Abstract
Background
The aim of this study was to assess homologous recombination deficiency (HRD) status and its correlation with carboplatin treatment response in early triple-negative breast cancer (TNBC) patients.
Methods
Tumor tissues from 225 consecutive TNBC patients were evaluated with an HRD panel and homologous recombination-related (HRR) gene expression data. HRD positivity was defined as a high HRD score and/or BRCA1/2 pathogenic or likely pathogenic mutation. Clinicopathological factors, neoadjuvant treatment response, and prognosis were analyzed with respect to HRD status in these TNBC patients.
Results
HRD positivity was found in 53.3% of patients and was significantly related to high Ki67 levels (P = 0.001). In patients who received neoadjuvant chemotherapy, HRD positivity (P = 0.005) or a high HRD score (P = 0.003) was significantly associated with a greater pathological complete response (pCR) rate, especially in those treated with carboplatin-containing neoadjuvant regimens (HRD positivity vs. negativity: 50.00% vs. 17.65%, P = 0.040). HRD positivity was associated with favorable distant metastasis-free survival (hazard ratio HR 0.49, 95% confidence interval CI 0.26–0.90, P = 0.022) and overall survival (HR 0.45, 95% CI 0.20–0.99, P = 0.049), irrespective of carboplatin treatment.
Conclusion
TNBC patients with high HRDs had high Ki67 levels and BRCA mutations. HRD-positive TNBC patients treated with carboplatin had a higher pCR rate. Patients with HRD positivity had a better prognosis, irrespective of carboplatin treatment, warranting further evaluation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the most common malignancy worldwide [1]. Increasing efforts have been made in searching for tools to guide physicians in making precise therapeutic choices and in predicting the prognosis of the disease. Among these new tools, homologous recombination deficiency (HRD), which is known to be a vital motif in the pathogenesis, progression, and treatment efficacy of breast cancer, could be of interest [2]. Mutations in BRCA1 and BRCA2 are the most well-known causes of HRD, and HRD can occur secondary to germline or somatic alterations of other homologous recombination (HR)-related genes or BRCA1 promoter methylation [3].
Current data revealed that HRD positivity occurs in approximately 30% of breast cancer patients, and triple-negative breast cancer (TNBC) has the highest rate of HRD, which is more than 60% according to several small cohort analyses [4, 5]. Through whole-genome sequencing analyses of triple-negative breast cancer (TNBC) patients, Staaf et al. revealed that among TNBC patients with a high HRDetect mutational signature, 67% of cases were caused by germline/somatic BRCA1/2, as well as by other genomic/epigenic abnormalities, such as BRCA1 promoter hypermethylation, RAD51C hypermethylation, or biallelic loss of PALB2, illustrating the existence of many alternative alterations that may lead to HRD tumor status [6]. In a pancancer study based on TCGA data, Su et al. demonstrated inferior overall survival (OS) in HRD score-high patients compared to HRD score-low patients [7]. However, another study based on the Swedish database revealed better invasive disease-free survival (iDFS) in patients with HRD-high tumors than in those with HRD-low tumors [8]. Therefore, the prognostic value of HRD status in TNBC patients remains unclear.
In terms of treatment approaches, homologous recombination repair (HRR) is an important DNA repair pathway for DNA damage and mostly involves DNA double strands and interstrand cross-links [9]. Thus, tumors with HRD are considered to be more genomically unstable and immunogenic and therefore potentially have a greater number of nonsynonymous mutations, leading to more tumor neoantigens [10,11,12]. Given the high prevalence of HRD in TNBC and the mechanism of synthetic lethality, the use of additional therapies targeting HRD, such as platinum salts and poly ADP-ribose polymerase (PARP) inhibitors, has greatly improved the treatment response. In neoadjuvant chemotherapy (NAC), adding carboplatin to anthracycline/taxane significantly increased the pathological complete response (pCR) rate in TNBC patients [13, 14]. However, according to several NAC trials, BRCA mutations were not correlated with a higher pCR rate in TNBC patients receiving platinum agents [15,16,17]. In addition to BRCA mutation, Telli et al. found that a high HRD is associated with a better response to platinum-containing NAC in a pooled analysis of three neoadjuvant trials [18]. Moreover, exploratory analysis of the BrighTNess and GeparSixto trials demonstrated that HRD status was an independent predictor of the pCR rate but not the carboplatin treatment response [19, 20]. Taken together, the current evidence is insufficient to support routine testing of HRD status to guide the use of platinum salt in daily practice.
In the present study, we aimed to analyze the frequency of HRD positivity and to assess the association between HRD status and carboplatin treatment response in early TNBC patients.
Methods
Patients and samples
We retrospectively screened consecutive breast cancer patients treated at the Comprehensive Breast Health Center, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine (RJBC-CBHC) from January 1, 2012 to July 31, 2022. Patients who met the following eligibility criteria were included: (1) had invasive breast cancer, (2) were pathologically diagnosed with TNBC, (3) had available formalin-fixed paraffin-embedded (FFPE) tissues, and (4) were evaluated with HRR genotyping and HRD assays. The exclusion criteria were as follows: (1) male breast cancer, (2) de novo stage IV, and (3) incomplete immunohistochemistry (IHC) information. All tumor size, lymph node status, comorbidities, and adjuvant therapy strategies were permitted.
Archival FFPE blocks were selected from the biobank at the Department of Pathology, Ruijin Hospital. Elaborate clinical data were retrieved from the Shanghai Jiao Tong University Breast Cancer Database (SJTU-BCDB). All patients provided informed consent, and our study was approved by the Ethical Committees of Ruijin Hospital, Shanghai Jiao Tong University School of Medicine. All procedures were in accordance with the 1964 Helsinki Declaration and its later amendments.
Assessment of clinicopathological information
At least two experienced pathologists (A. Li and M. Ruan) from the Department of Pathology, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, contributed to the tumor histopathological analysis. IHC was used to determine the status of the estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER2) as well as the proliferation index (Ki67). ER and PR positivity were defined as no less than 1% stained nuclei, as described in our previous publications [21, 22]. Ki67 > 30% was classified as high expression. HER2 status was classified as “HER2 low” if the IHC results were HER2 1 + or HER2 2 + /fluorescence in situ hybridization (FISH) negative and HER2 0 patients were defined as “HER2 negative.”
Evaluation of the efficacy of neoadjuvant chemotherapy
Among the patients included, patients who underwent neoadjuvant chemotherapy composed the neoadjuvant cohort for neoadjuvant chemotherapy efficacy analysis. The efficacy of neoadjuvant chemotherapy was evaluated according to the pathological result of the final surgical resection sample. A pathological complete response was defined as the disappearance of the target lesion(s).
Follow-up
All patients underwent regular outpatient follow-ups or follow-up calls once every 3 months within the first 2 years after surgery, once every 6 months through the third to the fifth year, and once every year thereafter, in accordance with the American Society of Clinical Oncology Guidelines (ASCO guidelines) [23]. DMFS was defined as the time interval between surgery and the event of distant recurrence, death from breast cancer, death from nonbreast cancer, or death from an unknown cause. OS was defined as the time interval between surgery and death from breast cancer, death from nonbreast cancer or death from an unknown cause [24]. For patients with no events, DMFS and OS were defined as the time interval between surgery and the last follow-up date (June 30, 2020).
HRD Testing
FFPE tumor tissues were stained with hematoxylin and eosin (H&E) and assessed by two pathologists (A. Li, M. Ruan) to determine tumor purity. For patients received neoadjuvant chemotherapy, pre-treatment samples collected by core needle biopsy were used for HRD test. If the tumor purity was ≥ 20%, the tumor tissue was subjected to DNA extraction. The ReliaPrep FFPE gDNA Miniprep System (Promega) was used to extract genomic DNA from FFPE tumor tissues. A total of 100 ng of DNA was used for library construction and hybrid capture with the AmoyDx® HRD complete panel, which selected coding sequence (CDS) regions for 24,000 single-nucleotide polymorphisms (SNPs) and 20 genes (ATM, BARD1, BRCA1, BRCA2, BRIP1, CDH1, CDK12, CHEK1, CHEK2, FANCA, FANCL, HDAC2, PALB2, PPP2R2A, PTEN, RAD51B, RAD51C, RAD51D, RAD54L, TP53). DNA libraries were sequenced on an Illumina NovaSeq 6000 platform with 150 paired-end reads.
The processed raw data were mapped to a reference (human reference genome, hg19) and corrected by UMI for statistical analysis. According to the procedures recommended by the manufacturer [25], a genomic scar (GS) model was built to predict homologous recombination deficiency events via a novel machine learning-based algorithm. A genomic scar score (GSS) ≥ 50.0 was considered to indicate GSS positivity. The gene variations involved in the CDS regions of 20 genes included base substitutions and small insertions and deletions. In addition, 20 gene variants were classified according to the American College of Medical Genetics guidelines [26]. HRD positivity was defined as a GSS ≥ 50.0 and/or BRCA1/2 pathogenic or likely pathogenic mutation.
Statistical analysis
The data distribution was characterized by frequency tabulation and summary statistics. Differences in continuous data were assessed through Student’s t test or the Wilcoxon rank sum test. Differences in categorical data were assessed through the chi-square test or Fisher’s exact test. Correlations between categorical data were assessed through the Spearman correlation coefficient. K‒M curves and the log-rank test were used to compare unadjusted survival between study groups. Cox proportional hazards models were used to evaluate hazard ratios across subgroups and to adjust for patients’ clinicopathological and therapeutic parameters. A two-sided P < 0.05 was considered to indicate statistical significance. All the graphs were generated using GraphPad Prism version 10.0.0 (GraphPad Software, San Diego). All the statistical analyses were performed using Stata version 16.1 (StataCorp LP, College Station, Texas).
Results
Baseline characteristics
A total of 225 patients who received radical surgery (Fig. 1), HRR genotyping, and HRD assays were included in the final analysis, of whom 120 (53.3%) were HRD positive (Table 1). Baseline clinical features and pathological characteristics according to HRD status are shown in Table 1. A higher percentage of HRD-positive patients was observed in TNBC patients younger than 35 (16.67% vs 7.62%). HRD positivity was significantly associated with high tumor grade (71.67% vs 51.43%, P = 0.001) and high Ki67 levels (83.33% vs 54.29%, P < 0.001), indicating more progressive disease. Multivariate analysis revealed that only Ki67 > 30% was independently associated with a high HRD (odds ratio OR 3.21, 95% confidence interval CI 1.66–6.20; P = 0.001).
HRD score and non-HRR gene mutation status
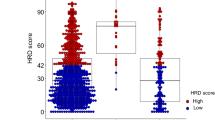
Regarding the non-HRR gene mutations tested in the current panel, 83.11% (n = 187) and 8.00% (n = 18) of patients were found to carry TP53 and PTEN mutations, respectively. TP53 (P < 0.001, Fig. 2A) and PTEN (P < 0.010, Fig. 2B) mutations were significantly related to the HRD score. Compared with wild-type TP53, mutated TP53 was related to a greater HRD score (56.48 ± 6.52 vs 24.86 ± 13.37, P < 0.001), while PTEN-mutated tumors had a lower HRD score than wild-type PTEN (23.17 ± 18.94 vs 53.37 ± 6.28, P = 0.008). Specifically, BRCA-TP53 comutation carriers had significantly greater HRD scores than those who had only TP53 mutations (78.82 ± 12.63 vs 50.97 ± 7.31, P < 0.001; Fig. 2C), while PTEN-TP53 comutation carriers had significantly lower HRD scores than those who had only TP53 mutations (26.04 ± 21.08 vs 59.33 ± 6.76, P < 0.01; Fig. 2D).
Distribution of HRD scores stratified by non-HRR gene mutation status. A Distribution of HRD scores stratified by TP53 mutation status. B Distribution of HRD scores stratified by PTEN mutation status. C Distribution of HRD scores stratified by BRCA-TP53 comutation status. D Distribution of HRD scores stratified by PTEN-TP53 comutation status HRR homologous recombination repair, HRD homologous recombination deficiency *P < 0.05; **P < 0.01; ***P < 0.001; NS not significant
HRD status and neoadjuvant treatment response
Overall, 60 of 225 patients received NAC (Fig. 1) and 15 (25.00%) patients achieved pCR. Both HRD positivity and an HRD score ≥ 50.0 were significantly associated with a higher pCR rate after receiving NAC. The pCR rate was 41.38% in HRD-positive patients and 9.68% in HRD-negative patients (P = 0.005, Fig. 3A). Patients with HRDs ≥ 50.0 had higher pCR rates than did those with HRDs ≤ 50.0 (42.86% vs 9.38%, P = 0.003; Fig. 3B).
Association between HRD and neoadjuvant chemotherapy efficacy. A Distribution of neoadjuvant chemotherapy efficacy stratified by HRD status in all patients (P = 0.005). B Distribution of neoadjuvant chemotherapy efficacy stratified by HRD score groups in all patients (P = 0.003). C Distribution of neoadjuvant chemotherapy efficacy stratified by HRD status in patients treated with carboplatin (P = 0.040). D Distribution of neoadjuvant chemotherapy efficacy stratified by HRD status in patients treated without carboplatin (P = 0.111). HRD homologous recombination deficiency, pCR pathological complete response. *P < 0.05; **P < 0.01; ***P < 0.001; NS not significant
Regarding HRD status and platinum neoadjuvant efficacy, in 37 patients who received carboplatin-containing NAC regimens, HRD positivity was significantly associated with a higher pCR rate (50.00% vs 17.65%, P = 0.040; Fig. 3C), while in the remaining 23 patients who were not treated with carboplatin, only a trend toward a greater pCR rate was observed in HRD-positive patients (25.00% vs 0.00%, P = 0.111; Fig. 3D).
HRD status and survival
After a median follow-up of 50.9 months, 29 deaths and 54 disease relapse events occurred in the whole population. As shown by the Kaplan‒Meier curves in Fig. 4, HRD-positive carriers had a significantly better DMFS (P = 0.040, Fig. 4A) and a trend toward better OS (P = 0.060, Fig. 4B) than HRD-negative carriers. Multivariate analysis revealed that after adjustment for other clinicopathological characteristics including clinical T stage, clinical N stage, and HRD status was independently associated with improved DMFS (hazard ratio HR 0.49, 95% CI 0.26–0.90, P = 0.022; Table 2) and OS (HR 0.45, 95% CI 0.20–0.99, P = 0.049; Table 3). Considering the presence of carboplatin, HRD positivity was associated with a trend toward superior DMFS (with carboplatin, P = 0.235; Fig. 4C; without carboplatin, P = 0.172; Fig. 4E) and OS (with carboplatin, P = 0.053; Fig. 4D; without carboplatin, P = 0.339; Fig. 4F) in the whole population. There was no significant interaction effect on the prognostic value of HRD status and carboplatin treatment (P interaction = 0.730 for DMFS, Fig. 4C; P interaction = 1.000 for OS, Fig. 4D). Moreover, HRD status had a similar effect on OS and DMFS in the other subgroups (all P values > 0.05, Supplementary Fig. S2). Taking neoadjuvant treatment setting into account, we found that HRD status has consistent significant prognostic value not matter in patients received NAC or underwent upfront surgery (DMFS, P for interaction 0.719; OS, P for interaction 0.452, Supplementary Fig. S2).
Prognostic significance of HRD status. A Distant metastasis-free survival in all patients. B Overall survival in all patients. C Distant metastasis-free survival in patients treated with carboplatin. D Overall survival in patients treated with carboplatin. E Distant metastasis-free survival in patients treated without carboplatin. F Overall survival in patients treated without carboplatin. HRD homologous recombination deficiency, DMFS Distant metastasis-free survival, OS Overall survival
Discussion
The present study analyzed the frequency of HRD and its association with carboplatin treatment response in early-stage TNBC patients. We found that HRD was identified in 53.3% of early TNBC patients, which was similar to previous reports [18, 27, 28]. Additionally, we also found that HRD status was significantly associated with high Ki67 levels in TNBC patients, similar to the results reported for breast cancer [5] and other solid tumors [29], which indicated high proliferation potential in HRD cancer cells. Moreover, we found that HRD positivity or a high HRD score was significantly associated with a higher pCR rate, especially in patients treated with carboplatin-containing neoadjuvant regimens. Furthermore, we found that HRD positivity was associated with favorable DMFS and OS, irrespective of carboplatin treatment.
The HRD score was calculated on the basis of loss of heterozygosity, telomeric allelic imbalance, and large-scale state transitions, which indicate the effects of the HRD pathway. HRD can be caused by several genetic and epigenetic changes, including BRCA1/2 gene mutations, BRCA1 promoter methylation, HRR gene mutations, and epigenetic changes in HRR genes [30, 31]. However, the associations between non-BRCA HRR genes and HRD scores have not been well established. Our study analyzed the association of HRR gene mutations with HRD scores. The BRCA1/2-mutated group had a greater HRD score than the wild-type group, indicating the core role of the BRCA protein in maintaining the process of homologous recombination [18]. Interestingly, there was no significant difference in the HRD score between the non-BRCA HRR gene mutation group and the wild-type group, indicating that BRCA1/2 are the predominant driver genes for constructing the HRD score. Our results showed that the genomic level of non-BRCA HRR genes, such as PALB2 and CHEK2, did not greatly contribute to a high HRD score. These results indicated that HRD positivity may also be caused by epigenetic alterations or posttranslational modifications of non-BRCA HRR genes [32,33,34], rather than mutations of these genes, which needs further validation in future studies.
Notably, in the neoadjuvant cohort, patients with HRD-positive tumors had a greater pCR rate than did those with HRD-negative tumors, supporting the superior chemotherapy response and survival benefit in patients with HRD. In the GeparSixto trial, TNBC patients with HRD were independently related to a higher pCR rate after receiving neoadjuvant therapy [20]. Another pooled analysis of five phase II studies also showed that HRD was significantly associated with a higher pCR rate and RCB 0/I in TNBC patients [32]. Thus, HRD status could serve as a potential predictor of NAC efficacy in TNBC patients. Moreover, in patients who received the carboplatin-containing NAC regimen, HRD was significantly associated with a higher pCR rate. Our results were in line with those of the PrECOG 0105 study, which concluded that TNBC patients with higher HRDs were more likely to achieve pCR after neoadjuvant therapy [33]. Increasing evidence has demonstrated that HRD could predict both greater response and survival in favor of carboplatin over other common chemotherapy agents, including docetaxel and epirubicin [34]. There is a strong association between BRCA1 mutation and basal-like cancer [35], which shares similar features with high degrees of chromosomal genomic instability [36], which might indicate the high platinum sensitivity we found in the HRD-positive carriers. However, in patients who received the noncarboplatin NAC regimen, the pCR rate tended to increase in the HRD-positive group, indicating that HRD status was not an ideal factor for predicting carboplatin treatment efficacy.
Regarding prognosis, we found that a high HRD score was associated with favorable disease outcomes in early TNBC patients, which was consistent with the findings of previous studies of other solid tumors, including ovarian cancer [37, 38], colorectal cancer [39], and pancreatic adenocarcinoma [40]. Possible mechanisms could be explained as follows: first, HRD-positive patients showed a better response to chemotherapy, including anthracycline/taxane-based and platinum-containing regimens. Since more than 97% of the enrolled TNBC patients received chemotherapy in the present study, patients with HRD would have a better chemotherapy response and prognosis. Second, patients with high HRDs were found to have greater immune cell infiltration [41, 42]. DNA damage can regulate the cGAS-STING pathway and recruit tumor-infiltrating lymphocytes, consequently activating antitumor immune responses and leading to favorable patient survival [40]. Interestingly, we found that HRD-positive carriers had significantly better DMFS and OS regardless of carboplatin usage. A meta-analysis encompassing more than 300 BRCA-mutated patients demonstrated that germline BRCA mutation carriers would receive no benefit from platinum usage [43], which could be explained by the fact that current standard adjuvant chemotherapy for TNBC patients already contains DNA-damaging agents, such as alkylants or anthracyclines, which may decrease the efficacy of carboplatin in BRCA-mutated patients.
Overall, the main findings of our research have several clinical and translational implications for TNBC treatment. Since there is no officially approved HRD test for breast cancer, our study enrolled a large number of early TNBC patients and supported that the original HRD test system is feasible for these patients. Potential limitations may exist in this study due to its retrospective design. First, our research is only a single-center study that continuously included TNBC patients who have undergone surgery and (neo)adjuvant therapy, and further multicenter studies are needed to confirm the major findings. Second, relatively small amount of patients with special histological type were included in the current analysis and the intrinsic subtype information was also lacked due to retrospective data, and further exploration is needed. Besides, chemotherapy backbone of patients received platinum agents were not consistent in the current study, and sample size was relatively small for further subgroup analysis. Furthermore, since there is no standard method for detecting HRD status in breast cancer patients, comparisons between this original HRD testing method and other detection methods were not performed, and future prospective studies are needed to integrate these methods to evaluate their clinical significance.
Conclusion
High HRDs were related to high Ki67 levels and BRCA mutations in TNBC patients. HRD-positive TNBC patients treated with carboplatin had a higher pCR rate. Patients with HRD positivity had a better prognosis, irrespective of carboplatin treatment, warranting further evaluation.
Data availability
The data analyzed in the current study are available from the corresponding authors on reasonable request.
References
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics 2020. CA Cancer J Clin 70:7–30
Galland L, Roussot N, Desmoulins I et al (2023) Clinical utility of genomic tests evaluating homologous recombination repair deficiency (HRD) for treatment decisions in early and metastatic breast cancer. Cancers. https://doi.org/10.3390/cancers15041299
Stewart MD, Merino Vega D, Arend RC et al (2022) Homologous recombination deficiency: concepts, definitions, and assays. Oncologist 27:167–174
Imanishi S, Naoi Y, Shimazu K et al (2019) Clinicopathological analysis of homologous recombination-deficient breast cancers with special reference to response to neoadjuvant paclitaxel followed by FEC. Breast Cancer Res Treat 174:627–637
Feng C, Zhang Y, Wu F et al (2023) Relationship between homologous recombination deficiency and clinical features of breast cancer based on genomic scar score. Breast 69:392–400
Staaf J, Glodzik D, Bosch A et al (2019) Whole-genome sequencing of triple-negative breast cancers in a population-based clinical study. Nat Med 25:1526–1533
Su R, Liu Y, Wu X et al (2021) Dynamically accumulating homologous recombination deficiency score served as an important prognosis factor in high-grade serous ovarian cancer. Front Mol Biosci 8:762741
Davies H, Glodzik D, Morganella S et al (2017) HRDetect is a predictor of BRCA1 and BRCA2 deficiency based on mutational signatures. Nat Med 23:517–525
O’Kane GM, Connor AA, Gallinger S (2017) Characterization, detection, and treatment approaches for homologous recombination deficiency in cancer. Trends Mol Med 23:1121–1137
Litton JK, Rugo HS, Ettl J et al (2018) Talazoparib in patients with advanced breast cancer and a germline BRCA mutation. New Engl J Med 379:753–763
Nolan E, Savas P, Policheni AN et al (2017) Combined immune checkpoint blockade as a therapeutic strategy for BRCA1-mutated breast cancer. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aal4922
Ballot E, Galland L, Mananet H et al (2022) Molecular intrinsic subtypes, genomic, and immune landscapes of BRCA-proficient but HRD-high ER-positive/HER2-negative early breast cancers. Breast Cancer Res 24:80
Alba E, Chacon JI, Lluch A et al (2012) A randomized phase II trial of platinum salts in basal-like breast cancer patients in the neoadjuvant setting. The results from the GEICAM/2006-03, multicenter study. Breast Cancer Res Treat 136:487–493
Sikov WM, Berry DA, Perou CM et al (2015) Impact of the addition of carboplatin and/or bevacizumab to neoadjuvant once-per-week paclitaxel followed by dose-dense doxorubicin and cyclophosphamide on pathologic complete response rates in stage II to III triple-negative breast cancer: CALGB 40603 (Alliance). J Clin Oncol 33:13–21
Hahnen E, Lederer B, Hauke J et al (2017) Germline Mutation status, pathological complete response, and disease-free survival in triple-negative breast cancer: secondary analysis of the geparsixto randomized clinical trial. JAMA Oncol 3:1378–1385
Loibl S, O’Shaughnessy J, Untch M et al (2018) Addition of the PARP inhibitor veliparib plus carboplatin or carboplatin alone to standard neoadjuvant chemotherapy in triple-negative breast cancer (BrighTNess): a randomized, phase 3 trial. Lancet Oncol 19:497–509
Metzger-Filho O, Collier K, Asad S et al (2021) Matched cohort study of germline BRCA mutation carriers with triple negative breast cancer in brightness. NPJ Breast Cancer 7:142
Telli ML, Timms KM, Reid J et al (2016) Homologous recombination deficiency (HRD) score predicts response to platinum-containing neoadjuvant chemotherapy in patients with triple-negative breast cancer. Clin Cancer Res 22:3764–3773
Telli ML, Metzger O, Timms K et al (2018) Evaluation of homologous recombination deficiency (HRD) status with pathological response to carboplatin +/− veliparib in BrighTNess, a randomized phase 3 study in early-stage TNBC. J Clin Oncol 36:519–519
Loibl S, Weber KE, Timms KM et al (2018) Survival analysis of carboplatin added to an anthracycline/taxane-based neoadjuvant chemotherapy and HRD score as predictor of response-final results from GeparSixto. Ann Oncol 29:2341–2347
Lu Y, Tong Y, Fei X et al (2023) Clinical characteristics, tumor-infiltrating lymphocytes, and prognosis in HER2-low breast cancer: a comparison study with HER2-zero and HER2-positive disease. Cancer Med. https://doi.org/10.1002/cam4.6290
Lu Y, Zhu S, Tong Y et al (2022) HER2-Low status is not accurate in breast cancer core needle biopsy samples: an analysis of 5610 consecutive patients. Cancers. https://doi.org/10.3390/cancers14246200
Khatcheressian JL, Wolff AC, Smith TJ et al (2006) American Society of Clinical Oncology 2006 update of the breast cancer follow-up and management guidelines in the adjuvant setting. J Clin Oncol 24:5091–5097
Tolaney SM, Garrett-Mayer E, White J et al (2021) Updated standardized definitions for efficacy end points (STEEP) in adjuvant breast cancer clinical trials: STEEP version 2.0. J Clin Oncol 39:2720–2731
Yuan W, Ni J, Wen H et al (2022) Genomic scar score: a robust model predicting homologous recombination deficiency based on genomic instability. BJOG 129(Suppl 2):14–22
Richards S, Aziz N, Bale S et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–424
Llop-Guevara A, Loibl S, Villacampa G et al (2021) Association of RAD51 with homologous recombination deficiency (HRD) and clinical outcomes in untreated triple-negative breast cancer (TNBC): analysis of the GeparSixto randomized clinical trial. Ann Oncol 32:1590–1596
Ueno T, Kitano S, Masuda N et al (2022) Immune microenvironment, homologous recombination deficiency, and therapeutic response to neoadjuvant chemotherapy in triple-negative breast cancer: Japan breast cancer research group (JBCRG)22 TR. BMC Med 20:136
An J, Oh JH, Oh B et al (2023) Clinicogenomic characteristics and synthetic lethal implications of germline homologous recombination-deficient hepatocellular carcinoma. Hepatology 78:452–467
Hoppe MM, Sundar R, Tan DSP, Jeyasekharan AD (2018) Biomarkers for homologous recombination deficiency in cancer. J Natl Cancer Inst 110:704–713
van der Wiel AMA, Schuitmaker L, Cong Y et al (2022) Homologous recombination deficiency scar: mutations and beyond-implications for precision oncology. Cancers. https://doi.org/10.3390/cancers14174157
Telli ML, Chu C, Badve SS et al (2020) Association of tumor-infiltrating lymphocytes with homologous recombination deficiency and BRCA1/2 status in patients with early triple-negative breast cancer: a pooled analysis. Clin Cancer Res 26:2704–2710
Telli ML, Jensen KC, Vinayak S et al (2015) Phase II study of gemcitabine, carboplatin, and iniparib as neoadjuvant therapy for triple-negative and BRCA1/2 mutation-associated breast cancer with assessment of a tumor-based measure of genomic instability: PrECOG 0105. J Clin Oncol 33:1895–1901
Tutt A, Tovey H, Cheang MCU et al (2018) Carboplatin in BRCA1/2-mutated and triple-negative breast cancer BRCAness subgroups: the TNT trial. Nat Med 24:628–637
Turner NC, Reis-Filho JS (2006) Basal-like breast cancer and the BRCA1 phenotype. Oncogene 25:5846–5853
Curtis C, Shah SP, Chin SF et al (2012) The genomic and transcriptomic architecture of 2,000 breast tumors reveals novel subgroups. Nature 486:346–352
Garsed DW, Alsop K, Fereday S et al (2018) Homologous recombination DNA repair pathway disruption and retinoblastoma protein loss are associated with exceptional survival in high-grade serous ovarian cancer. Clin Cancer Res 24:569–580
Candido-dos-Reis FJ, Song H, Goode EL et al (2015) Germline mutation in BRCA1 or BRCA2 and ten-year survival for women diagnosed with epithelial ovarian cancer. Clin Cancer Res 21:652–657
Moretto R, Elliott A, Zhang J et al (2022) Homologous recombination deficiency alterations in colorectal cancer: clinical, molecular, and prognostic implications. J Natl Cancer Inst 114:271–279
Park W, Chen J, Chou JF et al (2020) Genomic methods identify homologous recombination deficiency in pancreas adenocarcinoma and optimize treatment selection. Clin Cancer Res 26:3239–3247
Morse CB, Toukatly MN, Kilgore MR et al (2019) Tumor infiltrating lymphocytes and homologous recombination deficiency are independently associated with improved survival in ovarian carcinoma. Gynecol Oncol 153:217–222
Chen Q, Sun L, Chen ZJ (2016) Regulation and function of the cGAS-STING pathway of cytosolic DNA sensing. Nat Immunol 17:1142–1149
Wang CJ, Xu Y, Lin Y et al (2020) Platinum-based Neoadjuvant Chemotherapy for breast cancer with BRCA mutations: A meta-analysis. Front Oncol 10:592998
Acknowledgements
The authors would like to thank the assistance of Ms. Yidong Du in inputting SJTU-BCDB.
Funding
This work was supported by the National Natural Science Foundation of China (82002773, 82072937 and 82072897), the Innovative Research Team of High-Level Local Universities in Shanghai (SHSMU-ZDCX20212200), the Shanghai Municipal Education Commission-Gaofeng Clinical Medicine Grant Support (20172007), and the Natural Science Foundation Project of Science and Technology Commission of Shanghai Municipality (23ZR1439500).
Author information
Authors and Affiliations
Contributions
Conceptualization: Z Wang, L Dong, and X Chen; Data curation: Z Wang, A Li, Y Lu, and C Zhu; Formal analysis: Z Wang, M Han, and C Yang; Funding acquisition: Z Wang, K Shen, and X Chen; Investigation: Z Wang, A Li, Y Lu, M Ruan, and C Zhu; Methodology: Z Wang, A Li, Y Lu, M Ruan, X Zhang, and C Zhu; Project administration: K Shen, L Dong, and X Chen; Resources: C Zhu and K Shen; Software: A Li, M Han, Y Lu, M Ruan, and Y Tong; Supervision: K Shen, L Dong, and X Chen; Validation: Y Lu, M Ruan, and Y Tong; Visualization: Z Wang, M Han, C Yang, and C Zhu; Roles/Writing-original draft: Z Wang, Y Lu, and M Han; Writing-review & editing: K Shen, L Dong, and X Chen.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Ethical approval
This study was approved by the independent Ethical Committees of Ruijin Hospital and Shanghai Jiao Tong University School of Medicine. All procedures were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. (Approval code: 2020-309, date of approval: 17 September 2020).
Consent for publication
Consent for publication from all participants are available from the corresponding authors on reasonable request.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Z., Lu, Y., Han, M. et al. Association between homologous recombination deficiency status and carboplatin treatment response in early triple-negative breast cancer. Breast Cancer Res Treat (2024). https://doi.org/10.1007/s10549-024-07436-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10549-024-07436-1