Abstract
Beaked whales are cryptic and difficult to study species, often distributed in deep offshore waters and only briefly visible at the surface. A diverse range of cetacean species has been documented in the Bay of Biscay, including several species of beaked whales. However, little is known about how persistent their presence is. Citizen science data collected during ferry-based surveys between 2006 and 2018 were analysed to investigate how encounter rates varied across space and time, and their drivers for beaked whale species. Approximately 244,400 km were surveyed, and there were 419 encounters with beaked whales recorded including Cuvier’s beaked whales, (n = 260), Northern bottlenose whales (n = 19), Sowerby’s beaked whales (n = 13), and True’s beaked whales (n = 1). Generalized Additive Models revealed that encounters were generally more frequent in the southern bay, on northern facing slopes, with all species except Sowerby’s showing a preference for both deep waters and shallow shelf waters. Animals were recorded in each of the eight surveyed months, suggesting that beaked whales may be present year-round, with increased encounter rates in summer months. This study is the first to indicate that beaked whales may persist in this area throughout the year, which is key information for appropriate management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Due to consistent over-use of marine resources in recent decades, conservation is paramount to safeguard the environment for present and future generations (Lester et al., 2009; Roff & Zacharias, 2011). Research is essential for effective conservation, especially concerning species abundance and distribution parameters which are key for informing appropriate management (Arcangeli et al., 2016; Laran et al., 2017).
Globally, cetaceans (whales, dolphins, and porpoises) are under threat (Hammond et al., 2013). In Europe, cetacean conservation has been a topical issue especially since the implementation of the Habitats Directive and Marine Strategy Framework Directive (Hammond et al., 2013; Laran et al., 2017). Whilst there are previous studies that provided foundations for a diversity of observations and recommendations to support cetacean conservation in Europe, key research gaps remain (Hammond et al., 2009, 2013; Laran et al., 2017; Rogan et al., 2017; Matear et al., 2019). Insufficient knowledge regarding habitat preference and cetacean distribution impedes management and limits effective conservation (Kiszka et al., 2007). This partly reflects aspects of cetacean ecology, but also logistical limitations such as offshore surveys being expensive (Barlow & Gisiner, 2006; Arcangeli et al., 2016; Hooker et al., 2019).
Widespread geographies hinder cetacean management and research efforts because conservation boundaries are normally tied to administrative limits, which cetaceans do not adhere to (Spitz et al., 2012). Beaked whales (Family Ziphiidae) are widely regarded as the least known cetacean families (Cox et al., 2006; Rogan et al., 2017). Extensive dive times and relatively short periods at the surface, combined with a preference for deep offshore waters results in a paucity of knowledge about these cryptic animals, which can greatly inhibit effective conservation (Johnson et al., 2004; Schorr et al., 2014).
Beaked whales are a diverse family with at least 23 species over six genera classified, and further species being recently described (Cox et al., 2006; Rogan et al., 2017; Hooker et al., 2019; Yamada et al., 2019; Carroll et al., 2021). The diversity of beaked whales is credited to their selection of deep-ocean environments, which is exploited by relatively few other mammalian species (Claridge, 2006). However, this habitat preference represents a considerable logistical challenge for researchers (Johnson et al., 2004; Claridge, 2006; Tyack et al., 2006; Spitz et al., 2011; Schorr et al., 2014). Consequently, species-specific in situ field studies are often lacking, with stranding and whaling data representing a significant proportion of current knowledge (Faerber & Baird, 2010; Arcangeli et al., 2016; Hooker et al., 2019). However, this leaves considerable data gaps with respect to beaked whale distribution, habitat-use, behaviour, and broader ecology.
There is significant concern regarding the conservation of beaked whales due to a multitude of issues, including bycatch, entanglement, trawling, overfishing of prey, ship-strike, chemical pollution, noise, climate change and habitat disruption, such as marine development and tourism (Spitz et al., 2011; Ketten, 2014; Arcangeli et al., 2016; Laran et al., 2017; Matear et al., 2019). These threats may result in lethal and nonlethal responses leading to stranding (Parsons et al., 2008). Mass strandings, often caused by anthropogenic noise, are widely considered the predominant threat to beaked whales (Johnson et al., 2004; Parsons et al., 2008; D’Amico et al., 2009; Tyack et al., 2011; Arcangeli et al., 2016; Rogan et al., 2017; Hooker et al., 2019). Thus, there is a need to better understand beaked whale distribution and ecology to support effective management and conservation planning.
The Bay of Biscay is a deep-water gulf with complex topography and hydrography, known to support a diversity of cetacean species including several species of beaked whales (Weir et al., 2004; Kiszka et al., 2007; Spitz et al., 2011, 2012; Matear et al., 2019). It also experiences high levels of commercial vessel traffic, with major shipping lanes running through the Bay (OSPAR, 2019). However, these vessels provide “platforms of opportunity” for researchers, allowing regular access to offshore waters. This has resulted in an increased availability of sightings data from previously difficult-to-access sites, including data on the occurrence of beaked whales (Evans & Hammond, 2004; Moura et al., 2012). Previous studies have highlighted the Bay of Biscay as a suitable habitat for beaked whales, including Cuvier’s beaked whales (Kiszka et al., 2007), Northern bottlenose whales (Hyperoodon ampullatus) and Sowerby’s beaked whales (Matear et al., 2019), and occasionally True’s beaked whales (Weir et al., 2004; Robbins et al., 2019). However, little is known about the seasonality of these species in the Bay of Biscay, or whether they may be present year-round.
The aim of this study is to examine spatio-temporal patterns of beaked whale occurrence within the Bay of Biscay. We used a 12-year citizen-science dataset traversing the Bay to model counts of beaked whale sightings in regard to spatial, temporal, and environmental variables. These results are important for not only furthering our understanding of beaked whale geographic distribution and seasonality, but also the drivers behind such patterns. This information is particularly relevant given increasing levels of shipping traffic in this area.
Materials and methods
Study site
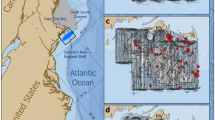
The Bay of Biscay is located in the north-eastern Atlantic Ocean, stretching in a curve from north-west France to north-west Spain. The eastern area is coastal and relatively shallow, with bathymetry increasing to 5000 m at the shelf break to the west (Certain et al., 2008). The southern bay features a narrow continental shelf, characterised by numerous narrow, steep-sided, deep-sea canyons; whereas the shelf widens in the northern reaches of the Bay and is intersected by coastal inlets and estuaries (Lavin et al., 2003). There are marked seasonality in the hydrodynamic features of the Bay, with alternation between winter mixing and summer stratification driving high phytoplankton biomass in the spring and a secondary autumnal peak, followed by lagged peaks in zooplankton (Lavin et al., 2003). Additionally, the Bay is home to a considerable diversity of fish species (~ 200 demersal and benthic fish species), with spring marking the spawning season of small pelagic fish such as sardine and anchovy (Lavin et al., 2003).
Two ferries (MV Pont-Aven & MV Cap Finistère) operated by Brittany Ferries were used as platforms of opportunity to collect data on the occurrence of beaked whales. The Pont-Aven travels between Portsmouth (UK)—Santander (Spain)—Plymouth (UK)—Roscoff (France)—Cork (Ireland)—Plymouth—Santander—Portsmouth (Fig. 1), with a full cycle taking 7 days. The Cap Finistère travels between Portsmouth—Santander—Portsmouth—Bilbao (Spain)—Portsmouth (Fig. 1), with a full cycle also taking 7 days.
Data collection followed established methods (Matear et al., 2019; Robbins et al., 2019, 2020) in the form of two protocols, which are summarised below.
Distance sampling protocol
Data on environmental conditions (e.g. sea state or precipitation), vessel location (‘effort’ data), and sightings data on cetacean occurrence were collected from the bridge of the Pont-Aven (21.6 m height) at the front of the vessel, with a team of four trained observers following distance sampling methodologies outlined in Robbins et al. (2020). Data were collected from March to October between 2006 and 2017, for as long as daylight hours allowed for one return crossing approximately once per month, between Plymouth—Santander—Portsmouth only.
Surveys were conducted by four surveyors. Two surveyors were on watch, with one on the port side and the other on the starboard side. The starboard surveyor scanned from 90° off the starboard side to 10° to port, whilst the port surveyor scanned from 90° off the port side to 10° to starboard (20° overlap to increase sighting potential). Surveyors on watch used the naked eye to scan, with binoculars to supplement and for identification of species. Scan cycles were from side to side then near to distant. The third surveyor recorded the sightings and effort data. Only sightings collected by surveyors on watch were recorded, other observations were classed as “incidental sightings”. The fourth surveyor was on a rest break. Surveyors followed a 30-min rotation period, with 30/60 min breaks recommended after long observation periods. Surveys were conducted from dawn to dusk, weather and route stage permitting, and data were only collected in good weather conditions (i.e. data were not recorded above sea state 6, or in poor visibility).
When a cetacean was sighted, the time, sighting number, watch number, GPS location, reticle distance, angle, eye distance, species code, certainty, school size, calve number, cue, behaviour, animal heading, observer code and comments/mixed group were documented. Effort readings were recorded every 30 min, along with environmental records. Data recorded included time, effort, watch number, GPS location, speed, vessel course, sea state, swell, precipitation, glare, visibility, observer codes and comments. All surveyors received ORCA training prior to surveys, and survey teams consist of experienced observers that have surveyed on other ferry routes. Data for sightings and effort were entered into pre-prepared data sheets and afterwards the survey was logged into a Microsoft Excel spreadsheet for collation with other ORCA data.
Wildlife officer protocol
Data were collected by one or two seasonal ‘Wildlife Officers’ across the full cycle of the routes of each of the Cap Finistère and Pont-Aven between 2014 and 2018. Two trained observers were on-board the Pont-Aven constantly from approximately the end of March–early July each year. There were two trained observers on the Cap Finistère between late March and the end of June, reducing to one trained observer and one less experienced ‘trainee’ until late September. In 2016 both ships entered dry dock, with the Pont-Aven for three weeks in May, and the Cap Finistère for two weeks in April.
Wildlife Officer surveys occur most days, but for shorter periods than distance sampling surveys. They are conducted from the open decks, Deck 10 on the Pont-Aven (24.1 m height), and 10 on the Cap Finistère (22.1 m height). Surveys were conducted from the starboard side on the Cap Finistère, and could be conducted from either both, or one of the port and starboard sides of the Pont-Aven, often depending on wind direction and glare, with the side with more favourable conditions chosen. Environmental conditions (sea state (Beaufort), swell height (in metres), glare, precipitation and GPS positions) were recorded at 30-min intervals at minimum, or whenever conditions changed. Details of sightings (location, species identity if known or highest known classification, number of animals, behaviour, distance, and position in relation to the vessel) were recorded for all cetacean, seal, and shark species; however only beaked whale species are included here. Data were collected on various models of Microsoft Surface tablets running Logger 2010 software, with GPS position collected by Canmore GT-730FL USB dongle every second.
Wildlife Officers also perform public engagement roles on-board, and therefore may be distracted by passengers during surveys; however, with two observers and the additional survey effort of passengers, minimal missed encounters beyond those normally anticipated at sea are expected.
Data processing
All data were used initially, except effort records where sea state was not recorded, and associated sightings during those periods. Distances of effort tracks were calculated using the geosphere package (Hijmans, 2019; see Table 1 for data processing packages) in R software (R Core Team, 2020). Encounter rates per 100 km were calculated with:
For maps of encounter rate, cells with less than 45 km effort (1st quantile) were removed as there was a single sighting on the northern slope of the Bay of Biscay with little effort which caused high encounter rates; however there was minimal data to identify this area as important.
Environmental covariates which have been found to influence beaked whale occurrence in previous studies (Cañadas et al., 2002; Tepisch et al., 2014; Arcangeli et al., 2016) were assigned to each track line using the centroid: mean depth (obtained from GEBCO at approximately 500 m resolution), slope (value calculated QGIS, with GRASS plugins), aspect (calculated with GRASS). Sightings were assigned to each effort line by vessel name, protocol type (Wildlife Officer or distance sampling), and date/time.
As beaked whales are cryptic species and can be difficult to visually identify (Hildebrand et al., 2015), records were prioritised where species identification certainty was ‘definite’ and survey conditions were favourable (below sea state 3). This allows a more accurate representation of encounter rate, given worse conditions reduce detection probability (Buckland et al., 2001; Kizska et al., 2007), and that lower certainties may introduce false-positive records. When removing any animals encountered in conditions above sea state 2, and only accepting those classed as definitely assigned the correct species identification, there were three encounters with Northern bottlenose whales (six animals), 107 encounters with Cuvier’s beaked whales (215 animals), six encounters with Sowerby’s beaked whales (13 animals), and 61 encounters with 101 animals not identified to species level which were confidently identified as beaked whales. Due to small sample sizes of all but Cuvier’s beaked whales in this subset, we retained all sightings regardless of identification certainty or conditions for analyses. However, all analyses of Cuvier’s beaked whale encounter rates use this subset to maximise accuracy based on the most reliable data available.
Modelling
Generalized Additive Models (GAMs; Wood, 2006) allow for non-normal response data, such as count of animals, to be related to the predictor variables using non-parametric smooths, and were created with the mgcv package (Wood, 2017) for R (R Core team, 2020) with the number of encounters as the response. The explanatory variables initially considered for inclusion in models were mean depth (hereafter depth), mean slope (hereafter slope), mean aspect (with penalized cubic regression splines; hereafter aspect), year, month, latitude, and longitude. Circadian patterns were not investigated as the vessels followed approximately the same schedules, and the time suitable habitat was passed is likely to be within a few hours. Depth and slope were correlated (− 0.59 Spearman correlation) and therefore slope was not included in the models. Similarly, depth was correlated (0.75) with latitude, and therefore depth was retained, and latitude was modelled as a bivariate term with longitude.
Response data possessed large numbers of zeroes, resulting in a left-skewed distribution. Data distributions were investigated, and negative binomial, Poisson, Gaussian, Tweedie, and scaled t GAM families were tried. For all species, the scaled t family which deals with heavy tailed data (Wood et al., 2016) performed best, judged on diagnostic plots (QQ-Plots, histograms, residuals versus fitted values, fitted values versus observed values; (Zuur and Ieno 2016). The double penalty approach (Marra & Wood, 2011) was adopted, which introduces an extra penalty to remove terms from the model and used Restricted Maximum Likelihood (REML) estimation to select smoothness. Knots (k) of smooths were varied up to k = 7 for all univariate terms, and up to k = 15 for bivariate smooth of longitude and latitude to avoid overfitting. Diagnostic plots, and the percentage of deviance explained were used to quantify each model’s goodness of fit. Four optimal models were used for inference, for overall beaked whales, Cuvier’s beaked whales, Sowerby’s beaked whales, and Northern bottlenose whales.
Results
Between 2006 and 2018, 244,395 km were travelled whilst on effort (Fig. 2A), 96,565 km of which occurred in conditions better than sea state 3 (Fig. 2B). The distance surveyed was similar between vessels, with 124,745 km on-board the Cap Finistère, and 119,650 km on-board the Pont-Aven. Of the total effort, 190,394 km was undertaken by Wildlife Officers across 841 days, and 54,001 km by distance sampling teams over 217 days. Effort was relatively evenly distributed across the route, with a slight increase in the Southern regions of the Bay of Biscay. Effort increased considerably from 2014 when Wildlife Officers were introduced (Fig. 3A). The majority of survey effort occurred between April and September, with minimal effort in March and October (Fig. 3B).
There were 419 encounters with 816 beaked whales, with a mean group size of 1.95 (± 1.13 SD). Of these encounters, 260 included Cuvier’s beaked whales, 19 Northern bottlenose whales, 13 Sowerby’s beaked whales, and one included True’s beaked whales. Animals in 130 encounters were not able to be identified to species level. The majority of Cuvier’s beaked whales were identified with high confidence (72.7%), followed by Sowerby’s (53.8%); however, certainty in identification was considerably lower for Northern bottlenose whales (21.1% classed as ‘definite’; Supplementary material Fig. 1).
All beaked whales
When beaked whales were not split into separate species, a mean encounter rate of 0.195 (± 0.0264 SE) per 100 km was calculated. The final scaled-t distribution model retained a 2-way smooth of longitude and latitude (P < 0.001), and 1-way smooths of month (P < 0.001), year (P < 0.001), aspect (P < 0.001), and depth (P < 0.001), and explained 11% deviance (Fig. 4; see Table 2 for significance of variables for each modelled species). Encounter rate increased in summer months, and generally increased throughout the study period, albeit with considerable inter-annual variation. Animals preferred northern-facing slopes and both shallow waters < 1000 m depth, and deeper waters > 3000 m. Beaked whales were constrained to areas of the transect in the Bay of Biscay (Fig. 5), with the exception of three sightings in the English Channel for which species identification was either unknown (e.g. generic beaked whale) or recorded with low confidence.
Cuvier’s beaked whales
Cuvier’s beaked whales were encountered on average 0.069 (± 0.0067 SE) per 100 km. The final scaled-t distribution model retained a 2-way smooth of longitude and latitude (P < 0.001), and 1-way smooths of year (P < 0.001), aspect (P < 0.001), and depth (P < 0.001), with month becoming penalized with little effect. The model explained 10.7% deviance. Cuvier’s beaked whales were predominantly encountered in the southern Bay of Biscay close to sub-sea canyons, and in a small area of the northern slope (Fig. 6). Northern-facing slopes were preferred, as were a mixture of shallow depths (< 1000 m) and deeper waters (> 4000 m). Encounters appear to have broadly increased since 2006, with inter-annual changes including a decrease between 2009 and 2012, and since 2016.
Northern bottlenose whales
Northern bottlenose whales were encountered at a mean rate of 0.0059 (± 0.001 SE) per 100 km. The final scaled-t distribution model retained a 2-way smooth of longitude and latitude (P < 0.001), and 1-way smooths of month (P < 0.001), year (P < 0.001), aspect (P < 0.001), and depth (P < 0.001), and explained 11.3% deviance. Northern bottlenose whales were only recorded in proximity to canyons on the approach to Santander, and not on the eastern route to Bilbao (Fig. 6B). Preference was evident for northern-facing slopes, whereas depth preference varied with peaks in shallow waters < 1000 m, and > 3500 m. There was an increase in encounters during summer months, and generally throughout the study period, with inter-annual variation causing some reductions between 2008 and 2012, and since 2016.
Sowerby’s beaked whales
Sowerby’s beaked whales were encountered at a mean rate of 0.0044 (± 0.001 SE) per 100 km. The final scaled-t distribution model retained a 2-way smooth of longitude and latitude (P < 0.001), and 1-way smooths of month (P = 0.026), year (P < 0.001), and depth (P < 0.001), with aspect being penalized out of the model. The model explained only 5.55% deviance. Patterns in the occurrence of Sowerby’s beaked whales were less clear compared to other species, with a weakly informative model. Encounters appeared marginally more likely later in the year, and have been increasing over the years. Animals were less likely to be recorded in deeper waters. Encounter rates were highest in a small area of the northern slopes, but more widely spread in the southern areas (Fig. 6C).
True’s beaked whales
Represented by a single sighting, habitat preferences of True’s beaked whales were not modelled; although the group of four individuals was sighted at approximately 3000 m depth, and 44.05416 N, 3.92118 W (Fig. 6D). Details of this sighting have been previously published in Robbins et al. (2019).
Discussion
This study aimed to use citizen science data to investigate spatio-temporal patterns in beaked whale occurrence within the Bay of Biscay. Overall, beaked whales were regularly observed in all months surveyed (i.e., March to October), indicating likely year-round presence within this area and a particular peak in sightings during the summer months. Aspect and depth were identified as significant environmental variables, with beaked whales typically preferring northern-facing slopes in both shallower (< 1000 m depth) and deeper waters (> 3000 m). However, there were some species-specific variations to these general patterns (Figs. 7, 8, 9).
The Bay of Biscay is characterised by weak oceanic anticyclonic circulation, poleward-flowing slope current, coastal upwelling, northward direction of the Mediterranean waters, shelf circulation, and cross-shelf transport. These features exhibit seasonal fluctuations (Forest et al., 2006); however, there is conflicting evidence regarding the seasonal movements of beaked whales. Laran et al. (2017), suggested that beaked whales have limited seasonal variations, whilst Giorli & Neuheimer (2016) proposed that beaked whales may seasonally migrate to optimise prey capture, evidenced through the increased strandings of Cuvier's beaked whales along select parts of the Irish coast during spring and summer. In the present study, significant seasonal patterns were observed for overall beaked whale sightings, as well as at the species-level for Nothern bottlenose whales and Sowerby’s beaked whales, with a strong peak in sightings during the summer months. The Bay of Biscay experiences coastal upwelling during the summer and early autumn (Lavin et al., 2003), which may lead to increased prey availability and thus drive this peak in whale sightings.
Regardless, beaked whales were observed in all months surveyed, representing 75% coverage throughout the year. Additonally, Cuvier’s beaked whales did not show seasonal variation, with encounter rates being relatively stable throughout the year, regardless of month. Therefore, it seems likely that beaked whales are present in the Bay of Biscay year-round, with the Bay having particular significance during the summer months when prey availability may be higher (Spitz et al., 2018). Unfortunately, winter survey effort was not possible; however, this would be an important avenue for future research to confirm year-round presence and allow study of any seasonal variation in spatial occurrence.
Previous studies have demonstrated relationships between beaked whale spatial distribution and physical environmental features such as depth, aspect, and slope (Davis et al., 1998, 2002; Cañadas, et al., 2002; Claridge, 2006; Kiszka et al., 2007; Azzellino et al., 2008; Arcanglei et al., 2016; Rogan et al., 2017). Our findings align with this and show more sightings in the southern Bay of Biscay, which is characterised by offshore canyon systems. In comparison, the northern Bay of Biscay appears to contain habitat that is less favourable to beaked whales, being characterised by a wide shelf and several estuaries, which are the main fluvial inputs into the basin and result in variable salinity—which has been found to influence beaked whale presence in other studies (Hazen et al., 2011). Thus, the spatial distribution of beaked whales within the Bay of Biscay reflects the underlying physical features of this region. However, it is worth remembering that the primary driver of beaked whale distribution is likely prey availability, which is linked to these physical features as well as more transient and seasonal environmental variables (Cañadas, et al., 2002; Kowarski et al., 2018).
It is generally accepted that beaked whales prefer deeper waters due to their foraging behaviours and diet preference (Cañadas, et al., 2002). Analysis of stomach contents in previous studies alludes to a diet of prey species that are found below 200 m and do not vertically migrate (MacLeod et al., 2006; Schorr et al., 2014). However, whilst most beaked whales are considered teutophagic in the Bay of Biscay (Spitz et al., 2011), Sowerby’s beaked whales are documented to primarily feed on fish (Waring et al., 2001; MacLeod et al., 2003). These fish are widely reported to exhibit vertical migration which may impact the depth at which Sowerby's beaked whales are encountered (Kowarski et al., 2018); thus, potentially explaining our sightings of beaked whales in shallower waters. Future studies aimed at improving understanding of beaked whale foraging ecology within the Bay of Biscay would be beneficial for further explaining animal distribution patterns.
Studies of beaked whale distribution and ecology are often restricted due to challenging logistics and the financial cost of offshore fieldwork. The current study was able to overcome this to a degree by using a combination of monthly citizen science surveys and daily employee surveys (i.e., Wildlife Officers) conducted from platforms of opportunity, representing a considerable amount of survey effort and leading to a substantial number of sightings of these cryptic animals. Although the utilisation of such platforms comes with some caveats, for example limited control over the survey area, this study still illustrates the usefulness of these methods. Similarly, the use of citizen scientists and Wildlife Officers as observers also comes with potential limitations; the former may be relatively inexperienced when compared with professionals, but are extensively trained and act as dedicated observers; whereas the latter may have greater experience, but are surveying on publicly available open-decks and have a primary job of public engagement. This may result in some missed detections and consequently lower encounter rates, due to overlooked animal cues or distractions. Conversely, by engaging passengers with the process of searching for animals, Wildlife Officers may actually benefit from extra eyes being available, as well as the invaluable benefit of educating members of the public on whale ecology and marine conservation. Indeed, the number of beaked whale encounters increased markedly from 2014 onwards (i.e., the year that Wildlife Officers began collecting effort-related data which were included in this study); even when accounting for increased survey effort, this still represents an increase in detections and thus the effectiveness of using Wildlife Officers. Previous work has modelled occurrence of other cetacean species collected by citizen scientists on these same vessels and found that relationships were comparable to those found by professional scientists (Robbins et al., 2020); however, it would be prudent to perform similar comparisons with the wildlife officer datasets. Regardless, the considerable efforts of these observers provide a wealth of data that would be otherwise unobtainable.
In conclusion, the Bay of Biscay appears to be an area of relevance for beaked whales, as suggested by previous studies (Kiszka et al., 2007; Spitz et al., 2011; Matear et al., 2019). Our findings strengthen the knowledge of beaked whale spatial distribution within the Bay of Biscay, but also add an important temporal component by illustrating seasonal patterns in occurrence and the likelihood of year-round use of this region. Given the numerous anthropogenic threats faced by beaked whales (García-Barón et al., 2019) and the expanding presence of human activities within the Bay, future research focusing on addressing the paucity of winter survey effort in this area would be beneficial. It is crucial to understand not only where these animals occur, but also the relevance of temporal scales with regard to diurnal, seasonal, and long-term occupancy patterns to inform successful management decisions.
Data availability
Data are held by ORCA, and can be made available upon fair request on a case by case basis.
Code availability
Code is held by the corresponding author, and will be shared upon request.
References
Arcangeli, A., L. Campana, L. Marini & C. MacLeod, 2016. Long-term presence and habitat use of Cuvier’s beaked whale (Ziphius cavirostris) in the Central Tyrrhenian Sea. Marine Ecology 37(2): 269–282.
Azzellino, A., S. Gaspari, S. Airoldi & B. Nani, 2008. Habitat use and preferences of cetaceans along the continental slope and the adjacent pelagic waters in the western Ligurian Sea. Deep Sea Research Part 1 55: 296–323.
Barlow, J. & R. Gisiner, 2006. Mitigating, monitoring and assessing the effects of anthropogenic sound on beaked whales. Journal of Cetacean Research and Management 7(3): 239–249.
Buckland, S. T., D. R. Anderson, K. P. Burnham, J. L. Laake, D. L. Borchers & L. Thomas, 2001. Introduction to distance sampling: estimating abundance of biological populations.
Cañadas, A., R. Sagarminaga & S. Garcıa-Tiscar, 2002. Cetacean distribution related with depth and slope in the Mediterranean waters off southern Spain. Deep Sea Research Part 1: Oceanographic Research Papers 49(11): 2053–2073.
Carroll, E. L., M. R. McGowen, M. L. McCarthy, F. G. Marx, N. Aguilar, M. L. Dalebout & M. T. Olsen, 2021. Speciation in the deep: genomics and morphology reveal a new species of beaked whale Mesoplodon eueu. Proceedings of the Royal Society B 288(1961): 20211213.
Certain, G., V. Ridoux, O. Van Canneyt & V. Bretagnolle, 2008. Delphinid spatial distribution and abundance estimates over the shelf of the Bay of Biscay. ICES Journal of Marine Science 65(4): 656–666
Claridge, D., 2006. Fine-scale distribution and habitat selection of beaked whales. Unpublished dissertation. Aberdeen: University of Aberdeen.
Cox, T., T. Ragen, A. Read, E. Vos, R. Baird, K. Balcomb & L. Benner, 2006. Understanding the impacts of anthropogenic sound on beaked whales. Journal of Cetacean Research and Management 7(3): 177–187.
Davis, R.W., G. S. Fargion, N. May, T. D. Leming, M. Baumgartner, W. E. Evans, L. J. Hansen, & K. Mullin, 1998. Physical habitat of cetaceans along the continental slope in the northcentral and western Gulf of Mexico. Marine Mammal Science 14(3): 490–507.
Davis, R. W., J. G. Ortega-Ortiz, C. A. Ribic, W. E. Evans, D. C. Biggs, P. H. Ressler, R. B. Cady, R. R. Leben, K. D. Mullin, & B. Würsig, 2002. Cetacean habitat in the northern oceanic Gulf of Mexico. Deep Sea Research Part I: Oceanographic Research Papers 49(1): 121–142.
D’Amico, A., R. C. Gisiner, D. R. Ketten, J. A. Hammock, C. Johnson, P. L. Tyack, & J. Mead, 2009. Beaked whale strandings and naval exercises. Space and naval warfare systems center, San Diego, CA
Evans, P. & P. Hammond, 2004. Monitoring cetaceans in European waters. Mammal Review 34(1): 131–156.
Fasiolo, M., R. Nedellec, Y. Goude, & S. N. Wood, 2018. Scalable visualisation methods for modern Generalized Additive Models. arXiv preprint arXiv:abs/1809.10632.
Faerber, M. & R. Baird, 2010. Does a lack of observed beaked whale strandings in military exercise areas mean no impacts have occurred? A comparison of stranding and detection probabilities in the Canary and main Hawaiian Islands. Marine Mammal Science 26(3): 602–613.
Forest, A., J. Boucher, P. Lazure & A. Jegou, 2006. The Bay of Biscay: the encountering of the ocean and the shelf. In Lavín, L., L. Valdés, F. Sánchez, P. Abaunza, A. Forest, J. Boucher & A. Jegou (eds), The Sea. Harvard University Press, Cambridge.
García-Barón, I., M. B. Santos, A. Uriarte, et al., 2019. Which are the main threats affecting the marine megafauna in the Bay of Biscay? Continental Shelf Research 186: 1–12.
Giorli, G. & N. Neuheimer, 2016. Temporal and spatial variation of beaked and sperm whales foraging activity in Hawai’i, as determined with passive acoustics. The Journal of the Acoustical Society of America 140(4): 2333–2343.
Hammond, P., K. Macleod, D. Gillespie, R. Swift, A. Winship & A. Cañadas, 2009. Cetacean Offshore Distribution and Abundance in the European Atlantic (CODA). CODA 2007: 1–44.
Hammond, P., K. Macleod, P. Berggren, D. Borchers, L. Burt, A. Cañadas & J. Vázquez, 2013. Cetacean abundance and distribution in European Atlantic shelf waters to inform conservation and management. Biological Conservation 164: 107–122.
Hazen, E. L., D. P. Nowacek, L. St. Laurent, et al., 2011. The relationship among oceanography, prey fields, and beaked whale foraging habitat in the tongue of the ocean. PLoS ONE 6: e19269.
Hijmans, R., 2019. geosphere: spherical trigonometry. R package version 1.5-10. https://CRAN.R-project.org/package=geosphere
Hijmans, R., 2020. raster: geographic data analysis and modeling. R package version 3.3-13. https://CRAN.R-project.org/package=raster
Hildebrand, J. A., S. Baumann-Pickering, K. E. Frasier, J. S. Trickey, K. P. Merkens, S. M. Wiggins, M. A. McDonald, et al., 2015. Passive acoustic monitoring of beaked whale densities in the Gulf of Mexico. Science Reports 5(1): 16343.
Hooker, S., N. De Soto, R. Baird, E. Carroll, D. Claridge, L. Feyrer & H. Whitehead, 2019. Future directions in research on beaked whales. Frontiers in Marine Science 5(514): 1–16.
Johnson, M., P. Madsen, W. Zimmer, N. De Soto & P. Tyack, 2004. Beaked whales echolocate on prey. Biological Letters 271: 383–386.
Ketten, D., 2014. Sonars and strandings: are beaked whales the aquatic acoustic canary? Acoustics Today 10: 46–56.
Kiszka, J., K. Macleod, O. Canneyt, D. Walker & V. Ridoux, 2007. Distribution, encounter rates, and habitat characteristics of toothed cetaceans in the Bay of Biscay and adjacent waters from platform-of-opportunity data. ICES Journal of Marine Science 64(5): 1033–1043.
Kowarski, K., J. Delarue, B. Martin, et al., 2018. Signals from the deep: spatial and temporal acoustic occurrence of beaked whales off western Ireland. PLoS ONE 13: e0199431.
Lavín, A., X. Moreno-Ventas, P. Abaunza, & J. M. Cabanas, 2003. Environmental variability in the Atlantic and Iberian waters and its influence on horse mackerel recruitment. In ICES Marine Science Symposia, Vol. 219, No. 403, 07.
Laran, S., M. Authier, A. Blanck, G. Dorémus, H. Falchetto, P. Monestiez & V. Ridoux, 2017. Seasonal distribution and abundance of cetaceans within French waters—Part II: the Bay of Biscay and the English Channel. Deep Sea Research Part II 141: 31–40.
Lester, S., B. Halpern, K. Grorud-Kolvert, J. Lubchenco, B. Ruttenberg & S. Gaines, 2009. Biological effects within no-take marine reserves: a global synthesis. Marine Ecology Progress Series 384: 33–46.
MacLeod, C. D., M. B. Santos & G. J. Pierce, 2003. Review of data on diets of beaked whales: evidence of niche separation and geographic segregation. Journal of Marine Biological Association of the United Kingdom 83: 651–665.
MacLeod, C., W. Perrin, R. Pitman, J. Barlow, L. Balance, A. D’Amico & G. Waring, 2006. Known and inferred distributions of beaked whale species (Cetacea: Ziphiidae). Journal of Cetacean Research and Management 7(3): 271–286.
Marra, G. & S. N. Wood, 2011. Practical variable selection for generalized additive models. Computational Statistics and Data Analysis 55(7): 2372–2387.
Matear, L., J. R. Robbins, M. Hale & J. Potts, 2019. Cetacean biodiversity in the Bay of Biscay: suggestions for environmental protection derived from citizen science data. Marine Policy 109: 103672.
Moura, A., N. Sillero & A. Rodrigues, 2012. Common dolphin (Delphinus delphis) habitat preferences using data from two platforms of opportunity. Acta Oecologica 38: 24–32.
OSPAR, 2019. Region IV: Bay of Biscay and Iberian Coast. Tratto da OSPAR Ministerial: https://www.ospar.org/convention/the-north-east-atlantic/iv.
Pante, E. & B. Simon-Bouhet, 2013. marmap: a package for importing, plotting and analyzing bathymetric and topographic data in R. PLoS ONE 8(9): e73051.
Parsons, E., S. Dolman, A. Wright, N. Rose & W. Burns, 2008. Navy sonar and cetaceans: just how much does the gun need to smoke before we act? Marine Pollution Bulletin 56(7): 1248–1257.
R Core Team, 2020. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna
Robbins, J. R., T. Park & E. J. Coombs, 2019. Supernumerary teeth observed in a live True’s beaked whale in the Bay of Biscay. PeerJ 7: e7809.
Robbins, J. R., L. Babey & C. B. Embling, 2020. Citizen science in the marine environment: estimating common dolphin densities in the north-east Atlantic. PeerJ 8: e8335.
Roff, J. & M. Zacharias, 2011. Marine Conservation Ecology. Routledge, Oxon.
Rogan, E., A. Cañadas, K. Macleod, M. Santos, B. Mikkelsen, A. Uriarte & P. Hammond, 2017. Distribution, abundance and habitat use of deep diving cetaceans in the North-East Atlantic. Deep Sea Research 141: 8–19.
Schorr, G., E. Falcone, D. Moretti & R. Andrews, 2014. First long-term behavioral records from cuvier’s beaked Whales (Ziphius cavirostris) reveal record-breaking dives. PLoS ONE 9(3): 1–10.
Spitz, J., Y. Cherel, S. Bertin, J. Kiszka, A. Dewez & V. Ridoux, 2011. Prey preferences among the community of deep-diving odontocetes from the Bay of Biscay, Northeast Atlantic. Deep Sea Research 58(3): 273–282.
Spitz, J., A. Trites, V. Becquet, A. Brind’Amour, Y. Cherel, R. Galois & V. Ridoux, 2012. Cost of living dictates what whales, dolphins and porpoises eat: the importance of prey quality on predator foraging strategies. PLoS ONE 7(11): e50096.
Spitz, J., V. Ridoux, A. W. Trites, et al., 2018. Prey consumption by cetaceans reveals the importance of energy-rich food webs in the Bay of Biscay. Progress in Oceanography 166: 148–158.
Tepsich, P., M. Rosso, P. N. Halpin & A. Moulins, 2014. Habitat preferences of two deep-diving cetacean species in the northern Ligurian Sea. Marine Ecology Progress Series 508: 247–260.
Tyack, P., M. Johnson, N. Aguilar De Soto, A. Sturlese & P. Madsen, 2006. Extreme diving of beaked whales. The Journal of Experimental Biology 209: 4238–4253.
Tyack, P., W. Zimmer, D. Moretti, B. Southall, D. Claridge, J. Durban & I. Boyd, 2011. Beaked whales respond to simulated and actual navy sonar. PLoS ONE 6(3): 1–15.
Waring, G. T., T. Hamazaki, D. Sheehan, et al., 2001. Characterization of beaked whale (ziphiidae) and sperm whale (Physeter Macrocephalus) summer habitat in shelf-edge and deeper waters off the northeast US. Society for Marine Mammal Science 17: 703–717.
Wickham, H., 2016. ggplot2: Elegant Graphics for Data Analysis. Springer, New York.
Wickham, H., R. François, L. Henry, & K. Müller, 2020. dplyr: a Grammar of Data Manipulation. R package version 1.0.0. https://CRAN.R-project.org/package=dplyr.
Wood, S. N. 2006. Generalized additive models: an introduction with R. chapman and hall/CRC.
Wood, S. N., 2017. Generalized Additive Models: An Introduction with R, 2nd ed. Chapman and Hall/CRC, Boca Raton.
Wood, S. N., N. Pya & B. Saefken, 2016. Smoothing parameter and model selection for general smooth models. Journal of the American Statistical Association 111: 1548–1575.
Weir, C., J. Stokes, C. Martin & P. Cermeño, 2004. Three sightings of Mesoplodon species in the Bay of Biscay: first confirmed True’s beaked whales (M. mirus) for the north-east Atlantic? Journal of Marine Biological Association of the United Kingdom 84(5): 1095–1099.
Yamada, T., S. Kitamura, S. Abe, Y. Tajima, A. Matsuda, J. Mead & T. Matsuishi, 2019. Description of a new species of beaked whale (Berardius) found in the North Pacific. Science Reports 9(1): 12723.
Zuur, A. F. & E. N. Ieno, 2016. A protocol for conducting and presenting results of regression-type analyses. Methods in Ecology and Evolution 7: 636–645.
Acknowledgments
Fieldwork was supported by Brittany Ferries who provided access to their vessels. The crew of the Cap Finistère and Pont-Aven have been supportive of survey work in the Bay of Biscay for decades. This work would not be possible without the contributions of volunteer Marine Mammal Surveyors and seasonally employed Wildlife Officers, and importantly ORCA’s members.
Funding
Fieldwork was supported by ORCA’s members and partners. JUSTONEOCEAN provided funding for associated work.
Author information
Authors and Affiliations
Contributions
EB, JR and LB conceived the study, with input from JP and SM, EB and JR conducted analyses, and EB, JR and SM drafted the manuscript. All authors contributed to manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest are known.
Ethical approval
Work was approved by ORCA and Brittany Ferries, with no additive disturbance to animals during research proceedings. All data were collected from platforms of opportunity which would be travelling regardless of researcher presence, and no course alterations were made to approach animals.
Consent to participate
No human participants were studied in this research.
Consent for publication
All authors have approved the submitted manuscript.
Additional information
Handling editor: Grazia Pennino
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Robbins, J.R., Bell, E., Potts, J. et al. Likely year-round presence of beaked whales in the Bay of Biscay. Hydrobiologia 849, 2225–2239 (2022). https://doi.org/10.1007/s10750-022-04822-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-04822-y