Abstract
Background
The pathogenic processes in the preclinical phase of inflammatory bowel disease (IBD) are mainly unknown.
Aims
To study typical antibodies for IBD in the preclinical phase in a cohort of Northern Sweden.
Methods
Antibodies typical for IBD (ASCA, pANCA, lactoferrin-ANCA, antibodies to goblet cells, and pancreas antigen) were analyzed in 123 subjects with preclinical ulcerative colitis (UC), 54 subjects with preclinical Crohn's disease (CD) and in 390 sex- and age-matched controls. In addition, in a subset of subjects, inflammatory markers (CRP, albumin, calprotectin and ferritin) were measured in plasma.
Results
The mean years between blood samples and IBD diagnosis were for UC 5.1 (SD 3.5) years and CD 5.6 (SD 3.5) years. There was no difference in the proportion of overall positive antibodies between subjects who later developed IBD compared to controls (16.9% vs. 12.3%; p = 0.137). The subjects who later developed CD had a significantly higher proportion of positive ASCA compared to controls (9.3% vs 2.8%; p = 0.034), but for all other antibodies, there were no differences compared to control subjects. Subjects with preclinical IBD and elevated antibodies showed significantly higher plasma calprotectin levels compared to subjects without antibodies (980 μg/L vs 756 μg/L; p = 0.042), but there was no difference in the levels of CRP, albumin and ferritin.
Conclusions
We found no significant increase in antibodies typical for IBD years before diagnosis except for ASCA, which was slightly more common in subjects who later developed CD. Very few subjects had detectable antibodies to goblet cells and pancreas antigen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Inflammatory bowel disease, including ulcerative colitis (UC) and Crohn's disease (CD), is a chronic and clinical relapsing disease [1] with an estimated prevalence in Sweden of 0.65% [2]. The cause of IBD is not fully known, but according to the present knowledge, the pathogenesis involves an autoimmune response to the gut mucosa where genetic factors, environmental factors and microbiota contribute and interact with the immune system [3]. Several antibodies have been associated with IBD. Anti-Saccharomyces cerevisiae antibodies (ASCA), an antibody directed to the mannan protein from the yeast Saccharomyces cerevisiae, has been detected in up to 40–70% of patients with CD [4,5,6]. An antibody directed to the nuclear lamina of neutrophils named perinuclear antineutrophil cytoplasmic antibody (pANCA) has been found to be abundant in patients with UC, with a prevalence of approximately 40–70% [7]. Besides ASCA and pANCA, several other antibodies have been associated with IBD. For example, there are increased levels of antibodies against DNA-bound lactoferrin (LFS-ANCA) in patients with UC [8]. Lactoferrin is an iron-binding protein produced in neutrophil granulocytes and secretory epithelial cells. Goblet cells are responsible for producing the protective mucus layer by secreting mucins. Autoantibodies against intestinal goblet cells have been associated with inflammatory bowel disease [9,10,11]. Also, antibodies against exocrine pancreatic antigens (rPag1 and rPag2) have been associated with IBD [10, 12] and is also elevated in unaffected first relatives to patients with IBD [13]. rPag1 is CUB (C1r/C1s, Uegf, BMp1) and zona pellucida-like domain containing protein-1 (CUZD1) and rPag 2 is synonymous to Glycoprotein-2 (GP2) [14].
However, the role of the mentioned antibodies in the pathogenesis of IBD is unknown. The presence of p-ANCA has been associated with a more unfavorable disease course in UC [15] and ASCA and other antibodies with a more complicated CD [9, 16, 17]. Studying the presence of antibodies in the preclinical phase of IBD is one way to better understand the role of antibodies in the pathogenesis of IBD. In the literature, some studies used this approach [16, 18,19,20], and these studies have demonstrated that a significant number of patients have detected antibodies years before diagnosis.
The prevalence of antibodies might differ between ethnically divergent populations [7]. This study aimed to explore to what extent antibodies associated with IBD are present in a Swedish population. In addition to ASCA and pANCA, we also analyzed antibodies to LFS-ANCA, goblet cells and recombinant pancreas antigen 1 and 2 (rPag1, rPag2). A secondary aim was to study if subjects with detected antibodies also had increased inflammatory markers.
Methods
Study Population
Study participants in this nested case–control study were recruited from the Västerbotten intervention project (VIP) [21], the Mammography screening project and the Northern Sweden Maternity cohort [22]. The VIP, started in 1985, is a large health survey focused on the prevention of cardiovascular disease. All residents of the Region of Västerbotten are offered to participate in the study the year they turn 30, 40, 50 and 60 years. In VIP, blood samples from over 150 000 visits are stored in a central biobank. The Mammography screening project invited women in the Region of Västerbotten who were scheduled for mammography screening between 1995 and 2006 and includes approximately 54,000 blood samples. The Maternity cohort comprises blood samples from pregnant women from the Northern part of Sweden (including the Region of Västerbotten) who had been screened for Rubella since 1975, and this biobank includes approximately 70,000 serum samples.
All study participants were linked through the National Patient Registry for IBD diagnosis according to the International Classification of Diseases (Codes K.50.1-9 and K51.0-9). Medical records were then checked for all patients coded for IBD to verify the diagnosis and classify the person according to the Montreal Classification for IBD [23]. The date of diagnosis was set when the patient, for the first time, demonstrated macroscopic and/or histologic evidence of IBD (i.e. endoscopy or radiologic evidence for IBD). Subjects diagnosed with IBD before or within one year after he or she donated blood samples were excluded from the study. For each case, two controls, matched for age, gender, time, center and each study cohort were randomly selected among persons not diagnosed with IBD. Table 1 shows characteristics of the including patients from each project.
Blood Samples Collection and Blood Analyses
In VIP and the Mammography cohort, venous blood samples were obtained after overnight fasting. Subjects rested for 15 min before sampling. The blood was collected in 10 mL Na-heparin tubes, centrifuged at 1500 G for 15 min, and aliquots of plasma were frozen within 1 hour and stored at − 80 °C. In the Maternity cohort, venous blood samples were obtained in clinical routine, and sera was frozen at − 20 °C.
Plasma samples were diluted 1:10 in phosphate-buffered saline for analyses of antibodies against goblet cells, rPAg1, rPAg2, pANCA, and Anti-DNA bound lactoferrin. For analyses of antibodies against Saccharomyces cerevisiae IgG, plasma was diluted 1:1000 in phosphate-buffered saline according to the manufacturer's description. The samples, including positive and negative controls, were incubated with fixed antigens of goblet cells, rPAg1, rPAg2, pANCA, and Anti-DNA bound lactoferrin, neutrophils and Saccharomyces cerevisiae (CIBD-Mosaics, Euroimmun, Lübeck, Germany) for 30 min. Slides were then rinsed, submerged in phosphate-buffered saline for 5 min, and then incubated with fluorescent conjugate for 30 min in a chamber. Slides were washed again as described above, and coverslips were applied with a mounting medium (Euroimmun, Lübeck, Germany) for analyses in a fluorescence microscope (Leica DC 300F). All lab work was performed by the same two individuals (LBZ, CE). The same two persons assessed all the samples by the microscope and were blinded to the case/control status of the participants. Samples were pooled in pairs and analyzed. All pools that were not clearly negative were further analyzed separately.
For subjects in the VIP and the Mammography cohort, we also had data from previous studies performed in these cohorts for albumin, CRP, ferritin and calprotectin in plasma samples [24, 25].
Statistical Analyses
IBM SPSS version 27.0 was used for statistical analyses. Continuous data, such as age, time to diagnosis, body mass index, and inflammatory markers, are presented as means with standard deviations. To compare continuous data Student's t test was used. Categorical data are presented as frequencies and were compared using Person chi2 test or Fisher's exact test when appropriate. p values < 0.05 were considered statistically significant. There were no corrections for multiple testing.
Ethics
The study was approved by the regional ethical board, Umeå, Sweden (Dnr 06-024M, 2010 284-31M). The participants gave written consent before they participated in the projects. There were no interventions related to this study.
Results
Baseline Characteristics
Table 1 shows the characteristics of each study cohort, and Table 2 shows the baseline characteristics of all the subjects included in the study. The youngest person in the study was 17 years old, and there were only subjects with onset of IBD after the age of 18 years (Montreal A2 and A3) included in the study. There were significantly more smokers among the subjects that later developed CD compared to subjects that later developed UC (p < 0.001) and controls (p < 0.001).
Among the controls, 45 persons (11.5%) had one positive antibody, and three (0.8%) had two positive antibodies. In subjects who later developed UC, 17 patients (13.8%) had one positive antibody, and one patient (0.8%) had three positive antibodies. Among the subjects who later developed CD and with positivity for antibodies, no one had more than one positive antibody. The number of subjects with any antibody in case/controls for each study cohort were: VIP 12/15, Mammography screening project 1/4 and the Maternity cohort 17/29.
The Prevalence of Antibodies in Preclinical IBD
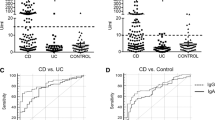
The proportion of overall positive antibodies was not significantly higher in subjects who later developed IBD compared to controls, and the difference was only approximately 6% (Table 3). The risk for a person with any antibody to develop IBD was low (odds ratio 1.45; 95th confidence interval 0.88–2.39). The subjects who later developed CD had a significantly higher proportion of positive ASCA and a significantly higher proportion of overall positive antibodies compared to controls, but for all other antibodies, there were no differences compared to control subjects.
Figure 1 shows the cumulative proportion of subjects who later developed IBD with positive antibodies at different time points before diagnosis. There was no difference in median time from sample to diagnosis between subjects with and without antibodies and who later developed IBD (5.0 years vs 5.0 years, p = 0.758). The prevalence of antibodies was at similar levels from eight years to 1 year before subjects were diagnosed with IBD.
Factors Associated with Antibodies for IBD
Among the cases, there were no gender differences in the proportion of any of the antibodies (women vs men; 17.5% vs 15.7%; p = 0.829), but among the controls, women had a higher rate of antibodies (15.6% vs 3.7%; p = 0.001).
There was no difference in the presence of antibodies between patients with preclinical IBD with a sample taken before or after 40 years of age (18.9% vs 15.0%; p = 0.490) or between patients with IBD diagnosis before 40 years (Montreal classification A2) and after 40 years (Montreal classification A3) (20.2% vs 13.6%; p = 0.243).
There was no difference in the proportion of smokers among subjects who later developed IBD with and without antibodies (25.0% vs 32.1%; p = 0.510). In addition, among the controls, there were no differences in smoking between subjects with antibodies versus no antibodies (8.3% vs 12.6%; p = 0.481).
The subjects who later developed IBD and controls with and without any positivity for antibodies did not differ in the level of inflammatory markers in plasma, except for mean plasma calprotectin, which was significantly higher among the subjects who later developed IBD (Table 4). The subjects with detected antibodies did not differ regarding disease behavior, location and extension of IBD compared to those without antibodies.
Discussion
This study aimed to explore the presence of antibodies typical for IBD in subjects several years before they were diagnosed with IBD. We found that the total number of patients with any antibody was significantly higher than in controls, but separately only ASCA showed a significant increase (in patients with CD) compared to control subjects. In addition, we found that the difference in the proportion of detected antibodies between subjects who later developed IBD and controls was only modest (6%).
Overall, the proportion of patients with IBD who had antibodies before diagnosis in our study was lower compared to earlier published studies (Table 5). The choice of cut-off levels, handling of samples, the age difference, differences in sample time in relation to diagnosis, and the choice of “wash-out period” before the diagnosis might partly explain the differences between studies. For example, in the study by van Schaik et al. [19], lower cut-off levels were used for the antibodies compared to that recommended for clinical practice. In the study of van Schaik et al. [19] and by Israeli et al. [18], the risk of being diagnosed with IBD for subjects with elevated antibodies was most prominent the three years before diagnosis, but Choung et al. [16] could demonstrate a high proportion of antibodies six years before clinical diagnosis. In addition, none of the previous studies excluded patients tested within 1 year before diagnosis. In our study, a “wash-out period” of one year before diagnosis was used, the same increase in the detection of antibodies toward the time of diagnosis was not seen, and the prevalence of antibodies was stable from approximately eight years to 1 year before diagnosis.
Choung et al. showed that patients with CD who had antibodies before diagnosis also had a more complicated disease[16]. This association could not be confirmed in our study, but the number of patients with positive antibodies was lower than in the study by Choung et al., and we also lacked longitudinal samples.
We found a significantly higher proportion of ASCA in subjects that later developed CD than in control subjects. The presence of ASCA is also common in patients with an established CD diagnosis and first-degree relatives of patients with CD [26,27,28]. The role of Saccharomyces cerevisiae in the pathogenesis of CD is unclear. This yeast species is considered an opportunistic low virulent pathogen and widespread at home (baking) and in the food industry. Approximately, 6% of the normal population has Saccharomyces cerevisiae in the digestive tract [29]. However, because Saccharomyces cerevisiae originates from food and its occurrence in the gastrointestinal tract is transient, it has been debated if the yeast should be considered to belong to the commensal flora of the gut or not [29, 30]. Saccharomyces cerevisiae is rarely found among the CD patients with ASCA, indicating that these antibodies exist without continuous exposure to Saccharomyces cerevisiae [31]. Previously, an increased gut permeability has been suggested to be involved in the production of ASCA in patients with CD, but this hypothesis has later been questioned since no association has been seen between ASCA levels and the degree of gut permeability in patients with CD [26].
Except for ASCA, no other antibody alone was associated with developing IBD in our study. This is consistent with a recent study by Torres et al. [20], who found that no single specific antibody, including pANCA, the most specific marker for UC, could accurately predict UC.
To our knowledge, this is the first study that explores the presence of goblet cell antibodies and antibodies to pancreas antigen in the preclinical phase of IBD. We found that the number of these antibodies was low in our study. Antibodies against the exocrine pancreas have been associated with younger age at onset of IBD [32]. In our study, a higher proportion of patients with later onset could perhaps explain the low prevalence of pancreatic antigen antibodies.
We investigated the plasma concentration of inflammatory markers in subjects with and without antibodies. In the subjects who later developed IBD, the mean plasma calprotectin levels were significantly higher in the persons with detected antibodies compared to persons without antibodies, but there were no differences in the levels of CRP, albumin or ferritin. This finding may reflect that antibody positivity is associated with subclinical systemic inflammation in the preclinical phase of IBD, but need to be tested in a larger cohort.
There are some limitations to our study. First, our data is based on a single sample for each subject, and we lack longitudinal information on the serological markers. Second, our study includes three cohorts with different characteristics. For example, VIP and the Mammography screening cohort mainly include older subjects and the Mammography screening cohort and the Maternity cohort only include women. The sample in the Maternity cohort is performed during pregnancy which can influence antibody levels. All samples had been frozen for several years, which could impact the quality of the sera. In addition, other antibodies not analyzed in our study have been associated with IBD [7]. For example, antibodies against the outer membrane protein (OmpC) of Escherichia coli and antibodies against subtypes of flagellins (anti-FlaX, anti-Fla2, anti-CBir1) have been shown to be increased in patients with CD years before diagnosis [16].
To conclude, as in line with previous studies, we found an increase in antibodies typical for IBD years before diagnosis. However, in our population, the increase was only modest. Very few subjects had detectable antibodies to goblet cells and pancreas antigen.
Data accessibility
Anonymized subject-level data can be made available upon request after approval by the Department of Biobank Research at Umeå University.
References
Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:s1–s106.
Busch K, Ludvigsson JF, Ekstrom-Smedby K, Ekbom A, Askling J, Neovius M. Nationwide prevalence of inflammatory bowel disease in Sweden: a population-based register study. Aliment Pharmacol Ther. 2014;39:57–68.
Chang JT. Pathophysiology of Inflammatory Bowel Diseases. N Engl J Med. 2020;383:2652–2664.
Sendid B, Colombel JF, Jacquinot PM, Faille C, Fruit J, Cortot A et al. Specific antibody response to oligomannosidic epitopes in Crohn’s disease. Clin Diagn Lab Immun. 1996;3:219–226.
Quinton JF, Sendid B, Reumaux D, Duthilleul P, Cortot A, Grandbastien B et al. Anti-Saccharomyces cerevisiae mannan antibodies combined with antineutrophil cytoplasmic autoantibodies in inflammatory bowel disease: prevalence and diagnostic role. Gut. 1998;42:788–791.
Walker LJ, Aldhous MC, Drummond HE, Smith BRK, Nimmo ER, Arnott IDR et al. Anti-Saccharomyces cerevisiae antibodies (ASCA) in Crohn’s disease are associated with disease severity but not NOD2/CARD15 mutations. Clin Exp Immunol. 2004;135:490–496.
Prideaux L, De Cruz P, Ng SC, Kamm MA. Serological antibodies in inflammatory bowel disease: a systematic review. Inflamm Bowel Dis. 2012;18:1340–1355.
Peen E, Almer S, Bodemar G, Ryden BO, Sjolin C, Tejle K et al. Anti-Lactoferrin Antibodies and Other Types of Anca in Ulcerative-Colitis, Primary Sclerosing Cholangitis, and Crohns-Disease. Gut. 1993;34:56–62.
Papp M, Altorjay I, Dotan N, Palatka K, Foldi I, Tumpek J et al. New Serological Markers for Inflammatory Bowel Disease Are Associated With Earlier Age at Onset, Complicated Disease Behavior, Risk for Surgery, and NOD2/CARD15 Genotype in a Hungarian IBD Cohort. American Journal of Gastroenterology. 2008;103:665–681.
Homsak E, Micetic-Turk D, Bozic B. Autoantibodies pANCA, GAB and PAB in inflammatory bowel disease: prevalence, characteristics and diagnostic value. Wien Klin Wochenschr. 2010;122:19–25.
Kovacs M, Lakatos PL, Papp M, Jacobsen S, Nemes E, Polgar M et al. Pancreatic Autoantibodies and Autoantibodies Against Goblet Cells in Pediatric Patients With Inflammatory Bowel Disease. J Pediatr Gastr Nutr. 2012;55:429–435.
Lawrance IC, Hall A, Leong R, Pearce C, Murray K. A comparative study of goblet cell and pancreatic exocine autoantibodies combined with ASCA and pANCA in Chinese and Caucasian patients with IBD. Inflammatory Bowel Diseases. 2005;11:890–897.
Joossens S, Vermeire S, Van Steen K, Godefridis G, Claessens G, Pierik M et al. Pancreatic autoantibodies in inflammatory bowel disease. Inflammatory Bowel Diseases. 2004;10:771–777.
Papp M, Sipeki N, Tornai T, Altorjay I, Norman GL, Shums Z et al. Rediscovery of the Anti-Pancreatic Antibodies and Evaluation of their Prognostic Value in a Prospective Clinical Cohort of Crohn’s Patients: The Importance of Specific Target Antigens [GP2 and CUZD1]. J Crohns Colitis. 2015;9:659–668.
Hoie O, Aamodt G, Vermeire S, Bernklev T, Odes S, Wolters FL, et al. Serological markers are associated with disease course in ulcerative colitis. A study in an unselected population-based cohort followed for 10 years. Journal of Crohns & Colitis. 2008;2:114–22.
Choung RS, Princen F, Stockfisch TP, Torres J, Maue AC, Porter CK et al. Serologic microbial associated markers can predict Crohn’s disease behaviour years before disease diagnosis. Aliment Pharmacol Ther. 2016;43:1300–1310.
Ferrante M, Henckaerts L, Joossens M, Pierik M, Joossens S, Dotan N et al. New serological markers in inflammatory bowel disease are associated with complicated disease behaviour. Gut. 2007;56:1394–1403.
Israeli E, Grotto I, Gilburd B, Balicer RD, Goldin E, Wiik A et al. Anti-Saccharomyces cerevisiae and antineutrophil cytoplasmic antibodies as predictors of inflammatory bowel disease. Gut. 2005;54:1232–1236.
van Schaik FD, Oldenburg B, Hart AR, Siersema PD, Lindgren S, Grip O et al. Serological markers predict inflammatory bowel disease years before the diagnosis. Gut. 2013;62:683–688.
Torres J, Petralia F, Sato T, Wang P, Telesco SE, Choung RS et al. Serum Biomarkers Identify Patients Who Will Develop Inflammatory Bowel Diseases Up to 5 Years Before Diagnosis. Gastroenterology. 2020;159:96–104.
Norberg M, Wall S, Boman K, Weinehall L. The Vasterbotten Intervention Programme: background, design and implications. Global Health Action. 2010;3.
Rantapaa-Dahlqvist S, de Jong BAW, Berglin E, Hallmans G, Wadell G, Stenlund H et al. Antibodies against cyclic citrullinated peptide and IgA rheumatoid factor predict the development of rheumatoid arthritis. Arthritis Rheum. 2003;48:2741–2749.
Satsangi J, Silverberg MS, Vermeire S, Colombel JF. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut. 2006;55:749–753.
Widbom L, Ekblom K, Karling P, Hultdin J. Patients developing inflammatory bowel disease have iron deficiency and lower plasma ferritin years before diagnosis: a nested case-control study. Eur J Gastroen Hepat. 2020;32:1147–1153.
Lundgren D, Widbom L, Hultdin J, Karling P. Preclinical Markers in Inflammatory Bowel Disease. A Nested Case-Control Study. Crohns Colitis 360. 2021 Oct;3(4).
Vermeire S, Peeters M, Vlietinck R, Joossens S, Den Hond E, Bulteel V et al. Anti-Saccharomyces cerevisiae antibodies (ASCA), phenotypes of IBD, and intestinal permeability: A study in IBD families. Inflammatory Bowel Diseases. 2001;7:8–15.
Annese V, Andreoli A, Andriulli A, D’Inca R, Gionchetti P, Latiano A et al. Familial expression of anti-Saccharomyces cerevisiae mannan antibodies in Crohn’s disease and ulcerative colitis: A GISC study. American Journal of Gastroenterology. 2001;96:2407–2412.
Seibold F, Stich O, Hufnagl R, Kamil S, Scheurlen M. Anti-Saccharomyces cerevisiae antibodies in inflammatory bowel disease: a family study. Scand J Gastroentero. 2001;36:196–201.
Hallen-Adams HE, Suhr MJ. Fungi in the healthy human gastrointestinal tract. Virulence. 2017;8:352–358.
Enache-Angoulvant A, Hennequin C. Invasive Saccharomyces infection: A comprehensive review. Clin Infect Dis. 2005;41:1559–1568.
Mallant-Hent RC, Mooij M, von Blomberg BME, Linskens RK, van Bodegrave AA, Savelkoul PHM. Correlation between Saccharomyces cerevisiae DNA in intestinal mucosal samples and anti-Saccharomyces cerevisiae antibodies in serum of patients with IBD. World J Gastroentero. 2006;12:292–297.
Pavlidis P, Shums Z, Koutsoumpas AL, Milo J, Papp M, Umemura T et al. Diagnostic and clinical significance of Crohn’s disease-specific anti-MZGP2 pancreatic antibodies by a novel ELISA. Clin Chim Acta. 2015;20:176–181.
Acknowledgments
We thank everyone who participated in the NSHDS for contributing to the study. We also like to thank the Department of Biobank Research at Umeå University, the Västerbotten Intervention Project and the Region of Västerbotten for providing data and samples and acknowledge the contribution from Biobank Sweden.
Funding
Open access funding provided by Umea University. This work was supported by the Region of Västerbotten, Biobank Sweden supported by the Swedish Research Council (VR 2017-00650) and grants from Mag-Tarmfonden, Sweden.
Author information
Authors and Affiliations
Contributions
PK, JH and CH designed the trial. LBZ, PK and LW analyzed the data. LBZ and PK drafted the manuscript, but all authors contributed to the final draft of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Bodecker-Zingmark, L., Widbom, L., Hultdin, J. et al. Anti-Saccharomyces cerevisiae Antibodies Are Only Modestly More Common in Subjects Later Developing Crohn's Disease. Dig Dis Sci 68, 608–615 (2023). https://doi.org/10.1007/s10620-022-07630-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-022-07630-5