Abstract
Risk stratification for sudden cardiac death in dilated cardiomyopathy is a field of constant debate, and the currently proposed criteria have been widely questioned due to their low positive and negative predictive value. In this study, we conducted a systematic review of the literature utilizing the PubMed and Cochrane library platforms, in order to gain insight about dilated cardiomyopathy and its arrhythmic risk stratification utilizing noninvasive risk markers derived mainly from 24 h electrocardiographic monitoring. The obtained articles were reviewed in order to register the various electrocardiographic noninvasive risk factors used, their prevalence, and their prognostic significance in dilated cardiomyopathy. Premature ventricular complexes, nonsustained ventricular tachycardia, late potentials on Signal averaged electrocardiography, T wave alternans, heart rate variability and deceleration capacity of the heart rate, all have both some positive and negative predictive value to identify patients in higher likelihood for ventricular arrhythmias and sudden cardiac death. Corrected QT, QT dispersion, and turbulence slope–turbulence onset of heart rate have yet to establish a predictive correlation in the literature. Although ambulatory electrocardiographic monitoring is frequently used in clinical practice in DCM patients, no single risk marker can be used for the selection of patients at high-risk for malignant ventricular arrhythmic events and sudden cardiac death who could benefit from the implantation of a defibrillator. More studies are needed in order to establish a risk score or a combination of risk factors with the purpose of selecting high-risk patients for ICD implantation in the context of primary prevention.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the World Health Organization, cardiovascular disease remains epidemiologically the most frequent cause of death worldwide [1]. It is estimated that 17 million people die each year from cardiovascular disease, and 25–50% of them die suddenly, often without having had any previous history of cardiovascular disease. Incidence of sudden cardiac death (SCD) in Europe is estimated at 1/1000 people per year, increasing with age [2,3,4]. Men exhibit approximately twice the risk for ACS when compared to women, but this difference decreases as age progresses [5]. With the ongoing evolution in treatment and diagnosis of the aforementioned disease, survival of patients seems to increase and SCD rates decline. According to a sub-study of the Rotterdam study, incidence of SCD in people over 40 years of age decreased from 4.7 per 1000 person-years in the 1990s to 2.1 in the 2000s [6].
Definitions
Sudden cardiac death
According to the most recent definition of the World Health Organization, SCD is the unexpected death that occurs within 1 h of symptom onset (in witnessed cases) or within 24 h since the last time the person was seen alive and asymptomatic (in unwitnessed cases) [7]. SCD occurs when a trigger acts on an anatomic or functional electrophysiological substrate, with the final pathway being elicitation of ventricular fibrillation (VF) or ventricular tachycardia (VT) that degenerates into VF, causing hemodynamic collapse and cessation of mechanical activity of the heart. Asystole and electromechanical dissociation are also causes of SCD [8]. Polymorphic VT and torsade de pointes occur more frequently in patients with QT interval prolongation, either genetically inherited or drug induced. It is worth emphasizing that SCD is also caused by arrhythmic causes such as massive pulmonary embolism, aortic dissection and rupture, narcotic substances, etc. (Fig. 1). According to an older study of patients who suffered SCD while wearing a Holter monitor, VT/VF/multiform VT were the initial rhythms of cardiac arrest, with VT being the most common [9]. Electromechanical dissociation is also a common arrest rhythm, occurring mostly in noncardiac causes of death although it also commonly occurs in patients with advanced heart failure [10].
Dilated cardiomyopathy/nonischemic cardiomyopathy
The term dilated cardiomyopathy (DCM) denotes the structural heart disease characterized by dilatation and systolic dysfunction of the left and often the right ventricle, which cannot be attributed to coronary artery disease, valvular heart disease, or arterial hypertension [11]. It is the third overall cause of heart failure in the general population and the first cause of heart transplantation worldwide [12]. DCM is an “umbrella” term as it includes numerous inherited or acquired myocardial diseases that lead to the same phenotypic outcome (e.g., myocarditis, substance and drug toxicity, autoimmune disease, gene mutations) (Fig. 2). Prevalence is estimated at 1:2500 people and is probably underestimated, as the clinical status of patients varies from asymptomatic to end-stage heart failure [13]. An often overlooked subtype of DCM is hypokinetic nondilated cardiomyopathy, characterized by left ventricular systolic dysfunction without accompanying dilatation [11]. It is underdiagnosed due to initially mild symptoms and less-pronounced imaging findings.
Risk stratification for sudden cardiac death
Despite the reduction in mortality that has been achieved with early diagnosis, medical therapy, and cardiovascular implantable devices, a significant percentage of patients will suffer the most dreaded complication of the disease, SCD. SCD occurs more often in young and mildly symptomatic patients. It is responsible for 30% of total DCM mortality, and 1/3 of patients are classified as functional class I according to the New York Heart Association (NYHA) classification, i.e., they are younger with mildly affected functional capacity [14]. Ventricular arrhythmias are the main cause of SCD followed by bradyarrhythmias, as myocardial fibrosis often causes conduction disturbances [15]. Apropos of VT, the main mechanism appears to be scar related re-entry followed by bundle branch re-entry [16].
Currently, primary prevention of SCD, as suggested by the European Society of Cardiology and American Heart Association guidelines is based on left ventricular ejection fraction (EF) and NYHA functional class [17, 18]. Patients qualify for an implantable cardioverter defibrillator (ICD) if their EF is below 35% and are in NYHA II-III class. The recommendations mainly considered studies such as DEFINITE, SCD-HEFT, CAT, AMIOVIRT, and COMPANION, whose patients were recruited more than 20 years ago, without receiving currently indicated pharmacotherapy. Also, several of them included a large percentage of patients with ischemic heart failure [19, 20]. Greater contribution to the level of evidence for these recommendations was given by meta-analyses of those studies, such as the one by Theuns et al. who reported a reduction of SCD along with a reduction in total mortality (RR:0.73) [21]. It is therefore reasonable to conclude that the aforementioned indicators are not characterized by sufficient sensitivity and specificity to adequately identify patients at high risk for SCD [22, 23]. Many patients with EF < 35% in whom, according to guidelines, implantation of an ICD is recommended for primary prevention of SCD, never do experience major arrhythmic events while undergoing the risks of device implantation, such as inappropriate shocks and infection. Results of the most recent DANISH study point in this direction, where ICD implantation did not reduce overall mortality in patients with DCM and low EF. The vast majority of patients were receiving optimal pharmacological and device treatment, including cardiac resynchronization devices. Patients younger than 65 years showed benefit, likely due to their fewer comorbidities, leading to the conclusion that device implantation should be reserved for those meeting multifactorial criteria beyond EF and NYHA class [24]. Conversely, an increased arrhythmic risk truly exists in some patients with EF > 35%. This was proven by SCD registries in the Oregon and Maastricht regions, in which 80% of victims had an EF that would have excluded them from ICD implantation [25, 26]. Based on the above, it is clear that the so far commonly accepted predictors of major arrhythmic events are not sufficient to unveil patients at high risk for SCD.
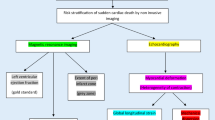
In the context of offering a more solid arrhythmic stratification for DCM patients, the ReCONSIDER study was conceived [27]. The ReCONSIDER study is an observational study, where imaging parameters, by cardiac magnetic resonance imaging (cMRI—such as late gadolinium enhancement) and echocardiography, along with electrocardiographic parameters (derived through 24 h Holter monitoring and signal averaged electrocardiography) are used to identify patients with EF > 35% at low or moderate risk for SCD. Those at moderate risk will undergo programmed ventricular stimulation and according to the results, they will be offered an ICD.
Materials and methods
Aim of the study
The purpose of this manuscript is to describe noninvasive electrocardiographic risk factors that have been studied in DCM patients, as well as their prevalence and prognostic significance in unveiling subsets at higher risk for ventricular arrhythmias and SCD.
Inclusion criteria
(1) Studies enrolling patients aged over 18 years old, where one or more noninvasive electrocardiographic risk factors (premature ventricular complexes, nonsustained ventricular tachycardia, late potentials on signal averaged electrocardiography, T wave alternans, heart rate variability and deceleration capacity of the heart rate, corrected QT, QT dispersion and turbulence slope–turbulence onset of heart rate) were studied concerning the occurrence of VT/VF or sudden death; (2) DCM/SCD/VT were defined in accordance with the current and commonly accepted criteria [7, 11].
Literature search strategy
This systematic review and meta-analysis is reported according to the Preferred Reporting Items for Systematic reviews and Meta-Analysis (PRISMA) guidelines. We searched MEDLINE (via PubMed) and Cochrane Central Register of Controlled Trials with search terms: dilated cardiomyopathy; sudden cardiac death; ventricular arrhythmias; noninvasive risk factors (Fig. 3).
Results
Forty-four manuscripts were finally included. Several observational studies have attempted to identify noninvasive and invasive predictors of major arrhythmic events such as late gadolinium enhancement on cardiac MRI, genetic testing, electrocardiographic testing, and programmed ventricular stimulation. In the context of this literature review, only noninvasive risk factors resulting from conventional or specialized electrocardiographic techniques are included. Each one of the arrhythmic risk markers that are going to be mentioned has been tied to at least one of the three conventional mechanisms of arrhythmogenesis: re-entry, triggered activity, and automaticity (Fig. 4).
Microvolt TWA
T wave alternans (TWA) describes the beat-to-beat variability in time, shape, and/or amplitude of the T wave on the electrocardiogram (ECG). It is a rate-dependent phenomenon that is the result of disturbances in the management of intracellular calcium, predisposing to ventricular arrhythmias [28]. It is derived through either the spectral or the modified moving average technique. The first technique is used in treadmill stress testing while the second one allows TWA estimation in 24-h electrocardiographic recordings (Holter monitoring). It cannot be accurately measured in the presence of persistent atrial fibrillation, frequent spontaneous ventricular contractions (PVCs) and in patients with chronotropic incompetence who cannot achieve a heart rate above 110 beats per minute. Seven prospective studies with > 100 patients with DCM each examined TWA with follow-up for sudden death. In two of these, the patients were not on beta-blockers (Table 1). The common result was a high negative predictive value of TWA for sudden death, exceeding 88% in all studies [29,30,31,32,33,34,35]. The largest study (ALPHA) included 446 patients in whom TWA were analyzed and correlated with the endpoints of total mortality, major arrhythmic events, and sudden death. Patients with abnormal TWA were usually older, with a lower EF and advanced NYHA class. The negative predictive value was 98% for the three endpoints in a follow-up of 18 months [33].
Based on these publications, one can assume that this risk factor may safely point towards patients with low risk for life threatening arrhythmias who may benefit little from implantation of an ICD, given its consistently high negative predictive value. The TWA study was recommended by the ISHNE guidelines in 2011 as a useful predictor of cardiovascular mortality and sudden death in patients with DCM beyond EF [36] but was excluded from their most recent guidelines in 2017 [37].
Ventricular ectopy and nonsustained ventricular tachycardia
Premature ventricular contractions (PVCs) and nonsustained ventricular tachycardia (NSVT) are thought to arise from regions of the myocardium with increased automaticity (Table 2). It has been estimated that in 40–60% of patients with DCM NSVT is recorded while 90% show polymorphic PVCs in 24-h Holter monitoring [38]. The prognostic value of PVCs has been recognized mainly in post-infarction patients, with the presence of > 10 PVCs/h being an independent indicator of mortality [39]. At present, a study examined the presence of > 1000 PVCs/24 h in 285 patients with newly diagnosed DCM but failed to demonstrate an association with SCD/VT/VF [40]. In a subgroup analysis of the recent DANISH trial that included DCM patients with a low EF < 35% and elevated NT-ProBNP levels, the presence of > 30 PVCs per hour on Holter monitoring significantly correlated with total mortality and cardiovascular death, but not with SCD[41]. Due to the high prevalence of PVCs in this population, one cannot safely draw conclusions about their value as risk stratifiers or asses their prognostic significance. However, there are several publications about NSVT detected in 24-h electrocardiographic recordings, with often conflicting results. In the same subgroup of patients in the DANISH trial, NSVT presence was significantly associated with all-cause mortality and cardiovascular death but not with SCD. Also, in interaction analysis, no benefit would be gained from ICD implantation in these patients [41]. The most recent Leiden Nonischemic Cardiomyopathy Study demonstrated that the presence of NSVT, regardless of the presence of late gadolinium enhancement in cardiac MRI, can predict the occurrence of sustained ventricular tachyarrhythmias in patients with DCM and palpitations, out-of-hospital arrest, or a history of syncope [42]. The Marburg cardiomyopathy study highlighted 3 risk groups in patients with DCM according to the number of NSVT QRS complexes. Patients without NSVT had 2% VT/VF/SCD at 52 ± 21 months of observation, in patients with 5–9 beat NSVT runs the end point was calculated as 5%, while in those with NSVT runs consisting of more than 10 complexes, 10%. NSVT rate was not associated with the primary endpoint [43]. The multivariate analysis of a study of 319 patients with DCM by Zecchin et. al demonstrated an increased risk for SCD/VT/VF in the presence of NSVT only for patients with an EF > 35% [44]. The presence of NSVT predicted with statistical significance the total mortality and SCD even after adjustment for EF, NYHA class and age in a prospective study of 157 patients [45]. Finally, in a meta-analysis of 18 studies that examined the association of NSVT with SCD, Goldberger et al. calculated the total odds ratio to be 2.92, concluding that detection of NSVT on Holter monitoring is associated with the endpoint of SCD but is not sufficient as a single risk stratification criterion [22]. Based on the above, the presence of PVCs or NSVT is frequent in patients with DCM and this may be the reason for their only modest performance as risk stratifiers in this subset of patients.
Late potentials on single averaged electrocardiography (Table 3)
Single averaged electrocardiography (SAECG) is a method for identifying areas of slow conduction in the ventricular myocardium, which is a necessary substrate for re-entrant ventricular arrhythmias to occur (Table 3). After collection of the ECG signals, they are processed and noise is removed revealing signals in the μV range, which are not visible in the surface ECG [46]. For this test to be considered positive, at least 2 of 3 standard criteria with abnormal values must be detected. The criteria are as follows: (1) the width of the overall filtered QRS (fQRS) to be greater than 114 ms; (2) the detected electrical activity of low potentials < 40 μV within the last portion of the QRS is maintained for a period of time of at least 38 ms; (3) this electrical activity actually corresponds to a current of low potentials < 20 μV [47]. In patients with intraventricular conduction delay and QRS > 120 ms, modified criteria are applied [48]. Late potentials have been applied mainly in postmyocardial infarction patients, and their use has been declining over time [49]. Regarding DCM, positive late potentials are acquired in 25% of patients with increasing percentage in those with a history of sustained VT (70–90%) [50]. In an older study of 79 DCM patients, positive late potentials were an independent indicator for SCD (3.7-fold higher risk) and cardiac mortality (2.1-fold higher risk) [51]. These results are comparable to those reported by Mancini et al. in their study of 114 DCM patients. Two groups were created based on the presence or absence of late potentials. Sixty-six patients with negative late potentials did not suffer ventricular tachyarrhythmias or SCD in a follow-up of about 1 year. On the other hand, in 20 patients with abnormal SAECG, 4 presented with sustained VT and 5 suffered SCD. Therefore, the annual survival rate without occurrence of VT or SCD was estimated to be 95% in the absence of late potentials and only 39% in their presence [52]. A positive correlation of abnormal late potentials with the occurrence of SCD and/or VT has been detected in most relevant studies unfortunately without as high positive predictive value [53,54,55,56,57]. Furthermore, in a relevant meta-analysis of 10 such studies, the calculated odds ratio was 2.11 for the presence of abnormal SAECG and the primary outcome of SCD or major ventricular arrhythmic events [22]. In summary, late potentials show a correlation with SCD and VT in patients with DCM, but their use as stand-alone risk stratification tools is not recommended due to their low positive and negative predictive value [58, 59].
QT interval and repolarization dispersion
Ventricular repolarization is considered to be a critical period of the cardiac cycle for the pathogenesis of malignant arrhythmias (Table 4). The QT interval is the measured ECG criterion regarding the duration of ventricular repolarization. Formulas have emerged that adjust the duration of this interval according to the heart rate (QTc), with the most commonly used being the Fridericia and Bazzet formulas. Prolongation of repolarization has been thoroughly investigated and tied to major arrhythmic events in ischemic cardiomyopathy, heart failure and primary “electrical” heart diseases, such as long QT syndromes [60]. The underlying mechanism of arrhythmogenesis is commonly considered to be triggered activity. In the Marburg cardiomyopathy study, QTc prolongation on surface ECG failed to reveal a tie between SCD or ventricular tachy-arrhythmias in DCM patients [30]. Of note, QTc measurement on 24-h Holter monitoring is considered to be a better reflector of this complex repolarization process when different dynamic components such as the autonomic nervous system interaction are taken into account [61]. Unfortunately, such studies could not be identified in the literature to date.
QT dispersion (QTd) is another repolarization marker that was correlated with major arrhythmic events in various clinical settings [62]. Regarding DCM, results are conflicting. Fei et al. found no significant difference in QTd in a sample of 60 patients some of which died or received heart transplants [63]. In the aforementioned Marburg study, QTc dispersion was of no prognostic value in 343 DCM patients in regard to major arrhythmic events and SCD [30]. Same results were also reported regarding the long term risk for such endpoints by Fauchier [64]. On the other hand, in the study of Galinier et al., a QTd > 80 ms was an independent predictor of sudden death, VT, VF with correlation to other arrhythmic risk markers such as SAECG late potentials [65]. Once again, repolarization prolongation and dispersion although commonly prolonged in DCM patients, failed to predict patients in risk of worse arrhythmic outcome. It remains to be established if such repolarization indices will earn their way in assessing arrhythmic risk in DCM patients.
Heart rate variability
Cardiovascular diseases cause disturbance in the balance of the autonomic nervous system that can predispose to ventricular arrhythmias and increased overall mortality. Whether this is due to sympathetic nervous system overload or loss of some of the activity of the parasympathetic nervous system remains unknown. Heart rate variability (HRV) quantifies the range of this variability and possibly reflects the activity of the autonomic nervous system. The classical methods of HRV analysis are (a) analysis of cardiac time series over time and (b) spectral analysis of the contained frequencies of the signal [66]. HRV is estimated in a 24-h Holter recording usually via the standard deviation of all normal RR intervals over a period of time (SDNN). HRV is implicated in numerous conditions such as arterial hypertension, diabetes mellitus, and coronary artery disease. Studies exist mainly on mixed populations with a diagnosis of heart failure, but with equivocal results (Table 5). In the literature, a meta-analysis of 4 studies, for a total of 630 patients with DCM, reported on the endpoint of SCD. Sensitivity and specificity of the index did not exceed 60% and Odds ratio was calculated as 1.72 with p = 0.13 [22]. Overall, the above mentioned meta-analysis did not highlight any of the autonomic nervous system assessment indices (as discussed below) as promising noninvasive risk factors for predicting death in DCM patients. In the DEFINITE trial, DCM patients with preserved HRV had an excellent prognosis on follow up and may gain little benefit from an ICD implantation. Patients were subdivided in 3 groups according to their SDNN values as SDNN < 81, 81 < SDNN < 113, SDNN > 113 with a mortality of 10%, 7%, and 0%, respectively, on a 3-year follow-up [20]. Similar results were published by Fauchier et al. where a reduced SDNN was an independent predictor of SCD as well as VT,VF (p = 0.01) [63]. Conversely, HRV failed to predict which patients could benefit from ICD implantation in the Marburg study [30]. Although it is now widely known that DCM patients have decreased HRV, this pathology is not formally associated with worse arrhythmic outcomes in this population and cannot serve as a risk marker for such.
Heart rate turbulence, deceleration capacity, turbulence onset, and turbulence slope
Early PVCs in a healthy population are followed by a normal biphasic sinus node depolarization response. More specifically, each PVC is followed by a short period of acceleration followed by a period of deceleration of the heart rate. In a normal functioning autonomic nervous system, heart rate turbulence (HRT) is present and can be quantified. In cardiovascular disease, however, as previously mentioned, this turbulence is reduced or even lost. HRT is considered as an index that quantifies reflex parasympathetic activity [67]. As a risk indicator, it can be measured in a 24-h Holter recording with the assumption of the existence of a sufficient number of PVCs. HRT is quantified by turbulence onset (TO) and turbulence slope (TS) which represent the initial acceleration and subsequent deceleration of the sinus after a PVC. The phenomenon was first discovered in post myocardial infarction patients and the combination of pathological TO and TS was the strongest predictor of total mortality in the study by Schmidt et al. [68]. Regarding DCM patients, the analysis of the Marburg and Frankfurt DCM registries did not reveal a predictive value of HRT regarding major arrhythmic events [30, 69]. A meta-analysis of 3 studies by Goldberger et al. yielded similar results in 434 patients [22]. The study included the two aforementioned studies as well as a Japanese registry of patients with DCM and ischemic heart disease, in which patients with pathological HRT showed a clearly increased incidence of SCD and VT [70]. HRT was considered positive when both TO was ≥ 0% and TS was ≤ 2.5 ms/R-R interval.
Finally, deceleration capacity (DC) is thought to assess the effect of the parasympathetic nervous system on heart rate independent of sympathetic activity. Deceleration of heart rate must be the final result of the tonic sympathetic–parasympathetic interactions on the sinus node level with the reflex vagal activity to be added each moment on this tonic status [71, 72]. This is also a method of assessing the autonomic nervous system and is calculated from time series of R-R intervals in 24-h electrocardiographic monitoring. It was introduced by Bauer et al. in a study of patients after myocardial infarction [73]. Only a few studies have examined the association of this novel marker with cardiac mortality in patients with DCM. In 201 patients with a 40-month follow-up, DC below 4.5 ms powerfully and independently predicted mortality [74]. Similar results were also published by Yang, where in 65 male patients, a DC < 4.72 ms was a significant predictor of cardiac mortality on a 60-month follow-up [75]. The conclusions drawn from these studies (Table 6) are as follows: (a) patients with DCM have a reduced DC compared to their healthy counterparts and (b) a reduction in DC is strongly associated with an increase in cardiac and total mortality as well.
Discussion
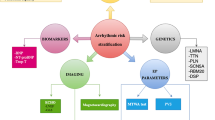
As previously mentioned, arrhythmia occurrence risk and sudden death risk stratification remain suboptimal in DCM. This is now widely known and proven by many investigators. On our everyday clinical practice, we encounter patients with an EF < 35% where the implantable defibrillator has never been activated, as well as patients with relatively preserved EF > 35% who present with sustained VT, VF, or suffer sudden death. Many previous and ongoing studies try to unveil a better risk stratification tool but unfortunately no single factor proved to have better sensitivity/specificity than EF. In this manner, clinicians worldwide usually rely on 24-h electrocardiographic monitoring in order to achieve a better understanding of the patients arrhythmia burden and risk. Electrocardiographic Holter monitoring can also be utilized in order to observe the therapeutic effects of antiarrhythmic agents such as beta blockers and amiodarone in heart failure. The clinical practice dissents current ISHNE guidelines on Holter monitoring, where it is stated that prognostic value is rather low and remains controversial in DCM [37]. Additionally, there is no mention of electrocardiographic monitoring in the recently published guidelines on ventricular arrhythmias and SCD of the European Society of Cardiology. Patients with an EF < 35% receive an implantable defibrillator based on a class IIa recommendation. Patients with an EF > 35% are risk layered based on 4 factors: unexplained syncope, pathogenic mutations on LMNA, PLN, FLNC, and RBM20 genes, presence of late gadolinium enhancement on cardiac MRI and inducible sustained monomorphic VT on programmed ventricular stimulation. Patients with 2 or more of these criteria qualify for primary prevention with implantation of an ICD based on a class IIa recommendation [17].
Conclusion
Unlike ischemic cardiomyopathy, where a well-defined scar serves as a substrate for re-entrant ventricular arrhythmias and current risk stratification parameters for sudden cardiac death are adequately defined, this is not the case for DCM. Despite the scientific community’s efforts, no single risk marker stands out besides left ventricular EF which has moderate negative and positive predictive values. It remains yet unclear whether a pooling of electrocardiographic parameters or a risk score containing noninvasive risk markers can be successfully used to this end in this population.
Availability of data and materials
Not applicable.
References
Goldberger JJ, Buxton AE, Cain M, Costantini O, Exner DV, Knight BP et al (2011) Risk stratification for arrhythmic sudden cardiac death: identifying the roadblocks. Circulation 123(21):2423–2430. https://doi.org/10.1161/CIRCULATIONAHA.110.959734
Tan HL, Dagres N, Böttiger BW, Schwartz PJ (2018) ESCAPE-NET Investigators, european sudden cardiac arrest network: towards prevention, education and new effective treatments (ESCAPE-NET): a major european horizon 2020 project focused on cardiac arrest. Eur Heart J 39(2):86–88. https://doi.org/10.1093/eurheartj/ehx758
Mendis S, Puska P, Norrving BE (2011) Global atlas on cardiovascular disease prevention and control, Geneva: World Health Organisation
Myerburg RJ, Junttila MJ (2012) Sudden cardiac death caused by coronary heart disease. Circulation 125(8):1043–1052. https://doi.org/10.1161/CIRCULATIONAHA.111.023846
Straus SM, Bleumink GS, Dieleman JP, van der Lei J, Stricker BH, Sturkenboom MC (2004) The incidence of sudden cardiac death in the general population. J Clin Epidemiol 57(1):98–102. https://doi.org/10.1016/S0895-4356(03)00210-5
Niemeijer MN, van den Berg ME, Leening MJ, Hofman A, Franco OH, Deckers JW, Heeringa J, Rijnbeek PR et al (2015) Declining incidence of sudden cardiac death from 1990–2010 in a general middle-aged and elderly population: the Rotterdam Study. Heart Rhythm 12(1):123–129. https://doi.org/10.1016/j.hrthm.2014.09.054
Chugh SS, Jui J, Gunson K, Stecker EC, John BT, Thompson B et al (2004) Current burden of sudden cardiac death: multiple source surveillance versus retrospective death certificate-based review in a large U.S. community. J Am Coll Cardiol 44:1268–1275. https://doi.org/10.1016/j.jacc.2004.06.029
Cobb LA, Fahrenbruch CE, Olsufka M, Copass MK (2002) Changing incidence of out-of-hospital ventricular fibrillation, 1980–2000. JAMA 288:3008–3013. https://doi.org/10.1001/jama.288.23.3008
Bayés de Luna A, Coumel P, Leclercq JF (1989) Ambulatory sudden cardiac death: mechanisms of production of fatal arrhythmia on the basis of data from 157 cases. Am Heart J 117(1):151–9. https://doi.org/10.1016/0002-8703(89)90670-4
Luu M, Stevenson WG, Stevenson LW, Baron K, Walden J (1989) Diverse mechanisms of unexpected cardiac arrest in advanced heart failure. Circulation 80(6):1675–1680. https://doi.org/10.1161/01.cir.80.6.1675
Pinto YM, Elliott PM, Arbustini E, Adler Y, Anastasakis A, Böhm M et al (2016) Proposal for a revised definition of dilated cardiomyopathy, hypokinetic non-dilated cardiomyopathy, and its implications for clinical practice: a position statement of the ESC working group on myocardial and pericardial diseases. Eur Heart J 37(23):1850–1858. https://doi.org/10.1093/eurheartj/ehv727
Maron BJ, Towbin JA, Thiene G et al (2006) Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation 113(14):1807–1816. https://doi.org/10.1161/CIRCULATIONAHA.106.174287
Bozkurt B, Colvin M, Cook J, Cooper LT, Deswal A, Fonarow GC et al (2016) American heart association committee on heart failure and transplantation of the council on clinical cardiology; council on cardiovascular disease in the young; council on cardiovascular and stroke nursing; council on epidemiology and prevention; and council on quality of care and outcomes research. current diagnostic and treatment strategies for specific dilated cardiomyopathies: a scientific statement from the american heart association. Circulation. 134(23):e579-e646. https://doi.org/10.1161/CIR.0000000000000455
Kjekshus J (1990) Arrhythmias and mortality in congestive heart failure. Am J Cardiol 65(19):42I-48I. https://doi.org/10.1016/0002-9149(90)90125-k
McElwee SK, Velasco A, Doppalapudi H (2016) Mechanisms of sudden cardiac death. J Nucl Cardiol 23(6):1368–1379. https://doi.org/10.1007/s12350-016-0600-6
Blanck Z, Jazayeri M, Dhala A, Deshpande S, Sra J, Akhtar M (1993) Bundle branch reentry: a mechanism of ventricular tachycardia in the absence of myocardial or valvular dysfunction. J Am Coll Cardiol 22(6):1718–1722. https://doi.org/10.1016/0735-1097(93)90602-w
Zeppenfeld K, Tfelt-Hansen J, de Riva M, Winkel BG, Behr ER, Blom NA et al (2022) ESC Scientific Document Group. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J 26:ehac262. https://doi.org/10.1093/eurheartj/ehac262
Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB et al (2018) 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation 138(13):e272–e391. https://doi.org/10.1161/CIR.0000000000000549
Bardy GH, Lee KL, Mark DB, Poole JE, Packer DL, Boineau R et al (2005) Sudden cardiac death in heart failure trial (SCD-HeFT) investigators. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med 352:225–237. https://doi.org/10.1056/NEJMoa043399
Kadish A, Dyer A, Daubert JP, Quigg R, Estes NA, Anderson KP et al (2004) Defibrillators in non-ischemic cardiomyopathy treatment evaluation (DEFINITE) investigators. Prophylactic defibrillator implantation in patients with nonischemic dilated cardiomyopathy. N Engl J Med 350(21):2151–8. https://doi.org/10.1056/NEJMoa033088
Theuns DA, Smith T, Hunink MG, Bardy GH, Jordaens L (2010) Effectiveness of prophylactic implantation of cardioverter-defibrillators without cardiac resynchronization therapy in patients with ischaemic or non-ischaemic heart disease: a systematic review and meta-analysis. Europace 12(11):1564–1570. https://doi.org/10.1093/europace/euq329
Goldberger JJ, Subacius H, Patel T, Cunnane R, Kadish AH (2014) Sudden cardiac death risk stratification in patients with nonischemic dilated cardiomyopathy. J Am Coll Cardiol 63(18):1879–1889. https://doi.org/10.1016/j.jacc.2013.12.021
Disertori M, Quintarelli S, Mazzola S, Favalli V, Narula N, Arbustini E (2013) The need to modify patient selection to improve the benefits of implantable cardioverter-defibrillator for primary prevention of sudden death in non-ischaemic dilated cardiomyopathy. Europace 15(12):1693–1701. https://doi.org/10.1093/europace/eut228
Køber L, Thune JJ, Nielsen JC, Haarbo J, Videbæk L, Korup E et al (2016) Defibrillator implantation in patients with nonischemic systolic heart failure. N Engl J Med 375:1221–1230. https://doi.org/10.1056/NEJMoa1608029
Stecker EC, Vickers C, Waltz J, Socoteanu C, John BT, Mariani R et al (2006) Population-based analysis of sudden cardiac death with and without left ventricular systolic dysfunction: two-year findings from the Oregon Sudden Unexpected Death Study. J Am Coll Cardiol 47:1161–1166. https://doi.org/10.1016/j.jacc.2005.11.045
Gorgels AP, Gijsbers C, de Vreede-Swagemakers J, Lousberg A, Wellens HJ (2003) Out-of-hospital cardiac arrest–the relevance of heart failure. The maastricht circulatory arrest registry. Eur Heart J 24:1204–1209. https://doi.org/10.1016/s0195-668x(03)00191-x
Gatzoulis KA, Dilaveris P, Arsenos P, Tsiachris D, Antoniou CK, Sideris S et al (2021) Arrhythmic risk stratification in nonischemic dilated cardiomyopathy: the ReCONSIDER study design - a two-step, multifactorial, electrophysiology-inclusive approach. Hellenic J Cardiol 62(2):169–172. https://doi.org/10.1016/j.hjc.2020.03.008
Arsenos P, Gatzoulis K, Dilaveris P, Manis G, Tsiachris D, Archontakis S et al (2013) Arrhythmic sudden cardiac death: substrate, mechanisms and current risk stratification strategies for the post-myocardial infarction patient. Hellenic J Cardiol 54(4):301–15
Kitamura H, Ohnishi Y, Okajima K, Ishida A, Galeano E, Adachi K et al (2002) Onset heart rate of microvolt-level T-wave alternans provides clinical and prognostic value in nonischemic dilated cardiomyopathy. J Am Coll Cardiol 39:295–300. https://doi.org/10.1016/s0735-1097(01)01718-1
Grimm W, Christ M, Bach J, Muller HH, Maisch B (2003) Noninvasive arrhythmia risk stratification in idiopathic dilated cardiomyopathy: results of the Marburg Cardiomyopathy Study. Circulation 108:2883–2891. https://doi.org/10.1161/01.CIR.0000100721.52503.85
Hohnloser Sh, Klingenheben T, Bloomfield D, Dabbous O, Cohen RJ (2003) Usefulness of microvolt T-wave alternans for prediction of ventricular tachyarrhythmic events in patients with dilated cardiomyopathy: results from a prospective observational study. J Am Coll Cardiol 41:2220–2224. https://doi.org/10.1016/s0735-1097(03)00467-4
Bloomfield DM, Bigger JT, Steinman RC, Namerow PB, Parides MK, Curtis AB et al (2006) Microvolt T-wave alternans and risk of death or sustained ventricular arrhythmias in patients with left ventricular dysfunction. J Am Coll Cardiol 47:456–463. https://doi.org/10.1016/j.jacc.2005.11.026
Salerno-Uriarte JA, De Ferrari GM, Klersy C, Pedretti RF, Tritto M, Sallusti L et al (2007) Prognostic value of T-wave alternans in patients with heart failure due to nonischemic cardiomyopathy. Results of the ALPHA study. J Am Coll Cardiol 50:1896 –904. https://doi.org/10.1016/j.jacc.2007.09.004
Gold MR, Ip JH, Costantini O, Poole JE, McNulty S, Mark DB et al (2008) Role of microvolt T-wave alternans in assessment of arrhythmia vulnerability among patients with heart failure and systolic dysfunction: primary results from the T-wave alternans Sudden Cardiac Death in Heart Failure Trial substudy. Circulation 118:2022–2028. https://doi.org/10.1161/CIRCULATIONAHA.107.748962
Shizuta S, Ando K, Nobuyoshi M, Ikeda T, Yoshino H, Hiramatsu S et al (2012) Prognostic utility of T-wave alternans in a real-world population of patients with left ventricular dysfunction: the PREVENT-SCD study. Clin Res Cardiol 101:89–99. https://doi.org/10.1007/s00392-011-0368-2
Verrier RL, Klingenheben T, Malik M, El-Sherif N, Exner DV, Hohnloser SH et al (2011) Microvolt T-wave alternans. Physiological basis, methods of measurement, and clinical utility—Consensus guideline by international society for holter and noninvasive electrocardiology. J Am Coll Cardiol 58:1309 –24. https://doi.org/10.1016/j.jacc.2011.06.029
Steinberg JS, Varma N, Cygankiewicz I, Aziz P, Balsam P, Baranchuk A et al (2017) ISHNE-HRS expert consensus statement on ambulatory ECG and external cardiac monitoring/telemetry. Ann Noninvasive Electrocardiol 22(3):e12447. https://doi.org/10.1111/anec.12447
Liuba I, Marchlinski FE (2013) The substrate and ablation of ventricular tachycardia in patients with nonischemic cardiomyopathy. Circ J 77(8):1957–1966. https://doi.org/10.1253/circj.cj-13-0758
Maggioni AP, Zuanetti G, Franzosi MG, Rovelli F, Santoro E, Staszewsky L et al (1993) Prevalence and prognostic significance of ventricular arrhythmias after acute myocardial infarction in the fibrinolytic era. GISSI-2 results. Circulation. 87(2):312–22. https://doi.org/10.1161/01.cir.87.2.312
Spezzacatene A, Sinagra G, Merlo M, Barbati G, Graw SL, Brun F et al (2015) Familial cardiomyopathy registry. arrhythmogenic phenotype in dilated cardiomyopathy: natural history and predictors of life-threatening arrhythmias. J Am Heart Assoc 4(10):e002149. https://doi.org/10.1161/JAHA.115.002149
Boas R, Thune JJ, Pehrson S, Køber L, Nielsen JC, Videbæk L et al (2021) Prevalence and prognostic association of ventricular arrhythmia in non-ischaemic heart failure patients: results from the DANISH trial. Europace 23(4):587–595. https://doi.org/10.1093/europace/euaa341
Piers SR, Androulakis AF, Yim KS, van Rein N, Venlet J, Kapel GF et al (2022) Nonsustained ventricular tachycardia is independently associated with sustained ventricular arrhythmias in nonischemic dilated cardiomyopathy. Circ Arrhythm Electrophysiol 15(2):e009979. https://doi.org/10.1161/CIRCEP.121.009979
Grimm W, Christ M, Maisch B (2005) Long runs of non-sustained ventricular tachycardia on 24-hour ambulatory electrocardiogram predict major arrhythmic events in patients with idiopathic dilated cardiomyopathy. Pacing Clin Electrophysiol 28(Suppl 1):S207–S210. https://doi.org/10.1111/j.1540-8159.2005.00035.x
Zecchin M, DI Lenarda A, Gregori D, Merlo M, Pivetta A, Vitrella G et al (2008) Are nonsustained ventricular tachycardias predictive of major arrhythmias in patients with dilated cardiomyopathy on optimal medical treatment?. Pacing Clin Electrophysiol 31:290–299 https://doi.org/10.1111/j.1540-8159.2008.00988.x
Becker R, Haass M, Ick D et al (2003) Role of nonsustained ventricular tachycardia and programmed ventricular stimulation for risk stratification in patients with idiopathic dilated cardiomyopathy. Basic Res Cardiol 98:259–266. https://doi.org/10.1007/s00395-003-0398-7
Breithardt G, Cain ME, el-Sherif N, Flowers NC, Hombach V, Janse M et al (1991) Standards for analysis of ventricular late potentials using high-resolution or signal-averaged electrocardiography. A statement by a task force committee of the european society of cardiology, the american heart association, and the american college of cardiology. Circulation 83(4):1481–8. https://doi.org/10.1161/01.cir.83.4.1481
Goldberger JJ, Cain ME, Hohnloser SH, Kadish AH, Knight BP, Lauer MS et al (2008) American heart association; American college of cardiology foundation; Heart rhythm society. American heart association/american college of cardiology foundation/heart rhythm society scientific statement on noninvasive risk stratification techniques for identifying patients at risk for sudden cardiac death. A scientific statement from the american heart association council on clinical cardiology committee on electrocardiography and arrhythmias and council on epidemiology and prevention. J Am Coll Cardiol 52(14):1179–99. https://doi.org/10.1016/j.jacc.2008.05.003
Gatzoulis KA, Carlson MD, Biblo LA, Rizos I, Gialafos J, Toutouzas P et al (1995) Time domain analysis of the signal averaged electrocardiogram in patients with a conduction defect or a bundle branch block. Eur Heart J 16(12):1912–1919. https://doi.org/10.1093/oxfordjournals.eurheartj.a060847
Gatzoulis KA, Arsenos P, Trachanas K, Dilaveris P, Antoniou C, Tsiachris D et al (2018) Signal-averaged electrocardiography: Past, present, and future. J Arrhythm 34(3):222–229. https://doi.org/10.1002/joa3.12062
Fauchier JP, Fauchier L, Babuty D, Cosnay P (1996) Time-domain signal-averaged electrocardiogram in nonischemic ventricular tachycardia. Pacing Clin Electrophysiol 19(2):231–244. https://doi.org/10.1111/j.1540-8159.1996.tb03315.x
Goedel-Meinen L, Hofmann M, Ryba S, Schömig A (2001) Prognostic value of an abnormal signal-averaged electrocardiogram in patients with nonischemic dilated cardiomyopathy. 87(6):809–812. https://doi.org/10.1016/s0002-9149(00)01514-9
Mancini DM, Wong KL, Simson MB (1993) Prognostic value of an abnormal signal-averaged electrocardiogram in patients with nonischemic congestive cardiomyopathy. Circulation 87:1083–1092. https://doi.org/10.1161/01.cir.87.4.1083
Denereaz D, Zimmermann M, Adamec R (1992) Significance of ventricular late potentials in non-ischaemic dilated cardiomyopathy. Eur Heart J 13(7):895–901. https://doi.org/10.1093/oxfordjournals.eurheartj.a060289
Marconi P, Ciaccheri M, Castelli G, Nannini M, Arcangeli C, Dolara A (1993) Late ventricular potentials in patients with dilated cardiomyopathy. Correlations with spontaneous ventricular tachycardia and clinical course. G Ital Cardiol 23(10):979–83
Winters SL, Goldman DS, Banas Jr JS (1993) Prognostic impact of late potentials in nonischemic dilated cardiomyopathy. Potential signals for the future. Circulation 87:1405–1407. https://doi.org/10.1161/01.CIR.87.4.1405
Turitto G, Ahuja RK, Bekheit S, Caref EB, Ibrahim B, el-Sherif N (1994) Incidence and prediction of induced ventricular tachyarrhythmias in idiopathic dilated cardiomyopathy. Am J Cardiol 73(11):770–3. https://doi.org/10.1016/0002-9149(94)90879-6
Yi G, Keeling PJ, Goldman JH, Jian H, Poloniecki J, McKenna WJ (1995) Prognostic significance of spectral turbulence analysis of the signal-averaged electrocardiogram in patients with idiopathic dilated cardiomyopathy. Am J Cardiol 75(7):494–497. https://doi.org/10.1016/s0002-9149(99)80588-8
Karcz M, Chojnowska L, Zareba W, Ruzyłło W (2003) In dilated cardiomyopathy normal signal-averaged electrocardiogram does not identify low mortality risk patients. Pol Merkur Lekarski 15(90):537–539
Grimm W, Hoffmann J, Knop U, Winzenburg J, Menz V, Maisch B (1996) Value of time- and frequency-domain analysis of signal-averaged electrocardiography for arrhythmia risk prediction in idiopathic dilated cardiomyopathy. Pacing Clin Electrophysiol 19(11 Pt 2):1923–1927. https://doi.org/10.1111/j.1540-8159.1996.tb03254.x
Brooksby P, Batin PD, Nolan J, Lindsay SJ, Andrews R, Mullen M et al (1999) The relationship between QT intervals and mortality in ambulant patients with chronic heart failure. The united kingdom heart failure evaluation and assessment of risk trial (UK-HEART). Eur Heart J 20(18):1335–41. https://doi.org/10.1053/euhj.1999.1542
Arsenos P, Gatzoulis KA, Dilaveris P, Gialernios T, Sideris S, Lazaros G et al (2011) The rate-corrected QT interval calculated from 24-hour Holter recordings may serve as a significant arrhythmia risk stratifier in heart failure patients. Int J Cardiol 147(2):321–323. https://doi.org/10.1016/j.ijcard.2010.12.076
Malik M, Batchvarov VN (2000) Measurement, interpretation and clinical potential of QT dispersion. J Am Coll Cardiol 36:1749–1766. https://doi.org/10.1016/s0735-1097(00)00962-1
Fei L, Goldman JH, Prasad K et al (1996) QT dispersion and RR variations on 12-lead ECGs in patients with congestive heart failure secondary to idiopathic dilated cardiomyopathy. Eur Heart J 17:258–263. https://doi.org/10.1093/oxfordjournals.eurheartj.a014843
Fauchier L, Douglas J, Babuty D, Cosnay P, Fauchier JP (2005) QT dispersion in nonischemic dilated cardiomyopathy. A long-term evaluation. Eur J Heart Fail 7:277–282. https://doi.org/10.1016/j.ejheart.2004.07.009
Galinier M, Vialette JC, Fourcade J et al (1998) QT interval dispersion as a predictor of arrhythmic events in congestive heart failure. Importance of aetiology Eur Heart J 19:1054–1062. https://doi.org/10.1053/euhj.1997.0865
Malik M (1996) Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 93(5):1043–65. https://doi.org/10.1111/j.1542-474X.1996.tb00275.x
Bauer A, Malik M, Schmidt G, Barthel P, Bonnemeier H, Cygankiewicz I et al (2008) Heart rate turbulence: standards of measurement, physiological interpretation, and clinical use: International Society for Holter and Noninvasive Electrophysiology Consensus. J Am Coll Cardiol 52(17):1353–1365. https://doi.org/10.1016/j.jacc.2008.07.041
Schmidt G, Malik M, Barthel P, Schneider R, Ulm K, Rolnitzky L et al (1999) Heart-rate turbulence after ventricular premature beats as a predictor of mortality after acute myocardial infarction. Lancet 353(9162):1390–1396. https://doi.org/10.1016/S0140-6736(98)08428-1
Klingenheben T, Ptaszynski P, Hohnloser SH (2008) Heart rate turbulence and other autonomic risk markers for arrhythmia risk stratification in dilated cardiomyopathy. J Electrocardiol 41:306–311. https://doi.org/10.1016/j.jelectrocard.2007.10.004
Miwa Y, Ikeda T, Sakaki K, Miyakoshi M, Ishiguro H, Tsukada T et al (2009) Heart rate turbulence as a predictor of cardiac mortality and arrhythmic events in patients with dilated cardiomyopathy: a prospective study. J Cardiovasc Electrophysiol 20(7):788–795. https://doi.org/10.1111/j.1540-8167.2009.01438.x
Hohnloser SH, Klingenheben T, van de Loo A et al (1994) Reflex versus tonic vagal activity as a prognostic parameter in patients with sustained ventricular tachycardia or ventricular fibrillation. Circulation 89:1068–1073. https://doi.org/10.1161/01.CIR.89.3.1068
Arsenos P, Manis G, Gatzoulis KA, Dilaveris P, Gialernios T, Angelis A et al (2016) Deceleration capacity of heart rate predicts arrhythmic and total mortality in heart failure patients. Ann Noninvasive Electrocardiol 21(5):508–518. https://doi.org/10.1111/anec.12343
Bauer A, Kantelhardt JW, Barthel P, Schneider R, Mäkikallio T, Ulm K et al (2006) Deceleration capacity of heart rate as a predictor of mortality after myocardial infarction: cohort study. Lancet 367(9523):1674–1681. https://doi.org/10.1016/S0140-6736(06)68735-7
Demming T, Sandrock S, Kuhn C, Kotzott L, Tahmaz N, Bonnemeier H (2016) Deceleration capacity: A novel predictor for total mortality in patients with non-ischemic dilated cardiomyopathy. Int J Cardiol 15(221):289–293. https://doi.org/10.1016/j.ijcard.2016.06.205
Yang Y, Wang F, Zou C, Dong H, Huang X, Zhou B et al (2018) Male patients with dilated cardiomyopathy exhibiting a higher heart rate acceleration capacity or a lower deceleration capacity are at higher risk of cardiac death. Front Physiol 7(9):1774. https://doi.org/10.3389/fphys.2018.01774
Funding
Open access funding provided by HEAL-Link Greece.
Author information
Authors and Affiliations
Contributions
M.N., G.K., S.S.: proposed the idea, proposed the structure of the paper and formulated the paper. D.P., D.I., S.Z, N.V., X.A., L.A., S.S.: independent reviewer. Z.P., K.P., A.S., A.C., A.P.: creators of tables and figures. T.D., D.P., T.K.: critically appraised the paper, made final suggestions.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Milaras, N., Dourvas, P., Doundoulakis, I. et al. Noninvasive electrocardiographic risk factors for sudden cardiac death in dilated ca rdiomyopathy: is ambulatory electrocardiography still relevant?. Heart Fail Rev 28, 865–878 (2023). https://doi.org/10.1007/s10741-023-10300-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-023-10300-x