Abstract
We compared a fast, single breath-hold three dimensional LGE sequence (3D LGE) with an established two dimensional multi breath-hold sequence (2D LGE) and evaluated image quality and the amount of myocardial fibrosis in patients with acute and chronic myocarditis. 3D LGE and 2D LGE (both spatial resolution 1.5 × 1.5 mm2, slice-thickness 8 mm, field of view 350 × 350 mm2) were acquired in 25 patients with acute myocarditis (mean age 40 ± 18 years, 7 female) and 27 patients with chronic myocarditis (mean age 44 ± 22 years, 9 female) on a 1.5 T MR system. Image quality was evaluated by two independent, blinded readers using a 5-point Likert scale. Total myocardial mass, fibrotic mass and total fibrotic tissue percentage were quantified for both sequences in both groups. There was no significant difference in image quality between 3D und 2D acquisitions in patients with acute (p = 0.8) and chronic (p = 0.5) myocarditis. No significant differences between 3D and 2D acquisitions could be shown for myocardial mass (acute p = 0.2; chronic p = 0.3), fibrous tissue mass (acute p = 0.7; chronic p = 0.1) and total fibrous percentage (acute p = 0.4 and chronic p = 0.2). Inter-observer agreement was substantial to almost perfect. Acquisition time was significantly shorter for 3D LGE (24 ± 5 s) as compared to 2D LGE (350 ± 58 s, p < 0.001). In patients with acute and chronic myocarditis 3D LGE imaging shows equal diagnostic quality compared to standard 2D LGE imaging but with significantly reduced acquisition time.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cardiac magnetic resonance imaging (MRI) has an exclusive role in the noninvasive detection of myocarditis [1]. Late gadolinium enhancement (LGE) imaging comprises a high sensitivity for the detection of focal fibrotic tissue, which can be commonly found in myocarditis [2]. In acute myocarditis, clinical presentation and outcome strongly vary, ranging from asymptomatic to fulminant myocarditis with acute heart failure and even sudden cardiac death [3,4,5]. It has been shown that the presence of myocardial fibrosis has diagnostic and prognostic value in patients with acute and chronic myocarditis [6,7,8,9]. After acute inflammation has declined, residual myocardial scars can persist and trigger arrhythmias or cardiovascular complications [10]. Therefore diagnosing fibrous tissue is of critical importance, even if symptoms of acute myocarditis have already vanished or have never occurred in the first place.
Cardiac MRI and especially LGE imaging became a key evaluation tool in the challenging diagnosis of myocarditis and prognosis estimation. While current guidelines still propose histological proof as gold standard for diagnosing acute myocarditis, endomyocardial biopsy is rarely performed in everyday clinical practice [11, 12].
Since CMR is used for risk stratification in patients with suspected myocarditis, patients with suspected acute myocarditis should be examined as timely as possible [13,14,15].
However, due to the relatively long CMR acquisition times and often limited availability of examination slots in the acute hospital setting, further efforts are required to shorten and streamline LGE protocols.
The use of three dimensional (3D), single breath-hold acquisition techniques in LGE imaging has been established in patients with ischemic scars and cardiomyopathies [16,17,18,19], offering abbreviated and simplified exams when compared to two dimensional (2D) LGE protocols. However, the value and utility of 3D LGE remains to be demonstrated in patients with myocarditis.
The purpose of this study was the qualitative and quantitative assessment of LGE in patients with acute and chronic myocarditis using a single breath-hold 3D LGE versus standard multiple breath-hold 2D LGE.
Methods
Study population
Between July 2017 and September 2018 58 patients (mean age 42 ± 18 years, 16 female) with subepicardial or focal midmyocardial fibrosis were included in this retrospective study. Six male patients that additionally showed subendocardial or transmural scars suspective of coronary artery disease were excluded. Twenty-five of the remaining 52 patients showed focal T2 hyperintensity accompanying scar tissue and presented symptoms of acute myocarditis (chest pain, elevated biomarkers, ECG alterations). The remaining 27 patients were categorized as cases with “chronic myocarditis”, because they displayed scars in subepicardial or focal midmyocardial location without edema (“chronic fibrosis”) and showed no acute clinical symptoms. This study was conducted in accordance to the Declaration of Helsinki and its later amendments and was approved by the institutional review board. All included patients gave written informed consent.
CMR data acquisition
CMR was performed on a 1.5 T MR system (Achieva, Philips Healthcare, Best, the Netherlands) using a dedicated 5-channel phased array coil. All data were acquired during breath holding in end expiration. After scout and reference scans, functional and geometric assessment was performed using cine balanced steady- state free precession (SSFP) images in standard long- axis geometries (two-, three- and four-chamber view) as well as in short-axis orientation covering the entire left ventricle (LV) (field of view: 350 × 350 mm2, matrix: 300 × 300, repetition time/echo time: 3.0/1.5 ms, in-plane resolution, 1.5 × 1.5 mm2; number of cardiac phases: 20, section thickness: 8 mm). Edema- sensitive black blood T2-weighted images with and without fat saturation in five short- axis slices were acquired for visualizing myocardial edema [20].
2D LGE images covering the entire LV were acquired 15 min after administration of a bolus of 0.2 mmol of gadobutrol (Gadovist; Bayer Schering Pharma, Zurich, Switzerland) per kilogram body weight. 2D LGE images were acquired in short-axis views by using an inversion recovery gradient-echo sequence: field of view: 350 × 350 mm2; matrix: 256 × 256; repetition time/echo time: 7.4/4.4; inversion time: 190–270 ms (individually optimized with a Look-Locker sequence); flip angle: 20°; in-plane resolution: 1.5 × 1.5 mm2; section thickness: 8 mm. 3D LGE images were acquired with the following parameters: field of view: 350 × 350 mm2; matrix: 256 × 256; repetition time/echo time: 3.6/1.8; inversion time: 190–270 ms (individually optimized with a Look- Locker sequence); flip angle: 15°; in-plane resolution: 1.5 × 1.5 mm2; section thickness: 8 mm.
2D and 3D LGE sequences were performed in random order and the acquisition times for both sequences were measured and noted.
CMR data analysis
For all quantitative analyses, commercially available software (IntelliSpace Portal, Philips, Version 8.0.3) was used. CMR data analysis assessment was performed by an experienced radiologist (4 years of experience in cardiac imaging), blinded to patient characteristics (Table 1). For the acquisition of cardiac volume and function, endocardial contours were drawn in end-systolic and end-diastolic short-axis balanced SSFP images excluding papillary muscle. Subjective image quality analysis and quantitative cardiac MR imaging data analysis of 2D and 3D LGE images were performed in random order. For 33 randomly chosen patients, quantitative analysis of 2D and 3D LGE images was repeated by the first reader after 4 months to assess intra-observer agreement and by a second reader (3 years of experience in cardiac imaging) to assess inter-observer agreement.
Image quality scoring
The image quality of each dataset was graded by the same two blinded independent readers using a five-point Likert scale, in which a score of 1 indicated excellent, a score of 2 good, a score of 3 moderate, a score of 4 poor and a score of 5 nondiagnostic image quality. Reference standard was the 2D LGE image. In case of a Likert score greater than 1 (score: 2–5) the reason for impaired image quality was noted and categorized as being due to motion artifacts, inadequate myocardial nulling, low contrast/high noise or folding artifacts.
Quantitative evaluation
A semi-automatic approach was used to quantify the amount of LGE. Endocardial and epicardial contours were manually drawn on 2D and 3D LGE images and ROIs were placed in hyperenhanced and normal appearing remote myocardium by the software and reviewed by the reader. Subsequently, the areas of unenhanced and hyperenhanced myocardium were automatically segmented by using a full-width at half-maximum algorithm [21]. After segmentation, myocardial and fibrous tissue masses were calculated and the percentage of fibrous tissue mass relative to total myocardial LV mass was computed. The calculated areas of fibrosis as well as myocardial tissue mass were double-checked by the reader and focal corrections could be performed manually if necessary.
Statistical analyses
Continuous data are expressed as means ± standard deviations, and categoric data are expressed as numbers and percentages. Image quality scores of the 2D and 3D data sets were compared by using the Wilcoxon signed rank test. Two-tailed paired t-tests were used to compare normally distributed continuous data and the Wilcoxon signed rank was used for non-parametric distributed continuous data where appropriate. Agreement between the results of the two acquisition techniques regarding total myocardial mass, fibrous mass and percentage of fibrous mass was assessed by using the Pearson correlation coefficient and Bland–Altman analysis with calculation of the limits of agreement (± 1.96 standard deviation). Intra- and inter-observer variability were assessed by calculating Cohen’s kappa coefficients. Kappa coefficients were considered as follows: ‘slight’ < 0.21, ‘fair’ 0.21–0.4, ‘moderate’ 0.41–0.60, ‘substantial’ 0.61–0.80 and ‘almost perfect’ 0.81–1.0 [22]. All statistical analyses were performed using commercially available software (IBM SPSS Statistics, release 25.0; SPSS, Armonk, NY). Statistical significance of difference was assumed for a p-value below 0.05.
Results
Qualitative image analysis
In patients with acute myocarditis, 16 out of 25 (64%) from the 3D datasets and 19 out of 25 (76%) from the 2D datasets had excellent image quality (Likert score: 1) (Figs. 1, 2). Average image quality did not vary significantly between 3D and 2D acquisitions (1.4 ± 0.6 vs. 1.3 ± 0.5, p = 0.8). Inter-observer agreement was substantial (κ = 0.76 and 0.71 for 3D and 2D, respectively).
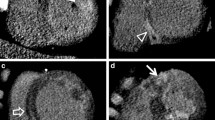
Upper part: Image quality of 3D and 2D LGE datasets of patients with acute myocarditis and chronic inflammatory scars assessed on a 5- point Likert scale from 1 (= excellent image quality) to 5 (= non-diagnostic). Lower part: Short-axis 3D LGE images of three different patients with epicardial fibrosis (arrowheads). Left image demonstrates excellent image quality (= 1), middle image good image quality (= 2, subtle motion artifacts) and right image moderate image quality (= 3, inadequate myocardial nulling, fibrosis still detectable). Poor (score: 4) or non-diagnostic (score: 5) image quality was found in neither acute nor chronic datasets
Short-axis LGE CMR images of a 23-year-old male patient with acute myocarditis. The upper panel shows basal, midventricular and apical sections from 3D LGE acquisition, the middle panel shows the same slices from 2D LGE acquisition and the lower panel shows the same slices in a T2 weighted sequence. High signal intensity in LGE images (arrowheads) indicating epicardial fibrous tissue in basal and midventricular anterolateral/inferolateral segments as well as apical lateral segment could be noted in both acquisitions with concomitant hyperintense signal in T2-weighted images
In patients with chronic fibrosis, 20 out of 27 (74%) from the 3D datasets and 21 out of 27 (78%) from the 2D datasets had excellent image quality (Likert score: 1) (Figs. 1 and 3). There was no significant difference in average image quality between 3D and 2D acquisitions (1.3 ± 0.7 vs. 1.2 ± 0.5, p = 0.5). Inter-observer agreement was substantial to almost perfect (κ = 0.76 and 0.81 for 3D and 2D, respectively).
Short-axis LGE CMR images of a 47-year-old male patient with chronic myocarditis. The upper panel shows basal, midventricular and apical sections from 3D LGE acquisition, the middle panel shows the same slices from 2D LGE acquisition and lower panel shows the same slices in a T2 weighted sequence. High signal intensity in LGE images (arrowheads) indicating epicardial fibrous tissue in basal and midventricular inferior segments could be noted in both acquisitions with no signal alterations in T2-weighted images
Poor (score: 4) or non-diagnostic (score: 5) image quality was found in neither acute nor chronic datasets. Impaired image quality (Likert score 2 and 3) occurred most frequently due to inadequate myocardial nulling and was as frequent in 3D as in 2D datasets (12/52 and 11/52, respectively).
Quantitative image analysis
Acute myocarditis
There were no significant differences between 3D and 2D acquisitions for myocardial mass (111.9 g ± 33.7 vs. 111.4 g ± 34.1, p = 0.2), fibrous tissue mass (9.2 g ± 5.3 vs. 9.2 g ± 5.2, p = 0.7) and total fibrous percentage (8.3% ± 4.8 vs. 8.2% ± 4.6, p = 0.4) (Table 2) with substantial inter-observer agreement (κ = 0.84, 0.76 and 0.74, respectively) and intra-observer agreement (κ = 0.82, 0.80 and 0.79, respectively).
A significant correlation was found between 3D and 2D datasets for myocardial mass (r = 0.91, p = 0.001), fibrous tissue mass (r = 0.99, p = 0.001) and total fibrous percentage (r = 0.95, p = 0.001). Bland–Altman analysis showed good agreement between 3D and 2D datasets for myocardial mass (mean difference: − 0.49 g; limits of agreement: − 4.0 to 3.51 g), fibrous tissue mass (mean difference: 0.01 g; limits of agreement: − 0.8 to 0.81 g) and total fibrous percentage (%) (mean difference: 0.07%; limits of agreement: − 0.9 to 1.1%) (Fig. 4).
Chronic myocarditis
There were no significant differences between 3D and 2D acquisitions for myocardial mass (108.7 g ± 25.9 vs. 109.1 g ± 25.8, p = 0.3), fibrous tissue mass (5.1 g ± 4.7 vs. 5.1 g ± 4.6, p = 0.1) and total fibrous percentage (4.4% ± 4.3 vs. 4.3% ± 3.1, p = 0.2) (Table 2) with substantial or almost perfect inter-observer agreement (κ = 0.75, 0.81 and 0.70, respectively) and intra-observer agreement (κ = 0.78, 0.83 and 0.81, respectively).
A significant correlation was found between 3D and 2D datasets for myocardial mass (r = 0.97, p < 0.001), fibrous tissue mass (r = 0.96, p < 0.001) and total fibrous percentage (r = 0.96, p = 0.001). Bland–Altman analysis showed good agreement between 3D and 2D datasets for myocardial mass (mean difference: 0.31 g; limits of agreement: − 3.13 to 3.75 g), fibrous tissue mass (mean difference: 0.04 g; limits of agreement: − 0.34 to 0.25 g) and total fibrous percentage (mean difference: 0.05%; limits of agreement: − 0.34 to 0.3%) (Fig. 4).
Acquisition time
Acquisition time was significantly shorter for 3D LGE (24 ± 5 s) as compared to 2D LGE (350 ± 58 s, p < 0.001).
Discussion
This study compared a fast, single breath-hold 3D LGE imaging sequence with a clinically established multi breath-hold 2D LGE sequence in patients with acute and chronic myocarditis. In both groups 3D LGE showed equivalent image quality and quantitative information in comparison with the 2D LGE sequence but with significantly reduced acquisition time. Single breath-hold 3D LGE imaging has been established for ischemic scars and cardiomyopathies, providing comparable image quality to multi breath-hold 2D LGE [18, 23, 24].
However, fibrosis in inflammatory disease has lower signal intensity compared with ischemic scars, with usually smaller amounts of fibrotic tissue in typically subepicardial location and lack of concomitant wall motion abnormalities [25, 26]. Optimal image quality of LGE images is of utmost importance in order not to miss these subtle areas of fibrosis and the established multi breath-hold 2D LGE sequences are still preferably used in myocarditis protocols [27,28,29].
All fibrotic areas in patients with acute and chronic myocarditis in 2D LGE were visible in 3D LGE. In patients with acute and chronic myocarditis image quality in 3D datasets was comparable to 2D datasets and inter-observer agreement was substantial to almost perfect. Impaired image quality was mostly due to inadequate myocardial nulling and occurred with same frequency in 3D and 2D datasets.
Quantitative LGE evaluation between 3D and 2D datasets in both patient groups showed no significant differences with substantial to almost perfect inter- and intra-observer agreement, even though some patients—especially those with chronic inflammatory scars—had subtle LGE areas with very low scar burden.
For a short-axis one breath-hold 3D LGE sequence acquisition time was approximately 24 s compared to nearly 6 min for a multi breath-hold 2D acquisition, this is a time reduction of more than 90%. Reduced scan time serves patient comfort and is economically favorable, especially in regard to patients that will receive follow-up MRIs. Moreover, if image quality of LGE is low, e.g. due to wrong inversion time, 3D LGE can easily be repeated without significantly prolonging overall exam times.
Some limitations have to be mentioned. 2D LGE was considered the reference standard for qualitative and quantitative LGE evaluation, but histological proof of LGE areas was not conducted. LGE appearance (subepicardial or focal midmyocardial fibrosis) in 2D LGE and T2-weighted image, respectively, was the main criterion for study inclusion and patients with a history of ischemic heart disease or other non-ischemic cardiomyopathies have not been explicitly excluded.
In conclusion, single breath-hold 3D LGE shows equal diagnostic quality in acute and chronic inflammatory scars compared to standard 2D LGE imaging at significantly reduced acquisition time. Therefore, single breath-hold 3D LGE can be used instead of multi breath-hold 2D LGE in patients with acute and chronic myocarditis.
Abbreviations
- 2D:
-
Two dimensional
- 3D:
-
Three dimensional
- ECG:
-
Electrocardiogramm
- LGE:
-
Late gadolinium enhancement
- LV:
-
Left ventricle/left-ventricular
- LVEDV:
-
Left ventricular end-diastolic volume
- LVEF:
-
Left ventricular ejection fraction
- LVESV:
-
Left ventricular end-systolic volume
- LVSV:
-
Left ventricular stroke volume
- MR/MRI:
-
Magnetic resonance/magnetic resonance imaging
- ms:
-
Milliseconds
- s:
-
Seconds
- SSFP:
-
Steady-state free precession
- T:
-
Tesla
References
Friedrich MG, Sechtem U, Schulz-Menger J, Holmvang G, Alakija P, Cooper LT et al (2009) Cardiovascular magnetic resonance in myocarditis: a JACC white paper. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2009.02.007
Ferreira VM, Schulz-Menger J, Holmvang G, Kramer CM, Carbone I, Sechtem U et al (2018) Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol 72:3158–3176. https://doi.org/10.1016/j.jacc.2018.09.072
Ammirati E, Veronese G, Brambatti M, Merlo M, Cipriani M, Potena L et al (2019) Fulminant versus acute nonfulminant myocarditis in patients with left ventricular systolic dysfunction. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2019.04.063
Sagar S, Liu PP, Cooper LT (2012) Myocarditis. Lancet. https://doi.org/10.1016/S0140-6736(11)60648-X
Berg J, Kottwitz J, Baltensperger N, Kissel CK, Lovrinovic M, Mehra T et al (2017) Cardiac magnetic resonance imaging in myocarditis reveals persistent disease activity despite normalization of cardiac enzymes and inflammatory parameters at 3-month follow-up. Circ Hear Fail. https://doi.org/10.1161/CIRCHEARTFAILURE.117.004262
Grigoratos C, Di Bella G, Aquaro GD (2019) Diagnostic and prognostic role of cardiac magnetic resonance in acute myocarditis. Heart Fail Rev 24:81–90. https://doi.org/10.1007/s10741-018-9724-x
Aquaro GD, Perfetti M, Camastra G, Monti L, Dellegrottaglie S, Moro C et al (2017) Cardiac MR with late gadolinium enhancement in acute myocarditis with preserved systolic function: ITAMY study. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2017.08.044
Lagan J, Schmitt M, Miller CA (2018) Clinical applications of multi-parametric CMR in myocarditis and systemic inflammatory diseases. Int J Cardiovasc Imaging 34:35–54. https://doi.org/10.1007/s10554-017-1063-9
Blissett S, Chocron Y, Kovacina B, Afilalo J (2019) Diagnostic and prognostic value of cardiac magnetic resonance in acute myocarditis: a systematic review and meta-analysis. Int J Cardiovasc Imaging. https://doi.org/10.1007/s10554-019-01674-x
Cooper LT (2009) Myocarditis. N Engl J Med. https://doi.org/10.1056/NEJMra0800028
Friedrich MG, Chetrit M (2018) The unique role of cardiovascular magnetic resonance imaging in acute myocarditis. F1000Research. https://doi.org/10.12688/f1000research.14857.1
Caforio ALP, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB et al (2013) Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. https://doi.org/10.1093/eurheartj/eht210
Sanguineti F, Garot P, Mana M, O’h-Ici D, Hovasse T, Unterseeh T et al (2015) Cardiovascular magnetic resonance predictors of clinical outcome in patients with suspected acute myocarditis. J Cardiovasc Magn Reson. https://doi.org/10.1186/s12968-015-0185-2
Schumm J, Greulich S, Wagner A, Grün S, Ong P, Bentz K et al (2014) Cardiovascular magnetic resonance risk stratification in patients with clinically suspected myocarditis. J Cardiovasc Magn Reson. https://doi.org/10.1186/1532-429X-16-14
Patriki D, Gresser E, Manka R, Emmert MY, Lüscher TF, Heidecker B (2018) Approximation of the incidence of myocarditis by systematic screening with cardiac magnetic resonance imaging. JACC Hear Fail. https://doi.org/10.1016/j.jchf.2018.03.002
Dewey M, Laule M, Taupitz M, Kaufels N, Hamm B, Kivelitz D (2007) Myocardial viability: assessment with three-dimensional MR imaging in pigs and patients. Radiology. https://doi.org/10.1148/radiol.2393050586
Goetti R, Kozerke S, Donati OF, Sürder D, Stolzmann P, Kaufmann PA et al (2011) Acute, subacute, and chronic myocardial infarction: quantitative comparison of 2D and 3D late gadolinium enhancement MR imaging. Radiology. https://doi.org/10.1148/radiol.11102216
Peters DC, Appelbaum EA, Nezafat R, Dokhan B, Han Y, Kissinger KV et al (2009) Left ventricular infarct size, peri-infarct zone, and papillary scar measurements: a comparison of high-resolution 3D and conventional 2D late gadolinium enhancement cardiac MR. J Magn Reson Imaging. https://doi.org/10.1002/jmri.21897
Morita K, Utsunomiya D, Oda S, Komi M, Namimoto T, Hirai T et al (2013) Comparison of 3D phase-sensitive inversion-recovery and 2D inversion-recovery MRI at 3.0 T for the assessment of late gadolinium enhancement in patients with hypertrophic cardiomyopathy. Acad Radiol. https://doi.org/10.1016/j.acra.2013.01.014
Simonetti OP, Finn JP, White RD, Laub G, Henry DA (1996) “Black blood” T2-weighted inversion-recovery MR imaging of the heart. Radiology. https://doi.org/10.1148/radiology.199.1.8633172
Amado LC, Gerber BL, Gupta SN, Rettmann DW, Szarf G, Schock R et al (2004) Accurate and objective infarct sizing by contrast-enhanced magnetic resonance imaging in a canine myocardial infarction model. J Am Coll Cardiol. https://doi.org/10.1016/j.jacc.2004.09.020
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics. https://doi.org/10.2307/2529310
Foo TKF, Stanley DW, Castillo E, Rochitte CE, Wang Y, Lima JAC et al (2004) Myocardial viability: breath-hold 3D MR imaging of delayed hyperenhancement with variable sampling in time. Radiology. https://doi.org/10.1148/radiol.2303021411
Morsbach F, Gordic S, Gruner C, Niemann M, Goetti R, Gotschy A et al (2016) Quantitative comparison of 2D and 3D late gadolinium enhancement MR imaging in patients with Fabry disease and hypertrophic cardiomyopathy. Int J Cardiol. https://doi.org/10.1016/j.ijcard.2016.04.175
Cummings KW, Bhalla S, Javidan-Nejad C, Bierhals AJ, Gutierrez FR, Woodard PK (2009) A pattern-based approach to assessment of delayed enhancement in nonischemic cardiomyopathy at MR imaging. Radiographics. https://doi.org/10.1148/rg.291085052
Mahrholdt H, Goedecke C, Wagner A, Meinhardt G, Athanasiadis A, Vogelsberg H et al (2004) Cardiovascular magnetic resonance assessment of human myocarditis: a comparison to histology and molecular pathology. Circulation. https://doi.org/10.1161/01.CIR.0000118493.13323.81
Muehlberg F, Arnhold K, Fritschi S, Funk S, Prothmann M, Kermer J et al (2018) Comparison of fast multi-slice and standard segmented techniques for detection of late gadolinium enhancement in ischemic and non-ischemic cardiomyopathy: a prospective clinical cardiovascular magnetic resonance trial. J Cardiovasc Magn Reson. https://doi.org/10.1186/s12968-018-0434-2
Gallegos C, Rottmann D, Nguyen VQ, Baldassarre LA (2019) Myocarditis with checkpoint inhibitor immunotherapy: case report of late gadolinium enhancement on cardiac magnetic resonance with pathology correlate. Eur Hear J Case Rep. https://doi.org/10.1093/ehjcr/yty149
Ammirati E, Moroni F, Sormani P, Peritore A, Milazzo A, Quattrocchi G et al (2017) Quantitative changes in late gadolinium enhancement at cardiac magnetic resonance in the early phase of acute myocarditis. Int J Cardiol. https://doi.org/10.1016/j.ijcard.2016.11.282
Funding
Open access funding provided by University of Zurich
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors of this manuscript has declared any conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Polacin, M., Kapos, I., Gastl, M. et al. Comparison of 3D and 2D late gadolinium enhancement magnetic resonance imaging in patients with acute and chronic myocarditis. Int J Cardiovasc Imaging 37, 305–313 (2021). https://doi.org/10.1007/s10554-020-01966-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-020-01966-7