Abstract
In cocoa agroforestry systems, cycling of leaves, pods, and branches are key for organic matter sustenance. We investigated annual total litterfall, annual nutrient stocks in total litterfall, cocoa pods and beans, as well as cocoa leaf decomposition rates in cocoa agroforestry systems under conventional and organic management in Suhum Municipality, Eastern Region of Ghana. The study was conducted using six cocoa agroforests for each management selected from a total of four villages. Litterfall was collected monthly using litterboxes and a litterbag technique was employed to study the rates of leaf decomposition and nutrient release for 12 months. In June and July, total litterfall in organic farms were 94% and 65%, respectively, higher than in conventional farms, but management had no effect on average annual total litterfall of 8.8 t ha−1 yr−1 litterfall. Due to the trees’ reduced transpiration, 61% of the annual total litterfall occurred during the dry season. Whereas average leaf litter nitrogen (N) concentration was 17% higher in the rainy season than dry season, potassium (K) concentration was 38% higher during the dry season than rainy season. This likely reflected the contribution of N rich green leaves to litterfall in the rainy season and plant coping strategy to drought leading to K accumulation. Cocoa leaf decomposition was not affected by management. Annual potassium (K) and calcium (Ca) stocks in cocoa pod husk were four and nine-fold, respectively, higher than in cocoa beans. We conclude that organic versus conventional management had no effect on litterfall and cocoa leaf decomposition rather season influenced litterfall quantity and chemistry. Irrespective of management the spreading of cocoa pod husk after harvest will improve internal nutrient cycling in cocoa agroforestry systems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cocoa (Theobroma cacao L.) is an important perennial crop, whose mode of cultivation is currently at large scale converted from agroforestry to monoculture systems given higher yields under the latter (Armengot et al. 2016; Niether et al. 2020). Because of its dependence on fertile forest soil, cocoa monoculture causes deforestation and CO2 emissions (Gockowski and Sonwa 2011) while cocoa agroforests are known to conserve biodiversity (Asase and Tetteh 2010) and sequester carbon (Nijmeijer et al. 2018). In these cocoa agroforestry systems, cycling of litterfall and decomposition acts as an input-output system for organic matter sustenance (Fontes et al. 2014). Due to reduction in biodiversity when cocoa agroforests are converted into monocultures, this matter cycling is reduced (Schneidewind et al. 2019). This is problematic especially in West Africa where nutrient reserves on heavily leached soils are low and nutrient exports in the harvest are rarely replaced by application of mineral fertilizers.
Litterfall in West African cocoa agroforests is estimated to amount to 10 t ha−1 yr−1 (Dawoe et al. 2010; Saj et al. 2021) whereby its distribution throughout the year depends on climatic conditions. It is generally higher in the dry than in the wet season (Saj et al. 2021), however, thunderstorms and high wind speeds in the rainy season may cause occasional peaks (Becker et al. 2015). Litterfall chemistry, such as nutrient concentrations, in cocoa agroforestry systems is influenced greatly by climate and shade tree diversity (Saj et al. 2021; Sari et al. 2022). In a cocoa agroforestry system in Mexico, magnesium (Mg) concentrations were higher in litterfall during the rainy season, whereas phosphorus (P) and calcium (Ca) concentrations were higher in dry season litter (Pérez-Flores et al. 2018).
The decomposition of litter biomass releases carbon and nutrients, whose turnover and plant availability is greatly influenced by climate, soil organisms, and litter chemistry (Aerts 1997). In most agroecosystems, initial N, P, lignin, cellulose, and polyphenols are important regulators of litter decomposition (Dawoe et al. 2010; Fontes et al. 2014). Decomposition begins with physical breakdown processes by soil macro-organisms such as ants, termites, and earthworms (Li et al. 2015). Thereafter the fragmented litter is colonized by soil microbes, which alter the litter chemistry through enzymatic activities further breaking down the litter into simple forms (Hobara et al. 2014). However, usage of chemical herbicides and pesticides in agriculture can have adverse effects on soil organisms, which alters the decomposition process (Afolabi and Muoghalu 2018).
Hartemink (2005) estimated that on average cocoa agroforests across the world loose 24–4–30 kg ha−1 of N–P–K annually through bean harvest. A low external input solution for resource poor cocoa agroforesters to reduce these losses is the spreading of cocoa pod husks after separation from beans. With every ton of dry cocoa beans produced approximately 349 kg of dry cocoa pod husk are generated (Kilama et al. 2019). However, poor management of the pods favors the spread of Phytophthora palmivora and Phytophthora megakarya, which cause cocoa black pod disease (Adejumo 2005).
Under organic management, cocoa agroforestry systems in Ghana are managed without synthetic agrochemicals and fertilizers and are more diversified in terms of shade trees (Asigbaase et al. 2019) and arthropods (Akesse-Ransford et al. 2021) than those under conventional management. Such differences imply that litterfall and decomposition processes in cocoa agroforests under organic management will differ from conventionally managed ones. However, studies comparing litterfall and decomposition of cocoa agroforests under conventional versus organic managements are scarce. The few existing studies from South America (Fontes et al. 2014; Schneidewind et al. 2019) indicated no difference between organic and conventional management concerning nutrient cycling. For West Africa, however, such comparisons are unavailable which may be at least partly due to the only recent development of organic cocoa certification. In Ghana, for example, the first certified organic cocoa production system was established in Suhum Municipality in 2005 (Glin et al. 2015). Globally organic cocoa is produced on 363,000 ha, which constitute 3.1% of the total area under cocoa (Willer et al. 2021). In 2011, West Africa produced 70% of the world’s conventional cocoa, but only 3.3% were certified organic (Potts et al. 2014).
Against this background the overall aim of this study was to determine the extent to which management and season determine nutrient cycling in cocoa agroforestry systems in West Africa. Such information will improve our knowledge on how to effectively manage soil fertility in cocoa production. Specifically, our study seeks to (a) determine the role of seasonality on litterfall and its nutrient concentrations in cocoa agroforests under organic and conventional management, (b) estimate annual litterfall and nutrient recycling under the two management systems, (c) compare the rate of cocoa leaf decomposition and nutrient release over 12 months, and (d) assess nutrient stocks in cocoa beans and pod husks.
Materials and methods
Study site
Our study was conducted in Suhum Municipality (6°2 × 3.84″ N, 0°27 × 8.64″ W) in the Eastern Region of Ghana. The natural vegetation in the Municipality is classified as a semi-deciduous forest, however, human encroachment has reduced it to secondary forests, thickets, and grasslands (MOFA 2017). The area has a bi-modal rainfall distribution with the major rainy season lasting from March to July and the minor rainy season from September to November (MOFA 2017). For 10 years (2010-2020) Suhum Municipality had an average annual precipitation ranging from 199 to 288 mm and an average temperature from 25 to 30 °C. (World Weather Online 2021). Suhum Municipality was chosen as the study location because both conventional and organic cocoa agroforestry systems exist in close vicinity. Local soils are predominantly classified as Lixisols (IUSS Working Group WRB 2015) with pH ranging from 5.83 to 6.85, major nutrients ranging from 0.55 to 2.29 µg N g−1 soil, 2–6 µg P g−1 soil, 21–84 µg K g−1 soil and a texture of 54% sand, 22% silt, and 24% clay (Agbotui unpublished data). Within an aerial distance of 6 km four villages were selected, two for each cocoa management system. In the villages Kuano and Oboadeka located on Haplic and Ferric Lixisols, respectively, cocoa farms were managed conventionally. The villages Nsuta and Adimediem, also located on Haplic and Ferric Lixisols, farms were under organic management. Within each village we randomly selected three farms at an age range of 8–25 years for litterfall collection and two for cocoa leaf decomposition studies, which represents the most productive period of cocoa trees (Obiri et al. 2007). In this selection, care was taken that in each village farms with similar ages were represented.
Description of management systems
Both organic and conventional management grow a mixture of hybrid (improved cocoa cultivar) and Amazonia cocoa cultivars planted in a haphazard manner. The cocoa tree densities were 1299 stems ha−1 and 1395 stems ha−1 for organic and conventional management, respectively. Shade tree composition and structure varied for both management systems (Table 1). In all farms, weeds were controlled 2–3 times per year. I organic farms they were slashed, whereas conventional farmers used both slashing and glyphosate-based herbicides (Agbotui et al. 2023). The six selected organic farms did not apply any mineral fertilizer during our study period. However, farmers are recommended to use 800 kg ha−1 Elite organic fertilizer™ which supplies 24–32–32 kg of N–P–K ha−1. Capsid (Distantiella theobroma (Dist.) and mirids (Sahlbergella singularis) in organic management were controlled using Agropy 5 EW™ with pyrethrum (a natural insecticide) as an active ingredient applied at the rate of 500 ml ha−1 once annually. Conventional farmers fertilized their farms using 50 kg ha−1 of Nitrabor™ (8 kg N ha−1) and Asaase Wura™ (33 kg P ha−1; 12 kg K ha−1), although the recommended rate is twice the farmers' application. Capsid and mirid in conventional farms were controlled once yearly using Akate Master™ (Bifenthrin as active ingredient), Confidor™ (Imidacloprid as active ingredient) and Actara™ (Thiamethoxam as active ingredient) at rates of 500 ml ha−1, 150 ml ha−1, and 85 ml ha−1, respectively.
Litter collection
Litterfall was determined by collection in five randomly distributed 50 cm × 50 cm × 30 cm open wooden boxes per farm raised 30 cm above the ground (Dawoe et al. 2010). Nylon netting with mesh size of 2 mm was used to close the boxes below and allow aeration and draining of rainwater. Litterbox positions were randomly relocated every two weeks to cover on-farm heterogeneity, and results were summed per month. Litter trapped within the boxes was collected fortnightly and irrespective of the litter origin (shade or cocoa trees) were sorted into leaves, reproductive parts, and twigs.
Due to the small quantities obtained reproductive parts and twigs were again pooled annually for nutrient determination.
Litterbag experiment
Litter decomposition was studied by selecting two fields aged 8–12 years and 20–25 years in each village. To this end freshly fallen cocoa leaves on the ground were collected in each farm, thoroughly mixed, and air-dried. Of these, 50 g dry matter (DM) were filled into bags of 30 cm × 30 cm and 2 mm mesh size. In each farm, 36 litterbags were placed on the soil surface and pinned with nails to assure close contact with the soil. Litterbags were then covered with litter placed on the soil surface to simulate the conditions of natural litter decomposition in cocoa agroforests. A set of six bags were randomly placed at six different locations per farm. The bags were sampled after 1, 2, 4, 6, 8, and 12 months of decomposition. At each sampling time, one bag from each location was randomly picked amounting to 6 bags (replicates) per farm. Upon bags retrieval, litter was cleaned from soil and other debris, dried, and stored until analysis. The initial characteristics of cocoa leaves used for decomposition study can be found in Table 2.
Annual cocoa pod and husk estimation
For biomass estimation of dry cocoa pod husks (CPH), twenty fruits from each farm were harvested and cocoa beans were separated from CPH. Beans and CPH were dried at 60 °C to constant weight and DM was determined for calculation of bean/CPH ratio. Cocoa bean yield in kg ha−1 was estimated by taking the 3-year average of the whole farm documented in the farmers’ Cocoa Passbook, which is the official document in Ghana, where the quantity of dried cocoa beans is recorded at the time of sale by the purchasing clerk (Asare et al. 2019).
Chemical analysis
Prior to chemical analysis, all collected samples were oven-dried at 60 °C for 72 h, weighed, and milled. The C and N concentration of milled samples were determined using a Vario Max CN analyzer (Elementar Analysensysteme GmbH, Langenselbold, Germany). For major and trace nutrient analyses, milled samples were dry-ashed at 550ºC overnight, followed by generating an ash solution with 32% HCl (Motsara and Roy 2008). Major and trace nutrient concentrations in ash solution were measured using a Spectrogreen ICP-OES (SPECTRO Analytical Instruments GmbH, Kleve, Germany). Acid detergent fiber (ADF) and acid detergent lignin (ADL) were analysed as described by Van Soest (1963) to determine lignin and cellulose, which was estimated as the difference between ADF and ADL. Total polyphenols were quantified by using the Folin-Ciocalteau method with aqueous acetone (70% v/v) as extractant (FAO and IAEA 2000). After addition of polyvinyl polypyrrolidone (PVPP) to bind the total tannins in the extract, the supernatant was used to determine the non-tannin phenols with the above-mentioned method. Total tannins were calculated as the difference between polyphenols and non-tannin phenols. Condensed tannins were measured using the butanol-HCl method (butanol-HCl 95:5 v/v; FAO and IAEA, 2000). Data were reported as means with their respective standard errors (Table 2).
Data analysis
Linear mixed effect models were used to analyse all data. Interaction between management and monthly total litterfall was tested by using management and month as fixed effects and Lixisol soil type as random effect with aboveground biomass (AGB) as covariate. Aboveground biomass was calculated with allometric equations reported in (Agbotui et al. 2023). In accordance to the Köppen Climate Classification for Tropical Regions (Belda et al. 2014), months were divided into dry and the rainy seasons, whereby months with a precipitation < 60 mm (December, January, February, and August) were assorted to the dry season. To test management and season interaction on leaf litter quantity and nutrient concentration, management and season were considered as fixed effects and soil type as a random effect with AGB as covariate. The effect of management on the quantity and nutrient content of annual total litterfall, cocoa pod husks, and beans were analysed with management as fixed effect, Lixisol soil type as a random effect and AGB as a covariate.
For the decomposition study, the ash concentration was used to correct for mineral soil contamination of bagged cocoa leaves. To this end, the following equations modified after Potthoff and Loftfield (1998) were used:
The two equations were combined, resulting in Eq. 3, which was used to estimate the contamination of litterbag content with soil.
whereby \({{DM}}_{{s}}{ }\left({g}\right)\) = Soil DM (this is the actual contamination) \({{DM}}_{{LB}}{ }\left({g}\right)\)= Litterbag DM \({{C}}_{{LB}}{ }\left(\frac{{mg}}{{g}}\right)\) = Concentration in litterbags (ash) \({{C}}_{{s}}\left(\frac{{mg}}{{g}}\right)\) = Concentration in soil (ash) \({{C}}_{{L}}{ }\left(\frac{{mg}}{{g}}\right)\) = Concentration in litter/leaves (ash)
The estimated soil contamination was used to correct the litterbag sample biomass and the nutrient concentrations of litterbag samples.
Constants of decomposition and nutrient release rates were estimated using the three parameter exponential model according of Olson (1963): \({{Y}}_{{t}}{=A+}{{Be}}^{{-}{kt}}\) where \({{Y}}_{{t}}\) is the mass or nutrient remaining at time t in months, A is the recalcitrant pool fraction, B is the labile fraction and k is the decay or nutrient release rate. The three-parameter exponential model was fitted using Sigmaplot 14.0 (Systat Software, Inc., San Jose, CA, USA). The half-life, representing the time until 50% of the cocoa leaf is decomposed, was calculated as \(\frac{{In (2)}}{{K}}\) (Olson 1963). Management effects on mass loss and nutrient release at each sampling time as well as on rates of mass loss and nutrient release were analysed with management as fixed effect and Lixisol type as random effect. Data normality and homoscedasticity were checked using Shapiro-Wilk test and visual inspection of model’s residuals versus fitted values, respectively. Data violating these assumptions were transformed using either log, square root, or Tukey ladder of powers. All statistics were performed with R (R Core Team 2020).
Results
Litterfall quantity and quality
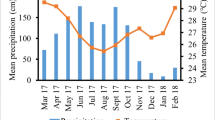
Seasonality influenced monthly total litterfall quantity under both managements. On average, the highest monthly total litterfall was recorded in December and it was three-fold higher than July which recorded lowest. (Fig. 1). The total litterfall peak at the onset of the dry season (December) in organic farms was 16% higher than in conventional farms, but this difference was not significant. In June and July, total litterfall in organic farms were 94% and 65%, respectively, higher (p < 0.05) than in conventional farms. Leaf litter contributed 82% to total litterfall and the difference in total litterfall between seasons resulted from 60% higher leaves shedding in the dry season than in the rainy season (Fig. 2b). Annual total litterfall amounted to 8 t ha−1 yr−1 and 9 t ha−1 yr−1 for conventional and organic management, respectively, whereby the difference was non-significant (Fig. 2a).
Monthly total litterfall (mean ± one standard error of the mean) for cocoa agroforests under organic and conventional management in Suhum Municipality, Eastern Region of Ghana. 2019–2020 monthly precipitation (World Weather Online 2021) indicated in grey bars. * and ** indicate significant differences between management systems (n = 6) at p = 0.05 and 0.01, respectively
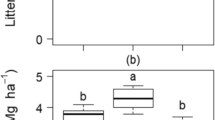
A Annual litterfall averaged within the two management systems (n = 6) and B seasonal litterfall (dry season; n = 4, rainy season; n = 8) averaged across the two management systems separated into leaves, reproductive parts (Repro), and twigs, and total litterfall in Suhum Municipality, Eastern Region of Ghana. Based on monthly precipitation, December to February and August were categorized as dry season months. Error bars indicate +/- one standard error of the mean. Lower case letters in B indicate significant differences of means between seasons
Management and season affected some of the leaf litter nutrient concentrations, whereby no interaction of the two factors was observed. The concentrations of P and Ca were 30% and 4%, respectively, greater in leaf litter of organically managed farms than in leaf litter of conventional farms (p < 0.05; Table 3). Contrarily, Mg concentration of leaf litter from conventional management was 10% higher than from organic management (p < 0.01). During the rainy season, C and N concentrations of leaf litter were 2% and 15%, respectively, higher than during the dry season. In contrast, K concentration of leaf litter in the dry season was 38% greater than in the rainy season.
No statistical differences of annual nutrient stocks in litterfall fractions were observed between organic and conventional management, except for K stocks in leaf litter, N, Ca, and Mg stocks in reproductive parts litter, and Ca stocks in total litterfall (Table 4). The annual quantity of K in leaf litter of farms under conventional management was 21% lower than of farms under organic management (p = 0.03). Likewise, annual N, Ca, and Mg quantity in reproductive parts litter of farms under conventional management were 36%, 43%, and 38%, respectively, lower (p < 0.05) than for their organic counterparts. Annual Ca quantity in total litterfall of organically managed farms was 13% higher (p < 0.05) than those in conventionally managed ones. Leaf litter contributed 71% of N, 65% of P, 61% of K, 82% of Ca, and 85% of Mg to annual nutrient stocks of total litterfall. Annual C and nutrient stocks in cocoa beans and cocoa pod husk were not significantly affected by management type. While N and P stocks in cocoa beans were 2 and 3-fold greater than in cocoa pod husks (Table 4), the annual K and Ca stocks in cocoa pod husk were four and nine-fold, respectively, greater than those in cocoa beans. On average, 19 kg N ha−1, 3 kg P ha−1, 30 kg K ha−1, 4 kg Ca ha−1 and 4 kg Mg ha−1 exported via annual harvest (cocoa pod husk and beans) represented 5%, 7%, 29%, 2%, and 4%, respectively, of the annual nutrient stocks recycled in total litterfall (Table 4).
Decomposition of cocoa leaf
Within the first 2 months, 50% of the initial litter DM disappeared under organic and conventional management (Fig. 3a). After 1 month of cocoa leaves placement, P and Ca release under conventional management were 20% and 21%, respectively, higher (p < 0.05) than under organic management (Fig. 3c and e). Similarly, Mg release at months one and two after cocoa leaves placement were 24% and 10%, respectively, lower (p < 0.05) under organic management than under conventional management (Fig. 3f). At the end of the 12 months study period, on average 15% of the initial litter remained in the litterbags. Potassium was rapidly released from litter leading to the disappearance of 98% of the initial content. Although DM disappearance and N release rates were 35% greater under organic management compared with conventional management, these differences were not statistically significant (Table 5). Phosphorus, K, and Ca release rates were 13%, 33%, and 41%, respectively, higher in conventional farms than organic farms, but these differences were not significant. For P, K, Ca, and Mg 93–100% of nutrients belong to the labile fraction of the cocoa leaves. Among the nutrient release rates, it was only Mg that under conventional management was significantly (51%) greater than under organic management.
Decay and nutrient release patterns of cocoa leaves during 12 months in cocoa agroforests under conventional (Con) and organic (Org) management in Suhum Municipality, Eastern Region of Ghana. Error bars indicate +/− one standard error of the mean. * and ** above the error bars indicate significant differences at p = 0.05 and 0.01, respectively, between management systems (n = 4)
Discussion
Seasonality affects litterfall quantity and quality
Our data did not indicate any effect of management type (organic versus conventional) on annual total litterfall, confirming findings for cocoa agroforests under organic and conventional management in Bolivia (Schneidewind et al. 2019). Average annual total litterfall of our study (8.8 t ha−1 yr−1) falls within the upper range of 5–10 t ha−1 yr−1 reported for other cocoa agroforests in Ghana (Dawoe et al. 2010), Brazil (Fontes et al. 2014), Cameroun (Saj et al. 2021), and Indonesia (Sari et al. 2022). Contribution of leaf litter to annual total litterfall (82%) in our study was within the range of 60–92% reported for cocoa agroforests (Dawoe et al. 2010; Fontes et al. 2014; Sari et al. 2022). High litterfall caused by dry seasons is well documented in cocoa agroforests (Saj et al. 2021) and was also observed in our study. This phenomenon is explained by trees’ transpiration adaptation with reduced foliage during the dry season, which is triggered by stimulation of abscisic acid in plant foliage (Yang et al. 2003). Another reason might be due to rapid leaf ageing during this period, which could be caused by photoinhibition, leaf overheating, and stomatal closure (Wright and Cornejo 1990). Average annual C stocks in total litterfall in our study (3.8 t ha−1 yr−1) falls within the upper limit of 1.5–4.0 t ha−1 yr−1 reported for homegardens in Tanzania (Becker et al. 2015) and cocoa agroforest in Cote d´Ivoire (Yao et al. 2021). This subsequently leads to high accumulation of organic matter on the soil surface, which increases C sequestration in the soil of cocoa agroforests (Gama-Rodrigues et al. 2010).
Higher N concentrations of leaf litter in the rainy season compared with those of the dry season confirms findings of Teklay (2004). During the rainy season thunderstorms mechanically force young N rich leaves to fall, resulting in litterfall with high N content (Paudel et al. 2015) compared with more senescent leaves shed during dry season (Teklay 2004). Additionally, higher N availability of soils during the rainy season could have increased N uptake by trees (Teklay 2004). Potassium concentrations in litterfall were lower during the rainy season compared with the dry season. On one hand, K is an important osmotic agent, accumulated by plants in reaction to drought. On the other hand, growth rates of leaves are reduced during dry periods, generally leading to an increase in mineral leaf components due to concentration effects. Moreover, because K is not bound to organic compounds, it might have (partly) leached from leaves in litter traps during rainfall events (Wood et al. 2005), leading to an underestimation of K concentration in leaf litter. Additionally, the dry season is characterised by high amounts of dust deposition on the plant leaves, which has been reported to have high K concentrations (Stoorvogel et al. 1997; Dawoe et al. 2018). Shade tree composition influences litterfall quantity and quality (Triadiati et al. 2011). But despite the diversity in the species composition of shade trees between the two managements there was no difference between annual nutrient stocks. There were no N-fixing shade tree species in the investigated farms, which is is typical for the study area (Asase and Tetteh 2010). Despite the absence of shade trees, the average annual total N stocks of 125 kg ha−1 year−1 was within the range of 84–175 kg ha−1 year−1 for cocoa agroforests with Erythrina glauca Lour. and Gliricidia sepium. And this can be explained by shade tree species recovering leached N from deeper soil depths. Although pruned litter from shade and cocoa trees influences the nutrient cycling process in cocoa agroforests, it could not be quantified with litterboxes.
Management did not significantly alter cocoa leaf decomposition
Initial rapid decomposition, as observed under both managements, was likely caused by leaching and break down of soluble compounds like sugar, starch, and amino acids (Wieder and Lang 1982). The disappearance of 50% of the initial material in the first 2 months is common in tropical agroforestry systems (Yao et al. 2021). Particularly cocoa agroforests with high litter diversity on the soil surface host a diverse community of soil and litter macro- and microfauna (da Silva Moço et al. 2009) accelerating litter fragmentation. Later stages of decomposition were more gradual because of accumulation of recalcitrant compounds like cellulose, lignin, and tannins (Wieder and Lang 1982). Our average decomposition rate of 0.40 g month−1 falls within the range of 0.30–0.58 g month−1 reported for cocoa leaves in Nigeria (Afolabi and Muoghalu 2018) and Cote d´Ivoire (Yao et al. 2021). However, Asigbaase et al. (2020) reported an average decomposition rate of 1.69 g month−1 in cocoa agroforests of the same municipality as ours, but it was 300% higher than ours. This disparity could be due to the above authors mixing cocoa leaves with shade tree leaves at a 1:1 rate. In our study decomposition and nutrient release rates were determined for cocoa leaves alone without including leaves from surrounding shade trees. A mixture of leaves from different species alters litter chemistry, which could be synergistic or antagonistic to decomposition processes and nutrient release (Cuchietti et al. 2014). Because of the high shade tree variability even between farms of the same management, we only considered cocoa leaves for the decomposition study. Hence, our decomposition results were not a complete reflection of litter decomposition in cocoa agroforests. In addition, the different methods that exist to study litter decomposition can lead to discrepancies when comparing studies. For example, in litterbag experiments mesh sizes > 2 mm have been reported to increase litter decomposition due to the increased activity of macro fauna which enhances litter fragmentation exposing them to higher microbial activity. On the other hand, increasing mesh size leads to high loss of fragmented litter and removal of litter from litterbag by macro fauna (Bradford et al. 2002; Smith and Bradford 2003). The tethered-leaf technique, where leaves are tied together with a nylon string and placed on the ground has the advantage of direct contact of the material with soil micro and macro fauna without altering the microclimate (Vitousek et al. 1994). However, this method is only suitable for initial decomposition studies, as it becomes difficult to distinguish between decomposition and comminution, when fragmentation of the material occurs. These drawbacks made us use a mesh size of 2 mm which is considered to allow for most soil faunal species activities in the bag (Li et al. 2015). Although not significant, our results of higher litter decomposition rate under organic management confirm the observation of higher decomposition of untreated cocoa leaves compared with those treated with pesticides (Afolabi and Muoghalu 2018; Asigbaase et al. 2020). The higher decomposition rate of cocoa leaves under organic management compared to conventional management may thus be due to the restriction of herbicides and pesticides in certified organic agriculture. Agrochemicals are well known to have adverse effects on the growth and population density of macro decomposers such as ants and earthworms (García-Pérez et al. 2016; Masoni et al. 2017) as well as on bacteria, and fungal populations (Afolabi and Muoghalu 2018). An indirect effect of agrochemicals on litter decomposition is their ability to alter the chemical composition of the litter or microclimate of the decomposition subsystem. The slower weight loss under conventional management coupled with faster P, Ca, and Mg release was similar to findings of Hendrix and Parmelee (1985) on John grass leaves (Sorghum halepense L.) treated with paraquat and glyphosate. Similarly, da Costa et al. (2019) reported on average 34% higher Mg and Ca release rates from Crotalaria spectabilis sprayed with glyphosate. According to Mertens et al. (2018) these pesticides contain chelating agents with the ability to bind P, Ca, and Mg from litter. Thus it is likely that high P, Ca, and Mg release rates under conventional management were triggered by the use of pesticides such as glyphosate and roundup.
Recycling cocoa pod husk is important for C and nutrient retention
The recorded cocoa yields of 559 kg ha−1 yr−1 were higher than the 477 kg ha−1yr−1 reported for the mid zone of Ghana (Abdulai et al. 2020), which has similar climatic conditions as Suhum. The average annual nutrient stocks of 19 kg N ha−1, 3 kg P ha−1, 30 kg K ha−1, 4 kg Ca ha−1 and 4 kg Mg ha−1 in the cocoa fruit (beans and pods) falls within the range of 19–29 kg N ha−1, 4–5 kg P ha−1, 15–51 kg K ha−1 (Hartemink 2005), 3–6 kg Ca ha−1, and 6–10 kg Mg ha−1 (Fontes et al. 2014) for other cocoa agroforests across the world. Farmers could reduce C export by 43% and nutrient export from harvest by 33% for N and 25% for P, if they spread the cocoa pod husks in their farms after harvest. This is even more important for nutrients such as K and Ca, where 81% and 90%, respectively, can be retained after harvest if cocoa pod husk were returned. However, the spreading cocoa pod husk is often avoided to prevent the spread of diseases from disease infested pods (Opoku-Ameyaw et al. 2010). Composting of cocoa pod husk could be an option to control disease pathogens (Opoku-Ameyaw et al. 2010) and return nutrients to the cocoa trees. Cocoa farmers in the study area may therefore benefit from training on composting to be able to use cocoa pod husk for improvement of their internal nutrient cycling.
Conclusions
Our findings show that litterfall follows a seasonal pattern with peaks at the onset of the dry season. Seasonality affected litterfall K and N concentrations, but organic versus conventional management had no effect on annual litterfall. We found that management had no significant effect on decomposition and nutrient release rates of N, P, K, and Ca. Only the Mg release rate under conventional management was 51% greater than under organic management, which may be attributed to the chelating capacity of herbicides used. We also conclude that spreading of cocoa pods husks in farms after harvest, particularly if composted or charred, can improve the internal carbon and nutrient cycle of cocoa agroforestry systems.
References
Abdulai I, Hoffmann MP, Jassogne L et al (2020) Variations in yield gaps of smallholder cocoa systems and the main determining factors along a climate gradient in Ghana. Agric Syst 181:102812. https://doi.org/10.1016/j.agsy.2020.102812
Adejumo TO (2005) Crop protection strategies for major diseases of cocoa, coffee and cashew in Nigeria. Afr J Biotechnol 4:143–150
Aerts R (1997) Climate, leaf litter chemistry and decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439–449. https://doi.org/10.2307/3546886
Afolabi O, Muoghalu J (2018) Effect of pesticides on microorganisms involved in litter decomposition in cacao plantation in Ile-Ife, Nigeria. Agrofor Syst 92:511–524. https://doi.org/10.1007/s10457-016-0032-y
Agbotui DK, Ingold M, Wiehle M, Buerkert A (2023) Can carbon payments improve profitability of traditional conventional and organic cocoa agroforests? A case study in the Eastern Region of Ghana. Agrofor Syst. https://doi.org/10.1007/s10457-023-00828-0
Akesse-Ransford G, Owusu EO, Kyerematen R, Adu-Acheampong S (2021) Arthropod diversity of cocoa farms under two management systems in the Eastern and Central regions of Ghana. Agrofor Syst 95:791–803. https://doi.org/10.1007/s10457-020-00568-5
Armengot L, Barbieri P, Andres C et al (2016) Cacao agroforestry systems have higher return on labor compared to full-sun monocultures. Agron Sustain Dev 36:70. https://doi.org/10.1007/s13593-016-0406-6
Asare R, Markussen B, Asare RA et al (2019) On-farm cocoa yields increase with canopy cover of shade trees in two agro-ecological zones in Ghana. Clim Dev 11:435–445. https://doi.org/10.1080/17565529.2018.1442805
Asase A, Tetteh DA (2010) The role of complex agroforestry systems in the conservation of forest tree diversity and structure in southeastern Ghana. Agrofor Syst 79:355–368. https://doi.org/10.1007/s10457-010-9311-1
Asigbaase M, Sjogersten S, Lomax BH, Dawoe E (2019) Tree diversity and its ecological importance value in organic and conventional cocoa agroforests in Ghana. PLoS ONE 14:e0210557. https://doi.org/10.1371/journal.pone.0210557
Asigbaase M, Dawoe E, Sjogersten S, Lomax BH (2020) Decomposition and nutrient mineralisation of leaf litter in smallholder cocoa agroforests: a comparison of organic and conventional farms in Ghana. J Soils Sediments. https://doi.org/10.1007/s11368-020-02844-4
Becker J, Pabst H, Mnyonga J, Kuzyakov Y (2015) Annual Litterfall dynamics and nutrient deposition depending on elevation and land use at Mt. Kilimanjaro Biogeosci 12:5635–5646. https://doi.org/10.5194/bg-12-5635-2015
Belda M, Holtanová E, Halenka T, Kalvová J (2014) Climate classification revisited: from Köppen to Trewartha. Clim Res 59:1–13. https://doi.org/10.3354/cr01204
Bradford MA, Tordoff GM, Eggers T et al (2002) Microbiota, fauna, and mesh size interactions in litter decomposition. Oikos 99:317–323. https://doi.org/10.1034/j.1600-0706.2002.990212.x
Cuchietti A, Marcotti E, Gurvich DE et al (2014) Leaf litter mixtures and neighbour effects: low-nitrogen and high-lignin species increase decomposition rate of high-nitrogen and low-lignin neighbours. Appl Soil Ecol 82:44–51. https://doi.org/10.1016/j.apsoil.2014.05.004
da Costa JVT, Junior MDAL, Saraiva ACG, et al (2019) Decomposition and nutrient release from Crotalaria spectabilis with glyphosate application. Cienc del Suelo 37:238–245
da Silva Moço MK, da Gama-Rodrigues EF, da Gama-Rodrigues AC et al (2009) Soil and litter fauna of cacao agroforestry systems in Bahia, Brazil. Agrofor Syst 76:127–138. https://doi.org/10.1007/s10457-008-9178-6
Dawoe EK, Isaac ME, Quashie-Sam J (2010) Litterfall and litter nutrient dynamics under cocoa ecosystems in lowland humid Ghana. Plant Soil 330:55–64. https://doi.org/10.1007/s11104-009-0173-0
Dawoe EK, Barnes VR, Oppong SK (2018) Spatio-temporal dynamics of gross rainfall partitioning and nutrient fluxes in shaded-cocoa (Theobroma cocoa) systems in a tropical semi-deciduous forest. Agrofor Syst 92:397–413. https://doi.org/10.1007/s10457-017-0108-3
FAO, IAEA (2000) Quantification of tannins in tree foliage: A laboratory manual for the FAO/IAEA co-ordinated research project on ’Use of nuclear and related techniques to develop simple tannin assays for predicting and improving the safety and efficiency of feeding rumin. Vienna
Fontes AG, Gama-Rodrigues AC, Gama-Rodrigues EF et al (2014) Nutrient stocks in litterfall and litter in cocoa agroforests in Brazil. Plant Soil 383:313–335. https://doi.org/10.1007/s11104-014-2175-9
Gama-Rodrigues EF, Nair PKR, Nair VD et al (2010) Carbon storage in soil size fractions under two cacao agroforestry systems in Bahia, Brazil. Environ Manag 45:274–283. https://doi.org/10.1007/s00267-009-9420-7
García-Pérez JA, Alarcón E, Hernández Y, Hernández C (2016) Impact of litter contaminated with glyphosate-based herbicide on the performance of Pontoscolex corethrurus, soil phosphatase activities and soil pH. Appl Soil Ecol 104:31–41. https://doi.org/10.1016/j.apsoil.2016.03.007
Glin LC, Oosterveer PJM, Mol APJ (2015) Governing the organic cocoa network from Ghana: towards hybrid governance arrangements? J Agrar Chang 15:43–64. https://doi.org/10.1111/joac.12059
Gockowski J, Sonwa D (2011) Cocoa intensification scenarios and their predicted impact on CO2 emissions, biodiversity conservation, and rural livelihoods in the Guinea rain forest of West Africa. Environ Manag 48:307–321. https://doi.org/10.1007/s00267-010-9602-3
Hartemink AE (2005) Nutrient stocks, nutrient cycling, and soil changes in cocoa ecosystems: a review. Adv Agron 86:227–253. https://doi.org/10.1016/S0065-2113(05)86005-5
Hendrix PF, Parmelee RW (1985) Decomposition, nutrient loss and microarthropod densities in herbicide-treated grass litter in a Georgia piedmont agroecosystem. Soil Biol Biochem 17:421–428. https://doi.org/10.1016/0038-0717(85)90003-3
Hobara S, Osono T, Hirose D et al (2014) The roles of microorganisms in litter decomposition and soil formation. Biogeochemistry 118:471–486. https://doi.org/10.1007/s10533-013-9912-7
IUSS Working Group WRB (2015) World Reference Base for Soil Resources. World Soil Resources Reports 106
Kilama G, Lating PO, Byaruhanga J, Biira S (2019) Quantification and characterization of cocoa pod husks for electricity generation in Uganda. Energy Sustain Soc 9:1–11
Li X, Yin X, Wang Z, Fan W (2015) Litter mass loss and nutrient release influenced by soil fauna of Betula Ermanii forest floor of the Changbai Mountains, China. Appl Soil Ecol 95:15–22. https://doi.org/10.1016/j.apsoil.2015.05.008
Masoni A, Frizzi F, Brühl C et al (2017) Management matters: a comparison of ant assemblages in organic and conventional vineyards. Agric Ecosyst Environ 246:175–183. https://doi.org/10.1016/j.agee.2017.05.036
Mertens M, Höss S, Neumann G et al (2018) Glyphosate, a chelating agent-relevant for ecological risk assessment? Environ Sci Pollut Res 25:5298–5317. https://doi.org/10.1007/s11356-017-1080-1
MOFA (2017) Suhum Municipal Assembly–Ministry of Food and Agriculture. http://mofa.gov.gh/site/?page_id=1526. Accessed 24 Mar 2019
Motsara MR, Roy RN (2008) Guide to laboratory establishment for plant nutrient analysis. Rome
Niether W, Jacobi J, Blaser WJ et al (2020) Cocoa agroforestry systems versus monocultures: a multi-dimensional meta-analysis. Environ Res Lett 15:104085. https://doi.org/10.1088/1748-9326/abb053
Nijmeijer A, Lauri PÉ, Harmand JM, Saj S (2018) Carbon dynamics in cocoa agroforestry systems in Central Cameroon: afforestation of savannah as a sequestration opportunity. Agrofor Syst. https://doi.org/10.1007/s10457-018-0204-z
Obiri BD, Bright GA, McDonald MA et al (2007) Financial analysis of shaded cocoa in Ghana. Agrofor Syst 71:139–149. https://doi.org/10.1007/s10457-007-9058-5
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331. https://doi.org/10.2307/1932179
Opoku-Ameyaw K, Baah F, Gyedu-Akoto E et al (2010) Cocoa manual: a source book for sustainable cocoa production. Cocoa Research Institute of Ghana, Tafo, Ghana
Paudel E, Dossa GGO, Xu J, Harrison RD (2015) Litterfall and nutrient return along a disturbance gradient in a tropical montane forest. Ecol Manag 353:97–106. https://doi.org/10.1016/j.foreco.2015.05.028
Pérez-Flores J, Pérez AA, Suárez YP et al (2018) Leaf litter and its nutrient contribution in the cacao agroforestry system. Agrofor Syst 92:365–374. https://doi.org/10.1007/s10457-017-0096-3
Potthoff M, Loftfield N (1998) How to quantify contamination of organic litter bag material with soil? Pedobiologia (Jena) 42:147–153
Potts J, Lynch M, Wilkins A et al (2014) The state of sustainability initiatives review 2014: standards and the green economy. In: International Institute for Sustainable Development and International Institute for Environment and Develoment, Winnipeg, Canada and London, UK
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing Vienna, Austria. https://www.R-project.org/
Saj S, Nijmeijer A, Nieboukaho JE, Harmand PLJ-M (2021) Litterfall seasonal dynamics and leaf-litter turnover in cocoa agroforests established on past forest lands or savannah. Agrofor Syst 95:583–597. https://doi.org/10.1007/s10457-021-00602-0
Sari RR, Rozendaal DMA, Saputra DD et al (2022) Balancing litterfall and decomposition in cacao agroforestry systems. Plant Soil 473:251–271. https://doi.org/10.1007/s11104-021-05279-z
Schneidewind U, Niether W, Armengot L et al (2019) Carbon stocks, litterfall and pruning residues in monoculture and agroforestry cacao production systems. Exp Agric 55:452–470. https://doi.org/10.1017/S001447971800011X
Smith VC, Bradford MA (2003) Litter quality impacts on grassland litter decomposition are differently dependent on soil fauna across time. Appl Soil Ecol 24:197–203. https://doi.org/10.1016/S0929-1393(03)00094-5
Stoorvogel JJ, Van Breemen N, Janssen BH (1997) The nutrient input by harmattan dust to a forest ecosystem in Cote d’Ivoire. Afr Biogeochem 37:145–157. https://doi.org/10.1023/A:1005734225727
Teklay T (2004) Seasonal dynamics in the concentrations of macronutrients and organic constituents in green and senesced leaves of three agroforestry species in southern Ethiopia. Plant Soil 267:297–307. https://doi.org/10.1007/s11104-005-0124-3
Triadiati S, Tjitrosemito E, Guhardja E et al (2011) Litter production and leaf litter decomposition at natural forest and cocoa agroforestry in Central Sulawesi, Indonesia. Asian J Biol Sci 4:221–234
Van Soest PJ (1963) Use of detergents in the analysis of fibrous feeds. II. A rapid method for the determination of fiber and lignin. J Assoc off Agric Chem 46:829–835
Vitousek PM, Turner DR, Parton WJ, Sanford RL (1994) Litter decomposition on the Mauna Loa environmental matrix, Hawai’i: patterns, mechanisms, and models. Ecology 75:418–429
Wieder RK, Lang GE (1982) A critique of the analytical methods used in examining decomposition data obtained from litter bags. Ecology 63:1636–1642. https://doi.org/10.2307/1940104
Willer H, Travnicek J, Meier C, Schlatter B (2021) The World of organic agriculture statistics and emerging trends 2021. Research Institute of Organic Agriculture FiBL and IFOAM - Organics International, Frick, Switzerland
Wood TE, Lawrence D, Clark DA (2005) Variation in leaf litter nutrients of a Costa Rican rain forest is related to precipitation. Biogeochemistry 73:417–437. https://doi.org/10.1007/s10533-004-0563-6
World Weather Online (2021) Suhum monthly climate averages. https://www.worldweatheronline.com/suhum-weather-averages/ashanti/gh.aspx. Accessed 4 Mar 2021
Wright JS, Cornejo FH (1990) Seasonal drought and leaf fall in a tropical forest. Ecol Soc Am 71:1165–1175. https://doi.org/10.2307/1937384
Yang Y, Guo G, Chen G et al (2003) Effect of slash burning on nutrient removing and soil fertility in Chinese fir and evergreen broadleaved forests of mid-subtropical China. Pedosphere 13:87–96
Yao MK, Koné AW, Otinga AN et al (2021) Carbon and nutrient cycling in tree plantations vs. natural forests: implication for an efficient cocoa agroforestry system in West Africa. Reg Enviromental Chang 21:44. https://doi.org/10.1007/s10113-021-01776-0
Acknowledgements
We are thankful to the German Academic Exchange Service (DAAD) for supporting the first author with a scholarship. We are also grateful to Eva Wiegard and Claudia Thieme-Fricke at the laboratory of Organic Plant Production and Agroecosystems Research in the Tropics and Subtropics, University of Kassel for technical assistance and thank Yayra Glover Ltd. for logistical support during data collection. Finally, the co-operation and support of cocoa farmers in Suhum is deeply appreciated.
Funding
Open Access funding enabled and organized by Projekt DEAL. The research leading to these results received funding from German Academic Exchange Service (DAAD) under Grant Agreement No. 57299294.
Author information
Authors and Affiliations
Contributions
DKA, MI and AB conceived and planned the experiments. DKA carried out the experiment. MI contributed to the interpretation of the results. DKA wrote the manuscript with support from MI and AB, who both supervised the project. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Agbotui, D.K., Ingold, M. & Buerkert, A. Carbon and nutrient cycling in cocoa agroforests under organic and conventional management. Nutr Cycl Agroecosyst 129, 7–20 (2024). https://doi.org/10.1007/s10705-024-10349-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10705-024-10349-6