Abstract
The European Biodiversity Strategy has set the key goal to maintain and promote biodiversity. Managed forests here play a key role, as they are among the most diverse ecosystems. To create biodiversity rich managed forest landscapes, we need a deep understanding on how management affects forest structure and subsequently habitat quality on the local and landscape level. However, to date a confusingly large amount of different terms for various management systems exist and it remains unclear how forest structure and composition affected by specific management systems affect biodiversity. Here, we first aim to clarify forest management systems terminology. Second, we link existing management systems with forest structure and review at European level how these structures affect local α-, as well as landscape-scale β- and γ-biodiversity. We found that research and derived management guidelines have a strong focus on local forest stand heterogeneity and related α-diversity, while ignoring the importance of landscape-scale heterogeneity and therefore β- and γ-diversity. Instead of promoting one management system as an all-in-one-solution, a diversity of different forest management systems seems the most promising way to create biodiversity rich forest landscapes. We finally discuss how a combination of different management systems might help to create structurally rich forest habitats and landscapes, simulating different successional stages and promoting species rich communities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forests provide important ecosystem services such as regulating the water-, carbon- and nutrient cycle, providing resources like wood, supporting soil formation, and supplying cultural services through recreation and education (Felipe-Lucia et al. 2018). Moreover, due to their three-dimensional complexity and richness in habitat features (e.g., dead wood, habitat trees, canopy gaps), natural forests harbor tremendously diverse species communities (Eckelt et al. 2018; Miklín et al. 2018).
Today, most of the European forests are classified as “seminatural” and only a few are considered undisturbed by humans (FOREST EUROPE 2020). In fact, only 0.7% of Europe’s forest area exhibits the structural attributes characteristic of primary forests (Dieler et al. 2017; Sabatini et al. 2018). Nonetheless, also seminatural, managed forests can be inhabited by species-rich communities if diverse habitat conditions exist, and necessary resources are available (Sebek et al. 2015). However, most forests in Europe are still characterized by intense resource exploitation and forest biodiversity is still declining at large scale (EEA 2020). Therefore, given the large amount of managed forest areas all over Europe and the high demand for wood, forest management approaches that maintain biodiversity are still urgently needed (Augustynczik et al. 2020; Kraus and Krumm 2013; Selva et al. 2020).
Multifunctional forest management, which simultaneously guarantees continuous wood supply and biodiversity conservation, has been aspired for a long time. As a reaction to the forest dieback in the 1980s, the Ministerial Conference on the protection of forests in Europe (MCPFE) was established. The goal of the MCPFE was to elaborate sustainable forest management concepts and hence, protect forests all over Europe (Rametsteiner and Mayer 2004). They introduced three different management classification types, of which “class 1” is mainly focusing on conserving biodiversity, “class 2” on protecting forests as landscape elements, and “class 3” on keeping the protective functions (e.g., water supply) of forests (MCPFE Expert Level Meeting 2002). These classes, until today, are used to evaluate the state of European forests and the proportion of protected forests in Europe (FOREST EUROPE 2020). Although this classification aims the multifunctional use of European forests, only minor parts of the European forests are protected under MCPFE class 1, hinting that biodiversity conservation still is a subordinate goal in forest management (FOREST EUROPE 2020; Uhl et al. 2022a). In addition, the MCPFE gives no clear definition of how to create or manage a biodiversity rich forest.
At the beginning of the 1990s, Natura 2000, a network of important areas for biodiversity covering Europe has been established. It is the largest coordinated network of areas of conservation concern in the world, extending across all 27 EU Member States. Natura 2000 has the goal to preserve biodiversity at European scale. However, there is broad evidence that after decades the goals of Natura 2000 are still not reached (Princé et al. 2021; Rada et al. 2019). One reason might be that forest management is still possible within Natura 2000 areas, as long as “favorable habitat conditions” are kept. The focus is often set on keeping the current status, which might on the one hand prevent habitat degradation, but on the other hand also hampers interventions to enhance habitat quality. Especially on the landscape scale, this could result in homogenized forest landscapes with low habitat heterogeneity.
Taken together, many efforts to protect forest biodiversity are still ineffective in promoting biodiversity, which might be partly due to the fact that often no clear guidelines—apart from keeping the status quo—for conservation are set. This might also stem from existing knowledge gaps. Indeed, there seems an overly strong focus on local (α-) diversity, while landscape-scale heterogeneity in the form of β- and γ-diversity patterns are mainly ignored. Furthermore, indicators that are used to evaluate forest diversity (Barbati et al. 2014) often do not reflect multi-taxon biodiversity (Paillet et al. 2024). As a result, existing management recommendations to promote biodiversity are not strongly evidence based (Domínguez and Shannon 2011). Despite the need to evaluate effects of different forest management systems on biodiversity, a broad review encompassing all different management systems and their effects on multi-taxonomic diversity is still missing. Therefore, we here aim to give an overview of European forest management systems and review the current knowledge about how these systems affect forest structure and biodiversity at local and landscape scale (Table 1). On the local scale, which is the forest stand unit in which foresters are operating, we discuss different forest management systems and their effects on biodiversity. We classify the type of forest management system according to Trentanovi et al. (2023). Hence, we consider the effects on biodiversity of clear-cuts, clear-cuts with retention, shelterwood systems, selection cutting, single coppice and coppice with standards. We also discuss management concepts that are suggested beyond management systems, viz. retention forestry, close-to-nature forestry, forest reserves, and forest conversion in times of climate change. Lastly, we provide recommendations about how different management combinations can promote biodiversity in managed forests.
Inclusive vs. segregative forest management concepts
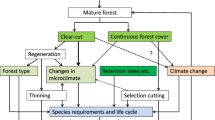
Today, forest management has the goal to provide different ecosystem services, yet the main management goal can depend on the specific locality (Blanco et al. 2015). For example, while management of urban forests may primarily focus on recreation and aesthetics, in other contests timber production or biodiversity conservation could be more important (Blanco et al. 2015). Therefore, management systems are shaped by their main focus of interest. In practice clear trade-offs often exist. One important example is the trade-off between timber production and forest biodiversity (Cosyns et al. 2020). From this well-known debate, existing forest management systems can be sorted into a larger theoretical concept: Similar to the land-sparing vs. land-sharing debate in agricultural systems (Loconto et al. 2020), inclusive vs. segregative concepts for forest management are discussed (Boncina 2011; Suda and Pukall 2014). Yet, the definition of what is “inclusive” and what is “segregative” often is unclear. In fact, these terms are only part of a larger sociological classification system, ranging from “Inclusion”, “Integration”, “Segregation”, “Exclusion”, to finally “Extinction” (Fig. 1, Infobox 1). Within our review, we will assign each management system to the according concept, in order to sort them into the existing theoretical framework for all the different systems discussed.
Theoretical management concepts and categories that either try to address all management objectives in one area, or address different objectives in different areas. Inclusive concepts seek to find an all-in-one solution, while integrative concepts acknowledge conflicts of interest between economic interests (wood supply) and biodiversity conservation. Segregative and exclusive concepts use different areas for different interests, while extinction describes the banishment of one interest. The different concepts are implemented through different forest management systems, which in turn can affect local- and landscape-scale heterogeneity as well as biodiversity
Infobox 1: Definitions and examples for inclusive, integrative, segregative, and exclusive forest management concepts.

The theoretical concept on which a management system is based can be linked to different biodiversity measures. Within the biodiversity conservation debate, there is often an overly strong focus on α-diversity patterns. Inclusive concepts and associated management systems such as selection cutting often focus on the local characteristics of a forest. Mainly the local stand structural heterogeneity is promoted, leading to high α-diversity. Landscape-scale heterogeneity yet is rather low, which results in neglectable variation in β-diversity. Landscape-scale γ-diversity is, therefore, rather driven by α-diversity patterns. Segregative and exclusive concepts, in contrast, address also the landscape scale. Here, not only the management within one stand is considered, but also the spatial arrangement of stands with different objectives. Such concepts can have high variation in local α-diversity, as stand management and therefore structure is varying due to the local objective. Varying management results in high landscape-scale heterogeneity and, as a result, in differences in species composition (β-diversity). Landscape-scale γ-diversity, consequently, is rather driven by β-diversity patterns (Schall et al. 2018; Fig. 1). To maintain α- as well as β-diversity, and therefore conserve regional γ-diversity, local habitat quality as well as landscape-scale heterogeneity need to be promoted. Integrative forest management concepts here try to find a compromise between wood supply and biodiversity conservation by using management to mimic natural disturbances (Kraus and Krumm 2013). Also combining different management systems to promote landscape-scale habitat heterogeneity is suggested to maintain landscape-scale diversity (Schall et al. 2018). However, the multitude of different management systems and inconsistent definitions make it until today difficult to get a good overview of how to manage forests biodiversity friendly. Although there is some all-encompassing literature presenting different forest management systems (e.g. Gayer 1898; Matthews 1989; Nyland 2002), the effects of different management systems on biodiversity are not discussed in detail as the main focus is the management system itself and how it is applied.
Forest management systems
Within the following chapters, we introduce the different forest management systems, sort them into the broader context of inclusive vs. segregative gradient, and give an overview on their application. We also review the current knowledge on how these systems affect α-, β-, and γ- diversity and identify knowledge gaps concerning management-biodiversity relations. We will start with segregative concepts that are focusing on wood supply, then discuss integrative concepts in which interests of wood supply and biodiversity conservation are balanced, and finally focus on segregative and exclusive concepts where the focus today is more on biodiversity conservation interests.
Clearcutting without and with retention (even-aged, rotational management)
Clearcutting, or even-aged, rotational management is a segregative management concept, where the management has a strong focus on wood supply (Fig. 1). Clearcutting describes the all-in-one harvest of wood on a certain area of forest, leaving an open area that is then again planted or recolonized with seedlings. By regular rotation of clearcutting and regeneration areas, a partitioned forest develops, with homogeneous age classes within each of the blocks. The term “Rotational (regular) forest management” yet is not only used for clearcuts, but also for regular shelterwood systems with block or strip felling (following Matthews 1989). Clearcutting and even-aged management (“Altersklassenwald” following Gayer 1898) was long seen as unsustainable and invasive management method, as it has major effects on the environment (Lindenmayer et al. 2006; Nolet et al. 2018). The drastic change of the habitat structure from closed canopy to open areas not only is a major disturbance for biotic communities, but also alters soil processes (Dahlgren and Driscoll 1994; Vitousek 1981), can promote erosion (Borrelli et al. 2017), and leads to drastic changes in microclimate (Kovács et al. 2020). When the focal tree species is to be changed e.g. when a spruce forest should be replaced by more climate-tolerant tree species, or when light demanding species like oaks or pines are to be promoted, clear-cutting and replanting can be an option. Another way to achieve regeneration after a clear-cut is the “seed tree method”. Here, the clear-cut does not include all trees of an area, but some solitary trees or groups of trees are kept (clear-cutting with retention). Especially for wind-dispersed, light-demanding tree species, this is the preferred regeneration method.
The α-diversity of even-aged forest areas is rather low compared to uneven-aged forest management systems (Savilaakso et al. 2021). Yet, and despite its reputation, clearcutting not only has negative effects on local biodiversity (α-diversity). While the harvest itself is a major disturbance, there are also very long regeneration times with no interventions. Clearcutting events can also simulate natural disturbances like bark beetle infestations and therefore positively affect structural heterogeneity and biodiversity (Blixt et al. 2015; Heinrichs and Schmidt 2009). In fact, many species of conservation concern have been found to profit from clearcutting events. However, clearcut species communities are often quite distinct from communities of closed forests and often are affiliated to more open habitat types (Hilmers et al. 2018; Jokela et al. 2019; Lõhmus 2011; Żmihorski et al. 2016).
Schall et al. (2018) found high landscape-scale multi-diversity (γ-diversity) in even-aged managed forests, mainly due to increased β-diversity among the whole management system. Even-aged management might, therefore, even help promote regional biodiversity by creating heterogeneous landscapes. When clear-cuts are additionally combined with retention forestry (see below) and some harvest residuals (e.g., branches, but also low quality stems and upheavals) remain at clearcuts, the resulting habitat can serve as highly valuable and biodiversity-rich early successional stage area (Fedrowitz et al. 2014; Uhl et al. 2022a).
Shelterwood systems
“Shelterwood” is an umbrella term for different management systems, in which young trees are growing “sheltered” beneath older trees (“Schirmschlagform” following Gayer 1898). Most shelterwood systems can be divided into different phases, starting with an initial thinning to initiate or sustain tree regeneration and successive further fellings. The partial opening of the canopy in the midst of the forest can help young trees develop under constant forest microclimate but with enough available light (Raymond et al. 2009). In addition, depending on the shade tolerance of the focal tree species, sheltering trees are thinned more quickly (for light demanding species like oak) or very slowly (for shade tolerant species like beech). The selection which trees are felled and their spatial distribution differs between shelterwood systems. Shelterwood systems can follow the objective to establish either a homogeneous forest structure (regular shelterwood) or multi-species cohorts with diverse tree layers, age-classes and light regimes (irregular shelterwood) (Raymond et al. 2009).
Regular shelterwood (even-aged, rotational management)
Regular shelterwood, like clearcutting, is considered a segregative concept that focuses on wood production (Fig. 1). It is also called the “uniform system” as it is the most homogeneously structured type of shelterwood. Other terms for this management system are “shelterwood compartment system”, “system of successive regeneration fellings”, “Schirmschlag” (following the original definition of Gayer 1898), or “rotational (regular) forest management” (see “clearcuts”). The latter term is sometimes also used for clearcutting. In fact, the transition between clearcuts, clearcuts with retention and regular shelterwood can blur, as the degree of thinning depends on the focal tree species that is to be promoted afterwards. In regular shelterwood systems, the whole forest or a large forest block is thinned uniformly to achieve an evenly distributed age-structure of trees (Nyland 2010). As young trees are growing under sheltered conditions, regular shelterwood is a suitable management system for shade tolerant tree species like beech, fir and linden. By rotating thinning and harvesting through growing up forest blocks while other blocks are regenerating, regular wood supply is guaranteed (Nyland 2010). Besides uniform shelterwood, also strip shelterwood (“Saumschlag” after Gayer 1898) is sometimes defined as regular shelterwood system (Matthews 1989). Here, the forest is separated in strips instead of blocks. Yet, the rotation principle is the same in both systems. In contrast to clear-cutting management, the re-establishment is usually achieved by natural seed dispersal so that there is no need for plantings. For this reason, regular shelterwood systems after the final removal of old trees might resemble the regeneration phase of natural forest succession. Due to the block/strip management, there can be a high horizontal structural heterogeneity on a large spatial scale. Yet, the vertical structure and age heterogeneity in regular shelterwood systems is rather low (Raymond and Bédard 2017). Due to the low vertical heterogeneity it was long believed that regular shelterwood mainly focuses on the production of wood and therefore neglects some ecosystem services and biodiversity conservation (Mason et al. 2022; Puettmann et al. 2015; Raymond et al. 2009).
On the local scale, thinning like applied in regular shelterwood systems have heterogeneous effects on α-biodiversity, depending on the thinning intensity and the species group considered (de Groot et al. 2016; Tinya et al. 2023). Vascular plant and bryophyte diversity does temporarily profit from thinning, as light availability in the understory is increased after the initial fellings (Tullus et al. 2019). However, birds and fungi seem to react negatively on thinning (de Groot et al. 2016). Brunet et al. (2010) state that regular shelterwood systems negatively affect biodiversity, yet, the negative effects in their study could mostly be explained by missing deadwood structures and the rapidly changing microclimate after thinning. However, the reduced amount of deadwood is a problem in all silvicultural systems, and a rapid change in microclimatic conditions only is true for the thinning cuts, but not for the retention times in shelterwood systems. In addition, the study seems to have focused on α-diversity, without taking into account that diverse communities might be found throughout the whole rotation system due to the high landscape heterogeneity between different management blocks.
In fact, regular shelterwood promotes γ-multidiversity (including i.e. lichens, vascular plants, spiders, hymenopterans, true bugs, and birds) in management combination experiments on the landscape scale. Uniform regular shelterwood performed even better than uneven aged forest, recently unmanaged forest or management combinations with these two latter forest types (Schall et al. 2020). At the same time, the occurrence of economically important species (viz. bark beetles) is reduced when regular shelterwood systems are applied (Williams et al. 2017).
Irregular shelterwood
Irregular shelterwood systems are following an integrative concept, where both wood supply and ecosystem function is to be sustained (Fig. 1). In contrast to clearcuttings and regular shelterwood systems, the irregular shelterwood system does not divide a forest into partitions or blocks of management. Irregular shelterwood systems are more inspired by natural disturbance regimes and therefore try to imitate such. The cuts are not applied to a whole block, but small groups of trees are removed irregularly, so that a heterogeneous forest structure with multiple layers develops (“Femelschlag” or “Femelhieb” following Gayer 1898). In contrast to the regular shelterwood, the regeneration period is extended, adding at least 20% to the regular rotation length (Raymond et al. 2009). Irregular shelterwood systems therefore allow more flexibility concerning the spatial arrangement of cuttings and time of regeneration, making them more adaptable to the requirements of different tree species (Raymond et al. 2009). Yet, the long regeneration time also reduces heterogeneity over time, as the forest becomes more and more shaped by dominant tree species. So, the heterogeneity of irregular shelterwood systems is only sustained on a landscape scale by constant management interventions at different sites. Irregular shelterwood systems can resemble selection cutting systems, yet, the main focus of the two management systems differs: The main focus of irregular shelterwood systems is to create multi-cohort stands, while selection cutting systems aim to balance different age classes across the stand (Raymond et al. 2009). So, irregular shelterwood systems are meant to create forests with high horizontal and vertical heterogeneity, while selection cutting systems only focus on high vertical heterogeneity. Apart from these slightly different objectives, the structure of irregular shelterwood systems and selection cutting systems can be very similar.
There are three different forms of irregular shelterwood: Expanding-gap, Continuous cover, and Extended (Raymond et al. 2009). The expanding-gap method aims to remove small groups of trees. Starting at these small canopy openings (normally the size of one-two tree lengths, summing up to about 20% of the whole stand), the gaps are extended step by step (always with extension cuts on 20% of the area), until the whole stand is replaced. In the continuous cover method not only gap cuts, but also single tree removals are possible, enabling the maintenance of a closed canopy. The management can therefore be adapted to the ecological needs of the focal tree species that should be promoted: Gaps for light demanding species like oak, spruce and pine, and single tree removals for sheltered growth of shade tolerant species like beech, fir and linden trees. The third irregular shelterwood method, the Extended method, “aims to regenerate the whole stand while keeping a more regular structure” (Raymond et al. 2009). It resembles the regular shelterwood system (Nyland 2002), but has a longer regeneration period (> 20% of the rotation length). The final removal cut is delayed, for which reason the forest longer remains in a two-cohort-state. Irregular shelterwood systems are believed to have low impact on the local ecosystem, as there are long periods without intervention. Yet, the scale on which no intervention takes place differs between the methods: In the expanding-gap cutting and the continuous cover method interventions are small scaled, i.e. at gap cuts, whereas in the surroundings, further gap cuts or gap extensions are optional. For the Extended method, the whole stand is thinned once, and then there is a long period of no intervention for the whole forest stand (Raymond et al. 2009). Horizontal heterogeneity, therefore, is higher in continuous cover and especially expanding gap methods, while extended irregular shelterwood is a more uniform system with just elongated regeneration phases.
For promoting local biodiversity (α-diversity), irregular shelterwood systems can be very suitable. Especially the expanding gap management might be effective for biodiversity conservation, as here both—vertical and horizontal canopy structure—are promoted. Forest gaps, as created by the expanding gap method, can serve as biodiversity hotspots (Muscolo et al. 2014). Starting with an initial gap cut of 1–2 tree lengths in diameter, the canopy openings are extended with successive fellings, enabling also the creation of larger forest gaps. Due to the increased light availability, forest gaps have a high diversity of flowering plants (Heinrichs and Schmidt 2009; Lanta et al. 2019; Steinert et al. 2018; Ujházy et al. 2017). In addition, many rare and protected species like orchids are dependent on forest gaps (Bertolini et al. 2012; Uhl et al. 2022b). Based on the high diversity of flowering plants, forest clearings provide also important resources for insect pollinators (Korpela et al. 2015; Rodríguez and Kouki 2017) and herbivores (Blixt et al. 2015; Jokela et al. 2019). In addition, deadwood-dwelling beetles prefering sun-exposed deadwood can profit from forest gaps (Lettenmaier et al. 2022). The natural resilience might also profit from the expanding-gap management as for birds and bats, forest clearings provide important foraging habitats (Müller-Kroehling et al. 2020; Tena et al. 2020), and the number of parasitoids is also increasing (Eckerter et al. 2022).
On the landscape scale, irregular shelterwood methods might also pose an opportunity to promote regional γ-diversity, as the irregular thinning results in increased microclimatic variation, which allows for more diverse communities preferring different habitats (Lettenmaier et al. 2022; von Felten et al. 2020). Horizontal heterogeneity and therefore the diversity of different ecological niches is diversified, especially in the expanding-gap and the continuous method. Yet, studies on the effects of irregular shelterwood systems on γ-diversity are mostly missing. Also, there are recently no studies comparing γ-diversity of irregular shelterwood systems with other forest management systems.
Selection cutting (single tree selection and group selection/gap cutting)
Selection cutting systems are sometimes promoted as inclusive concepts addressing economic (e.g., wood production) and ecological interests (e.g., biodiversity conservation, ecosystem services) simultaneously (Fig. 1). Due to the persistent trade-off between wood extraction or e.g., deadwood enrichment, it more likely should be sorted within the integrative concepts (Fig. 1). While shelterwood systems are based on the thought of growing a new tree stock below the shelter of older trees, selection cutting systems are more flexible, with the possibility of cutting single trees or groups, replanting new tree species or including natural regeneration. The main objective here lies on generating a multilayered forest comprising similar percentages of all age classes, so that a constant wood supply in each block is all the time guaranteed (Gayer 1898). Selection cutting is sometimes also called continuous cover forestry. Yet, this term is also used for shelterwood systems like continuous cover shelterwood (Mason et al. 2022). Both systems have in common that a continuous canopy cover is maintained: In the first case by promoting all age classes at the small scale through removal of single trees. In the second case by establishing the next tree generation before the final harvest of the preceding generation. The transition between these systems is fluent and the choice of how much thinning is done often depends on the tree species that is to be promoted. Shade tolerant tree species profit from single tree removals, while light demanding species need larger-scale thinning to increase light availability in the understory. Some authors also differentiate between continuous cover forestry cutting trees of a target diameter (“Zielstärkennutzung”), and single-tree selection cutting trees of multiple age classes (Aszalós et al. 2022). Other terms for selection cutting are perpetual forest (“Dauerwald”), stem-by-stem removal or plenter forestry (“Plenterwald”). The term “plenter forestry” can be misleading, as the term “plenter” is also used as a successional stage in natural forest ecosystems, in which many old grown trees and a high amount of deadwood can be found (Remmert 1991). Forests under plenter forestry and natural forests in the plenter successional stage might have a similar age variance, yet the amount of deadwood and old trees in plenter forestry is rather low, like for other managed forests.
For shade-tolerant tree species like beech and fir trees, which require a closed canopy and a stable microclimate for natural regeneration, selection cutting with a continuous cover might be the right choice, as selection cutting systems are known to maintain a relatively stable microclimate (Kovács et al. 2020). If more light demanding tree species are to be promoted in selection cutting forests, group selection cuts can be applied. Doing so, the proportion of species like maple and ash can for example be enhanced in beech forests. Group selection cutting is another form of selection cutting, where harvests are performed by creating forest gaps of varying size. In contrast to the extended-gap shelterwood system, an existing gap is not extended during subsequent cuttings, but new gaps are created (Raymond et al. 2009). The gap size plays a major role when it comes to tree regeneration and biodiversity conservation. For the promotion of light demanding tree species, a gap should be twice the size of the tallest surrounding trees, while for shade tolerant species a gap with the size of the tallest surrounding trees is sufficient (Muscolo et al. 2014). Brang et al. (2014) defines a gap size range of 0.05–0.5 ha in the group selection system, with shade tolerant species growing also at gaps of 0.05 ha and smaller, while light demanding species profit from larger gaps up to about 0.3 ha size. Group selection cutting results in slightly higher horizontal heterogeneity than single tree selection, which might promote biodiversity and especially forest gap species (Eckerter et al. 2022; Jokela et al. 2019). Yet, landscape heterogeneity within selection cutting systems is rather low, especially when larger gaps are missing due to a strong focus of single tree removals in selection cutting systems.
Selection cutting systems can be rich in α-diversity, as the focus on vertical heterogeneity results in a variety of canopy layers at the small scale. In fact, responses of birds to multilayered versus single layered stands lead to the habitat heterogeneity hypothesis (MacArthur and MacArthur 1961), which originally applies to the local scale. But also Brunet et al. (2010) conclude that, in terms of biodiversity conservation, selection cutting has advantages compared to regular shelterwood systems. Due to the more stable microclimatic conditions, species diversity was better preserved with continuous cover selection cutting, while regular shelterwood systems were believed to harm most forest species (Brunet et al. 2010). In fact, forest species seem to be mostly unaffected by selection cutting, as concluded by a large review from Ekholm et al. (2023). Selection cutting, therefore, is a suitable method to maintain and promote the local α-diversity of continuous cover forest species.
On a landscape scale, however, selection cutting systems are characterized by low heterogeneity in horizontal forest structure. All age classes are homogeneously distributed at a small scale, while the small scale patches are ‘identically’ repeated in space. As a result, there is low landscape heterogeneity and low structural β-diversity (Schall et al. 2018). Selection cutting systems, therefore, can result in rather low γ-biodiversity compared to other silvicultural systems like regular shelterwood (Schall et al. 2020).
Short rotation and coppice forestry
(Modern) short rotation and (historical) coppice forestry both describe management practices, where trees are harvested quite regularly, with interventions every 2–20 years (e.g. for short rotation plantations according to Lindegaard et al. 2016) or every 5–30 years (for European coppice stands according to Nicolescu et al. 2017). Yet, there are large differences between the two management methods: Modern short rotation has an overly strong focus on fast growing wood production, for which reason short rotation areas are not considered as forests in some countries (= Exclusion or even extinction concept, Fig. 1). Historical coppice forestry, in contrast, can create habitats for highly endangered species whilst not being of economic interest. They are, therefore, nowadays only kept out of conservation interest (= segregative concept, Fig. 1). Modern short rotation plantations are often planted in dense rows for maximizing the yield. Plantation fields are either cut down as a whole or in blocks, making the blocks their self even-aged. Typical short rotation forestry trees are eucalyptus, poplar and willow. In contrast, coppice forestry (also called traditional coppicing or “Niederwald”) describes a traditional forest management, where trees are cut selectively or in small parcels to grow again from stock shoots. The arrangement of cuts is normally very small scaled, resulting in an uneven-aged and heterogeneous stock. Typically, a traditional coppice forest is characterized by a diverse herb layer and by a well-developed shrub layer, consisting of stock shoots from the target trees (e.g. hornbeam, linden or hazel) as well as of accompanying, light-demanding species of early successional forest stages, like rowanberry, service tree, ash, birch and whitebeam. Despite these obvious differences, literature often does not discriminate between traditional coppice and short rotation plantations. For example, Duncker et al. (2012), did not mention traditional management regimes like coppicing, summing up coppice management and plantations together within “Intensive short rotation forestry”. Both management practices are, therefore, regarded as relatively intensive management strategy, where frequent disturbances hinder the establishment of stable biotic communities (Vanbeveren and Ceulemans 2019).
Short rotation forestry most likely has negative effects on biodiversity, when plantations are situated in forest landscapes (Vanbeveren and Ceulemans 2019). As a consequence, short rotation forestry is in some countries like Germany only allowed on agricultural areas and not within forest sites. Yet, some studies advocate for the consideration of short rotation forestry as part of the sustainable transformation of the energy market and enhanced energy security (Lindegaard et al. 2016; Quinkenstein and Jochheim 2016; Verlinden et al. 2015). Conversion of agricultural areas to short rotation plantations might even have positive effects on the local water cycle (Wahren et al. 2015) and biodiversity (Müller-Kroehling et al. 2020; Vanbeveren and Ceulemans 2019). Finally, they might also be the right management choice for agricultural areas at forest edges, which might help to buffer edge effects such as nitrogen influx and pesticide drift from arable fields (Remy et al. 2016; Weathers et al. 2001) as both—nitrogen and pesticides—can have negative effects on forest biodiversity (De Vries and Schulte-Uebbing 2019; Gove et al. 2007; Kurze et al. 2018).
Traditional coppice management might, compared to other forest management systems, mark the oldest management practice, as it is believed to have been practiced even in the prehistoric age (Kirby et al. 2017). With its long history and therefore the persistence of that specific forest type, specific species communities have developed inside coppice forest sites (Kirby et al. 2017). Rare and endangered species like the scarce fritillary (Euphydryas maturna) were found to depend on coppice forest stands and the associated structural attributes (Freese et al. 2006). Yet, also other species that are normally characteristic for early successional stage forests can be found in traditional coppice stands (Buckley 2020). The abandonment of traditional coppice areas mainly results in the disappearance of red-listed species and therefore in a drop of the forests conservation value (Müllerová et al. 2015). Therefore, maintaining or even reestablishing traditional coppice forestry, though also being regarded as “intensive management system” (Duncker et al. 2012), can provide valuable habitats for a variety of different species and therefore are considered a chance for biodiversity conservation (Vacik et al. 2009). For this reason, traditional coppice stands are nowadays protected in many countries.
For both, short rotation and traditional coppice management, there are no studies on γ-biodiversity, as both are normally only applied on the small scale. Yet, the establishment of short rotation stands within agricultural landscapes is believed to positively affect landscape-scale heterogeneity and therefore γ-diversity (Müller-Kroehling et al. 2020). With more woody structures in agricultural areas, connectivity between forest areas might be ameliorated and habitats especially for forest edges species might be supplied (Müller-Kroehling et al. 2020). Yet, this might especially true for coppice stands with longer rotation periods (10 years and more).
Coppice with standards and other open forest management systems
Apart from the widely used forest management systems that result in a mainly closed canopy (e.g. selection cutting or continuous cover shelterwood), there are also some traditional managements that promote an open canopy structure and are nowadays mainly maintained due to their importance for biodiversity (Segregative concept for biodiversity conservation; Fig. 1). An exception are wood pastures, as these are, in some countries, no more allowed in forested ecosystems and therefore need extra permission to be reintroduced again for biodiversity reasons (Exclusive concept, Fig. 1). Open forests once were created by combined use. Coppice with standards as an example was once a widely applied forest management system, which resulted in open canopy forests with rich shrub vegetation and some very old trees in between. Wood pasture similarly results in forest gaps and open canopies as here large herbivores are thinning the understory vegetation. These habitat types are recognized as biodiversity hotspots in Central Europe (Horák et al. 2018; Miklín and Čížek 2014; Sebek et al. 2015), yet, they have vanished in many areas, as these traditional managements cannot cope with modern forest management systems when it comes to economical gain. Remaining open forest areas are threatened by nutrient input, non-native species, and natural succession processes (Chudomelová et al. 2017).
The open canopy structure of coppice with standards and wood pasture areas result in a species-rich herb and shrub layer. Plant diversity as well as functional divergence here are promoted (Chelli et al. 2021; Šebesta et al. 2017), yet the gaps in the canopy do also lead to warmer temperatures, which is assumed to make open forests more prone to climate change (Zellweger et al. 2020). However, the claims of Zellweger et al. (2020) that a climatic depth in the herb layer is developing, was found to be based on circular reasoning (Schall and Heinrichs 2020). Forest understory plants, therefore, seem to be well adapted to microclimatic changes due to canopy openings and rather benefit from more light availability than to struggle with increased temperatures (Schall and Heinrichs 2020). With the high diversity in plant species, open forests provide also diverse niches for higher trophic levels. Many endangered insect species are dependent on open forest structures, like the red-listed butterflies Euphydryas maturna and Lopinga achine (Bergman 2001; Freese et al. 2006) as well as specialized birds (Zasadil et al. 2020). The positive effects of open forest management on biodiversity can not only be found on the short term, but last for many years, so that positive effects of former open forest management can still be found long time after the management was abandoned. Miklín et al. (2018) found that endangered beetle species, associated with veteran trees, can still often be found in forests with open forest history, 71 years after the abandonment of the traditional management. This, however, might also indicate, that there is an extinction dept within these former open forest sites.
It is difficult to evaluate how coppice with standards and other open forest systems would affect biodiversity on a landscape scale, as open forests nowadays are rare habitat types that are only persistent in small areas. Yet, some theories exist, stating that the natural vegetation of Central Europe once was a park-like landscape with open areas and woody vegetation mixed up (Vera et al. 2006). As a result, most Central European species should be adapted to this kind of habitat, resulting in high γ-diversity throughout open forest landscapes. In fact, Vollmuth (2021) describes that the high biodiversity of many German beech forests is likely due to the former open forest management and should not be seen as evidence that closed-canopy forestry is good for biodiversity. The high biodiversity might be the result of an extinction dept, posing a challenge to future efforts to halter biodiversity loss. Also in mediterranean forest ecosystems, open forest areas are considered especially biodiversity rich (Bagella et al. 2020; Torralba et al. 2016), while ongoing succession due to the abandonment of traditional management leads to severe biodiversity loss (Micó et al. 2022; Otero et al. 2015). Extensive management systems, therefore, should at least be reintroduced and promoted in forests, where biodiversity conservation is the main objective (Horák et al. 2018; Miklín and Čížek 2014).
Additional forest management strategies
While the above mentioned forest management systems precisely give guidelines on how to manage a forest stand particularly at the local scale, there are also other forest management strategies that are more focused on the landscape scale. These strategies are independent of the local forest management system and, therefore, can often be applied in coherence with the focal management to support biodiversity conservation (Lindenmayer et al. 2006). The following chapters will give an overview of these strategies: Retention forestry and close-to-nature management have long been applied to promote key habitat structures within forests. These strategies often act on an integrative basis. Thus, the aim is to enrich structures within the management system by retaining single trees or smaller groups of trees. Another possibility would be the segregative concept with the aim to manage forests for wood supply, but also create forest reserves for biodiversity. Either way, there is urgent need to prepare our forests for future challenges such as climate change. Disturbance regimes here might not only be seen as threat to constant wood supply, but also as a chance to create resilient forests and heterogeneous landscapes.
Retention forestry and active deadwood enrichment
One integrative strategy to counteract forest simplification and to promote key structures is the so called retention forestry (Franklin 1989). It was introduced 1989 in North America, and aims to retain key structural elements like veteran and hollow trees, as well as large-volume standing and lying deadwood. All these structures are rarely found in managed forests and therefore need to be promoted (Gossner et al. 2013; Lindenmayer et al. 2006). By doing so, biodiversity (Gossner et al. 2016; Seibold et al. 2015; Tomao et al 2020; Wetherbee et al. 2021) as well as biodiversity related ecosystem services (Wetherbee et al. 2020) can be effectively supported. At the moment, 5–10 habitat trees per hectare (including standing deadwood) are suggested to preserve biodiversity (Kraus and Krumm 2013). In addition, the amount of deadwood needs to be increased in most European managed forests above the current average of 10 m3/ha, providing also areas with significantly larger volumes (50 m3/ha and more) (depending on the respective forest type; see Uhl et al. 2022a). The taxonomic and structural heterogeneity of deadwood objects hereby is as important as the amount itself. The more different structures (standing and lying deadwood, sun-exposed and shaded deadwood, deadwood of different tree species) are enriched, the more diverse species groups can be promoted (Gossner et al. 2016; Uhl et al. 2022b; Vogel et al. 2020). At the stand level, setting aside key structures like veteran trees and deadwood can promote biodiversity to levels comparable with those of primary forests (Mori and Kitagawa 2014).
When retention forestry is applied, important structures like deadwood and old trees are retained within the forest stand. The so created “deadwood or old tree islands” can serve as stepping stones within managed forest stands and between protected forest patches and therefore are believed to help biodiversity on a landscape scale (Gustafsson et al. 2012). The set-aside structures can, in addition, serve as “lifeboat” for species that are dependent on old grown and deadwood structures, enabling the persistence of such species within a managed forest landscape (Gustafsson et al. 2012; Komonen and Müller 2018). While retention forestry has well-studied positive effects on biodiversity, it has nearly no impact on the financial value of a managed forest (Augustynczik et al. 2018).
While deadwood retention focuses on the passive enrichment of deadwood, by keeping dying and dead trees in a forest stand, there are also newer, integrative approaches to actively enrich or even create deadwood. A combination of both, passive deadwood retention and actively introducing tree senescence (e.g. via girdling or cutting the tree crown) is believed to create diverse deadwood resources, which in turn effectively promotes saproxylic diversity in forests (Vítková et al. 2018). One possibility to integrate active deadwood enrichment in forest management would be the girdling or crown removing of low quality trees, instead of removing the whole tree during thinning. The active enrichment of deadwood can be an opportunity to reintroduce otherwise critically endangered or even extinct deadwood-dependent species (Nordén et al 2013). There are, as an example, several approaches to reintroduce endangered fungi on actively enriched deadwood structures (Heilmann-Clausen et al. 2017; Nordén et al. 2020). Deadwood enrichment, therefore, is not only an effective tool to promote the existing biodiversity of a forest, but also to reestablish focal species that otherwise might go extinct within the next decades.
Close-to-nature forestry
Close-to-nature forestry is nowadays the most widely applied, integrative forest management strategy in Central Europe (Bürgi 2015). The terms close-to-nature forestry or near-to-nature forestry both describe the same principle, where the aim is to manage a forest for wood production by simultaneously keeping its multifunctionality (referring to an all-encompassing, inclusive concept, Fig. 1). In fact, close-to-nature forestry is one term for a globally observable phenomenon, where conventional silvicultural systems are rethought to create multifunctional forest ecosystems (Puettmann et al. 2015). While close-to-nature forestry is originating from Europe, similar approaches in other parts of the world are called “New Forestry”, “Natural disturbance based management”, “Ecological forestry”, “Nature oriented” or “Multi-purpose forestry” (see Puettmann et al. 2015 for details). However, these terms are not directly linked to a specific management system, although various systems like continuous cover selection cutting, group selection and irregular shelterwood systems are mentioned in connection with close to nature forestry (Brang et al. 2014; Mason et al. 2022; Schütz et al. 2016).
In fact, in close-to-nature forestry, the goal to achieve a multifunctional, heterogeneously structured forest is more important than the management system, which is implemented to reach this goal (Schütz et al. 2016). Puettmann et al. (2015) summarizes five silvicultural principles that can be found for all close-to-nature approaches: Applying only partial harvests, promoting structural diversity and natural regeneration, creating mixed species stands and avoiding intensive operations (Fig. 2). Often, mimicking natural disturbances is set as a guideline to manage forests in a biodiversity friendly way (viz. “Natural dynamics sylviculture”, following Aszalós et al. 2022). However, the goal of “mimicking natural disturbances” is far from the actual management, as none of the management practices considered in close-to-nature forestry is able to resemble natural disturbance dynamics (Aszalós et al. 2022; Bässler et al. 2014; Schütz et al. 2016). Natural forest dynamics take place over long time periods with the oldest trees reaching 300–400 years of age, e.g., in mountain beech and fir forests. In managed forests, these dynamics are shortened by 2.7–3.6 times, resulting in a rotation length of approximately 120 years (Schütz et al. 2016).
As a consequence, large forest areas are artificially kept in a stable mid-optimum stage, where wood production is highest, but similarly the diversity of many species is lowest, compared to other forest successional stages (Hilmers et al. 2018). Old growth forest species, that are dependent on trees reaching a minimum threshold age of 100–220 years are at a clear disadvantage here and cannot persist in such managed forests (Moning and Müller 2009; Nagel et al. 2017). The large scale implementation of close-to-nature forestry (viz. selection cutting), therefore, results in homogenized landscapes with perpetual mid-optimum stage forests and neglects the importance of other forest structures/successional stages for biodiversity. The promoted high within-stand heterogeneity hereby cannot compensate for the rather low landscape-scale heterogeneity, resulting in low γ-multi-diversity (Schall et al. 2018).
Based on these shortcomings, the EU redefined the principles of close-to-nature forestry and extended the concept under the umbrella term “closer-to-nature forest management”, in order to reach the goal set by the EU Forest Strategy 2030 (European Commission 2021; Larsen et al. 2022). The features with which stand structural heterogeneity can be promoted are formulated more clearly under the “closer-to-nature” approach: The retention of habitat trees and deadwood, as well as the promotion of native tree species and site-adapted species gives clear advice how to enhance structural heterogeneity in the forest (Fig. 2). In addition, not only stand structural diversity is promoted, but also landscape level heterogeneity and special habitat types. The closer-to-nature approach, therefore, does not implement the use of one particular suited and sustainable management type to promote a continuous cover, but promotes landscape-scale heterogeneity and, therefore, diversity in management approaches (Larsen et al. 2022) (Fig. 2).
Comparison of the silvicultural principles under close-to-nature forestry (following Puettmann et al. 2015, left) and under closer-to-nature forestry (following Larsen et al. 2022, right). Light green background indicates redundant principles that can be found in both concepts (linked through an arrow). Lime green background indicates principles, that were added/extended under the closer-to-nature principle. The retention of habitat trees and deadwood (closer-to-nature) is partly represented by “structural diversity” (close-to-nature), yet, the inclusion of special habitats is a new aspect that can only be found in closer-to-nature principles
Forest reserves
While sustainable forest management is crucial for biodiversity conservation on the large scale, some high quality forest habitats also need to be protected (Segregative concept for biodiversity conservation, Fig. 1). The EU biodiversity strategy states that primary and old-growth forests should be strictly protected to halt biodiversity loss (European Commission 2020). Yet, only 46% of the of Europe’s primary forests (which themselves make up only 0.7% of the European forest cover) are protected, showing that there is still a long way to go until valuable old-growth forests are sufficiently protected (Sabatini et al. 2018). Some forest types are known for their sensitivity to (anthropogenic) disturbance. Mountain forests, as an example are already facing environmental pressure due to climate change (Albrich et al. 2020) and eutrophication (Mauchamp et al. 2016; Ruiz-Labourdette et al. 2012). Hygrophilous forests, like riparian forests and swamp forests likewise are sensitive to environmental change (Havrdová et al. 2023). As these forest types also serve as habitat for many endangered species, they have a high conservation value and therefore should be prioritized when forest reserves are established.
Especially in old-growth forests, non-intervention approaches are required to not disrupt natural dynamics. Forest national parks here play an important role. With their main principle “benign neglect”, national parks can give insight into natural succession (Hilmers et al. 2018) and disturbance dynamics (Heurich et al. 2010; Müller et al. 2008, 2010; Sommerfeld et al. 2020). Abandoning management and therefore allowing natural forest succession, can gradually increase biodiversity (Langridge et al. 2023; Paillet et al. 2010). Yet, these positive effects might not be visible immediately, as succession processes take decades of time (Hilmers et al. 2018). In fact, some studies show that in an initial phase of secondary forest succession, canopy closure can even lead to a loss in species, and only after this initial phase, positive effects of management abandonment occur (Braunisch et al. 2019). After approximately 200 years of natural succession (depending on the initial situation and constitution of the forest), forests enter the plenter and later the terminal stage of succession, with many old grown trees, high amounts of deadwood and species rich communities (Hilmers et al. 2018; Tomao et al. 2020). Deadwood is a key component of forest ecosystems, and especially large logs in advanced stages of decay are crucial for the protection of specialized and endangered saproxylic species (Hagge et al. 2021; Thorn et al. 2020). In large unmanaged reserves, hands off strategies result not only in old grown plenter/terminal stages, but also enable natural disturbance dynamics. Storm and fire events, diseases and drought can result in large areas of decaying trees. The developing open forest habitats of the decay, gap and regeneration stage serve as habitat for multiple species (Müller et al. 2008). Likewise, these succession stages are underrepresented in most managed forest (Hilmers et al. 2018), as here, disturbance events result in massive economic losses and therefore are either preventively avoided, or deadwood is cleared and young trees are replanted soon after the disturbance event to compensate for the economic losses and to rehabilitate the forest, following national reforestation guidelines. Forest reserves therefore are urgently needed to preserve especially late successional and early successional stages.
Forest conversion in times of climate change
Integrative and segregative strategies to support biodiversity are undoubtedly important to reach the goals set by the European Biodiversity Strategy. Yet, new challenges are arising that force us to take action, as our forests will locally and regionally be affected by climate change (Albrich et al. 2020; Lindner et al. 2014; Venäläinen et al. 2020). Forests are changing continuously due to natural succession and disturbance processes, elucidating that forests are not stable, but dynamic systems. Also natural disturbances need to be considered as a part of the forest systems and, therefore, need to be tolerated. Storm events, mass occurrences of herbivorous insects, or bark beetle outbreaks, all of which will amplify with ongoing climate change (Dale et al. 2001; Sommerfeld et al. 2020), are often only considered under economic aspects, and hence regarded as threats to timber yields (FOREST EUROPE 2020). Yet, such disturbances can positively affect structural and compositional heterogeneity and promote biodiversity (Müller et al. 2008, 2010), for which reason they should be considered a part of sustainably managed forests. The intensity of salvage logging therefore could be reduced, with at least some low-quality deadwood structures remaining in disturbed areas or even leaving some areas unprocessed. Large disturbance areas can also be seen as a chance to diversify a forest stand. A bark beetle infested spruce forest e.g. could be diversified by dividing disturbance areas into different zones: One quarter with replanted deciduous trees, one natural succession zone with pioneer species such as birch and rowanberry, one natural succession zone with spruce, and one with drought tolerant species such as Douglas fir.
In fact, the promotion of mixed species stands or stands of different species gives foresters the opportunity to spread risks of forest damage (Sebald et al. 2021). With different tree species growing within a forest stand, potential disturbances such as pest outbreaks but also drought events do not affect all trees similarly. For European beech forests, co-occurring species like service tree, field maple or even fir can be promoted with the right forest management system, as these species are relatively drought tolerant and therefore might withstand climate change (Hemery et al 2010; Koch et al. 2022; Tinner et al. 2013). Another approach would be the introduction of non-native trees. Foresters constantly are looking for “new” tree species that can better cope with the altered climatic conditions (Pötzelsberger et al. 2020). But as exotic trees are not a part of native ecological networks, their potential impact on local biodiversity always has to be investigated carefully before the species is introduced.
For biodiversity, the tree species composition of a forest surely is crucial. A forest consisting of native species is usually the best solution, as neophytic trees often are useless for specialized organisms (Vogel et al. 2021) and can lower predator diversity (Schuldt and Scherer-Lorenzen 2014). As an example, the richness of southern European understory plants is negatively affected as soon as the exotic tree-of-heaven (Ailanthus altissima) is introduced (Montecchiari et al. 2020). Similar effects can be seen when Red Oak (Quercus rubra) is introduced in Central European forests (Woziwoda et al. 2014). Also for fungi, non-native tree species can have negative effects, reducing the relative abundance of some functional groups (Likulunga et al. 2021) and being a deadwood resource only for a small fraction of saproxylic fungi (Schmid et al. 2014). For forest insects, mixtures with non-native tree species can alter community composition, which might undermine efforts to preserve especially rare and specialized species (Kriegel et al. 2021). Yet, some studies also hint that not all exotic tree species show negative effects on biodiversity. Non-native Douglas fir (Pseudotsuga menziesii) stands in Europe, as an example, have been found to be inhabited by communities that are quite similar to native forest stands (Glatthorn et al. 2023). Douglas firs are accepted by many herbivores as alternative to native conifer trees (Schmid et al. 2014) and Douglas fir deadwood is inhabited by diverse saproxylic communities (Purahong et al. 2018). In addition, the admixture of Douglas fir within pure beech stands has been found to even promote understory plant species richness (Bärmann et al. 2023). The effect of introduced tree species, therefore, might depend on the phylogenetic distinctiveness between the introduced and the native tree species. The introduction of non-native tree species hence might be a solution to create resilient forests in times of climate change. But the decision on whether to introduce a species or not should always be based on extensive scientific research and only be used when other options to create resilient forests resembling the natural vegetation of a region can no longer be uphold.
Outlook: finding the best management combinations for biodiversity
Our review revealed high redundancy among commonly used forest system terms, which makes it difficult to get an overview on the effects of management on biodiversity. Also, there are some basic knowledge gaps concerning forest management and biodiversity. Effects on β- and γ- diversity are rarely investigated. Especially for irregular shelterwood systems and traditional management systems, such as coppice and open forest management, no analyses on β- and γ-diversity are available to the best of our knowledge. Insights from studies focusing on even-aged (regular shelterwood) vs. uneven-aged (selection cutting) systems have shown that especially landscape-scale heterogeneity can be crucial for biodiversity conservation. In this context, clearcuts seem to pose a chance to increase β- and subsequent γ-diversity, although this system has long been seen as intensive and biodiversity-unfriendly management system.
However, an all-in-one, inclusive concept, presenting one management system, that helps manage forests for sustainable wood supply and simultaneously promotes biodiversity, does not exist. Instead, multiple recent studies came to the conclusion that a combination of different “biodiversity friendly” management systems seem the best approach to increase stand- and landscape-scale heterogeneity (Himes et al. 2022; Kuuluvainen 2002; Murray et al. 2017; Nolet et al. 2017; Rodríguez et al. 2019; Schall et al. 2018; Sing et al. 2018). Habitat heterogeneity is linked to biodiversity, as the diversity in ecological niches promotes the establishment of a species-rich community (Heidrich et al. 2020; Tews et al. 2004), which in turn can also promote forest resilience (Messier et al. 2022) on the stand and landscape scale. Therefore, the main objective of forest biodiversity conservation should be a management to create compositionally and structurally rich forests (Felipe-Lucia et al. 2018). “Compositionally and structurally rich” hereby should not only be considered important on the local level, but also on the landscape level.
On the local level, the composition of different tree species can be crucial (Felton et al. 2010). Choosing a suitable dominant tree species is determinant for the subsequent forest type and can orientate on the natural vegetation of a region, which is determined by the local abiotic conditions (Bohn and Wels 2004). Yet, also non-native species which complement natural vegetation in their resource requirements can be included, as long as they have proven to be non-invasive and useful also for native biota. The dominant tree species (e.g., beech, spruce, pine) of a forest can serve as ecosystem engineer and create a specific forest type (e.g., beech forests, spruce forests, pine forests) with a typical microclimate and providing habitat for specialized communities. The according management system can support the creation of different forest types (Fig. 3). Shade tolerant forest trees like beech can be promoted by continuous cover shelterwood or by selection cutting, while light demanding trees like oak might benefit from irregular shelterwood, gap cutting and open forest management. Within the different forest types, the thinning grade could additionally be varied keeping some patches closed while opening others and even incorporating gaps (Fig. 3). Doing so, also different amounts of co-occurring species can be promoted, resulting in heterogeneous stands with different forest types and heterogeneous forest types with different amounts of co-occurring species. By mixing co-occurring tree species to create multi-species stands, understory plant diversity (Gong et al. 2021), forest resilience (Jactel et al. 2017; MacLean und Clark 2021) and forest productivity can be enhanced (Jactel et al. 2018; Liang et al. 2016), with trees growing faster as soon as they are surrounded by different species (Fan et al. 2020; Liang et al. 2016). The occurrence of different tree species also builds up the basis for diverse species communities of higher trophic levels, as many forest species are host specific (Brändle and Brandl 2001). To further promote biodiversity on the local scale, retention forestry and other forms of deadwood and old tree enrichment should be considered at least in parts of the forest stands. By integrating these management system independent methods, the amount of key structural elements like deadwood and veteran trees can further be enhanced. Artificially creating or enriching deadwood might be an additional opportunity to create diverse deadwood structures (Vítková et al. 2018). Such deadwood enrichment methods can, in addition, be used when rare saproxylic species are to be reintroduced in areas with conservation goal (Nordén et al. 2020).
Schematic overview showing how horizontal and vertical forest heterogeneity can be influenced by different forest management systems. Horizontal heterogeneity, which also refers to the “grain size” of disturbance/cutting areas, is defined by the thinning level, which in turn determines which tree species (shade tolerant vs. light demanding species) are to be promoted. Vertical heterogeneity describes the local structural configuration of a forest, which is strongly dependent on how forests are managed
On the landscape scale, there are three different levels, which can be used to create heterogeneity: The forest type level, the succession, and the thinning level (Fig. 4). A mix of various forest types, e.g. beech and oak, pine and fir forests can be essential to increase γ-diversity (Heinrichs et al. 2019). Also here, the establishment of different forest types is always dependent on the soil and macroclimatic conditions. Within the forest types, different succession stages should be present. In natural, old-grown forests, high structural heterogeneity among forest patches is achieved by natural succession dynamics, which are separated into nine stages: the gap stage, then regeneration, establishment, early optimum, mid-optimum, late optimum, plenter, terminal, and finally the decay stage. With different management systems in managed forests, the different succession stages can artificially be created, helping to provide habitats for diverse species groups that normally can be found within natural forests underlying forest succession (Fig. 5). While continuous cover, selection cutting systems favor the establishment of mid- to late-optimum forest stages, clearcuts and expanding gap shelterwood systems can help to create forest gaps and early successional stages. Regeneration stage forests can be created by applying regular and extended shelterwood systems. Traditional coppice systems can resemble the establishment phase of a forest (Fig. 5). To protect late successional stages, forest nature reserves are essential (Fig. 5). Open forest habitats, as achieved by traditional systems such as coppice with standard and wood pasture, might additionally serve as substitute for terminal stage forests, where few old grown trees are still present, but light availability is high (Fig. 5). For example, an oligotrophic to mesotrophic beech forest, which is a quite common forest type for central Europe, might profit from selective cutting as young, shade-tolerant beech trees can then grow sheltered under a closed canopy (Fig. 3, Table 1). Beech forests that are managed this way resemble the optimal stage. Gap-cuttings, or irregular shelterwood approaches can further be used to enhance the proportion of co-occuring tree species like oak, and promote a species rich understory at some places. These stands will resemble the gap stage within a beech forest (Fig. 4). Unmanaged patches (either smaller fractions from retention forestry or larger forest reserve areas) would finally be needed to enhance the proportion of old and hollow trees as well as the proportion of deadwood, similar to the structure of an old grown, plenter stage beech forest (Fig. 4). Within the successional stages, different levels of thinning can finally be used to promote different proportions of co-occuring species. Patches of low level thinning will promote shade-tolerant beech regrowth on the one hand, and high intensity thinning patches will promote light demanding, co-occurring species on the other hand (Fig. 4). In total, there are five different thinning levels, which can be mixed to create heterogeneity within the succession stages of the different forest types (Pretzsch 2009).
Theoretical framework of how to create heterogeneous forest landscapes, implementing three levels of heterogeneity creation: 1 Mix of different forest types, which might demand different forest management systems, 2 Mix of different succession stages, which can artificially be created by different management systems, 3 Variance in regeneration mode, thinning, retention, and gap size
Focusing on a diversity-in-management-approach gives foresters many possibilities to create heterogeneous, managed forest landscapes. There might be not only multiple ecological niches created, but also many stepping stones and refuges for habitat specialists. A forest gap within an irregular shelterwood forest hereby can serve as a stepping stone for open forest and forest edge species (Kozel et al. 2021), while e.g. tree retention after a clearcut can help forest species, such as cavity nesting birds (Lindbladh et al. 2022). Apart from the positive effects on biodiversity, the diversity in different tree species and forest types also strenghens forest resilience and builds up a natural insurance against future challenges such as climate change (Sebald et al. 2021). With these heterogeneous forest landscapes, which not only are characterized by high structural heterogeneity on a local level, but also a diverse structure on the landscape level, forest biodiversity and ecosystem function might be most effectively preserved.
Data availability
No datasets were generated or analysed during the current study.
References
Albrich K, Rammer W, Seidl R (2020) Climate change causes critical transitions and irreversible alterations of mountain forests. Glob Change Biol 26(7):4013–4027. https://doi.org/10.1111/gcb.15118
Aszalós R, Thom D, Aakala T et al (2022) Natural disturbance regimes as a guide for sustainable forest management in Europe. Ecol Appl 32(5):e2596. https://doi.org/10.1002/eap.2596
Augustynczik ALD, Gutsch M, Basile M, Suckow F, Lasch P, Yousefpour R, Hanewinkel M (2020) Socially optimal forest management and biodiversity conservation in temperate forests under climate change. Ecol Econ 169:106504. https://doi.org/10.1016/j.ecolecon.2019.106504
Augustynczik ALD, Yousefpour R, Rodriguez LCE, Hanewinkel M (2018) Conservation costs of retention forestry and optimal habitat network selection in southwestern Germany. Ecol Econ 148:92–102. https://doi.org/10.1016/j.ecolecon.2018.02.013
Bagella S, Caria MC, Seddaiu G, Leites L, Roggero PP (2020) Patchy landscapes support more plant diversity and ecosystem services than wood grasslands in Mediterranean silvopastoral agroforestry systems. Agric Syst 185:102945. https://doi.org/10.1016/j.agsy.2020.102945
Barbati A, Marchetti M, Chirici G, Corona P (2014) European Forest Types and Forest Europe SFM indicators: tools for monitoring progress on forest biodiversity conservation. For Ecol Manage 321:145–157. https://doi.org/10.1016/j.foreco.2013.07.004
Bärmann L, Kaufmann S, Weimann S, Hauck M (2023) Future forests and biodiversity: Effects of Douglas fir introduction into temperate beech forests on plant diversity. For Ecol Manage 545:121286. https://doi.org/10.1016/j.foreco.2023.121286
Bässler C, Ernst R, Cadotte M, Heibl C, Müller J (2014) Near-to-nature logging influences fungal community assembly processes in a temperate forest. J Appl Ecol 51(4):939–948. https://doi.org/10.1111/1365-2664.12267
Bergman KO (2001) Population dynamics and the importance of habitat management for conservation of the butterfly Lopinga achine. J Appl Ecol 38(6):1303–1313. https://doi.org/10.1046/j.0021-8901.2001.00672.x
Bergmeier E, Petermann J, Schröder E (2010) Geobotanical survey of wood-pasture habitats in Europe: diversity, threats and conservation. Biodivers Conserv 19:2995–3014. https://doi.org/10.1007/s10531-010-9872-3
Bertolini V, Damon A, Mora JV, Rojas Velázques AN (2012) Distribution and ecological patterns of orchids in Monte Pelegrino Reserve, Palermo (Sicily, Italy). Biodivers J 3:375–384
Blanco V, Brown C, Rounsevell M (2015) Characterising forest owners through their objectives, attributes and management strategies. Eur J Forest Res 134:1027–1041. https://doi.org/10.1007/s10342-015-0907-x
Blixt T, Bergman K, Milberg P, Westerberg L, Jonason D (2015) Clear-cuts in production forests: From matrix to neo-habitat for butterflies. Acta Oecologica 69:71–77. https://doi.org/10.1016/j.actao.2015.09.006
Bohn U, Wels W (2004) Die potenzielle natürliche Vegetation. Nationalatlas Bundesrepublik Deutschland—Klima, Pflanzen- und Tierwelt: pp 84–87. https://archiv.nationalatlas.de/wp-content/art_pdf/Band3_84-87_archiv.pdf
Boncina A (2011) Conceptual approaches to integrate nature conservation into forest management: a Central European perspective. Int Rev 13(1):13–22. https://doi.org/10.1505/146554811798201152
Borrelli P, Panagos P, Märker M, Modugno S, Schütt B (2017) Assessment of the impacts of clear-cutting on soil loss by water erosion in Italian forests: first comprehensive monitoring and modelling approach. CATENA 149:770–781. https://doi.org/10.1016/j.catena.2016.02.017
Brändle M, Brandl R (2001) Species richness of insects and mites on trees: expanding Southwood. J Anim Ecol 70(3):491–504. https://doi.org/10.1046/j.1365-2656.2001.00506.x
Brang P, Spathelf P, Larsen JB et al (2014) Suitability of close-to-nature silviculture for adapting temperate European forests to climate change. Forestry 87(4):492–503
Braunisch V, Roder S, Coppes J, Froidevaux JSP, Arlettaz R, Bollmann K (2019) Structural complexity in managed and strictly protected mountain forests: effects on the habitat suitability for indicator bird species. For Ecol Manage 448:139–149. https://doi.org/10.1016/j.foreco.2019.06.007
Brunet J, Fritz Ö, Richnau G (2010) Biodiversity in European beech forests - a review with recommendations for sustainable forest management. Ecol Bull 53:77–94
Buckley P (2020) Coppice restoration and conservation: a European perspective. J for Res 25(3):125–133. https://doi.org/10.1080/13416979.2020.1763554
Bürgi M (2015) Close-to-nature Forestry. In: Kirby K, Watkins C (eds) Europe’s changing woods and forests: from wildwood to managed landscapes. CABI, Oxfordshire, pp 107–115
Chelli S, Bricca A, Cutini M, Campetella G, Cervellini M, Tsakalos JL, Canullo R (2021) Large standard trees and deadwood promote functional divergence in the understory of beech coppice forests. For Ecol Manage 494:119324. https://doi.org/10.1016/j.foreco.2021.119324
Chudomelová M, Hédl R, Zouhar V, Szabó P (2017) Open oakwoods facing modern threats: will they survive the next fifty years? Biol Cons 210:163–173. https://doi.org/10.1016/j.biocon.2017.04.017
Cosyns H, Joa B, Mikoleit R, Krumm F, Schuck A, Winkel G, Schulz T (2020) Resolving the trade-off between production and biodiversity conservation in integrated forest management: comparing tree selection practices of foresters and conservationists. Biodivers Conserv 29:3717–3737. https://doi.org/10.1007/s10531-020-02046-x
Dahlgren RA, Driscoll CT (1994) The effects of whole-tree clear-cutting on soil processes at the Hubbard Brook Experimental Forest, New Hampshire, USA. Plant Soil 158:239–262. https://doi.org/10.1007/BF00009499
Dale VH, Joyce LA, McNulty S et al (2001) Climate change and forest disturbances: climate change can affect forests by altering the frequency, intensity, duration, and timing of fire, drought, introduced species, insect and pathogen outbreaks, hurricanes, windstorms, ice storms, or landslides. Bioscience 51(9):723–734. https://doi.org/10.1641/0006-3568(2001)051[0723:CCAFD]2.0.CO;2
de Groot M, Eler K, Flajšman K, Grebenc T, Marinšek A, Kutnar L (2016) Differential short-term response of functional groups to a change in forest management in a temperate forest. For Ecol Manage 376:256–264. https://doi.org/10.1016/j.foreco.2016.06.025
de Vries W, Schulte-Uebbing L (2019) Impacts of nitrogen deposition on forest ecosystem services and biodiversity. In: Schröter M, Bonn A, Klotz S, Seppelt R, Baessler C (eds) Atlas of ecosystem services. Springer, Cham
Dieler J, Uhl E, Biber P, Müller J, Rötzer T, Pretzsch H (2017) Effect of forest stand management on species composition, structural diversity, and productivity in the temperate zone of Europe. Eur J Forest Res 136(4):739–766. https://doi.org/10.1007/s10342-017-1056-1
Domínguez G, Shannon M (2011) A wish, a fear and a complaint: understanding the (dis)engagement of forest owners in forest management. Eur J Forest Res 130:435–450. https://doi.org/10.1007/s10342-009-0332-0
Duncker PS, Barreiro SM, Hengeveld GM, Lind T, Mason WL, Ambrozy S, Spiecker H (2012) Classification of forest management approaches: a new conceptual framework and its applicability to European forestry. E&S 17(4):51. https://doi.org/10.5751/ES-05262-170451
Eckelt A, Müller J, Bense U et al (2018) Primeval forest relict beetles of Central Europe: a set of 168 umbrella species for the protection of primeval forest remnants. J Insect Conserv 22(1):15–28. https://doi.org/10.1007/s10841-017-0028-6
Eckerter T, Braunisch V, Pufal G, Klein AM (2022) Small clear-cuts in managed forests support trap-nesting bees, wasps and their parasitoids. For Ecol Manage 509:120076. https://doi.org/10.1016/j.foreco.2022.120076
EEA (2020) State of nature in the EU - Results from reporting under the nature directives 2013–2018. European Environment Agency Technical report No 10/2020. https://doi.org/10.2800/088178
Ekholm A, Lundqvist L, Axelsson EP, Egnell G, Hjältén J, Lundmark T, Sjögren J (2023) Long-term yield and biodiversity in stands managed with the selection system and the rotation forestry system: A qualitative review. For Ecol Manage 537:120920. https://doi.org/10.1016/j.foreco.2023.120920
European Commission (2020) Communication from the commission to the European parliament, the council, the European economic and social committee and the committee of the regions- EU Biodiversity Strategy for 2030. Brussels. https://eur-lex.europa.eu/resource.html?uri=cellar:a3c806a6-9ab3-11ea-9d2d-01aa75ed71a1.0001.02/DOC_1&format=PDF. Accessed 15 March 2024
European Commission (2021): New EU Forest Stragegy for 2030. Communication from the commission to the european parliament, the council, the european economic and social committee and the committee of the regions. Brussels. https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52021DC0572. Accessed 15 March 2024
Fan C, Tan L, Zhang C, Zhao X, Gao L, von Gadow K (2020) Scale-dependent effects of neighborhood biodiversity on individual tree productivity in a coniferous and broad-leaved mixed forest in China. Ecol Evol 10:8225–8234. https://doi.org/10.1002/ece3.6530
Fedrowitz K, Koricheva J, Baker SC et al (2014) Can retention forestry help conserve biodiversity? A meta-analysis. J Appl Ecol 51(6):1669–1679. https://doi.org/10.1111/1365-2664.12289
Felipe-Lucia MR, Soliveres S, Penone C et al (2018) Multiple forest attributes underpin the supply of multiple ecosystem services. Nat Commun 9:4839. https://doi.org/10.1038/s41467-018-07082-4
Felton A, Lindbladh M, Brunet J, Fritz Ö (2010) Replacing coniferous monocultures with mixed-species production stands: an assessment of the potential benefits for forest biodiversity in northern Europe. For Ecol Manage 260:939–947. https://doi.org/10.1016/j.foreco.2010.06.011
Franklin JF (1989) Towards a new forestry. Am for 5:1–8
Freese A, Benes J, Bolz R et al (2006) Habitat use of the endangered butterfly Euphydryas maturna and forestry in Central Europe. Anim Conserv 9(4):388–397. https://doi.org/10.1111/j.1469-1795.2006.00045.x
FOREST EUROPE (2020) State of Europe's forests 2020. https://foresteurope.org/wp-content/uploads/2016/08/SoEF_2020.pdf. Accessed 15 March 2024
Gayer K (1898) Der Waldbau. Verlagsbuchhandlung Paul Parey, Berlin
Glatthorn J, Appleby S, Balkenhol N et al (2023) Species diversity of forest floor biota in non-native Douglas-fir stands is similar to that of native stands. Ecosphere 14:e4609. https://doi.org/10.1002/ecs2.4609
Gong C, Tan Q, Liu G, Xu M (2021) Impacts of tree mixtures on understory plant diversity in China. For Ecol Manage 498:119545. https://doi.org/10.1016/j.foreco.2021.119545
Gossner MM, Lachat T, Brunet J, Isacsson G, Bouget C, Brustel H, Brandl R, Weisser WW, Mueller J (2013) Current near-to-nature forest management effects on functional trait composition of saproxylic beetles in beech forests. Conserv Biol 27(3):605–614. https://doi.org/10.1111/cobi.12023
Gossner MM, Wende B, Levick S, Schall P, Floren A, Linsenmair KE, Steffan-Dewenter I, Schulze E, Weisser WW (2016) Deadwood enrichment in European forests—which tree species should be used to promote saproxylic beetle diversity? Biol Cons 201:92–102. https://doi.org/10.1016/j.biocon.2016.06.032
Gove B, Power SA, Buckley GP, Ghazoul J (2007) Effects of herbicide spray drift and fertilizer overspread on selected species of woodland ground flora: comparison between short-term and long-term impact assessments and field surveys. J Appl Ecol 44(2):374–384. https://doi.org/10.1111/j.1365-2664.2007.01261.x
Gustafsson L, Baker SC, Bauhus J et al (2012) Retention forestry to maintain multifunctional forests: a world perspective. Bioscience 62(7):633–645. https://doi.org/10.1525/bio.2012.62.7.6
Hagge J, Müller J, Birkemoe T et al (2021) What does a threatened saproxylic beetle look like? Modelling extinction risk using a new morphological trait database. J Anim Ecol 90(8):1934–1947. https://doi.org/10.1111/1365-2656.13512
Havrdová A, Douda J, Doudová J (2023) Threats, biodiversity drivers and restoration in temperate floodplain forests related to spatial scales. Sci Total Environ 854:158743. https://doi.org/10.1016/j.scitotenv.2022.158743
Heilmann-Clausen J, Adamčík S, Bässler C, Halme P, Krisai-Greilhuber I, Holec J (2017) State of the art and future directions for mycological research in old-growth forests. Fungal Ecol 27:141–144. https://doi.org/10.1016/j.funeco.2016.12.005
Heidrich L, Bae S, Levick S et al (2020) Heterogeneity–diversity relationships differ between and within trophic levels in temperate forests. Nat Ecol Evol 4(9):1204–1212. https://doi.org/10.1038/s41559-020-1245-z
Heinrichs S, Schmidt W (2009) Short-term effects of selection and clear cutting on the shrub and herb layer vegetation during the conversion of even-aged Norway spruce stands into mixed stands. For Ecol Manage 258:667–678. https://doi.org/10.1016/j.foreco.2009.04.037
Heinrichs S, Ammer C, Mund M et al (2019) Landscape-Scale mixtures of tree species are more effective than stand-scale mixtures for biodiversity of vascular plants, bryophytes and lichens. Forests 10:73. https://doi.org/10.3390/f10010073
Hemery GE, Clark JR, Aldinger E, Claessens H, Malvolti ME, O’connor E, Raftoyannis Y, Savill PS, Brus R (2010) Growing scattered broadleaved tree species in Europe in a changing climate: a review of risks and opportunities. Forestry 83(1):65–81. https://doi.org/10.1093/forestry/cpp034
Heurich M, Beudert B, Rall H, Křenová Z (2010) National Parks as model regions for interdisciplinary long-term ecological research: the Bavarian Forest and Šumavá National Parks underway to transboundary ecosystem research. In: Müller F, Bässler C, Schubert H, Klotz S (eds) Long term ecological research. Dordrecht Springer, Netherlands, pp 327–344
Hilmers T, Friess N, Bässler C, Heurich M, Brandl R, Pretzsch H, Seidl R, Müller J (2018) Biodiversity along temperate forest succession. J Appl Ecol 55(6):2756–2766. https://doi.org/10.1111/1365-2664.13238
Himes A, Betts M, Messier C, Seymour R (2022) Perspectives: thirty years of triad forestry, a critical clarification of theory and recommendations for implementation and testing. For Ecol Manage 510:120103. https://doi.org/10.1016/j.foreco.2022.120103
Horák J, Pavlíček J, Kout J, Halda JP (2018) Winners and losers in the wilderness: response of biodiversity to the abandonment of ancient forest pastures. Biodivers Conserv 27:3019–3029. https://doi.org/10.1007/s10531-018-1585-z
Jactel H, Bauhus J, Boberg J et al (2017) Tree diversity drives forest stand resistance to natural disturbances. Curr for Rep 3:223–243. https://doi.org/10.1007/s40725-017-0064-1
Jactel H, Gritti ES, Drössler L, Forrester DI, Mason WL, Morin X, Pretzsch H, Castagneyrol B (2018) Positive biodiversity-productivity relationships in forests: climate matters. Biol Let 14(4):20170747. https://doi.org/10.1098/rsbl.2017.0747
Jokela J, Siitonen J, Koivula M (2019) Short-term effects of selection, gap, patch and clear cutting on the beetle fauna in boreal spruce-dominated forests. For Ecol Manage 446:29–37. https://doi.org/10.1016/j.foreco.2019.05.027
Kirby KJ, Buckley GP, Mills J (2017) Biodiversity implications of coppice decline, transformations to high forest and coppice restoration in British woodland. Folia Geobot 52:5–13. https://doi.org/10.1007/s12224-016-9252-1
Koch O, de Avila AL, Heinen H, Albrecht AT (2022) Retreat of major European tree species distribution under climate change—minor natives to the rescue? Sustainability 14(9):5213. https://doi.org/10.3390/su14095213
Komonen A, Müller J (2018) Dispersal ecology of deadwood organisms and connectivity conservation. Conserv Biol 32(3):535–545. https://doi.org/10.1111/cobi.13087
Korpela EL, Hyvönen T, Kuussaari M (2015) Logging in boreal field-forest ecotones promotes flowervisiting insect diversity and modifies insect community composition. Insect Conserv Divers 8:152–162. https://doi.org/10.1111/icad.12094
Kovács B, Tinya F, Németh C, Ódor P (2020) Unfolding the effects of different forestry treatments on microclimate in oak forests: results of a 4-yr experiment. Ecol Appl 30(2):e02043. https://doi.org/10.1002/eap.2043
Kozel P, Sebek P, Platek M et al (2021) Connectivity and succession of open structures as a key to sustaining light-demanding biodiversity in deciduous forests. J Appl Ecol 58(12):2951–2961. https://doi.org/10.1111/1365-2664.14019
Kraus D, Krumm F (2013) Integrative Ansätze als Chance für die Erhaltung der Artenvielfalt in Wäldern. European Forest Institute.
Kriegel P, Matevski D, Schuldt A (2021) Monoculture and mixture-planting of non-native Douglas fir alters species composition, but promotes the diversity of ground beetles in a temperate forest system. Biodivers Conserv 30:1479–1499. https://doi.org/10.1007/s10531-021-02155-1
Kurze S, Heinken T, Fartmann T (2018) Nitrogen enrichment in host plants increases the mortality of common Lepidoptera species. Oecologia 188(4):1227–1237. https://doi.org/10.1007/s00442-018-4266-4
Kuuluvainen T (2002) Natural variability of forests as a reference for restoring and managing biological diversity in boreal Fennoscandia. Silva Fennica 36(1):97–125
Langridge J, Delabye S, Gilg O, Paillet Y, Reyjol Y, Sordello R, Touroult J, Gosselin F (2023) Biodiversity responses to forest management abandonment in boreal and temperate forest ecosystems: a meta-analysis reveals an interactive effect of time since abandonment and climate. Biol Conserv. https://doi.org/10.1016/j.biocon.2023.110296
Lanta V, Mudrák O, Liancourt P et al (2019) Active management promotes plant diversity in lowland forests: A landscape-scale experiment with two types of clearings. For Ecol Manag 448:94–103. https://doi.org/10.1016/j.foreco.2019.05.073
Larsen JB, Angelstam P, Bauhs J et al (2022) Closer-to-nature forest management: from science to policy 12. Eur for Inst. https://doi.org/10.36333/fs12
Lettenmaier L, Seibold S, Bässler C, Brandl R, Gruppe A, Müller J, Hagge J (2022) Beetle diversity is higher in sunny forests due to higher microclimatic heterogeneity in deadwood. Oecologia 198(3):825–834. https://doi.org/10.1007/s00442-022-05141-8
Liang J, Crowther TW, Picard N et al (2016) Positive biodiversity-productivity relationship predominant in global forests. Science 354(6309):aaf8957. https://doi.org/10.1126/science.aaf8957
Likulunga E, Rivera Pérez CA, Schneider D, Daniel R, Polle A (2021) Forest tree species composition and abiotic site conditions drive soil fungal communities and functional groups. bioRxiv. https://doi.org/10.1101/2021.07.21.453256
Lindbladh M, Elmberg J, Hedwall PO, Holmström E, Felton A (2022) Broadleaf retention benefits to bird diversity in mid-rotation conifer production stands. For Ecol Manage 515:120223. https://doi.org/10.1016/j.foreco.2022.120223
Lindegaard KN, Adams PW, Holley M, Lamley A, Henriksson A, Larsson S, von Engelbrechten H, Lopez GE, Pisarek M (2016) Short rotation plantations policy history in Europe: lessons from the past and recommendations for the future. Food Energy Secur 5(3):125–152. https://doi.org/10.1002/fes3.86
Lindenmayer DB, Franklin JF, Fischer J (2006) General management principles and a checklist of strategies to guide forest biodiversity conservation. Biol Cons 131(3):433–445. https://doi.org/10.1016/j.biocon.2006.02.019
Lindner M, Fitzgerald JB, Zimmermann NE et al (2014) Climate change and European forests: what do we know, what are the uncertainties, and what are the implications for forest management? J Environ Manage 146:69–83. https://doi.org/10.1016/j.jenvman.2014.07.030
Loconto A, Desquilbet M, Moreau T, Couvet D, Dorin B (2020) The land sparing—land sharing controversy: tracing the politics of knowledge. Land Use Policy 96:103610. https://doi.org/10.1016/j.landusepol.2018.09.014
Lõhmus A (2011) Silviculture as a disturbance regime: the effects of clear-cutting, planting and thinning on polypore communities in mixed forests. J for Res 16(3):194–202. https://doi.org/10.1007/s10310-011-0256-7
MacArthur RH, MacArthur JW (1961) On bird species diversity. Ecology 42(3):594–598. https://doi.org/10.2307/1932254
MacLean DA, Clark KL (2021) Mixedwood management positively affects forest health during insect infestations in eastern North America. Can J for Res 51:910–920. https://doi.org/10.1139/cjfr-2020-046
Mason WL, Diaci J, Carvalho J, Valkonen S (2022) Continuous cover forestry in Europe: usage and the knowledge gaps and challenges to wider adoption. Forestry 95(1):1–12. https://doi.org/10.1093/forestry/cpab038
Matthews JD (1989) Silvicultural systems. Clarendon Press, Oxford
Mauchamp L, Mouly A, Badot P, Gillet F (2016) Impact of nitrogen inputs on multiple facets of plant biodiversity in mountain grasslands: does nutrient source matter? Appl Veg Sci 19:206–217. https://doi.org/10.1111/avsc.12214
MCPFE Expert Level Meeting (2002) MCPFE Assessment guidelines for protected and protective forest and other wooded land in Europe. https://unece.org/fileadmin/DAM/timber/publications/2002-guidelines-protected-forest.pdf. Accessed 15 March 2024
Messier C, Bauhus J, Sousa-Silva R et al (2022) For the sake of resilience and multifunctionality, let’s diversify planted forests! Conserv Lett 15(1):e12829. https://doi.org/10.1111/conl.12829
Micó E, Martínez-Pérez S, Jordán-Núñez J, Galante E, Mico-Vicent B (2022) On how the abandonment of traditional forest management practices could reduce saproxylic diversity in the mediterranean region. For Ecol Manage 520:120402. https://doi.org/10.1016/j.foreco.2022.120402
Miklín J, Čížek L (2014) Erasing a European biodiversity hot-spot: open woodlands, veteran trees and mature forests succumb to forestry intensification, succession, and logging in a UNESCO Biosphere Reserve. J Nat Conserv 22:35–41
Miklín J, Sebek P, Hauck D, Konvicka O, Cizek L (2018) Past levels of canopy closure affect the occurrence of veteran trees and flagship saproxylic beetles. Divers Distribut 24(2):208–218. https://doi.org/10.1111/ddi.12670
Moning C, Müller J (2009) Critical forest age thresholds for the diversity of lichens, molluscs and birds in beech (Fagus sylvatica L.) dominated forests. Ecol Indic 9(5):922–932. https://doi.org/10.1016/j.ecolind.2008.11.002
Montecchiari S, Tesei G, Allegrezza M (2020) Ailanthus altissima forests determine a shift in herbaceous layer richness: a paired comparison with hardwood native forests in sub-mediterranean Europe. Plants 9:1404. https://doi.org/10.3390/plants9101404
Mori AS, Kitagawa R (2014) Retention forestry as a major paradigm for safeguarding forest biodiversity in productive landscapes: a global meta-analysis. Biol Cons 175:65–73. https://doi.org/10.1016/j.biocon.2014.04.016
Müller J, Bußler H, Goßner M, Rettelbach T, Duelli P (2008) The European spruce bark beetle Ips typographus in a national park: from pest to keystone species. Biodivers Conserv 17(12):2979–3001. https://doi.org/10.1007/s10531-008-9409-1
Müller J, Noss RF, Bussler H, Brandl R (2010) Learning from a “benign neglect strategy” in a national park: Response of saproxylic beetles to dead wood accumulation. Biol Cons 143(11):2559–2569. https://doi.org/10.1016/j.biocon.2010.06.024
Müller-Kroehling S, Hohmann G, Helbig C et al (2020) Biodiversity functions of short rotation coppice stands-results of a meta study on ground beetles (Coleoptera: Carabidae). Biomass Bioenerg 132:105416. https://doi.org/10.1016/j.biombioe.2019.105416
Müllerová J, Hédl R, Szabó P (2015) Coppice abandonment and its implications for species diversity in forest vegetation. For Ecol Manage 343:88–100. https://doi.org/10.1016/j.foreco.2015.02.003
Murray BD, Holland JD, Summerville KS, Dunning JB, Saunders MR, Jenkins MA (2017) Functional diversity response to hardwood forest management varies across taxa and spatial scales. Ecol Appl 27:1064–1081. https://doi.org/10.1002/eap.1532
Muscolo A, Bagnato S, Sidari M, Mercurio R (2014) A review of the roles of forest canopy gaps. J for Res 25:725–736. https://doi.org/10.1007/s11676-014-0521-7
Nagel TA, Firm D, Pisek R, Mihelic T, Hladnik D, de Groot M, Rozenbergar D (2017) Evaluating the influence of integrative forest management on old-growth habitat structures in a temperate forest region. Biol Cons 216:101–107. https://doi.org/10.1016/j.biocon.2017.10.008
Nicolescu VN, Carvalho J, Hochbichler E et al (2017) Silvicultural guidelines for European coppice forests. Silviculture 7:46–63. https://doi.org/10.5555/20193063061
Nolet P, Kneeshaw D, Messier C, Béland M (2018) Comparing the effects of even-and uneven-aged silviculture on ecological diversity and processes: a review. Ecol Evol 8(2):1217–1226. https://doi.org/10.1002/ece3.3737
Nordén J, Penttilä R, Siitonen J, Tomppo E, Ovaskainen O (2013) Specialist species of wood-inhabiting fungi struggle while generalists thrive in fragmented boreal forests. J Ecol 101(3):701–712. https://doi.org/10.1111/1365-2745.12085
Nordén J, Abrego N, Boddy L et al (2020) Ten principles for conservation translocations of threatened wood-inhabiting fungi. Fungal Ecol 44:100919. https://doi.org/10.1016/j.funeco.2020.100919
Nyland RD (2002) Silviculture: concept and applications, 3rd edn. Waveland Press, Long Grove
Nyland RD (2010) The shelterwood method: adapting to diverse management objectives. J for 108(8):419–420. https://doi.org/10.1093/jof/108.8.419
Otero I, Marull J, Tello E, Diana GL, Pons M, Coll F, Boada M (2015) Land abandonment, landscape, and biodiversity: questioning the restorative character of the forest transition in the Mediterranean. Ecol Soc 20(2):7
Paillet Y, Bergès L, Hjältén J et al (2010) Biodiversity differences between managed and unmanaged forests: Meta-analysis of species richness in Europe. Conserv Biol 24(1):101–112. https://doi.org/10.1111/j.1523-1739.2009.01399.x
Paillet Y, Zapponi L, Schall P et al (2024) One to rule them all? Assessing the performance of Forest Europe’s biodiversity indicators against multitaxonomic data. Biorxiv. https://doi.org/10.1101/2024.02.12.579875
Pötzelsberger E, Spiecker H, Neophytou C, Mohren F, Gazda A, Hasenauer H (2020) Growing non-native trees in European forests brings benefits and opportunities but also has its risks and limits. Curr for Rep 6:339–353. https://doi.org/10.1007/s40725-020-00129-0
Pretzsch H (2009) Forest dynamics, growth and yield. Springer, Heidelberg. https://doi.org/10.1007/978-3-540-88307-4
Princé K, Rouveyrol P, Pellissier V, Touroult J, Jiguet F (2021) Long-term effectiveness of Natura 2000 network to protect biodiversity: a hint of optimism for common birds. Biol Cons 253:108871. https://doi.org/10.1016/j.biocon.2020.108871
Puettmann KJ, Wilson SM, Baker SC et al (2015) Silvicultural alternatives to conventional even-aged forest management-what limits global adoption? For Ecosyst 2(1):1–16. https://doi.org/10.1186/s40663-015-0031-x
Purahong W, Wubet T, Krüger D, Buscot F (2018) Molecular evidence strongly supports deadwood-inhabiting fungi exhibiting unexpected tree species preferences in temperate forests. ISME J 12(1):289–295. https://doi.org/10.1038/ismej.2017.177
Quinkenstein A, Jochheim H (2016) Assessing the carbon sequestration potential of poplar and black locust short rotation coppices on mine reclamation sites in Eastern Germany-Model development and application. J Environ Manage 168:53–66. https://doi.org/10.1016/j.jenvman.2015.11.044
Rada S, Schweiger O, Harpke A, Kühn E, Kuras T, Settele J, Musche M (2019) Protected areas do not mitigate biodiversity declines: a case study on butterflies. Divers Distrib 25(2):217–224. https://doi.org/10.1111/ddi.12854
Rametsteiner E, Mayer P (2004) Sustainable forest management and pan: European forest policy. Ecological Bulletins (51): 51–57.
Raymond P, Bédard S (2017) The irregular shelterwood system as an alternative to clearcutting to achieve compositional and structural objectives in temperate mixedwood stands. For Ecol Manage 398:91–100. https://doi.org/10.1016/j.foreco.2017.04.042
Raymond P, Bédard S, Roy V, Larouche C, Tremblay S (2009) The Irregular Shelterwood System: Review, Classification, and Potential Application to Forests Affected by Partial Disturbances. J Forest 107:405–413. https://doi.org/10.1093/jof/107.8.405
Remmert H (1991) Das Mosaik-Zyklus-Konzept und seine Bedeutung für den Naturschutz - eine Übersicht. Laufener Seminarbeiträge 5/1991: 5–15. https://www.zobodat.at/pdf/Laufener-Spez-u-Seminarbeitr_5_1991_0005-0015.pdf
Remy E, Wuyts K, Boeckx P, Ginzburg S, Gundersen P, Demey A, Van Den Bulcke J, Van Acker J, Verheyen K (2016) Strong gradients in nitrogen and carbon stocks at temperate forest edges. For Ecol Manage 376:45–58. https://doi.org/10.1016/j.foreco.2016.05.040
Rodríguez A, Kouki J (2017) Disturbance-mediated heterogeneity drives pollinator diversity in boreal
managed forest ecosystems. Ecological Applications 27: 589–602. https://doi.org/10.1002/eap.1468
Rodríguez A, Pohjoismäki JLO, Kouki J (2019) Diversity of forest management promotes parasitoid functional diversity in boreal forests. Biol Cons 238:108205. https://doi.org/10.1016/j.biocon.2019.108205
Ruiz-Labourdette D, Nogués-Bravo D, Ollero HS, Schmitz MF, Pineda FD (2012) Forest composition in Mediterranean mountains is projected to shift along the entire elevational gradient under climate change. J Biogeogr 39:162–176. https://doi.org/10.1111/j.1365-2699.2011.02592.x
Sabatini FM, Burrascano S, Keeton WS et al (2018) Where are Europe’s last primary forests? Diversity and Distribution 24(10):1426–1439. https://doi.org/10.1111/ddi.12778
Savilaakso S, Johansson A, Häkkilä M, Uusitalo A, Sandgren T, Mönkkönen M, Puttonen P (2021) What are the effects of even-aged and uneven-aged forest management on boreal forest biodiversity in Fennoscandia and European Russia? A Systematic Review Environmental Evidence 10:1. https://doi.org/10.1186/s13750-020-00215-7
Schall P, Gossner MM, Heinrichs S et al (2018) The impact of even-aged and uneven-aged forest management on regional biodiversity of multiple taxa in European beech forests. J Appl Ecol 55(1):267–278. https://doi.org/10.1111/1365-2664.12950
Schall P, Heinrichs S (2020) Comment on “Forest microclimate dynamics drive plant responses to warming”. Science 370(6522): eabd9920. https://doi.org/10.1126/science.abd9920
Schall P, Heinrichs S, Ammer C et al (2020) Can multi-taxa diversity in European beech forest landscapes be increased by combining different management systems? J Appl Ecol 57(7):1363–1375. https://doi.org/10.1111/1365-2664.13635
Schmid M, Pautasso M, Holdenrieder O (2014) Ecological consequences of Douglas fir (Pseudotsuga menziesii) cultivation in Europe. Eur J Forest Res 133:13–29. https://doi.org/10.1007/s10342-013-0745-7
Schuldt A, Scherer-Lorenzen M (2014) Non-native tree species (Pseudotsuga menziesii) strongly decreases predator biomass and abundance in mixed-species plantations of a tree diversity experiment. For Ecol Manage 327:10–17. https://doi.org/10.1016/j.foreco.2014.04.036
Schütz JP, Saniga M, Diaci J, Vrška T (2016) Comparing close-to-naturesilviculture with processes in pristine forests: lessons from Central Europe. Ann for Sci 73:911–921. https://doi.org/10.1007/s13595-016-0579-9
Sebald J, Thrippleton T, Rammer W, Bugmann H, Seidl R (2021) Mixing tree species at different spatial scales: The effect of alpha, beta and gamma diversity on disturbance impacts under climate change. J Appl Ecol 58:1749–1763. https://doi.org/10.1111/1365-2664.13912
Sebek P, Bace R, Bartos M et al (2015) Does a minimal intervention approach threaten the biodiversity of protected areas? A multi-taxa short-term response to intervention in temperate oak-dominated forests. For Ecol Manage 358:80–89. https://doi.org/10.1016/j.foreco.2015.09.008
Šebesta J, Maděra P, Řepka R, Matula R (2017) Comparison of vascular plant diversity and species composition of coppice and high beech forest in the Banat region, Romania. Folia Geobot 52:33–43. https://doi.org/10.1007/s12224-016-9279-3
Seibold S, Bässler C, Brandl R, Gossner MM, Thorn S, Ulyshen MD, Müller J (2015) Experimental studies of dead-wood biodiversity: a review identifying global gaps in knowledge. Biol Cons 191:139–149. https://doi.org/10.1016/j.biocon.2015.06.006
Selva N, Chylarecki P, Jonsson BG, Ibisch PL (2020) Misguided forest action in EU biodiversity strategy. Science 368(6498):1438–1439. https://doi.org/10.1126/science.abc9892
Sing L, Metzger MJ, Paterson JS, Ray D (2018) A review of the effects of forest management intensity on ecosystem services for northern European temperate forests with a focus on the UK. Forestry 91:151–164. https://doi.org/10.1093/forestry/cpx042
Sommerfeld A, Rammer W, Heurich M, Hilmers T, Müller J, Seidl R (2020) Do bark beetle outbreaks amplify or dampen future bark beetle disturbances in Central Europe? J Ecol 109:737–749. https://doi.org/10.1111/1365-2745.13502
Steinert M, Moe SR, Sydenham MAK, Eldegard K (2018) Different cutting regimes improve species and functional diversity of insect-pollinated plants in power-line clearings. Ecosphere 9:e02509. https://doi.org/10.1002/ecs2.2509
Suda M, Pukall K (2014) Multifunktionale Forstwirtschaft zwischen Inklusion und Extinktion (Essay). Schweiz Z Forstwes 165(11):333–338. https://doi.org/10.3188/szf.2014.0333
Tena E, de Paz Ó, de La Peña R, Fandos G, Redondo M, Tellería JL (2020) Mind the gap: effects of canopy clearings on temperate forest bat assemblages. For Ecol Manage 474:118341. https://doi.org/10.1016/j.foreco.2020.118341
Tews J, Brose U, Grimm V, Tielbörger K, Wichmann MC, Schwager M, Jeltsch F (2004) Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. J Biogeogr 31(1):79–92. https://doi.org/10.1046/j.0305-0270.2003.00994.x
Thorn S, Seibold S, Leverkus AB, Michler T, Müller J, Noss RF, Stork N, Vogel S, Lindenmayer DB (2020) The living dead: acknowledging life after tree death to stop forest degradation. Front Ecol Environ 18(9):505–512. https://doi.org/10.1002/fee.2252
Tinner W, Colombaroli D, Heiri O et al (2013) The past ecology of Abies alba provides new perspectives on future responses of silver fir forests to global warming. Ecol Monogr 83(4):419–439. https://doi.org/10.1890/12-2231.1
Tinya F, Doerfler I, de Groot M et al (2023) A synthesis of multi-taxa management experiments to guide forest biodiversity conservation in Europe. Global Ecol Conserv 46:e02553. https://doi.org/10.1016/j.gecco.2023.e02553
Tomao A, Antonio Bonet J, Castaño C, de Miguel S, (2020) How does forest management affect fungal diversity and community composition? Current knowledge and future perspectives for the conservation of forest fungi. For Ecol Manage 457:117678. https://doi.org/10.1016/j.foreco.2019.117678
Torralba M, Fagerholm N, Burgess PJ, Moreno G, Plieninger T (2016) Do European agroforestry systems enhance biodiversity and ecosystem services? A meta-analysis. Agr Ecosyst Environ 230:150–161. https://doi.org/10.1016/j.agee.2016.06.002
Trentanovi G, Campagnaro T, Sitzia T et al (2023) Words apart: Standardizing forestry terms and definitions across European biodiversity studies. For Ecosyst 10:100128. https://doi.org/10.1016/j.fecs.2023.100128
Tullus T, Tishler M, Rosenvald R, Tullus A, Lutter R, Tullus H (2019) Early responses of vascular plant and bryophyte communities to uniform shelterwood cutting in hemiboreal Scots pine forests. For Ecol Manage 440:70–78. https://doi.org/10.1016/j.foreco.2019.03.009
Uhl B, Pouska V, Červenka J, Karasch P, Bässler C (2022a) Biodiversitätsschutz in Zentraleuropäischen Wäldern—ein Konzept für Waldbewirtschafter und politische Entscheidungsträger. Verwaltung des Nationalparks Sumava, Vimperk
Uhl B, Krah FS, Baldrian P, Brandl R, Hagge J, Müller J, Thorn S, Vojtech T, Bässler C (2022b) Snags, logs, stumps, and microclimate as tools optimizing deadwood enrichment for forest biodiversity. Biol Cons 270:109569. https://doi.org/10.1016/j.biocon.2022.109569
Ujházy K, Hederová L, Máliš F, Ujházyová M, Bosela M, Čiliak M (2017) Overstorey dynamics controls plant diversity in age-class temperate forests. For Ecol Manage 391:96–105. https://doi.org/10.1016/j.foreco.2017.02.010
Vacik H, Zlatanov T, Trajkov P, Dekanic S, Lexer MJ (2009) Role of coppice forests in maintaining forest biodiversity. Silva Balcanica 10(1):35–45
Vanbeveren SP, Ceulemans R (2019) Biodiversity in short-rotation coppice. Renew Sustain Energy Rev 111:34–43. https://doi.org/10.1016/j.rser.2019.05.012
Venäläinen A, Lehtonen I, Laapas M, Ruosteenoja K, Tikkanen OP, Viiri H, Ikonen V, Peltola H (2020) Climate change induces multiple risks to boreal forests and forestry in Finland: a literature review. Glob Change Biol 26(8):4178–4196. https://doi.org/10.1111/gcb.15183
Vera FW, Bakker ES, Olff H (2006) Large herbivores: missing partners of western European light-demanding tree and shrub species? Conserv Biol Ser Camb 11:203
Verlinden MS, Broeckx LS, Ceulemans R (2015) First vs. second rotation of a poplar short rotation coppice: above-ground biomass productivity and shoot dynamics. Biomass Bioenerg 73:174–185. https://doi.org/10.1016/j.biombioe.2014.12.012
Vítková L, Bače R, Kjučukov P, Svoboda M (2018) Deadwood management in Central European forests: key considerations for practical implementation. For Ecol Manage 429:394–405. https://doi.org/10.1016/j.foreco.2018.07.034
Vitousek PM (1981) Clear-cutting and the nitrogen cycle. Ecol Bull 33:631–642
Vogel S, Gossner MM, Mergner U, Müller J, Thorn S (2020) Optimizing enrichment of deadwood for biodiversity by varying sun exposure and tree species: an experimental approach. J Appl Ecol 57:2075–2085. https://doi.org/10.1111/1365-2664.13648
Vogel S, Bussler H, Finnberg S, Müller J, Stengel E, Thorn S (2021) Diversity and conservation of saproxylic beetles in 42 European tree species: an experimental approach using early successional stages of branches. Insect Conserv Divers 14(1):132–143. https://doi.org/10.1111/icad.12442
Vollmuth DW (2021) Die Nachhaltigkeit und der Mittelwald. Göttinger Forstwissenschaften 10:568. https://doi.org/10.17875/gup2021-1602
von Felten S, Berney C, Erb B, Baumann P, Korner-Nievergelt F, Senn-Irlet B (2020) Habitat enhancements for reptiles in a beech forest may increase fungal species richness. Biodivers Conserv 29(6):1805–1819. https://doi.org/10.1007/s10531-020-01949-z
Wahren A, Richter F, Julich S, Jansen M, Feger KH (2015) The influence of more widespread cultivation of short rotation coppice on the water balance: from the site to the regional scale. In: Manning DB, Bemmann A, Bredemeier M, Lamersdorf N, Ammer C (eds) Bioenergy from dendromass for the sustainable development of rural areas. Wiley, New York
Wetherbee R, Birkemoe T, Sverdrup-Thygeson A (2020) Veteran trees are a source of natural enemies. Sci Rep 10(1):18485. https://doi.org/10.1038/s41598-020-75723-0
Wetherbee R, Birkemoe T, Burner RC, Sverdrup-Thygeson A (2021) Veteran trees have divergent effects on beetle diversity and wood decomposition. PLoS ONE 16(3):e0248756. https://doi.org/10.1371/journal.pone.0248756
Weathers KC, Cadenasso ML, Pickett ST (2001) Forest edges as nutrient and pollutant concentrators: potential synergisms between fragmentation, forest canopies, and the atmosphere. Conserv Biol 15(6):1506–1514. https://doi.org/10.1046/j.1523-1739.2001.01090.x
Williams DT, Straw N, Fielding N, Jukes M, Price J (2017) The influence of forest management systems on the abundance and diversity of bark beetles (Coleoptera: Curculionidae: Scolytinae) in commercial plantations of Sitka spruce. For Ecol Manage 398:196–207. https://doi.org/10.1016/j.foreco.2017.05.014
Woziwoda B, Kopec D, Witkowski J (2014) The negative impact of intentionally introduced Quercus rubra L. on a forest community. Acta Soc Bot Pol 83(1):39–49. https://doi.org/10.5586/asbp.2013.035
Zasadil P, Romportl D, Horák J (2020) Disentangling the roles of topography, patch, and land use on conservation trait status of specialist birds in marginal forest land use types. Forests 11:103. https://doi.org/10.3390/f11010103
Zellweger F, de Frenne P, Lenoir J et al (2020) Forest microclimate dynamics drive plant responses to warming. Science 368:772–775. https://doi.org/10.1126/science.aba6880
Żmihorski M, Berg Å, Pärt T (2016) Forest clear-cuts as additional habitat for breeding farmland birds in crisis. Agr Ecosyst Environ 233:291–297. https://doi.org/10.1016/j.agee.2016.09.023
Acknowlegements
The position of BU was partly funded by the European Union (Interreg project BioDiv 316). We additionally want to thank the Editor and the two anonymous reviewers for their constructive comments on previous manuscript versions.
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was supported by the European Union (Project Interreg Biodiv 316). Besides, there were no other funds, grants or support during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Claus Bässler and Britta Uhl had the idea for writing the review. Britta Uhl performed the literature search, wrote the first draft and designed figures and tables. Claus Bässler and Peter Schall critically revised the work, commented on previous versions of the manuscript and added information. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Mauro Agnoletti.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Uhl, B., Schall, P. & Bässler, C. Achieving structural heterogeneity and high multi-taxon biodiversity in managed forest ecosystems: a European review. Biodivers Conserv (2024). https://doi.org/10.1007/s10531-024-02878-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10531-024-02878-x