Abstract
Ecological restoration is considered a tool for repairing anthropogenic habitat disturbances, but the biodiversity recovering needs to be monitored. Here we evaluate a comprehensive approach for biodiversity: Biodiversity Quality, which comprises a set of 10 indices representing different components of diversity and providing an holistic overview. This approach was tested in a hyper-diverse insect group, the Ichneumonidae family in three different levels of conservation, i.e., a degraded area, a well-conserved area and an area undergoing ecological restoration for 10–15 years. Comparisons were done in three different ecosystems from southern Ecuador, i.e., Andean forest, rainforest and dry forest. We also compared the species assemblages through beta diversity indices. A total of 36 Townes style white Malaise traps were installed at three different conservation levels in 12 natural reserves, and all Ichneumonid insects collected were sorted, mounted and identified to operational taxonomic units (OTUs). A total of 2929 individuals in 708 OTUs were collected, which represented 1264.78 g of biomass. No differences were found between conservation levels, but all indices showed significant differences when comparing ecosystem types. Andean forests had significantly more richness, diversity, population and biomass than the other ecosystems, and less dominance and rarity than dry forests. Species composition of Ichneumonidae assemblages were also different between ecosystems and not so between conservation levels. When comparing in every ecosystem separately, degraded areas in dry forest had significantly more density and biomass than conserved areas. This represents a first attempt of applying this comprehensive approach in such a species-rich family.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Current habitat fragmentation processes may be leading to an extinction debt (i.e., time-delayed extinctions) (Krauss et al. 2010). Its repairing requires not only conservation efforts but also the reversal of this fragmentation. Ecological restoration may help but suitable indicators are needed to monitor its performance. Tropical ecosystems undergoing ecological restoration, especially those in Latin America, have been poorly monitored, and vegetation structure and diversity-related indicators are commonly used (Mazón et al. 2019). When evaluating how human-induced disturbances are impacting on tropical ecosystems and how they are recovering afterwards, it is important to include as many indicator groups as possible (Whitworth et al. 2018). Often there are not enough specialists or time for monitoring all groups, so indicator groups should be tested for suitability in evaluating restoration trajectory (Massi et al. 2021).
Insects may be suitable ecological indicators, with some groups having a fundamental role in ecosystem functions. Hymenoptera are probably the most diverse group of organisms in the world thanks to species-rich (and mostly unknown) parasitoid wasps (Forbes et al. 2018). Pest control functions provided by insects have been estimated to be more than 4 billion dollars per year just for the United States (Losey and Vaughan 2006). These natural enemies of pests are the most affected by the land-use changes in tropical forests (Barnes et al. 2014). Among them, parasitoid wasps have been shown to be highly sensitive to ecosystem disturbances (Marrec et al. 2018) and to climate change (Kankaanpää et al. 2020) making them suitable for monitoring changes. To evaluate how parasitoid assemblages are affected by environmental changes, alpha diversity assessment (i.e., the inherent diversity in every habitat) has been widely used. Other indicators may provide more useful information. Beta diversity (i.e., species turnover between habitats) may be more helpful: even in apparently homogeneous landscapes, beta diversity may vary significantly in parasitoids (Torné-Noguera et al. 2020). A recent approach proposes using a set of biodiversity indices to give a comprehensive understanding of the assemblage’s functionality in a changing environment (Feest et al. 2010). This set of indices, known as Biodiversity Quality, includes biomass, which is an essential variable for food webs (Lister and Garcia 2018; Orihuela-Torres et al. 2018). Although this approach has been used once before on tropical insects (Juen and Feest 2019), it has not been tested with such a species-rich group as Darwin wasps (Hymenoptera: Ichneumonidae), one of the largest insect families (Klopfstein et al. 2019).
The aims of this paper are: (1) to evaluate how Ichneumonidae Biodiversity Quality differs in areas with different conservation levels, (2) to determine if those differences are found in different ecosystems, (3) to assess if Ichneumonidae biomass is more impacted by perturbation than taxonomic-based biodiversity indices, and (4) to evaluate if Ichneumonidae assemblages are more related to the conservation level or to the ecosystem type. We hypothesized (H1) that Biodiversity Quality in areas under ecological restoration will be similar (i.e., non-significantly different, with significance levels > 0.05) to that from the well-conserved forests, and different from that in degraded areas. We expected (H2) that differences in Biodiversity Quality will be less significant, or not significant at all, in the dry forest ecosystems. This hypothesis is supported by the severe abiotic conditions in this ecosystem, that causes a slower ecosystem recovery (Ceccon et al. 2006). We also hypothesized (H3) that Ichneumonidae assemblages will be more determined by the ecosystem type than by the conservation level, but in all ecosystems, we expected that assemblages from restoration areas will be like the forests rather than the open areas. When conducting ecological restoration, efforts should lead to not only recover biodiversity metrics, but also the species composition in order to assure the functionality of the ecosystem (Ramírez et al. 2015). Therefore, both biodiversity and species composition should be more and more resembling that of the reference ecosystem (i.e., conserved forests). Furthermore, we expected (H4) the biomass will be more sensitive (i.e., more significant differences will be found when comparing biomass) to conservation level than taxonomic-based indices.
Methods
Study area
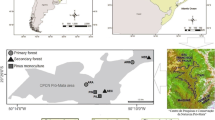
The research took place in 12 natural reserves (one of them belonging to the Ecuadorian National System of Protected Areas, and the rest of them belonging to NGO’s: Naturaleza y Cultura Internacional NCI, Jocotoco Foundation, or private reserves) from three different tropical ecosystems: Andean forest, rainforest and dry forest. In every reserve, three areas in different conservation status were identified: an area clearly degraded (open pasture-like), an area undergoing ecological restoration (either active or passive, with about 10–15 years of recovery) and a well-conserved area (unperturbed forest) (Table 1).
Sampling and identification
Sampling was done in different sampling periods from November 2015 to January 2019. In each conservation area, a Townes style white Malaise trap was installed and kept in the field for six consecutive weeks. Sampling pots were filled with ethanol 70% and replaced every two weeks, having a total of three samples per Malaise trap and a total of 36 traps. The three traps in every reserve were working simultaneously, and traps for the same ecosystem were also simultaneous for most of the reserves (except for Tapichalaca, Buenaventura and Arenillas reserves that were installed in different years).
All insects from the Ichneumonidae family were separated, mounted, and identified to subfamily. Then, individuals were sorted to operational taxonomic units (OTUs), based on external morphological characters. All specimens are preserved in LOUNAZ collection (Universidad Nacional de Loja, Ecuador).
Data analyses
To clarify the nature of biodiversity and improve its measurement, Feest et al. (2010) introduced the concept of biodiversity as a quality defined by a number of functional indices since the Convention on Biological diversity definition does not create this possibility (the term variability in the convention is undefined) (UN 1992). Changes or differences in any of the indices will show how Biodiversity Quality has changed or differs and one reviews the data holistically.
Biodiversity Quality was calculated as described in Feest et al. (2010), which includes the species richness, the population density, the species conservation value index (SCVI), the biomass index, the Shannon-Wiener and Simpson indices for alpha diversity, the Berger-Parker dominance index, and non-parametric species richness estimators (Chao 1, Chao 2 and Jackknife), as explained in Feest et al. (2010) and in Juen and Feest (2019). The SCVI is generally assigned according to the “conservation value” that each species has. However, considering that information on tropical Ichneumonidae species is scarce, and that most species were not identified, we have utilized this index as a rarity/commonness index. We used the reciprocal of the OTUs population multiplied by 100 such that a sample population of 1 had a value of 100 ((1/1) x 100) and 5 a value of 20 ((1/5) x100) and 100 a value of 1 ((1/100) x 100). The SCVI was then the mean (and Standard Deviation) of all OTUs recorded in every site, meaning that we considered the SCVI at a regional scale, i.e., the SCVI of each OTU is calculated based on all individuals collected in this research. All indices were calculated for every conservation level in every ecosystem type and separately for every conservation level in every reserve to make statistical comparisons.
Insect weights were evaluated from the fore wing length based on the allometric equation presented by Mazón et al. (2020), so the forewing length of all OTUs (male and female separately) was measured from the base to the apex using a ZEISS Stemi 2000-C with an ocular micrometer.
Due to the great number of rare OTUs represented by single individuals (nearly 50%) the data were not normal or transformable and non-parametric statistical tests were used (Kruskal-Wallis and post-hoc Dunn tests) to compare all Biodiversity Quality indices according to (a) conservation level, (b) ecosystem type, and (c) conservation level separately in every ecosystem type. Also, to represent the sampling completeness, the proportion of OTUs predicted by the richness estimators from the observed richness is included.
For beta diversity, we conducted a non-metric multidimensional scaling (NMDS), which represents in a 2D plot differences in assemblage composition in every disturbance level and every reserve from the three ecosystem types. Furthermore, statistical differences among Ichneumonidae assemblages were tested with a 9999 permutations PERMANOVA test. Differences were checked among conservation levels and among reserves, but separately for every ecosystem type. Due to the high proportion of singletons and doubletons, both NMDS and PERMANOVA were based on Jaccard similarity index, which only considers presence/absence of species. Beta diversity analyses and Kruskal-Wallis comparisons were done with software Past version 3.0 (Hammer et al. 2001).
Results
We collected a total of 2929 individuals belonging to 708 OTUs, with a total biomass of 1264.78 g (Table S1). The highest species richness, biomass and densities were found in Andean forests. Rarity was high in all samples but specially in the rainforest samples. Regarding sampling completeness, proportions of collected OTUs from the total estimated ranged from 64.38 to 69.53%, according to the Jackknife estimator (Table S2).
None of the variables followed the same patterns in the three ecosystems (Table S2). Areas undergoing restoration showed intermediate values between degraded and well-conserved areas in Andean forests in most indices, except for SCVI. In rainforests, restoration areas showed the lowest values in almost all indices except for biomass, which was intermediate between the other two. In dry forests, indices behaved very differently: while richness and diversity were the lowest in the degraded areas, they had a higher biomass than the other areas.
When running Kruskal-Wallis tests, we observed that there were no statistical differences among conservation levels in any index, but differences were all significant between ecosystem types (Table 2). According to the post-hoc tests, Andean forest had significantly (p = < 0.05) more species richness (observed and estimated), diversity, population and biomass than the other ecosystems, less dominance than dry forest and less rarity than rainforest (Fig. 1). When testing for differences among conservation levels in each ecosystem type, no significant differences were found (Table 2). However, post-hoc tests showed some: in Andean forests, diversity in conserved areas was significantly higher (p = < 0.05) than in degraded areas (Fig. 2 A) and in dry forests both population (Fig. 2B) and biomass (Fig. 2 C) were significantly higher (p = < 0.05) in degraded areas than in conserved forests.
Box plots comparing differences among ecosystem types (AF = Andean Forest, RF = Rainforest, DF = Dry Forest) in every Biodiversity Quality index of Ichneumonidae assemblages: observed richness (A), Shannon-Wiener index (B), Simpson index (C), Berger-Parker Dominance (D), Density (E), SCVI (F), Biomass (G) and Jackknife’s richness estimation (H). Different letters show significant differences (p < 0.05)
Box plots comparing differences among conservation levels (DEG = degraded, RES = undergoing restoration, CON = conserved) in Shannon-Wiener index in Andean forests (AF) (A) and Density (B) and Biomass (C) in dry forests (DF) of Ichneumonidae assemblages. Different letters show significant differences (p < 0.05)
In Beta diversity, assemblages were clearly more similar between the same ecosystem type than between conservation types, and even within the same ecosystem type assemblages did not aggregate by conservation level (Fig. 3). Assemblages were significantly different between reserves (Table 3).
Discussion
Here we have shown it is possible to conduct a comprehensive analysis of such a highly speciose data set as of Darwin wasps, including a wide range of biodiversity measures in different habitats and conservation status.
Of the research hypotheses, H1 is partly supported by the data. Sites undergoing ecological restoration had diversity and dominance intermediate between the other two treatments, with values like those from high conserved areas rather than from degraded lands, at least in Andean and dry forest. However, none of these results were statistically significant. A relatively high diversity in open or degraded areas seems to be the consequence of a higher occurrence of vagrant species, especially when using interception traps (Sverdrup-Thygeson and Birkemoe 2009) such as Malaise traps. Open areas may favour biodiversity in closed, forest habitats, and they are promoted as conservation management strategies in temperate ecosystems (Korpela et al. 2015; Plewa et al. 2020), although not fully demonstrated in tropical habitats (Peh et al. 2006). However, non-significant differences might be due to a relatively low proportion of species collected regarding the estimated richness, although the Jackknife estimator, which usually does not tend to overestimate sampling (Mazón and Bordera 2008), gave proportions proximate to 70%. The apparently low efficiency is a common issue when collecting Ichneumonidae, and it seems hard to improve even when increasing the sampling effort because of the high proportion of singletons that are usually found. For instance, Gómez et al. (2018) had a 7710 Malaise trap days effort in Peruvian Amazonia and collected about 60% of potential Ichneumonidae richness, and Saunders and Ward (2018) in Australia, with a sampling effort of 840 Malaise trap days, reached about 66% of estimated richness. Considering that we had a sampling effort of c. 1620 Malaise trap days, a 65–70% of completeness might be assumed as respectable.
Furthermore, the use of OTUs based on morphological characters may be underestimating species richness. Some Ichneumonidae subfamilies have cryptic species (Veijalainen et al. 2012), and DNA analyses or genitalia treatment are needed to disclose them (e.g., for Ophioninae species, Gauld 1985, Fernández-Triana 2005). However, this kind of analyses requires time and money, which is not always available, and OTUs also called morphospecies, are commonly used in Ichneumonidae diversity assessments (Mazón 2016, Saunders and Ward 2018).
Therefore, the absence of differences between conservation levels seems to be mainly masked by the ecosystem type, which gave significant results in all Biodiversity Quality Indices. Andean forests showed significantly higher species richness, diversity, density and biomass, and less dominance and rarity than the other ecosystems. Rainforests have been associated with a large richness of Darwin wasps (Veijalainen et al. 2012, Gómez et al. 2018), but in this research we found that Andean forests are even more diverse and richer. Parasitoid distribution and richness are highly related to that of their hosts (Barbosa & Caldas 2004; Nascimento et al. 2015), most of them herbivores. The high diversity of herbivorous insects in rainforest seems to be also related to a high diversity of plants (Novotny et al. 2006), however, plant species richness in tropical Andes seems to increase with altitude up to about 1500 masl (Malizia et al. 2020), so a higher plant diversity in Andean forests might harbour a higher diversity of potential hosts and therefore a higher richness of parasitoids. Plant diversity was one of the main features explaining Ichneumonidae diversity in a Peruvian rainforest (Sääksjärvi et al. 2006).
On the other hand, rainforests from southern Ecuador have experienced an intense deforestation in the last decades, more than the other ecosystem types (Tapia-Armijos et al. 2015), so they may be having a lower arthropod diversity than expected in this type of ecosystem. Andean forests in southern Ecuador also harboured a higher richness of leaf beetles compared to rainforests in this region (Thormann et al. 2018). Further, to evaluate how human-induced disturbances are impacting on rainforests, it would be important to sample in the different vertical strata, since invertebrates in the canopy seem to be more sensitive to these perturbances than those from the understory (Whitworth et al. 2018). In parasitoid Hymenoptera, the height from the ground where the trap was installed may significantly influence the species composition (Chan-Canché et al. 2020).
When evaluating the conservation level effect in every ecosystem type separately, significant differences were only found at higher diversity in well conserved areas of Andean forests, and at higher density and biomass in degraded areas of dry forests. These results do not support H2, and even showed dynamics opposite to that expected in dry forests. Dry forests in southern Ecuador are subjected to a high human impact (Graefe et al. 2020). Also, the strong seasonality might be biasing results, depending on the synchrony or asynchrony of sampling and the rainy season, which is not always easy to establish. Ultimately, Ichneumonidae abundance or richness are not easy to predict. For instance, Gómez et al. (2018) found more individuals in secondary and in altered areas than in primary forests, and Saunders and Ward (2018) found that neither habitat nor season or surrounding vegetation explained catches made by each trap.
Ecosystems rather than conservation level determined differences, not only in Biodiversity Quality but also in the species composition, supporting H3, although the statistical tests were somewhat compromised by the very high number of very rare species (those occurring only once). These ecosystems, despite being relatively close to each other, seem to be very particular in their insect composition. For instance, in this region the biodiversity of ants and a predation function associated with them was better explained by habitat conditions than by abiotic variables in an altitudinal gradient (Wallis et al. 2021), and Thormann et al. (2018) showed that species composition of leaf beetles clearly changed between Andean forests and rainforests even when they were connected and part of the same mountain range. SCVI reached nearly 100 at some of the reserves in dry forests and rainforests, meaning that nearly 100% of individuals in those samples were represented by only one species.
Finally, H4 was not supported since biomass did not show different results than the taxonomic-base diversity indices. However, the dry forest showed a differing pattern than the other two ecosystem types, where biomass increased when conservation level decreased, opposite to the rising biomass observed in the other ecosystems when conservation level increased. A high plant diversity helps to reduce the herbivore pressure by enhancing the biomass of their natural enemies (Barnes et al. 2020), but in some cases plant traits instead of plant diversity affects the plant-herbivore interactions more in tropical and subtropical high diverse ecosystems (Wang et al. 2020), and biomass might be altered. Also, in degraded areas, the dominance of a relatively few generalist species will be translated into a low diversity but a high abundance and consequently a high biomass (Ruan et al. 2021) For instance, dung beetles showed a higher biomass in areas with high level of deforestation (Cultid-Medina and Escobar 2016) as seen here in dry forests. Therefore, although biomass gives much information useful for food webs and the conservation status of ecosystems, it may not be reflecting all diversity changes occurring in insect assemblages (Vereecken et al. 2021), and will not represent the loss of small, rare species (Seibold et al. 2019). Furthermore, dry forest functions recover more slowly than wet forests after a perturbation (Poorter et al. 2021), so the 10–15 years of restoration might not be enough for representing significant changes in this ecosystem.
Conclusion
Here we found limited support to demonstrate habitat recovering effects on Ichneumonidae Biodiversity Quality in the three ecosystems along the Southern Ecuador. However, this was a first attempt of representing an extensive sampling of Darwin wasps in this region, with an impressive 700 OTUs set in a 54 trap months sampling effort, and it is approximately a 60–70% of the estimated richness for this region. Also, we found a high proportion of rarity in many of the studied reserves, which is critical for species conservation and the ecosystem services they provide. Habitat fragmentation in tropical forests seems to alter Ichneumonidae richness and abundance but also the species turnover (Ruiz-Guerra et al. 2013) and impacts of land use changes on arthropod diversity are expected to increase with climate change (Sohlström et al. 2022), so there is an urgent need to recover unused degraded lands.
Data availability
The datasets analyzed during the current study are available from the corresponding author on request.
References
Barbosa P, Caldas A (2004) Patterns of parasitoid-host associations in differentially parasitized macrolepidopteran assemblages on black willow Salix nigra (Marsh) and Box elder Hacer negundo L. Basic Appl Ecol 5(1):75–85. https://doi.org/10.1078/1439-1791-00213
Barnes AD, Jochum M, Mumme S, Haneda NF, Farajallah A, Widarto TH, Brose U (2014) Consequences of tropical land use for multitrophic biodiversity and ecosystem functioning. Nat Commun 5:5351. https://doi.org/10.1038/ncomms6351
Barnes AD, Scherber C, Brose U, Borer ET, Ebeling A, Gauzens B, Giling DP, Hines J, Isbell F, Ristok C, Tilman D, Weisser WW, Eisenhauer N (2020) Biodiversity enhances the multitrophic control of arthropod herbivory. Sci Adv 6:eabb6603. https://doi.org/10.1126/sciadv.abb6603
Ceccon E, Huante P, Rincón E (2006) Abiotic factors influencing tropical dry forests regeneration. Braz Arch Biol Technol 49(2):305–312. https://doi.org/10.1590/S1516-89132006000300016
Chan-Canché R, Ballina-Gómez H, Leirana-Alcocer J, Bordera S, González-Moreno A (2020) Sampling of parasitoid Hymenoptera: influence of the height on the ground. J Hymenopt Res 78:19–31. https://doi.org/10.3897/jhr.78.54309
Cultid-Medina CA, Escobar F (2016) Assessing the ecological response of dung beetles in an agricultural landscape using number of individuals and biomass in diversity measures. Environ Entomol 45(2):310–319. https://doi.org/10.1093/ee/nvv219
Feest A, Aldred TD, Jedamzik K (2010) Biodiversity quality: a paradigm for biodiversity. Ecol Indic 10:1077–1082. https://doi.org/10.1016/j.ecolind.2010.04.002
Fernández-Triana JL (2005) The taxonomy and biogeography of cuban Ophioninae (Hymenoptera: Ichneumonidae). Zootaxa 1007:1–60. https://doi.org/10.11646/zootaxa.1007.1.1
Forbes AA, Bagley RK, Beer MA, Hippee AC, Widmayer HA (2018) Quantifying the unquantifiable: why Hymenoptera, not Coleoptera, is the most speciose animal order. BMC Ecol 18:21. https://doi.org/10.1186/s12898-018-0176-x
Gauld ID (1985) The phylogeny, classification and evolution of parasitic wasps of the subfamily Ophioninae (Ichneumonidae). Bull Br Mus Nat 51(2):62–71
Gómez IC, Sääksjärvi IE, Mayhew PJ, Pollet M, Rey del Castillo C, Nieves-Aldrey JL, Broad GR, Roininen H, Tuomisto H (2018) Variation in the species richness of parasitoid wasps (Ichneumonidae: Pimplinae and Rhyssinae) accross sites on different continents. Insect Conserv Divers 11(3):305–316. https://doi.org/10.1111/icad.12281
Graefe S, Rodrigo R, Cueva E, Butz P, Werner F, Homeier J (2020) Impact of disturbance on forest structure and tree species composition in a tropical dry forest of South Ecuador. Ecotropica 22:202002. https://doi.org/10.30427/ecotrop202002
Hammer O, Harper DAT, Ryan PD (2001) Past: Paleontological Statistics software package for education and data analysis. Paleontol Electron 4(1):1–9
Juen L, Feest A (2019) Evaluating the biodiversity quality response of tropical odonata to tree clearance. Int J Trop Insect Sci 39:45–52. https://doi.org/10.1007/s42690-019-00011-y
Kankaanpää T, Vesterinen E, Hardwick B, Schmidt NM, Andersson T, Aspholm PE, Barrio IC, Beckers N, Bêty J, Birkemoe T, DeSiervo M, Drotos KHI, Ehrich D, Gilg O, Gilg V, Hein N, Hoye TT, Jakobsen KM, Jodouin C, Jorna J, Kozlov MV, Kresse JC, Leandri-Breton DJ, Lecomte N, Loonen M, Marr P, Monckton SK, Olsen M, Otis JA, Pyle M, Roos RE, Raundrup K, Rozhkova D, Sabard B, Sokolov A, Sokolova N, Solecki AM, Urbanowicz C, Villeneuve C, Vyguzova E, Zverev V, Roslin T (2020) Parasitoids indicates major climate-induced shifts in arctic communities. Glob Change Biol 26(11):6276–6295. https://doi.org/10.1111/gcb.15297
Klopfstein S, Santos BF, Shaw MR, Alvarado M, Bennett AM, Dal Pos D, Giannotta M, Herrera Florez AF, Karlsson D, Khalaim AI, Lima AR, Mikó I, Sääksjärvi IE, Shimizu S, Spasojevic T, van Noort S, Vilhelmsen L, Broad GR (2019) Darwin wasps: a new name heralds renewed efforts to unravel the evolutionary history of Ichneumonidae. Entomol Commun 1:ec01006. https://doi.org/10.37486/2675-1305.ec01006
Korpela EL, Hyvönen T, Kuussaari M (2015) Logging in boreal field-forest ecotones promotes flower-visiting insect diversity and modifies insect community composition. Insect Conserv Divers 8(2):152–162. https://doi.org/10.1111/icad.12094
Krauss J, Bommarco R, Guardiola M, Heikkinen RK, Helm A, Kuussaari M, Lindborg R, Öckinger E, Pärtel M, Pino J, Pöyry J, Raatikainen KM, Sang A, Stefanescu C, Teder T, Zobel M, Steffan-Dewenter I (2010) Habitat fragmentation causes immediate and time-delayed biodiversity loss at different trophic levels. Ecol Lett 13:597–605. https://doi.org/10.1111/j.1461-0248.2010.01457.x
Lister BC, Garcia A (2018) Climate-driven declines in arthropod abundance restructure a rainforest food web. PNAS 115:E10397–E10406. https://doi.org/10.1073/pnas.1722477115
Losey JE, Vaughan M (2006) The economic value of ecological services provided by insects. Bioscience 56(4):311–323. https://doi.org/10.1641/0006-3568(2006)56[311:TEVOES]2.0.CO;2
Malizia A, Blundo C, Carilla J, Acosta AO, Cuesta F, Duque A, Aguirre N, Aguirre Z, Ataroff M, Baez S, Calderón-Loor M, Cayola L, Cayuela L, Ceballos S, Cedillo H, Ríos WF, Feeley KJ, Fuentes AF, Gámez LE, Grau R, Homeier J, Jadan O, Llambi LD, Loza MI, Macía MJ, Malhi Y, Malizia L, Peralvo M, Pinto E, Tello S, Silman M, Young KR (2020) Elevation and latitude drives structure and tree species composition in Andean forests: results from a large-scale plot network. PLoS ONE 15(4):e0231553. https://doi.org/10.1371/journal.pone.0231553
Marrec R, Pontbriand-Paré O, Legault S, James PMA (2018) Spatiotemporal variation in drivers of parasitoid metacommunity structure in continuous forest landscapes. Ecosphere 9(1):e02075. https://doi.org/10.1002/ecs2.2075
Massi KG, Chaves RB, Tambosi LR (2021) Simple indicators are good proxies for ecological complexity when assessing Atlantic Forest restoration success. Rest Ecol 30(3):e13520. https://doi.org/10.1111/rec.13520
Mazón M (2016) Taking shortcuts to measure species diversity: Parasitoid Hymenoptera subfamilies as surrogates of species richness. Biodivers Conserv 25(1):67–76. https://doi.org/10.1007/s10531-015-1029-y
Mazón M, Aguirre N, Echeverría C, Aronson J (2019) Monitoring attributes for ecological restoration in Latin America and the caribbean region. Rest Ecol 27:992–999. https://doi.org/10.1111/rec.12986
Mazón M, Bordera S (2008) Effectiveness of two sampling methods used for collecting Ichneumonidae (Hymenoptera) in the Cabañeros National Park (Spain). Eur J Entomol 105:879–888. https://doi.org/10.14411/eje.2008.116
Mazón M, Nuñez-Penichet C, Cobos ME (2020) Relationship between body mass and forewing length in Neotropical Ichneumonidae (Insecta: Hymenoptera). Neotrop Entomol 49:713–721. https://doi.org/10.1007/s13744-020-00784-9
Nascimento AR, Almeida-Neto M, Almeida AM, Fonseca CR, Lewinsohn TM, Penteado-Dias A (2015) Disentagling the influence of plants and herbivores on the local diversity of parasitoids in the brazilian Cerrado. Insect Conserv Divers 8(4):313–321. https://doi.org/10.1111/icad.12108
Novotny V, Drozd P, Miller SE, Kulfan M, Janda M, Basset Y, Weiblen GD (2006) Why are there so many species of herbivorous insects in tropical rainforests? Science 313:1115–1118. https://doi.org/10.1126/science.1129237
Orihuela-Torres A, Ordóñez-Delgado L, Brito J, López F, Mazón M, Freile JF (2018) Ecología trófica del búho terrestre Athene cunicularia punensis (Strigiformes: Strigidae) en el archipiélago de Jambelí, provincia de El Oro, suroeste de Ecuador. Rev Peru Biol 25:123–130. https://doi.org/10.15381/rpb.v25i2.13376
Peh KSH, Sodhi NS, de Jong J, Sekercioglu CH, Yap CAM, Lim SLH (2006) Conservation value of degraded habitats for forest birds in southern Peninsular Malaysia. Divers Distrib 12:572–581. https://doi.org/10.1111/j.1366-9516.2006.00257.x
Plewa R, Jaworski T, Tarwacki G, Gil W, Horak J (2020) Establishment and maintenance of power lines are important for insect diversity in Central Europe. Zool Stud 59:e3. https://doi.org/10.6620/ZS.2020.59-3
Poorter L, Rozendaal DMA, Bongers F, Almeida JS, Álvarez FS, Andrade JL, Arreola LF, Becknell JM, Bhaskar R, Boukili V, Brancalion PHS, César RG, Chave J, Chazdon RL, Colletta GD, Craven D, de Jong BHJ, Denslow JS, Dent DH, DeWalt SJ, Díaz E, Dupuy JM, Durán SM, Espírito Santo MM, Fernandes GW, Finegan B, Granda Moser V, Hall JS, Hernández-Stefanoni JL, Jakovac CC, Kennard D, Lebrija-Trejos E, Letcher SG, Lohbeck M, Lopez OR, Marín-Spiotta E, Martínez-Ramos M, Meave JA, Mora F, Moreno VS, Müller SC, Muñoz R, Muscarella R, Nunes YRF, Ochoa-Gaona S, Oliveira RS, Paz H, Sanchez-Azofeifa A, Sanaphre-Villanueva L, Toledo M, Uriarte M, Utrera LP, van Breugel M, van der Sande MT, Veloso MDM, Wright SJ, Zanini KJ, Zimmerman JK, Westoby M (2021) Functional recovery of secondary tropical forests. PNAS 118(49):e2003405118. https://doi.org/10.1073/pnas.2003405118
Ramírez W, Aguilar-Garavito M, Cabrera M (2015) Definición de objetivos, metas, indicadores y cuantificadores para el monitoreo a procesos de restauración ecológica. In: Aguilar-Garavito M, Ramírez W (eds) Monitoreo a procesos de restauración ecológica, aplicado a ecosistemas terrestres, 1st edn. Instituto de Investigación de Recursos Biológicos Alexander von Humboldt, Bogotá DC, pp 33–41
Ruan H, Wu X, Wang S, Yang J, Zhu H, Guo Q, Wang L, Wang D (2021) The responses of different insect guilds to grassland degradation in northeastern China. Ecol Indic 133:108369. https://doi.org/10.1016/j.ecolind.2021.108369
Ruiz-Guerra B, Hanson P, Guevara R, Dirzo R (2013) Richness and abundance of Ichneumonidae in a fragmented tropical rain forest. Neotrop Entomol 42(5):458–465. https://doi.org/10.1007/s13744-013-0143-3
Sääksjärvi IE, Ruokolainen K, Tuomisto H, Haataja S, Fine PVA, Cárdenas G, Mesones I, Vargas V (2006) Comparing composition and diversity of parasitoid wasps and plants in an amazonian rain-forest mosaic. J Trop Ecol 22:167–176. https://doi.org/10.1017/S0266467405002993
Saunders TE, Ward DF (2018) Variation in the diversity and richness of parasitoid wasps based on sampling effort. PeerJ 6:e4642. https://doi.org/10.7717/peerj.4642
Seibold S, Gossner MM, Simons NK, Blüthgen N, Müller J, Ambarli D, Ammer C, Bauhus J, Fischer M, Habel JC, Linsenmair KE, Nauss T, Penone C, Prati D, Schall P, Schulze ED, Vogt J, Wöllauer S, Weisser WW (2019) Arthropod decline in grasslands and forests is associated with landscape-level drivers. Nature 574:671–674. https://doi.org/10.1038/s41586-019-1684-3
Sohlström EH, Brose U, van Klink R, Rall BC, Rosenbaum B, Schädler M, Barnes AD (2022) Future climate and land-use intensification modify arthropod community structure. Agric Ecosyst Environ 327:107830. https://doi.org/10.1016/j.agee.2021.107830
Sverdrup-Thygeson A, Birkemoe T (2009) What window traps can tell us: effect of placement, forest openness and beetle reproduction in retention trees. J Insect Conserv 13:183–191. https://doi.org/10.1007/s10841-008-9141-x
Tapia-Armijos MF, Homeier J, Espinosa CI, Leuschner C, de la Cruz M (2015) Deforestation and forest fragmentation in South Ecuador since the 1970s – losing a hotspot of biodiversity. PLoS ONE 10(9):e0133701. https://doi.org/10.1371/journal.pone.0133701
Thormann B, Ahrens D, Espinosa CI, Armijos DM, Wagner T, Wägele JW, Peters MK (2018) Small-scale topography modulates elevational a-, b- and g-diversity of Andean leaf beetles. Oecologia 187(1):181–189. https://doi.org/10.1007/s00442-018-4108-4
Torné-Noguera A, Arnan X, Rodrigo A, Bosch J (2020) Spatial variability of hosts, parasitoids and their interactions across a homogeneous landscape. Ecol Evol 10(8):3696–3705. https://doi.org/10.1002/ece3.6158
UN (1992) Convention on Biological Diversity. https://www.cbd.int/doc/legal/cbd-en.pdf.Accessed 25 April 2022
Veijalainen A, Wahlberg N, Broad GA, Erwin TL, Longino JT, Sääksjärvi IE (2012) Unprecedented ichneumonid parasitoid wasp diversity in tropical forests. Proc R Soc B 279:4694–4698. https://doi.org/10.1098/rspb.2012.1664
Vereecken NJ, Weekers T, Leclercq N, De Greef S, Hainaut H, Molenberg JM, Martin Y, Janssens X, Noël G, Pauly A, Roberts SPM, Marshall L (2021) Insect biomass is not a consistent proxy for biodiversity metrics in wild bees. Ecol Indic 121:107132. https://doi.org/10.1016/j.ecolind.2020.107132
Wallis CIB, Tiede YC, Beck E, Böhning-Gaese K, Brandl R, Donoso DA, Espinosa CI, Fries A, Homeier J, Inclan D, Leuschner C, Maraun M, Mikolajewski K, Neuschulz EL, Scheu S, Schleuning M, Suárez JP, Tinoco BA, Farwig N, Bendix J (2021) Biodiversity and ecosystem functions depend on environmental conditions and resources rather than the geodiversity of a tropical biodiversity hotspot. Sci Rep 11(1):24530. https://doi.org/10.1038/s41598-021-03488-1
Wang MQ, Li Y, Chesters D, Bruelheide H, Ma K, Guo PF, Zhou QS, Staab M, Zhu CD, Schuldt A (2020) Host functional and phylogenetic composition rather than host diversity structure plant-herbivore networks. Mol Ecol 29:2747–2762. https://doi.org/10.1111/mec.15518
Whitworth A, Huarcaya RP, Whittaker L, Braunholtz L (2018) Are we using the most appropriate methodologies to assess the sensitivity of rainforest biodiversity to habitat disturbance? Tropic Conserv Sci 11:1–4. https://doi.org/10.1177/1940082918788445
Acknowledgements
We would like to express our gratitude to the staff in every reserve that helped us with the permissions and facilities: Hugo Tapia (Madrigal Reserve), Joerg Zeilinger (ECSF), Arturo Jiménez (Arcoíris Reserve and Foundation), Michael Möens and Jocotoco Foundation’s staff (Tapichalaca and Buenaventura Reserves), Catherine Vits (Copalinga Reserve), Robert Guerrero (Zapotepamba Reserve), Felipe Serrano and Naturaleza y Cultura Internacional’s staff (Laipuna, La Ceiba and Jamboé Reserves), Lenín Ramón and MAATE’s staff (Arenillas Reserve), and Diego Loaiza (El Padmi Reserve). We also thank Ecosulis Ltd. for funding Alan Feest’s research programme and the publication of this paper. Samplings were done under MAATE permissions 001-2018-IC-VS-UPN-DPAL-MAE, 011-2014-IC-FLO-DPL-MA, 013-2018-IC-FLO-FAU-DPAZCH-UPN-VS/MA and 005-2019-IC-FLO/FAU-DPAEO-MAE.
Funding
This research was supported by the Prometeo Project of Secretaría de Educación Superior, Ciencia, Tecnología e Innovación from Ecuador, and the projects DI-10-FARNR (2017–2019) and DI-10-FARNR-2019 from Universidad Nacional de Loja, Ecuador.
Author information
Authors and Affiliations
Contributions
Conceptualization and identification of the OTUs were performed by Marina Mazón. Methodology and field work were carried out by Marina Mazón and Oscar Romero. The analyses were conducted by Marina Mazón and Alan Feest. The first draft of the manuscript was written by Marina Mazón, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Communicated By: Anurag Chaurasia
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mazón, M., Romero, O. & Feest, A. Ecosystem type might mask the effect of ecosystem recovery on parasitoids’ biodiversity quality. Biodivers Conserv 32, 509–522 (2023). https://doi.org/10.1007/s10531-022-02509-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-022-02509-3