royen | Sapotaceae |
22 | Melicoccus bijugatus Jacq | Sapindaceae |
23 | Muntingia calabura L. | Muntingiaceae |
24 | Musa x paradisiaca L. | Musaceae |
25 | Persea americana Mill | Lauraceae |
26 | Pouteria campechiana Baehni | Sapotacae |
27 | Pouteria sapota (Jacq.) H.E. Moore & Stearn | Sapotaceae |
28 | Prunus persica (L.) Stokes | Rosaceae |
29 | Psidium guajava L. | Myrtaceae |
30 | Punica granatum L. | Lythraceae |
31 | Spondias purpurea L. | Anacardiaceae |
32 | Syzygium malaccense (L.) Merr. & L.M. Perry | Myrtaceae |
List of the species used as grains.
Scientific name | Family | |
|---|---|---|
1 | Arachys hypogaea L. | Leguminosae |
2 | Oryza sativa L. | Poaceae |
3 | Phaseolus lunatus L. | Leguminosae |
4 | Phaseolus vulgaris L. | Leguminosae |
5 | Sesamum indicum L. | Pedaliaceae |
6 | Vigna umbellata (Thunb.) Ohwi & H. Ohashi | Leguminosae |
7 | Vigna unguiculata L. Walp | Leguminosae |
8 | Zea mays L. | Poaceae |
List of wood and timber species.
1 | Scientific name | Family |
|---|---|---|
2 | Abarema obovalis (A. Rich.) Barneby & J.W. Grimes | Leguminosae |
3 | Acrocomia aculeata (Jacq.) Lodd. ex Mart | Arecaceae |
4 | Alchornea latifolia Sw | Euphorbiaceae |
5 | Amphitecna latifolia (Mill.) A. H. Gentry | Bignoniaceae |
6 | Amyris balsamifera L. | Rutaceae |
7 | Andira inermis (W. Wright) DC | Leguminosae |
8 | Boconia frutescens L. | Papaveraceae |
9 | Ehretia cassinifolia A. Rich | Boraginaceae |
10 | Buchenavia capitata (Aubl.) Howard | Combrataceae |
11 | Calophyllum antillanum Britton | Calophyllaceae |
12 | Calyptrantes capitulata C. Wright | Myrtaceae |
13 | Casearia hirsuta Sw | Salicaceae |
14 | Cedrela odorata L. | Meliaceae |
15 | Celtis trinervia Lam | Ulmaceae |
16 | Citharexylum caudatum L. | Verbanaceae |
17 | Coccoloba retusa Griseb | Polygonaceae |
18 | Cojoba arborea (L.) Britton & Rose | Leguminosae |
19 | Colubrina arborescens (Mill.) Sarg | Rhamnaceae |
20 | Comocladia dentata Jacq | Anacardiaceae |
21 | Cordia nitida Vahl | Boraginaceae |
22 | Cupania americana L. | Sapindaceae |
23 | Cupania glabra Sw | Sapindaceae |
24 | Cupania macrophylla C. Mart | Sapindaceae |
25 | Deherainia cubensis (Radlk.) Mez | Primulaceae |
26 | Dendropanax arboreus (L) Decne. & Planch | Araliaceae |
27 | Dichrostachys cinerea (L.) Wight & Arn | Leguminosae |
28 | Diospyros caribaea (A. DC.) Standl | Ebenaceae |
29 | Diospyros crassinervis (Krug &Urb.) Standl | Ebenaceae |
30 | Drypetes alba Poit | Euphorbiaceae |
31 | Drypetes serrata (Maycock) Krug & Urb | Euphorbiaceae |
31 | Ehretia cassinifolia A. Rich | Boraginaceae |
33 | Ehretia tinifolia L. | Boraginaceae |
34 | Erythroxylon alternifolium M. Gomez | Erythroxylaceae |
35 | Erythroxylon areolatum L. | Erythroxylaceae |
36 | Eucaliptus resinifera Sm | Myrtaceae |
37 | Eugenia glabra Alston | Myrtaceae |
38 | Eugenia maleolens Pers | Myrtaceae |
39 | Eugenia rigidifolia A. Rich | Myrtaceae |
40 | Eugenia rimosa C. Wright | Myrtaceae |
41 | Ficus aurea Nutt | Moraceae |
42 | Ficus cassinervia Desf. ex Willd | Moraceae |
43 | Ficus citrifolia Mill | Moraceae |
44 | Ficus combissi Warb | Moraceae |
45 | Ficus maxima Mill | Moraceae |
46 | Ficus membranacea C. Wright | Moraceae |
47 | Garcinia aristata (Griseb.) Borhidi | Clusiaceae |
48 | Garcinia serpentini Borhidi | Clusiaceae |
49 | Garcinia ruscifolia (Griseb.) Borhidi | Clusiaceae |
50 | Genipa americana L. | Rubiaceae |
51 | Gerascanthus gerascanthioides | Boraginaceae |
52 | Guarea guidonia (L.) Sleumer | Meliaceae |
53 | Guettarda combsii Urb | Rubiaceae |
54 | Guettarda lindeniana A. Rich | Rubiaceae |
55 | Guettarda valenzuelana Rich | Rubiaceae |
56 | Gymnanthes lucida Sw | Euphorbiaceae |
57 | Hibiscus cordifolius Mill | Malvaceae |
58 | Hibiscus elatus Sw | Malvaceae |
59 | Hibiscus tiliaceus L. var. pernambucensis (Arruda) I.M. Johnst | Malvaceae |
60 | Hura crepitans L. | Euphorbiaceae |
61 | Ilex cassine L. | Aquifoliaceae |
62 | Ilex repanda Griseb | Aquifoliaceae |
63 | Juglans jamaicensis D. DC | Fagaceae |
64 | Khaya senegalensis (Desr.) A. Juss | Meliaceae |
65 | Krugiodendron ferreum (Vahl) Urb | Rhamnaceae |
66 | Laplacea curtyana A. Rich | Theaceae |
67 | Licaria triandra (Sw.) Kosterm | Lauraceae |
68 | Lonchocarpus pentaphyllus (Poir.) D.C | Leguminosae |
69 | Lonchocarpus sericeus (Poir.) Kunth ex DC | Leguminosae |
70 | Lysiloma sabicu Benth | Leguminosae |
71 | Malpighia urens L. | Malpighiaceae |
72 | Manilkara albescens (Griseb.) Cronquist | Sapotaceae |
73 | Manilkara jaimiqui (C. Wright ex Griseb.) Dubard | Sapotaceae |
74 | Mappia racemosa Jacq | Icacinaceae |
75 | Margaritaria nobilis L.f | Phyllantaceae |
76 | Matayba oppositifolia (A.Rich.) Britton | Sapindaceae |
77 | Melia azedarach L | Meliaceae |
78 | Micropholis guyanensis (A. DC.) Pierre | Sapotaceae |
79 | Myrcia valenzuelana (A. Rich.) Griseb | Rubiaceae |
80 | Myrica cerifera L. | Myricaceae |
81 | Nectandra coriacea (Sw.) Griseb | Lauraceae |
82 | Nectandra hihua (Ruiz & Pav.) Rohwe | Lauraceae |
83 | Nectandra minima Rohwer | Lauraceae |
84 | Neobracea valenzuelana (A. Rich.) Urb | Apocynaceae |
85 | Ocotea cuneata (Griseb.) Urb | Lauraceae |
86 | Ocotea floribunda (Sw.) Mez | Lauraceae |
87 | Ocotea leucoxylon (Sw.) Laness | Lauraceae |
88 | Peltophorum adnatum Griseb | Leguminosae |
89 | Phoebe elongata (Vahl) Nees | Lauraceae |
90 | Picramnia pentandra Sw | Leguminosae |
91 | Picramnia reticulata Griseb | Leguminosae |
92 | Piscidia piscipula (L.) Sarg | Leguminosae |
93 | Samanea saman (Jacq.) Merr | Leguminosae |
94 | Pouteria dictyoneura (Griseb.) Radlk | Sapotaceae |
95 | Pouteria dominigensis (C.F. Gaertn.) Baehni | Sapotaceae |
96 | Protium cubense (Rose) Urb | Burseraceae |
97 | Prunus myrtifolia (L.) Urb | Rosaceae |
98 | Prunus occidentalis Sw | Rosaceae |
99 | Pseudolmedia spuria (Sw.) Griseb | Moraceae |
100 | Psychotria horizontalis Sw | Rubiaceae |
101 | Psychotria undata Jacq | Rubiaceae |
102 | Rauvolfia cubana A. DC | Apocynaceae |
103 | Rauvolfia nitida Jacq | Apocynaceae |
104 | Reynosia wrightii Urb | Rhamnaceae |
105 | Richardia brasiliensis Gomes | Rubiaceae |
106 | Sabal parviflora Becc | Arecaceae |
107 | Sapindus saponaria L. | Sapindaceae |
108 | Savia bahamensis Britt | Euphorbaceae |
109 | Savia clusiifolia Griseb | Euphorbaceae |
110 | Schefflera morototoni (Aubl.) Maguire, Steyerm. & Frodin | Araliaceae |
111 | Sideroxylon foetidissimum Jacq | Sapotaceae |
112 | Simarouba glauca DC | Simaroubaceae |
113 | Suberanthus neriifolius (A. Rich.) Borhidi & M. Fernández | Rhamnaceae |
114 | Swietenia macrophylla King | Meliaceae |
115 | Swietenia mahagoni (L.) Jacq | Meliaceae |
116 | Symplocos martinicensis (Krug & Urb.) Mai | Symplocaceae |
117 | Tabebuia shaferi Britton | Bignoniaceae |
118 | Tabernaemontana citrifolia L. | Apocynaceae |
119 | Tapura obovata Britton & Wilson | Dichapetalaceae |
120 | Tectona grandis L. | Verbenaceae |
121 | Terminalia catappa L. | Combretaceae |
122 | Ternstroemia peduncularis DC | Penthapylaceae |
123 | Trema micrantha (Roem. & Scult.) Blume | Ulmaceae |
124 | Trichilia havanensis Jacq | Meliaceae |
125 | Trichilia hirta L. | Meliaceae |
126 | Trichospermum grewiifolius (A.Rich.) Kosterm | Malvaceae |
127 | Zanthoxylon cubense P. Wilson | Rutaceae |
128 | Zanthoxylon elephantiasis Macfad | Rutaceae |
129 | Zanthoxylon fagara (L.) Sarg | Rutaceae |
130 | Zanthoxylon martinicense (Lam.) DC | Rutaceae |
131 | Zuelania guidonia (Sw.) Britton & Millsp. | Salicaceae |
List of medicinal species.
Scientific Name | Family | |
|---|---|---|
1 | Abrus precatorius L. | Leguminosae |
2 | Adenoropium multifidum (L.) Poh | Euphorbiaceae |
3 | Adianthus capillus veneris L. | Adianthaceae |
4 | Agalinis albida Britton & Pennell | Orobanchaceae |
5 | Ageratum conyzoides L. | Asteraceae |
6 | Allophyllus cominia (L.) Sw | Sapindaceae |
7 | Aloe vera (L.) Burm.f | Xanthorrhoeaceae |
8 | Alternanthera caracasana Kunth | Amaranthaceae |
9 | Ambrosia artemisiifolia L. | Asteraceae |
10 | Andropogon glomeratus (Walter) Britton | Poaceae |
11 | Argemone mexicana L. | Papaveraceae |
12 | Artemisa absinthium L. | Asteraceae |
13 | Arthrostylidium capillifolium Griseb | Poaceae |
14 | Asclepias curassavica L. | Asclepiadaceae |
15 | Asclepias nívea L. | Asclepiadaceae |
16 | Bahuinia cumanensis Kunth | Caesalpinaceae |
17 | Bahuinia divaricata L. | Caesalpinaceae |
18 | Banksea speciosa J. König | Proteaceae |
19 | Bidens pilosa L. | Asteraceae |
20 | Blechum pyramidatum (Lam.) Urb | Acanthaceae |
21 | Byrsonima spicata (Cav.) DC | Malpighiaceae |
22 | Caesalpinia vesicaria L. | Leguminosae |
23 | Caesalpinia pulcherrima (L.) Sw | Leguminosae |
24 | Cassia diphylla (L.) Greene | Leguminosae |
25 | Catharanthus roseus (L.) G. Don | Apocynaceae |
26 | Cayaponia racemosa (Mill.) Cogn | Cucurbitaceae |
27 | Cecropia schreberiana Miq | Urticaceae |
28 | Celtis iguanea (Jacq.) Sarg | Ulmaceae |
29 | Chromolaena odorata (L.) R.M. King & H. Rob | Asteraceae |
30 | Cissus verticillata (L.) Nicolson & C.E.Jarvis | Vitaceae |
31 | Citrus x bergamia Risso & Poit | Rutaceae |
32 | Cladium jamaicensis Crantz | Cyperaceae |
33 | Clematis dioica L. | Ranunculaceae |
34 | Clidemia hirta (L.) D.Don | Melastomataceae |
35 | Clusia minor L | Clusiaceae |
36 | Coix lacryma-jobi L. | Poaceae |
37 | Costus sp. | Costaceae |
38 | Costus scaber Ruiz & Pav | Costaceae |
39 | Crescencia cujete L. | Bignoniaceae |
40 | Cymbopogon citratus (DC) Stapf | Poaceae |
41 | Dalbergia ecastaphyllum (L.) Taubert | Leguminosae |
42 | Davilla rugosa Poir | Dilleniaceae |
43 | Echites umbellatus Jacq | Apocynaceae |
44 | Elephantopus scaber L. | Asteraceae |
45 | Erigeron quercifolius Lam | Asteraceae |
46 | Faramea occidentalis (L.) A.Rich | Rubiaceae |
47 | Gaya occidentalis (L.) Sweet | Malvaceae |
48 | Gossypium sp | Malvaceae |
49 | Guzmania monostachya Rusby ex Mez | Bromeliaceae |
50 | Hamelia patens Jacq | Rubiaceae |
51 | Hedychium coronarium J. Koening | Zyngiberceae |
52 | Heliotropium indicum L. | Asteraceae |
53 | Hibiscus sabdariffa L. | Malvaceae |
54 | Hippobroma longiflora (L.) Don | Campanulaceae |
55 | Hohenbergia penduliflora (A. Rich.) Mez | Bromeliaceae |
56 | Imperata brasiliensis Trin | Poaceae |
57 | Ixora floribunda (A. Rich.) Griseb | Rubiaceae |
58 | Jacquinia brunnescens Urb | Primulaceae |
59 | Justicia pectoralis Jacq | Acanthaceae |
60 | Koanophyllon dolicholepis (Urb.) R.M. King & H. Rob | Asteraceae |
61 | Lantana camara L. | Verbenaceae |
62 | Lantana involucrata L. | Verbenaceae |
63 | Lepidium virginicum L. | Brassicaceae |
64 | Lippia alba (Mill.) N.E. Br. Ex Britton & Wilson | Verbenaceae |
65 | Matricaria recutita L. | Asteraceae |
66 | Mentha sp. | Lamiaceae |
67 | Mentha spicata L. | Lamiaceae |
68 | Mentha x piperita L. | Lamiaceae |
69 | Miconia laeviagata (L.) D.Don | Melastomataceae |
70 | Mikania cordifolia (L. f.) Willd | Asteraceae |
71 | Mikania hastata (L.) Willd | Asteraceae |
72 | Mikania ranunculifolia A. Rich | Asteraceae |
73 | Mimosa pudica L. | Leguminosae |
74 | Momordica charantia L. | Cucurbitaceae |
75 | Morinda royoc L. | Rubiaceae |
76 | Mucuna pruriens (L.) DC | Leguminoseae |
77 | Neurolaena lobata (L.) R.Br. ex Cass | Asteraceae |
78 | Ocimum basilicum L. | Lamiaceae |
79 | Ocimum sanctum L. | Lamiaceae |
80 | Olyra latifolia L. | Poaceae |
81 | Origanum majorana L. | Lamiaceae |
82 | Oxandra lanceolata (Sw.) Baill | Lauraceae |
83 | Parthenium hysterophorus L. | Asteraceae |
84 | Paspalum conjugatum | Poaceae |
85 | Paspalum notatum Fluggé | Poaceae |
86 | Passiflora sexflora Juss | Passifloraceae |
87 | Passiflora suberosa L. | Passifloraceae |
88 | Pavonia fruticosa (Mill.) Fawc. & Rendle | Malvaceae |
89 | Petiveria alliacea L. | Phytolacaceae |
90 | Phylla scaberrima (Trevir.) Moldenke | Verbenaceae |
91 | Piper aduncum L. | Piperaceae |
92 | Piper auritum Kunth | Piperaceae |
93 | Piper peltatum L. | Piperaceae |
94 | Piper umbellatum L. | Piperaceae |
95 | Pisonia aculeata L. | Nyctaginaceae |
96 | Plantago major L. | Plantaginaceae |
97 | Platygine hexandra (Jacq) | Euphorbiaceae |
98 | Plectranthus sp | Lamiaceae |
99 | Pluchea carolinensis (Jacq.) G. Don | Asteraceae |
100 | Pleopeltis polypodioides (L.) E. G. Andrews & Windham | Polypodiaceae |
101 | Psychotria grandis Sw | Rubiaceae |
102 | Psychotria revoluta DC | Rubiaceae |
103 | Renealmia aromatica L.f | Zingiberaceae |
104 | Rondeletia odorata Jacq | Rubiaceae |
105 | Rosmarinus officinalis L. | Lamiaceae |
106 | Ruta graveolens L. | Rutaceae |
107 | Securidaca virgata Sw | Polygalaceae |
108 | Senna alata (L.) Roxb | Leguminosae |
109 | Senna occidentalis (L.) Link | Leguminosae |
110 | Setaria geniculata (Lam.) Beauv | Poaceae |
111 | Sida aculeata Burm | Malvaceae |
112 | Sida rhombifolia L. | Malvaceae |
113 | Sloanea curatelifolia Griseb | Elaeocarpaceae |
114 | Solanum torvum Sw | Solanaceae |
115 | Stachytarpheta jamaicensis (L.) Vahl | Verbenaceae |
116 | Stevia rebaudiana (Bert.) Bertoni | Asteraceae |
117 | Stygmaphyllum sagreanum A. Juss | Malpighiaceae |
118 | Satureja brownei (Sw.) Briq | Lamiaceae |
119 | Tabernaemontana citrifola L. | Apocynaceae |
120 | Tagetes erecta L. | Asteraceae |
121 | Tagetes lucida Cav | Asteraceae |
122 | Dysphania ambrosioides (L.) Mosyakin et Clemonts | Amaranthaceae |
123 | Tillandsia usneoides (L.) L. | Bromeliaceae |
124 | Tillandsia valenzuelana A. Rich | Bromeliaceae |
125 | Tournefortia hirsutissima L. | Boraginaceae |
126 | Tradescantia spathacea Sw | Commelinaceae |
126 | Turnera ulmifolia L. | Passifloraceae |
List of species used as roots and tubers.
Scientific Name | Family | |
|---|---|---|
1 | Colocasia esculenta (L.) Schott | Araceae |
2 | Dioscorea alata L. | Dioscoraceae |
3 | Ipomoea batatas (L) Lam | Convolvulaceae |
4 | Manhiot esculenta Crantz | Euphorbiaceae |
5 | Xanthososma sagittifolium (L.) Schott | Araceae |
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Agnoletti, M., Pelegrín, Y.M. & Alvarez, A.G. The traditional agroforestry systems of Sierra del Rosario and Sierra Maestra, Cuba. Biodivers Conserv 31, 2259–2296 (2022). https://doi.org/10.1007/s10531-021-02348-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-021-02348-8