Abstract
Subthalamic deep brain stimulation (STN-DBS) is known to improve motor function in advanced Parkinson’s disease (PD) and to enable a reduction of anti-parkinsonian medication. While the levodopa challenge test and disease duration are considered good predictors of STN-DBS outcome, other clinical and neuroanatomical predictors are less established. This study aimed to evaluate, in addition to clinical predictors, the effect of patients’ individual brain topography on DBS outcome. The medical records of 35 PD patients were used to analyze DBS outcomes measured with the following scales: Part III of the Unified Parkinson’s Disease Rating Scale (UPDRS-III) off medication at baseline, and at 6-months during medication off and stimulation on, use of anti-parkinsonian medication (LED), Abnormal Involuntary Movement Scale (AIMS) and Non-Motor Symptoms Questionnaire (NMS-Quest). Furthermore, preoperative brain MRI images were utilized to analyze the brain morphology in relation to STN-DBS outcome. With STN-DBS, a 44% reduction in the UPDRS-III score and a 43% decrease in the LED were observed (p<0.001). Dyskinesia and non-motor symptoms decreased significantly [median reductions of 78,6% (IQR 45,5%) and 18,4% (IQR 32,2%) respectively, p=0.001 – 0.047]. Along with the levodopa challenge test, patients’ age correlated with the observed DBS outcome measured as UPDRS-III improvement (ρ= -0.466 – -0.521, p<0.005). Patients with greater LED decline had lower grey matter volumes in left superior medial frontal gyrus, in supplementary motor area and cingulum bilaterally. Additionally, patients with greater UPDRS-III score improvement had lower grey matter volume in similar grey matter areas. These findings remained significant when adjusted for sex, age, baseline LED and UPDRS scores respectively and for total intracranial volume (p=0.0041- 0.001). However, only the LED decrease finding remained significant when the analyses were further controlled for stimulation amplitude. It appears that along with the clinical predictors of STN-DBS outcome, individual patient topographic differences may influence DBS outcome. Clinical Trial Registration Number: NCT06095245, registration date October 23, 2023, retrospectively registered
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Subthalamic deep brain stimulation (STN-DBS) is an evidence-based treatment option for advanced Parkinson’s disease (PD) (Deuschl et al. 2006, Mansouri et al. 2018). DBS treatment usually alleviates cardinal Parkinsonian motor symptoms, such as rigidity, tremor, and bradykinesia (Krack et al. 2003, Deuschl, Paschen et al. 2013).
Even after careful patient selection with well-established guidelines (Bronstein et al. 2011, Hartmann et al. 2019, Deuschl, Antonini et al. 2022), DBS response may vary among PD patients. A levodopa challenge test is considered to be a good predictor of DBS response (Charles et al. 2002, Lang et al. 2006, Lachenmayer et al. 2021, Lin et al. 2022). More than 33% improvement in Unified Parkinson’s Disease Rating Scale part III (UPDRS-III) score during the ON phase of the levodopa challenge test is required for STN-DBS treatment (Charles et al. 2002, Lang et al. 2006, Lachenmayer et al. 2021, Lin et al. 2022). Several studies have suggested that younger age may be a predictor of DBS response and quality of life after DBS operation (Charles et al. 2002, Lang et al. 2006, Groiss et al. 2009, Hartmann et al. 2019, Deuschl et al. 2022). In other studies, the relationship between aging and DBS response has not been confirmed (Geraedts et al. 2020, Lin et al. 2022). Additionally, the disease duration prior to the initiation of DBS treatment and fewer levodopa-resistant symptoms may be predictors of DBS response (Lachenmayer et al. 2021). On neuroimaging, vascular changes have been recognized as clinical predictors of poorer long-term DBS outcome (Cavallieri et al. 2021).
Versatile theories on the actions of DBS on Parkinsonian symptoms have been proposed. DBS may affect neuronal firing rates locally (Lozano and Lipsman 2013, Chiken and Nambu 2016) but may also modulate more remote connected brain regions (McIntyre and Hahn 2010, Lozano and Lipsman 2013, Horn et al. 2017, Johnson et al. 2020). This connectivity may predict DBS outcome in PD patients (Horn et al. 2017). An individual patient’s brain topographic features may also have an impact on DBS response (Frizon et al. 2020, Chen et al. 2022, Jergas et al. 2023). However, to date, reliable topographic predictors have not been identified. While some studies suggest that cortical thickness, especially in the motor and supplementary motor cortex, could predict motor outcome in DBS, other studies have provided contradictory evidence (Wang et al. 2022). Additionally, the volumes of various brain regions have been linked to DBS response, but consistent findings have not been reported (Younce et al. 2019, Yim et al. 2020). The aim of this study was to evaluate possible clinical patient features affecting DBS response and to investigate whether patients’ individual cortical volumes have an impact on DBS response.
Materials and Methods
Clinical Data
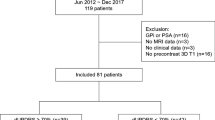
The medical records of 48 consecutive PD patients treated with STN-DBS at Helsinki University Hospital (HUS) were reviewed. MRI imaging was performed on 35 patients with the same MRI machine (3T Magnetom Skyra, Siemens Healthineers, Erlangen, Germany), and these patients were selected for this retrospective study. Staging of advanced PD in all patients had been determined by a treating movement disorder specialist according to Delphi criteria (Antonini et al. 2018). All patients had undergone a routine DBS screening (including a thorough neuropsychological examination, the levodopa challenge test and brain MRI scan without observed significant vascular changes). All patients received a directional DBS system (Abbott Infinity DBS™, Abbott Neuromodulation, Austin, TX, USA). The patients had completed a full DBS programming routine with programming visits at 1, 1.5, 3 and 6 postoperative months as described earlier (Koivu et al. 2022). Since the MRI imaging protocol for DBS was revised at HUS in 2019, only patients operated on between 2020 – 2022 were selected for this study.
The following clinical data were collected from the DBS screening visit (baseline) and the in-hospital six-month programming visit at HUS: UPDRS-III in the medication off state and in the medication ON state (during the levodopa challenge test) and in the medication off, stimulation on state (at the six-month visit), Abnormal Involuntary Movements Scale (AIMS), Parkinson’s Disease Questionnaire 39 (PDQ-39), Mini Mental State Examination (MMSE), Non-Motor Symptoms Questionnaire (NMS-Quest), and Beck Depression Inventory (BDI). Additionally, data on the use of antiparkinsonian drugs and other comorbidities were obtained. The levodopa equivalent dose (LED) was calculated as previously proposed (Tomlinson et al. 2010, Schade et al. 2020). The DBS programming settings (amplitudes, pulse width, frequency, impedance) as well as the stimulation mode and possible alterations in electrode activation during the six-month follow-up were collected. The research permit was approved by the medical director responsible for the academic research at HUS without additional approval from the ethics committee according to Finnish laws.
MRI Data Acquisition
All neuroimaging data were collected at the Department of Radiology. High-resolution magnetization-prepared rapid acquisition gradient-recalled T1 images were obtained (TR = 2000 ms, TE = 2.74 ms, voxel size = 1 × 1 × 1). A neuroradiologist checked the MRI images for incidental findings.
Voxel-based Morphometry
Morphometric analysis was carried out using Statistical Parametric Mapping (SPM12, Wellcome Department of Cognitive Neurology, UCL) in MATLAB 9.10.0 (The MathWorks, Inc., Natick, MA, USA; version R2021a). First, the individual T1 images were reoriented using the anterior commissure as the origin. The new segmentation algorithm with default parameters, except for the affine regularization set to the International Consortium for Brain Mapping (ICBM) template for the brains of European participants, was subsequently applied to the T1 images, segmenting them precisely into grey matter, white matter, and cerebrospinal fluid probability maps. The tissue probability maps were then normalized to the Montreal Neurological Institute (MNI) space using the Diffeomorphic Anatomical Registration Through Exponentiated Lie Algebra (DARTEL) registration process implemented in SPM12. During the normalization process, the data were resampled to a 1.5 × 1.5 × 1.5 mm3 voxel size and modulated, allowing evaluation of regional volumetric differences. Finally, grey matter and white matter maps were smoothed with an isotropic Gaussian kernel of 8 mm full width at half maximum (FMWH). During each step, the images were visually checked for potential segmentation and registration errors. The total intracranial volume for each patient was calculated by combining the grey matter, white matter, and cerebrospinal fluid images generated during the segmentation.
Statistical Analyses
All the statistical analyses were performed with IBM SPSS Statistics, version 27 (IBM Corporation, Armonk, New York, USA). The data are presented as medians with interquartile ranges (IQRs), and p values less than 0.05 were considered significant. Clinical data analyses were conducted using the Wilcoxon signed rank test and Mann‒Whitney test considering the small patient cohort, and for correlations, the Spearman correlation test or Pearson two-tailed correlation when applicable, contingency coefficient, and linear regression were used for DBS outcome analysis.
In the voxel-based morphometry analysis, two linear regression models evaluating the relationship between pre-DBS regional grey matter volume and changes (6 months > preoperative) in i) LED and ii) UPDRS-III scores were calculated using SPM12. The results were thresholded using the “Threshold and transform spmT-maps” function in the CAT12 toolbox at a default cluster-forming threshold (uncorrected p < 0.001) and a familywise error rate (FWE) corrected p < 0.05 at the cluster level (alpha-level) and corrected for non-isotropic smoothness (Hayasaka et al. 2004). All voxel-based morphometry analyses were adjusted for age, sex, and total intracranial volume (Barnes et al. 2010) as well as for baseline LED and UPDRS-III scores (Hope et al. 2019). Neuroanatomical regions were identified using the Automated Anatomical Labeling Atlas (Tzourio-Mazoyer et al. 2002) included in the xjView toolbox (http://www.alivelearn.net/xjview/).
Results
Clinical Data
The baseline demographic data of the 35 PD patients are presented in Table 1.
During the levodopa challenge test at the DBS screening visit, a median decrease of 23,0 points (IQR 10,0 points) in the UPDRS-III score was observed. This corresponds to a median improvement of 65% (IQR 20,5%) (Table 2). After 6 months of DBS treatment, the median UPDRS-III score was reduced by 15,0 points (IQR 15,0 points) when assessed during medication off, stimulation on, corresponding to a 43,9% (IQR 25,6%) decrease in the UPDRS-III score (Table 2). In addition, non-motor symptoms, dyskinesia (as evaluated with the AIMS) and motor symptoms (as evaluated with the H&Y scale in medication off condition) also improved significantly with DBS treatment. The postoperative decrease in LED was also significant, with a median reduction of 43,0% (IQR 33,1%) (p<0.001).
Patients’ age correlated with the duration of PD (ρ=0.505, p=0.002) but not with the duration of the advanced stage of PD (ρ=0.040, p=0.834). Furthermore, age did not correlate with baseline UPDRS-III score or LED, sex, or PDQ-39 SI score, nor did the aforementioned variables correlate with each other (ρ=-0.294 – 0.443, p=0.086 – 0.945). Age correlated with longitudinal UPDRS-III score improvement with STN-DBS and with observed improvement in the levodopa challenge test score (ρ=-0.466 – -0.521, p<0.005) but not with longitudinal LED decrease (ρ=-0.072, p=0.682). Younger patients had greater improvements in the UPDRS-III scores with STN-DBS and greater improvements in motor symptoms the levodopa challenge test. Age appeared to predict UPDRS-III score improvement with DBS stimulation (β=-0.595, p<0.001) but did not predict LED decrease after DBS therapy (β=-0.070, p=0.688).
Motor improvement in the levodopa challenge test correlated with longitudinal UPDRS-III score changes (ρ=0.478, p=0.004) but not with disease duration, sex, or the reported duration of advanced PD (ρ=0,024 – -0.286, p=0.101 –0.898). The longitudinal UPDRS-III score improvement did not correlate with the baseline LED, disease duration or reported duration of advanced PD (ρ=-0.289 – 0.014, p=0.097-0.936) or with the baseline PDQSI or NMS-Quest scores (ρ=-0.170– 0.244, p=0.097 – 0.871). Disease duration and self-reported duration of advanced PD were not associated with longitudinal UPDRS-III score changes (β=-0.299–0.023, p=0.165–0.913).
DBS data
After six months, directional stimulation was used in 87,1% of the dDBS leads (in 61 out of 70 dDBS leads); see Table 3. With 27 patients, directional single-segment stimulation (SSA) was used bilaterally, and with others, SSA was activated in one of the dDBS leads, as was omnipolar and/or two-segment activation in the other leads. Only two patients received omnidirectional stimulation bilaterally after directionality testing.
There were no operation-related complications (infection or intracranial bleeding) or severe stimulation-related adverse effects (e.g., dysarthria or depression). One patient had deep vein thrombosis postoperatively treated with a regimen of transient anticoagulation therapy.
Morphometric analysis
First, the relationships between longitudinal changes in LED and UPDRS-III scores and global brain measurements (i.e., grey matter, white matter, cerebrospinal fluid and total brain volume) as defined by the percentage out of total intracranial volume (e.g.,\(\frac{grey matter volume}{total intracranial volume}\)) were evaluated with Pearson correlations (two-tailed). No statistically significant associations were found between the global brain measures and LED change (grey matter r=-0.255, p=0.140; white matter r=0.290, p=0.082; cerebrospinal fluid r=-0.103, p=0.555; and total brain volume r=0.103, p=0.555) or UPDRS-III score change (grey matter r=0.032, p=0.853; white matter r=0.064, p=0.717; cerebrospinal fluid r=-0.237, p=0.171; and total brain volume r=0.237, p=0.171).
In the whole-brain voxel-based morphometry VBM analyses adjusted for total intracranial volume, age, sex and baseline LED, a greater longitudinal decrease in LED was associated with lower grey matter volume in one cluster comprising the left superior medial frontal gyrus as well as the supplementary motor area and cingulum bilaterally (Fig. 1A, Table 4). The finding remained relatively unchanged when the analysis was further adjusted for the stimulation amplitude used (Fig. 1B). The duration of PD or the duration of advanced PD did not show correlations with aforementioned grey matter clusters observed with LED decrease (disease duration r=0.014, p=0.938; advanced PD r=0.004, p=0.983).
Morphometric results showing grey matter volume associations with longitudinal changes in LED and UPDRS-III scores. (A) and (B) lower baseline grey matter volume predicting greater longitudinal decrease in LED, and (C) lower baseline grey matter volume predicting greater longitudinal decrease in the UPDRS-III score. N = 35. The results are reported using MNI coordinates at the cluster-forming threshold (uncorrected p < 0.001) and a FWE-corrected p < 0.05 at the cluster level (alpha level) and corrected for non-isotropic smoothness. L = Left, R = Right, TIV = Total intracranial volume
A greater decrease in the UPDRS-III score was associated with a lower grey matter volume in a similar cluster comprising the superior medial frontal gyrus and the cingulum bilaterally (Fig. 1C, Table 4) when the analysis was adjusted for total intracranial volume, age, sex, and baseline UPDRS score. However, when the analysis was further adjusted for the stimulation current amplitude, no significant findings were observed. Observed grey matter findings associated with UPDRS-III improvements did not correlate with the disease duration nor with the duration of advanced PD (disease duration r=0.040, p=0.822; advanced PD r=0.018, p=0.926). Figure 2 shows the structural connectivity of the STN in relation to the significant grey matter findings.
Visualization of the significant morphometric findings in relation to the structural connectivity of the subthalamic nucleus. (A) and (B) lower baseline grey matter volume predicting greater longitudinal decrease in LED, and (C) lower baseline grey matter volume predicting greater longitudinal decrease in UPDRS score. GM = Grey matter, STN = subthalamic nucleus, TIV = total intracranial volume
Discussion
Using clinical variables and grey matter morphometric MRI analyses, this study aimed to evaluate predictors of STN-DBS outcome in advanced PD patients. Our findings indicate that both age and the levodopa challenge test score significantly predict STN-DBS outcome, measured as UPDRS-III score improvement. Furthermore, lower grey matter volume in the left superior frontal/supplementary motor areas (SMA) and cingulum predicted greater longitudinal UPDRS-III improvement and LED decrease in this patient cohort. The disease duration or the duration of advanced stage of PD did not correlate with the morphometric findings in this study. Overall, the previous morphometric studies on DBS effect have been heterogeneous, presenting various brain regions’ role in connectivity with STN in the DBS outcome. Previous studies have shown that multiple brain regions are both positively and negatively correlated with postsurgical outcome (Wang et al. 2022). This can be explained to some extent by the various research methods used. In some studies, the thickness of the motor cortex predicted motor outcome after DBS operation, and in others, these results could not be replicated (Wang et al. 2022). In a study by Muthuraman (Muthuraman et al. 2017), the thickness of the frontal cortex, principally in the paracentral area and superior frontal region, was found to be a predictor of motor improvement following STN-DBS treatment. In another study, a positive association between STN-DBS outcome and stimulated area’s connections to the SMA and the right precentral cortex was noted (Chen et al. 2022). Additionally, in the same study, a negative association between DBS outcome and connectivity to the right superior frontal, middle and inferior frontal cortices, anterior and middle cingulum and caudate was reported (Chen et al. 2022). Preoperative STN connectivity to the frontal and prefrontal cortex and cingulate gyrus has been correlated with postoperative outcomes (Koirala et al. 2018, Gonzalez-Escamilla et al. 2022), and long-term STN-DBS outcomes have been associated with connectivity to the SMA (Chen et al. 2022).
Posterior and middle STN have strongest connectivity to the primary motor cortex and SMA (Obeso et al. 2008, DiRisio et al. 2023), and the SMA activation is speculated to result from the activation of the fibers in the hyperdirect pathway (Accolla et al. 2016, Horn et al. 2017). In neuroimaging studies with ongoing subthalamic stimulation, increased cerebral blood flow has been noted in SMA, anterior cingulate and prefrontal cortex (Ceballos-Baumann et al. 1999, Sestini et al. 2005). In a study with functional MRI, an augmented interconnectedness in the motor cortices bilaterally and enhanced cerebello-thalamic-cortical network connectivity were observed with STN DBS treatment but not with oral levodopa treatment (Mueller et al. 2018). It has been suggested that broad frontal and premotor cortical thinning can be found in patients with PD compared to healthy controls (Ibarretxe-Bilbao et al. 2012, Pereira et al. 2014), and this phenomenon may have influenced results in this study. In addition to frontal cortical connections, studies have demonstrated associations with other brain regions and STN-DBS outcomes. A greater thickness of the left lateral occipital lobe has been reported to be associated with better UPDRS-III score improvement after STN-DBS (Frizon et al. 2020). This observation was noted in region of interest (ROI) analysis as well as in vertex-by-vertex analysis. In the same study, a thicker frontal and temporal cortex was found to be associated with UPDRS-III score improvements in ROI analysis but not in vertex-by-vertex analysis (Frizon et al. 2020). In a recent study, grey matter atrophy in the medial prefrontal cortex, cingulate gyrus, paracingulate gyrus and parietal lobe was related to a suboptimal response to DBS, although a significant association with brain atrophy and the levodopa response could not be detected (Jergas et al. 2023).
Some studies suggest that subcortical brain structures are linked to DBS outcome (Price et al. 2011, Younce et al. 2019, Yim, Kim et al. 2020). Smaller thalamus volumes and larger ventricle volumes have been proposed to be predictors of poor motor improvements after DBS surgery (Younce et al. 2019, Yim et al. 2020), although in another study, ventricle volumes lacked predictive value (Price et al. 2011). Moreover, previous studies lack consistency in the utilization of relevant covariates necessary for volumetric analyses (Wang et al. 2022). Furthermore, to the best of our knowledge, only one previous study has adjusted the analyses for baseline LED and UPDRS-III score (Younce et al. 2019), as recommended (Hope et al. 2019). However, to the best of our knowledge, not all of those previous studies controlled the stimulation amplitude. Taken together, the previous heterogeneous findings may be partly explained by the methodological and statistical variation as well as the lack of consistency in adjusting the analyses for confounding effects in the studies, although the demographics of the patients were relatively homogeneous.
In this study, the data were corrected for non-isotropic smoothness (Hayasaka et al. 2004) and all analyses were adjusted for age, sex, and total intracranial volume (Barnes et al. 2010) as well as for baseline levodopa equivalent daily dose and UPDRS-III scores (Hope et al. 2019). Greater declines in the UPDRS-III score were observed with lower grey matter volume in the superior medial frontal gyrus and cingulum bilaterally (see Fig. 1C); however, when these changes were further controlled by the stimulation current amplitude, no significant changes were observed. In some studies, higher voltages or amplitudes were needed for an optimal stimulation-induced improvement in those with thinner frontal cortex (Muthuraman et al. 2017, Koirala et al. 2018, Gonzalez-Escamilla et al. 2022). In the present study, with current stimulation, no relationship was observed between the required stimulation current for optimal motor control and the DBS response. In all the patients, the amplitudes used were rather moderate (median, 2.2 mA; IQR, 0.9 mA). Similarly, a longitudinal decrease in LED seemed to be associated with a decrease in grey matter volume in the left superior medial frontal gyrus as well as in the supplementary motor area and cingulum bilaterally (see Fig. 1A), even when controlling stimulation amplitude. These observations may be related to our small patient cohort, though its size is comparable to that of many previous morphometric studies (Muthuraman et al. 2017, Frizon et al. 2020). Therefore, additional studies are warranted to reproduce these results.
The clinical results of the six-month STN-DBS treatment in this study paralleled those of previous studies; approximately 44% improvement in the UPDRS-III score and a 43% decrease in the LED were observed, as was a significant reduction in dyskinesia. With STN-DBS, 43–51% improvements in UPDRS-III scores and 50–64% LED reductions have been reported (Bronstein et al. 2011, Dembek et al. 2017, Shao et al. 2020, Schnitzler et al. 2022). For 27 patients (77%), bilateral directional single-segment stimulation was used, only two patients preferred bilateral ring-mode stimulation, and all the stimulation parameters used resembled previous reports (Rammo et al. 2022, Schnitzler et al. 2022).
In the present study, age and the levodopa response in the levodopa challenge test predicted a significantly positive DBS response as assessed by the UPDRS-III score. However, age did not seem to influence the reduction in LED with DBS treatment. Earlier reports on the impact of aging on DBS response have been heterogeneous. In some studies, age has been shown to be negatively correlated with postoperative DBS responsiveness, as measured by the reduction in the UPDRS-III score (Charles et al. 2002, Russmann et al. 2004), and improved motor function has been noted with younger age and shorter disease duration (Welter et al. 2002, Hartmann et al. 2019). According to a recently published meta-analysis of STN- and GPi-DBS-treated PD patient cohorts, preoperative levodopa responsiveness predicted short-term (6- to 12-month) DBS responsiveness, whereas age did not have predictive value (Lin et al. 2022). Similar results for aging have been published previously (Kleiner-Fisman et al. 2006, Weaver et al. 2009, Hartmann et al. 2019, Geraedts et al. 2020, Lin et al. 2022).
This study has limitations due to its retrospective nature. The patient population was relatively small; therefore, an extensive statistical analysis of DBS outcomes could not be performed.
Another limitation to the study is that post-operational survey of the DBS lead location was not conducted. With these patients, all electrode contacts had been tested systematically with previously presented routine (Koivu et al. 2022). Each electrode contact had been tested initially in omnipolar stimulation mode during 1-month programming visit and then in directional mode at 1.5-month visit for determining the most optimal contact for stimulation. Patients had a notable clinical improvement (as measured with UPDRS-III scores) and a significant LED reduction. Based on that, it could be assumed that the active electrode was within or directly adjacent to the motor region of STN and its fibers connected to cortico-striato-thalamocortical motor loop. The methodology of this study is comparable to the methods used in previous reports (Muthuraman et al. 2017, Frizon et al. 2020). Some studies have shown that a DBS electrode’s connectivity profile depends on the shape, size and position of the volume of tissue activated (VTA) (Horn et al. 2017, Chen et al. 2022). Conversely, a study with insignificant correlation between the postoperative UPDRS-III and the position of the VTA has been published (Koirala et al. 2018). The method of calculating VTA also has some limitations, including being biased on assumptions about white matter fiber orientation and tissue homogeneity which are not always met (Koirala et al. 2018, Duffley et al. 2019).
In conclusion, individual patient morphometric properties, especially in cortical areas, may predict motor outcomes in STN-DBS in combination with clinical predictors and DBS settings.
Data Availability
Data available upon a request.
References
Accolla EA, Herrojo Ruiz M, Horn A, Schneider G-H, Schmitz-Hübsch T, Draganski B, Kühn AA (2016) Brain networks modulated by subthalamic nucleus deep brain stimulation. Brain 139(9):2503–2515
Antonini A, Stoessl AJ, Kleinman LS, Skalicky AM, Marshall TS, Sail KR, Onuk K, Odin PLA (2018) Developing consensus among movement disorder specialists on clinical indicators for identification and management of advanced Parkinson’s disease: a multi-country Delphi-panel approach. Curr Med Res Opin 34(12):2063–2073
Barnes J, Ridgway GR, Bartlett J, Henley SM, Lehmann M, Hobbs N, Clarkson MJ, MacManus DG, Ourselin S, Fox NC (2010) Head size, age and gender adjustment in MRI studies: a necessary nuisance? Neuroimage 53(4):1244–1255
Bronstein JM, Tagliati M, Alterman RL, Lozano AM, Volkmann J, Stefani A, Horak FB, Okun MS, Foote KD, Krack P, Pahwa R, Henderson JM, Hariz MI, Bakay RA, Rezai A, Marks WJ Jr, Moro E, Vitek JL, Weaver FM, Gross RE, DeLong MR (2011) Deep Brain Stimulation for Parkinson Disease: An Expert Consensus and Review of Key Issues. Arch Neurol 68(2):165–165
Cavallieri F, Fraix V, Bove F, Mulas D, Tondelli M, Castrioto A, Krack P, Meoni S, Schmitt E, Lhommée E, Bichon A, Pélissier P, Chevrier E, Kistner A, Seigneuret E, Chabardès S, Moro E (2021) Predictors of Long-Term Outcome of Subthalamic Stimulation in Parkinson Disease. Ann Neurol 89(3):587–597
Ceballos-Baumann AO, Boecker H, Bartenstein P, von Falkenhayn I, Riescher H, Conrad B, Moringlane JR, Alesch F (1999) A positron emission tomographic study of subthalamic nucleus stimulation in Parkinson disease: enhanced movement-related activity of motor-association cortex and decreased motor cortex resting activity. Arch Neurol 56(8):997–1003
Charles PD, Van Blercom N, Krack P, Lee SL, Xie J, Besson G, Benabid AL, Pollak P (2002) Predictors of effective bilateral subthalamic nucleus stimulation for PD. Neurology 59(6):932–934
Chen Y, Zhu G, Liu D, Liu Y, Zhang X, Du T, Zhang J (2022) Seed-Based Connectivity Prediction of Initial Outcome of Subthalamic Nuclei Deep Brain Stimulation. Neurotherapeutics 19(2):608–615
Chiken S, Nambu A (2016) Mechanism of Deep Brain Stimulation: Inhibition, Excitation, or Disruption? Neuroscientist 22(3):313–322
Dembek TA, Reker P, Visser-Vandewalle V, Wirths J, Treuer H, Klehr M, Roediger J, Dafsari HS, Barbe MT, Timmermann L (2017) Directional DBS increases side-effect thresholds-A prospective, double-blind trial. Mov Disord 32(10):1380–1388
Deuschl G, Schade-Brittinger C, Krack P, Volkmann J, Schäfer H, Bötzel K, Daniels C, Deutschländer A, Dillmann U, Eisner W, Gruber D, Hamel W, Herzog J, Hilker R, Klebe S, Kloss M, Koy J, Krause M, Kupsch A, Lorenz D, Lorenzl S, Mehdorn HM, Moringlane JR, Oertel W, Pinsker MO, Reichmann H, Reuss A, Schneider GH, Schnitzler A, Steude U, Sturm V, Timmermann L, Tronnier V, Trottenberg T, Wojtecki L, Wolf E, Poewe W, Voges J (2006) A randomized trial of deep-brain stimulation for Parkinson’s disease. N Engl J Med 355(9):896–908
Deuschl G, Paschen S, Witt K (2013) Clinical outcome of deep brain stimulation for Parkinson’s disease. Handb Clin Neurol 116:107–128
Deuschl G, Antonini A, Costa J, Śmiłowska K, Berg D, Corvol JC, Fabbrini G, Ferreira J, Foltynie T, Mir P, Schrag A, Seppi K, Taba P, Ruzicka E, Selikhova M, Henschke N, Villanueva G, Moro E (2022) European Academy of Neurology/Movement Disorder Society-European Section Guideline on the Treatment of Parkinson’s Disease: I. Invasive Therapies. Mov Disord 37(7):1360–1374
DiRisio AC, Avecillas-Chasin JM, Platt S, Jimenez-Shahed J, Figee M, Mayberg HS, Choi KS, Kopell BH (2023) White matter connectivity of subthalamic nucleus and globus pallidus interna targets for deep brain stimulation. J Neurosurg pp 1–10
Duffley G, Anderson DN, Vorwerk J, Dorval AD, Butson CR (2019) Evaluation of methodologies for computing the deep brain stimulation volume of tissue activated. J Neural Eng 16(6):066024
Frizon LA, Gopalakrishnan R, Hogue O, Floden D, Nagel SJ, Baker KB, Isolan GR, Stefani MA, Machado AG (2020) Cortical thickness in visuo-motor areas is related to motor outcomes after STN DBS for Parkinson’s disease. Parkinsonism Relat Disord 71:17–22
Geraedts VJ, Feleus S, Marinus J, van Hilten JJ, Contarino MF (2020) What predicts quality of life after subthalamic deep brain stimulation in Parkinson’s disease? A systematic review. Eur J Neurol 27(3):419–428
Gonzalez-Escamilla G, Koirala N, Bange M, Glaser M, Pintea B, Dresel C, Deuschl G, Muthuraman M, Groppa S (2022) Deciphering the Network Effects of Deep Brain Stimulation in Parkinson’s Disease. Neurol Ther 11(1):265–282
Groiss SJ, Wojtecki L, Südmeyer M, Schnitzler A (2009) Deep brain stimulation in Parkinson’s disease. Ther Adv Neurol Disord 2(6):20–28
Hartmann CJ, Fliegen S, Groiss SJ, Wojtecki L, Schnitzler A (2019) An update on best practice of deep brain stimulation in Parkinson’s disease. Ther Adv Neurol Disord 12:1756286419838096
Hayasaka S, Phan KL, Liberzon I, Worsley KJ, Nichols TE (2004) Nonstationary cluster-size inference with random field and permutation methods. Neuroimage 22(2):676–687
Hope TMH, Friston K, Price CJ, Leff AP, Rotshtein P, Bowman H (2019) Recovery after stroke: not so proportional after all? Brain 142(1):15–22
Horn A, Reich M, Vorwerk J, Li N, Wenzel G, Fang Q, Schmitz-Hübsch T, Nickl R, Kupsch A, Volkmann J, Kühn AA, Fox MD (2017) Connectivity Predicts deep brain stimulation outcome in Parkinson disease. Ann Neurol 82(1):67–78
Ibarretxe-Bilbao N, Junque C, Segura B, Baggio HC, Marti MJ, Valldeoriola F, Bargallo N, Tolosa E (2012) Progression of cortical thinning in early Parkinson’s disease. Mov Disord 27(14):1746–1753
Jergas H, Petry-Schmelzer JN, Dembek TA, Dafsari HS, Visser-Vandewalle V, Fink GR, Baldermann JC, Barbe MT (2023) Brain Morphometry Associated With Response to Levodopa and Deep Brain Stimulation in Parkinson Disease. Neuromodulation 26(2):340–347
Johnson LA, Wang J, Nebeck SD, Zhang J, Johnson MD, Vitek JL (2020) Direct Activation of Primary Motor Cortex during Subthalamic But Not Pallidal Deep Brain Stimulation. J Neurosci 40(10):2166–2177
Kleiner-Fisman G, Herzog J, Fisman DN, Tamma F, Lyons KE, Pahwa R, Lang AE, Deuschl G (2006) Subthalamic nucleus deep brain stimulation: summary and meta-analysis of outcomes. Mov Disord 21(Suppl 14):S290-304
Koirala N, Fleischer V, Glaser M, Zeuner KE, Deuschl G, Volkmann J, Muthuraman M, Groppa S (2018) Frontal Lobe Connectivity and Network Community Characteristics are Associated with the Outcome of Subthalamic Nucleus Deep Brain Stimulation in Patients with Parkinson’s Disease. Brain Topogr 31(2):311–321
Koivu M, Scheperjans F, Eerola-Rautio J et al (2022) Real-life experience on directional deep brain stimulation in patients with advanced Parkinson's Disease. J Pers Med 12(8):1224. https://doi.org/10.3390/jpm12081224
Krack P, Batir A, Van Blercom N, Chabardes S, Fraix V, Ardouin C, Koudsie A, Limousin PD, Benazzouz A, LeBas JF, Benabid AL, Pollak P (2003) Five-year follow-up of bilateral stimulation of the subthalamic nucleus in advanced Parkinson’s disease. N Engl J Med 349(20):1925–1934
Lachenmayer ML, Mürset M, Antih N, Debove I, Muellner J, Bompart M, Schlaeppi JA, Nowacki A, You H, Michelis JP, Dransart A, Pollo C, Deuschl G, Krack P (2021) Subthalamic and pallidal deep brain stimulation for Parkinson’s disease-meta-analysis of outcomes. NPJ Parkinsons Dis 7(1):77
Lang AE, Houeto JL, Krack P, Kubu C, Lyons KE, Moro E, Ondo W, Pahwa R, Poewe W, Tröster AI, Uitti R, Voon V (2006) Deep brain stimulation: preoperative issues. Mov Disord 21(Suppl 14):S171-196
Lin Z, Zhang C, Li D, Sun B (2022) Preoperative Levodopa Response and Deep Brain Stimulation Effects on Motor Outcomes in Parkinson’s Disease: A Systematic Review. Mov Disord Clin Pract 9(2):140–155
Lozano AM, Lipsman N (2013) Probing and regulating dysfunctional circuits using deep brain stimulation. Neuron 77(3):406–424
Mansouri A, Taslimi S, Badhiwala JH, Witiw CD, Nassiri F, Odekerken VJJ, De Bie RMA, Kalia SK, Hodaie M, Munhoz RP, Fasano A, Lozano AM (2018) Deep brain stimulation for Parkinson’s disease: meta-analysis of results of randomized trials at varying lengths of follow-up. J Neurosurg 128(4):1199–1213
McIntyre CC, Hahn PJ (2010) Network perspectives on the mechanisms of deep brain stimulation. Neurobiol Dis 38(3):329–337
Mueller K, Jech R, Růžička F, Holiga Š, Ballarini T, Bezdicek O, Möller HE, Vymazal J, Růžička E, Schroeter ML, Urgošík D (2018) Brain connectivity changes when comparing effects of subthalamic deep brain stimulation with levodopa treatment in Parkinson’s disease. NeuroImage Clinical 19:1025–1035
Muthuraman M, Deuschl G, Koirala N, Riedel C, Volkmann J, Groppa S (2017) Effects of DBS in parkinsonian patients depend on the structural integrity of frontal cortex. Sci Rep 7:43571
Obeso JA, Marin C, Rodriguez-Oroz C, Blesa J, Benitez-Temiño B, Mena-Segovia J, Rodríguez M, Olanow CW (2008) The basal ganglia in Parkinson’s disease: current concepts and unexplained observations. Ann Neurol 64(Suppl 2):S30-46
Pereira JB, Svenningsson P, Weintraub D, Brønnick K, Lebedev A, Westman E, Aarsland D (2014) Initial cognitive decline is associated with cortical thinning in early Parkinson disease. Neurology 82(22):2017–2025
Price CC, Favilla C, Tanner JJ, Towler S, Jacobson CE, Hass CJ, Foote KD, Okun MS (2011) Lateral ventricle volume is poor predictor of post unilateral DBS motor change for Parkinson’s disease. Parkinsonism Relat Disord 17(5):343–347
Rammo RA, Ozinga SJ, White A, Nagel SJ, Machado AG, Pallavaram S, Cheeran BJ, Walter BL (2022) Directional Stimulation in Parkinson’s Disease and Essential Tremor: The Cleveland Clinic Experience. Neuromodulation 25(6):829–835
Russmann H, Ghika J, Villemure JG, Robert B, Bogousslavsky J, Burkhard PR, Vingerhoets FJ (2004) Subthalamic nucleus deep brain stimulation in Parkinson disease patients over age 70 years. Neurology 63(10):1952–1954
Schade S, Mollenhauer B, Trenkwalder C (2020) Levodopa Equivalent Dose Conversion Factors: An Updated Proposal Including Opicapone and Safinamide. Mov Disord Clin Pract 7(3):343–345
Schnitzler A, Mir P, Brodsky MA, Verhagen L, Groppa S, Alvarez R, Evans A, Blazquez M, Nagel S, Pilitsis JG, Pötter-Nerger M, Tse W, Almeida L, Tomycz N, Jimenez-Shahed J, Libionka W, Carrillo F, Hartmann CJ, Groiss SJ, Glaser M, Defresne F, Karst E, Cheeran B, Vesper J (2022) Directional Deep Brain Stimulation for Parkinson’s Disease: Results of an International Crossover Study With Randomized, Double-Blind Primary Endpoint. Neuromodulation 25(6):817–828
Sestini S, Ramat S, Formiconi AR, Ammannati F, Sorbi S, Pupi A (2005) Brain networks underlying the clinical effects of long-term subthalamic stimulation for Parkinson’s disease: a 4-year follow-up study with rCBF SPECT. J Nucl Med 46(9):1444–1454
Shao MM, Liss A, Park YL, DiMarzio M, Prusik J, Hobson E, Adam O, Durphy J, Sukul V, Danisi F, Feustel P, Slyer J, Truong H, Pilitsis JG (2020) Early Experience With New Generation Deep Brain Stimulation Leads in Parkinson’s Disease and Essential Tremor Patients. Neuromodulation 23(4):537–542
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE (2010) Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord 25(15):2649–2653
Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, Mazoyer B, Joliot M (2002) Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 15(1):273–289
Wang F, Lai Y, Pan Y, Li H, Liu Q, Sun B (2022) A systematic review of brain morphometry related to deep brain stimulation outcome in Parkinson’s disease. NPJ Parkinsons Dis 8(1):130
Weaver FM, Follett K, Stern M, Hur K, Harris C, Marks WJ Jr, Rothlind J, Sagher O, Reda D, Moy CS, Pahwa R, Burchiel K, Hogarth P, Lai EC, Duda JE, Holloway K, Samii A, Horn S, Bronstein J, Stoner G, Heemskerk J, Huang GD (2009) Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: a randomized controlled trial. Jama 301(1):63–73
Welter ML, Houeto JL, Tezenas du Montcel S, Mesnage V, Bonnet AM, Pillon B, Arnulf I, Pidoux B, Dormont D, Cornu P, Agid Y (2002) Clinical predictive factors of subthalamic stimulation in Parkinson’s disease. Brain 125(Pt 3):575–583
Yim Y, Kim SJ, Jung SC, Kim HS, Choi CG, Lee JK, Lee CS, Lee SH, Shim WH, Cheong EN, Park SC (2020) Pretreatment brain volumes can affect the effectiveness of deep brain stimulation in Parkinson’s disease patients. Sci Rep 10(1):22065
Younce JR, Campbell MC, Perlmutter JS, Norris SA (2019) Thalamic and ventricular volumes predict motor response to deep brain stimulation for Parkinson’s disease. Parkinsonism Relat Disord 61:64–69
Acknowledgments
The authors would like to thank neuroradiologist Antti Korvenoja for his expertise and assistance throughout the study.
Funding
Open Access funding provided by University of Helsinki (including Helsinki University Central Hospital). MK has received a grant from Finnish Parkinson Foundation for this study and funding from Government research grant (TYH).
KAMP is supported in part by the Research Council of Finland (grant number 350242), the Sigrid Juselius Foundation and the Finnish Medical Foundation.
EP has received funding from Government research grant (TYH).
Author information
Authors and Affiliations
Contributions
MK: Study design, data collection and analyses, original manuscript drafting and manuscript editing AJS: Study design, data collection and analyses, original manuscript editing JE-R: Manuscript revision KAP: Manuscript revision JR-N: Manuscript revision NV: Manuscript revision RK: Manuscript revision FS: Manuscript revision EP: Study design, original manuscript editing, supervision, manuscript revision
Corresponding author
Ethics declarations
Competing interests
MK: None
AJS: None
JE-R: None
KAMP: None
JR-N: None
NV: None
RK: None
FS: None
EP: None
Additional information
Handling Editor: Cristina Granziera
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koivu, M., Sihvonen, A.J., Eerola-Rautio, J. et al. Clinical and Brain Morphometry Predictors of Deep Brain Stimulation Outcome in Parkinson’s Disease. Brain Topogr 37, 1186–1194 (2024). https://doi.org/10.1007/s10548-024-01054-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-024-01054-2